Introduction
In the fourth chapter of Geoffrey Harris’ widely acclaimed 1955 monograph (Harris 1955), which now 60 years later provides the corner stone of this special issue of the journal, he succinctly and systematically presented his views, and the evidence upon which they were based, on the neural mechanisms controlling the pituitary-gonadal axis. That gonadal function was under control by the central nervous system was well established at the time of Harris’ monograph, as was the recognition of the gonad-stimulating properties of pituitary gonadotropin, the relative insignificance of gonadal nerves to gonadal function and the concept of neurosecretion. The problem for Harris and his fellow neuroendocrinologists was how did the hypothalamus regulate the secretion of the anterior pituitary hormones, specifically gonadotropin in the context of this review, and what was the role of the hypopophysial portal system in this regard. After elegantly interpreting and summarizing the extant data, Harris proposed that of the hypotheses that were being debated at the time “the most likely seems to be that nerve fibres from the hypothalmus liberate some humoral substance(s) into the capillaries of the primary plexus in the median eminence and that this substance is carried by the portal vessels to excite or inhibit the cells of the pars distalis.” It is to be recalled, that this idea had been proposed by Harris and Green and Harris on several occasions prior to publication of the monograph (see Harris, 1955). It is also worth noting that the evidence upon which Harris’ hypothesis was based had been obtained primarily from studies of the female, most likely because ovulation was a discrete and readily detected event and, at the time, the only reliable surrogate marker of acute hypothalamic activation.
The main purpose of the present chapter is to describe the essential refinements and additional complexities that have been added to the fundamental model Harris put forward in 1955 for the neuroendocrine control of gonadal function (Figure 1). The major additions to the neuroendocrinologist’s armamentarium that have facilitated the development of the ideas of Harris are, according to an historical timeline, the introduction of radioimmunoassay to measure concentrations of pituitary and gonadal hormones in the peripheral circulation and GnRH in portal blood, the development of immunohistochemical techniques to localize neuropeptides and neurotransmitters in the hypothalamus, the application of techniques in molecular endocrinology to the study of gene expression in the hypothalamus and pituitary, the introduction of transgenic models, the advances in molecular genetics, and the arrival of the “omics” era. After discussing refinements to the Harris model resulting from the application of the foregoing technologies, the review will close with a brief outline of ways in which knowledge of the operation of this neuroendocrine axis has been applied to clinical practice, and a personal glimpse into the future of this field.
Figure 1.
Model proposed by Harris in his 1955 monograph to illustrate the relationship between the external environment and the reproductive organs (reprinted from Harris, 1955).
Harris’ “humoral” substance (gonadotropin releasing hormone, GnRH) and its mode of release
After many years of herculean effort, a struggle between two rival laboratories and many setbacks1, the isolation of the humoral substance of hypothalamic origin that is secreted into the hypophysial portal circulation to regulate the synthesis and secretion of the gonadotopins, luteinizing hormone (LH) and follicle stimulating hormone (FSH), was independently achieved from bovine and ovine brain by the groups of Schally and Guilleman, respectively; ironically this work was published in 1971, the year of Harris’ death (Amoss, et al. 1971; Matsuo, et al. 1971). It was a decapeptide that was initially termed luteinizing hormone releasing hormone (LHRH) or luteinizing hormone releasing factor (LRF), but the molecule is now generally referred to as GnRH12. For their labors, Schally and Guilleman were awarded the Nobel Prize in Physiology or Medicine in 1977. Had it not been for Harris’ untimely death at an age of 58 he surely would have shared this most prestigious of awards for laying the conceptual underpinnings for the work of Schally and Guilleman!
In 1969, two years before Schally and Guilleman reported the isolation of GnRH, Knobil’s laboratory studying the ovariectomized rhesus monkey had found that LH was secreted in a pulsatile or episodic manner with a frequency in the agonadal condition of approximately 1 pulse per hour: they proposed that this pulsatile mode of LH secretion may be due to intermittent signals from the central nervous system that are relayed to the pituitary by an LH releasing factor (Dierschke, et al. 1970; Knobil 1992). Knobil’s laboratory went on to demonstrate in 1978 that intermittent GnRH stimulation of the pituitary was essential for sustained secretion of both LH and FSH (Belchetz, et al. 1978) (Figure 2). Interestingly, it was not until 1982, after Clarke and Cummins had developed a technique to sample portal blood in the un-sedated ewe, that the episodic nature of hypothalamic GnRH release was empirically demonstrated by these investigators (Clarke and Cummins 1982) (Figure 3). Together, the foregoing findings provided the foundation for the concept that pulsatile GnRH release was driven by a neural timing mechanism resident in the hypothalamus (Karsch 1980) (Pohl and Knobil 1982) that subsequently came to be known as the hypothalamic GnRH pulse generator. However, it was the later observation that the episodes of LH release in ovariectomized monkeys were tightly coupled to brief increases in multiunit electrophysiological activity (MUA) recorded in the mediobasal hypothalamus (MBH) (Wilson, et al. 1984), that led to the general acceptance of the notion of a GnRH pulse generator. This hypothalamic mechanisms drives “basal” or “tonic” gonadotropin secretion that, in the female, is responsible for folliculogenesis, maintenance of the corpus luteum and the synthesis of ovarian hormones and, in the male, for maintaining spermatogenesis and testosterone secretion (Plant and Marshall 2001; Zeleznik 2015).
Figure 2.
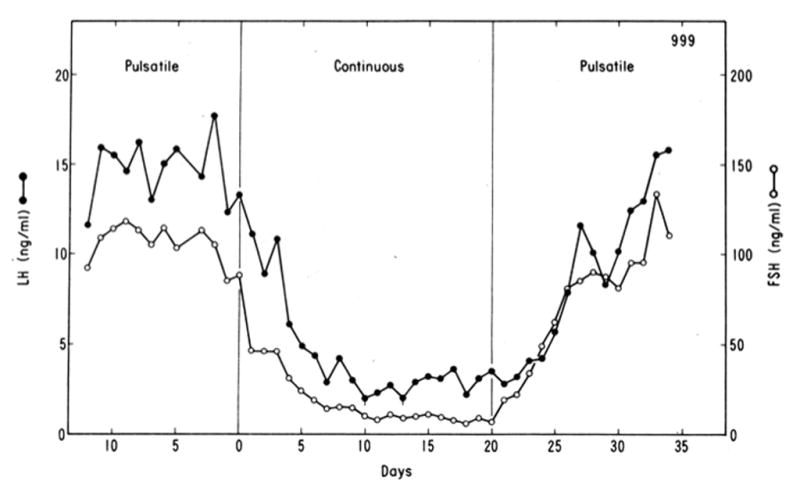
Demonstration of the need of intermittent GnRH stimulation of the pituitary for sustained secretion of LH (closed data points) and FSH (open data points). A monkey rendered hypogonadotropic by a hypothalamic lesion was treated initially with an intermittent iv infusion of GnRH (1 μg/min for 6 min every h). On day 0 the pulsatile regimen was terminated and replaced with a continuous GnRH infusion (1 μg/min). On day 20 the pulsatile mode of GnRH stimulation was re-instituted (reprinted from Belchetz et al, 1978).
Figure 3.

Relationship between pulsatile secretion of GnRH (open data points) into hypophysial portal blood and corresponding episodes of pituitary LH secretion (closed data points) into the systemic circulation in an ovariectomized ewe. Modified from Clarke and Cummins, 1982 – permission pending (Copyright 1982, The Endocrine Society).
Ovulation at the end of the follicular phase of the ovarian cycle is triggered by a massive and relatively prolonged discharge of gonadotropin, a mode of release termed “surge” secretion. The view held by Harris that ovulation was under neural control was, in part, based on the classical studies in 1950 by Everett and Sawyer (Everett and Sawyer 1950) indicating that, in the rat, a recurring daily neural signal was generated by the hypothalamus during a brief critical period in the light phase of the 24 h cycle, and which, on the day of proestrus, was responsible for eliciting the pre-ovulatory gonadotropin surge from the pituitary. The prediction that this neural signal was relayed to the pituitary by Harris’ humoral substance was confirmed when Fink and his colleagues in 1976 demonstrated a large surge in the concentration of GnRH in portal blood during the critical period of the proestrus rat (Sarkar, et al. 1976). The neural mechanism responsible for the preovulatory surge of GnRH that occurs over a period of several hours is now conceptualized as the GnRH surge generator. The precise relationship between the GnRH neurons mediating surge and pulsatile release of the decapeptide, however, remains to be determined.
