Abstract
Objective
To develop a porous, biodegradable scaffold for mastoid air cell regeneration.
Study Design
In vitro development of a temperature-sensitive poly(DL-lactic acid-co-glycolic acid)/poly(ethylene glycol) (PLGA/PEG) scaffold tailored for this application.
Methods
Human mastoid bone microstructure and porosity was investigated using micro-computed tomography. PLGA/PEG-alginate scaffolds were developed and scaffold porosity was assessed. Human bone marrow mesenchymal stem cells (hBM-MSCs) were cultured on the scaffolds in vitro. Scaffolds were loaded with ciprofloxacin and release of ciprofloxacin over time in vitro was assessed.
Results
Porosity of human mastoid bone was measured at 83% with an average pore size of 1.3mm. PLGA/PEG-alginate scaffold porosity ranged from 43–78% depending on the alginate bead content. hBM-MSCs proliferate on the scaffolds in vitro, and release of ciprofloxacin from the scaffolds was demonstrated over 7–10 weeks.
Conclusion
The PLGA/PEG-alginate scaffolds developed in this study demonstrate similar structural features to human mastoid bone, support cell growth and display sustained antibiotic release. These scaffolds may be of potential clinical use in mastoid air cell regeneration. Further in vivo studies to assess the suitability of PLGA/PEG-alginate scaffolds for this application are required.
Keywords: Scaffold, PLGA, alginate, mastoid, ciprofloxacin
1. Introduction
The mastoid portion of the temporal bone is composed of interconnected air-filled chambers. This mastoid air cell system is reported to have a number of important functions including sound conduction, maintaining pressure regulation, gas exchange, material secretion, waste excretion and middle ear cavity aeration1–3. Following mastoidectomy, mastoid obliteration procedures are focussed on filling the mastoid cavity and do not repair the mastoid bone structure. However, the only way to restore the vital functions of the mastoid is to fully regenerate the air cells within the bone.
Various polymers have been investigated for use as scaffolds in bone regeneration, including poly(DL-lactic acid-co-glycolic acid) (PLGA), which has a long history of use as a degradable implant material4. As previously described by Dhillon et al, blending PLGA with a plasticiser such as poly(ethylene glycol) (PEG) allows the production of temperature-sensitive particles with a reduced glass transition temperature (Tg) of 37°C5. When these particles are mixed with a carrier solution at room temperature a paste is formed that can be moulded into any size or shape. The paste then hardens into a solid scaffold at 37°C due to temperature-induced PEG-leaching. The ability of this scaffold system to regenerate bone in vivo has recently been demonstrated in a mouse calvarial defect model6.
We describe the modification of this scaffold technology for potential use in mastoid air cell regeneration. A key advantage is the ability to paste the material into a cavity of any size or shape followed by scaffold solidification at body temperature. To optimise the scaffold structure we describe the inclusion of alginate beads in the formulation to act as a porogen, creating scaffolds with a high level of macroporosity. We also investigate in vitro release of ciprofloxacin from the scaffolds with the intention of providing local, sustained antibiotic delivery to decrease post-operative infections or continuation of underlying infections of the middle ear.
2. Methods
2.1 Mastoid bone sample preparation
A section of mastoid bone approximately 3 cm × 3 cm was excised from fresh frozen human cadaveric temporal bone.
2.2 PLGA/PEG particle production
Particles were fabricated from blends of poly(DL-lactic acid-co-glycolic acid) (PLGA) 85:15 DLG 4CA, 53 kDa (Lakeshore Biomaterials, USA) and poly(ethylene glycol) 400 (PEG) (Sigma Aldrich, UK) as previously described (Dhillon et al., 2011). Briefly, a mixture of 93.5%:6.5% PLGA:PEG (w/v) was blended at 80–90°C, mixed and allowed to cool. Cooled polymer was ground into particles and sieved to obtain the 100–200 μm size fraction.
2.3 Alginate bead fabrication
A solution of 1% (w/v) sodium alginate (Acros organics, UK) was prepared in phosphate buffered saline. 10 ml of alginate solution was loaded into a syringe and ejected from a 23 G needle into a gently agitated bath of 25 ml 5% (w/v) calcium chloride (Sigma-Aldrich, UK). The beads were strained using a 100 μm sieve and rinsed with dH2O.
