Abstract
Chronic pain may be related to cardiovascular disease (CVD) risk. The current study examined whether persistent bodily pain was related to cardiovascular disease risk factors, whether these effects were moderated by body mass index (BMI), and, if not, whether chronic pain accounted for unique variance in CVD risk factors. Participants were women (N=2135) in the Study of Women's Health Across the Nation. A High Pain Frequency variable (high pain in 0 through 4 assessments) was coded to reflect the frequency of high levels of bodily pain across the first 3 years of the study. Six CVD risk factors and BMI were measured at follow-up year 3. High Pain Frequency and BMI were correlated significantly with risk factors, although effects for the former were small. Hierarchical multiple regressions revealed High Pain Frequency × BMI interactions for 5 of 6 CVD risk factors. Dissecting the interactions revealed a similar pattern across 4 risk factors: for women with normal BMI, there was a “dose-response” in which increasing frequency of high pain revealed increasingly worse CVD risk factor levels, whereas for women with obese BMI, high pain frequency was unrelated to risk factors. For obese women, increasing frequency of high pain was associated with higher blood glucose. Although BMI is a well-established CVD risk factor, evaluation of CVD risk level may be improved by considering the incidence of persistent pain, particularly in normal weight women (BMI<25kg/m2) lower BMI.
Keywords: Persistent pain, BMI, CVD risk factors, moderation
Chronic pain has detrimental effects on health and quality of life beyond the influence of pain itself. It has long been recognized that non-pain medical disorders are often comorbid with systemic inflammatory diseases associated with chronic pain, such as rheumatoid arthritis and systemic lupus erythematosus. However, the impact of chronic pain not arising from systemic inflammatory disease or cancer on comorbid medical disorders has only recently been revealed. Evidence points toward an association between chronic pain from musculoskeletal or other nonmalignant conditions and cardiovascular disease (CVD) risk factors (Bruehl, Chung, Jirjis & Biridepalli, 2005; Goodson et al., 2013; Gureje, Akinpelu, Uwakwe, Udofia & Wakil, 2007; Olsen et al., in press; Thommasen & Zhang, 2006; Van den Bussche et al, 2011; Von Korff et al., 2005). If chronic pain influences the development or worsening of CVD, important implications for our understanding of chronic pain and its clinical treatment would emerge.
Studies examining a link between chronic pain and CVD risk have focused almost exclusively on elevated blood pressure (BP) or hypertension (HTN). Von Korff and colleagues (Von Korff et al., 2005) reported that individuals with chronic spinal pain in the past 12 months were significantly more likely to be diagnosed with HTN than individuals without chronic spinal pain, with a 50% greater risk of HTN in the former group. In a Nigerian sample, the risk of having HTN was three times greater in individuals reporting chronic spinal pain in the past 12 months than in those who were pain-free (Gureje et al., 2007). A study conducted in a German population indicated that people with back pain had a significantly greater lifetime prevalence of HTN than those without pain (Schneider, Mohnen, Schiltenwolf, & Rau, 2007). In a recent study of 10,000 Norwegian adults, individuals reporting chronic pain at any body location for at least the past 90 days were at 23% higher risk of experiencing HTN than individuals without pain, even adjusting for age, sex, and BMI (Olsen et al., in press). Finally, evidence suggests there may be a dose-response relationship between chronic pain and CVD risk. In a cross-sectional sample from the Framingham study (aged 72 years and older), frequent musculoskeletal pain was associated with higher resting SBP, and SBP increased linearly with the number of painful body sites (Leveille, Zhang, McMullen, Kelly-Hayes & Felson, 2005). Similarly, among individuals with chronic musculoskeletal pain, mortality due to CVD increased in direct proportion to the number of body sites affected by pain (McBeth et al., 2009).
Other evidence suggests that chronic pain is related to CVD risk factors other than HTN. Findings from population-based and clinic studies indicate that presence of chronic low back pain is related to an elevated incidence of metabolic syndrome (Duruoz, Turan, Gurgan & Deveci, 2013; Ha, 2011). High BMI, and in particular greater waist circumference, are also associated with chronic pain (Hitt, McMillen, Thorton-Neaves, Koch & Cosby, 2007; Goodson et al., 2013; Ray, Lipton, Zimmerman, Katz, Derby, 2011; Stone & Broderick, 2012; Urquhart et al., 2011; Wright et al., 2010). Other results indicate that chronic pain not due directly to inflammation is related to low-grade elevations in a number of systemic inflammatory markers including CRP (Ray et al., 2011), tumor necrosis factor- α (Wang et al., 2010), interleukin-6, (Livshits et al., 2009), and dyslipidaemia (Goodson et al., 2013). A definitive connection between chronic pain and CVD risk cannot be concluded from extant studies, but these limited data are consistent with such a relationship. Additional work based on population studies that include factors comprising the metabolic syndrome, and inflammatory and coagulation markers are needed.
It is also not clear whether the reported link between chronic pain and CVD risk markers, such as HTN, are due to unique aspects of chronic pain, overlap with other, well-established risk factors for CVD, or work in concert with other factors to magnify risk. A chief candidate is obesity. Numerous findings indicate that obesity adversely affects cardiac function, increases CVD risk factors, and is an independent risk factor for development of CVD (Bogers et al., 2007; de Konig, Merchant, Pogue & Anand, 2007; Klein et al, 2004; Lavie et al., 2009; Poirier et al., 2006). Body mass index (BMI), as an index of obesity, is related to elevated SBP, circulating CRP and fibrinogen, dyslipidaemia, and decreased HDL (Klein et al., 2004; Goodson et al., 2013; Lavie et al., 2009; Poirier et al., 2006). BMI and other measures of adiposity have also been linked to inflammation (Santos, Oliveira, Lopes, 2013), which is a known contributor to CVD risk.
Given that people with chronic pain tend to be more obese than those without chronic pain, it may be the case that chronic pain is related to CVD risk factors primarily through its association with obesity. Alternatively, chronic pain may have a relationship with CVD risk factors that depends on level of BMI; that is, BMI may moderate links between chronic pain and CVD risk. Because BMI is a robust and well-established CVD risk factor (Bogers et al., 2007; de Konig, Merchant, Pogue & Anand, 2007; Klein et al, 2004; Lavie et al., 2009; Poirier et al., 2006), we speculated that significant interactions between may be decomposed to show that chronic pain is positively related to CVD risk primarily at lower versus higher levels of BMI.
In the present study, we examined the degree to which persistent pain and BMI among women participating in the Study of Women's Health Across the Nation (SWAN; Sowers et al., 2000) were related to CVD risk markers as indexed by SBP, triglycerides, HDL, fasting glucose, and blood levels of CRP (a measure of inflammation) (Clearfield, 2005), and fibrinogen (a measure of platelet aggregability) (Stec et al., 2000). In addition, analyses were conducted to determine the degree to which persistent pain interacted with BMI to predict CVD risk factors. In the case of non-significant interactions, analyses were conducted to determine the degree to which variance accounted for in CVD risk factors by persistent pain was unique from or shared with BMI.
