Abstract
Metformin is an oral biguanide used for type II diabetes. Epidemiologic studies suggest a link between metformin use and reduced risk of breast and other types of cancers. ErbB2-expressing breast cancer is a subgroup of tumors with poor prognosis. Previous studies demonstrated that metformin is a potent inhibitor of ErbB2-overexpressing breast cancer cells; metformin treatment extends the life span and impedes mammary tumor development in ErbB2 transgenic mice in vivo. However, the mechanisms of metformin associated antitumor activity, especially in prevention models, remain unclear. We report here for the first time that systemic administration of metformin selectively inhibits CD61high /CD49fhigh subpopulation, a group of tumor-initiating cells (TIC) of mouse mammary tumor virus (MMTV)-ErbB2 mammary tumors, in preneoplastic mammary glands. Metformin also inhibited CD61high /CD49fhigh subpopulation in MMTV-ErbB2 tumor-derived cells, which was correlated with their compromised tumor initiation/development in a syngeneic tumor graft model. Molecular analysis indicated that metformin induced downregulation of ErbB2 and EGFR expression and inhibited the phosphorylation of ErbB family members, insulin-like growth factor-1R, AKT, mTOR, and STAT3 in vivo. In vitro data indicate that low doses of metformin inhibited the self-renewal/proliferation of cancer stem cells (CSC)/TICs in ErbB2-over-expressing breast cancer cells. We further demonstrated that the expression and activation of ErbB2 were preferentially increased in CSC/TIC-enriched tumorsphere cells, which promoted their self-renewal/ proliferation and rendered them more sensitive to metformin. Our results, especially the in vivo data, provide fundamental support for developing metformin-mediated preventive strategies targeting ErbB2-associated carcinogenesis.
Introduction
Breast cancer is the leading cause of cancer-related deaths among women, with as many as 40% of cases ending in relapse and metastatic disease (1). Growing evidence suggests that cancer stem cells (CSC) play a critical role in breast cancer initiation, metastasis, and therapeutic resistance. According to the CSC theory, cancers are driven by a rare group of tumor cells with stem cell properties, including self-renewal and multilineage differentiation capacity (2). Al-Hajj and colleagues first reported that ESA+CD44+CD24−/low Lin− human breast cancer cells were significantly enriched for tumor-forming ability in nonobese diabetic/severe combined immunodeficient mice compared with Lin− cells with other phenotypes. Self-renew and differentiation potential of the CD44+CD24−/low Lin−cells was demonstrated by serial in vivo passages and the heterogeneity of the derived tumors (3,4). The stem cell-like properties of these cancer cells were similar to the bipotent human mammary epithelial progenitors (5–7). Later, Ginestier and colleagues demonstrated that breast cancer cells with high ALDH1 activity, which contain a small fraction of cells overlapping with CD44+CD24−/low Lin− cells, were also capable of self-renewal and generating tumors that recapitulate the heterogeneity of the parental tumor (8). Recently, Lo and colleagues identified CD61high/CD49fhigh subpopulation as tumor-initiating cells (TIC) in mammary tumors developed in mouse mammary tumor virus (MMTV)-ErbB2 transgenic mice (9). These studies not only provide solid evidence supporting “CSC theory” but also establish breast CSC markers for studies aiming at clinical implications.
ErbB2, also known as HER2/neu, is a 185 kDa transmembrane glycoprotein that belongs to the epidermal growth factor receptor (EGFR) family. It is amplified/overexpressed in 20% to 30% of breast cancers, which has been correlated with aggressive phenotypes and poor prognosis (10). ErbB2 is a receptor tyrosine kinase (RTK) with intrinsic tyrosine kinase activity. As the only EGFR family member that has no known ligand, ErbB2 can be activated by homodimerization and/or heterodimerization with the other ErbB members upon cognate ligand binding (11). It has been well established that dysregulation of the ErbB2 pathway disrupts homeostasis of normal cell-control mechanisms and gives rise to aggressive tumor cells (12–14). In particular, recent evidence indicates that overexpression of ErbB2 induces the expansion of stem/progenitor subpopulation of breast cancer cells, which promote metastasis and drug resistance (15). In vivo data also showed that luminal progenitor cell populations in the preneoplastic mammary glands of MMTV-ErbB2 transgenic mice were significantly expanded (9). Therefore, ErbB2 signaling may drive carcinogenesis through regulation of the mammary stem/progenitor cell populations.
Metformin is the most commonly used therapy in patients with type II diabetes (16). Epidemiologic studies suggest that metformin may lower cancer risk in diabetics and improve outcomes of various types of cancers (17). In particular, metformin treatment was associated with lower breast cancer incidence among patients with diabetes, and higher pathologic complete response in patients with earlystage breast cancer who were receiving neoadjuvant therapy (18). Previous cell line- and xenograft tumor-based experiments have shown that metformin selectively kills CSCs in different types of breast tumors (19). It regulates breast CSC ontogeny by transcriptional regulation of the epithelial-mesenchymal transition (EMT) machinery (20) and targets Stat3 to inhibit cell growth and induce apoptosis in basal-like breast cancer cells (21). Metformin was also reported to overcome trastuzumab resistance by specifically killing breast cancer initiating CD44+CD24−/low cells (22) and by inhibiting ErbB2/insulin-like growth factor (IGF)-1R receptor interactions (23). In contrast to extensive studies on breast cancer cell lines, little data are available on the metformin-mediated cancer-preventive effect on nondiabetic models. In this regard, reports by Anisimov and colleagues showed that systemic treatment with metformin extended life span and delayed spontaneous breast tumor development in MMTV-ErbB2 transgenic mice (24, 25). Mechanistic insight into metformin-mediated inhibition of mammary tumor development in this clinically relevant model will facilitate the development of metformin-mediated breast cancer prevention.
In this study, we investigated the mechanisms of metformin-associated prevention/inhibition of ErbB2-mediated breast cancer development by focusing on the potential effect of metformin on CSCs/TICs in the MMTV-ErbB2 transgenic mouse model and its context with existing in vitro models. We show here for the first time that systemic administration of metformin selectively inhibits CSC/TIC subpopulations in the preneoplastic mammary glands of MMTV-ErbB2 transgenic mice, which was associated with metformin-induced downregulation of ErbB2/phosphoi-nositide 3-kinase (PI3K)/Akt pathway and the inhibition of IGF-1R, mTOR, and Stat3 activation. These data will have significant impact on the implication of metformin in breast cancer prevention.
Materials and Methods
Animals and treatment
The FVB/N-Tg MMTV-ErbB2 transgenic mice used in this study were obtained from The Jackson Laboratory. They were housed in a temperature-controlled room with a 12-hour light-dark cycle, and fed an estrogen-free AIN-93G diet (Harlan Teklad). With 10 mice in each of the control and treatment groups, 8-week-old virgin mice were intraperitoneally (i.p.) injected daily with 250 mg/kg of metformin (Sigma) or vehicle (saline) as control for 10 weeks. The dosage used was based on previous studies (26), as well as human therapeutic doses (27). Mammary glands were collected 18 hours after last injection for subsequent experiments. For tissue collection, the fourth pairs of mammary gland from 4 mice of each group were harvested for isolation of primary mammary epithelial cells, which were used for flow cytometry analysis and mammosphere assay. Protein lysate was also prepared from the treated mammary glands for Western blot analysis. The 4th inguinal mammary glands from 3 mice in each group were processed for whole mount staining. All procedures involving mice were performed with the approval of university’s Institutional Animal Care and Use Committee and conducted in accordance with the NIH Guide for the Care and Use of Laboratory Animals.
Cell culture and treatments
Human breast cancer cell lines BT-474 and SKBR3 were purchased from American Type Culture Collection. The 78617 cell line was a mammary tumor cell line derived from MMTV-ErbB2 transgenic mice as previously described (28). All the cells were maintained in Dulbecco’s Modified Eagle Medium: Nutrient Mix F12 (DMEM/F12 1:1; Life Technologies) supplemented with 10% FBS, and cultured in a 37°C humidified atmosphere containing 5% CO2.
For the cell-proliferation assays, 3 × 103 cells were seeded into 96-well plates and cultured in DMEM/F12 containing 10% FBS and graded metformin for 6 days. The medium was changed once throughout the process. Cells cultured for other assays were treated as indicated in individual experiments.
Mammary gland whole mount analysis
The inguinal mammary glands of MMTV-ErbB2 transgenic mice were dissected and spread onto slides followed by overnight fixation in Carnoy’s solution at room temperature (29). The glands were rehydrated and stained overnight with carmine alum. The stained glands were then dehydrated, cleared with xylene, and mounted. Photographs were taken under a digital camera mounted on a Nikon C-LEDS microscope.