2.4 Scaffold preparation
Triplicate scaffolds were prepared in PTFE moulds producing cylindrical scaffolds of 12 mm length and 6 mm diameter. PLGA/PEG particles were mixed with saline and varying alginate bead masses to give a range of weight percentages from 0% to 80% alginate. Ratio of PLGA/PEG particles to saline was 1:0.6 (particles:saline). Particle paste was packed into the mould which was placed at 37°C for 4 hours to allow scaffold formation. For cell seeding experiments scaffolds were UV sterilised for 80 minutes.
Ciprofloxacin-loaded scaffolds were prepared for drug release assays using two different methods, A and B. In method A, ciprofloxacin solution (Sigma-Aldrich, UK) was mixed with the PLGA/PEG particles and alginate beads to produce a paste which was used to prepare scaffolds as described above (100 μg ciprofloxacin per scaffold). In method B, ciprofloxacin was added into the PLGA/PEG melt-blend at 80–90°C on a hotplate. The ciprofloxacin-PLGA/PEG particles were then fabricated as described in section 2.2 and used to produce scaffolds as described above (100 μg ciprofloxacin per scaffold).
2.5 Porosity measurements
Triplicate scaffolds prepared using 40% PLGA/PEG-60% alginate were sintered for 4 hours at 37°C followed by freeze-drying for 24 hours causing the alginate beads to dehydrate. The porosity of the scaffolds was calculated using the bulk density and particle density values as follows: 1 − BD / PD × 100 = % Porosity.
2.6 X-ray micro-computed tomography
High-resolution three-dimensional images of the mastoid bone and scaffold samples were acquired using a laboratory X-ray micro-computed tomography system (μCT-40, Scanco Medical AG, Brüttisellen, Switzerland). The reconstructed field of view was 12.3mm for the scaffold specimens and 30.7mm for the mastoid bone specimen, with isotropic nominal resolutions of 12μm and 15μm, respectively. Approximately 1cm in length was scanned, spanning 800 slices (scaffolds) or 667 slices (bone). The bone or scaffold structure was binarized using a visually selected fixed global threshold. The surface of the 3D structure was triangulated and displayed using the manufacturer’s software (μCT Ray v3.8, Scanco Medical AG).
2.7 Mechanical properties
PLGA/PEG particles sinter over time at 37°C temperature to form scaffolds. Assessing the mechanical properties of the scaffolds over time at 37°C reveals if solidification due to particle sintering has occurred. Triplicate scaffolds prepared using 40% PLGA/PEG-60% alginate were sintered at 37°C for 3, 4 or 5 hours. Compressive strength was tested using a TA.HD+ texture analyser (Stable Microsystems).
2.8 Scanning electron microscopy
Scaffolds were mounted on aluminium stubs and sputter-coated with gold at an argon current rate of 30 mA for 3 minutes. Scaffold structural morphology was examined using a scanning electron microscope (JEOL JSM-6060LV) at 10 kV.
2.9 Human bone marrow mesenchymal stem cell culture
Human bone marrow mesenchymal stem cells were cultured in Mesenchymal Stem Cell Media (TCS Cell Works, UK) supplemented with 5% foetal calf serum (FCS), 1% Mesenchymal stem cell growth supplement, 1% penicillin/streptomycin solution (all TCS Cell Works, UK), 1% L-Glutamine (GibcoBRL, UK) (200 mM) and 1% non-essential amino acids (Sigma-Aldrich, Poole, UK). Cells were maintained in a humidified tissue-culture incubator at 37 °C and with 5 % CO2.
2.10 Cell seeding on scaffolds
Triplicate scaffolds for both test and control groups were pre-wet with 100 μl media and placed in the incubator at 37°C for 1 hour. 2 × 105 cells in a 30 μl suspension was seeded per scaffold. The scaffolds were incubated for 2 hours at 37°C prior to the addition of 1ml media to each well.
2.11 Prestoblue metabolic activity assay
The Prestoblue metabolic activity assay was performed on triplicate scaffolds 1, 2, 3, 5 and 7 days post-seeding. Each scaffold was submerged in 1ml of media. 111 μl of Prestoblue (Invitrogen Life Sciences, UK) was added to each well and the scaffolds were incubated at 37°C for 25 minutes. Triplicate 100 μl media samples from each well were read on a Tecan plate reader with the excitation wavelength set to 535 nm and the emission wavelength set at 615 nm.
