Abstract
Background
Sleepiness and fatigue are commonly reported by family members of intensive care unit (ICU) patients. Sleep deprivation may result in cognitive deficits. Sleep deprivation and cognitive blunting have not been quantitatively assessed in this population. We sought to determine the proportion of family members of ICU patients that experience excessive daytime sleepiness, sleep-associated functional impairment, and cognitive blunting.
Methods
Multicenter, cross-sectional survey of family members of patients admitted to ICUs at the University of Maryland Medical Center, Johns Hopkins University Hospital, and Christiana Hospital. Family members of ICU patients were evaluated using the Epworth Sleepiness Scale, a validated survey assessing sleepiness in everyday situations (normal, less than 10); the Functional Outcomes of Sleep Questionnaire-10 (FOSQ-10), a questionnaire quantifying the impact of sleepiness on daily activities (normal, at least 17.9); and psychomotor vigilance testing, a test of cognitive function, in relation to sleep deprivation (normal mean reaction time less than 500 ms).
Results
A total of 225 family members were assessed. Of these, 50.2 % (113/225) had Epworth scores consistent with excessive daytime sleepiness. Those with sleepiness experienced greater impairment in performing daily activities by FOSQ-10 (15.6 ± 3.0 vs 17.4 ± 2.2, p < 0.001). Cognitive blunting was found in 13.3 % (30/225) of family members and 15.1 % (14/93) of surrogate decision-makers. Similar rates of cognitive blunting as reported by mean reaction time of at least 500 ms were found among family members whether or not they reported sleepiness (15.0 % (17/113) vs. 11.6 % (13/112), p = 0.45).
Conclusions
Half of the family members of ICU patients suffer from excessive daytime sleepiness. This sleepiness is associated with functional impairment, but not cognitive blunting.
Keywords: Critical care, Sleep deprivation, Family caregivers, Sleepiness, End of life, Surrogate decision-maker
Introduction
Sleep deprivation is common among family members across a spectrum of clinical settings, including family members of cancer patients [1], patients with acute coronary syndrome [2], surgical candidates, and critically ill infants and adults [3]. Nearly half of intensive care unit (ICU) family decision-makers report feeling sleepy and sleep deprived [3]. Despite demonstrating that a large proportion of family members of critically ill patients suffer sleepiness, prior studies did not quantify its extent or impact [3–5]. In other environments, sleep deprivation results in cognitive impairment in family members of hospitalized patients [4], but it is unclear whether this occurs in family members of patients in the ICU. Since a large proportion of medical decisions in the ICU are made by surrogates—including the vast majority of decision-making at the end of life (between 90 and 94 %) performed by someone other than the patient [1, 2]—this is concerning.
The greatest impact of sleepiness in family members of ICU patients may be the deleterious effects it has on performing activities of daily living and cognitive function [6]. Sleepiness not only results in fatigue, but may impair one’s ability to perform efficiently and effectively in their daily responsibilities with respect to work or leisure, and can result in decreased alertness, reduced attention span, and lack of vigilance. Given the responsibilities family members of ICU patients have as surrogates and decision-makers, this potential functional and cognitive impairment combined with the stress of having a critically ill loved one can have a tremendous negative impact on family function and psyche.
We hypothesized that, among family members of the critically ill, sleep deprivation results in impaired functionality and cognitive blunting. Our primary outcome was excessive daytime sleepiness. We sought to assess the relationship between sleepiness and associated functional impairment and cognitive blunting in this population. We performed a multicenter, cross-sectional study of family members, spouses, and partners of patients admitted to various ICUs. Excessive daytime sleepiness, resultant functional impairment, and cognitive blunting in family members of critically ill patients were assessed using validated surveys.
Methods
Environment
We conducted a multicenter, cross-sectional study on a convenience sample surveyed across eight ICUs at the University of Maryland Medical Center (UMMC), The Johns Hopkins University Hospital (JHU) medical ICU, and the Christiana Hospital (CH) medical ICU. Data were collected from family members of patients admitted between 1 February 2011 and 1 May 2012. The ICU composition was as follows: UMMC (29 medical ICU beds, 19 surgical ICU beds, 19 cardiac surgical ICU beds, 15 complex cardiology ICU beds, 12 neurology ICU beds, and 30 trauma ICU beds); JHU (16 medical ICU beds); CH (22 medical ICU beds). UMMC and JHU are academic, tertiary care facilities located in Baltimore, MD, with 705 and 1,025 beds, respectively. Christiana Hospital is a 913-bed, community-based tertiary care referral center located in Newark, DE.