Method
Participants
SWAN, a cohort study of community-based midlife women, enrolled subjects in 7 sites in the United States. Baseline examination, conducted between 1995 and 1997, recruited 3302 women from 5 ethnic/racial groups (Caucasian, African American, Japanese, Chinese, Hispanic). Each site had approximately 50% Caucasian and 50% Non-Caucasian enrollment, with 1 Non-Caucasian ethnic/racial group per site. Women aged 42 to 52 years with an intact uterus and at least 1 ovary were invited to participate in SWAN if they had menstruated in the previous 3 months, were not currently pregnant or breast feeding, and had not used reproductive hormone preparations affecting ovarian or pituitary function in the past 3 months. Several population sampling techniques were used by the 7 sites, as previously described (Sowers et al., 2000). All women provided written informed consent. The Institutional Review Board at each site approved the study. We performed a cross-sectional study of persistent pain and CVD risk factor markers starting with the SWAN participants (N = 2135) who completed their fourth annual assessment (baseline and 3 follow-up assessments). See Table 1 for characteristics of the sample.
Table 1.
Characteristics of Sample Participants from the Study of Women's Health Across the Nation (SWAN)
| Age (years) M (SD) |
48.99 (2.68) |
| Education n (%) | |
| < High school | 107 (5.0%) |
| High school grad | 336 (15.7%) |
| Some college | 670 (31.4%) |
| College grad | 468 (21.9%) |
| Grad school | 540 (25.3%) |
| Race/ethnicity n (%) | |
| African American | 488 (22.9%) |
| Caucasian | 1079 (40.5%) |
| Chinese | 217 (10.2%) |
| Hispanic | 112 (5.2%) |
| Japanese | 239 (11.2%) |
| Menopausal Status n (%) | |
| pre-menopausal | 265 (12.4%) |
| early peri-menopausal | 1124 (52.6%) |
| late peri-menopausal | 184 (8.6%) |
| post-menopausal | 250 (11.7%) |
| surgical menopausal | 67 (3.1%) |
| undetermined but not post-menopausal | 242 (11.3%) |
| Pain medications (yes) | 677 (31.7%) |
| Anticoagulants (yes) | 29 (1.4%) |
| Cholesterol medication (yes) | 80 (3.7%) |
| Blood pressure medication (yes) | 359 (16.8%) |
| Insulin (yes) | 82 (3.8%) |
| BMI M (SD) | 28.50 kg/m2 (7.32) |
| SBP | 115.59 mm Hg (16.38) |
| HDL | 59.79 mg/dl (15.93) |
| Triglycerides (log) | 4.69 mg/dl (.51) |
| Fasting Glucose | 95.87 mg/dl (28.60) |
| C-reactive protein (log) | .56 mg/l (1.34) |
| Fibrinogen | 276.54 mg/dl (53.01) |
Measures
Trained interviewers conducted annual exams via clinic or home visits. Survey exams (both self-assessments and interviews) were completed in-person and via telephone. Common protocols were standardized and used across the 7 sites. Information obtained at baseline and annual follow-up assessments included demographics (age, ethnicity, level of education, etc.), and anthropometric data.
Chronic Pain
The item from the Medical Outcomes Study Short Form 36 (SF-36; Ware, Kosinski, & Keller, 1994) regarding bodily pain intensity during the past 4 weeks (“How much bodily pain have you had during the past 4 weeks?” range 1-6) was used as a basis for identifying people with varying frequency of reporting high severity of pain. The number of assessments at which people scored in the upper tertile (scored 5 or 6 on a 6-point scale, corresponding to “severe” and “very severe”) of this item's score distribution was tabulated. The variable “High Pain Frequency” was constructed to reflect whether people had not scored at or above the upper tertile in any of the four assessments (n = 1074), scored once at or above the upper tertile in any of the four assessments (n = 451), scored twice at or above the upper tertile in any of the four assessments (n = 285), scored three times at or above the upper tertile in any of the four assessments (n = 180), or they scored at or above the upper tertile in all four assessments (n = 145).
Body Mass Index (BMI)
BMI was measured with the participant in nonrestrictive undergarments. BMI was calculated as weight (kg)/[height (m)2]. These measures were collected at the Year 3 assessment.
Anthropometric Measures
BP was measured according to a standardized protocol, with readings taken on the right arm and with the participant seated with feet flat on the floor for at least 5 minutes before measurement. Respondents had not smoked or consumed any caffeinated beverage within 30 minutes of BP measurement. A standard mercury sphygmomanometer was used to record systolic and diastolic pressures at the first and fifth Korotkoff sounds. Two sequential BP values, with a minimum 2-minute rest between measures, were obtained and averaged.
Phlebotomy was performed in the morning after an overnight fast. Subjects were scheduled for venipuncture on days 2 to 5 of a spontaneous menstrual cycle within 60 days of assessment date. All samples were maintained at 4°C until separated and then were frozen at −80°C and shipped on dry ice to a central laboratory (Medical Research Laboratories, Highland Heights, KY, USA), which is certified by the National Heart Lung and Blood Institute, Centers for Disease Control Lipid Standardization Part III program. Total cholesterol and triglyceride levels were analyzed using enzymatic methods (Hitachi747analyzer; Boehringer Mannheim Diagnostics, Indianapolis, Indiana) as previously described (Steiner, Freidel, Bremner, & Stein 1981). HDL-C was isolated using heparin–manganese chloride (Warnick, & Albers, 1978).
These measures were collected at the Year 3 assessment.
Markers of Inflammation and Coagulation
Fibrinogen was measured in frozen citrated plasma using a clot-based turbidometric detection system. CRP was measured using an ultra-sensitive rate immunonephelometry (Dade- Behring, Marburg, Germany). Both of these indexes were also collected at Year 3 assessment.
Covariates
To control for the potential confounding effects of demographic factors, menopausal status and medication use, variables reflecting relevant factors were used in analyses. Demographic information included age, ethnicity, and education. Education was self-reported as 1 of 5 categories (less than high school, high school, some college, college, graduate school). Less than high school and high school were combined (referent group; coded “1”), and some college, college, graduate school were combined (coded “0”). Race/ethnicity was self-identified as 1 of 5 categories (Caucasian, African American, Hispanic, Japanese, or Chinese) and modeled in analyses with “Caucasian” as the referent group (coded “1”; other groups coded “0”). Menopausal status was assessed as pre-menopausal (referent group; coded “1”), early perimenopausal, late peri-menopausal, post-menopausal, surgical menopausal, and undetermined due to hormone therapy use but not post-menopausal (all coded “0”). Medication use was assessed at the Year 3 visit as whether or not (“yes” or “no”) participants had taken anticoagulants, lipid lowering agents, antihypertensive medications, insulin or oral medications to control blood glucose, and prescription or over-the-counter pain medications since the last interview.
Data Analyses
Triglycerides and CRP levels were log-transformed to improve distributions. Pearson zero-order correlations were generated to examine the associations between the High Pain Frequency variable, BMI, and CVD risk factors.
Multiple regression analyses were performed separately for SBP, fasting glucose levels, HDL, triglycerides, and plasma CRP levels and fibrinogen levels. Interaction terms were computed by multiplying High Pain Frequency by BMI values. Analyses involved three steps. First, general covariates (age, ethnicity, education, menopausal status, use of analgesic medication) and specific covariates (e.g., use of cholesterol lowering medication when the dependent varabiles were HDL or triglycerides) were entered first. Second, the High Pain Frequency and BMI main effect terms were entered simultaneously. Third, the High Pain frequency × BMI interaction term was entered. Significant interactions were dissected by deriving regression equations for BMI values regressed on the CVD risk factors separately for participants with each High Pain Frequency score (i.e., 0 – 4). To illustrate these effects, the separate regression equations were solved for hypothetical values that were based on clinical norms for BMI using the procedure described by Aiken and West (1991). Namely, the value for “Normal” was defined as the mean of the subsample of people falling within the BMI range 18.5 – 24.9 (M = 22.01; n = 701). The value for “Overweight” was defined as the mean of the subsample of people falling within the BMI range 25 – 29.9 (M = 27.29; n = 643). The value for “Obese” was defined as the mean of the subsample of people falling within the BMI range 30 and above (M = 36.89; n = 791). The interactions were further probed by testing the relationships between the High Pain Frequency variable and CVD risk factor for the normal, overweight and obese groups, as defined within the BMI intervals described above.
