Abstract
Increased levels of monocyte–platelet aggregates (MPAs) are reported to be highly correlated with cardiovascular events. In this study, the MPA levels in different monocyte subsets and the associations between MPA levels, HIV-1 viremia and monocyte activation were evaluated during HIV-1 infection. The results showed that the percentages of MPAs in all three monocyte subsets were higher in HIV-1-infected subjects than in healthy controls, and were associated with the plasma viral load in the non-classical and intermediate monocyte subsets. The plasma levels of sCD14 and sCD163 were upregulated in HIV-1 infection and were positively associated with viral loads and negatively associated with CD4 counts. P-selectin glycoprotein ligand-1 (PSGL-1) was shown to be expressed at significantly lower levels on all three monocyte subsets and was negatively correlated with the sCD163 level. The MPA level was correlated with the levels of plasma sCD163 but negatively correlated with CD163 and PSGL-1 on all three monocyte subsets. An elevated immune activation status was correlated with increased MPA formation, underlying the potential interaction between monocyte activation and MPA formation. This interaction may be related to a higher thromboembolic risk in patients infected with HIV-1.
Keywords: HIV-1, immune activation, monocytes, platelets, sCD163
Introduction
Chronic inflammation and immune activation are implicated in HIV-1 persistence and disease progression. As markers of immune activation, the plasma sCD14 and sCD163 levels are elevated during HIV-1 infection and are useful in predicting the activation status of the monocyte response, HIV activity and pathogenesis.1,2,3,4,5,6 Toll-like receptors (TLRs) are abundantly expressed on monocytes and are involved in innate monocyte responses. Many studies suggested that TLR responses contribute to the disease progression and clinical outcomes of viral infections, including HIV infections.7,8,9,10 As a natural ligand of the programmed death 1 (PD-1) molecule, increased levels of PD-L1 expression on antigen-presenting cells (APCs, including monocytes, dendritic cells and macrophages) have also been frequently reported during HIV-1 infection. This molecule can deliver signals that inhibit T-cell activation and proliferation.11,12
Human monocytes in the peripheral blood can be divided into the following three subsets: CD14dimCD16+ (non-classical monocytes), CD14highCD16+ (intermediate monocytes) and CD14highCD16− (classical monocytes).13,14 Each subset has a unique genetic signature and distinct functions regarding inflammation and the immune response.14,15 Increased proportions of non-classical and intermediate monocytes have been indicated in HIV-1 infected patients.16,17 However, the phenotypic characteristics of specific monocyte subsets and their association with immune activation markers have not been fully defined. Recently, monocyte subset alterations and the formation of monocyte–platelet aggregates (MPAs) have been reported to be highly correlated with cardiovascular events.18 MPA is regarded as a more sensitive marker of platelet activation than P-selectin (CD62P), a molecule that binds to P-selectin glycoprotein ligand-1 (PSGL-1) on monocytes, and is closely correlated with thromboembolic events.19,20,21 Cardiovascular risks are increased in HIV-1-infected patients,22,23,24 and this increase may be due to a mechanism involving monocyte subset distribution, activation, and MPA formation disorders. Increased MPA formation was previously reported in HIV-1-infected patients receiving antiretroviral therapy and in simian immunodeficiency virus infection.25,26 However, the MPA levels in HIV-1-infected, treatment naive patients and the changes in MPA levels as the disease progresses have not been previously examined. Although monocyte activation is a typical characteristic of HIV-1 infection, the association between monocyte activation and MPA levels in HIV-1 infection is still unclear. Investigating the circulating MPA levels and their association with monocyte immune activation markers will contribute to our understanding of the monocyte-related activation/inflammation status and thromboembolic risk during HIV-1 infection.
In this study, we investigated the distribution and phenotypic characteristics of three monocyte subsets in HIV-1-infected individuals. We specifically focused on the formation of MPA during HIV-1 infection and the association of these aggregates with monocyte activation and disease progression. The findings of this study provide a helpful surrogate marker for predicting cardiovascular risks in HIV-1 infection, and in addition, understanding the association between MPA formation and monocyte activation may implicate potential targets for intervention.
Materials and methods
Subjects
Fifty-eight HIV-1-infected subjects from a cohort of men who have sex with men in Chaoyang District, Beijing, China and 26 healthy, male controls were recruited in this study. The establishment of HIV-1 infection was diagnosed by the presence of HIV-1 antibodies using an anti-HIV-1 ELISA kit (Wantai, Beijing, China) and an anti-HIV-1 western blotting kit (Aoya, Hangzhou, China). Of the HIV-1 infected subjects, 38 had a primary HIV-1 infection (PHI), as defined by a diagnosis of HIV-1 infection within 12 months from the last negative detection, and 20 were chronically infected with HIV-1 infection (CHI), as defined by a diagnosis of HIV-1 infection over 3 years. All of the HIV-1-infected patients enrolled in this study were treatment naive. The demographic characteristics of the studied subjects are listed in Table 1. The study was approved by the Institutional Review Board of the National Center for AIDS/STD Control and Prevention, China CDC. Written informed consents were signed before blood sample collection by all study participants. The CD4+ T-cell counts were measured by subjecting EDTA-treated whole blood stained for CD3/CD4/CD8/CD45 in TruCount tubes to analysis with a FACSCalibur flow cytometer (Becton Dickinson, San Jose, CA, USA). The absolute numbers of CD4+ T lymphocytes were analyzed using MultiSET software (BD Bioscience, San Jose, CA, USA). The plasma levels of HIV-1 RNA were measured with the Standard Amplicor HIV Monitor assay, version 2.0 (Roche Diagnostics, Indianapolis, IN, USA), according to the manufacturer's protocols. The lower limit of detection was 40 RNA copies/ml.