Data sources and patient selection
Family members were approached in patients’ rooms and in common areas non-consecutively to comprise our convenience sample. Family members were eligible regardless of their loved one’s length of stay, allowing for enrollment of subjects at various time points in the course of their family member’s critical illness. We assessed all members of the family, consistent with prior studies [7,8]. In order to maximize recruitment and minimize selection bias, we did not link participants to their family member in the ICU. Subjects independently completed a 39-question survey instrument, with the investigator present to address any issues. The survey consisted of 14 questions, capturing subject demographics, subject-reported patient clinical characteristics, the Epworth Sleepiness Scale (ESS) [8], the Functional Outcomes of Sleep Questionnaire–short form (FOSQ-10) [9], and seven sleep-related questions inquiring about sleep habits, preexisting sleep disorders, and use of hypnotic medications. The ESS is an eight-question measure of daytime sleepiness, rating the likelihood of falling asleep in eight different scenarios. A score of at least 10 is representative of significant daytime sleepiness [10]. The FOSQ-10 is a validated short version (10 items) of the FOSQ [11], quantifying the impact of daytime sleepiness on activities of daily living. Subjects were queried regarding “poor physician communication” as per their perceptions, and labeled those who answered the question “I feel like my loved one’s doctors do a good job explaining things to me” with “disagree” or “strongly disagree” as reporting “poor physician communication.” This term was used to encompass family perception of adequacy of time, clarity, and completeness of information conveyed by the health care team, which have been assessed previously [12].
Following collection of the completed survey, the research team instructed the subject on the procedure for the psychomotor vigilance test and then administered the test to the subject on a personal data assistant (Palm Tungsten or Palm Zire, Sunnyvale, CA USA) [13]. The psychomotor vigilance test is a validated test of cognitive function, assessing loss of sustained attention in response to sleep deprivation [14]. In the psychomotor vigilance test, the subject was asked to press a button in response to a visual prompt, which appeared repeatedly at varying time intervals for a total of 5 min. Subjects were allowed to participate in the study only once.
The research team evaluated families during the morning (9:00 a.m.–12:00 p.m.) or evening (7:00 p.m.–9:00 p.m.) every day. These times were chosen for recruitment of subjects with conventional work hours and to avoid testing during the circadian physiologic dip in alertness that coincides with early afternoon sleepiness (2:00 p.m.–4:00 p.m.) [15]. This study was approved by the institutional review boards of the participating centers with a waiver of informed consent (University of Maryland, Baltimore HP-00047937; JHU, NA-00047939; CH, CCC# 32028).
Statistical analysis
Baseline characteristics of patients admitted to the ICUs were presented as counts and percentages or mean values with standard deviations (SDs). Differences were evaluated using two-sample t tests for normally distributed continuous variables, comparison of proportions for categorical variables, and Wilcoxon–Mann–Whitney tests for ordinal variables [16]. Non-parametric testing was used for non-normally distributed variables. Our primary outcome was subjective sleepiness by ESS (ESS ≥ 10) [10]. Our secondary outcomes were cognitive blunting, defined as a mean reaction time of at least 500 ms on psychomotor vigilance testing [17], and impairment with respect to daily function as measured by a FOSQ-10 score of less than 17.9 [18].
Characteristics of subjects by score were presented as mean values with standard deviations and compared using two-sample t tests for continuous variables and comparisons of proportions for categorical variables. A Pearson’s product-moment correlation coefficient and linear regression was assessed between the variables of ESS and psychomotor vigilance testing mean reaction time. In selecting variables for the multivariable logistic regression, all baseline characteristics were assessed and those with a significance test of at most 0.20 in univariate testing were included in the models for sleepiness, functional impairment, and cognitive blunting [19].
Analyses were performed using SAS v.9.1.3. We applied the traditional definition of p ≤ 0.05 for statistical significance.
Results
Among respondents, 99.6 % (224/225) completed the ESS and 100 % (225/225) completed the FOSQ-10. Psychomotor vigilance testing was completed on 100 % of subjects (225). Given low missingness, listwise deletion was used for missing data. The composition of respondents by ICU type was as follows: medical, 28.0 % (63/225); surgical, 12.4 % (28/225); cardiothoracic, 8.4 % (19/225); neurologic, 9.3 % (21/225); cardiac, 9.8 % (22/225); and trauma, 30.7 % (69/225). Three respondents did not report ICU type, 1.3 % (3/225). Of all subjects, 7.1 % (16/225) reported a preexisting sleep disorder.
Excessive daytime sleepiness (ESS ≥ 10) was found in 50.2 % (113/225) of respondents (Table 1). Subjects with excessive daytime sleepiness reported greater difficulty conducting daily activities (mean FOSQ-10 scores 15.6 ± 3.0 vs. 17.4 ± 2.2, p < 0.001). Age, gender, race, relationship to patient, status as surrogate decision-maker, hours slept the night prior, patient ICU length of stay, and the presence of an advanced directive were not associated with increased risk of excessive daytime sleepiness. The frequency of cognitive blunting in the sleepy subjects (ESS ≥ 10) was similar to that of the subjects without excessive daytime sleepiness [15.0 % (17/113) vs. 11.6 % (13/112), p = 0.45].
Table 1.