In the case of nonsignificant interactions, the semi-partial correlations of High Pain Frequency and BMI were examined as indexes of unique relationships. That is, a significant semi-partial correlation for High Pain Frequency and/or BMI would signal a significant unique effect.
Results
Correlations among All Variables
Correlation coefficients were generated among High Pain Frequency, BMI, SBP, fasting glucose, HDL, log-transformed triglycerides, log-transformed CRP and fibrinogen levels. Small to moderate relationships were found among variables indicating that greater frequency of having high bodily pain and greater BMI were associated with increased CVD risk markers (see Table 2). In addition, High Pain Frequency was related significantly to BMI (r = .26; p < .01), indicating that women with greater frequency of high pain had higher BMI.
Table 2.
Zero-Order Correlations
| SBP | HDL | Triglycerides | Glucose | CRP | Fibrinogen | |
|---|---|---|---|---|---|---|
| High Pain Frequency | .17 | −.12 | .13 | .16 | .17 | .09 |
| BMI | .37 | −.37 | .27 | .32 | .60 | .40 |
Note. All r's significant at p < .05. Triglycerides = log transformed triglycerides. Glucose = fasting glucose. CRP = log transformed CRP.
Tests of Moderation or Unique and Shared Effects
SBP
Adjusted for covariates, the High Pain Frequency × BMI interaction was nonsignificant (see Table 3) for SBP as the criterion. Note that relationships between covariates and all the CVD risk factors were in expected directions (e.g., higher education was related negatively to SBP levels). To ascertain the unique relationships between High Pain Frequency and SBP and between BMI and SBP, we examined the semi-partial correlations when both variables were entered simultaneously (Step 2). The semi-partial correlation between High Pain Frequency and SBP was nonsignificant (sr = .01), whereas the semi-partial correlation for BMI was significant (sr = .27; p < .01). Despite a significant zero-order correlation between High Pain Frequency and SBP, the unique association of this factor with SBP was rendered nonsignificant with covariates and BMI controlled. BMI, in contrast, accounted for a unique portion of variance even after controlling for covariates and High Pain Frequency.
Table 3.
Results of Hierarchical Regressions Testing High Pain Frequency × BMI Interactions
| SBP | B | SE | t | p-value |
|---|---|---|---|---|
| Step 1: Covariates | ||||
| Education | 3.20 | .82 | 3.91 | .001 |
| Age | .52 | .13 | 4.07 | .001 |
| Pain Meds | 2.12 | .70 | 3.01 | .003 |
| Ethnicity | −4.40 | .67 | −6.55 | .001 |
| Menopausal status | .60 | .94 | .64 | >.10 |
| BP meds | 14.19 | .89 | 15.91 | .001 |
| R2 of Step 1 = .17; p < .001 | ||||
| Step 2: Main effects | ||||
| High Pain Frequency | .32 | .26 | 1.22 | >.10 |
| BMI | .67 | .05 | 14.62 | .001 |
| R2 of Steps 1, 2 = .25; p < .001 | ||||
| Step 3: Interaction term | ||||
| High Pain Frequency x BMI | −.05 | .03 | −1.48 | >.10 |
| R2 of Steps 1, 2, 3 = .25; p < .001 | ||||
| HDL | B | SE | t | p-value |
|---|---|---|---|---|
| Step 1: Covariates | ||||
| Education | −4.21 | .85 | 4.93 | .001 |
| Age | .72 | .13 | 5.39 | .001 |
| Pain Meds | −1.01 | .74 | −1.37 | >.10 |
| Ethnicity | .45 | .70 | .65 | >.10 |
| Menopausal status | 2.02 | .99 | 2.04 | >.10 |
| Cholesterol meds | −9.20 | 1.80 | −5.11 | .001 |
| R2 of Step 1 = .04; p < .001 | ||||
| Step 2: Main effects | ||||
| High Pain Frequency | −.39 | .27 | −1.45 | > .10 |
| BMI | −.79 | .05 | −17.43 | .001 |
| R2 of Steps 1, 2 = .17; p < .001 | ||||
| Step 3: Interaction term | ||||
| High Pain Frequency × BMI | .14 | .03 | 4.35 | .001 |
| R2 of Steps 1, 2, 3 = .18; p < .001 | ||||
| Triglycerides (log) | B | SE | t | p-value |
|---|---|---|---|---|
| Step 1: Covariates | ||||
| Education | .11 | .03 | 3.73 | .001 |
| Age | .02 | .01 | 4.07 | .001 |
| Pain Meds | .03 | .02 | 1.26 | >.10 |
| Ethnicity | .04 | .02 | 1.86 | .07 |
| Menopausal status | .03 | .03 | 1.03 | >.10 |
| Cholesterol meds | .41 | .06 | 7.15 | .001 |
| R2 of Step 1 = .05; p < .001 | ||||
| Step 2: Main effects | ||||
| High Pain Frequency | .02 | .01 | 2.41 | .02 |
| BMI | .02 | .01 | 9.78 | .001 |
| R2 of Steps 1, 2 = .10; p < .001 | ||||
| Step 3: Interaction term | ||||
| High Pain Frequency × BMI | −.01 | .01 | −2.16 | .03 |
| R2 of Steps 1, 2, 3 = .11; p < .001 | ||||
| CRP (log) | B | SE | t | p-value |
|---|---|---|---|---|
| Step 1: Covariates | ||||
| Education | .33 | .07 | 4.48 | .001 |
| Age | .01 | .01 | −.04 | >.10 |
| Pain Meds | .34 | .06 | 5.35 | .001 |
| Ethnicity | .13 | .06 | 2.18 | .03 |
| Menopausal status | .16 | .02 | 1.84 | .07 |
| R2 of Step 1 = .03; p < .001 | ||||
| Step 2: Main effects | ||||
| High Pain Frequency | .01 | .02 | .15 | >.10 |