2.12 Live/Dead Staining
Live/Dead staining was performed on triplicate scaffolds 3 days post-seeding. A staining solution was prepared in PBS containing 20 μg ml−1 propidium iodide (Sigma-Aldrich, UK) and 1 μg ml−1 fluorescein diacetate (Calbiochem, UK). 1 ml of the staining solution was added to each scaffold and incubated at room temperature for 15 minutes before visualisation using a Leica LCS confocal macroscope. Live cells stain green and dead cells stain red.
2.13 Ciprofloxacin release assay
Triplicate scaffolds loaded with 100 μg ciprofloxacin prepared using method A or B as described in section 2.4 were placed in 3 ml PBS and incubated at 37°C. At various time intervals the PBS was replaced with 3 ml fresh PBS and 100 μl of PBS containing released drug was sampled in triplicate at 315 nm on a Tecan plate reader and concentration of drug measured. Non-drug loaded scaffolds containing only PBS were used as a control for background absorbance values. Data is presented as cumulative drug release as a function of time.
3. Results
3.1 Microstructure of human mastoid bone
Human mastoid bone microstructure was analysed using micro-computed tomography (micro-CT). The porosity of the bone was 83%. The physiology of the bone was observed in both X-ray images and 3D reconstruction (Figure 1). Pores were observed throughout the structure with sizes averaging 1.3mm, as measured by micro-CT.
Figure 1. Microstructure of human mastoid bone.
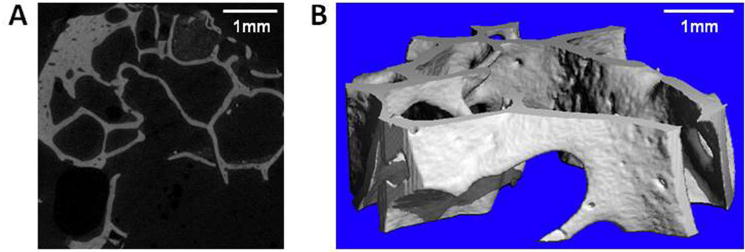
(A) Micro-CT x-ray slice image of human mastoid bone. (B) Micro-CT 3D reconstruction of human mastoid bone.
3.2 Microstructure of PLGA/PEG-alginate composite scaffolds
Scaffolds containing different amounts of alginate beads were sintered for 4 hours. At 37°C, alginate degrades in 3–4 days whereas PLGA/PEG particles degrade in 2–3 months. Therefore the alginate beads will degrade rapidly in vivo leaving a highly porous PLGA/PEG scaffold structure. To mimic this degradation effect the scaffolds were freeze-dried for 24 hours prior to porosity measurements to dehydrate the alginate beads. Porosity was assessed by density measurements and increased from 43% (100% PLGA/PEG-0% alginate) to 78% (20% PLGA/PEG-80% alginate) (Figure 2A). 20% PLGA/PEG-80% alginate scaffolds were fragile when handled during cell culture experiments, therefore 40% PLGA/PEG-60% alginate scaffolds were used in all subsequent experiments (herein referred to as ‘PLGA/PEG-alginate’ scaffolds).
Figure 2. Microstructure of PLGA/PEG-alginate scaffolds.
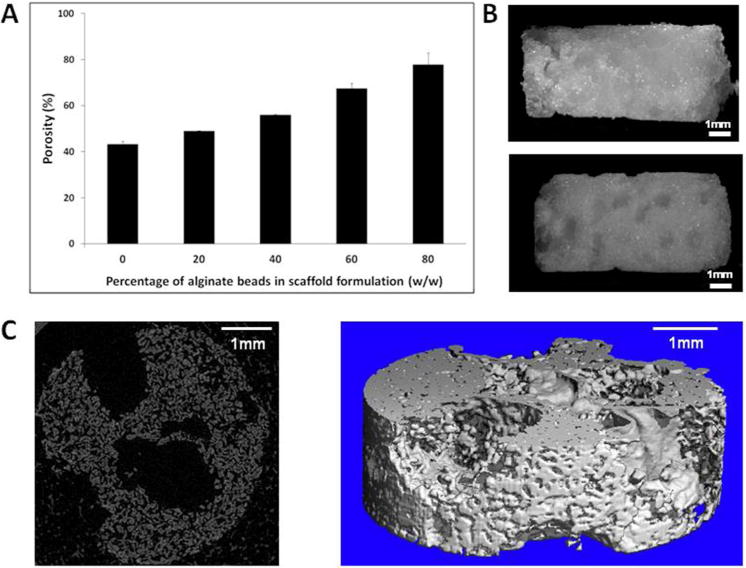
(A) Porosity of PLGA/PEG-alginate scaffolds calculated by density measurements. (B) Light microscope images of 40% PLGA/PEG-60% alginate scaffolds before freeze-drying (top image) and after freeze-drying for 24 hours (bottom image). (C) Micro-CT x-ray slice and 3D reconstruction of 40% PLGA/PEG-60% alginate scaffold after freeze-drying for 24 hours.
