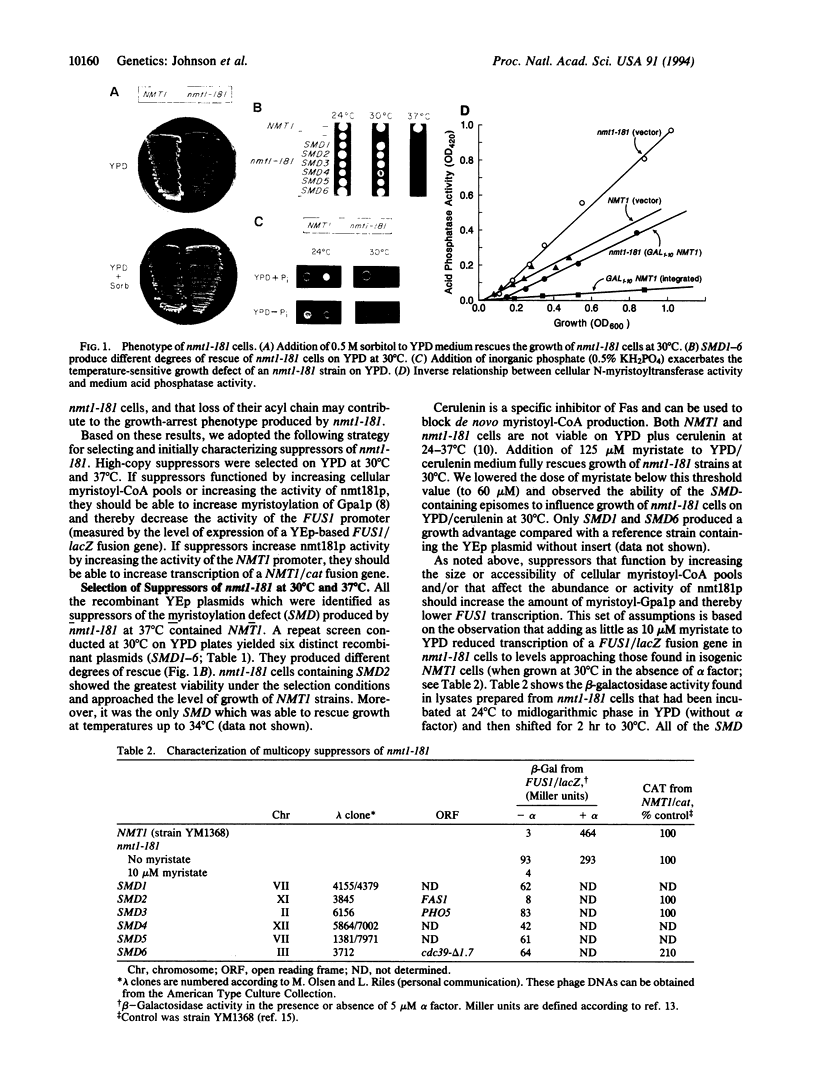
Abstract
Several essential Saccharomyces cerevisiae proteins require myristate to be covalently bound to their amino-terminal glycine for biological activity. Protein N-myristoylation is catalyzed by myristoyl-CoA:protein N-myristoyl-transferase, Nmt1p. nmt1-181 encodes a mutant enzyme with a Gly451-->Asp substitution. nmt181p has a reduced affinity for myristoyl-CoA and produces global defects in protein N-myristoylation at > or = 30 degrees C. nmt1-181 results in growth arrest at various stages of the cell cycle within 1 hr after cells are shifted to > or = 30 degrees C and lethality within 8 hr. The growth-arrest phenotype and loss of viability do not require components of the mating pathway and are associated with lysis sensitivity that may be related to undermyristoylation of two protein phosphatases, Ppz1p and Ppz2p. Growth can be rescued at 30 degrees C by adding myristate or sorbitol to the medium or by removing inosine. Cells can be rescued at 37 degrees C by overexpressing nmt1-181p or Nmt1p or by adding myristate to the medium. Selection of high-copy suppressors of the myristate auxotrophy and lethality observed at 37 degrees C yielded only NMT1, whereas six unlinked suppressors of the myristoylation defect (SMD1-6) were obtained when the screen was conducted at 30 degrees C. The protein products of three SMD loci were identified: (i) cdc39-delta 1.7p, which transactivates NMT1; (ii) Fas1p, the beta subunit of the fatty acid synthetase complex, activates FAS2's promoter and increases myristoylation of Gpa1p; and (iii) Pho5p, the major secreted acid phosphatase produced by this yeast. PHO5 is normally induced when yeast are grown in phosphate-depleted medium. Removal of inorganic phosphate from the medium also rescues nmt1-181 cells at 30 degrees C. PHO5's mechanism of suppression of nmt1-181 appears to involve, at least in part, activation of FAS2 transcription and a resulting effect on FAS1 expression. There is an inverse relationship between cellular N-myristoyltransferase and secreted acid phosphatase activities. These observations provide a potential mechanism for coupling phosphate metabolism with the regulation of myristoyl-CoA synthesis and protein N-myristoylation.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bhatnagar R. S., Jackson-Machelski E., McWherter C. A., Gordon J. I. Isothermal titration calorimetric studies of Saccharomyces cerevisiae myristoyl-CoA:protein N-myristoyltransferase. Determinants of binding energy and catalytic discrimination among acyl-CoA and peptide ligands. J Biol Chem. 1994 Apr 15;269(15):11045–11053. [PubMed] [Google Scholar]
- Carlson M., Botstein D. Two differentially regulated mRNAs with different 5' ends encode secreted with intracellular forms of yeast invertase. Cell. 1982 Jan;28(1):145–154. doi: 10.1016/0092-8674(82)90384-1. [DOI] [PubMed] [Google Scholar]