| BMI | .11 | .01 | 32.18 | .001 |
| R2 of Steps 1, 2 = .37; p < .001 | ||||
| Step 3: Interaction term | ||||
| High Pain Frequency × BMI | −.01 | .01 | −5.09 | .001 |
| R2 of Steps 1, 2, 3 = .38; p < .001 | ||||
| Fibrinogen | B | SE | t | p-value |
|---|---|---|---|---|
| Step 1: Covariates | ||||
| Education | 13.06 | 3.03 | 4.32 | .001 |
| Age | .85 | .47 | 1.81 | .07 |
| Pain Meds | 7.82 | 2.61 | 2.48 | .01 |
| Ethnicity | −4.24 | 2.46 | −1.72 | .09 |
| Menopausal status | 3.62 | 3.54 | 1.02 | > .10 |
| Anti-coagulant meds | 35.72 | 10.31 | 3.46 | .001 |
| R2 of Step 1 = .03; p < .001 | ||||
| Step 2: Main effects | ||||
| High Pain Frequency | −1.63 | .94 | −1.70 | >.10 |
| BMI | 2.88 | .16 | 18.11 | .001 |
| R2 of Steps 1, 2 = .17; p < .001 | ||||
| Step 3: Interaction term | ||||
| High Pain Frequency × BMI | −.27 | .11 | −2.42 | .02 |
| R2 of Steps 1, 2, 3 = .18; p < .001 | ||||
| Fasting Glucose | B | SE | t | p-value |
|---|---|---|---|---|
| Step 1: Covariates | ||||
| Education | 9.54 | 1.59 | 6.01 | .001 |
| Age | .18 | .25 | .73 | >.01 |
| Pain Meds | 3.37 | 1.37 | 2.47 | .02 |
| Ethnicity | −3.92 | 1.29 | −3.04 | .002 |
| Menopausal status | .60 | 1.84 | .32 | >.10 |
| Insulin | 14.73 | 5.59 | 2.64 | .008 |
| R2 of Step 1 = .03; p < .001 | ||||
| Step 2: Main effects | ||||
| High Pain Frequency | 1.34 | .51 | 2.65 | .008 |
| BMI | 1.24 | .09 | 14.68 | .001 |
| R2 of Steps 1, 2 = .14; p < .001 | ||||
| Step 3: Interaction term | ||||
| Persistent Pain × BMI | .13 | .06 | 2.21 | .03 |
| R2 of Steps 1, 2, 3 = .15; p < .001 | ||||
HDL, Triglycerides, C-Reactive Protein and Fibrinogen
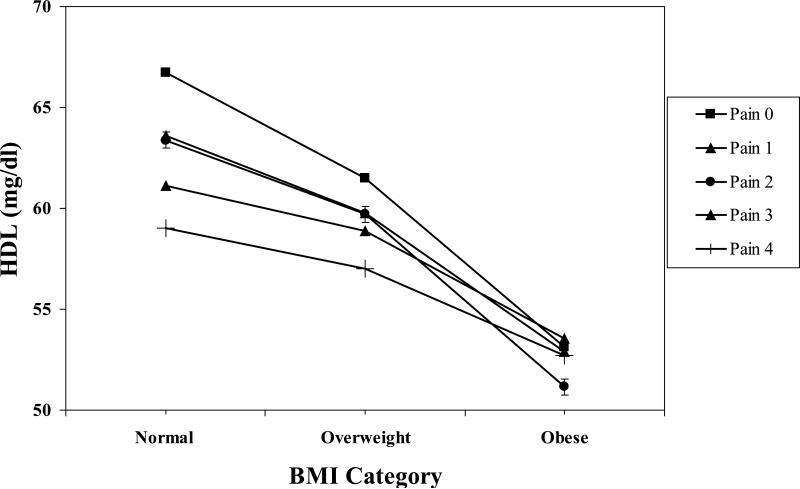
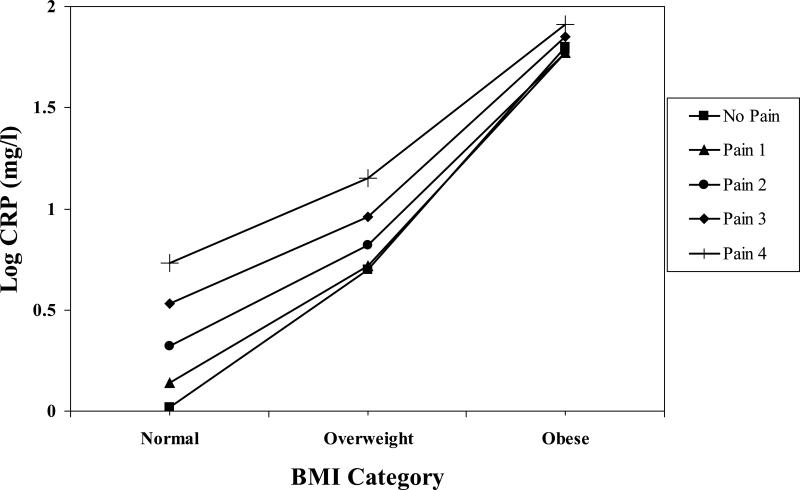
The High Pain Frequency × BMI interactions were significant (see Table 3) for HDL, triglycerides, CRP and fibrinogen after adjusting for covariates. To illustrate the significant interactions, BMI was first regressed on the CVD risk factor values separately for participants with each High Pain Frequency score (i.e., 0 – 4). With covariates entered first, regressions showed that BMI was related significantly to all the CVD factors at different levels across the High Pain Frequency distribution. For example, for HDL, the beta weights for the five High Pain Frequency values were −.39, −.34, −.42, −.22, −.31, respectively. The pattern of beta weights was similar across HDL, triglycerides, CRP and fibrinogen.
Next, we solved the five regression equations using hypothetical BMI values (22.01, 27.29, 36.89) approximately corresponding to clinical norms for normal, overweight and obese. The resulting hypothetical values for HDL are shown in Figure 1. The pattern of values for triglycerides, CRP and fibrinogen showed a similar pattern, and are illustrated with the single example of CRP values in Figure 2. To further probe the interaction, the other simple effects – namely, the relationships between the High Pain Frequency variable and the CVD risk factors for the normal, overweight and obese groups – were examined. Again, a similar pattern of simple effects emerged across the four CVD risk factors. For example, the beta weights for the relationship between High Pain Frequency and HDL levels for the normal group, after entering covariates, was −.10 (p < .04), whereas the beta weights for the overweight (−.02) and obese groups (−.02) were nonsignificant. This pattern was repeated for triglycerides, CRP and fibrinogen.
Figure 1.
High Pain Frequency × BMI interaction for HDL. The value for Normal BMI = 22.01. The value for Overweight BMI = 27.29. The value for Obese BMI = 36.89.
Figure 2.
High Pain Frequency × BMI interaction for C-Reactive Protein (log). The value for Normal BMI = 22.01. The value for Overweight BMI = 27.29. The value for Obese BMI = 36.89.
Results suggest that among women with normal BMI, there is a “dose response” for HDL, triglycerides, CRP and fibrinogen levels depending on frequency of high pain report. Namely, the relationship between High Pain Frequency for women with normal BMI was significant and suggested that those with no reports of frequent high pain had more favorable CVD risk factor levels than women with normal weight but increasingly frequent reports of high pain. Those women with the most frequent high pain (high pain at all four assessments) had the least favorable CVD risk factor values. Relationships between High Pain Frequency and the CVD risk factors for the overweight and obese women were nonsignificant, suggesting that differences in these factors across high pain reports appeared to diminish among these groups.
Finally, because the interactions were significant, thus describing multiplicative relationships between high pain frequency and BMI, unique and shared contributions were not examined.
