Abstract
Background
Cytomegalovirus (CMV) is one of the most important opportunistic infections in transplant recipients. Currently sero-positivity for CMV IgG before solid organ transplantation is the laboratory test of choice for stratifying the risk of CMV reactivation after solid organ transplantation. Theoretically, CMV-specific cell-mediated immune responses before solid organ transplantation should further categorize patients as high or low risk of CMV development. We therefore evaluated the usefulness of the CMV-specific enzyme-linked immunospot (ELISPOT) assay in kidney transplant (KT) candidates for predicting the development of CMV infections after transplantation.
Materials and Methods
All adult CMV IgG (+) recipients admitted to the KT institute between March 2014 and June 2014 were enrolled, and CMV infections after KT were observed between March 2014 and December 2014. All patients underwent CMV pp65 and IE1-specific ELISPOT assays before transplantation. CMV infection was defined in the presence of CMV antigenemia, CMV syndrome, or tissue-invasive CMV disease. We used the data to select optimal cut-off values for pp65 and IE1, respectively, on ROC curves.
Results
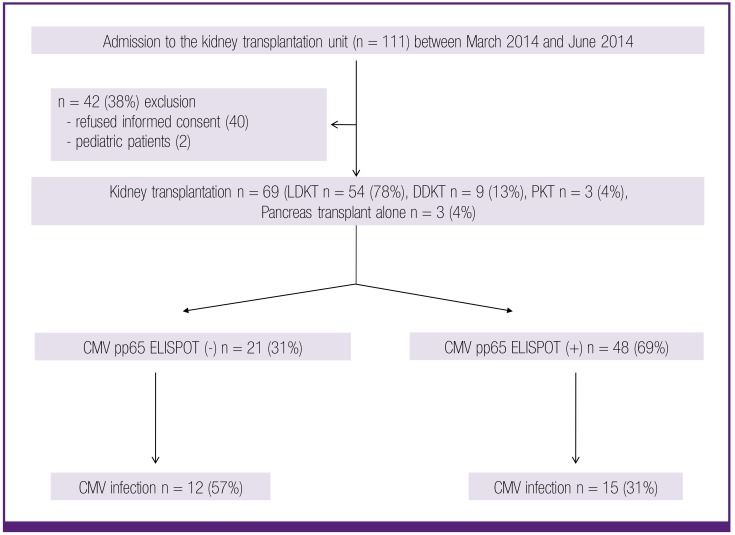
A total of 69 transplant recipients involving 54 (78%) living-donor KT, 9 (13%) deceased-donor KT, 3 (4%) kidney-pancreas transplants, and 3 (4%) pancreas transplants were enrolled. Of the 69 patients, 27 (39%) developed CMV infections. There was no association between the IE1-specific ELISPOT assay and CMV infection. However, only 15 (31%) of the 48 patients with positive pp65-specific ELISPOT results (>10 spots/2.0 × 105 cells) developed CMV infections, whereas 12 (57%) of the 21 patients with negative pp65-specific ELISPOT results developed CMV infection (P = 0.04).
Conclusion
Negative pp65-specific ELISPOT assay results before transplantation appear to predict the subsequent development of CMV infections after transplantation in CMV IgG (+) KT recipients. Therefore, risk stratification of CMV IgG (+) recipients using the CMV-specific ELISPOT, together with preventive strategies, may further reduce CMV development.
Keywords: Cytomegalovirus, Enzyme-linked immunospot assay, Kidney transplantation
Introduction
Cytomegalovirus (CMV) is one of the most important opportunistic infections in transplant recipients [1]. Currently sero-positivity for CMV IgG before solid organ transplantation is the laboratory test of choice for stratifying the risk of CMV reactivation after solid organ transplantation (SOT) [2]. It is known that D+/R (donor seropositive and recipient seronegative) has the highest risk of CMV infection after SOT, followed by D+/R+ and D-/R+. D-/R- has the least risk [1]. Experts recommend universal ganciclovir prophylaxis for the highest risk group [1]. Either universal prophylaxis or preemptive ganciclovir therapy based on monitoring for early CMV replication is recommended for the moderate risk groups [1], into which more than 95% of Korean SOT candidates fall [3]. Theoretically, CMV-specific cell-mediated immune responses before SOT would further categorize the moderate risk groups (D+/R+; D-/R+) into patients with higher or lower risk of CMV development after SOT. If that were the case, risk stratification of recipients using the CMV-specific enzyme-linked immunospot (ELISPOT) together with preventive strategies might further reduce CMV development. We therefore evaluated in kidney transplantation (KT) candidates the usefulness of the CMV-specific ELISPOT assay for predicting the development of CMV infections after KT. We report the results of a preliminary proof-of-concept study.