Subject characteristics by sleepiness (sleepiness denoted by Epworth Sleepiness Score ≥10)
| Characteristicsa | Total (n = 225) | Sleepy (n = 113) | Not sleepy (n = 112) |
p value* |
|---|---|---|---|---|
| Age (years) | 48.0 ± 15.1 (n = 223) | 46.1 ± 15.5 (n = 112) | 49.8 ± 14.5 (n = 111) | 0.07 |
| Sex: male, n (%) | 72 (32.0) | 38 (33.6) | 34 (30.4) | 0.60 |
| Race: white, n (%) | 128 (56.9) | 59 (52.2) | 69 (61.6) | 0.16 |
| Patient relationship | ||||
| Spouse/partner | 41 (18.2) | 19 (16.8) | 22 (19.6) | 0.48 |
| Parent | 38 (16.9) | 17 (15.0) | 21 (18.8) | |
| Child | 52 (23.1) | 26 (23.0) | 26 (23.2) | |
| Sibling | 34 (15.1) | 15 (13.3) | 19 (17.0) | |
| Other | 60 (26.7) | 36 (31.9) | 24 (21.4) | |
| Surrogate (yes) | 93 (41.3) | 42 (37.2) | 51 (45.5) | 0.20 |
| Education (college or more) | 116 (51.6) | 59 (52.1) | 57 (50.9) | 0.84 |
| Hours slept prior night | 6.7 ± 2.3 (n = 213) | 6.5 ± 2.5 (n = 105) | 6.9 ± 2.1 (n = 108) | 0.24 |
| ICU length of stay | 9.7 ± 20.5 | 9.8 ± 20.1 | 9.6 ± 21.1 | 0.94 |
| Previous ICU admit (yes) | 88 (39.3) | 40 (35.4) | 48 (43.2) | 0.23 |
| Advance directive (yes) | 57 (25.9) | 27 (24.3) | 30 (27.5) | 0.59 |
| Clarity of physician explanation | ||||
| Strongly disagree | 6 (2.8) | 3 (2.7) | 3 (2.7) | 0.34 |
| Disagree | 14 (6.2) | 10 (8.8) | 4 (3.6) | |
| Agree | 109 (48.4) | 49 (43.4) | 60 (53.5) | |
| Strongly agree | 93 (41.3) | 50 (44.2) | 43 (38.4) | |
| Unknown | 3 (1.3) | 1 (0.9) | 2 (1.8) | |
| Perceived severity of illness | ||||
| Not very sick | 4 (1.8) | 2 (1.8) | 2 (1.8) | 0.78 |
| Sick but will get well | 101 (44.9) | 50 (44.2) | 51 (45.5) | |
| Very sick–may die | 68 (30.2) | 37 (32.7) | 31 (27.7) | |
| Extremely sick–high chance of death |
25 (11.1) | 14 (12.4) | 11 (9.8) | |
| Other | 27 (12.0) | 11 (8.8) | 16 (15.2) | |
| Psychomotor vigilance test (mean reaction time in milliseconds) |
387.7 ± 130.0 | 397.5 ± 107.8 | 377.8 ± 148.6 | 0.26 |
| Functional Outcome of Sleep Questionnaire-10 |
16.5 ± 2.8 | 15.6 ± 3.0 | 17.4 ± 2.2 | <0.001 |
For continuous variables, p values were obtained from two- sample t tests; for categorical variables, p values were obtained from tests of comparison of proportions
Continuous values are presented as mean ± SD, and categorical values are presented as number (%)
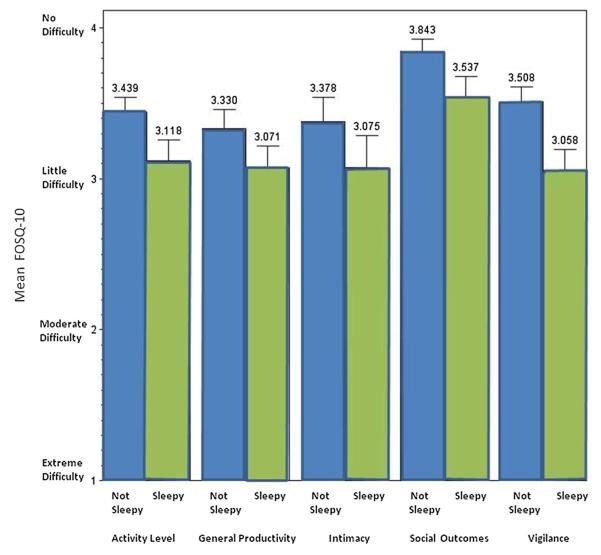
Overall, 62 % (140/225) of all respondents had a FOSQ-10 score less than 17.9 (mean FOSQ-10 16.5 ± 2.8), indicating impairment of daily activities due to sleepiness (Table 2) [18]. The decrement was not concentrated in a single component of the five domains of the FOSQ-10. The FOSQ-10 scores of those that were sleepy compared to those that were not were lower across all domains (Fig. 1). Accepted normal scores for each domain of the FOSQ have been noted in the literature (general productivity = 3.6, activity level = 3.5, vigilance = 3.5, social outcomes = 3.9, intimacy and sexual relationship = 3.5) [11, 20].
Table 2.