Primary mouse mammary epithelial cells (MECs) isolation and flow cytometric analysis
The inguinal mammary glands were harvested from 18-week-old mice. The glands were finely minced with a tissue chopper (Mickle Laboratory Engineering) and then digested with collagenase (Roche) and hyaluronidase (Sigma) for 2 hours at 37°C. The resultant organoids were further digested with 0.25% trypsin-EDTA (Sigma) and dispase (Stem Cell Technologies)/DNase I (Sigma), followed by filtration through a 40-mm mesh. The cells were stained with antibodies against CD49f, CD61, and the lineage markers, followed by flow cytometric analysis, as described by Shelton and colleagues (30).
Mammosphere and tumorsphere assays
Primary mouse MECs were seeded at 10 cells per well in triplicate in ultra-low attachment 6-well plates (Corning). BT-474, SKBR-3 and 78617 cells were seeded in triplicate in ultra-low attachment 6-well plates at a density of 500 cells/cm2. In the presence or absence of metformin, spheres were allowed to grow in DMEM/F12 media supplemented with 5 mg/mL insulin (Sigma), 0.5 µg/mL hydrocortisone (Sigma), 1 × B27 (Life Technologies), 20 ng/mL EGF (Sigma), 20 ng/mL hFGF (Stem Cell Technologies), and 4 µg/mL heparin (Stem Cell Technologies) for 6 days, followed by sphere counting and image documentation. For secondary sphere cultures, primary spheres were harvested, incubated with trypsin for 5 minutes at 37°C, and vigorously pipetted. Single cells were confirmed under a microscope and resuspended with sphere culture conditions for another 6 days. The sphere-forming efficiency (%) was calculated as (number of spheres per well/number of cells seeded per well) × 100.
Western blotting
Western blotting was performed as previously described (31). Antibodies against pErbB2 (Y877), pEGFR (S1406/ 1407), pErbB3 (Y1289), pAKT (S473), pERK1/2 (T202/ Y204), pAMPKα (T172), AMPKα, mTOR, and pMTOR (S2448) were purchased from Cell Signaling. Antibodies against EGFR, ErbB3, estrogen receptor (ER)-α, pER-α (S167), Stat3, pStat3 (Y705), Actin, and ERK2 were purchased from Santa Cruz (Santa Cruz Biotechnology). Antibodies against ErbB2 and AKT1 were purchased from Millipore and EPITMICS, respectively.
Syngeneic grafting of metformin-treated tumor cells
For the syngeneic grafting experiment, 78617 cells were pretreated with 1 mmol/L metformin in DMEM/F12/10% FBS or control medium for 72 hours. The cells were then harvested and examined for viability with trypan blue. Before grafting, cell number for each sample was adjusted based on viability. One million (1 × 106) viable cells were subcuta-neously injected into each flank of female MMTV-ErbB2 transgenic mice (n = 5 mice for each of the control and metformin groups). Tumor volumes were measured every other day after the initial injection for 14 days. The volume was calculated as long diameter × (short diameter)2 × 0.52.
Sulforhodamine B assay
For the proliferation assays, treated SKBR-3, BT-474, 78617, and MCF10A cells were fixed with 10% trichloroacetic acid at 4°C for 1 hour. The plates were then washed 4 times with tap water, followed by staining with 50 µL 0.4% sulforhodamine B (SRB; Sigma; in acetic acid: H2O = 1:100) for 30 minutes. Excess stain was washed 4 times with 1% acetic acid. The stained cells were dissolved in 10 mmol/L Tris. The optical density at 490 nm was determined using a Synergy Mx microplate reader (BioTek).
Clonogenic assay
As in the syngeneic grafting experiments, 78617 cells were pretreated with 1 mmol/L metformin in DMEM/F12/10% FBS or control medium for 72 hours. The harvested cells were adjusted based on viability. One thousand viable single cells were seeded in each well of 6-well plates and allowed to grow in complete DMEM/F12 media supplemented with 10% FBS for 2 weeks. Colonies were then washed twice with PBS and stained with 0.5% crystal violet (in methanol: H2O =1:1) for 30 min at room temperature. The extra stain was aspirated, and the plates were washed with tap water and air dried. Colonies were counted and photographed with a digital camera mounted on a Nikon C-LEDS microscope.
ALDH1 activity assay
ALDH1 activity was detected by flow cytometric analysis with an ALDEFLUOR Kit (Stem Cell Technologies) on Cell Lab Quanta SC (Bechman Coulter) according to the manufacturer’s instructions. Briefly, control and metformin-treated cells were incubated with ALDEFLUOR substrate. The specific inhibitor of ALDH, DEAB, was used to establish the baseline fluorescence and define the ALDEFLUOR-positive region. ALDEFLUOR-positive subpopulation data in triplicate were statistically analyzed.
RNA extraction and quantitative real-time PCR
Total RNA was isolated from monolayer attached and tumorsphere cells using a RNeasy Mini Kit (Qiagen). First-strand cDNA synthesis was performed using an iScript cDNA Synthesis Kit (Bio-Rad). Quantitative real-time PCR was performed on Bio-Rad CFX96 Touch Real-Time PCR Detection System using SsoFast EvaGreen Supermix (Bio-Rad). The primer sequences are listed in Supplementary Table S1.
Statistical analysis
A Student t test for comparison of 2 groups was used for statistical analysis. Calculations were performed using software from GraphPad Prism (GraphPad) and data were expressed as means ± SEM of at least 3 independent experiments. AP value of ≤0.05 was considered statistically significant.
Results
Metformin selectively inhibits CD61high/CD49fhigh subpopulation in preneoplastic mammary glands of MMTV-ErbB2 transgenic mice
A previous report showed that metformin treatment extended life span and delayed spontaneous breast tumor development in MMTV-ErbB2 transgenic mice (24), but the underlying mechanisms were not addressed. To study the in vivo effect of metformin on mammary ductal growth and tissue hierarchy in MMTV-ErbB2 transgenic mice at the premalignant stage, 8-week-old virgin mice were treated with saline (control) or metformin at a dose of 250 mg/kg/day for 10 weeks. We first examined the effect of metformin on mammary morphogenesis. Whole mount analysis showed that metformin treatment resulted in significantly decreased lateral branching and alveolar structures as compared to the control (Fig. 1A).
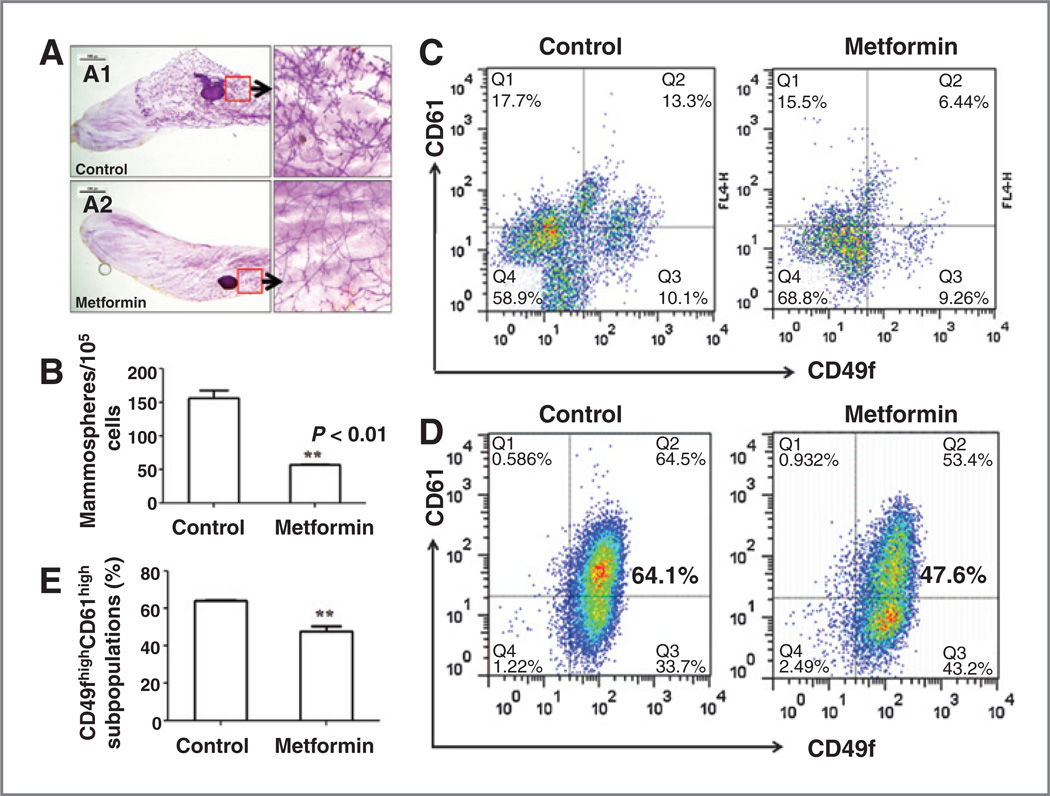
Figure 1.