Materials and Methods
1. Study population
All patients admitted for transplantation to a renal transplant unit between March 2014 and June 2014 in a 2,700-bed, tertiary-care hospital in Seoul, South Korea, were prospectively screened. Tests for CMV IgG were performed in the KT recipients and donorss. Exclusion criteria were refusal of informed consent and pediatric renal transplant candidates (<16 years old). The universal oral valgaciclovir for 3 months was given only to the highest CMV risk group (D+/R-). CMV antigenemia assays were performed weekly during the first month, bi-weekly during the 1-3 months after KT, and then monthly to 6 months after KT. CMV antigenemia of >50 cells per 200,000 cells were indications for preemptive therapy. Conventional-dose ganciclovir (5 mg/kg twice daily) as preemptive therapy was given daily for at least 2 weeks and until patients were negative for CMV antigenemia. All individuals were informed of the nature of the study, and all participants provided written informed consent. This investigation was approved by the Institutional Review Board of our hospital.
2. The CMV antigenemia assay and CMV ELISPOT assay
The CMV antigenemia assay was performed as previously described [4]. EDTA-treated whole blood samples were fractionated by dextran sedimentation and lysis of erythrocytes. The granulocytes were then centrifuged to prepare a cytospin slide. The cells were then fixed with formaldehyde, sequentially immunostained by monoclonal antibodies C10/C11 (Clonab CMV; Biotest, Dreieich, Germany). Counts are expressed as positive cells per 200,000 leukocytes.
A peripheral venous blood sample (~8 mL) was collected from each patient for the CMV ELISPOT assay for T cells producing IFN-γ (i.e. T-track CMV, Regensburg, Germany). Briefly, peripheral blood mononuclear cells (PBMC) were immediately (within 30 minutes) separated and collected. The collected cells were resuspended at 2.0 × 106 cells/mL, placed (2.0 × 105 cells/well) in wells pre-coated with anti-human IFN-γ antibody. The samples were stimulated with phytohemagglutinin (positive control), pp65, IE1, and media only (negative control) and incubated for 18 hours. The resulting spots were counted with an automated microscope (ELiSpot 04 HR; Autoimmune Diagnostika GmbH, Strassberg, Germany). Background counts, obtained in negative control wells, were subtracted.
3. Assessment of outcomes
The primary outcome was the development of CMV infection. In this study, patients with CMV antigenemia or CMV disease were considered to have a CMV infection [2,5,6]. CMV antienemia was defined as CMV antigenemia identified by pp65 antigenemia, and CMV disease was defined as CMV syndrome or tissue-invasive CMV disease. CMV syndrome was defined as CMV antigenemia and at least one of the following: fever >38℃; new onset severe malaise; leucopenia in two successive measurements (WBC count of <3,500 cells/mm3); atypical lymphocytes of >5%; thrombocytopenia of < 100,000/mm3. Tissue-invasive CMV was defined as evidence of localized CMV infection (cells with CMV inclusions, in situ detection of CMV antigen by immnunohistochemistry, or DNA) in a biopsy or other appropriate specimen (e.g., bronchoalveolar lavage, cerebral spinal fluid) and symptoms of organ dysfunction.