Light microscope images of PLGA/PEG-alginate scaffolds were taken before and after freeze-drying (Figure 2B). The residual pores created by the dehydrated alginate beads can be seen in the image after freeze-drying and in the micro-CT images in Figure 2C. PLGA/PEG-alginate scaffolds were submerged in saline for 2 weeks at 37°C to visually compare the physical structure of mastoid bone with the scaffolds following alginate bead degradation at this temperature (Figure 3).
Figure 3. Image comparison of human mastoid bone and PLGA/PEG scaffold.
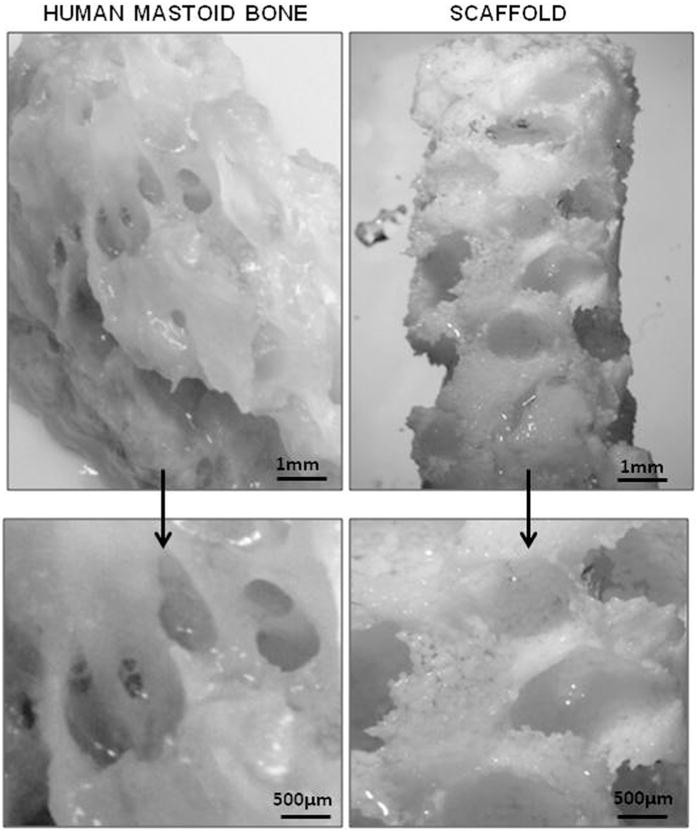
Light microscope image of human mastoid bone section (left) and 40% PLGA/PEG-60% alginate scaffold (right). Scaffold was submerged in saline at 37°C for 2 weeks prior to imaging.
3.3 Particle sintering within PLGA/PEG-alginate scaffolds
PLGA/PEG-alginate scaffolds were sintered for 3, 4 and 5 hours at 37°C. Compressive strength tests were performed to assess the ability of the PLGA/PEG particles to sinter in the presence of alginate beads (Figure 4). Following 3 hours at 37°C the compressive strength values were 20 kPa on average. This increased over the next hour at 37°C to 0.2 MPa and further increased to 0.38 MPa by 5 hours. The microstructure of a scaffold sintered for 4 hours at 37°C and freeze-dried for 24 hours is shown in the SEM image in Figure 4B.
Figure 4. PLGA/PEG particle sintering within PLGA/PEG-alginate scaffolds at 37C.