FOSQ-10 domains
| FOSQ subscale | Question domainsa | Total | Abnormal n (%) |
|---|---|---|---|
| General productivity | Concentration (n = 225) | 3.2 ± 0.8 | 160 (71) |
| Remembering (n = 225) | 3.2 ± 0.8 | ||
| Activity level | Activity in evening (n = 225) | 3.0 ± 0.9 | 127 (56) |
| Activity in morning (n = 225) | 3.2 ± 0.9 | ||
| Relationships affected (n = 219) | 3.7 ± 0.6 | ||
| Vigilance | Watching movies (n = 219) | 3.0 ± 0.8 | 119 (53) |
| Driving long distances (n = 202) | 3.3 ± 0.9 | ||
| Driving short distances (n = 206) | 3.7 ± 0.6 | ||
| Social outcomes | Visit in your home (n = 216) | 3.7 ± 0.6 | 52 (24) |
| Intimacy and sexual relationships | Desire intimacy (n = 191) | 3.2 ± 0.9 | 94 (49) |
| Total (scale 0–20) | 16.5 ± 2.8 | ||
| Impairment (yes) | 140 (62 %) |
Subjects answering “I don’t do this activity” are not included
Fig. 1.
Functional Outcomes of Sleep Questionnaire-10 by sleepiness
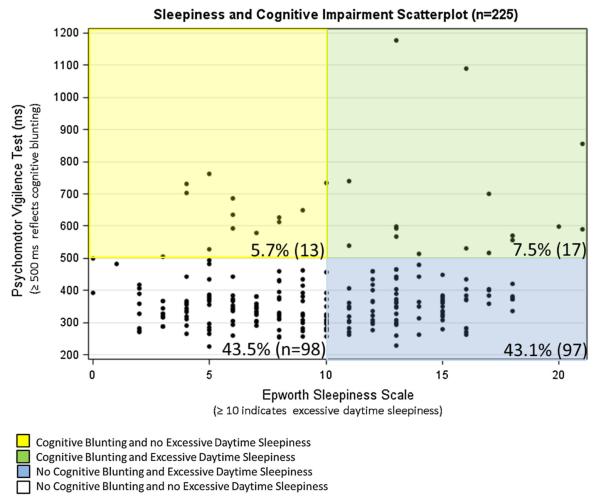
Cognitive blunting (psychomotor vigilance test mean reaction time of at least 500 ms) was found in 13.3 % (30/225) of subjects. Linear regression demonstrated a positive relationship between ESS and psychomotor vigilance test scores (Fig. 2), with a one-unit increase in ESS yielding a 4.4-ms increase in RT (p = 0.01).
Fig. 2.
Joint distribution of psychomotor vigilance testing values and Epworth Sleepiness Scale Scores
In multivariable analysis, no association was found between excessive daytime sleepiness and patient or subject characteristics. After adjusting for sleep hours and ICU length of stay, low FOSQ-10 score was independently associated with age (OR 0.98, 95 % CI 0.96–1.00, p = 0.05) and female gender (OR 2.13, 95 % CI 1.16–3.93, p = 0.020). In a multivariable model containing, gender, race, presence of advanced directive, quality of physician communication, cognitive blunting was independently associated with female gender (OR 4.40, 95 % CI 1.26–15.38, p = 0.02) and a perception of poor physician communication (OR 4.46, 95 % CI 1.48–13.43, p = 0.01). Additional analysis found that sleepiness was significantly associated with functional impairment (OR 2.341, 95 % CI 1.285–4.265, p = 0.005), when adjusting for gender, age, hours of sleep prior night, and ICU length of stay.
When reporting our outcomes in the context of being a surrogate decision-maker, the mean degree and frequency of excessive daytime sleepiness were similar among surrogates and non-surrogates (ESS 9.1 ± 5.0 vs. 10.0 ± 4.4, p = 0.19; 45.2 % (42/93) vs. 53.8 % (71/132), p = 0.20). Additionally, FOSQ-10 scores among surrogates and non-surrogates were similar (FOSQ-10 16.4 ± 3.1 vs. 16.6 ± 2.5, p = 0.7). Surrogates, however, demonstrated worse performance, with a higher overall median reaction time, or reaction time (353.0 ± 110.2 vs. 326.8 ± 64.5 ms, p = 0.04), a higher minimum reaction time (237.5 ± 54.7 vs. 222.0 ± 35.3 ms, p = 0.02), and a greater number of lapses (13.2 ± 15.5 vs. 9.0 ± 10.7, p = 0.02) when compared to non-surrogates (Appendix A, Electronic Supplementary Material). There were 17 subjects that reported having a prior diagnosis of a sleep disorder. No significant differences were noted between those with and those without a prior diagnosis of a sleep disorder when comparing demographic characteristics, surrogate status, sleepiness by ESS, functional impairment by FOSQ-10, or cognitive blunting by psychomotor vigilance testing results.
Discussion
We found that excessive daytime sleepiness is common among family members of the critically ill. Over half of our subjects reported significant daytime sleepiness [9], demonstrating sleepiness similar to patients with obstructive sleep apnea [11, 21]. Thus, the proportion of significant daytime sleepiness (50.2 %) in these subjects is striking and considerably greater than in the general population (10–20 %) [8]. Excessive daytime sleepiness and decreased functionality were found across the study population to a degree comparable to patients with chronic cardiac, pulmonary, and neurologic conditions [2,22–24]. Our study is the first to quantify the extent of sleepiness in family members of the critically ill, and assess its impact on functional impairment and cognitive blunting.