4. Statistical analysis
Since this study was a preliminary proof-of-concept study, the sample size was not calculated. We planned to enroll over a 4-month period and monitor the development of CMV infection for an additional 6 months. For each of the tests used here to predict CMV infection namely the pp65- and IE1-specific ELISPOT assays, we examined receiving operator characteristic (ROC) curves that plotted sensitivity against the rate of false-positive results over a range of cut-off values [7]. We chose the optimal cut-off value as the point on each ROC curve farthest from the diagonal line that maximized the sum of sensitivity and specificity. Diagnostic performance was expressed in terms of sensitivity, specificity, positive predictive value, and negative predictive value. Categorical variables were compared using Pearson's Chi-square tests. All tests of significance were two-tailed and a P-value of less than 0.05 was considered to indicate statistical significance. Calculations were performed using the SPSS for Windows software package, version 14.0K (SPSS Inc., Chicago, IL, USA) and MedCalc software (MedCalc Software bvba, Mariakerke, Belgium).
Results
1. Patients characteristics
Figure 1 is a flow diagram covering all the patients admitted to our hospital for KT between March 2014 and June 2014, and presents the results of observations on the development of CMV infections after KT between March 2014 and December 2014. A total of 111 patients were initially enrolled in the study. Of these, 42 (38%) were ultimately excluded; 40 for refusal of informed consent and 2 pediatric patients. The remaining 69 patients were enrolled in the final analysis. Demographic data for the study patients are shown in Table 1.
Figure 1. Flow chart of the study. LDKT, living-donor kidney transplantation; DDKT, deceased-donor kidney transplantation; PKT, pancreas-kidney transplantation.
CMV, cytomegalovirus; ELISPOT, enzyme-linked immunospot.
Table 1. Characteristics of transplant recipients.
| Patient characteristic | N = 69 (%) |
|---|---|
| Mean age, years (± SD) | 46 ± 12 |
| Male gender | 46 (67) |
| Primary reason for transplant | |
| Glomerulonephritis | 17 (25) |
| Hypertension | 10 (15) |
| Diabetes mellitus | 12 (17) |
| Unknown | 19 (28) |
| Polycystic kidney disease | 2 (3) |
| Others | 9 (13) |
| Transplant type | |
| Living donor kidney | 54 (79) |
| Deceased donor kidney | 9 (13) |
| Pancreas and kidney | 3 (4) |
| Pancreas alone | 3 (4) |
| ABO-mismatch transplantation | 18 (26) |
| Primary transplant induction therapy at transplantation | |
| Anti-IL2 receptor antibodies | 57 (83) |
| Antithymocyte antibodies | 6 (9) |
| Rituximab | 22 (32) |
| CMV serostatus | |
| D+/R+ | 63 (91) |
| R+ (donor serology unknown) | 5 (7) |
| D-/R+ | 1 (2) |
| CMV infection | |
| CMV antigenemia | 27 (39) |
| CMV antigenemia > 50 CMV-positive cell/2,000,000 leukocytes | 7 (10) |
| CMV syndrome | 0 |
| Tissue-invasive CMV | 4 (6) |
IL, interleukin; CMV, cytomegalovirus; D, donor; R, recipient.
2. Development of CMV infections and CMV ELISPOT
Of the 69 patients, 27 (39%) developed CMV infections after KT (Table 1). Of the 27 patients with CMV infections, 7 (10%) displayed significant levels of CMV antigenemia (>50 CMV-positive cells/200,000 cells, which was the threshold for ganciclovir preemptive therapy in our hospital). Of these 7 patients, 4 who had tissue-invasive CMV disease (all had CMV gastritis) and 1 without tissue-invasive CMV disease received ganciclovir therapy. The remaining 2 patients with >50 CMV antigenemia who did not received ganciclovir preemptive therapy recovered spontaneously without any complications.