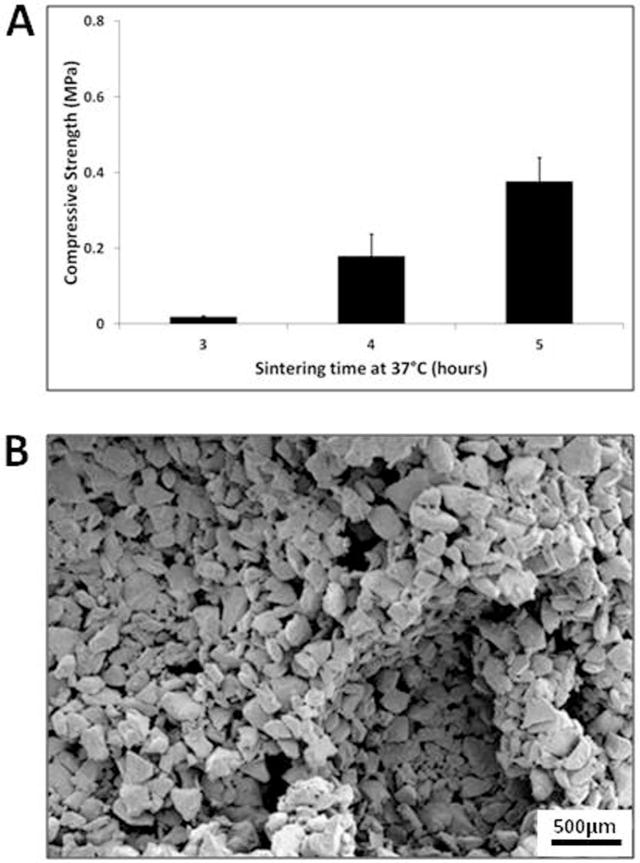
(A) Compressive strength of 40% PLGA/PEG-60% alginate scaffolds sintered for 3, 4 and 5 hours at 37°C. Error bars represent standard deviation. (B) Representative scanning electron microscopy image of a 40% PLGA/PEG-60% alginate scaffold sintered for 4 hours at 37°C and freeze-dried for 24 hours.
3.4 Cell proliferation on PLGA/PEG-alginate scaffolds
Human bone marrow mesenchymal stem cells (hBM-MSCs) were seeded onto the scaffolds and the Prestoblue cell viability assay was performed over 7 days (Figure 5A). The cell number increased during this time period from 2 × 105 cells per scaffold to 6.8 × 105 cells per scaffold, demonstrating hBM-MSC proliferation. Scaffolds were subjected to Live/Dead staining 3 days post-cell seeding in order to visualise viable (green) and non-viable (red) hBM-MSCs within the scaffolds (Figure 5B).
Figure 5. Proliferation of human bone marrow-derived mesenchymal stem cells on PLGA/PEG-alginate scaffolds.
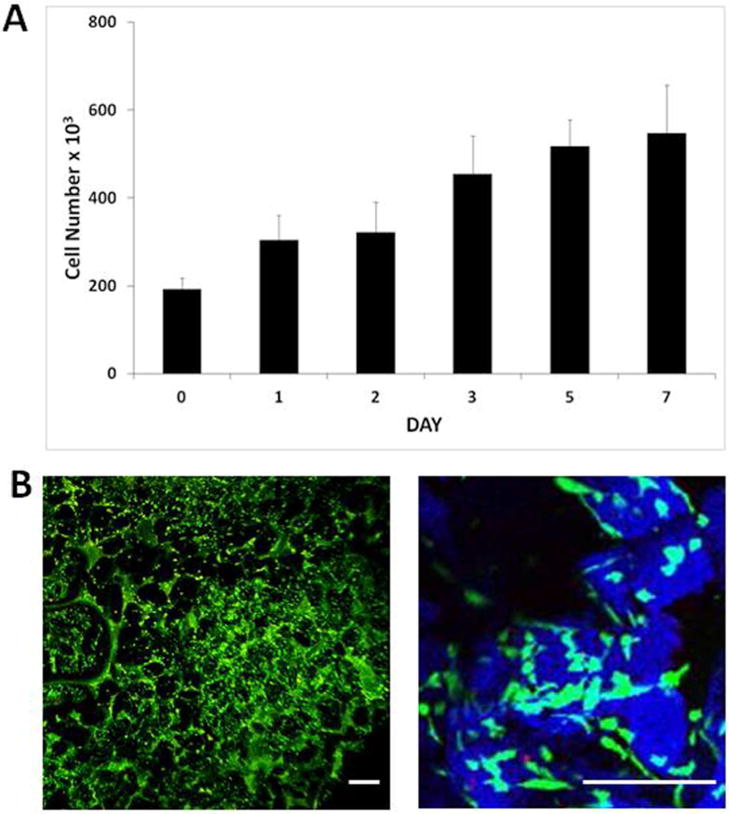
(A) Number of viable human bone marrow mesenchymal stem cells (hBM-MSCs) on 40% PLGA/PEG-60% alginate as measured by the Prestoblue metabolic activity assay over 7 days in culture. Error bars represent standard deviation. (B) Representative images of hBM-MSC-seeded 40% PLGA/PEG-60% alginate scaffold 3 days post-seeding following Live/Dead staining. PLGA/PEG particles rendered in blue using Leica LCS confocal macroscope software in order to visualise the particles (blue) and pore spaces between particles (black) Size bar = 200μm.