Metformin inhibits MEC mammosphere formation and CD61high/CD49fhigh subpopulation. A, representative mammary whole mounts of control (A1) and metformin-treated (A2) mice. MMTV-ErbB2 mice were treated with saline (control) or metformin (250 mg/kg/day via i.p.) starting from 8 weeks of age. Mammary glands were harvested and processed at 18 weeks of age. Whole mounts from 3 mice of each group were examined. B, mammosphere-forming efficiency of MECs from metformin-treated mice was reduced. Primary MECs were isolated from 18-week-old mice with above treatments, followed by mammosphere-formation assay. C, metformin selectively inhibits CD61high/CD49fhigh subpopulation in mammary tissues of MMTV-ErbB2 transgenic mice. MECs were isolated from 18-week-old mammary tissues with above treatments. CD49f and CD61 expression profile were analyzed by FACS. D, metformin inhibits CD61high/CD49fhigh subpopulation in 78617 cells. These MMTV-ErbB2 tumor-derived cells were cultured in the absence/presence of 1 mmol/L of metformin for 72 hours, followed by FACS analysis of CD49f and CD61 expression. E, quantitative analysis of CD61high/CD49fhigh subpopulation in 78617 cells as in D.
Mammosphere and tumorsphere assays have been developed and widely used for evaluation of mammary epithelial stem/progenitor cells (32, 33) and mammary CSCs/TICs (34–36), respectively. We performed quantitative sphere assay to examine the frequency of mammary epithelial stem/progenitor cells in mammary tissues with different treatments. As shown in Fig. 1B, the sphere-forming efficiency of the primary MECs from metformin-treated group was significantly decreased as compared with the control group, suggesting that metformin treatment inhibits mammary stem/progenitor cell subpopulation.
CD61high/CD49fhigh Lin− mammary cells have been recently identified as TICs of mammary tumors in MMTV-ErbB2 transgenic mice (9). Mammary tissues of MMTV- ErbB2 transgenic mice at 18 weeks, the endpoint of metformin treatment, are in the preneoplastic stage (37). To determine whether metformin modulates TIC-like MECs in the preneoplastic mammary glands, we analyzed CD61high/ CD49fhigh profiles in primary MECs isolated from each group. As shown in Fig. 1C, CD61high/CD49fhigh cells in metformin-treated samples were significantly decreased. In contrast to little or no decrease of the cells in the other quadrants, the data suggest that metformin may selectively target CD61high/CD49fhigh TICs.
It has been shown that cell lines derived from MMTV-ErbB2 tumors are enriched with CD61high/CD49fhigh TICs (9). To further demonstrate that metformin specifically targets CD61high/CD49fhigh TICs, we examined the effect of metformin on CD61high/CD49fhigh subpopulation in 78617 cells, an MMTV-ErbB2 tumor cell line established in our laboratory. We found that a 72-hour treatment with 1 mmol/L metformin significantly decreased TICs-enriched CD61high/ D49fhigh subpopulations in 78617 cells (Fig. 1D and E).
Molecular signaling in metformin-treated mammary tissues
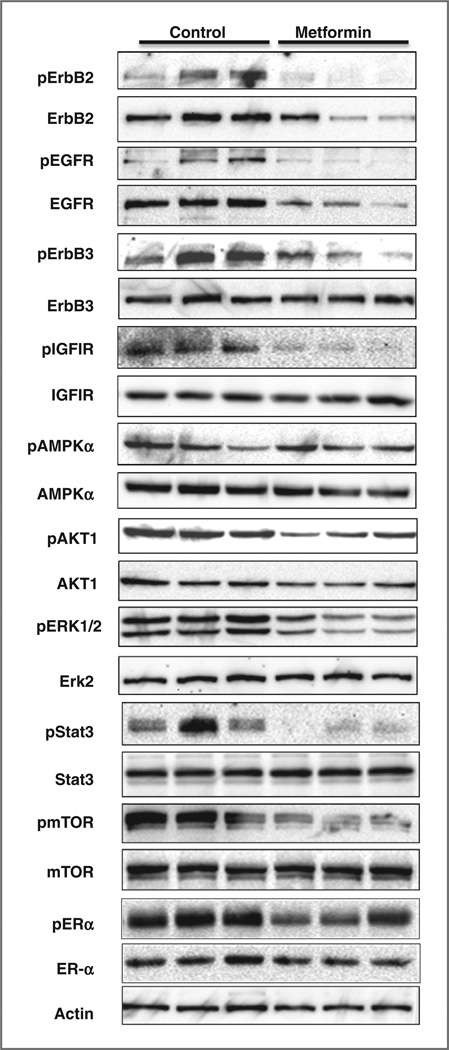
To understand the underlying mechanism of metformin-mediated inhibitionofstem/progenitor cells and mammary ductal growth, we examined the expression and activation of key markers involved in ErbB2 signaling and metformin actions (Fig. 2). We found that metformin treatment resulted in significant downregulation of ErbB2 and EGFR expression, the inhibition of ErbB2, EGFR, ErbB3 and IGF-1R phosphorylation/activation. Consistently, activation of AKT1 and ERK1/2; were also inhibited. Metformin-induced activation of AMPK, however, was not as significant as in in vitro studies, possibly because of the timing of tissue collection. Nevertheless, phosphorylation of Stat3 and mTOR was remarkably inhibited in metformintreated tissues. Moreover, we found that metformin-induced modest inhibition of estrogen receptor (ER)-a phosphorylation, but not expression, which might be associated with Akt and Erk inactivation. These findings suggested that metformin inhibits mammary epithelial growth through the inhibition of RTK and downstream AKT/ mTOR, MAPK/Erk, and Stat3 signaling, suggesting the critical role of these regulators in metformin-mediated suppression of TIC subpopulation.
Figure 2.
Metformin-induced in vivo inhibition of RTK signaling in mammary tissues of MMTV-ErbB2 transgenic mice. Protein lysates were prepared from mammary tissues of 18-week-old MMTV-ErbB2 mice treated with saline or metformin as specified in Fig. 1. Protein levels of indicated markers were detected by Western blot analysis.
Metformin pretreatment inhibits ErbB2–overexpressing tumor development in a syngeneic graft mouse model
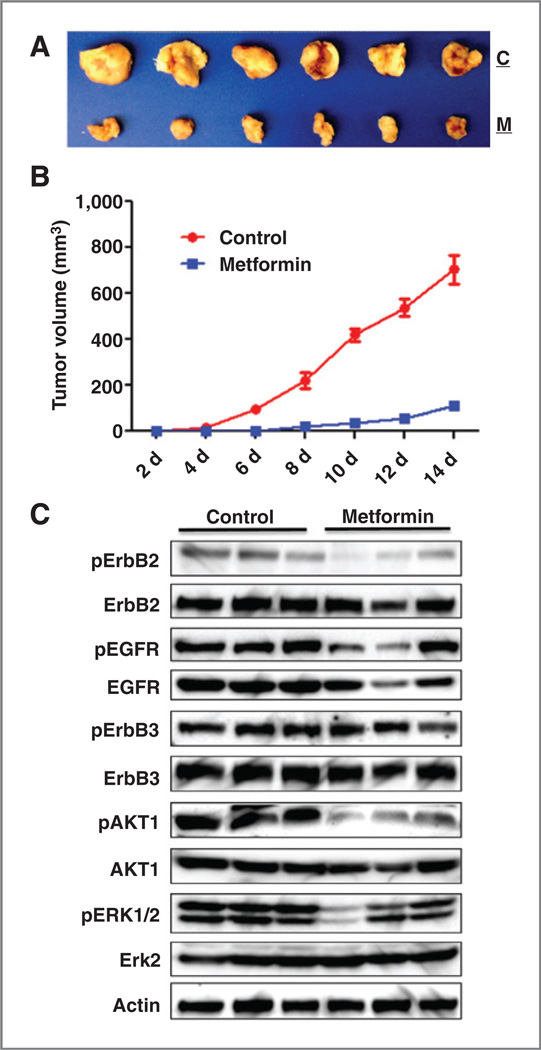
To gain more in vivo evidence that metformin suppresses the initiation and development of ErbB2–overexpressing breast cancer, we assessed in vivo tumor initiation/development of 78617 cells with metformin pretreatment before implantation. To this end, the cells were pretreated with 1 mmol/L metformin for 72 hours. One million of adjusted viable cells of each sample were subcutaneously injected into MMTV-ErbB2 mice. As shown in Fig. 3A and B, tumor development and growth from the cells with metformin pretreatment were remarkably inhibited. Western blot assays indicate that metformin pretreatment induced prolonged inhibition of the phosphorylation of ErbB2 and EGFR, as well as the signaling of downstream AKT and ERK1/2 signaling (Fig. 3C). In context with the decrease of CD61high/CD49fhigh cells in Fig. 1D and E, these results suggest that metformin-mediated inhibition of TICs/CSCs and ErbB2 signaling may contribute to slow development and growth of the syngeneic tumors and spontaneous tumors as well.
Figure 3.