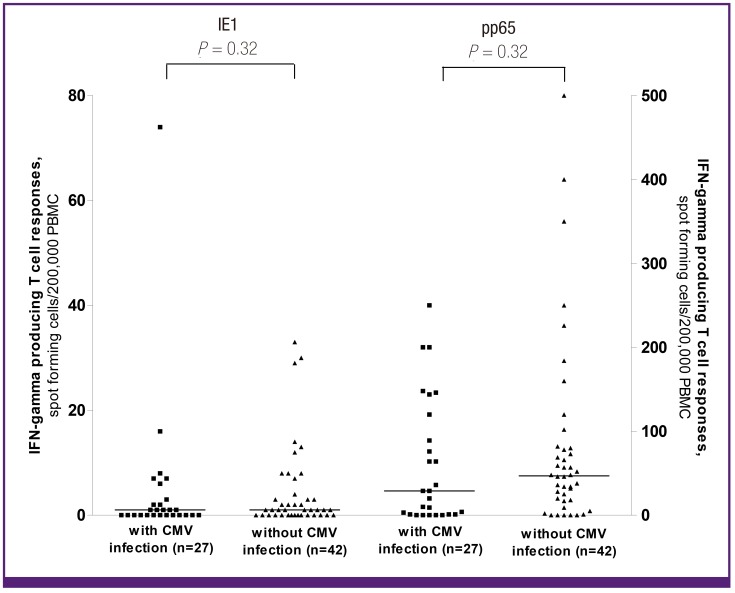
On the basis of the ROC curves (Supplemental Fig. 1), we determined that the optimal cut-off values were >10 spots and > 0 spots, for the pp65 and IE1 ELISPOT assays, respectively. When we used these cut-offs, only 15 (31%) of the 48 patients with positive pp65-specific ELISPOT results (>10 spots/2.0 × 105 cells) developed CMV infection, whereas 12 (57%) of the 21 patients with negative pp65-specific ELISPOT results developed CMV infection (P = 0.04). The sensitivity, specificity, positive predictive value, and negative predictive value of the pp65 ELISPOT for predicting CMV infection were 44% (95% CI, 25-65), 79% (95% CI, 63-89), 57% (95% CI, 34-78), and 69% (95% CI, 54-81), respectively. However, there was not any statistical significant association between the IE1-specific ELISPOT assay and CMV infection; 14 (33%) of the 42 patients with positive IE1-specific ELISPOT results (>0 spots/2.0 × 105 cells) developed CMV infection, and 13 (48%) of the 27 patients with negative IE1-specific ELISPOT results developed CMV infection (P = 0.22). The sensitivity, specificity, positive predictive value, and negative predictive value of the IE1 ELISPOT for predicting CMV infection were 48% (95% CI, 29-68), 67% (95% CI, 50-80), 48% (95% CI, 29-68), and 67% (95% CI, 50-80), respectively. Responses to IE1 and pp65 according to the presence of CMV infection after kidney transplantation are shown in Figure 2.
Figure 2. Response to IE1 and pp65 according to the presence of CMV infection after kidney transplantation. Bars indicate medians. The Mann-Whitney U test was used to compare the differences between groups.
IFN, interferon; PBMC, peripheral blood mononuclear cells; CMV, cytomegalovirus.
Discussion
Current clinical immune assessment of the CMV risk of infection before transplantation relies exclusively on donor and recipient CMV IgG serostatus [2]. However, given that most adults in Korea are CMV seropostive (>95%) [3], the serostatus of most SOT recipients is D+/R+. Thus, most Korean SOT recipients are homogenously classified as at moderate risk of CMV infection. In this clinical situation, a futher risk strification strategy is needed to reduce CMV development after SOT. In this study we showed that negative pp65-specific ELISPOT assay results before transplantation were associated with the subsequent development of CMV infections after transplantation in KT CMV IgG (+) recipients. So, our data indicate that risk stratification of CMV IgG (+) recipients using the CMV-specific ELISPOT, together with preventive strategies, may further reduce CMV development.
As for the interferon-gamma releasing assays (IGRAs) for tuberculosis, two commercial IGRAs for CMV are available; one is the ELISA-based Quantiferon-CMV (Cellestis, Valencia, CA, USA) and the other, the ELISPOT-based T-track CMV. Numerous studies using Quantiferon CMV have been reported in SOT recipients. Kumar, et al. [9] showed for 108 SOT recipients that those with detectable Quantiferon CMV responses after universal CMV prophylaxis were at lower risk of CMV disease [8]. Manuel, et al. also reported similar results in 127 D+/R- SOT recipients using Quantiferon CMV results after universal CMV prophylaxis [9]. Although there are a few studies of the utility of in-house ELISPOT assays for predicting CMV infection after SOT, there are no published data on the commercially available ELISPOT assay. To the best of our knowledge, this is the first effort to evaluate the clinical utility of the commercial CMV ELISPOT assay in transplant recipients.