Table 1. Demographic characteristics of HIV-1-infected subjects and healthy controls.
| Characteristics | PHI | CHI | Healthy controls |
|---|---|---|---|
| Cases | 38 | 20 | 26 |
| Age (years) | 27±6 | 33±7 | 28±5 |
| Years of infection | 0.6±0.2 | 6.2±3.1 | NA |
| CD4+ T-cell counts (cells/μl) | 432.5±147.2 | 474.0±195.7 | NA |
| Viral load (log10 copies/ml) | 4.21±0.95 | 1.30±1.68 | NA |
Abbreviations: CHI, chronic HIV-1 infection; NA, not applicable; PHI, primary HIV-1 infection.
Data are shown as the median±s.d.
Monocyte phenotype and MPA determination
EDTA anticoagulated peripheral blood was collected from HIV-1-infected subjects and healthy controls. Then, 100 µl of fresh whole blood was stained with CD3-Pacific Blue, anti-CD14 PE-Cy7, anti-CD16 APC-Cy7 and anti-CD61 APC for MPA determination. Additional combined antibodies, including anti-PSGL-1 Percp-eFluor710, anti-CD163 PE, anti-TLR2 Alex Fluor700, anti-TLR4 FITC, anti-PD-L1 FITC and anti-PD-1 PE were used for monocyte phenotype staining. The cells were stained for 25 min at room temperature in the dark. The red blood cells in the samples were lysed using FACS lysing solution (BD Bioscience) for 10 min in the dark. The remaining cells were washed twice and fixed with 2% paraformaldehyde. Compensation for fluorescence spillover was performed by using fluorochrome-conjugated calibration beads (OneComp eBeads; eBioscience, San Diego, CA, USA). With the exception of anti-CD16 APC-Cy7 and anti-PD-L1 FITC, which were purchased from BD Pharmingen, all of the other antibodies were purchased from eBioscience. The samples were acquired on a FACSAria cytometer (BD Bioscience, San Jose, CA, USA) equipped with three lasers (488 nm, 405 nm and 633 nm) and 9-color detecting capabilities and were analyzed using FlowJo software (TreeStar, Ashland, OR, USA). The monocyte subsets were defined by CD14 and CD16 expression, and MPAs were represented by CD61+ monocytes. The levels of phenotypic markers expressed on different monocyte subsets were recorded as mean fluorescence intensity (MFI).
Detection of sCD14 and sCD163 in the plasma
ELISA kits were utilized to quantify soluble CD14 (sCD14) and soluble CD163 (sCD163) in plasma samples according to the manufacturer's instruction (R&D System, Minneapolis, MN, USA).
Statistical analysis
Statistical analysis was performed with GraphPad Prism 5.0 software (San Diego, CA, USA). A non-parametric Mann–Whitney U-test was used to evaluate variations between corresponding groups. The Pearson correlation test was used to evaluate correlations among the CD4 counts, viral load, proportions of monocyte subsets, percentages of MPAs and the MFI of monocyte phenotypic markers. All analyses were two-tailed, and P values less than 0.05 were considered to be statistically significant.
Results
Monocyte subset frequencies are altered during HIV-1 infection
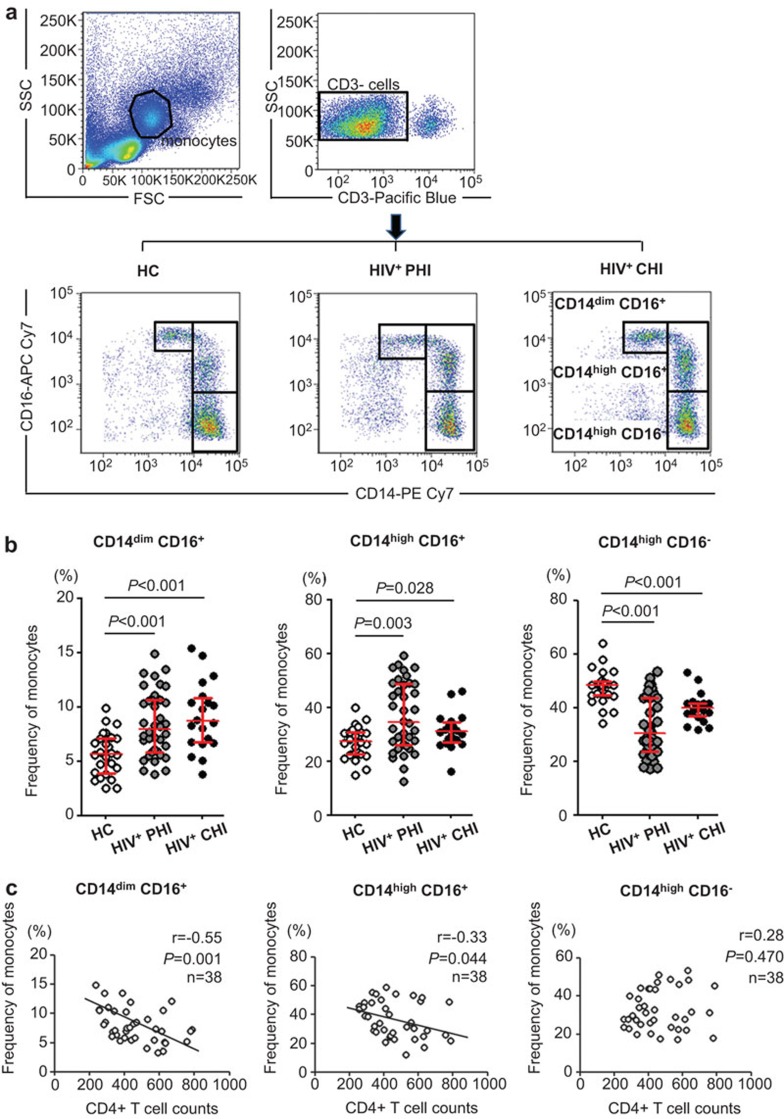
Whole blood samples were stained with anti-CD14 and anti-CD16 antibodies and analyzed by flow cytometry. The monocyte subsets were gated according to the expression levels of CD14 and CD16 (Figure 1a) using the following criteria: CD14dimCD16+ (non-classical monocytes), CD14highCD16+ (intermediate monocytes) and CD14highCD16− (classical monocytes). As shown in Figure 1b, the proportion of non-classical CD14dimCD16+ monocytes was increased in HIV-1-infected subjects in comparison with healthy controls, including both PHI and CHI patients (P<0.001 for both). Similarly, the proportion of intermediate CD14highCD16+ monocytes was also increased during HIV-1 infection (P=0.003 for PHI and P=0.028 for CHI). In contrast, the frequencies of classical CD14highCD16− monocytes were decreased during HIV-1 infection (P<0.001 for both PHI and CHI). No significant difference was observed between PHI and CHI regarding any of the three subsets of monocytes (Figure 1b).