Family members also suffered significant decreases in quality of life due to sleep deprivation as reflected by low FOSQ-10 scores, with almost two-thirds suffering significant functional impairment. The domain which was most notably affected was general productivity, which includes concentration, completing daily tasks and chores, remembering, working on hobbies, completing financial affairs and paperwork, employment tasks, and maintaining telephone conversations. Contextually, these scores were similar to those in individuals with mild sleep apnea [21] and worse than in patients suffering excessive daytime sleepiness due to congestive heart failure [23], chronic obstructive pulmonary disease with nocturnal hypoxemia [24], traumatic brain injury [22], and geriatric patients with self-reported sleepiness [25]. Additionally, the low FOSQ-10 scores may reflect the impact of sleepiness on daytime activities, including interactions with the ICU medical team, which is further evidenced by the association we found between perceived poor physician communication and cognitive blunting. Historically, guidelines for support of family members of ICU patients have focused on improving communication and addressing family members’ spiritual and emotional needs [26] as strategies to mitigate the stress experienced by their loved one’s admission to the ICU. Our data suggest that support for family members will also need to include interventions to alleviate sleep deprivation, such as providing family-friendly areas that may alleviate stress [27], quiet areas for napping, or educating family members on the importance of obtaining adequate and consolidated sleep by setting aside 7–9 h of scheduled time dedicated to sleep.
We differentiated degrees of cognitive blunting according to psychomotor vigilance testing mean reaction time and number of lapses (reaction time at least 500 ms), consistent with prior studies [17, 28]. The proportion and extent of cognitive blunting was marked. Family members of ICU patients exhibited greater cognitive blunting than patients with sleep-disordered breathing [28], sleep-deprived on-call resident physicians [29], and healthy subjects with a blood alcohol concentration of 0.05–0.10 % [29, 30].
Difficulty sleeping has been self-reported as one of the top stressors of ICU patients and their family members [31, 32]. Previous data demonstrate that up to 80 % of family members obtain less sleep, 70 % suffer worse quality sleep, and 20 % require hypnotic medications after their family member was admitted to an ICU [5]. Sleep disturbances and exhaustion have been identified as physiologic and behavioral responses to the stress of having a family member in the ICU [33]. Additionally, our objective findings concur with prior studies that subjectively identified sleepiness and fatigue as causes of psychological and physical morbidity in family members of the critically ill [34]. Our findings further support the theory that sleep deprivation may play a role in the development of the postintensive care syndrome-family [35], the constellation of symptoms experienced by family members after ICU discharge, which is manifested by anxiety, acute stress, posttraumatic stress, depression, and grief. Further work associating patient-specific data with respective family members may also be necessary to better understand the patient clinical characteristics associated with family members’ sleepiness and which families may benefit most from interventions.
This multicenter study evaluated a spectrum of ICU types across multiple hospitals types, making our results generalizable to a variety of settings. While our respondent selection sampling and the decision not to link to specific patients were designed to reduce selection bias, these still require consideration. Prior studies have shown that families more satisfied with their loved ones’ care are more likely to respond to survey instruments [36], are more likely to be at the bedside visiting [37], and may have higher socioeconomic status [38], thus potentially underestimating the severity of sleepiness-associated morbidity among this population. Additionally, prior studies have demonstrated that survey-based assessments of sleepiness often underestimate the magnitude of sleepiness [39], potentially further underestimating the severity of this problem. Although the possibility exists that some respondents had an undiagnosed sleep disorder, we screened for symptoms and prior diagnoses of sleep-disordered breathing and found a low proportion within our population. While the psychomotor vigilance test assesses sustained attention as a component of executive functioning, it is possible that we may have underestimated the prevalence of cognitive blunting, as the psychomotor vigilance test does not capture all realms of executive function [40]. Lastly, consistent with prior studies evaluating sleep deprivation among family members [23, 24, 34], we performed a cross-sectional design, which does not include a control group. While the possibility exists that our data reflect general sleepiness in a general population, the marked divergence from multiple prior studies that included normal subjects suggests otherwise [8, 10, 18].
Conclusion
Half of the family members of ICU patients suffer from excessive daytime sleepiness. Functional impairment due to sleep deprivation is common in this population, with a proportion of family members suffering cognitive blunting as a result of sleepiness. Surrogates suffer equal or greater dysfunction and cognitive impairment than those without decision-making capacities. Sleepiness may be both a challenge to patient-centered care and a contributing risk factor for postintensive care syndrome-family. Efforts should be directed towards better understanding the etiology of sleep deprivation as well as interventions to better support family members.
Supplementary Material
Take-home message.
Half of the family members of intensive care unit patients suffer from excessive daytime sleepiness. This sleepiness results in functional impairment, with a proportion of family members also demonstrating cognitive blunting.