Other “humoral” substances regulating gonadotroph function
The notion that regulation of anterior pituitary hormone secretion by humoral substances from the hypothalamus might be achieved by inhibiting the cells of the pars distalis was built into the Harris’ hypothesis (see above). In the context of the hypothalamo-pituitary-gonadal axis, evidence for such an inhibitory control system was obtained in studies of Japanese quail in 2000 reporting that a neuropeptide belonging to the RFamide related peptide (RFRP)3 family was found to be concentrated in the external zone of the median eminence of quail and, in vitro, inhibited LH secretion from the pituitary of these birds (Tsutsui, et al. 2000). RFRPs were known to exhibit a wide range of biological actions and Tsutsui and colleagues termed the peptide they had discovered in the quail brain, gonadotropin inhibitory hormone (GnIH). Subsequent studies have demonstrated that GnIH not only regulates gonadotropin secretion in avian species by a direct action on the pituitary but also by an indirect action at the hypothalamic level to modulate GnRH release (Tsutsui, et al. 2012). GnIH is not expressed in mammalian brain but two related peptides, RFRP-1 and RFRP-3, are encoded by a gene orthologous to that encoding avian GnIH (Tsutsui et al. 2012). Evidence that RFRPs serve a hypophysiotropic role in the control of the pituitary-gonadal axis of mammalian species in general, however, is tenuous (see Herbison, 2015).
The GnRH Neuron (birth, location, morphology, electrophysiology)
Two years after the structure of GnRH was announced, the first antibody to the peptide was reported (Barry, et al. 1973) and since then numerous studies of the immunohistochemical distribution of GnRH neurons in the developing and mature hypothalamus have been conducted (Herbison 2015). Based on careful examination of the embryonic mouse brain the groups of Pfaff and Wray in 1989 independently proposed that GnRH neurons are born in the olfactory placode and after entering the forebrain during early embryonic development migrate to the hypothalamus where several hundred of these cells are found diffusely distributed in the preoptic area (POA) and more caudal areas in the MBH (Schwanzel-Fukuda, et al. 1989) (Wray, et al. 1989). As may be expected from such an amazing journey through the brain, the neurobiology of GnRH neuron migration is complex and involves the interplay of guidance cues, adhesion molecules, growth factors and neurotransmitters (Wierman, et al. 2011).
In the adult hypothalamus, the typical GnRH neuron has two dendritic projections, which, as revealed by studies conducted with contemporary morphological techniques over the last 10 years, may extend remarkable distances (2–3 mm) from the cell body or perikaryon (Herbison 2015) (Figure 4). Not surprisingly, a principal target of hypothalamic GnRH neurons is the primary plexus of the hypophysial portal system in the median eminence. Interestingly, the latter projections combine characteristics of both dendrites and axons and have been termed “dendrons” by Herbison and his colleagues (Herde, et al. 2013). While considerable attention has been paid to the electrophysiology of the GnRH neuron and the underlying ion channels in the membrane of the cell body, a meaningful correlation between electrical activity and intermittent terminal release of GnRH, as classically demonstrated for oxytocin neurons, has yet to be obtained.
Figure 4.
A, B. Typical morphology of mouse GnRH neurons revealed by immunohistochemistry. C. Two GnRH neurons in sheep. D. A GnRH neuron from an adult mouse has been filled in situ with the dye, biocytin, in order to facilitate visualization of the entire length of the dendrites, which in the case of the lower process is seen to extend more than 500 μm. The two higher magnification insets on the right reveal a high density of dendritic spines. Reprinted with permission from Herbison (2015).
GnRH biosynthesis and action
GnRH is encoded by the GNRH1 gene, which was cloned by Seeburg and Adelman in 1984 (Seeburg and Adelman 1984). As pointed out by Herbison (2015), GnRH neurons maintain a high content of their peptide, and it is therefore unlikely that regulation of GNRH1 expression plays a critical role in moment to moment control of either GnRH surges or pulses. GnRH action on the pituitary gonadotroph is mediated by G-protein coupled receptors that signal via Gq and or G11 to activate phospholipase-C that leads to mobilization of Ca++ by inositol phosphate3 (McArdle 2015).
Neurobiology of the hypothalamic GnRH pulse generator
The concept of the hypothalamic GnRH pulse generator that emerged in the 1980s remained for more than 20 years in the realm of a “black box”. One school of thought proposed that pulsatility was intrinsic to the GnRH neurons themselves and that extensive inter-cellular mechanisms orchestrated synchrony within the network. A second school argued that non-GnRH neurons in the MBH target the GnRH neuronal network directly and drive intermittent release of GnRH (Herbison 2015). The later position has gained increasing support since 2003 when, in the context of the neuroendocrine axis governing the gonad, the power of molecular human genetics to identify novel signaling pathways was dramatically revealed. In that year, two independent groups, led by DeRoux and Seminara in Paris and Boston, respectively, reported that loss-of-function mutations in a G-protein coupled receptor, known as GPR54, was associated with hypogonadotropic hypogonadism in men and women (de Roux, et al. 2003; Seminara, et al. 2003). The cognate ligand of GPR54 turned out to be a 54 amino acid RFRP known as metastin or kisspeptin. The gene encoding kisspeptin, KISS1, had been of interest to cancer biologists since 1996 when it was found to be a tumor suppressor gene, but, quite remarkably, neither the gene nor the peptide was known to the neuroendocrine community in 2003. Since then, however, it has emerged in a volcanic manner that kisspeptin is an exceptionally potent GnRH secretagogue, GPR54 (the kisspeptin receptor, KISS1R) is expressed by GnRH neurons, kisspeptin neurons target the GnRH neuronal network, and one population of kisspeptin neurons is located in the arcuate nucleus (Herbison 2015), a periventricular structure located immediately above the median eminence that was originally argued by Knobil’s laboratory to be the primary structure mediating the hypothalamic control of gonadotropin secretion in the primate (Knobil 1980).
As a result of the foregoing findings, the idea that kisspeptin might be a component of the GnRH pulse generator, possible the output of the black box began to develop. The process was greatly reinforced by another signal observation resulting from the study of human genetics. In late 2009, Topoluglu and his colleagues reported that loss-of-function mutations in both ligand and receptor in another neuropeptide signaling pathway, namely that of neurokinin B, resulted in a phenotype similar to that described earlier for mutations of GPR54 (Topaloglu, et al. 2009). The significance of this finding was greatly amplified by the fact that Goodman and colleagues had 2 years earlier described the expression of neurokinin B in kisspeptin neurons in the arcuate nucleus of the ewe (Goodman, et al. 2007). Thus, in 2010 the community of reproductive neuroendocrinologists began to grapple with the fascinating observation that a subset of neurons in the arcuate nucleus co-express two peptides, each of which appeared to be obligatory for gonadal function in man. Goodman and his colleagues had also noted that the kisspepeptin/neurokinin B neurons in the arcuate nucleus of the ewe co-expressed a third peptide, dynorphin (Goodman et al. 2007), an endogenous opioid and one inhibitory to GnRH release. Based upon the first letter of each of these peptides, the acronym, KNDy, was coined to describe these neurons (Cheng, et al. 2010). In contrast to dynorphin, neurokinin B is generally stimulatory to GnRH release. Additional information obtained over the last decade has permitted the formulation of a model for GnRH pulse generation (Figure 5) that posits this hypothalamic timing mechanism is initiated in the KNDy neuronal network of the arcuate nucleus by a reciprocating interplay of stimulatory neurokinin B signals and inhibitory dynorphin inputs (Rance, et al. 2010) (Wakabayashi, et al. 2010) (Goodman 2015). As mentioned above, the output of the pulse generator is posited to be relayed from the arcuate nucleus to the GnRH neuronal network by release of kisspeptin from axonal terminals originating from KNDy neurons. According to this model, kisspeptin of arcuate nucleus origin should be viewed simply as a GnRH pulse generating peptide (Terasawa, et al. 2013).
Figure 5.