Figure 1.
Altered distributions of monocyte subsets in HIV-1 infection. (a) Gating strategies of monocyte subsets according to CD14 and CD16 expression. (b) Frequencies (%) of CD14dimCD16+, CD14highCD16+ and CD14highCD16− monocyte subsets in primary HIV-1 infection (PHI, n=38), chronic HIV-1 infection (CHI, n=20), and healthy controls (HC, n=26). The lines show the median with the interquartile range. (c) Correlation analyses for CD14dimCD16+, CD14highCD16+ and CD14highCD16− monocyte subsets vs. CD4 T-cell counts in PHI (n=38). P<0.05 indicates significance.
We then analyzed the relationship between the frequencies of the monocyte subsets and the CD4 counts and HIV-1 viral load. In PHI, the percentages of CD14dimCD16+ non-classical monocytes and CD14highCD16+ intermediate monocytes were negatively associated with the CD4 counts (P=0.001, R=−0.55 and P=0.044, R=−0.33, respectively) (Figure 1c). No correlation was found between the monocyte subsets and the viral load in the PHI group (data not shown). Unlike the PHI group, the proportions of the monocyte subsets in the CHI group had no relationship with either CD4 counts or HIV-1 viral load (data not shown).
Formation of MPAs was increased in all three monocyte subsets during HIV-1 infection
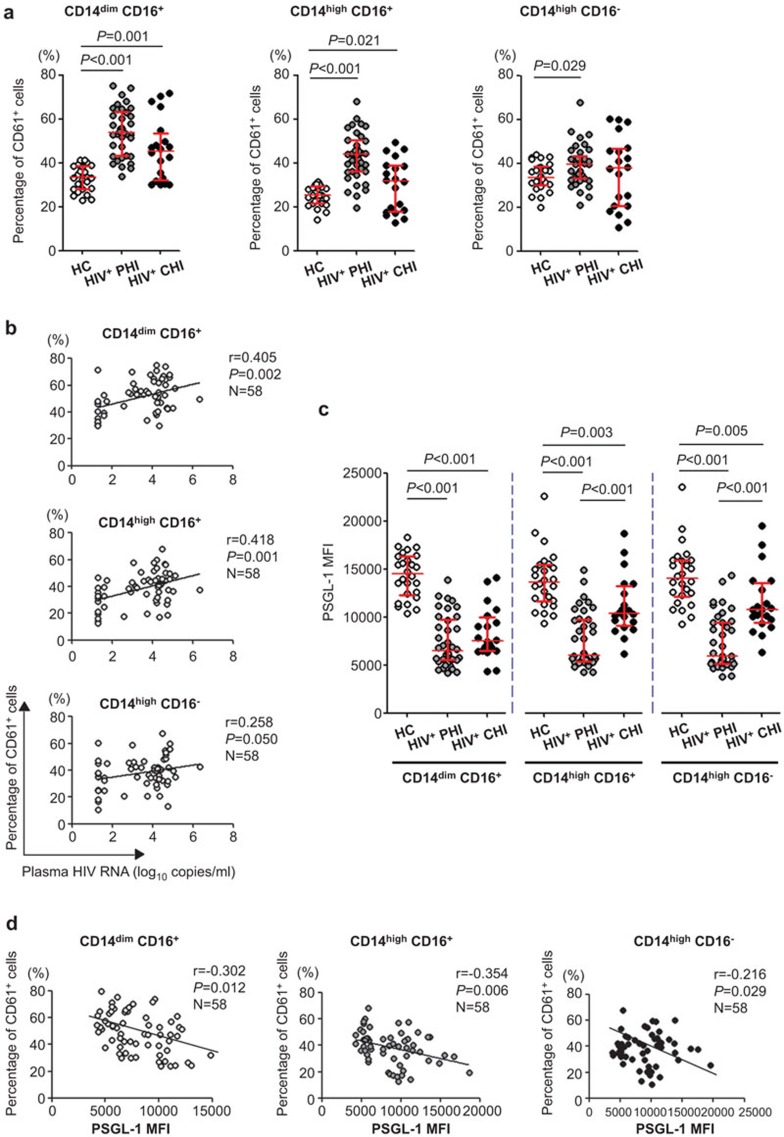
CD61 was expressed on platelets and can be used to gate and define MPA via the detection of CD61+ monocytes. The MPA levels were significantly increased in all three monocyte subsets in the PHI group (P<0.001, P<0.001 and P=0.029 for CD14dimCD16+, CD14highCD16+ and CD14highCD16− monocytes, respectively), while only CD14dimCD16+ non-classical monocytes and CD14highCD16+ intermediate monocytes exhibited increased MPA formation in the CHI group (P=0.001 and P=0.021, respectively) compared with healthy controls (Figure 2a). The proportions of MPAs in the CD14dimCD16+ non-classical and CD14highCD16+ intermediate monocyte subsets were found to be related to the HIV-1 viral load (P=0.002, r=0.405 and P=0.001, r=0.418, respectively) (Figure 2b). Because the formation of MPAs depends on the interactions between PSGL-1 on monocytes and P-selectin (CD62P) on platelets, PSGL-1 expression on monocyte subsets was determined. As shown in Figure 2c, PSGL-1 expression on all three monocyte subsets was decreased in the PHI group (P<0.001 for all monocyte subsets). It was partially restored in the CHI group but did not return to the level of the healthy controls (Figure 2c). PSGL-1 expression was negatively correlated with the MPA levels (P=0.012 and r=−0.302 for CD14dimCD16+ monocytes, P=0.006 and r=−0.354 for CD14highCD16+ monocytes and P=0.029 and r=−0.216 for CD14highCD16− monocytes; Figure 2d), indicating that PSGL-1 may be shed from the monocyte surface upon MPA formation.