Fasting Glucose
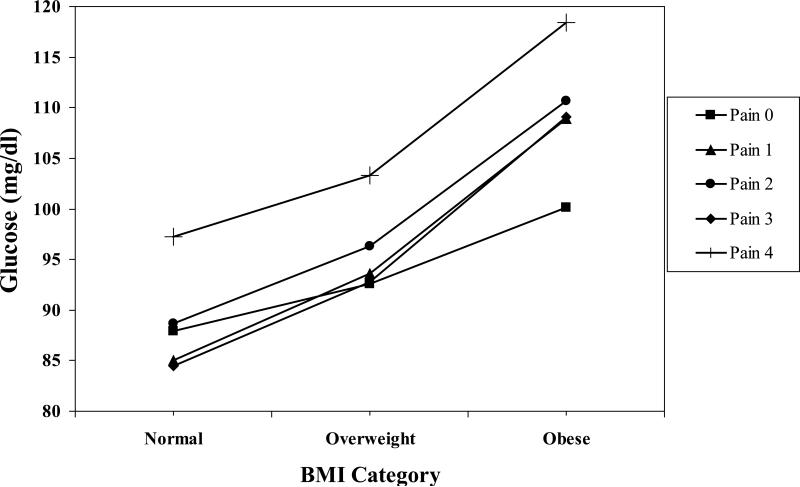
Adjusted for covariates, the High Pain Frequency × BMI interaction was significant (see Table 3) for fasting glucose as the criterion. The procedure described above to dissect and illustrate the interaction was used here. With covariates entered, regressions for each High Pain Frequency value showed that BMI was related significantly to glucose but at different levels across the High Pain Frequency distribution (beta weights were .30, .40, .30, .43, .22; respectively). Solving the five regression equations, as described above, gave the values shown in Figure 3. As described above, the relationships between the High Pain Frequency variable and glucose for the normal, overweight and obese groups were examined. The beta weights for the relationships for the normal (.05; p > .10) and overweight groups (.04; p > .10) were nonsignificant, whereas the beta weight for the obese group (.11; p < .005) was significant.
Figure 3.
High Pain Frequency × BMI interaction for Fasting Glucose. The value for Normal BMI = 22.01. The value for Overweight BMI = 27.29. The value for Obese BMI = 36.89.
Unlike HDL, triglycerides, CRP and fibrinogen, results suggest that among women with obese BMI, there is a “dose response” for glucose depending on frequency of high pain report. The relationship between High Pain Frequency for women with obese BMI was significant and positive, indicating that those with no reports of frequent high pain had lower glucose than women with obese BMI but increasingly frequent reports of high pain. Obese women with the most frequent high pain had the highest glucose. Relationships between High Pain Frequency and glucose for the normal and overweight women were nonsignificant, suggesting that differences in glucose across high pain reports were minimal among these groups.
Discussion
Findings from epidemiological and clinical studies suggest that chronic pain may be related to CVD risk factors. Whereas most studies have focused on elevated BP and established HTN, there are some data to support an association between chronic pain and a wider CVD risk profile. Here, in a large cohort sample of community-based women, we found in univariate analyses that the frequency in which women reported high bodily pain over three years was related significantly to a host of CVD risk factors in directions indicating that frequent pain was related to elevated CVD risk. We also evaluated the degree to which frequent high pain combined with BMI to affect CVD risk factors, either in moderation or in terms of unique and shared relationships. The results revealed that significant High Pain Frequency × BMI interactions qualified the univariate relationships for the CVD risk factors, except for SBP. Findings indicated that women who did not report high pain at all over the four years and had normal BMI were characterized by a low level of risk, whereas the otherwise optimal CVD risk profile for those with normal BMI was increasingly eroded as a function of how often they reported high bodily pain. For glucose, this increasing level of risk with increasing frequency of high bodily pain appeared for obese women.
The frequency in which women reported high bodily pain was correlated significantly in univariate analyses with all CVD risk factors included in this study. Results are consistent with past findings regarding links between chronic pain and blood pressure, and extend past findings by showing relationships between chronic pain and other CVD risk factors. Thus, chronic pain was correlated significantly with elevated CRP, fasting glucose, fibrinogen and triglycerides, and was related to lower levels of HDL. Although correlations were small, results hint that chronic pain is related to a wide range of CVD risk factors. At the same time, a close examination of the regression tables reveals that none of the beta weights for High Pain Frequency were significant with BMI and covariates also in the equations. These results hint that relationships between chronic pain and CVD risk factors may be primarily due to shared variance with other CVD risk factors such as BMI, low SES, etc.
The critical and novel finding was that the univariate relationships were qualified by significant interactions. Namely, the relationships between high pain frequency and HDL, triglycerides, glucose, CRP and fibrinogen were moderated by BMI. For HDL, triglycerides, CRP and fibrinogen, a similar pattern emerged where BMI altered the relationship between the frequency of high bodily pain and the risk factors such that women with increasingly frequent reports of high pain but normal BMI revealed an increasingly higher level of risk than normal BMI women who did not report high pain at any time across the four assessments. Put otherwise, the advantage gained by women with normal BMI was partly nullified in statistically significant “dose response” effects that depended on how often they reported high bodily pain over three years. The moderation relationships were also characterized by the differences on risk factors across high pain frequency scores generally decreasing for overweight and obese women. Indeed, for obese women, participants with different high pain frequencies nonetheless revealed similar levels of risk factors as evidenced by nonsignificant relationships between high pain frequency and the CVD risk factors. Thus, the moderating effects of BMI on relationships between high pain frequency and CVD risk factors featured the increasing frequency of high pain increasingly eroding the protective effects of normal BMI on CVD risk. Perhaps the importance of chronic pain in affecting CVD risk factors lies in the identification of favorable profiles (e.g., normal BMI and no high pain), and in illustrating how chronic pain may increase CVD risk even among women with normal BMI.
It should be noted, however, that the moderating effects of BMI on the relationship between high pain frequency and fasting glucose revealed a different pattern of findings. Women with BMI values in the obese range revealed the characteristic “dose response” where those with increasingly frequent reports of high pain revealed increasingly higher glucose concentrations than those measured in obese BMI women who did not report high pain at any time across the four assessments. Thus, the moderating effects of BMI on relationships between high pain frequency and fasting glucose featured increasing frequency of high pain increasingly magnifying the deleterious effects of obese BMI on this CVD risk factor. It is not clear why effects for glucose differed from those of the other risk factors, and this issue awaits further research.
These results suggest that the effects of chronic pain on CVD risk cannot be explained by reference solely to the fact that people with chronic pain also tend to have relatively high BMI. Other explanatory factors must therefore be examined. If chronic pain is conceptualized more broadly as a chronic physiological stressor, one possibility is that chronic stress-related activations of the sympathoadrenomedullary and hypothalamo-pituary-adrenocortical axes may contribute to increased incidence of metabolic syndrome (Nieuwenhuizen & Rutters, 2008; Rosmond, 2005). Other studies provide evidence for chronic pain-related reductions in baroreflex sensitivity indicating altered autonomic function (Chung et al., 2008; Furlan et al., 2005; Reyes Del Paso, Garrido, Pulgar, Martín-Vázquez, Duschek, 2010; Spaziani, 2008), absence of natural analgesia typically associated with BP elevations (Bruehl, Chung, Diedrich, Diedrich, Robertson, 2008; Olsen et al., in press; Bruehl, Chung, & Chont, 2010; Bruehl, Chung, Ward, Johnson, & McCubbin, 2002), and possible alterations in neurotransmitter function including opioid (Bruehl & Chung, 2004; Bruehl, McCubbin, & Harden, 1999; Spaziani et al., 2008) and alpha-2 adrenergic (Chung et al., 2008) systems. These findings suggest that chronic pain is associated with changes in both the central and peripheral nervous systems that could provide alternative pathways for elevated CVD risk.