The KNDy neuron model of the GnRH pulse generator proposed by Lehman, Coolen and Goodman. KNDy neurons reside within the arcuate nucleus (dotted purple circle) and express kisspeptin (Ks, green), neurokinin B (NKB, purple) and dynorphin (dyn, red). The model proposes that pulse generation by the network of KNDy neurons in the arcuate nucleus is achieved by a poorly understood reciprocating interplay of stimulatory NKB and inhibitory dyn inputs and an unidentified interneurone (gray). The output of the pulse generator is relayed to the GnRH neuronal network (blue) by a brief kisspeptin signal that evokes a discharge of GnRH into the hypophysial-portal circulation (shown in the lower portion of the figure). Note that the phenotype of each terminal indicates biologically relevant peptide and not selective transport of that peptide to the terminal. Similarly, the triple colored KNDy neurons indicate co-expression of the 3 peptides and not location within the cell body. RKs, Ks receptor; RNKB, NKB receptor, RDYN, dyn receptor. Reprinted with permission from Goodman and Inskeep (2015).
It is probably no exaggeration to say that the discovery of the impact of loss-of-function mutations in GPR54 on the reproductive axis led to a profound and much needed revitalization to the study of GnRH neuroendocrinology, a field that had begun to stagnate in the 1990s.
Neuroendocrine control of ovarian cycles
In spontaneously ovulating species of mammal, the typical ovarian cycle is characterized by the secretion of relatively low basal or tonic levels of LH and FSH, which are interrupted approximately once every 4–5 days in rats and mice and once every 28 days in women, by a massive discharge or surge of gonadotropin which triggers ovulation. Tonic gonadotropin secretion during the follicular phase of the cycle, which drives folliculogenesis, is governed by a negative feedback loop in accordance with the Harris model (Figure 1). The principal ovarian component of the loop is estradiol secreted by the developing follicle(s), and it is now established that such feedback occurs at the pituitary level as well as at the hypothalamus. At the hypothalamic level, estradiol negative feedback appears to be exerted primarily by modulating the amplitude of pulsatile GnRH release (see Zeleznik and Plant 2015). Two nuclear estrogen receptors (ERs) exist, ERα and ERβ, and studies using transgenic mice null for these proteins indicate that ERα is the likely ER mediating the negative feedback action of estradiol. Interestingly, GnRH neurons do not appear to express ERα, as initially demonstrated by binding studies (Shivers, et al. 1983) and subsequently confirmed by immunohistochemistry; therefore estradiol negative feedback at the level of the hypothalamus must be mediated indirectly either by non-GnRH neurons or glia. Classical studies have indicated that the locus of the negative feedback action of estradiol is within the MBH, and, intuitively, it might be expected that ERα expressing KNDy neurons in the arcuate nucleus are the neuronal phenotype targeted. Intriguingly, however, a recent study by Levine and his colleagues employing transgenic mice null for ERα in kisspeptin neurons (not restricted to KNDy neurons) has failed to support this notion (Dubois, et al. 2015). On the other hand, Herbison’s group using genetically engineered adult female mice and adeno-associated virus injection into the arcuate nucleus to delete approximately 75% of ERα positive cells in this nucleus abolished chronic negative feedback action of estradiol on LH secretion (Yeo and Herbison 2014)4. It would seem reasonable to conclude that, in the later study, the majority of KNDy neurons would be null for ERα, and therefore the results of the two studies are difficult to reconcile. One possible explanation is that embryonic loss of ERα in kisspeptin neurons may have led to the development of compensatory feedback mechanisms.
During the later half of the follicular phase of the menstrual cycle, the secretion of FSH is suppressed to a greater degree than that of LH, and this differential pattern of gonadotropin release plays a pivotal role in the selection of the pre-ovulatory follicle (Zeleznik and Plant 2015). The mechanism underlying differential suppression of gonadotropin secretion at this stage of the menstrual cycle has not been extensively studied but appears to involve estradiol negative feedback action at the level of the pituitary to regulate the constitutive component of FSH release, which is greater than that of LH (see Zeleznik and Plant 2015).
In those species with a prolonged luteal phase, such as primates and sheep, progesterone secretion by the corpus luteum markedly retards the frequency of the GnRH pulse generator (see Goodman and Inskeep 2015; Zeleznik and Plant 2015). As there is little evidence for progesterone receptor (PR) expression by GnRH neurons, the suppressive feedback action of progesterone on GnRH release, like that of estradiol, is probably indirect. The finding that KNDy neurons in the ewe hypothalamus express PR suggests that these cells may be the target for progesterone’s negative feedback action. However, the impact of ablating PR selectively in KNDy neurons has yet to be examined probably because such genetic approaches have been largely limited to mice, which do not have prolonged luteal phases. It should also be noted that the physiological significance of progesterone’s ability to decelerate the frequency of pulsatile GnRH/LH remains unclear because corpus luteum function appears to be normal in GnRH deficient monkeys and women receiving invariant intermittent GnRH replacement (see Zeleznik and Plant 2015). In this regard, estradiol and inhibin (primarily inhibin A) are also secreted by the corpus luteum and both are capable of inhibiting gonadotropin secretion.
The importance of ovarian steroid feedback for the surge in LH secretion required for ovulation at the end of the follicular phase was suspected at the time Harris’ monograph was published. Although Harris was appreciative of these ideas (Harris 1969), he was primarily focused on the nature of the hypothalamic factor that triggered the ovulatory discharge of LH, and it was not until 1969 that Goding and his colleagues convincingly demonstrated the “positive” feedback action of estrogen on gonadotropin secretion by showing that administration of estradiol to the anestrous ewe elicited an LH surge comparable to that seen spontaneously at the end of the follicular phase during the breeding season (Goding, et al. 1969). By this time, Halasz5 and Gorski had shown by surgically isolating the POA from the MBH of the rat with a bayonet shaped “Halasz” knife that the more rostral area was essential for ovulation in this species (Halasz and Gorski 1967; Szentagothai J 1968). Together, these findings suggested that a major site of the positive feedback action of estradiol in the rodent was in the POA, and Goodman in 1978 went on to verify this idea by demonstrating that implants of crystalline estradiol in the POA of the rat were more effective at eliciting LH surges than those implanted in the more caudal region of the hypothalamus (Goodman 1978). So after almost 30 years from the time that Everitt and Sawyer had demonstrated that the generation of a daily neural signal for ovulation in the rat was tightly coupled to the 24 h light dark cycle (Everett and Sawyer 1950), a comprehensive model for the control of ovulation in the rat had finally begun to emerge. It should also be pointed out that, at this time, evidence was accumulating to indicate that this model might not be applicable to other species. Interestingly, the model for the rat ovarian cycle remained essential unchanged until the recognition in 2003 of the significance of kisspeptin in the regulation of GnRH secretion. During the subsequent 5 years and largely as a result of work by Steiner’s laboratory it became apparent that a second hypothalamic population of kisspeptin neurons are located in the anteroventral periventricular nucleus (AVPV) of the POA and that this rostral population is a major site for the positive feedback action of estradiol in rodents (Oakley, et al. 2009). As with the negative feedback action of estradiol, knockout studies of ER in mice indicate that ERα mediates the positive feedback of the steroid (Dubois et al. 2015).
In the mid-1970s, Knobil’s group threw much of the field of reproductive neuroendocrinology into a state of partial shock when they demonstrated that ovulation occurred in the monkey after neural connections between the POA and MBH were severed using a Halasz knife (Krey, et al. 1975). The furor continued when this group proposed that the role of hypothalamic GnRH release in dictating the menstrual cycle was permissive (Knobil, et al. 1980). In other words, the only hypothalamic input that was needed for ovarian cycles and ovulation in highly evolved primates was an invariant intermittent stimulation of the pituitary by GnRH and that the characteristic cyclic pattern of gonadotropin secretion was dictated solely by the negative and positive feedback actions of estradiol directly at the pituitary: a positive feedback action of estradiol in the POA and a GnRH surge was not required. Since then, the most compelling evidence in support of this hypothesis has been obtained from GnRH deficient women, such as those with Kallmann syndrome, where replacement treatment with an invariant intermittent infusion of “physiological” pulse doses of GnRH will drive cycles and ovulation (see Zeleznik and Plant 2015). Moreover, indirect evidence indicates that spontaneous ovulation in normal women likely occurs in the absence of a GnRH surge (see Zeleznik and Plant 2015). Whatever the final resolution of this important comparative question, it appears that ovulation in the human female is largely emancipated from control by the POA (Plant 2012).