Figure 2.
Increased MPA formation in CD14dimCD16+, CD14highCD16+ and CD14highCD16− monocyte subsets during HIV-1 infection. (a) MPA levels, as represented by the percentage of CD61+ cells, in individual monocyte subsets in PHI, CHI and healthy controls. The lines show the median with the interquartile range. (b) Correlation analyses for MPA levels in each monocyte subsets vs. the plasma viral load in HIV-1 infection (n=58). (c) PSGL-1 expression on three monocyte subsets in PHI, CHI and healthy controls. The data are shown as the MFI, and the lines show the median with the interquartile range. (d) Correlation analyses for MPA levels vs. PSGL-1 expression intensity in each monocyte subset in HIV-1 infection (n=58). P<0.05 indicates significance. CHI, chronic HIV-1 infection; MFI, mean fluorescent intensity; MPA, monocyte–platelet aggregate; PHI, primary HIV-1 infection; PSGL-1, P-selectin glycoprotein ligand-1.
Immune activation markers on monocyte subsets were altered in HIV-1 infection
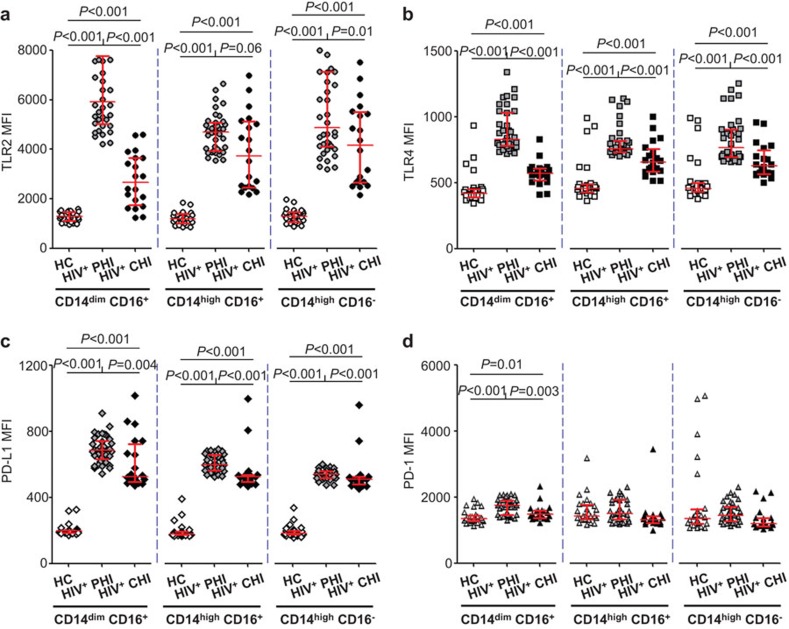
Differential expression of immune activation markers such as PD-1, PD-L1, TLR2 and TLR4 on monocytes has been previously reported. Here, we investigated the expression of these markers on different monocyte subsets in HIV-1-infected subjects. TLR2, TLR4 and PD-L1 expression levels were increased in all monocyte subsets in the PHI group (P<0.001 for all; Figure 3a–c). In the CHI group, the expression of these immune activation markers was decreased on all three monocyte subsets, but still could not return to the normal level of healthy controls (P<0.001 for all) (Figure 3a–c). In contrast, increased PD-1 expression was only observed on the CD14dimCD16+ monocyte subset (P<0.001 for PHI group and P=0.01 for CHI group; Figure 3d). These findings indicated that all three monocyte subsets were involved in immune activation during HIV-1 infection.
Figure 3.
Increased expression of surface immune activation markers on monocyte subsets in HIV-1 infection. Expression of TLR2 (a), TLR4 (b), PD-L1 (c) and PD-1 (d) on CD14dimCD16+, CD14highCD16+ and CD14highCD16− monocyte subsets in PHI, CHI and healthy controls. The data are shown as the MFI. The lines show the median with the interquartile range. P<0.05 indicates significance. CHI, chronic HIV-1 infection; MFI, mean fluorescent intensity; PD-1, programmed death 1; PHI, primary HIV-1 infection; TLR, Toll-like receptor.
The levels of sCD14 and sCD163 in the plasma are increased in HIV-1 infection and are related to disease progression
The plasma levels of soluble CD14 and CD163 were quantified in samples from 58 HIV-1-infected subjects and 26 healthy controls. Plasma sCD14 and sCD163 were elevated in both the PHI (P<0.001 for both sCD14 and sCD163) and CHI (P=0.002 for sCD14 and P<0.001 for sCD163) groups in comparison with healthy controls (Figure 4a). In addition, the sCD163 level in the CHI group was further increased relative to the PHI group (P=0.001) (Figure 4a). The levels of plasma sCD14 and sCD163 were negatively correlated with CD4 counts (P=0.028, r=−0.409 and P=0.017, r=−0.388, respectively) and were positively correlated with viral load (P=0.030, r=0.385 and P=0.034, r=0.406, respectively) (Figure 4b).
Figure 4.