3.5 Antibiotic release from PLGA/PEG-alginate scaffolds
Ciprofloxacin loaded scaffolds were prepared as described in Section 2.4 using two different loading methods for scaffold preparation, A and B. In method A, ciprofloxacin solution was mixed with the PLGA/PEG particles and alginate beads to produce a paste which was used to prepare scaffolds as described above (100 μg ciprofloxacin per scaffold). In method B, the appropriate amount of ciprofloxacin was added into the PLGA/PEG melt-blend. High burst release of ciprofloxacin after 24 hours (73%) was observed with the scaffolds prepared using method A (Figure 6). Burst release was minimised to 30% using method B where the drug was mixed within the PLGA/PEG at the melt-blend phase. Ciprofloxacin release from scaffolds prepared using method A slowed down to an average of 0.1% drug release per day from day 7 until day 53 when release stopped at a total of 97% ciprofloxacin released.
Figure 6. Ciprofloxain release from PLGA/PEG-alginate scaffolds.
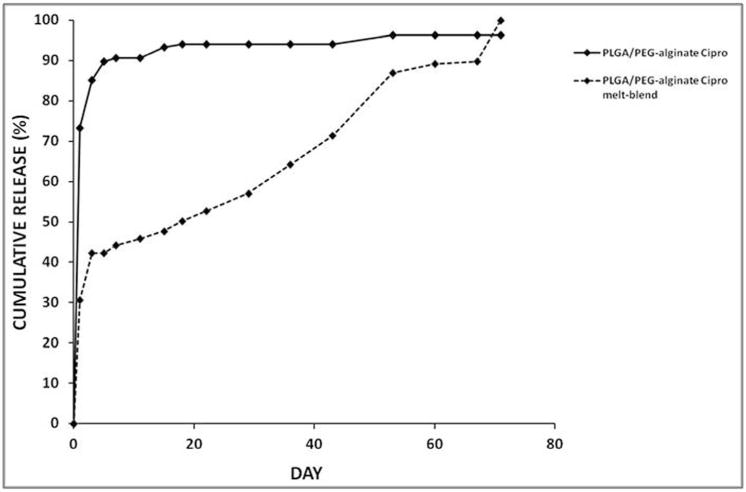
Cumulative release profiles for ciprofloxacin from 40% PLGA/PEG-60% alginate scaffolds represented as percentage of total loaded drug released. Two different drug loading methods were used, method A (‘PLGA/PEG-alginate cipro’) and method B (PLGA/PEG-alginate cipro melt-blend’).
4. Discussion
An ideal material for mastoid air cell regeneration has not yet been identified, therefore alternatives have been investigated in recent years including biphasic calcium phosphate granules mixed with fibrin sealant and polycaprolactone-tricalcium phosphate composites7,8. These materials contain ceramic components which may have similar disadvantages to hydroxyapatite for this application, such as extrusion and infection. Bioactive glasses such as BonAlive have recently been reported to show promise in mastoid bone repair procedures, however the glass granules occasionally leak into the ear canal9. Biodegradable materials have also been assessed including cell-loaded hyaluronic acid gel and growth factor-loaded collagen implants10,11. These materials show promise for bone repair, however they lack the macroporosity required to regenerate the highly porous structure of the mastoid air cell system.
Here we describe the development of a biodegradable polymer scaffold tailored for mastoid air cell regeneration. The scaffold is based on PLGA/PEG particle paste which can be moulded into any size or shape and hardens into a solid scaffold at 37°C5. To our knowledge this is the first description of a biodegradable particulate polymer scaffold that can be pasted into a cavity and hardens in vivo for this application. The ability to paste the scaffold around the mastoid cavity offers improved clinical application and tissue approximation when compared to implants.