Neuroendocrine control of testicular function
The study of the neuroendocrine control of the testis in the post Harris era has continued to play second fiddle to that of the ovary. As with early studies, upon which the foundations of reproductive neuroendocrinology were laid, ovulation and the pre-ovulatory LH surge, have provided a robust read-out of hypothalamic activation and one that could by easily harnessed to indirectly explore the neurobiological mechanism controlling GnRH secretion. As will be discussed later, the GnRH surge generator in the rodent is decommissioned during perinatal development, and the hypothalamic control of the testis can be accounted entirely by GnRH pulse generation. Since fundamental sex differences in the control of this mode of GnRH release have yet to be identified, it is to be anticipated that what we understand of GnRH pulse generation in the female will be largely translatable to the male. With regard to the negative feedback control of testicular function, the concept of an aqueous testicular feedback signal “inhibin” was proposed more than 20 years before the publication of Harris’ monograph. It was not until 1985, however, that the nature of inhibin was independently revealed by four groups (Vale, et al. 1988). Interestingly, follicular fluid not testicular tissue/fluid was used for isolation of the inhibins, which are heteromeric peptides consisting of an α-subunit and one of two β-subunits (Vale et al. 1988). Studies of the male monkey have provided the most convincing evidence to date for a major role of testicular inhibin B (αβB dimer) in regulating the secretion of FSH by a direct negative feedback action at the level of the pituitary (Majumdar, et al. 1995).
Neuroendocrine control of puberty
Although Harris’ proposal in 1955 that puberty is triggered by a hypothalamic stimulus that is transmitted via the hypophysial portal vessels to the pituitary is now well established, the mechanism underlying the timing of this critical development event in our own species remains an intriguing mystery. Major conceptual advances, however, have been achieved. Studies in the 1980s of pituitary and plasma gonadotropin content in the human and sheep fetus by Grumbach and his colleagues (Clark, et al. 1984; Sklar, et al. 1981) and of circulating testosterone and LH levels in infantile boys and monkeys by others (Plant 1980) (Forest, et al. 1974; Waldhauser, et al. 1981) led to the deduction that 1) GnRH pulse generation in these species develops in the fetus shortly after GnRH neurons complete their embryonic migration from the olfactory placode to the hypothalamus, and 2) robust pulsatile GnRH and gonadotropin release is manifest during infancy in highly evolved primates. Puberty, however, is not initiated during infancy because at this stage of development the somatic cells of the gonads that underpin gametogenesis are unable to respond fully to such gonadotropin stimulation. Although the ability of the prepubertal gonad to respond to gonadotropin stimulation is acquired during childhood, by then a hypogonadotropic state is in place as a result of the suppression of pulsatile GnRH release thereby guaranteeing the continued quiescence of the gonad in boys and girls. Interestingly, studies of the monkey indicate that the GnRH neuronal network of the juvenile primate, like the pituitary and gonad at this stage of development, is not limiting to the onset of puberty. This view is based on the finding that in the monkey intermittent neurochemical stimulation of the juvenile hypothalamus with a glutamate receptor agonist will lead with surprising ease to an adult like pattern of pulsatile GnRH release and precocious puberty (Figure 6) (Plant, et al. 1989). Moreover, compelling evidence is now at hand indicating that the proximal stimulus responsible for the activation of robust GnRH pulsatility at the onset of spontaneous puberty is indeed an intermittent release of kisspeptin that is generated by the re-awakening of the GnRH pulse generator in the arcuate nucleus (Terasawa et al. 2013). The mechanisms that turn the GnRH pulse generator off during infancy, maintain it in a state of suspended animation during juvenile development, and reawaken it at the termination of juvenile development are poorly understood (Plant 2015) and provide a major challenge for the future.
Figure 6.

Chronic intermittent neurochemical stimulation of juvenile male monkeys with N-methyl DL aspartate (NMDA) readily induces a precocious pubertal pattern of pulsatile GnRH release as reflected by the emergence of corresponding discharges of LH (open data points) and testicular testosterone (closed data points) secretion. Testicular and motile epididymal sperm were typically observed after 16–26 weeks of NMDA stimulation. Means±SE (N=4) are shown. Arrows indicate time of iv injections of NMDA. Reproduced from Plant et al (1989) “Copyright (1989) National Academy of Sciences, U.S.A.”
Perinatal programming of the GnRH surge generator
With regard to sexual differentiation of the hypothalamus, Harris stated in 1955, “In view of the fact that Pfeiffer (1936) showed prepubertal testis grafts in female rats produce a constant oestrous state after puberty, and that ovaries grafted into adult male rats castrated at birth do undergo cyclic changes, it seems that some neural structure in the male animal becomes differentiated and fixed in its function under the influence of androgens in early life.”: a position shared by Everett, Sawyer and Markee (1949) (Everett, et al. 1949). Examination of this hypothesis in the post-Harris era has proceeded somewhat in parallel with development of the model for the neural control of ovulation in the rodent (see above), and the target of testicular testosterone action to defeminize the rodent brain is considered to reside within the POA and to involve a negative impact of testicular androgen on the development of kisspeptin neurons in the AVPV (Oakley et al. 2009). An interesting feature of this perinatal decommissioning of the GnRH surge mechanism by testicular testosterone is that the action of the steroid in target neurons within the POA appears to be mediated by ER signaling after intracellular aromatization of testosterone to estradiol (Gonzalez-Martinez, et al. 2008).
One important comparative aspect of perinatal programming of the GnRH surge system in spontaneous ovulators that has emerged since the time of Harris is the recognition that, in the monkey and probably in man, the neural mechanism underlying the pre-ovulatory LH surge is not decommissioned by the testis during early development. This was first demonstrated by Knobil’s laboratory by the finding that unequivocal estrogen-induced LH surges were readily elicited in adult male monkeys castrated postpubertally and implanted with estradiol containing Silastic capsules that produced follicular phase levels of the steroid in the agonadal situation (Karsch, et al. 1973). A more sensational demonstration followed when Spies’ laboratory reported that ovarian cycles and ovulation unfolded in an adult male castrate bearing a sc ovarian graft (Figure 7) (Norman and Spies 1986).
Figure 7.
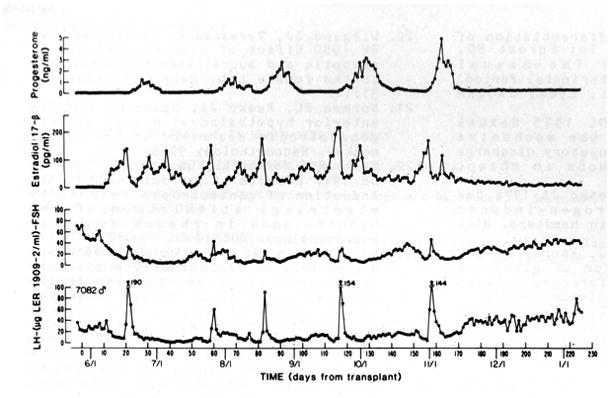
Ovarian tissue transplanted sc to the abdomen of an adult male rhesus monkey (#7082) castrated post-pubertally and receiving anti rejection therapy with cyclosporine A exhibits regular cycles of folliculogenesis and ovulation followed by a normal luteal phase. Numbers on the peaks in LH indicate maximum concentration of the gonadotropin on that day. Modified from Norman and Spies (1986) with permission (Copyright 1986, The Endocrine Society).
Modulation of GnRH pulsatility
At the time Harris wrote his monograph in 1955, he discussed the impact of food, temperature, light, social factors and emotional states (in women) on ovarian cycles and fertility, and argued that these modulators of gonadal function are registered by the central nervous system, which then relays the respective exteroceptive or interoceptive cue to the gonad by controlling gonadotropin secretion from the pituitary. Although considerable advances have been made in our understanding of the neural and neuroendocrine mechanisms involved in such modulation, a complete review of these fall beyond the scope of this chapter. Instead, two examples that are of particular conceptual significance are discussed.