Plasma levels of sCD14 and sCD163 in HIV-1 infection were altered and were positively correlated with HIV-1 viremia and negatively correlated with CD4 T-cell counts. (a) Plasma samples were tested for sCD14 and sCD163 levels by ELISA. The lines show the median with the interquartile range. (b) Correlation analyses of increased levels of sCD14 and sCD163 in the plasma vs. the CD4 T-cell counts (left panel) or vs. the plasma viral load (right panel) (n=58). P<0.05 indicates significance. CHI, chronic HIV-1 infection; HC, healthy control; PHI, primary HIV-1 infection.
MPA formation was differently associated with CD163 expression on monocytes and sCD163 levels in the plasma during HIV-1 infection
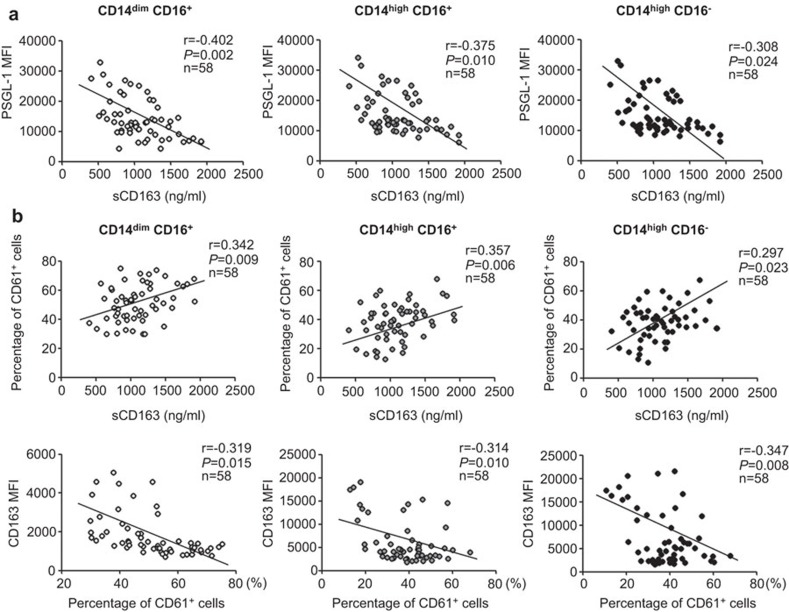
We have shown that PSGL-1 expression was negatively correlated with MPA formation (Figure 2d). Here, we further evaluated whether immune activation was responsible for MPA formation. As shown in Figure 5a, PSGL-1 expression was found to be negatively associated with the plasma levels of sCD163 (P=0.002 and r=−0.402 for CD14dimCD16+ monocytes, P=0.010 and r=−0.375 for CD14highCD16+ monocytes and P=0.024 and r=−0.308 for CD14highCD16− monocytes). Consistent with the inverse correlation between PSGL-1 expression and MPA levels (Figure 2d), the MPA levels in all three monocyte subsets were positively correlated with plasma sCD163 (P=0.009 and r=0.342 for CD14dimCD16+ monocytes, P=0.006 and r=0.357 for CD14highCD16+ monocytes and P=0.023 and r=0.297 for CD14highCD16− monocytes) (Figure 5b). Interestingly, we also found that the MPA level was negatively correlated with CD163 expression on each monocyte subset (P=0.015 and r=−0.319 for CD14dimCD16+ monocytes, P=0.010 and r=−0.314 for CD14highCD16+ monocytes, and P=0.008 and r=−0.347 for CD14highCD16− monocytes) (Figure 5b), although an inverse correlation between CD163 expression on monocytes and the plasma levels of sCD163 was not observed in this study as reported by other groups.27 Altogether, these findings suggest that the elevated immune activation status may be responsible for MPA formation during HIV-1 infection.
Figure 5.
Plasma levels of sCD163 were correlated with MPA levels and inversely correlated with PSGL-1 and CD163 expression in all three monocyte subsets. (a) Correlation analyses of PSGL-1 expression on CD14dimCD16+, CD14highCD16+ and CD14highCD16− monocyte subsets vs. sCD163 level in plasma (n=58). (b) Correlation analyses of MPA levels on CD14dimCD16+, CD14highCD16+ and CD14highCD16− monocyte subsets vs. sCD163 levels in the plasma (upper panel) or vs. CD163 expression on monocyte subsets (lower panel) (n=58). P<0.05 indicates significance. MPA, monocyte–platelet aggregate; PSGL-1, P-selectin glycoprotein ligand-1.
Discussion
HIV-1-infected individuals experience a higher risk of cardiovascular disease (CVD) compared with their uninfected counterparts.22,23,24 Increased immune activation of monocytes was reported to be involved in HIV-1 pathogenesis and thus associated with CVD risk.17,28,29 In the present study, we demonstrated that in contrast to the decreased percentage of CD14highCD16− monocyte subset, CD14dimCD16+ and CD14highCD16+ monocyte subsets were expanded during both primary and chronic HIV-1 infection. This was the first report of the monocyte subset distribution in primary HIV-1 infection, and it expanded previous observations that had focused on chronic HIV-1-infected patients, HARRT patients, and elite controllers.16,17,30 In contrast to the report by Han et al.,16 we found that CD14dimCD16+ and CD14highCD16− monocytes were negatively correlated with CD4 counts in PHI but not CHI. This difference might be due to our limited case numbers in the CHI group.
MPA levels have been previously associated with thrombosis and inflammation and are increased in CVD and autoimmune diseases.19,20,21,31 Here, we found that MPA levels were elevated in all three monocyte subsets in PHI and that they were increased in non-classical CD14dimCD16+ monocytes and intermediate CD14highCD16+ monocytes in CHI. To the best of our knowledge, this is the first report regarding the MPA status in primary and chronic HIV-1 infection without antiretroviral therapy. Because MPA formation increased CD16 expression on circulating CD14+CD16− monocytes,32 increased MPA levels may contribute to the increased proportions of CD14dimCD16+ and CD14highCD16+ monocyte subsets in HIV-1 infection. In addition, we found that the MPA levels in non-classical and intermediate monocytes were closely associated with HIV-1 viremia, indicating a potentially higher CVD risk in patients with active HIV-1 replication.