To aid scaffold design it is important to take into account the structure of the mastoid air cells, therefore mastoid bone was visualised using light microscopy and micro-CT. The overall porosity of the bone was measured at 83% with an average pore diameter of 1.3 mm. Our intention was to optimise scaffold structure with regard to generating large air cells mimicking those observed in mastoid bone. Alginate beads of 1mm–1.5mm diameter were successfully used as porogens to create pores in the PLGA/PEG scaffolds. Scaffold porosity was enhanced from 43% to 78% with increasing amount of alginate beads.
In the current study, the mastoid bone sample displayed a porosity level of 83%. The scaffold formulation with a porosity value closest to that of the mastoid bone sample was 20% PLGA/PEG-80% alginate, at 78%. Due to the fact scaffolds produced using this formulation were fragile when handled during cell culture experiments, 40% PLGA/PEG-60% alginate scaffolds (64% porous) were used in all subsequent experiments. It will be important to assess the ability of both formulations to regenerate mastoid air cells in future in vivo studies in order to determine the best formulation to take forward for clinical use.
The ability of the scaffold to harden at body temperature is an important property as it ensures the polymer is retained where it is applied. This hardening process could potentially allow the polymer to act as an obliteration material in addition to promoting air cell regeneration within the mastoid bone. As the formulation contained 60% alginate beads it was necessary to assess the ability of the PLGA/PEG particles to sinter in this formulation. Scaffold hardening at 37°C was demonstrated by an increase in scaffold compressive strength over time, confirming the ability of the PLGA/PEG particles to fuse into a solid scaffold in the presence of the alginate beads.
PLGA/PEG scaffolds are capable of regenerating bone in vivo. The formulation used in the current study contained 40% PLGA/PEG and 60% alginate, therefore the ability of human bone marrow mesenchymal stem cells (hBM-MSCs) to grow on the scaffolds in vitro was assessed. Our results demonstrate attachment and proliferation of hBM-MSCs on the scaffolds over a seven day time period, demonstrating the potential for cells to proliferate on the scaffolds which has implications for their successful use for bone repair in vivo.
To reduce the likelihood of secondary complications arising from post-operative infection or recurrence of underlying middle ear infections, we incorporated cirofloxacin into the PLGA/PEG-alginate scaffold formulation as it is commonly used in the treatment of middle ear infections. We used two different antibiotic loading methods. Method A involved mixing the drug into the PLGA/PEG and alginate bead paste prior to scaffold formation, trapping the drug inside the scaffold during solidification at 37°C. Method B involved mixing the drug into the PLGA/PEG at the melt-blend phase, therefore loading it inside the particles. Incorporation of ciprofloxacin into polymer melt-blends at high temperature (80–150°C) has been reported using polycaprolactone/poloxamine, with the antibiotic being shown to retain its antibacterial activity following exposure to these high temperatures12. In our method the ciprofloxacin was heated to 80°C. A high burst release of 73% ciprofloxacin after 24 hours was observed with the scaffolds prepared using method A, caused by drug on the surface of the particles releasing immediately. This burst release was minimised to 30% using method B, as the drug is trapped inside the particles therefore reducing the amount of drug available for immediate release. Scaffolds produced using method A released for 7 weeks, with a daily release rate of approximately 0.1% per day. Scaffolds produced using method B generated an improved release profile with a lower burst release phase followed by 10 weeks release of approximately 1% per day.
5. Conclusions
We describe the development of a porous, biodegradable PLGA/PEG-alginate scaffold that can be pasted into a cavity of any size or shape. Scaffold porosity can be controlled by varying the amount of alginate beads in the formulation, with a maximum of porosity of 78% achieved. The scaffolds support the growth of hBM-MSCs and display sustained release of ciprofloxacin for 7–10 weeks in vitro. Future studies will focus on the assessment of this scaffold formulation for mastoid air cell regeneration in vivo.
Acknowledgments
The authors would like to thank Andrew Burghardt (University of California San Francisco) for performing the micro-computed tomography. This work was in part funded by the EPSRC Centre for Innovative Manufacturing in Regenerative Medicine and the European Community’s FP7 project Biodesign EUFP7-NMP.20102.3-1 (Grant: 262948). The research leading to these results received funding from the European Research Council under the European Community’s Seventh FP7 project FP72007–2013 (Grant: 227845).