The first concerns the relationship between somatic and sexual development, and specifically the question of how information on growth is relayed to the reproductive axis. In 1963 Kennedy and Mitra (Kennedy and Mitra 1963) proposed that, in the rat, body weight was an initiating factor in the onset of puberty, and inspired by this idea, Frisch and Revelle examined the relationship between body weight and the timing of menarche in girls. The observations of the latter workers led them to propose in 1970 (Frisch and Revelle 1970) “that attainment of body weight in the critical range causes a change in metabolic rate, which, in turn, is responsible for an increase in hypothalamic drive to the pituitary gonadotroph.” An extension of this idea would be to propose that a “metabometer” in the hypothalamus tracks information on metabolism and sends a cue to the GnRH pulse generator when a pubertal metabolic state develops. While this hypothesis has not been systematically examined, interest in the idea of Frisch was re-awakened in 1994 by the cloning of the obese gene encoding leptin (Zhang, et al. 1994): an adipocyte hormone that plays a major role in regulating appetite and food intake. Although it is now generally recognized that leptin is not the trigger timing the pubertal re-awakening of the GnRH pulse generator, this hormone is essential, in a permissive sense, for robust GnRH pulsatility in general, and therefore for both pubertal and adult gonadal function. The latter view is best supported by the finding that in young children with leptin deficiency initiation of treatment with recombinant leptin does not induce puberty immediately but rather only after appropriate somatic development has been attained following prolonged exposure to the hormone (Farooqi and O’Rahilly 2014). Although KNDy neurons in the arcuate nucleus of the hypothalamus were initially thought to be the target of leptin’s hypothalamic action to modulate the pituitary-gonadal axis, more recent studies using transgenic mice suggest that the neurobiology underlying the action of leptin to modulate GnRH pulsatility may be considerably more complex (Donato, et al. 2011).
The second modulator of GnRH pulsatility to be discussed is photoperiod; a major factor determining annual changes in gonadal function in mammalian species that breed seasonally. Of all the areas of reproductive neuroendocrinology that interested Harris, the study of season in the post-Harris era has arguably uncovered the most novel of insights into neuroendocrine control systems governing the hypothalamic-pituitary-gonadal axis. The story begins with the recognition in the mid-1960s of the importance of the pineal gland in regulating seasonal changes in gonadal function in the Syrian hamster. Melatonin secreted by the pineal is the major signal relaying information on duration of daylight to the hypothalamus, and a major site of action of melatonin in the hypothalamus is the pars tuberalis, a portion of the anterior pituitary that forms a funnel like structure around the pituitary stalk and median eminence. The initial suggestion that the pars tuberalis was instrumental in mediating seasonal changes in anterior pituitary function came from a study by Lincoln and Clark employing an experimental preparation in which the pituitary of sheep was disconnected from neurovascular control by the hypothalamus (Lincoln and Clarke 1994). Such hypothalamo-pituitary disconnected animals continued to exhibit a seasonal rhythm in prolactin secretion. Importantly, gonadotropin levels on the other hand were routinely low due to the blockade of the GnRH signal by the surgical procedure. At about the same time, Karsch’s laboratory studying the ewe was systematically exploring an earlier idea of others that thyroid hormone was required for seasonal changes in hypothalamic-pituitary function: their work demonstrated that thyroid hormone was permissive for the transition to anovulation during the long days of spring, but not needed for return of reproductive function at the start of the fall breeding season (Karsch, et al. 1995). Insight into the neurobiology underlying the interaction between thyroid hormone and the seasonal melatonin signal came initially from the finding in Japanese quail (a long-day breeder), that expression in the MBH of a thyroid hormone metabolizing enzyme (type 2 diodinase) that increases the local concentration of triiodothyronine, the active cellular metabolite of thyroid hormone, was upregulated by exposure to long days (Yoshimura, et al. 2003). Interestingly, long days also result in an increase in hypothalamic thyroid hormone activity in short day breeders such as sheep. In both cases, the link between melatonin action in the pars tuberalis and deiodinase activity in the MBH appears to be provided by increased synthesis of thyroid stimulating hormone in the pars tuberalis triggered by long days (Figure 8). How low day induced increases in thyroid hormone activity in the MBH either terminate or reactivate pulsatile GnRH release in long and short day breeders, respectively, remains to be established, although KNDy neurons and RFRP expressing neurons have been implicated. As pointed out by Hazelrigg and his colleagues (Hanon, et al. 2008), information in this intriguing pathway moves in the opposite direction to that in Harris’ model (Figure 1), albeit without the need of the hypophysial portal circulation, and therefore is unlikely to have been envisioned by Harris. An excellent review of this fascinating control system has recently been published by Hazlerigg and Simonneauz (2015).
Figure 8.

A model for the seasonal control of pulsatile GnRH release. The duration of the photoperiod is relayed by melatonin to melatonin receptors (MT1) in the thyrotrophs of the pars tuberalis (PT), and further relayed by thyrotrophin (TSH) to the mediobasal hypothalamus, where the arcuate nucleus KNDy neurons are located. TSH upregulates the expression of the genes encoding deiodinase 2 and 3 (DIO2 and DIO3), in specialized ependymal cells (tanycytes) lining the base of the third ventricle (3v). The diodinase enzymes convert thyroid hormone (T4) into the active metabolite, tri-iodothyronine (T3), and the increase in thyroid hormone activity dictates the level of GnRH pulsatility, which in turn governs the gonadotrophin output from the gonadotrophs in the pars distalis (PD). TH is considered to reach the MBH via the 3v and/or from brain capillaries (Cp). Reprinted with permission from Hazelrigg and Simonneaux (2015).
Bench to bedside
The isolation and characterization of GnRH, together with the demonstration that sustained stimulation of gonadotropin secretion may be achieved with intermittent stimulation of the pituitary whereas continuous exposure of the gland to the peptide leads to desensitization and thus suppression of gonadotropin secretion, provides the conceptual framework for the current therapeutic uses of GnRH receptor analogs. As comprehensively reviewed by Millar and Newton (Millar and Newton 2013), GnRH agonists and antagonist are currently used widely to treat gonadal steroid dependent cancers, and disorders of reproductive development including GnRH dependent precocious puberty. These agents are also used extensively in fertility clinics where controlled cycles of follicular development and oocyte maturation are required for egg retrieval and in-vitro fertilization. Millar and Newton also speculate that in the future kisspeptin and neurokinin B analogs may offer additional or improved therapeutic interventions to modulate LH and FSH release. In this regard, a recent report describes successful oocyte maturation following an LH surge induced by a single sc injection of kisspeptin administered at the end of the follicular phase of a controlled cycle of ovarian stimulation (Jayasena, et al. 2014). It remains to be determined, however, whether kisspeptin induced LH release has any advantages over GnRH induced LH release for oocyte maturation in such a clinical setting.
The future
Harris’ model of the control of gonadal function has, over the last 60 years, served as a solid framework for those studying mechanisms that govern how the brain regulates the pituitary–gonadal axis: it has stood the test of time and revisions to the paradigm, while quite remarkable, have been primarily those at the level of cellular and molecular detail, rather than those with major conceptual implications. Perhaps an exception to this generalization is the further recognition that during evolution different mammalian species have co-opted different neuroendocrine control systems, or perhaps more precisely utilized a set of neuroendocrine controls to variable degrees, to achieve regulation of the gonad. The implication of the latter view to the future of the field in general is that diversity in the selection of animal models is beneficial, while the interrogation of problems that at the time appear immediately translatable to human health and well being to the exclusion of others is not. With regard to the issue of diversity in selecting animal models, the recent introduction of a technology known as CRISPR-Cas9, which is potentially capable of being used to edit the genome of any mammalian species (Hsu, et al. 2014), is likely to profoundly enhance the value of animal models hitherto considered to be genetically intractable. The power of contemporary molecular human genetics/genomics has already resulted in signal surprises and inevitably will lead to further major advances in our understanding of neural signaling pathways involved in regulating hypothalamic GnRH release. The application of global analyses of hypothalamic gene expression, and thereby the identification of gene networks, associated with neuroendocrine mechanisms governing GnRH release has been introduced by Ojeda’s laboratory (Lomniczi, et al. 2013), and it is likely that this systems biology approach will see wider use as high throughput sequencing techniques become less expensive.