Pretreatment with metformin in vitro inhibits in vivo development of ErbB2-overexpressing syngeneic tumor grafts. MMTV-ErbB2 tumor-derived 78617 cells were treated with control medium or 1 mmol/L metformin for 72 hours and then trypsinized. After adjusting cell number based on viability, 1 × 106 viable cells were injected subcutaneously to each flank of a MMTV-ErbB2 mouse. Tumor volumes were measured every other day for 14 days. Protein lysates were prepared from harvested tumors for Western blot analysis. A, images of grafted tumors with control medium (C) or metformin (M) treatment before grafting. B, tumor growth curves of control and metformin pretreatment groups. C, Western blot detection of RTK markers’ expression and activation in tissues harvested from syngeneic graft tumors 14 days after grafting.
Metformin inhibits the proliferation and RTK signaling of ErbB2–overexpressing breast cancer cells in vitro
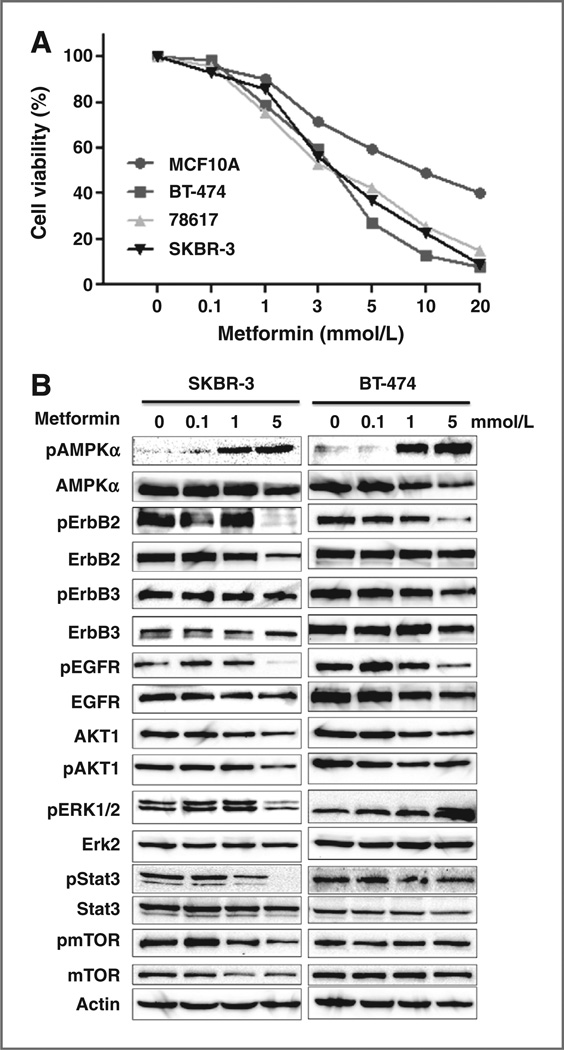
We next examined the effect of metformin on the proliferation of a series of cell lines with different ErbB2 expression profiles, including 3 ErbB2–overexpressing breast cancer cell lines (SKBR-3, BT-474, and 78617) and a line of nontransformed human MECs (MCF10A). SRB assay results demonstrated that metformin inhibited the growth of all 3 ErbB2–overexpressing breast cancer cell lines in a dose-dependent manner but its effect on the MCF10A cells was moderate (Fig. 4A). Examination of molecular signaling in metformin-treated SKBR-3 and BT-474 cells indicated that, under the given conditions, the most sensitive marker was AMPK phosphorylation/ activation, which could be detected in samples treated with 0.1 mmol/L metformin (Fig. 4B). Consistent with the in vivo data, phosphorylation of ErbB2, EGFR, and AKT1 was inhibited in both cell lines. The inhibition of Stat3, ERK1/2, and mTOR phosphorylation, however, was evident in SK-BR-3 cells but not in BT-474 cells. These data generally support the role of AMPK, ErbB2, EGFR, Akt, Stat3, and mTOR regulation in metformin-mediated inhibition of the 2 cell lines, although changes of certain makers were subtle in BT-474 cells. In context with above proliferation data, we considered 1 mmol/L as the upper threshold concentration being nontoxic to monolayer-cultured ErbB2–overexpressing breast cancer cells, which were used in the following experiments.
Figure 4.
Metformin inhibits growth and RTK signaling of ErbB2– overexpressing breast cancer cells in vitro. A, effect of metformin on the proliferation of the selected cell lines. MCF-10A, BT-474, 78617, and SK-BR-3 cells were treated with metformin at indicated concentrations for 6 days, followed by SRB assay. B, Western blot analysis of RTK expression, activation, and signaling in ErbB2–overexpressing breast cancer cells.
Metformin at low concentrations inhibits the self-renewal/proliferation of CSCs/TICs in ErbB2–overexpressing breast cancer cells in vitro
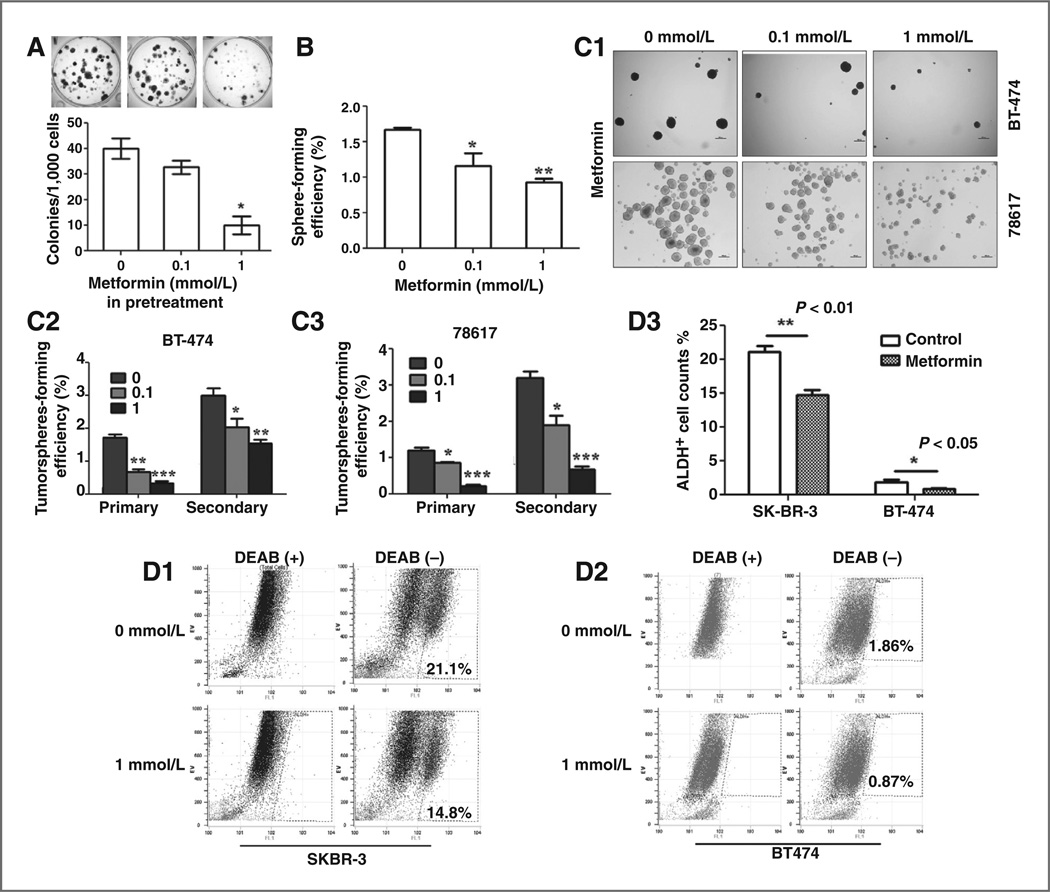
To support our in vivo findings, we performed a series of in vitro experiments to study the effect of metformin on the stemness and proliferation of established ErbB2–over-expressing breast cancer cell lines. In parallel to the in vivo experiments described in Fig. 3, we first examined the effect of metformin pretreatment on colony formation. As shown in Fig. 5A, pretreatment of 78617 cells with low-dose metformin significantly inhibited their colony formation efficiency.
Figure 5.
Metformin at low concentrations inhibits the stemness of ErbB2-overexpressing breast cancer cell lines in vitro. A, colony formation efficiency of control and metformin pretreated 78617 cells. The cells were pretreated with control medium or 1 mmol/L metformin for 72 hours, followed by trypsinization and viability assessment. One thousand viable cells were seeded in each well of 6-well plates and allowed them to grow for 2 weeks, followed by crystal violate staining. Colonies of each group in triplicates were statistically analyzed. B, low-dose metformin inhibits mammosphere formation from primary MECs in vitro. Primary MECs isolated from MMTV-ErbB2 mice were treated with metformin at indicated concentrations and assessed for mammosphere formation as specified in the methods. C, low-dose metformin inhibits tumorsphereformation in BT-474 and 78617 cells. C1, representative images of tumorsphere formation from each cell line. C2, C3, primary and secondary tumorsphere-forming efficiency of metformin-treated BT-474 (C2) and 78617 (C3) cells. D, low-dose metformin inhibits ALDEFLUOR-positive subpopulation in SKBR-3 and BT-474 breast cancer cells. Cells from each line were treated with 1 mmol/L metformin for 72 hours, followed by FACS analysis. D1, D2, representative FACS analysis of SKBR-3 (D1) and BT-474 (D2) cells using the ALDEFLUOR assay. D3, histogram of metformin induced changes of ALDEFLUOR-positive subpopulations in SKBR-3 and BT-474 cells. *P < 0.05; **P < 0.01.