Acknowledgments
We thank the nurses and family members of the participating ICUs for their time and participation in this study. Roy Brower, MD contributed to the questionnaire design and study conception. Helpful criticism was received from Theodore Iwashyna, MD, PhD. Dr. Netzer thanks Eric Zager, MD for making the completion of this study possible. Dr. Verceles receives support from NIA P30 AG028747, and Dr. Netzer is supported by NIH K12RR023250.
Footnotes
Conflicts of interest On behalf of all authors, the corresponding author states that there is no conflict of interest.
Electronic supplementary material The online version of this article (doi:10.1007/s00134-014-3347-z) contains supplementary material, which is available to authorized users.
Contributor Information
Avelino C. Verceles, Division of Pulmonary, Critical Care and Sleep Medicine, University of Maryland School of Medicine, 110 S. Paca St., Second Floor, Baltimore, MD 21201, USA; University of Maryland Claude D. Pepper Older Americans Independence Center, University of Maryland School of Medicine and the Baltimore VA Medical Center Geriatric Research, Education and Clinical Center, Baltimore, MD, USA
Douglas S. Corwin, Department of Medicine, University of Maryland School of Medicine, Baltimore, MD, USA
Majid Afshar, Division of Pulmonary, Critical Care and Sleep Medicine, University of Maryland School of Medicine, 110 S. Paca St., Second Floor, Baltimore, MD 21201, USA.
Eliot B. Friedman, Pulmonology Associates, Main Line Health Lankenau Hospital, Wynnewood, PA, USA
Michael T. McCurdy, Division of Pulmonary, Critical Care and Sleep Medicine, University of Maryland School of Medicine, 110 S. Paca St., Second Floor, Baltimore, MD 21201, USA
Carl Shanholtz, Division of Pulmonary, Critical Care and Sleep Medicine, University of Maryland School of Medicine, 110 S. Paca St., Second Floor, Baltimore, MD 21201, USA.
Karen Oakjones, Division of Pulmonary and Critical Care Medicine, Johns Hopkins University, Baltimore, MD, USA.
Marc T. Zubrow, Division of Pulmonary, Critical Care and Sleep Medicine, University of Maryland School of Medicine, 110 S. Paca St., Second Floor, Baltimore, MD 21201, USA
Jennifer Titus, Division of Pulmonary, Critical Care and Sleep Medicine, University of Maryland School of Medicine, 110 S. Paca St., Second Floor, Baltimore, MD 21201, USA.
Giora Netzer, Division of Pulmonary, Critical Care and Sleep Medicine, University of Maryland School of Medicine, 110 S. Paca St., Second Floor, Baltimore, MD 21201, USA; Department of Epidemiology and Public Health, University of Maryland School of Medicine, Baltimore, MD, USA.
References
- 1.Kinoshita S, Miyashita M. Evaluation of end-of-life cancer care in the ICU: perceptions of the bereaved family in Japan. Am J Hosp Palliat Care. 2012;30:225–230. doi: 10.1177/1049909112446805. [DOI] [PubMed] [Google Scholar]
- 2.Bedsworth JA, Molen MT. Psychological stress in spouses of patients with myocardial infarction. Heart Lung. 1982;11:450–456. [PubMed] [Google Scholar]
- 3.McAdam JL, Dracup KA, White DB, Fontaine DK, Puntillo KA. Symptom experiences of family members of intensive care unit patients at high risk for dying. Crit Care Med. 2010;38:1078–1085. doi: 10.1097/CCM.0b013e3181cf6d94. [DOI] [PubMed] [Google Scholar]
- 4.Halm MA, Titler MG, Kleiber C, Johnson SK, Montgomery LA, Craft MJ, Buckwalter K, Nicholson A, Megivern K. Behavioral responses of family members during critical illness. Clin Nurs Res. 1993;2:414–437. doi: 10.1177/105477389300200405. [DOI] [PubMed] [Google Scholar]
- 5.Van Horn E, Tesh A. The effect of critical care hospitalization on family members: stress and responses. Dimens Crit Care Nurs. 2000;19:40–49. doi: 10.1097/00003465-200019040-00014. [DOI] [PubMed] [Google Scholar]
- 6.Killgore WD. Effects of sleep deprivation on cognition. Prog Brain Res. 2010;185:105–129. doi: 10.1016/B978-0-444-53702-7.00007-5. [DOI] [PubMed] [Google Scholar]
- 7.Fumis RR, Deheinzelin D. Family members of critically ill cancer patients: assessing the symptoms of anxiety and depression. Intensive Care Med. 2009;35:899–902. doi: 10.1007/s00134-009-1406-7. [DOI] [PubMed] [Google Scholar]