Major questions remain to be addressed and, in the opinion of the writer, the two most important concern the nature of the neurobiological mechanisms that underlie the generation of the pulsatile mode of GnRH release and the mystery of human puberty. The two are related since human puberty is triggered by a re-awakening of GnRH pulse generation. In this regard, optogenetics (Deisseroth 2011), which allows the investigator to selectively activate specific signaling pathways in the brain, would appear to be particularly suitable for interrogating the KNDy neuron model of GnRH pulse generation. Examination of this model would also be greatly aided by a complete “wiring” diagram of the arcuate nucleus, and such a goal is on the horizon as technologies underlying connectomics (Eberle, et al. 2015) become more readily available. Finally, as with many other fields, it will also be of interest to witness over the next decade or so to what extent epigenetic mechanisms contribute to transmission of inheritable traits in hypothalamic control systems governing GnRH release.
Acknowledgments
The author is most grateful to Dr. Robert L Goodman, Department of Physiology and Pharmacology, West Virginia University, for kindly reading the manuscript and for his insightful comments and suggestions. Work conducted in the author’s laboratory was supported by the National Institutes of Health (Grants HD08610, HD13254 and HD16851).
Funding: Original research is not included in this review!
Footnotes
Recounted by Nicholas Wade in The Nobel Duel (Anchor Press/DoubleDay, 1981) and in a series of three articles published in Science in April/May, 1978.
In this review GnRH1 (mammalian GnRH) will be referred to as GnRH.
The carboxyl terminal of these peptides is preceded by the amino acid sequence, LP (LorQ)RFamide (Tsutsui et al. 2012).
Acute suppression of LH by estradiol, however, was preserved.
Halasz developed the use of a surgical knife to isolate the hypophysiotropic area (MBH) from more rostral areas of the rat hypothalamus at Pecs University Medical School. The group at Pecs was one of the major World centers studying the hypothalamic control of the anterior pituitary at the time of Harris, and is perhaps most famous for its studies of hypothalamic pathways and regions (including those that are sensitive to target hormones from the gonad, adrenal and thyroid) underlying the feedback control of anterior pituitary function (Szentagothai et al. 1968).
Declaration of interest: I declare that there is no conflict of interest that could be perceived as prejudicing the impartiality of my interpretation of the research reported in this review.
References
- Amoss M, Burgus R, Blackwell R, Vale W, Fellows R, Guillemin R. Purification, amino acid composition and N-terminus of the hypothalamic luteinizing hormone releasing factor (LRF) of ovine origin. Biochem Biophys Res Commun. 1971;44:205–210. doi: 10.1016/s0006-291x(71)80179-1. [DOI] [PubMed] [Google Scholar]
- Barry J, Dubois MP, Poulain P. LRF producing cells of the mammalian hypothalamus. A fluorescent antibody study. Z Zellforsch Mikrosk Anat. 1973;146:351–366. doi: 10.1007/BF02346227. [DOI] [PubMed] [Google Scholar]
- Belchetz PE, Plant TM, Nakai Y, Keogh EJ, Knobil E. Hypophysial responses to continuous and intermittent delivery of hypopthalamic gonadotropin-releasing hormone. Science. 1978;202:631–633. doi: 10.1126/science.100883. [DOI] [PubMed] [Google Scholar]
- Cheng G, Coolen LM, Padmanabhan V, Goodman RL, Lehman MN. The kisspeptin/neurokinin B/dynorphin (KNDy) cell population of the arcuate nucleus: sex differences and effects of prenatal testosterone in sheep. Endocrinology. 2010;151:301–311. doi: 10.1210/en.2009-0541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clark SJ, Ellis N, Styne DM, Gluckman PD, Kaplan SL, Grumbach MM. Hormone ontogeny in the ovine fetus. XVII. Demonstration of pulsatile luteinizing hormone secretion by the fetal pituitary gland. Endocrinology. 1984;115:1774–1779. doi: 10.1210/endo-115-5-1774. [DOI] [PubMed] [Google Scholar]
- Clarke IJ, Cummins JT. The temporal relationship between gonadotropin releasing hormone (GnRH) and luteinizing hormone (LH) secretion in ovariectomized ewes. Endocrinology. 1982;111:1737–1739. doi: 10.1210/endo-111-5-1737. [DOI] [PubMed] [Google Scholar]
- de Roux N, Genin E, Carel J-C, Matsuda F, Chaussain J-L, Milgrom E. Hypogonadotropic hypogonadism due to loss of function of the KiSS1-derived peptide receptor GPR54. Proc Natl Acad Sci U S A. 2003;100:10972–10976. doi: 10.1073/pnas.1834399100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deisseroth K. Optogenetics. Nat Methods. 2011;8:26–29. doi: 10.1038/nmeth.f.324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dierschke DJ, Bhattacharya AN, Atkinson LE, Knobil E. Circhoral oscillations of plasma LH levels in the ovariectomized rhesus monkey. Endocrinology. 1970;87:850–853. doi: 10.1210/endo-87-5-850. [DOI] [PubMed] [Google Scholar]
- Donato J, Jr, Cravo RM, Frazao R, Gautron L, Scott MM, Lachey J, Castro IA, Margatho LO, Lee S, Lee C, et al. Leptin’s effect on puberty in mice is relayed by the ventral premammillary nucleus and does not require signaling in Kiss1 neurons. J Clin Invest. 2011;121:355–368. doi: 10.1172/JCI45106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dubois SL, Acosta-Martinez M, DeJoseph MR, Wolfe A, Radovick S, Boehm U, Urban JH, Levine JE. Positive, But Not Negative Feedback Actions of Estradiol in Adult Female Mice Require Estrogen Receptor alpha in Kisspeptin Neurons. Endocrinology. 2015;156:1111–1120. doi: 10.1210/en.2014-1851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eberle AL, Selchow O, Thaler M, Zeidler D, Kirmse R. Mission (im)possible - mapping the brain becomes a reality. Microscopy (Oxf) 2015;64:45–55. doi: 10.1093/jmicro/dfu104. [DOI] [PubMed] [Google Scholar]
- Everett JW, Sawyer CH. A 24-hour periodicity in the “LH-release apparatus” of female rats, disclosed by barbiturate sedation. Endocrinology. 1950;47:198–218. doi: 10.1210/endo-47-3-198. [DOI] [PubMed] [Google Scholar]
- Everett JW, Sawyer CH, Markee JE. A neurogenic timing factor in control of the ovulatory discharge of luteinizing hormone in the cyclic rat. Endocrinology. 1949;44:234–250. doi: 10.1210/endo-44-3-234. [DOI] [PubMed] [Google Scholar]
- Farooqi IS, O’Rahilly S. 20 years of leptin: human disorders of leptin action. J Endocrinol. 2014;223:T63–70. doi: 10.1530/JOE-14-0480. [DOI] [PubMed] [Google Scholar]
- Forest MG, Sizonenko PC, Cathiard AM, Betrand J. Hypophysogonadal function in humans during the first year of life. I. Evidence for testicular activity in early infancy. J Clin Invest. 1974;53:819–828. doi: 10.1172/JCI107621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frisch RE, Revelle R. Height and weight at menarche and a hypothesis of critical body weights and adolescent events. Science. 1970;169:397–399. doi: 10.1126/science.169.3943.397. [DOI] [PubMed] [Google Scholar]
- Goding JR, Catt KJ, Brown JM, Kaltenbach CC, Cumming IA, Mole BJ. Radioimmunoassay for ovine luteinizing hormone. Secretion of luteinizing hormone during estrus and following estrogen administration in sheep. Endocrinology. 1969;85:133–142. doi: 10.1210/endo-85-1-133. [DOI] [PubMed] [Google Scholar]