To determine whether metformin impedes the self-renewal/proliferation of stem/progenitor cells at low concentrations, primary MECs isolated from ErbB2 transgenic mice were grown under sphere-culturing conditions in the presence of graded low doses of metformin for 6 days. We found that low dose of metformin reduced the sphere-forming efficiency of primary MECs (Fig. 5B). The results also showed that both primary and secondary tumor-sphere formation from BT-474 and 78617 cell lines were significantly inhibited by metformin in a similar manner, suggesting a potent inhibitory effect of metformin on tumorsphere-forming efficiency at low concentrations (Fig. 5C).
ALDH1 activation is an established marker of breast CSCs (8). To gain more evidence that metformin is able to act on TICs/CSCs at a low concentration, BT-474 and SKBR-3 cells were treated with 1 mmol/L metformin for 48 hours, followed by flow cytometry analysis of ALDH1 activity using an ALDEFLUOR Kit. As shown in Fig. 5D3, ALDH1-positive subpopulation was significantly decreased in both cell lines treated with metformin. Hence, results from multiple evaluation approaches demonstrated that metformin exerts potent inhibitory effect on the self-renewal/proliferation of CSCs/TICs at concentrations nonlethal to monolayer ErbB2–overexpressing breast cancer cells.
Metformin targets ErbB2 and its downstream signaling molecules that serve as key factors for maintaining self-renewal/proliferation of CSCs/TICs
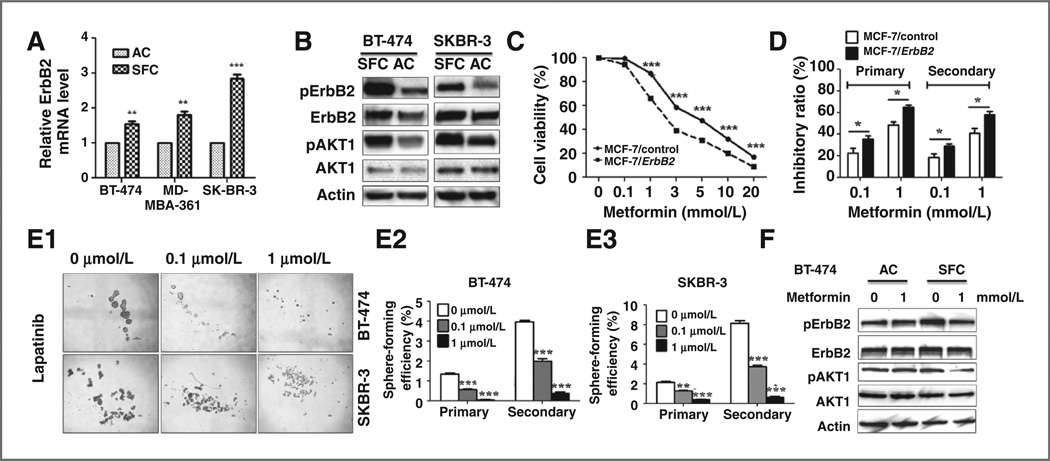
It has been reported that ErbB2 regulates the mammary stem/progenitor cell population that drives tumorigenesis and invasion (15). We compared the expression and activation of ErbB2 in monolayer attached cells and secondary tumorsphere cells, which are enriched in CSCs, to correlate ErbB2 status with metformin responsiveness (Fig. 6A). Results from multiple ErbB2–overexpressing breast cancer cell lines showed that ErbB2 mRNA and protein levels in sphere formation cells (SFC) were significantly higher than the attached cells (Fig. 6A and B), which is consistent with a previous report by Magnifico and colleagues (38). Importantly, phosphorylation/activation of ErbB2 and Akt in CSC/TIC-enriched SFCs was increased accordingly (Fig. 6B). These data underscore the role of ErbB2 deregulation in TIC/CSCs and the significance of metformin-mediated inhibition of ErbB2 signaling in its suppression of TICs/ CSCs.
Figure 6.
The role of ErbB2 expression and signaling in metformin-mediated inhibition of TICs/CSCs. A, relative mRNA levels of ErbB2 in attached monolayer cells and secondary tumorsphere of breast cancer cell lines. AC, attached cells. **,P< 0.01; ***P < 0.001. B, protein levels of ErbB2, pErbB2, Akt, and pAkt in attached monolayer cells and secondary tumorspheres detected by Western blot analysis. C, effect of metformin on MCF-7 with different ErbB2 statuses. MCF-7/control and MCF-7/ErbB2 cells were treated with metformin at indicated concentrations for 6 days. The survival fractions were detected by SRB assays. D, inhibitory ratio of low-dose metformin on MCF-7-derived primary and secondary tumorsphere. The ratio was expressed as the difference of tumorsphere numbers between the treated and untreated samples of the same cell line were divided by the tumorsphere number of the untreated cells. E, blocking ErbB2/EGFR signaling inhibits primary and secondary tumorsphere formation. BT-474 and SKBR-3 cells were treated with lapatinib at indicated concentrations in tumorsphere culture. E1, representative images of the 2 cell lines with different lapatinib treatments. E2, E3, tumorsphere-forming efficiencies of BT-474 (E2) and SKBR-3 (E3) cells with lapatinib treatments. F, ErbB2 expression and ErbB2/PI3K/AKT signaling in metformin-treated BT-474 cells that were in attached monolayer (AC) or secondary tumorspheres (SFC).
To determine metformin responsiveness in cancer cells with different ErbB2 status, MCF-7 control (MCF-7/con-trol) and ErbB2–overexpressing isogenic cells (MCF-7/ ErbB2) were treated with metformin at graded doses for 6 days, followed by SRB assays. Although ErbB2 overexpression enhanced MCF-7 cell proliferation and tumorsphere formation (Supplementary Figs. S1 and S2), MCF-7/ErbB2 cells were more sensitive to metformin (Fig. 6C). Similarly, metformin also induced a stronger inhibition of tumor sphere formation in MCF-7/ErbB2 cells as compared with MCF-7/control (Fig. 6D).
To determine the crucial role of ErbB2 signaling in CSCs/ TICs self-renewal, BT-474 and SKBR-3 cells were treated with lapatinib,a dual inhibitor that targets ErbB2 and EGFR, followed by tumorsphere assays. The results indicated that the blockage of ErbB2/EGFR signaling resulted in a consistent, sharp decrease of sphere-forming frequencies in primary and secondary tumorspheres of both cell lines (Fig. 6E). These data signify the role of RTK signaling in maintaining self-renewal/proliferation of CSCs/TICs and support the significance of metformin-mediated inhibition of ErbB2/EGFR expression/activation in its anti-CSC/TIC activities.
Finally, the differential effects of metformin on ErbB2 expression and activation in attached monolayer BT-474 cells and corresponding secondary tumorspheres were compared. Metformin treatment at 1 mmol/L for 72 hours resulted in little or no changes of ErbB2/Akt levels in the attached cells but caused a substantial reduction in both ErbB2 protein level and AKT phosphorylation in tumor-sphere cells (Fig. 6F). Taken together, these data indicate that metformin targets ErbB2 signaling that is preferentially expressed and activated in CSCs/TICs of human breast cancer, which serves as key factors in the maintenance of self-renewal/proliferation; this preference enables metformin to inhibit stem cell-like cells at nonlethal concentrations.
Discussion
Recent advances in metformin-associated antitumor studies hold great promise for improving cancer treatment and prevention with a blockbuster antidiabetes drug. A number of clinical trials are being carried out to test metformin’s efficacy in different clinical settings (39). However, the underlying mechanisms and factors that may affect metformin’s efficacy remain unclear. In particular, preclinical studies on metformin-mediated cancer prevention are limited. Pioneer work by Anisimov and colleagues demonstrated observational evidence indicating that metformin extends the life span and inhibits the development of spontaneous mammary tumors in MMTV-ErbB2 transgenic mice (24). In this report, we advanced Anisimov’s observation and demonstrated for the first time that systemic administration of metformin selectively targets TICs in a clinically relevant prevention model, which is a novel development of metformin-related implications for breast cancer prevention and treatment.