Financial Disclosure: This work was funded by (1) EPSRC Centre for Innovative Manufacturing in Regenerative Medicine (2) European Community’s FP7 project Biodesign EUFP7-NMP.20102.3-1 (Grant: 262948) (3) European Community’s FP7 ERC project (Grant: 227845).
Footnotes
Any financial interests the authors may have: None
Conflict of interest: None
Level of evidence: N/A
References
- 1.Cinamon U, Sade J. Mastoid and tympanic membrane as pressure buffers: a quantitative study in a middle ear cleft model. Otol Neurotol. 2003 Nov;24(6):839–842. doi: 10.1097/00129492-200311000-00002. [DOI] [PubMed] [Google Scholar]
- 2.Doyle WJ, Alper CM, Banks JM, Swarts JD. Rate of nitrous oxide exchange across the middle ear mucosa in monkeys before and after blockage of the mastoid antrum. Otolaryngol Head Neck Surg. 2003 May;128(5):732–741. doi: 10.1016/S0194-59980223309-4. [DOI] [PubMed] [Google Scholar]
- 3.Kanemaru S-i, Nakamura T, Omori K, Magrufov A, Yamashita M, Ito J. Regeneration of Mastoid Air Cells in Clinical Applications by In Situ Tissue Engineering. The Laryngoscope. 2005;115(2):253–258. doi: 10.1097/01.mlg.0000154728.23657.3a. [DOI] [PubMed] [Google Scholar]
- 4.Howard D, Buttery LD, Shakesheff KM, Roberts SJ. Tissue engineering: strategies, stem cells and scaffolds. J Anat. 2008 Jul;213(1):66–72. doi: 10.1111/j.1469-7580.2008.00878.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Dhillon A, Schneider P, Kuhn G, et al. Analysis of sintered polymer scaffolds using concomitant synchrotron computed tomography and in situ mechanical testing. J Mater Sci Mater Med. 2011 Dec;22(12):2599–2605. doi: 10.1007/s10856-011-4443-z. [DOI] [PubMed] [Google Scholar]
- 6.Rahman CV, Ben-David D, Dhillon A, et al. Controlled release of BMP-2 from a sintered polymer scaffold enhances bone repair in a mouse calvarial defect model. J Tissue Eng Regen Med. 2012 Jun 8; doi: 10.1002/term.1497. [DOI] [PubMed] [Google Scholar]
- 7.Bagot d’Arc M, Daculsi G, Emam N. Biphasic ceramics and fibrin sealant for bone reconstruction in ear surgery. Ann Otol Rhinol Laryngol. 2004 Sep;113(9):711–720. doi: 10.1177/000348940411300907. [DOI] [PubMed] [Google Scholar]
- 8.Jang CH, Cho YB, Kim JS, Lee HJ, Kim GH. Regeneration of mastoid air cells using polycaprolactone/β-tricalcium phosphate biocomposites: An experimental study. The Laryngoscope. 2012;122(3):660–664. doi: 10.1002/lary.22503. [DOI] [PubMed] [Google Scholar]
- 9.Silvola JT. Mastoidectomy Cavity Obliteration with Bioactive Glass. Otolaryngology – Head and Neck Surgery. 2012 Jul 1;147(1):119–126. doi: 10.1177/0194599812438168. 2012. [DOI] [PubMed] [Google Scholar]
- 10.Jang CH, Park H, Cho YB, Song CH. Mastoid obliteration using a hyaluronic acid gel to deliver a mesenchymal stem cells-loaded demineralized bone matrix: an experimental study. International journal of pediatric otorhinolaryngology. 2008 Nov;72(11):1627–1632. doi: 10.1016/j.ijporl.2008.07.017. [DOI] [PubMed] [Google Scholar]
- 11.Nishizaki K, Tsujigiwa H, Takeda Y, et al. Mastoid obliteration by BMP- 2/collagen composites: An experimental study using tissue engineering. American journal of otolaryngology. 2003;24(1):14–18. doi: 10.1053/ajot.2003.13. [DOI] [PubMed] [Google Scholar]
- 12.Puga AM, Rey-Rico A, Magarinos B, Alvarez-Lorenzo C, Concheiro A. Hot melt poly-epsilon-caprolactone/poloxamine implantable matrices for sustained delivery of ciprofloxacin. Acta Biomater. 2012 Apr;8(4):1507–1518. doi: 10.1016/j.actbio.2011.12.020. [DOI] [PubMed] [Google Scholar]