- Chirala S. S. Coordinated regulation and inositol-mediated and fatty acid-mediated repression of fatty acid synthase genes in Saccharomyces cerevisiae. Proc Natl Acad Sci U S A. 1992 Nov 1;89(21):10232–10236. doi: 10.1073/pnas.89.21.10232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collart M. A., Struhl K. CDC39, an essential nuclear protein that negatively regulates transcription and differentially affects the constitutive and inducible HIS3 promoters. EMBO J. 1993 Jan;12(1):177–186. doi: 10.1002/j.1460-2075.1993.tb05643.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dohlman H. G., Goldsmith P., Spiegel A. M., Thorner J. Pheromone action regulates G-protein alpha-subunit myristoylation in the yeast Saccharomyces cerevisiae. Proc Natl Acad Sci U S A. 1993 Oct 15;90(20):9688–9692. doi: 10.1073/pnas.90.20.9688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duronio R. J., Rudnick D. A., Johnson R. L., Johnson D. R., Gordon J. I. Myristic acid auxotrophy caused by mutation of S. cerevisiae myristoyl-CoA:protein N-myristoyltransferase. J Cell Biol. 1991 Jun;113(6):1313–1330. doi: 10.1083/jcb.113.6.1313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duronio R. J., Towler D. A., Heuckeroth R. O., Gordon J. I. Disruption of the yeast N-myristoyl transferase gene causes recessive lethality. Science. 1989 Feb 10;243(4892):796–800. doi: 10.1126/science.2644694. [DOI] [PubMed] [Google Scholar]
- Egner R., Thumm M., Straub M., Simeon A., Schüller H. J., Wolf D. H. Tracing intracellular proteolytic pathways. Proteolysis of fatty acid synthase and other cytoplasmic proteins in the yeast Saccharomyces cerevisiae. J Biol Chem. 1993 Dec 25;268(36):27269–27276. [PubMed] [Google Scholar]
- Griggs D. W., Johnston M. Promoter elements determining weak expression of the GAL4 regulatory gene of Saccharomyces cerevisiae. Mol Cell Biol. 1993 Aug;13(8):4999–5009. doi: 10.1128/mcb.13.8.4999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson D. R., Duronio R. J., Langner C. A., Rudnick D. A., Gordon J. I. Genetic and biochemical studies of a mutant Saccharomyces cerevisiae myristoyl-CoA:protein N-myristoyltransferase, nmt72pLeu99-->Pro, that produces temperature-sensitive myristic acid auxotrophy. J Biol Chem. 1993 Jan 5;268(1):483–494. [PubMed] [Google Scholar]
- Johnson D. R., Knoll L. J., Rowley N., Gordon J. I. Genetic analysis of the role of Saccharomyces cerevisiae acyl-CoA synthetase genes in regulating protein N-myristoylation. J Biol Chem. 1994 Jul 8;269(27):18037–18046. [PubMed] [Google Scholar]
- Kaffman A., Herskowitz I., Tjian R., O'Shea E. K. Phosphorylation of the transcription factor PHO4 by a cyclin-CDK complex, PHO80-PHO85. Science. 1994 Feb 25;263(5150):1153–1156. doi: 10.1126/science.8108735. [DOI] [PubMed] [Google Scholar]