This study was aimed to investigate mechanisms of metformin-associated prevention of mammary tumor development in MMTV-ErbB2 mice by focusing on its impact on mammary stem cells and/or TICs. We found that metformin treatment selectively inhibited CD61high/CD49fhigh subpop-ulation in the preneoplastic mammary tissues of MMTV-ErbB2 transgenic mice. It has previously been demonstrated that CD61high/CD49fhigh cells are the TICs of mammary tumor development in MMTV-ErbB2 transgenic mice, which are significantly increased during transition from preneoplasia to tumor formation (9). Therefore, our data suggest that metformin selectively targetsTICsinErbB2–mediatedtumor development. This is further supported by the decrease of CD61high/CD49fhigh cells in 78617 cells treated with metformin in vitro. As CD61high/CD49fhigh cells are potentially derived from luminal progenitors (9), and we showed that mammosphere formation efficiency of primary MECs from metformin-treated mice was significantly reduced, it is likely that metformin inhibits the transition of luminal progenitors to CD61high/CD49fhigh TICs and their further expansion. Moreover, results from the syngeneic transplantation experiments showed that pretreatment MMTV-ErbB2 tumor-derived 78617 cell line with low-dose metformin significantly inhibited graft tumor initiation/development after transplantation. Because the cell numbers for injection of the control and metformin-treated groups have been adjusted to be equal based on viability, inhibited tumor development/growth in metformin pretreated samples may reflect decreased TIC numbers (Fig. 1D and E) and tumor static effect of metformin, rather than cell death before transplantation.
Although metformin-mediated inhibition of CSCs has been reported in established breast cancer cell line models (40), our results advance our understanding of metformin-mediated inhibition of TICs in preneoplastic mammary tissues or at risk stages, which underscores its potential as a chemopreventive agent, especially for ErbB2–overexpressing breast cancer. Moreover, results from this study also suggest that analysis of CD61high/CD49fhigh TIC subpopulation could be a useful tool for the identification of other TIC/CSC targeting agents.
In support of the in vivo data, our in vivo results demonstrated that low-dose metformin inhibited primary and secondary tumorsphere formation of ErbB2–overexpressing breast cancer cells. Metformin-induced decrease of ALDEFLUOR-positive populations in BT-474 and SKBR-3 cells further demonstrated its capability in targeting TICs/ CSCs. Results from lapatinib-mediated inhibition of tumor-sphere formation supported the role of RTK blockade in metformin-mediated inhibition of TIC/CSC self-renewal and proliferation. Moreover, we demonstrated that ErbB2 expression and activation were enhanced in CSC-enriched tumorspheres, and ErbB2–overexpressing breast cancer cells were more sensitive to metformin-mediated inhibition of tumorsphere formation and proliferation, which may partially explain metformin’s selective effect on ErbB2– overexpressing TICs/CSCs.
Mechanisms of metformin-mediated antitumor activities have been studied in different cell line models, and the derived data indicate that LKB1/AMPK/mTOR/ p70S6K pathway (39) and ErbB2 expression are the major functional targets of metformin (41). However, in vivo effects of metformin on molecular signaling in mammary tissues have been rarely reported. Our results demonstrated that the activation/phosphorylation of ErbB2, EGFR, IGF-IR AKT1, ERK1/2, Stat3, and mTOR in metformin-treated mammary tissues was significantly inhibited, which was accompanied by downregulation of ErbB2 and EGFR expression. Of note, these changes were from tissues harvested 18 hours after the last metformin injection, suggesting a long-lasting effect induced by metformin. In context with the report that ErbB2–regulated mammary stem/progenitor cell population is a driving force of related tumorigenesis and invasion (15), and the regulation of PTEN/AKT/b-catenin signaling is vital in regulating mammary stem/progenitor cells (42), down-regulation of RTK-activated PI3K/Akt, MAPK/Erk, mTOR/ p70S6K, and Stat3 pathways may play a critical role in metformin-induced inhibition of CD61high/CD49fhigh subpopulation and the consequent tumor initiation.
How metformin induces the changes in molecular signaling and the interplay among those pathways is intriguing. It is generally believed that metformin exerts its effects mainly through the activation of LKB1/AMPK, which leads to the inhibition of mTOR/p70S6K pathway (43). Downregulation of mTOR/p70S6K signaling was also reported to suppress the expression of ErbB2 and IGF-1R expression (41). We observed that metformin inhibited both the expression and phosphorylation of ErbB2 and EGFR but mainly the phosphorylation of ErbB3 and IGF-1R in vivo. Because ErbB2 activation is closely regulated by its interaction with its family members and IGF-1R (44), the effect of metformin on ErbB2 signaling may involve multiple mechanisms yet to be defined. Similarly, metformin-induced inhibition of mTOR phosphorylation could have resulted from the inhibition of Akt pathway and activation of AMPK pathways and p53/ REDD1 axis (41, 45, 46). Interestingly, we observed that metformin-induced AMPK phosphorylation/activation was very sensitive to metformin in cell culture system (Fig. 4) but not in the harvested mammary tissues (Fig. 2). This is likely because AMPK activation is a short-lived response and its changes were not detectable when the tissues were harvested 18 hours after the last injection. Nevertheless, the significance of AMPK activation should not be overlooked. Moreover, metformin-induced inhibition of Stat3 phosphorylation was previously detected in triple-negative breast cancer cell lines (21). We found that Stat3 phosphorylation was also sensitive to metformin in vivo and in SKBR-3 cells, suggesting that regulation of IL-6/JAK2/Stat3 pathway may also play an important role in metformin-mediated inhibition of TICs/ CSCs. This is supported by a recent report demonstrating that Stat3 is required to maintain the full differentiation potential of mammary stem cells and the proliferative potential of mammary luminal progenitors (47). Moreover, because Stat3 could be activated/inhibited independent of ErbB2 signaling (21), the role of Stat3 inactivation and the interactions between Stat3 and ErbB2 in metformin induced inhibition of TICs/CSCs warrant further investigation. Taken together, our mechanistic studies support the role of RTK inactivation and Stat3 blockage in metformin-mediated inhibition of TICs/ CSCs in ErbB2 tumor development. However, the specifics that lead to metformin-induced RTK inactivation and pathway-crosstalk remain to be defined.
Although our results showed that the proliferation and self-renewal/sphere formation of ErbB2–overexpressing cells were more sensitive to metformin, given that metformin also suppresses CSCs of other type of cells/cancers, such as pancreatic and ovarian cancers, metformin may also target critical steps common to TIC/CSC regulatory machinery. For example, metformin was reported to represses the self-renewal of breast CSC via inhibition of ER-mediated OCT4 expression (48). Metformin-mediated regulation of E-Cadherin can efficiently prevent the TGF-β–induced EMT (49). Therefore, metformin-induced changes in these fundamental mechanisms may interact with the signaling pathways specific to certain cells type, such as ErbB2 overexpression, and give rise to various responses in different cell/ cancer types.
The dose of metformin used in anticancer studies has been a debating issue. After a dose–response test in relevant cell lines (Fig. 4A), we used relatively low doses, 0.1 to 1 mmol/L, in the subsequent in vivo experiments. Although the doses were much lower than that in many previous studies, which ranged from 1 to 30 mmol/L (45, 50, 51), we should acknowledge that the concentrations are much higher than the therapeutic plasma levels in clinical medicine(40,52). However, the discrepancy between in vivo and in vitro conditions could be smaller if the following factors are considered: (i) metformin levels in tissues could be several-fold higher than those in blood because of its tissue accumulation (53); (ii) supplements in the medium may affect metformin’s action in cell culture system. (iii) longer exposure to metformin, as it is in vivo, may lower the threshold for metformin-associated activities. Indeed, our data indicated that the dose–response curve for a 6-day treatment was significantly shifted to a lower threshold as compared with that of a 3-day treatment (Supplementary Fig. S3). Based on previous reports (46, 51), we used a dose of 250 mg/kg in our in vivo studies. Although it was well tolerated by the animals, it is apparently higher than the therapeutic doses for diabetes. This should also be cautiously considered when the data are used for clinical reference. Nevertheless, data from this study have provided proof of concept for metformin induced inhibition of TICs in ErbB2–mediated carcinogenesis, which will be tested with various doses.
In summary, we demonstrated that systemic administration of metformin selectively inhibited TICs subpopulation in MMTV-ErbB2 tumor model. Downregulation of RTK-mediated signaling through multiple mechanisms plays a critical role in this process, which warrant further investigation. Data from this in vivo study will be particularly valuable in developing novel strategies for the prevention of ErbB2–mediated carcinogenesis.
Supplementary Material
Acknowledgments
Grant Support
This work was supported in part by a Research Scholar Grant to X. Yang from American Cancer Society (RSG-08-138-01-CNE).
Footnotes
Note: Supplementary data for this article are available at Cancer Prevention Research Online (http://cancerprevres.aacrjournals.org/).
Disclosure of Potential Conflicts of Interest
No potential conflicts of interest were disclosed.