It is noteworthy that the pp65-specific but not the IE1-specific T cell response was associated with the development of CMV after SOT. Both pp65 and IE-1 are considered dominant T cell targets. Bunde et al. [10] demonstrated that only IE1-specific CD8 T cells were associated with protection against CMV diseases [10]. In contrast, a recent study demonstrated that pp65-specific immunity was crucial for controlling CMV dissemination in an animal model [11]. There are reports that the IE1-specific T cell response is associated with CMV development after transplantation SOT [2,12] whereas others have shown a positive correlation between pp65 T cell responses and CMV viremia [13,14]. The reasons for these differences are not clear. Possible explanations could be differences between the various cellular immune assays, and different outcome measure in response to variable CMV stimuli. Further studies are needed in this area.
This study has some limitations. It is a preliminary proof-of-concept study that provides the information necessary to be able to calculate the sample size needed for a confirmatory study. Hence the present sample size was not sufficient to meaure the true difference in CMV infection rate between positive IE1-specific ELISPOTs and negative IE1-specific ELISPOTs. Indeed, statistical power was only 40% in this cohort study. Therefore, we are planning to enroll 191 patients to confirm the association of IE1-specific ELISPOT with CMV infection at a 5% alpha and a power of 80%.
In conclusion, negative pp65-specific ELISPOT assay results before transplantation appear to predict the development of CMV infections after KT in CMV IgG (+) recipients with CMV IgG (+) donors. We believe that risk stratification using the CMV-specific ELISPOT assay, combined with preventive strategies, may further reduce CMV development.
Acknowledgement
This study is supported by the Korean Society of Infectious Diseases (study grant 2013).
Footnotes
Conflicts of Interest: No conflicts of interest.
Supplementary material
Supplementary data including one figure can be found with this article online http://www.icjournal.org/src/sm/ic-47-105-s001.pdf.
Receiver operating characteristics (ROC) curves of pp65- and IE1-specific ELISPOT assays for predicting CMV infection after transplantation. (A) pp65-specific ELISPOT assays. (B) IE1-specific ELISPOT assays.
References
- 1.Razonable RR, Humar A. AST Infectious Diseases Community of Practice. Cytomegalovirus in solid organ transplantation. Am J Transplant. 2013;13(Suppl 4):93–106. doi: 10.1111/ajt.12103. [DOI] [PubMed] [Google Scholar]
- 2.Bestard O, Lucia M, Crespo E, Van Liempt B, Palacio D, Melilli E, Torras J, Llaudó I, Cerezo G, Taco O, Gil-Vernet S, Grinyó JM, Cruzado JM. PRetransplant immediately early-1-specific T cell responses provide protection for CMV infection after kidney transplantation. Am J Transplant. 2013;13:1793–1805. doi: 10.1111/ajt.12256. [DOI] [PubMed] [Google Scholar]
- 3.Hahn JS, Lee SJ, Park WK, Ko YW, Kim HO, Lee SY. A survey on the cytomegalovirus antibodies in blood donors and the diseased. Korean J Blood Transfus. 1990;1:21–34. [Google Scholar]
- 4.Mori T, Okamoto S, Matsuoka S, Yajima T, Wakui M, Watanabe R, Ishida A, Iwao Y, Mukai M, Hibi T, Ikeda Y. Risk-adapted pre-emptive therapy for cytomegalovirus disease in patients undergoing allogeneic bone marrow transplantation. Bone Marrow Transplant. 2000;25:765–769. doi: 10.1038/sj.bmt.1702227. [DOI] [PubMed] [Google Scholar]