- 8.Johns MW. A new method for measuring daytime sleepiness: the Epworth Sleepiness Scale. Sleep. 1991;14:540–545. doi: 10.1093/sleep/14.6.540. [DOI] [PubMed] [Google Scholar]
- 9.Chasens ER, Ratcliffe SJ, Weaver TE. Development of the FOSQ-10: a short version of the Functional Outcomes of Sleep Questionnaire. Sleep. 2009;32:915–919. doi: 10.1093/sleep/32.7.915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Johns MW. Daytime sleepiness, snoring, and obstructive sleep apnea. The Epworth Sleepiness Scale. Chest. 1993;103:30–36. doi: 10.1378/chest.103.1.30. [DOI] [PubMed] [Google Scholar]
- 11.Weaver TE, Laizner AM, Evans LK, Maislin G, Chugh DK, Lyon K, Smith PL, Schwartz AR, Redline S, Pack AI, Dinges DF. An instrument to measure functional status outcomes for disorders of excessive sleepiness. Sleep. 1997;20:835–843. [PubMed] [Google Scholar]
- 12.Azoulay E, Pochard F, Kentish-Barnes N, Chevret S, Aboab J, Adrie C, Annane D, Bleichner G, Bollaert PE, Darmon M, Fassier T, Galliot R, Garrouste-Orgeas M, Goulenok C, Goldgran-Toledano D, Hayon J, Jourdain M, Kaidomar M, Laplace C, Larche J, Liotier J, Papazian L, Poisson C, Reignier J, Saidi F, Schlemmer B. Risk of post-traumatic stress symptoms in family members of intensive care unit patients. Am J Respir Crit Care Med. 2005;171:987–994. doi: 10.1164/rccm.200409-1295OC. [DOI] [PubMed] [Google Scholar]
- 13.Thorne DR, Johnson DE, Redmond DP, Sing HC, Belenky G, Shapiro JM. The Walter Reed palm-held psychomotor vigilance test. Behav Res Methods. 2005;37:111–118. doi: 10.3758/bf03206404. [DOI] [PubMed] [Google Scholar]
- 14.Dinges DI, Powell JW. Microcomputer analysis of performance on a portable, simple visual RT task sustained operations. Behav Res Methods Instrum Comput. 1985;17:652–655. [Google Scholar]
- 15.Monk TH, Buysse DJ, Reynolds CF, 3rd, Kupfer DJ. Circadian determinants of the postlunch dip in performance. Chronobiol Int. 1996;13:123–133. doi: 10.3109/07420529609037076. [DOI] [PubMed] [Google Scholar]
- 16.Rosner B. Fundamentals of biostatistics. Duxbury, Belmont: 1995. [Google Scholar]
- 17.Basner M, Dinges DF. Maximizing sensitivity of the psychomotor vigilance test (PVT) to sleep loss. Sleep. 2011;34:581–591. doi: 10.1093/sleep/34.5.581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Weaver TE, Maislin G, Dinges DF, Bloxham T, George CF, Greenberg H, Kader G, Mahowald M, Younger J, Pack AI. Relationship between hours of CPAP use and achieving normal levels of sleepiness and daily functioning. Sleep. 2007;30:711–719. doi: 10.1093/sleep/30.6.711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Mickey RM, Greenland S. The impact of confounder selection criteria on effect estimation. Am J Epidemiol. 1989;129:125–137. doi: 10.1093/oxfordjournals.aje.a115101. [DOI] [PubMed] [Google Scholar]
- 20.Barnes M, Houston D, Worsnop CJ, Neill AM, Mykytyn IJ, Kay A, Trinder J, Saunders NA, Douglas McEvoy R, Pierce RJ. A randomized controlled trial of continuous positive airway pressure in mild obstructive sleep apnea. Am J Respir Crit Care Med. 2002;165:773–780. doi: 10.1164/ajrccm.165.6.2003166. [DOI] [PubMed] [Google Scholar]
- 21.Weaver TE, Mancini C, Maislin G, Cater J, Staley B, Landis JR, Ferguson KA, George CF, Schulman DA, Greenberg H, Rapoport DM, Walsleben JA, Lee-Chiong T, Gurubhagavatula I, Kuna ST. Continuous positive airway pressure treatment of sleepy patients with milder obstructive sleep apnea: results of the CPAP Apnea Trial North American Program (CATNAP) randomized clinical trial. Am J Respir Crit Care Med. 2012;186:677–683. doi: 10.1164/rccm.201202-0200OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Beaulieu-Bonneau S, Morin CM. Sleepiness and fatigue following traumatic brain injury. Sleep Med. 2012;13:598–605. doi: 10.1016/j.sleep.2012.02.010. [DOI] [PubMed] [Google Scholar]
- 23.Carmona-Bernal C, Ruiz-Garcia A, Villa-Gil M, Sanchez-Armengol A, Quintana-Gallego E, Ortega-Ruiz F, Baron-Esquivias G, Capote F. Quality of life in patients with congestive heart failure and central sleep apnea. Sleep Med. 2008;9:646–651. doi: 10.1016/j.sleep.2007.11.008. [DOI] [PubMed] [Google Scholar]
- 24.Lewis CA, Fergusson W, Eaton T, Zeng I, Kolbe J. Isolated nocturnal desaturation in COPD: prevalence and impact on quality of life and sleep. Thorax. 2009;64:133–138. doi: 10.1136/thx.2007.088930. [DOI] [PubMed] [Google Scholar]