The formation of MPAs depends on the interaction of PSGL-1 on monocytes and P-selectin (CD62P) on platelets.33 Upon the PSGL-1/CD62P interaction, PSGL-1 induces the production of pro-inflammatory cytokines, such as monocyte chemoattractant protein 1 and tumor-necrosis factor-α, and coagulation-initiating tissue factor, thus enhancing the adhesion of MPA to endothelial cells.34,35 PSGL-1 was constitutively expressed on circulating monocytes and was decreased upon monocyte activation or monocyte–platelet interaction.36,37 Here, we demonstrated that PSGL-1 expression was decreased in all three monocyte subsets and was negatively associated with MPA levels. The decreased PSGL-1 expression and the associated decreased capacity of MPA formation may have been due to a feedback mechanism of the host immune system to prevent further lesions caused by MPA.
MPA formation resulted in monocyte activation and is associated with pro-inflammatory cytokine secretion, upregulated adhesion molecules and tissue factor expression.34,35,38,39,40 In our study, high levels of the immune activation markers TLR2, TLR4 and PD-L1 were found on all three monocyte subsets. sCD14 and sCD163 are monocyte/macrophage activation markers. They have been reported to be upregulated in HIV infection and can act as predictive markers of HIV disease progression.1,2,3,4,5,6 Increased coagulation biomarkers, such as D-dimer and/or thrombin-antithrombin complex, were also found to be correlated with sCD14 in pathogenic simian immunodeficiency virus infection.41 In addition, we found that increased plasma levels of sCD14 and sCD163 were positively correlated with HIV-1 viremia, but were negatively correlated with the CD4 counts. These data suggested that monocyte activation occurs in HIV-1 infection and plays a pivotal role in HIV disease progression. sCD163 was derived from the ectodomain shedding of membrane CD163 and was inversely correlated with CD163 expression.27,42 sCD163 was previously reported to be elevated and associated with CVD in HIV-infected or-uninfected patients.2,43 Interestingly, we found that the MPA levels of all three monocyte subsets were positively correlated with sCD163 levels, indicating that the increased formation of MPA in HIV-1 infection was associated with monocyte activation status. We could not determine whether MPA formation induced monocyte activation or whether monocyte activation enhanced MPA formation. However, other groups have reported that the formation of MPA is primarily dependent on platelet activation and to a lesser extent on monocyte activation.26,44,45 Therefore, we speculated that the increased monocyte activation in HIV-1 infection might be partially ascribed to MPA formation, which rendered monocytes more active, more adhesive, more procoagulant, and more inflammatory through the release of pro-inflammatory cytokines (such as IL-6 and tumor-necrosis factor-α) or the expression of tissue factor, resulting in an increased CVD risk.
In summary, we showed for the first time that MPA levels are increased on CD14dimCD16+, CD14highCD16+ and CD14highCD16− monocyte subsets and are correlated with viremia and plasma sCD163 levels in HIV-1 infection. The increased crosstalk between monocytes and platelets, together with increased HIV replication and monocyte activation, might contribute to the higher risk of CVD in HIV-1 infection.
Acknowledgments
We thank Jenny Hsi for her critical editing of the manuscript. This work was supported by the National Natural Science Foundation of China (31100126, 81271826, 81020108030, 81101281), SKLID Development grants (2011SKLID207, 2012SKLID103), The China National Major Projects for Infectious Diseases Control and Prevention (2012ZX10001008, 2014ZX10001001-002) and the Beijing Natural Science Foundation (7122108).
The authors declare that they have no competing financial interests.
References
- Burdo TH, Lentz MR, Autissier P, Krishnan A, Halpern E, Letendre S, et al. Soluble CD163 made by monocyte/macrophages is a novel marker of HIV activity in early and chronic infection prior to and after anti-retroviral therapy. J Infect Dis. 2011;204:154–163. doi: 10.1093/infdis/jir214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burdo TH, Lo J, Abbara S, Wei J, DeLelys ME, Preffer F, et al. Soluble CD163, a novel marker of activated macrophages, is elevated and associated with noncalcified coronary plaque in HIV-infected patients. J Infect Dis. 2011;204:1227–1236. doi: 10.1093/infdis/jir520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chevalier MF, Petitjean G, Dunyach-Remy C, Didier C, Girard PM, Manea ME, et al. The Th17/Treg ratio, IL-1RA and sCD14 levels in primary HIV infection predict the T-cell activation set point in the absence of systemic microbial translocation. PLoS Pathog. 2013;9:e1003453. doi: 10.1371/journal.ppat.1003453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knudsen A, Moller HJ, Katzenstein TL, Gerstoft J, Obel N, Kronborg G, et al. Soluble CD163 does not predict first-time myocardial infarction in patients infected with human immunodeficiency virus: a nested case–control study. BMC Infect Dis. 2013;13:230. doi: 10.1186/1471-2334-13-230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leeansyah E, Malone DF, Anthony DD, Sandberg JK. Soluble biomarkers of HIV transmission, disease progression and comorbidities. Curr Opin HIV AIDS. 2013;8:117–124. doi: 10.1097/COH.0b013e32835c7134. [DOI] [PubMed] [Google Scholar]
