Abstract
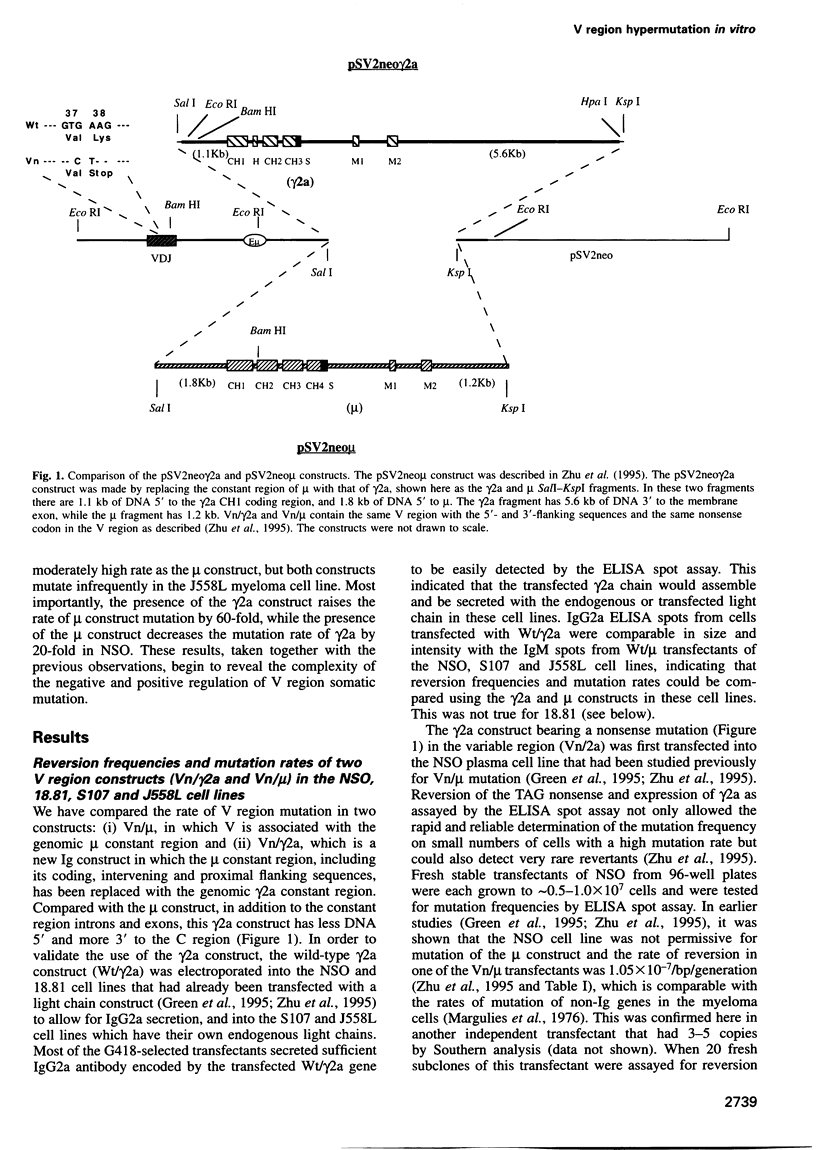
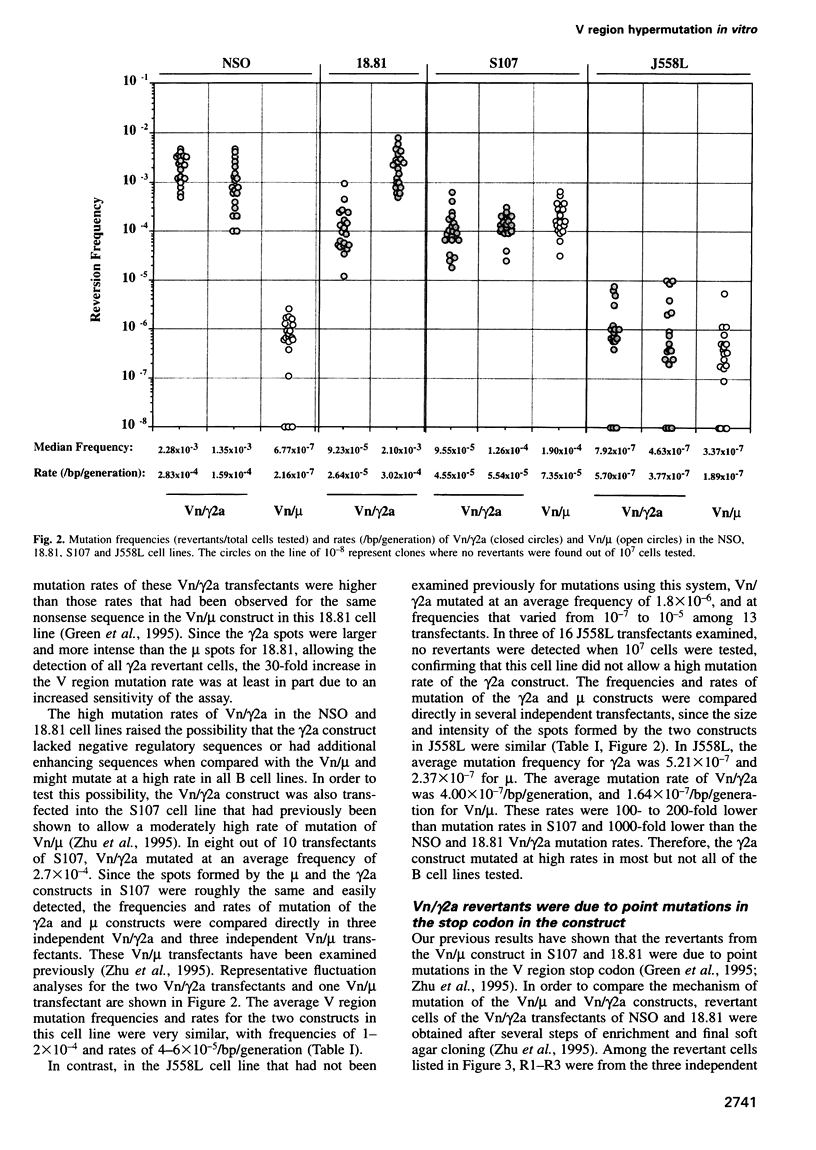
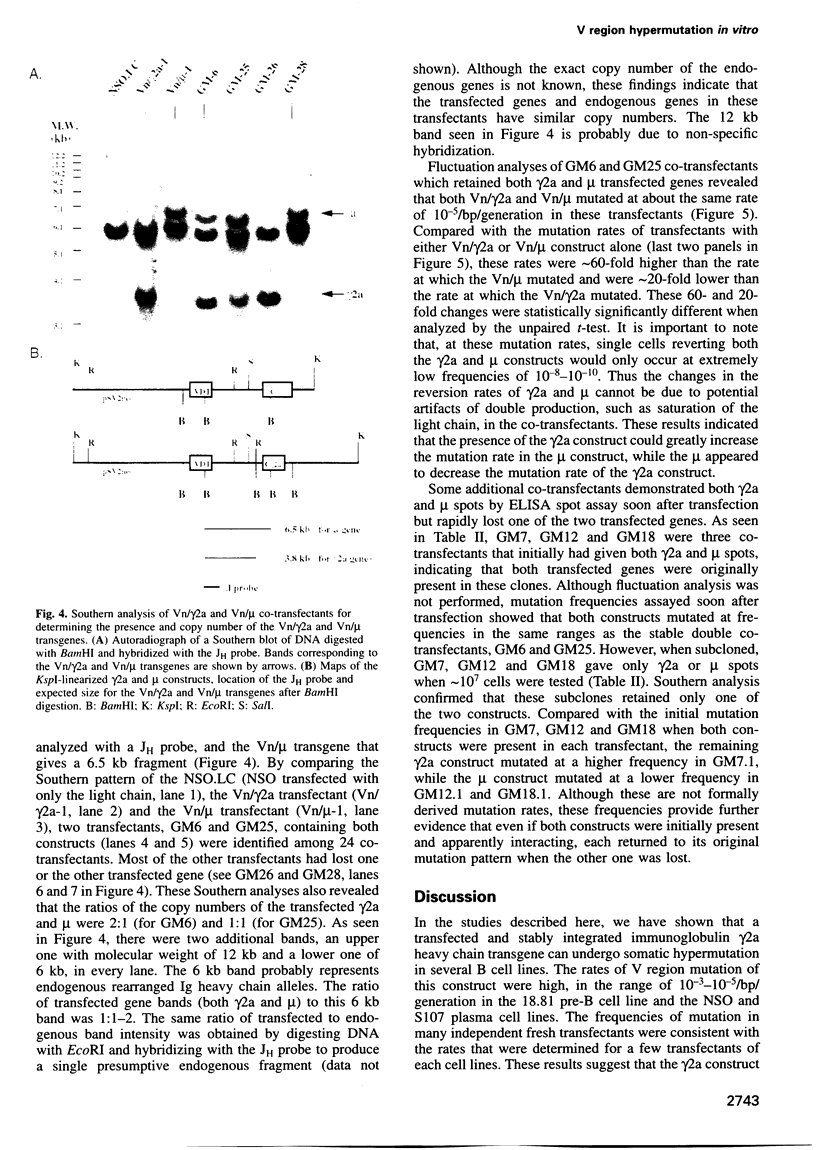
We have established B cell culture systems in which transfected and stably integrated Ig constructs spontaneously undergo high rates of variable (V) region mutation. Mutation rates were determined using reversion analysis of an Ig V region nonsense codon (Vn). A construct (Vn/gamma2a) in which a Vn was associated with the gamma2a constant region and its intervening and immediate flanking sequences mutated at a high rate of 2.2 x 10(-4)/bp/generation in the NSO myeloma cell line. This same Vn, when associated with the mu constant region (Vn/mu), mutated at a 1000-fold lower rate in NSO. The Vn/gamma2a construct also mutated at high rates in the 18.81 pre-B and the S107 myeloma cell lines and at a low rate in the J558 myeloma cell line. In NSO, the presence of the gamma2a construct raised the mutation rate of the mu construct and the mu decreased the mutation rate of gamma2a. These results suggest that there is both positive and negative regulation of V region mutation and that different cell lines express different combinations and/or amounts of trans-acting factors that are involved in the mutational process.