Authors' Contributions
Conception and design: X. Yang, P. Zhu
Development of methodology: P. Zhu, X. Yang, M. Davis, A.J. Blackwelder
Acquisition of data (provided animals, acquired and managed patients, provided facilities, etc.): P. Zhu, M. Davis, A.J. Blackwelder
Analysis and interpretation of data (e.g., statistical analysis, biostatistics, computational analysis): P. Zhu, M. Davis, X. Yang
Writing, review, and/or revision of the manuscript: X. Yang, P. Zhu, N. Bachman
Administrative, technical, or material support (i.e., reporting or organizing data, constructing databases): A.D. Thor, B. Liu, S. Edgerton, L.L. Williams
Study supervision: X. Yang
References
- 1.Rosen PP, Groshen S, Saigo PE, Kinne DW, Hellman S. Pathological prognostic factors in stage I (T1N0M0) and stage II (T1N1M0) breast carcinoma: a study of 644 patients with median follow-up of 18 years. J Clin Oncol. 1989;7:1239–1251. doi: 10.1200/JCO.1989.7.9.1239. [DOI] [PubMed] [Google Scholar]
- 2.Reya T, Morrison SJ, Clarke MF, Weissman IL. Stem cells, cancer, and cancer stem cells. Nature. 2001;414:105–111. doi: 10.1038/35102167. [DOI] [PubMed] [Google Scholar]
- 3.Al-Hajj M, Wicha MS, Benito-Hernandez A, Morrison SJ, Clarke MF. Prospective identification of tumorigenic breast cancer cells. Proc Natl Acad Sci U S A. 2003;100:3983–3988. doi: 10.1073/pnas.0530291100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ponti D, Costa A, Zaffaroni N, Pratesi G, Petrangolini G, Coradini D, et al. Isolation and in vitro propagation of tumorigenic breast cancer cells with stem/progenitor cell properties. Cancer Res. 2005;65:5506–5511. doi: 10.1158/0008-5472.CAN-05-0626. [DOI] [PubMed] [Google Scholar]
- 5.Stingl J, Eaves CJ, Kuusk U, Emerman JT. Phenotypic and functional characterization in vitro of a multipotent epithelial cell present in the normal adult human breast. Differentiation. 1998;63:201–213. doi: 10.1111/j.1432-0436.1998.00201.x. [DOI] [PubMed] [Google Scholar]
- 6.Stingl J, Eaves CJ, Zandieh I, Emerman JT. Characterization of bipotent mammary epithelial progenitor cells in normal adult human breast tissue. Breast Cancer Res Treat. 2001;67:93–109. doi: 10.1023/a:1010615124301. [DOI] [PubMed] [Google Scholar]
- 7.Gudjonsson T, Villadsen R, Nielsen HL, Ronnov-Jessen L, Bissell MJ, Petersen OW. Isolation, immortalization, and characterization of a human breast epithelial cell line with stem cell properties. Genes Dev. 2002;16:693–706. doi: 10.1101/gad.952602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ginestier C, Hur MH, Charafe-Jauffret E, Monville F, Dutcher J, Brown M, et al. ALDH1 is a marker of normal and malignant human mammary stem cells and a predictor of poor clinical outcome. Cell Stem Cell. 2007;1:555–567. doi: 10.1016/j.stem.2007.08.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lo PK, Kanojia D, Liu X, Singh UP, Berger FG, Wang Q, et al. CD49f and CD61 identify Her2/neu-induced mammary tumor-initiating cells that are potentially derived from luminal progenitors and maintained by the integrin-TGF-β signaling. Oncogene. 2012;31:2614–2626. doi: 10.1038/onc.2011.439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Slamon DJ, Clark GM, Wong SG, Levin WJ, Ullrich A, McGuire WL. Human breast cancer: correlation of relapse and survival with amplification of the HER-2/neu oncogene. Science. 1987;235:177–182. doi: 10.1126/science.3798106. [DOI] [PubMed] [Google Scholar]
- 11.Olayioye MA. Update on HER-2 as a target for cancer therapy: intracellular signaling pathways of ErbB2/HER-2 and family members. Breast Cancer Res. 2001;3:385–389. doi: 10.1186/bcr327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Tan M, Yao J, Yu D. Overexpression of the c-ErbB2 gene enhanced intrinsic metastasis potential in human breast cancer cells without increasing their transformation abilities. Cancer Res. 1997;57:1199–1205. [PubMed] [Google Scholar]
- 13.Moody SE, Sarkisian CJ, Hahn KT, Gunther EJ, Pickup S, Dugan KD, et al. Conditional activation of Neu in the mammary epithelium of transgenic mice results in reversible pulmonary metastasis. Cancer Cell. 2002;2:451–461. doi: 10.1016/s1535-6108(02)00212-x. [DOI] [PubMed] [Google Scholar]
- 14.Holbro T, Civenni G, Hynes NE. The ErbB receptors and their role in cancer progression. Exp Cell Res. 2003;284:99–110. doi: 10.1016/s0014-4827(02)00099-x. [DOI] [PubMed] [Google Scholar]
- 15.Korkaya H, Paulson A, Iovino F, Wicha MS. HER2 regulates the mammary stem/progenitor cell population driving tumorigenesis and invasion. Oncogene. 2008;27:6120–6130. doi: 10.1038/onc.2008.207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Pierotti MA, Berrino F, Gariboldi M, Melani C, Mogavero A, Negri T, et al. Targeting metabolism for cancer treatment and prevention: metformin, an old drug with multi-faceted effects. Oncogene. 2013;32:1475–1487. doi: 10.1038/onc.2012.181. [DOI] [PubMed] [Google Scholar]
- 17.Giovannucci E, Harlan DM, Archer MC, Bergenstal RM, Gapstur SM, Habel LA, et al. Diabetes and cancer: a consensus report. Diabetes Care. 2010;33:1674–1685. doi: 10.2337/dc10-0666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Jiralerspong S, Palla SL, Giordano SH, Meric-Bernstam F, Liedtke C, Barnett CM, et al. Metformin and pathologic complete responses to neoadjuvant chemotherapy in diabetic patients with breast cancer. J Clin Oncol. 2009;27:3297–3302. doi: 10.1200/JCO.2009.19.6410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hirsch HA, Iliopoulos D, Tsichlis PN, Struhl K. Metformin selectively targets cancer stem cells, and acts together with chemotherapy to block tumor growth and prolong remission. Cancer Res. 2009;69:7507–7511. doi: 10.1158/0008-5472.CAN-09-2994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Vazquez-Martin A, Oliveras-Ferraros C, Cufi S, Del Barco S, Martin-Castillo B, Menendez JA. Metformin regulates breast cancer stem cell ontogeny by transcriptional regulation of the epithelial-mesenchymal transition (EMT) status. Cell Cycle. 2010;9:3807–3814. [PubMed] [Google Scholar]
- 21.Deng XS, Wang S, Deng A, Liu B, Edgerton SM, Lind SE, et al. Metformin targets Stat3 to inhibit cell growth and induce apoptosis in triple-negative breast cancers. Cell Cycle. 2012;11:367–376. doi: 10.4161/cc.11.2.18813. [DOI] [PubMed] [Google Scholar]
- 22.Cufi S, Corominas-Faja B, Vazquez-Martin A, Oliveras-Ferraros C, Dorca J, Bosch-Barrera J, et al. Metformin-induced preferential killing of breast cancer initiating CD44+CD24—/low cells is sufficient to overcome primary resistance to trastuzumab in HER2+ human breast cancer xenografts. Oncotarget. 2012;3:395–398. doi: 10.18632/oncotarget.488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Liu B, Fan Z, Edgerton SM, Yang X, Lind SE, Thor AD. Potent antiproliferative effects of metformin on trastuzumab-resistant breast cancer cells via inhibition of ErbB2/IGF-1 receptor interactions. Cell Cycle. 2011;10:2959–2966. doi: 10.4161/cc.10.17.16359. [DOI] [PubMed] [Google Scholar]
- 24.Anisimov VN, Berstein LM, Egormin PA, Piskunova TS, Popovich IG, Zabezhinski MA, et al. Effect of metformin on life span and on the development of spontaneous mammary tumors in HER-2/neu transgenic mice. Exp Gerontol. 2005;40:685–693. doi: 10.1016/j.exger.2005.07.007. [DOI] [PubMed] [Google Scholar]
- 25.Anisimov VN, Egormin PA, Piskunova TS, Popovich IG, Tyndyk ML, Yurova MN, et al. Metformin extends life span of HER-2/neu transgenic mice and in combination with melatonin inhibits growth of transplantable tumors in vivo . Cell Cycle. 2010;9:188–197. doi: 10.4161/cc.9.1.10407. [DOI] [PubMed] [Google Scholar]
- 26.Zou MH, Kirkpatrick SS, Davis BJ, Nelson JS, Wiles WGt, Schlattner U, et al. Activation of the AMP-activated protein kinase by the antidiabetic drug metformin in vivo. Role of mitochondrial reactive nitrogen species. J Biol Chem. 2004;279:43940–43951. doi: 10.1074/jbc.M404421200. [DOI] [PubMed] [Google Scholar]