- Gonzalez-Martinez D, De Mees C, Douhard Q, Szpirer C, Bakker J. Absence of gonadotropin-releasing hormone 1 and Kiss1 activation in alpha-fetoprotein knockout mice: prenatal estrogens defeminize the potential to show preovulatory luteinizing hormone surges. Endocrinology. 2008;149:2333–2340. doi: 10.1210/en.2007-1422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goodman RL. The site of the positive feedback action of estradiol in the rat. Endocrinology. 1978;102:151–159. doi: 10.1210/endo-102-1-151. [DOI] [PubMed] [Google Scholar]
- Goodman RL, Inskeep EK. Control of the Ovarian Cycle of the Sheep. In: Plant TM, Zeleznik AJ, editors. Knobil and Neill’s Physiology of Reproduction. 4. San Diego, CA, USA: Elsevier Inc; 2015. pp. 1259–1305. [Google Scholar]
- Goodman RL, Lehman MN, Smith JT, Coolen LM, de Oliveira CV, Jafarzadehshirazi MR, Pereira A, Iqbal J, Caraty A, Ciofi P, et al. Kisspeptin neurons in the arcuate nucleus of the ewe express both dynorphin A and neurokinin B. Endocrinology. 2007;148:5752–5760. doi: 10.1210/en.2007-0961. [DOI] [PubMed] [Google Scholar]
- Halasz B, Gorski RA. Gonadotrophic hormone secretion in female rats after partial or total interruption of neural afferents to the medial basal hypothalamus. Endocrinology. 1967;80:608–622. doi: 10.1210/endo-80-4-608. [DOI] [PubMed] [Google Scholar]
- Hanon EA, Lincoln GA, Fustin JM, Dardente H, Masson-Pevet M, Morgan PJ, Hazlerigg DG. Ancestral TSH mechanism signals summer in a photoperiodic mammal. Curr Biol. 2008;18:1147–1152. doi: 10.1016/j.cub.2008.06.076. [DOI] [PubMed] [Google Scholar]
- Harris GW. Neural control of the Pituitary Gland. London: Edward Arnold; 1955. [Google Scholar]
- Harris GW. Ovulation. Am J Obstet Gynecol. 1969;105:659–669. doi: 10.1016/0002-9378(69)90002-7. [DOI] [PubMed] [Google Scholar]
- Hazlerigg D, Simonneaux V. Seasonal Reproduction in Mammals. In: Plant TM, Zeleznik AJ, editors. Knobil and Neill’s Physiology of Reproduction. 4. San Diego, CA, USA: Elsevier Inc; 2015. pp. 1575–1604. [Google Scholar]
- Herbison AE. Physiology of the Adult Gonadotropin-Releasing Hormone Neuronal Network. In: Plant TM, Zeleznik AJ, editors. Knobil and Neill’s Physiology of Reproduction. 4. San Diego, CA, USA: Elsevier Inc; 2015. pp. 399–467. [Google Scholar]
- Herde MK, Iremonger KJ, Constantin S, Herbison AE. GnRH neurons elaborate a long-range projection with shared axonal and dendritic functions. J Neurosci. 2013;33:12689–12697. doi: 10.1523/JNEUROSCI.0579-13.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hsu PD, Lander ES, Zhang F. Development and applications of CRISPR-Cas9 for genome engineering. Cell. 2014;157:1262–1278. doi: 10.1016/j.cell.2014.05.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jayasena CN, Abbara A, Comninos AN, Nijher GM, Christopoulos G, Narayanaswamy S, Izzi-Engbeaya C, Sridharan M, Mason AJ, Warwick J, et al. Kisspeptin-54 triggers egg maturation in women undergoing in vitro fertilization. J Clin Invest. 2014;124:3667–3677. doi: 10.1172/JCI75730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karsch FJ. Twenty-fifth Annual Bowditch Lecture. Seasonal reproduction: a sage of reversible fertility. Physiologist. 1980;23:29–38. [PubMed] [Google Scholar]
- Karsch FJ, Dahl GE, Hachigian TM, Thrun LA. Involvement of thyroid hormones in seasonal reproduction. J Reprod Fertil Suppl. 1995;49:409–422. [PubMed] [Google Scholar]
- Karsch FJ, Dierschke DJ, Knobil E. Sexual differentiation of pituitary function: apparent difference bewteen primates and rodents. Science. 1973;179:484–486. doi: 10.1126/science.179.4072.484. [DOI] [PubMed] [Google Scholar]
- Kennedy GC, Mitra J. Body weight and food intake as initiating factors for puberty in the rat. J Physiol. 1963;166:408–418. doi: 10.1113/jphysiol.1963.sp007112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knobil E. The neuroendocrine control of the menstrual cycle. Recent Prog Horm Res. 1980;36:53–88. doi: 10.1016/b978-0-12-571136-4.50008-5. [DOI] [PubMed] [Google Scholar]
- Knobil E. Remembrance: the discovery of the hypothalamic gonadotropin-releasing hormone pulse generator and of its physiological significance. Endocrinology. 1992;131:1005–1006. doi: 10.1210/endo.131.3.1505445. [DOI] [PubMed] [Google Scholar]
- Knobil E, Plant TM, Wildt L, Belchetz PE, Marshall G. Control of the rhesus monkey menstrual cycle: permissive role of hypothalamic gonadotropin-releasing hormone. Science. 1980;207:1371–1373. doi: 10.1126/science.6766566. [DOI] [PubMed] [Google Scholar]
- Krey LC, Butler WR, Knobil E. Surgical disconnection of the medial basal hypothalamus and pituitary function in the rhesus monkey. I. Gonadotropin secretion. Endocrinology. 1975;96:1073–1087. doi: 10.1210/endo-96-5-1073. [DOI] [PubMed] [Google Scholar]
- Lincoln GA, Clarke IJ. Photoperiodically-induced cycles in the secretion of prolactin in hypothalamo-pituitary disconnected rams: evidence for translation of the melatonin signal in the pituitary gland. J Neuroendocrinol. 1994;6:251–260. doi: 10.1111/j.1365-2826.1994.tb00580.x. [DOI] [PubMed] [Google Scholar]
- Lomniczi A, Wright H, Castellano JM, Sonmez K, Ojeda SR. A system biology approach to identify regulatory pathways underlying the neuroendocrine control of female puberty in rats and nonhuman primates. Horm Behav. 2013;64:175–186. doi: 10.1016/j.yhbeh.2012.09.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Majumdar SS, Mikuma N, Ishwad PC, Winters SJ, Attardi BJ, Perera AD, Plant TM. Replacement with recombinant human inhibin immediately after orchidectomy in the hypophysiotropically clamped male rhesus monkey (Macaca mulatta) maintains follicle-stimulating hormone (FSH) secretion and FSH beta messenger ribonucleic acid levels at precastration values. Endocrinology. 1995;136:1969–1977. doi: 10.1210/endo.136.5.7720645. [DOI] [PubMed] [Google Scholar]
- Matsuo H, Baba Y, Nair RM, Arimura A, Schally AV. Structure of the porcine LH- and FSH-releasing hormone. I. The proposed amino acid sequence. Biochem Biophys Res Commun. 1971;43:1334–1339. doi: 10.1016/s0006-291x(71)80019-0. [DOI] [PubMed] [Google Scholar]
- McArdle CA, Roberson MS. Gonadotropes and Gonadotropin-Releasing Hormone Signaling. In: Plant TM, Zeleznik AJ, editors. Knobil and Neill’s Physiology of Reproduction. 4. San Diego, CA, USA: Elsevier Inc; 2015. pp. 335–397. [Google Scholar]
- Millar RP, Newton CL. Current and future applications of GnRH, kisspeptin and neurokinin B analogues. Nat Rev Endocrinol. 2013;9:451–466. doi: 10.1038/nrendo.2013.120. [DOI] [PubMed] [Google Scholar]
- Norman RL, Spies HG. Cyclic ovarian function in a male macaque: additional evidence for a lack of sexual differentiation in the physiological mechanisms that regulate the cyclic release of gonadotropins in primates. Endocrinology. 1986;118:2608–2610. doi: 10.1210/endo-118-6-2608. [DOI] [PubMed] [Google Scholar]