- 25.Gooneratne NS, Weaver TE, Cater JR, Pack FM, Arner HM, Greenberg AS, Pack AI. Functional outcomes of excessive daytime sleepiness in older adults. J Am Geriatr Soc. 2003;51:642–649. doi: 10.1034/j.1600-0579.2003.00208.x. [DOI] [PubMed] [Google Scholar]
- 26.Davidson JE, Powers K, Hedayat KM, Tieszen M, Kon AA, Shepard E, Spuhler V, Todres ID, Levy M, Barr J, Ghandi R, Hirsch G, Armstrong D. Clinical practice guidelines for support of the family in the patient-centered intensive care unit: American College of Critical Care Medicine Task Force 2004–2005. Crit Care Med. 2007;35:605–622. doi: 10.1097/01.CCM.0000254067.14607.EB. [DOI] [PubMed] [Google Scholar]
- 27.Jongerden IP, Slooter AJ, Peelen LM, Wessels H, Ram CM, Kesecioglu J, Schneider MM, van Dijk D. Effect of intensive care environment on family and patient satisfaction: a before-after study. Intensive Care Med. 2013;39:1626–1634. doi: 10.1007/s00134-013-2966-0. [DOI] [PubMed] [Google Scholar]
- 28.Lee IS, Bardwell WA, Ancoli-Israel S, Dimsdale JE. Number of lapses during the psychomotor vigilance task as an objective measure of fatigue. J Clin Sleep Med. 2010;6:163–168. [PMC free article] [PubMed] [Google Scholar]
- 29.Arnedt JT, Owens J, Crouch M, Stahl J, Carskadon MA. Neurobehavioral performance of residents after heavy night call vs after alcohol ingestion. JAMA. 2005;294:1025–1033. doi: 10.1001/jama.294.9.1025. [DOI] [PubMed] [Google Scholar]
- 30.Williamson AM, Feyer AM, Mattick RP, Friswell R, Finlay-Brown S. Developing measures of fatigue using an alcohol comparison to validate the effects of fatigue on performance. Accid Anal Prev. 2001;33:313–326. doi: 10.1016/s0001-4575(00)00045-2. [DOI] [PubMed] [Google Scholar]
- 31.Novaes MA, Knobel E, Bork AM, Pavao OF, Nogueira-Martins LA, Ferraz MB. Stressors in ICU: perception of the patient, relatives and health care team. Intensive Care Med. 1999;25:1421–1426. doi: 10.1007/s001340051091. [DOI] [PubMed] [Google Scholar]
- 32.Novaes MA, Aronovich A, Ferraz MB, Knobel E. Stressors in ICU: patients’ evaluation. Intensive Care Med. 1997;23:1282–1285. doi: 10.1007/s001340050500. [DOI] [PubMed] [Google Scholar]
- 33.Tyrie LS, Mosenthal AC. Care of the family in the surgical intensive care unit. Anesthesiol Clin. 2012;30:37–46. doi: 10.1016/j.anclin.2011.11.003. [DOI] [PubMed] [Google Scholar]
- 34.Day A, Haj-Bakri S, Lubchansky S, Mehta S. Sleep, anxiety, and fatigue in family members of patients admitted to the intensive care unit: a questionnaire study. Crit Care. 2013;17:R91. doi: 10.1186/cc12736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Davidson JE, Jones C, Bienvenu OJ. Family response to critical illness: postintensive care syndrome-family. Crit Care Med. 2012;40:618–624. doi: 10.1097/CCM.0b013e318236ebf9. [DOI] [PubMed] [Google Scholar]
- 36.Kross EK, Engelberg RA, Shannon SE, Curtis JR. Potential for response bias in family surveys about end-of-life care in the ICU. Chest. 2009;136:1496–1502. doi: 10.1378/chest.09-0589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Schwarzkopf D, Behrend S, Skupin H, Westermann I, Riedemann NC, Pfeifer R, Günther A, Witte OW, Reinhart K, Hartog CS. Family satisfaction in the intensive care unit: a quantitative and qualitative analysis. Intensive Care Med. 2013;39(6):1071–1079. doi: 10.1007/s00134-013-2862-7. [DOI] [PubMed] [Google Scholar]
- 38.Bein T, Hackner K, Zou T, Schultes S, Bösch T, Schlitt HJ, Graf BM, Olden M, Leitzmann M. Socioeconomic status, severity of disease and level of family members’ care in adult surgical intensive care patients: the prospective ECSSTASI study. Intensive Care Med. 2012;38(4):612–619. doi: 10.1007/s00134-012-2463-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Miletin MS, Hanly PJ. Measurement properties of the Epworth Sleepiness Scale. Sleep Med. 2003;4:195–199. doi: 10.1016/s1389-9457(03)00031-5. [DOI] [PubMed] [Google Scholar]
- 40.Chan RC, Shum D, Toulopoulou T, Chen EY. Assessment of executive functions: review of instruments and identification of critical issues. Arch Clin Neuropsychol. 2008;23:201–216. doi: 10.1016/j.acn.2007.08.010. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.