- Sandler NG, Wand H, Roque A, Law M, Nason MC, Nixon DE, et al. Plasma levels of soluble CD14 independently predict mortality in HIV infection. J Infect Dis. 2011;203:780–790. doi: 10.1093/infdis/jiq118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dolganiuc A, Garcia C, Kodys K, Szabo G. Distinct Toll-like receptor expression in monocytes and T cells in chronic HCV infection. World J Gastroenterol. 2006;12:1198–1204. doi: 10.3748/wjg.v12.i8.1198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma Y, He B. Recognition of herpes simplex viruses: Toll-like receptors and beyond. J Mol Biol. 2014;426:1133–1147. doi: 10.1016/j.jmb.2013.11.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arpaia N, Barton GM. Toll-like receptors: key players in antiviral immunity. Curr Opin Virol. 2011;1:447–454. doi: 10.1016/j.coviro.2011.10.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lester RT, Yao XD, Ball TB, McKinnon LR, Kaul R, Wachihi C, et al. Toll-like receptor expression and responsiveness are increased in viraemic HIV-1 infection. AIDS. 2008;22:685–694. doi: 10.1097/QAD.0b013e3282f4de35. [DOI] [PubMed] [Google Scholar]
- Meier A, Bagchi A, Sidhu HK, Alter G, Suscovich TJ, Kavanagh DG, et al. Upregulation of PD-L1 on monocytes and dendritic cells by HIV-1 derived TLR ligands. AIDS. 2008;22:655–658. doi: 10.1097/QAD.0b013e3282f4de23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rodriguez-Garcia M, Porichis F, de Jong OG, Levi K, Diefenbach TJ, Lifson JD, et al. Expression of PD-L1 and PD-L2 on human macrophages is up-regulated by HIV-1 and differentially modulated by IL-10. J Leukoc Biol. 2011;89:507–515. doi: 10.1189/jlb.0610327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ziegler-Heitbrock L, Ancuta P, Crowe S, Dalod M, Grau V, Hart DN, et al. Nomenclature of monocytes and dendritic cells in blood. Blood. 2010;116:e74–e80. doi: 10.1182/blood-2010-02-258558. [DOI] [PubMed] [Google Scholar]
- van de Veerdonk FL, Netea MG. Diversity: a hallmark of monocyte society. Immunity. 2010;33:289–291. doi: 10.1016/j.immuni.2010.09.007. [DOI] [PubMed] [Google Scholar]
- Cros J, Cagnard N, Woollard K, Patey N, Zhang SY, Senechal B, et al. Human CD14dim monocytes patrol and sense nucleic acids and viruses via TLR7 and TLR8 receptors. Immunity. 2010;33:375–386. doi: 10.1016/j.immuni.2010.08.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han J, Wang B, Han N, Zhao Y, Song C, Feng X, et al. CD14highCD16+ rather than CD14lowCD16+ monocytes correlate with disease progression in chronic HIV-infected patients. J Acquir Immune Defic Syndr. 2009;52:553–559. doi: 10.1097/qai.0b013e3181c1d4fe. [DOI] [PubMed] [Google Scholar]
- Funderburg NT, Zidar DA, Shive C, Lioi A, Mudd J, Musselwhite LW, et al. Shared monocyte subset phenotypes in HIV-1 infection and in uninfected subjects with acute coronary syndrome. Blood. 2012;120:4599–4608. doi: 10.1182/blood-2012-05-433946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shantsila E, Lip GY. The role of monocytes in thrombotic disorders. Insights from tissue factor, monocyte–platelet aggregates and novel mechanisms. Thromb Haemost. 2009;102:916–924. doi: 10.1160/TH09-01-0023. [DOI] [PubMed] [Google Scholar]
- Furman MI, Barnard MR, Krueger LA, Fox ML, Shilale EA, Lessard DM, et al. Circulating monocyte-platelet aggregates are an early marker of acute myocardial infarction. J Am Coll Cardiol. 2001;38:1002–1006. doi: 10.1016/s0735-1097(01)01485-1. [DOI] [PubMed] [Google Scholar]
- Sarma J, Laan CA, Alam S, Jha A, Fox KA, Dransfield I. Increased platelet binding to circulating monocytes in acute coronary syndromes. Circulation. 2002;105:2166–2171. doi: 10.1161/01.cir.0000015700.27754.6f. [DOI] [PubMed] [Google Scholar]
- Michelson AD, Barnard MR, Krueger LA, Valeri CR, Furman MI. Circulating monocyte–platelet aggregates are a more sensitive marker of in vivo platelet activation than platelet surface P-selectin: studies in baboons, human coronary intervention, and human acute myocardial infarction. Circulation. 2001;104:1533–1537. doi: 10.1161/hc3801.095588. [DOI] [PubMed] [Google Scholar]
- Knobel H, Jerico C, Montero M, Sorli ML, Velat M, Guelar A, et al. Global cardiovascular risk in patients with HIV infection: concordance and differences in estimates according to three risk equations (Framingham, SCORE, and PROCAM) AIDS Patient Care STDS. 2007;21:452–457. doi: 10.1089/apc.2006.0165. [DOI] [PubMed] [Google Scholar]
- Edwards-Jackson N, Kerr S, Tieu H, Ananworanich J, Hammer S, Ruxrungtham K, et al. Cardiovascular risk assessment in persons with HIV infection in the developing world: comparing three risk equations in a cohort of HIV-infected Thais. HIV Med. 2011;12:510–515. doi: 10.1111/j.1468-1293.2011.00916.x. [DOI] [PubMed] [Google Scholar]
- Pullinger CR, Aouizerat BE, Gay C, Coggins T, Movsesyan I, Davis H, et al. Metabolic abnormalities and coronary heart disease risk in human immunodeficiency virus-infected adults. Metab Syndr Relat Disord. 2010;8:279–286. doi: 10.1089/met.2009.0094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Metcalf Pate KA, Lyons CE, Dorsey JL, Shirk EN, Queen SE, Adams RJ, et al. Platelet activation and platelet–monocyte aggregate formation contribute to decreased platelet count during acute simian immunodeficiency virus infection in pig-tailed macaques. J Infect Dis. 2013;208:874–883. doi: 10.1093/infdis/jit278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singh MV, Davidson DC, Kiebala M, Maggirwar SB. Detection of circulating platelet–monocyte complexes in persons infected with human immunodeficiency virus type-1. J Virol Methods. 2012;181:170–176. doi: 10.1016/j.jviromet.2012.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davis BH, Zarev PV. Human monocyte CD163 expression inversely correlates with soluble CD163 plasma levels. Cytometry B Clin Cytom. 2005;63:16–22. doi: 10.1002/cyto.b.20031. [DOI] [PubMed] [Google Scholar]