- Oakley AE, Clifton DK, Steiner RA. Kisspeptin signaling in the brain. Endocr Rev. 2009;30:713–743. doi: 10.1210/er.2009-0005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Plant TM. The effects of neonatal orchidectomy on the developmental pattern of gonadotropin secretion in the male rhesus monkey (Macaca mulatta) Endocrinology. 1980;106:1451–1454. doi: 10.1210/endo-106-5-1451. [DOI] [PubMed] [Google Scholar]
- Plant TM. A comparison of the neuroendocrine mechanisms underlying the initiation of the preovulatory LH surge in the human, Old World monkey and rodent. Front Neuroendocrinol. 2012;33:160–168. doi: 10.1016/j.yfrne.2012.02.002. [DOI] [PubMed] [Google Scholar]
- Plant TM, Gay VL, Marshall GR, Arslan M. Puberty in monkeys is triggered by chemical stimulation of the hypothalamus. Proc Natl Acad Sci U S A. 1989;86:2506–2510. doi: 10.1073/pnas.86.7.2506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Plant TM, Marshall GR. The functional significance of FSH in spermatogenesis and the control of its secretion in male primates. Endocr Rev. 2001;22:764–786. doi: 10.1210/edrv.22.6.0446. [DOI] [PubMed] [Google Scholar]
- Plant TM, Terasawa E, Witchel SF. Puberty in non-human primates and man. In: Plant TM, Zeleznik AJ, editors. Knobil and Neill’s Physiology of Reproduction. 4. San Diego, CA, USA: Elsevier Inc; 2015. pp. 1487–1536. [Google Scholar]
- Pohl CR, Knobil E. The role of the central nervous system in the control of ovarian function in higher primates. Annu Rev Physiol. 1982;44:583–593. doi: 10.1146/annurev.ph.44.030182.003055. [DOI] [PubMed] [Google Scholar]
- Rance NE, Krajewski SJ, Smith MA, Cholanian M, Dacks PA. Neurokinin B and the hypothalamic regulation of reproduction. Brain Res. 2010;1364:116–128. doi: 10.1016/j.brainres.2010.08.059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sarkar DK, Chiappa SA, Fink G, Sherwood NM. Gonadotropin-releasing hormone surge in pro-oestrous rats. Nature. 1976;264:461–463. doi: 10.1038/264461a0. [DOI] [PubMed] [Google Scholar]
- Schwanzel-Fukuda M, Bick D, Pfaff DW. Luteinizing hormone-releasing hormone (LHRH)-expressing cells do not migrate normally in an inherited hypogonadal (Kallmann) syndrome. Brain Res Mol Brain Res. 1989;6:311–326. doi: 10.1016/0169-328x(89)90076-4. [DOI] [PubMed] [Google Scholar]
- Seeburg PH, Adelman JP. Characterization of cDNA for precursor of human luteinizing hormone releasing hormone. Nature. 1984;311:666–668. doi: 10.1038/311666a0. [DOI] [PubMed] [Google Scholar]
- Seminara SB, Messager S, Chatzidaki EE, Thresher RR, Acierno JS, Jr, Shagoury JK, Bo-Abbas Y, Kuohung W, Schwinof KM, Hendrick AG, et al. The GPR54 gene as a regulator of puberty. N Engl J Med. 2003;349:1614–1627. doi: 10.1056/NEJMoa035322. [DOI] [PubMed] [Google Scholar]
- Shivers BD, Harlan RE, Morrell JI, Pfaff DW. Absence of oestradiol concentration in cell nuclei of LHRH-immunoreactive neurones. Nature. 1983;304:345–347. doi: 10.1038/304345a0. [DOI] [PubMed] [Google Scholar]
- Sklar CA, Mueller PL, Gluckman PD, Kaplan SL, Rudolph AM, Grumbach MM. Hormone ontogeny in the ovine fetus. VII. Circulating luteinizing hormone and follicle-stimulating hormone in mid- and late gestation. Endocrinology. 1981;108:874–880. doi: 10.1210/endo-108-3-874. [DOI] [PubMed] [Google Scholar]
- Szentagothai JFB, Mess B, Halasz B. Hypothalamic Control of the Anterior Pituitary. 3. Budapest: Akademiai Kiado; 1968. [Google Scholar]
- Terasawa E, Guerrier KA, Plant TM. Kisspeptin and puberty in mammals. Adv Exp Med Biol. 2013;784:253–273. doi: 10.1007/978-1-4614-6199-9_12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Topaloglu AK, Reimann F, Guclu M, Yalin AS, Kotan LD, Porter KM, Serin A, Mungan NO, Cook JR, Ozbek MN, et al. TAC3 and TACR3 mutations in familial hypogonadotropic hypogonadism reveal a key role for Neurokinin B in the central control of reproduction. Nat Genet. 2009;41:354–358. doi: 10.1038/ng.306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsutsui K, Saigoh E, Ukena K, Teranishi H, Fujisawa Y, Kikuchi M, Ishii S, Sharp PJ. A novel avian hypothalamic peptide inhibiting gonadotropin release. Biochem Biophys Res Commun. 2000;275:661–667. doi: 10.1006/bbrc.2000.3350. [DOI] [PubMed] [Google Scholar]
- Tsutsui K, Ubuka T, Bentley GE, Kriegsfeld LJ. Gonadotropin-inhibitory hormone (GnIH): discovery, progress and prospect. Gen Comp Endocrinol. 2012;177:305–314. doi: 10.1016/j.ygcen.2012.02.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vale W, Rivier C, Hsueh A, Campen C, Meunier H, Bicsak T, Vaughan J, Corrigan A, Bardin W, Sawchenko P, et al. Chemical and biological characterization of the inhibin family of protein hormones. Recent Prog Horm Res. 1988;44:1–34. doi: 10.1016/b978-0-12-571144-9.50005-3. [DOI] [PubMed] [Google Scholar]
- Wakabayashi Y, Nakada T, Murata K, Ohkura S, Mogi K, Navarro VM, Clifton DK, Mori Y, Tsukamura H, Maeda K, et al. Neurokinin B and dynorphin A in kisspeptin neurons of the arcuate nucleus participate in generation of periodic oscillation of neural activity driving pulsatile gonadotropin-releasing hormone secretion in the goat. J Neurosci. 2010;30:3124–3132. doi: 10.1523/JNEUROSCI.5848-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Waldhauser F, Weissenbacher G, Frisch H, Pollak A. Pulsatile secretion of gonadotropins in early infancy. Eur J Pediatr. 1981;137:71–74. doi: 10.1007/BF00441173. [DOI] [PubMed] [Google Scholar]
- Wierman ME, Kiseljak-Vassiliades K, Tobet S. Gonadotropin-releasing hormone (GnRH) neuron migration: initiation, maintenance and cessation as critical steps to ensure normal reproductive function. Front Neuroendocrinol. 2011;32:43–52. doi: 10.1016/j.yfrne.2010.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilson RC, Kesner JS, Kaufman JM, Uemura T, Akema T, Knobil E. Central electrophysiologic correlates of pulsatile luteinizing hormone secretion in the rhesus monkey. Neuroendocrinology. 1984;39:256–260. doi: 10.1159/000123988. [DOI] [PubMed] [Google Scholar]
- Wray S, Grant P, Gainer H. Evidence that cells expressing luteinizing hormone-releasing hormone mRNA in the mouse are derived from progenitor cells in the olfactory placode. Proc Natl Acad Sci U S A. 1989;86:8132–8136. doi: 10.1073/pnas.86.20.8132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yeo SH, Herbison AE. Estrogen-negative feedback and estrous cyclicity are critically dependent upon estrogen receptor-alpha expression in the arcuate nucleus of adult female mice. Endocrinology. 2014;155:2986–2995. doi: 10.1210/en.2014-1128. [DOI] [PubMed] [Google Scholar]
- Yoshimura T, Yasuo S, Watanabe M, Iigo M, Yamamura T, Hirunagi K, Ebihara S. Light-induced hormone conversion of T4 to T3 regulates photoperiodic response of gonads in birds. Nature. 2003;426:178–181. doi: 10.1038/nature02117. [DOI] [PubMed] [Google Scholar]
- Zeleznik AJ, Plant TM. Control of the Menstrual Cycle. In: Plant TM, Zeleznik AJ, editors. Knobil and Neill’s Physiology of Reproduction. 4. San Diego, CA, USA: Elsevier Inc; 2015. pp. 1307–1361. [Google Scholar]
- Zhang Y, Proenca R, Maffei M, Barone M, Leopold L, Friedman JM. Positional cloning of the mouse obese gene and its human homologue. Nature. 1994;372:425–432. doi: 10.1038/372425a0. [DOI] [PubMed] [Google Scholar]