- Kamiryo T., Parthasarathy S., Numa S. Evidence that acyl coenzyme A synthetase activity is required for repression of yeast acetyl coenzyme A carboxylase by exogenous fatty acids. Proc Natl Acad Sci U S A. 1976 Feb;73(2):386–390. doi: 10.1073/pnas.73.2.386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee K. S., Hines L. K., Levin D. E. A pair of functionally redundant yeast genes (PPZ1 and PPZ2) encoding type 1-related protein phosphatases function within the PKC1-mediated pathway. Mol Cell Biol. 1993 Sep;13(9):5843–5853. doi: 10.1128/mcb.13.9.5843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCaffrey G., Clay F. J., Kelsay K., Sprague G. F., Jr Identification and regulation of a gene required for cell fusion during mating of the yeast Saccharomyces cerevisiae. Mol Cell Biol. 1987 Aug;7(8):2680–2690. doi: 10.1128/mcb.7.8.2680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meyer K. H., Schweizer E. Saturated fatty acid mutant of Saccharomyces cerevisiae with an intact fatty acid synthetase. J Bacteriol. 1974 Feb;117(2):345–350. doi: 10.1128/jb.117.2.345-350.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neiman A. M., Chang F., Komachi K., Herskowitz I. CDC36 and CDC39 are negative elements in the signal transduction pathway of yeast. Cell Regul. 1990 Apr;1(5):391–401. doi: 10.1091/mbc.1.5.391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oliver S. G., van der Aart Q. J., Agostoni-Carbone M. L., Aigle M., Alberghina L., Alexandraki D., Antoine G., Anwar R., Ballesta J. P., Benit P. The complete DNA sequence of yeast chromosome III. Nature. 1992 May 7;357(6373):38–46. doi: 10.1038/357038a0. [DOI] [PubMed] [Google Scholar]
- Reed S. I. The selection of S. cerevisiae mutants defective in the start event of cell division. Genetics. 1980 Jul;95(3):561–577. doi: 10.1093/genetics/95.3.561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rubin G. M. The nucleotide sequence of Saccharomyces cerevisiae 5.8 S ribosomal ribonucleic acid. J Biol Chem. 1973 Jun 10;248(11):3860–3875. [PubMed] [Google Scholar]
- Rudnick D. A., McWherter C. A., Gokel G. W., Gordon J. I. MyristoylCoA:protein N-myristoyltransferase. Adv Enzymol Relat Areas Mol Biol. 1993;67:375–430. doi: 10.1002/9780470123133.ch5. [DOI] [PubMed] [Google Scholar]
- Stearns T., Kahn R. A., Botstein D., Hoyt M. A. ADP ribosylation factor is an essential protein in Saccharomyces cerevisiae and is encoded by two genes. Mol Cell Biol. 1990 Dec;10(12):6690–6699. doi: 10.1128/mcb.10.12.6690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stone D. E., Cole G. M., de Barros Lopes M., Goebl M., Reed S. I. N-myristoylation is required for function of the pheromone-responsive G alpha protein of yeast: conditional activation of the pheromone response by a temperature-sensitive N-myristoyl transferase. Genes Dev. 1991 Nov;5(11):1969–1981. doi: 10.1101/gad.5.11.1969. [DOI] [PubMed] [Google Scholar]
- TORRIANI A. Influence of inorganic phosphate in the formation of phosphatases by Escherichia coli. Biochim Biophys Acta. 1960 Mar 11;38:460–469. doi: 10.1016/0006-3002(60)91281-6. [DOI] [PubMed] [Google Scholar]
- Trueheart J., Boeke J. D., Fink G. R. Two genes required for cell fusion during yeast conjugation: evidence for a pheromone-induced surface protein. Mol Cell Biol. 1987 Jul;7(7):2316–2328. doi: 10.1128/mcb.7.7.2316. [DOI] [PMC free article] [PubMed] [Google Scholar]