- 27.Qu Z, Zhang Y, Liao M, Chen Y, Zhao J, Pan Y. In vitro and in vivo antitumoral action of metformin on hepatocellular carcinoma. Hepatol Res. 2012;42:922–933. doi: 10.1111/j.1872-034X.2012.01007.x. [DOI] [PubMed] [Google Scholar]
- 28.Kim A, Liu B, Ordonez-Ercan D, Alvarez KM, Jones LD, McKimmey C, et al. Functional interaction between mouse ErbB3 and wild-type rat c-neu in transgenic mouse mammary tumor cells. Breast Cancer Res. 2005;7:R708–R718. doi: 10.1186/bcr1281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Yang X, Edgerton SM, Kosanke SD, Mason TL, Alvarez KM, Liu N, et al. Hormonal and dietary modulation of mammary carcinogenesis in mouse mammary tumor virus-c-ErbB2 transgenic mice. Cancer Res. 2003;63:2425–2433. [PubMed] [Google Scholar]
- 30.Shelton DN, Fernandez-Gonzalez R, Illa-BochacaI , Ortiz-de-Solorzano C, Barcellos-Hoff MH, Welm BE. Use of stem cell markers in dissociated mammary populations. Methods Mol Biol. 2010;621:49–55. doi: 10.1007/978-1-60761-063-2_3. [DOI] [PubMed] [Google Scholar]
- 31.Tophkhane C, Yang SH, Jiang Y, Ma Z, Subramaniam D, Anant S, et al. p53 inactivation upregulates p73 expression through E2F-1 mediated transcription. PLoS ONE. 2012;7:e43564. doi: 10.1371/journal.pone.0043564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Liao MJ, Zhang CC, Zhou B, Zimonjic DB, Mani SA, Kaba M, et al. Enrichment of a population of mammary gland cells that form mammo-spheres and have in vivo repopulating activity. Cancer Res. 2007;67:8131–8138. doi: 10.1158/0008-5472.CAN-06-4493. [DOI] [PubMed] [Google Scholar]
- 33.Cicalese A, Bonizzi G, Pasi CE, Faretta M, Ronzoni S, Giulini B, et al. The tumor suppressor p53 regulates polarity of self-renewing divisions in mammary stem cells. Cell. 2009;138:1083–1095. doi: 10.1016/j.cell.2009.06.048. [DOI] [PubMed] [Google Scholar]
- 34.Liu JC, Deng T, Lehal RS, Kim J, Zacksenhaus E. Identification of tumorsphere- and tumor-initiating cells in HER2/Neu-induced mammary tumors. Cancer Res. 2007;67:8671–8681. doi: 10.1158/0008-5472.CAN-07-1486. [DOI] [PubMed] [Google Scholar]
- 35.Grimshaw MJ, Cooper L, Papazisis K, Coleman JA, Bohnenkamp HR, Chiapero-Stanke L, et al. Mammosphere culture of metastatic breast cancer cells enriches for tumorigenic breast cancer cells. Breast Cancer Res. 2008;10:R52. doi: 10.1186/bcr2106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Cioce M, Gherardi S, Viglietto G, Strano S, Blandino G, Muti P, et al. Mammosphere-forming cells from breast cancer cell lines as a tool for the identification of CSC-like- and early progenitor-targeting drugs. Cell Cycle. 2010;9:2878–2887. [PubMed] [Google Scholar]
- 37.Guy CT, Webster MA, Schaller M, Parsons TJ, Cardiff RD, Muller WJ. Expression of the neu protooncogene in the mammary epithelium of transgenic mice induces metastatic disease. Proc Natl Acad Sci U S A. 1992;89:10578–10582. doi: 10.1073/pnas.89.22.10578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Magnifico A, Albano L, Campaner S, Delia D, Castiglioni F, Gasparini P, et al. Tumor-initiating cells of HER2-positive carcinoma cell lines express the highest oncoprotein levels and are sensitive to trastuzumab. Clin Cancer Res. 2009;15:2010–2021. doi: 10.1158/1078-0432.CCR-08-1327. [DOI] [PubMed] [Google Scholar]
- 39.Vazquez-Martin A, Oliveras-Ferraros C, Del Barco S, Martin-Castillo B, Menendez JA. If mammalian target of metformin indirectly is mammalian target of rapamycin, then the insulin-like growth factor-1 receptor axis will audit the efficacy of metformin in cancer clinical trials. J Clin Oncol. 2009;27:e207–e209. doi: 10.1200/JCO.2009.24.5456. author reply e10. [DOI] [PubMed] [Google Scholar]
- 40.Vazquez-Martin A, Oliveras-Ferraros C, Del Barco S, Martin-Castillo B, Menendez JA. The anti-diabetic drug metformin suppresses self-renewal and proliferation of trastuzumab-resistant tumor-initiating breast cancer stem cells. Breast Cancer Res Treat. 2011;126:355–364. doi: 10.1007/s10549-010-0924-x. [DOI] [PubMed] [Google Scholar]
- 41.Vazquez-Martin A, Oliveras-Ferraros C, Menendez JA. The antidia-betic drug metformin suppresses HER2 (ErbB2) oncoprotein over-expression via inhibition of the mTOR effector p70S6K1 in human breast carcinoma cells. Cell Cycle. 2009;8:88–96. doi: 10.4161/cc.8.1.7499. [DOI] [PubMed] [Google Scholar]
- 42.Korkaya H, Paulson A, Charafe-Jauffret E, Ginestier C, Brown M, Dutcher J, et al. Regulation of mammary stem/progenitor cells by PTEN/Akt/β-catenin signaling. PLoS Biol. 2009;7:e1000121. doi: 10.1371/journal.pbio.1000121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Goodwin PJ, Ligibel JA, Stambolic V. Metformin in breast cancer: time for action. J Clin Oncol. 2009;27:3271–3273. doi: 10.1200/JCO.2009.22.1630. [DOI] [PubMed] [Google Scholar]
- 44.Huang X, Gao L, Wang S, McManaman JL, Thor AD, Yang X, et al. Heterotrimerization of the growth factor receptors ErbB2, ErbB3, and insulin-like growth factor-i receptor in breast cancer cells resistant to herceptin. Cancer Res. 2010;70:1204–1214. doi: 10.1158/0008-5472.CAN-09-3321. [DOI] [PubMed] [Google Scholar]
- 45.Gou S, Cui P, Li X, Shi P, Liu T, Wang C. Low concentrations of metformin selectively inhibit CD133(+) cell proliferation in pancreatic cancer and have anticancer action. PLoS ONE. 2013;8:e63969. doi: 10.1371/journal.pone.0063969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Memmott RM, Mercado JR, Maier CR, Kawabata S, Fox SD, Dennis PA. Metformin prevents tobacco carcinogen-induced lung tumorigenesis. Cancer Prev Res. 2010;3:1066–1076. doi: 10.1158/1940-6207.CAPR-10-0055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Staniszewska AD, Pensa S, Caffarel MM, Anderson LH, Poli V, Watson CJ. Stat3 is required to maintain the full differentiation potential of mammary stem cells and the proliferative potential of mammary luminal progenitors. PLoS ONE. 2012;7:e52608. doi: 10.1371/journal.pone.0052608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Jung JW, Park SB, Lee SJ, Seo MS, Trosko JE, Kang KS. Metformin represses self-renewal of the human breast carcinoma stem cells via inhibition of estrogen receptor-mediated OCT4 expression. PLoS ONE. 2011;6:e28068. doi: 10.1371/journal.pone.0028068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Cufi S, Vazquez-Martin A, Oliveras-Ferraros C, Martin-Castillo B, Joven J, Menendez JA. Metformin against TGFβ-induced epithelial-to-mesenchymal transition (EMT): from cancer stem cells to aging-associated fibrosis. Cell Cycle. 2010;9:4461–4468. doi: 10.4161/cc.9.22.14048. [DOI] [PubMed] [Google Scholar]
- 50.Bao B, Wang Z, Ali S, Ahmad A, Azmi AS, Sarkar SH, et al. Metformin inhibits cell proliferation, migration and invasion by attenuating CSC function mediated by deregulating miRNAs in pancreatic cancer cells. Cancer Prev Res. 2012;5:355–364. doi: 10.1158/1940-6207.CAPR-11-0299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ben Sahra I, Regazzetti C, Robert G, Laurent K, Le Marchand-Brustel Y, Auberger P, et al. Metformin, independent of AMPK, induces mTOR inhibition and cell-cycle arrest through REDD1. Cancer Res. 2011;71:4366–4372. doi: 10.1158/0008-5472.CAN-10-1769. [DOI] [PubMed] [Google Scholar]
- 52.Sum CF, Webster JM, Johnson AB, Catalano C, Cooper BG, Taylor R. The effect of intravenous metformin on glucose metabolism during hyperglycaemia in type 2 diabetes. Diabet Med. 1992;9:61–65. doi: 10.1111/j.1464-5491.1992.tb01716.x. [DOI] [PubMed] [Google Scholar]
- 53.Wilcock C, Bailey CJ. Accumulation of metformin by tissues of the normal and diabetic mouse. Xenobiotica. 1994;24:49–57. doi: 10.3109/00498259409043220. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.