Some limitations of the present study should be delineated. First, the current results can be generalized only to women. It is not known whether findings would be similar in men. Limited evidence suggests that the impact of chronic pain on CVD risk markers may be greater in women, but this possibility cannot be evaluated with the SWAN data. Second, although the High Pain Frequency variable was derived from four consecutive annual assessments, we do not treat it as a longitudinal factor, but as a cross-sectional factor representing the accumulation of pain over three years. Thus, the design is cross-sectional, and we must be mindful of all accompanying limitations regarding causal inferences. Third, while the stability of the High Pain Frequency variable was no doubt enhanced by a three-year assessment window, we still relied on the single bodily pain item of the SF-36. State-of-the-art chronic pain assessment via well-validated self-report instruments or quantitative sensory testing was not available in this dataset. Fourth, the High Pain Frequency variable did not make distinctions within the four-assessment periods regarding whether certain pain groups may have unique relationships with CVD risk factors. For instance, it may be that groups described by distinct temporal patterns of pain intensity, such as “initially low but increasing” versus “initially high but decreasing” versus “initially high and stable” may be characterized by different risk factor profiles. Examining groups defined by these patterns in future research may reveal additional information regarding pain-related CVD risk. Finally, all zero-order, semi-partial, interaction and simple effect relationships involving the High Pain Frequency variable were small. Although the interactions with chronic pain and BMI appear new and compelling, enthusiasm must be tempered by the fact that the interaction effects themselves and the simple effects (e.g., the relationship between the High Pain Frequency variable and HDL among normal BMI women) accounted for little variance in CVD risk factors.
Although a causal link remains to be demonstrated, even a small influence of chronic pain on CVD risk could have a meaningful impact on public health given the estimated 100 million individuals in the United States affected by chronic pain (Institute of Medicine, 2011). Once chronic pain is established, it tends to persist, with chronic pain unresolved in 75-85% of patients at 4-12 year follow-up (Anderson, 2004; Elliott, Smith, Hannaford, Smith, & Chambers, 2002). To the extent that chronic pain contributes to CVD risk, the often unremitting nature of chronic pain highlights its potential importance as a public health issue and as a treatment target for reducing cardiovascular morbidity and mortality. Improved recognition of the role that chronic pain may play in CVD risk – albeit in concert with BMI -- has two important clinical implications. First, because this issue crosses traditional boundaries of medical specialization, it may not be specifically addressed in typical treatment protocols. Pain physicians understandably tend to focus on addressing patients’ pain and functional complaints. On the other hand, physicians in specialties most likely to treat CVD may be unlikely to appreciate the role that chronic pain could be playing in fostering CVD risk. Results linking chronic pain and CVD risk could potentially stir recognition by researchers and clinicians alike of the pressing need to better manage chronic pain not just to limit suffering but to perhaps aid in preventing the development of CVD. Second, the possibility that the linear effects of BMI on CVD risk may be altered by the presence of chronic pain – particularly among normal BMI women -- could provide evidence to justify a change in medical management. That is, the CVD risks faced by normal BMI women with chronic pain may be overlooked because of their normal BMI. Present results suggest, however, that women with this profile may be at enhanced CVD risk, and thus interventions to reduce chronic pain may prove useful not only to minimize interference with daily functioning, but could be used to prevent the development of CVD. In short, improved understanding of connections between chronic pain and CVD risk could facilitate more timely assessments, diagnoses, earlier interventions, and improve CVD treatment in millions of individuals exhibiting the normal BMI/high pain profile (Von Korff et al., 2005), ultimately leading to reduced cardiovascular-related morbidity and mortality.
ACKNOWLEDGMENTS
John W. Burns, Phillip J. Quartana, Stephen Bruehl, Imke Janssen, Sheila A. Dugan, Bradley Appelhans, Karen A. Matthews, and Howard M. Kravitz declare that they have no conflict of interest.
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2000 (5). Informed consent was obtained from all participants for being included in the study.
The Study of Women's Health Across the Nation (SWAN) has grant support from the National Institutes of Health (NIH), DHHS, through the National Institute on Aging (NIA), the National Institute of Nursing Research (NINR) and the NIH Office of Research on Women's Health (ORWH) (Grants U01NR004061; U01AG012505, U01AG012535, U01AG012531, U01AG012539, U01AG012546, U01AG012553, U01AG012554, U01AG012495). The content of this manuscript is solely the responsibility of the authors and does not necessarily represent the official views of the NIA, NINR, ORWH or the NIH.
Clinical Centers: University of Michigan, Ann Arbor – Siobán Harlow, PI 2011 – present, MaryFran Sowers, PI 1994-2011; Massachusetts General Hospital, Boston, MA – Joel Finkelstein, PI 1999 – present; Robert Neer, PI 1994 – 1999; Rush University, Rush University Medical Center, Chicago, IL – Howard Kravitz, PI 2009 – present; Lynda Powell, PI 1994 – 2009; University of California, Davis/Kaiser – Ellen Gold, PI; University of California, Los Angeles – Gail Greendale, PI; Albert Einstein College of Medicine, Bronx, NY – Carol Derby, PI 2011 – present, Rachel Wildman, PI 2010 – 2011; Nanette Santoro, PI 2004 – 2010; University of Medicine and Dentistry – New Jersey Medical School, Newark – Gerson Weiss, PI 1994 – 2004; and the University of Pittsburgh, Pittsburgh, PA – Karen Matthews, PI.
NIH Program Office: National Institute on Aging, Bethesda, MD – Winifred Rossi 2012 - present; Sherry Sherman 1994 – 2012; Marcia Ory 1994 – 2001; National Institute of Nursing Research, Bethesda, MD – Program Officers.
Central Laboratory: University of Michigan, Ann Arbor – Daniel McConnell (Central Ligand Assay Satellite Services).
Coordinating Center: University of Pittsburgh, Pittsburgh, PA – Maria Mori Brooks, PI 2012 - present; Kim Sutton-Tyrrell, PI 2001 – 2012; New England Research Institutes, Watertown, MA - Sonja McKinlay, PI 1995 – 2001.
Steering Committee: Susan Johnson, Current Chair
Chris Gallagher, Former Chair
We thank the study staff at each site and all the women who participated in SWAN.
Source of Funding
The data used in this study are derived from The Study of Women's Health Across the Nation (SWAN). Grant support for SWAN is from the National Institutes of Health (NIH), DHHS, through the National Institute on Aging (NIA), the National Institute of Nursing Research (NINR) and the NIH Office of Research on Women's Health (ORWH) (Grants U01NR004061; U01AG012505, U01AG012535, U01AG012531, U01AG012539, U01AG012546, U01AG012553, U01AG012554, U01AG012495). The content of this manuscript is solely the responsibility of the authors and does not necessarily represent the official views of the NIA, NINR, ORWH or the NIH. The opinions or assertions contained herein are the private views of the authors, and are not to be construed as official, or as reflecting true views, of the Department of the Army or the Department of Defense.
Footnotes
Conflicts of Interest
None declared.