- 5.Jang EY, Park SY, Lee EJ, Song EH, Chong YP, Lee SO, Choi SH, Woo JH, Kim YS, Kim SH. Diagnostic performance of the cytomegalovirus (CMV) antigenemia assay in patients with CMV gastrointestinal disease. Clin Infect Dis. 2009;48:e121–e124. doi: 10.1086/599116. [DOI] [PubMed] [Google Scholar]
- 6.Moon SM, Sung H, Kim MN, Lee SO, Choi SH, Kim YS, Woo JH, Kim SH. Diagnostic yield of the cytomegalovirus (CMV) antigenemia assay and clinical features in solid organ transplant recipients and hematopoietic stem cell transplant recipients with CMV pneumonia. Transpl Infect Dis. 2012;14:192–197. doi: 10.1111/j.1399-3062.2011.00703.x. [DOI] [PubMed] [Google Scholar]
- 7.Kim SH, Cho OH, Park SJ, Lee EM, Kim MN, Lee SO, Choi SH, Kim YS, Woo JH, Lee SA, Kang JK. Rapid diagnosis of tuberculous meningitis by T cell-based assays on peripheral blood and cerebrospinal fluid mononuclear cells. Clin Infect Dis. 2010;50:1349–1358. doi: 10.1086/652142. [DOI] [PubMed] [Google Scholar]
- 8.Kumar D, Chernenko S, Moussa G, Cobos I, Manuel O, Preiksaitis J, Venkataraman S, Humar A. Cell-mediated immunity to predict cytomegalovirus disease in high-risk solid organ transplant recipients. Am J Transplant. 2009;9:1214–1222. doi: 10.1111/j.1600-6143.2009.02618.x. [DOI] [PubMed] [Google Scholar]
- 9.Manuel O, Husain S, Kumar D, Zayas C, Mawhorter S, Levi ME, Kalpoe J, Lisboa L, Ely L, Kaul DR, Schwartz BS, Morris MI, Ison MG, Yen-Lieberman B, Sebastian A, Assi M, Humar A. Assessment of cytomegalovirus-specific cell-mediated immunity for the prediction of cytomegalovirus disease in high-risk solid-organ transplant recipients: a multicenter cohort study. Clin Infect Dis. 2013;56:817–824. doi: 10.1093/cid/cis993. [DOI] [PubMed] [Google Scholar]
- 10.Bunde T, Kirchner A, Hoffmeister B, Habedank D, Hetzer R, Cherepnev G, Proesch S, Reinke P, Volk HD, Lehmkuhl H, Kern F. Protection from cytomegalovirus after transplantation is correlated with immediate early-1-specific CD8 T cells. J Exp Med. 2005;201:1031–1036. doi: 10.1084/jem.20042384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Malouli D, Hansen SG, Nakayasu ES, Marshall EE, Hughes CM, Ventura AB, Gilbride RM, Lewis MS, Xu G, Kreklywich C, Whizin N, Fischer M, Legasse AW, Viswanathan K, Siess D, Camp DG, 2nd, Axthelm MK, Kahl C, DeFilippis VR, Smith RD, Streblow DN, Picker LJ, Früh K. Cytomegalovirus pp65 limits dissemination but is dispensable for presistence. J Clin Invest. 2014;124:1928–1944. doi: 10.1172/JCI67420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Nickel P, Bold G, Presber F, Biti D, Babel N, Kreutzer S, Pratschke J, Schönemann C, Kern F, Volk HD, Reinke P. High levels of CMV-IE-1-specific memory T cells are associated with less alloimmunity and improved renal allograft function. Transpl Immunol. 2009;20:238–242. doi: 10.1016/j.trim.2008.11.002. [DOI] [PubMed] [Google Scholar]
- 13.Egli A, Binet I, Binggeli S, Jäger C, Dumoulin A, Schaub S, Steiger J, Sester U, Sester M, Hirsch HH. Cytomegalovirus-specific T-cell responses and viral replication in kindey transplant recipients. J Transl Med. 2008;6:29. doi: 10.1186/1479-5876-6-29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Pipeling MR, John ER, Orens JB, Lechtzin N, McDyer JF. Primary cytomegalovirus phosphoprotein 65-specific CD8+ T-cell responses and T-bet levels predict immune control during early chronic infection in lung transplant recipients. J Infect Dis. 2011;204:1663–1671. doi: 10.1093/infdis/jir624. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Receiver operating characteristics (ROC) curves of pp65- and IE1-specific ELISPOT assays for predicting CMV infection after transplantation. (A) pp65-specific ELISPOT assays. (B) IE1-specific ELISPOT assays.