- Funderburg NT, Jiang Y, Debanne SM, Storer N, Labbato D, Clagett B, et al. Rosuvastatin treatment reduces markers of monocyte activation in HIV infected subjects on antiretroviral therapy. Clin Infect Dis. 2014;58:588–595. doi: 10.1093/cid/cit748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alcaide ML, Parmigiani A, Pallikkuth S, Roach M, Freguja R, Della Negra M, et al. Immune activation in HIV-infected aging women on antiretrovirals—implications for age-associated comorbidities: a cross-sectional pilot study. PLoS One. 2013;8:e63804. doi: 10.1371/journal.pone.0063804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krishnan S, Wilson EM, Sheikh V, Rupert A, Mendoza D, Yang J, et al. Evidence for innate immune system activation in HIV-1 infected elite controllers. J Infect Dis. 2014;209:931–939. doi: 10.1093/infdis/jit581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sellam J, Proulle V, Jungel A, Ittah M, Miceli Richard C, Gottenberg JE, et al. Increased levels of circulating microparticles in primary Sjogren's syndrome, systemic lupus erythematosus and rheumatoid arthritis and relation with disease activity. Arthritis Res Ther. 2009;11:R156. doi: 10.1186/ar2833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rogacev KS, Cremers B, Zawada AM, Seiler S, Binder N, Ege P, et al. CD14++CD16+ monocytes independently predict cardiovascular events: a cohort study of 951 patients referred for elective coronary angiography. J Am Coll Cardiol. 2012;60:1512–1520. doi: 10.1016/j.jacc.2012.07.019. [DOI] [PubMed] [Google Scholar]
- Bournazos S, Rennie J, Hart SP, Fox KA, Dransfield I. Monocyte functional responsiveness after PSGL-1-mediated platelet adhesion is dependent on platelet activation status. Arterioscler Thromb Vasc Biol. 2008;28:1491–1498. doi: 10.1161/ATVBAHA.108.167601. [DOI] [PubMed] [Google Scholar]
- Celi A, Pellegrini G, Lorenzet R, de Blasi A, Ready N, Furie BC, et al. P-selectin induces the expression of tissue factor on monocytes. Proc Natl Acad Sci USA. 1994;91:8767–8771. doi: 10.1073/pnas.91.19.8767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weyrich AS, McIntyre TM, McEver RP, Prescott SM, Zimmerman GA. Monocyte tethering by P-selectin regulates monocyte chemotactic protein-1 and tumor necrosis factor-alpha secretion. Signal integration and NF-kappa B translocation. J Clin Invest. 1995;95:2297–2303. doi: 10.1172/JCI117921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davenpeck KL, Brummet ME, Hudson SA, Mayer RJ, Bochner BS. Activation of human leukocytes reduces surface P-selectin glycoprotein ligand-1 (PSGL-1, CD162) and adhesion to P-selectin in vitro. . J Immunol. 2000;165:2764–2772. doi: 10.4049/jimmunol.165.5.2764. [DOI] [PubMed] [Google Scholar]
- Ashman N, Macey MG, Fan SL, Azam U, Yaqoob MM. Increased platelet-monocyte aggregates and cardiovascular disease in end-stage renal failure patients. Nephrol Dial Transplant. 2003;18:2088–2096. doi: 10.1093/ndt/gfg348. [DOI] [PubMed] [Google Scholar]
- da Costa Martins PA, van Gils JM, Mol A, Hordijk PL, Zwaginga JJ. Platelet binding to monocytes increases the adhesive properties of monocytes by up-regulating the expression and functionality of beta1 and beta2 integrins. J Leukoc Biol. 2006;79:499–507. doi: 10.1189/jlb.0605318. [DOI] [PubMed] [Google Scholar]
- Yago T, Tsukuda M, Minami M. P-selectin binding promotes the adhesion of monocytes to VCAM-1 under flow conditions. J Immunol. 1999;163:367–373. [PubMed] [Google Scholar]
- Li G, Sanders JM, Bevard MH, Sun Z, Chumley JW, Galkina EV, et al. CD40 ligand promotes Mac-1 expression, leukocyte recruitment, and neointima formation after vascular injury. Am J Pathol. 2008;172:1141–1152. doi: 10.2353/ajpath.2008.070633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pandrea I, Cornell E, Wilson C, Ribeiro RM, Ma D, Kristoff J, et al. Coagulation biomarkers predict disease progression in SIV-infected nonhuman primates. Blood. 2012;120:1357–1366. doi: 10.1182/blood-2012-03-414706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moller HJ. Soluble CD163. Scand J Clin Lab Invest. 2012;72:1–13. doi: 10.3109/00365513.2011.626868. [DOI] [PubMed] [Google Scholar]
- Aristoteli LP, Moller HJ, Bailey B, Moestrup SK, Kritharides L. The monocytic lineage specific soluble CD163 is a plasma marker of coronary atherosclerosis. Atherosclerosis. 2006;184:342–347. doi: 10.1016/j.atherosclerosis.2005.05.004. [DOI] [PubMed] [Google Scholar]
- Furman MI, Benoit SE, Barnard MR, Valeri CR, Borbone ML, Becker RC, et al. Increased platelet reactivity and circulating monocyte-platelet aggregates in patients with stable coronary artery disease. J Am Coll Cardiol. 1998;31:352–358. doi: 10.1016/s0735-1097(97)00510-x. [DOI] [PubMed] [Google Scholar]
- Li N, Hu H, Lindqvist M, Wikstrom-Jonsson E, Goodall AH, Hjemdahl P. Platelet–leukocyte cross talk in whole blood. Arterioscler Thromb Vasc Biol. 2000;20:2702–2708. doi: 10.1161/01.atv.20.12.2702. [DOI] [PubMed] [Google Scholar]