References
- Aiken LS, West SG. Multiple regression: Testing and interpreting interactions. Sage; Thousand Oaks, CA: 1991. [Google Scholar]
- Andersson HI. The course of non-malignant chronic pain: a 12-year follow-up of a cohort from the general population. European Journal of Pain. 2004;8:47–53. doi: 10.1016/S1090-3801(03)00064-8. [DOI] [PubMed] [Google Scholar]
- Bingefors K, Isacson D. Epidemiology, co-morbidity, and impact on health-related quality of life of self-reported headache and musculoskeletal pain--a gender perspective. European Journal of Pain. 2004;8:435–450. doi: 10.1016/j.ejpain.2004.01.005. [DOI] [PubMed] [Google Scholar]
- Bogers RP, Bemelmans WJ, Hoogenveen RT, Boshuizen HC, Woodward M, Knekt P, van Dam RM, Hu FB, Visscher TL, Menotti A, Thorpe RJ, Jr, Jamrozik K, Calling S, Strand BH, Shipley MJ, the BMI-CHD Collaboration Investigators Association of overweight with increased risk of coronary heart disease partly independent of blood pressure and cholesterol levels: A meta-analysis of 21 cohort studies including more than 300 000 persons. Archives of Internal Medicine. 2007;167:1720–1728. doi: 10.1001/archinte.167.16.1720. [DOI] [PubMed] [Google Scholar]
- Bruehl S, Chung OY. Interactions between the cardiovascular and pain regulatory systems: an updated review of mechanisms and possible alterations in chronic pain. Neuroscience and Biobehavioral Review. 2004;28:395–414. doi: 10.1016/j.neubiorev.2004.06.004. [DOI] [PubMed] [Google Scholar]
- Bruehl S, Chung OY, Chont M. Chronic pain-related changes in endogenous opioid analgesia: a case report. Pain. 2010;148:167–171. doi: 10.1016/j.pain.2009.09.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bruehl S, Chung OY, Diedrich L, Diedrich A, Robertson D. The relationship between resting blood pressure and acute pain sensitivity: effects of chronic pain and alpha-2 adrenergic blockade. Journal of Behavioral Medicine. 2008;31:71–80. doi: 10.1007/s10865-007-9133-4. [DOI] [PubMed] [Google Scholar]
- Bruehl S, Chung OY, Jirjis JN, Biridepalli S. Prevalence of clinical hypertension in patients with chronic pain compared to nonpain general medical patients. Clinical Journal of Pain. 2005;21:147–153. doi: 10.1097/00002508-200503000-00006. [DOI] [PubMed] [Google Scholar]
- Bruehl S, Chung OY, Ward P, Johnson B, McCubbin JA. The relationship between resting blood pressure and acute pain sensitivity in healthy normotensives and chronic back pain sufferers: the effects of opioid blockade. Pain. 2002;100:191–201. doi: 10.1016/s0304-3959(02)00295-6. [DOI] [PubMed] [Google Scholar]
- Bruehl S, McCubbin JA, Harden RN. Theoretical review: altered pain regulatory systems in chronic pain. Neuroscience and Biobehavioral Review. 1999;23:877–890. doi: 10.1016/s0149-7634(99)00039-1. [DOI] [PubMed] [Google Scholar]
- Chung OY, Bruehl S, Diedrich L, Diedrich A, Chont M, Robertson D. Baroreflex sensitivity associated hypoalgesia in healthy states is altered by chronic pain. Pain. 2008;138:87–97. doi: 10.1016/j.pain.2007.11.011. [DOI] [PubMed] [Google Scholar]
- Clearfield MB. “C-reactive protein: a new risk assessment tool for cardiovascular disease”. Journal of the American Osteopathic Association. 2005;105:409–16. [PubMed] [Google Scholar]
- de Koning L, Merchant AT, Pogue J, Anand SS. Waist circumference and waist-to-hip ratio as predictors of cardiovascular events: Meta-regression analysis of prospective studies. European Heart Journal. 2007;28:850–856. doi: 10.1093/eurheartj/ehm026. [DOI] [PubMed] [Google Scholar]
- Duruoz MT, Turan Y, Gurgan A, Deveci H. Evaluation of metabolic syndrome in patients with chronic low back pain. Rheumatologyl International. 2013;15:215–220. doi: 10.1007/s00296-010-1693-x. [DOI] [PubMed] [Google Scholar]
- Elliott AM, Smith BH, Hannaford PC, Smith WC, Chambers WA. The course of chronic pain in the community: results of a 4-year follow-up study. Pain. 2002;99:299–307. doi: 10.1016/s0304-3959(02)00138-0. [DOI] [PubMed] [Google Scholar]
- Furlan R, Colombo S, Perego F, Atzeni F, Diana A, Barbic F, et al. Abnormalities of cardiovascular neural control and reduced orthostatic tolerance in patients with primary fibromyalgia. Journal of Rheumatology. 2005;32:1787–1793. [PubMed] [Google Scholar]
- Goodson NJ, Smith BH, Hocking LJ, McGilchrist MM, Dominiczak AF, Morris A, Porteous DJ, Goebel A. Cardiovascular risk factors associated with the metabolic syndrome are more prevalent in people reporting chronic pain: results from a cross-sectional general population study. Pain. 2013;154:1595–1602. doi: 10.1016/j.pain.2013.04.043. [DOI] [PubMed] [Google Scholar]
- Gureje O, Akinpelu AO, Uwakwe R, Udofia O, Wakil A. Comorbidity and impact of chronic spinal pain in Nigeria. Spine. 2007;32:E495–E500. doi: 10.1097/BRS.0b013e31810768fc. [DOI] [PubMed] [Google Scholar]
- Ha JY. Evaluation of metabolic syndrome in patients with chronic low back pain: Using the Fourth Korean National Health and Nutrition Examination survey data. Chonnam Medical Journal. 2011;47:160–164. doi: 10.4068/cmj.2011.47.3.160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hitt HC, McMillen RC, Thorton-Neaves T, Koch K, Cosby AG. Comorbidity of obesity and pain in a general population: results for the southern Pain Prevalence Study. Journal of Pain. 2007;8:430–436. doi: 10.1016/j.jpain.2006.12.003. [DOI] [PubMed] [Google Scholar]
- Institute of Medicine . Relieving pain in America: a blueprint for transforming prevention, care, education, and research. The National Academies Press; Washington, DC: 2011. [PubMed] [Google Scholar]
- Irwin MR, Wang M, Ribeiro D, Cho HJ, Olmstead R, Breen EC, et al. Sleep loss activates cellular inflammatory signaling. Biological Psychiatry. 2008;64:538–540. doi: 10.1016/j.biopsych.2008.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ishii S, Karlamangla AS, Bote M, Irwin MR, Jacobs DR, Cho HJ, Seeman TE. Gender, obesity and repeated elevation of C-Reactive Protein: Data from the CARDIA Cohort. PLoS One. 2012;7:e36062. doi: 10.1371/journal.pone.0036062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klein S, Burke LE, Bray GA, Blair S, Allison DB, Pi-Sunyer X, Hong Y, Eckel RH. Clinical implications of obesity with specific focus on cardiovascular disease: a statement for professionals from the American Heart Association Council on Nutrition, Physical Activity, and Metabolism: endorsed by the American College of Cardiology Foundation. American Heart Association Council on Nutrition, Physical Activity, and Metabolism. Circulation. 2004;110:2952–67. doi: 10.1161/01.CIR.0000145546.97738.1E. [DOI] [PubMed] [Google Scholar]
- Lavie CJ, Milani RV, Ventura HO. Obesity and cardiovascular disease: risk factor, paradox, and impact of weight loss. Journal of American College of Cardiology. 2009;53:1925–32. doi: 10.1016/j.jacc.2008.12.068. [DOI] [PubMed] [Google Scholar]
- Leveille SG, Zhang Y, McMullen W, Kelly-Hayes M, Felson DT. Sex differences in musculoskeletal pain in older adults. Pain. 2005;116:332–338. doi: 10.1016/j.pain.2005.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Livshits G, Zhai G, Hart D, Kato B, Wang H, Williams F, et al. Interleukin-6 is a significant predictor of radiographic knee osteoarthritis: the Chingford Study. Arthritis & Rheumatism. 2009;60:2037–2045. doi: 10.1002/art.24598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McBeth J, Symmons DP, Silman AJ, Allison T, Webb R, Brammah T, et al. Musculoskeletal pain is associated with a long-term increased risk of cancer and cardiovascular- related mortality. Rheumatology. 2009;48:74–77. doi: 10.1093/rheumatology/ken424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nieuwenhuizen AG, Rutters F. The hypothalamic-pituitary-adrenal-axis in the regulation of energy balance. Physiology and Behavior. 2008;94:169–177. doi: 10.1016/j.physbeh.2007.12.011. [DOI] [PubMed] [Google Scholar]
- O'Connor MF, Motivala SJ, Valladares EM, Olmstead R, Irwin MR. Sex differences in monocyte expression of IL-6: role of autonomic mechanisms. American Journal of Physiological Regulation and Integrated Comprehensive Physiology. 2007;293:R145–51. doi: 10.1152/ajpregu.00752.2006. [DOI] [PubMed] [Google Scholar]
- Olsen RB, Bruehl S, Nielsen CS, Rosseland LA, Eggen AE, Stubhaug A. Hypertension prevalence and diminished blood pressure-related hypoalgesia in individuals reporting chronic pain in a general population. The Tromsø study. Pain. doi: 10.1016/j.pain.2012.10.020. in press. [DOI] [PubMed] [Google Scholar]
- Poirier P, Giles TD, Bray GA, et al. Obesity and cardiovascular disease: pathophysiology, evaluation, and effect of weight loss: an update of the 1997 American Heart Association scientific statement on obesity and heart disease from the obesity committee of the council on nutrition, physical activity, and metabolism. Circulation. 2006;113:898–918. doi: 10.1161/CIRCULATIONAHA.106.171016. [DOI] [PubMed] [Google Scholar]
- Ray L, Lipton RB, Zimmerman ME, Katz MJ, Derby CA. Mechanisms of association between obesity and chronic pain in the elderly Pain. 2011;152:53–59. doi: 10.1016/j.pain.2010.08.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reyes Del Paso GA, Garrido S, Pulgar A, Martín-Vázquez M, Duschek S. Aberrances in autonomic cardiovascular regulation in fibromyalgia syndrome and their relevance for clinical pain reports. Psychosomatic Medicine. 2010;72:462–470. doi: 10.1097/PSY.0b013e3181da91f1. [DOI] [PubMed] [Google Scholar]
- Rosmond R. Role of stress in the pathogenesis of the metabolic syndrome. Psychoneuroendocrinology. 2005;30:1–10. doi: 10.1016/j.psyneuen.2004.05.007. [DOI] [PubMed] [Google Scholar]
- Santos S, Oliveira A, Lopes C. Systematic review of saturated fatty acids on inflammation and circulating levels of adipokines. Nutrition Research. 2013;33:687–95. doi: 10.1016/j.nutres.2013.07.002. [DOI] [PubMed] [Google Scholar]
- Schneider S, Mohnen SM, Schiltenwolf M, Rau C. Comorbidity of low back pain: representative outcomes of a national health study in the Federal Republic of Germany. European Journal of Pain. 2007;11:387–397. doi: 10.1016/j.ejpain.2006.05.005. [DOI] [PubMed] [Google Scholar]
- Sowers MC, Sternfeld B, Morganstein D, et al. SWAN: a multicenter, multiethnic, community-based cohort study of women and the menopausal transition. In: Lobo RMR, Kelsey J, editors. Menopause: Biology and Pathobiology. Academic Press; San Diego, CA: 2000. pp. 175–188.. [Google Scholar]
- Spaziani R, Bayati A, Redmond K, Bajaj H, Bienenstock J, Collins SM, Kamath MV. Vagal dysfunction in irritable bowel syndrome assessed by rectal distension and baroreceptor sensitivity. Neurogastroenterology & Motility. 2008;20:336–342. doi: 10.1111/j.1365-2982.2007.01042.x. [DOI] [PubMed] [Google Scholar]
- Stec JJ, Silbershatz H, Tofler GH, Matheney TH, Sutherland P, Lipinska I, Massaro JM, Wilson PF, Muller JE, D'Agostino RB., Sr Association of fibrinogen with cardiovascular risk factors and cardiovascular disease in the Framingham Offspring Population. Circulation. 2000;102:1634–8. doi: 10.1161/01.cir.102.14.1634. [DOI] [PubMed] [Google Scholar]
- Steiner P, Freidel J, Bremner W, Stein E. Standardization of micromethods for plasma cholesterol, triglyceride and HDL-cholesterol with the lipid clinics’ methodology. Journal of Clinical Chemistry and Clinical Biochemistry. 1981;19:850. [Google Scholar]
- Stone AA, Broderick JE. Obesity and pain are associated in the United States. Obesity. 2012;20:1491–1495. doi: 10.1038/oby.2011.397. [DOI] [PubMed] [Google Scholar]
- Thommasen HV, Zhang W. Impact of chronic disease on quality of life in the Bella Coola Valley. Rural Remote Health. 2006;6:528. [PubMed] [Google Scholar]
- Urquhart DM, Berry P, Wluka AE, et al. Young Investigator Award winner: Increased fat mass is associated with high levels of low back pain intensity and disability. Spine. 2011;36:1320–1325. doi: 10.1097/BRS.0b013e3181f9fb66. [DOI] [PubMed] [Google Scholar]
- Van den Bussche H, Koller D, Kolonko T, Hansen H, Wegscheider K, Glaeske G, von Leitner EC, Schafer I, Schon G. Which chronic diseases and disease combinations are specific to multimorbidity in the elderly? Results of a claims data based cross-sectional study in Germany. BMC Public Health. 2011;11:101. doi: 10.1186/1471-2458-11-101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Von Korff M, Crane P, Lane M, Miglioretti DL, Simon G, Saunders K, Stang P, Brandenburg N, Kessler R. Chronic spinal pain and physical-mental comorbidity in the United States: results from the national comorbidity survey replication. Pain. 2005;113:331–339. doi: 10.1016/j.pain.2004.11.010. [DOI] [PubMed] [Google Scholar]
- Wang H, Ahrens C, Rief W, Gantz S, Schiltenwolf M, Richter W. Influence of depression symptoms on serum tumor necrosis factor-ά of patients with chronic low back pain. Arthritis Research and Therapy. 2010;12:186–193. doi: 10.1186/ar3156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ware JE, Kosinski M, Keller SD. SF-36 Physical and Mental Health Summary Scales: A Users' Manual. The Health Institute; Boston: 1994. [Google Scholar]
- Warnick GR, Albers JJ. A comprehensive evaluation of the heparin-manganese precipitation procedure for estimating high density lipoprotein cholesterol. Journal of Lipid Research. 1978;19:65–76. [PubMed] [Google Scholar]
- Wright LJ, Schur E, Noonan C, et al. Chronic pain, overweight, and obesity: findings from a community-based twin registry. Journal of Pain. 2010;11:628–635. doi: 10.1016/j.jpain.2009.10.004. [DOI] [PMC free article] [PubMed] [Google Scholar]