Full text
PDF





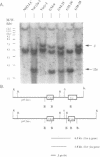
Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Apel M., Berek C. Somatic mutations in antibodies expressed by germinal centre B cells early after primary immunization. Int Immunol. 1990;2(9):813–819. doi: 10.1093/intimm/2.9.813. [DOI] [PubMed] [Google Scholar]
- Behar S. M., Lustgarten D. L., Corbet S., Scharff M. D. Characterization of somatically mutated S107 VH11-encoded anti-DNA autoantibodies derived from autoimmune (NZB x NZW)F1 mice. J Exp Med. 1991 Mar 1;173(3):731–741. doi: 10.1084/jem.173.3.731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berek C., Milstein C. Mutation drift and repertoire shift in the maturation of the immune response. Immunol Rev. 1987 Apr;96:23–41. doi: 10.1111/j.1600-065x.1987.tb00507.x. [DOI] [PubMed] [Google Scholar]
- Betz A. G., Milstein C., González-Fernández A., Pannell R., Larson T., Neuberger M. S. Elements regulating somatic hypermutation of an immunoglobulin kappa gene: critical role for the intron enhancer/matrix attachment region. Cell. 1994 Apr 22;77(2):239–248. doi: 10.1016/0092-8674(94)90316-6. [DOI] [PubMed] [Google Scholar]
- Betz A. G., Neuberger M. S., Milstein C. Discriminating intrinsic and antigen-selected mutational hotspots in immunoglobulin V genes. Immunol Today. 1993 Aug;14(8):405–411. doi: 10.1016/0167-5699(93)90144-a. [DOI] [PubMed] [Google Scholar]
- Betz A. G., Rada C., Pannell R., Milstein C., Neuberger M. S. Passenger transgenes reveal intrinsic specificity of the antibody hypermutation mechanism: clustering, polarity, and specific hot spots. Proc Natl Acad Sci U S A. 1993 Mar 15;90(6):2385–2388. doi: 10.1073/pnas.90.6.2385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Both G. W., Taylor L., Pollard J. W., Steele E. J. Distribution of mutations around rearranged heavy-chain antibody variable-region genes. Mol Cell Biol. 1990 Oct;10(10):5187–5196. doi: 10.1128/mcb.10.10.5187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng G. H., Skoultchi A. I. Rapid induction of polyadenylated H1 histone mRNAs in mouse erythroleukemia cells is regulated by c-myc. Mol Cell Biol. 1989 Jun;9(6):2332–2340. doi: 10.1128/mcb.9.6.2332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chu Y. W., Marin E., Fuleihan R., Ramesh N., Rosen F. S., Geha R. S., Insel R. A. Somatic mutation of human immunoglobulin V genes in the X-linked HyperIgM syndrome. J Clin Invest. 1995 Mar;95(3):1389–1393. doi: 10.1172/JCI117791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chui Y. L., Lozano F., Jarvis J. M., Pannell R., Milstein C. A reporter gene to analyse the hypermutation of immunoglobulin genes. J Mol Biol. 1995 Jun 9;249(3):555–563. doi: 10.1006/jmbi.1995.0318. [DOI] [PubMed] [Google Scholar]
- Cohn M., Notani G., Rice S. A. Characterization of the antibody to the C-carbohydrate produced by a transplantable mouse plasmacytoma. Immunochemistry. 1969 Jan;6(1):111–123. doi: 10.1016/0019-2791(69)90183-9. [DOI] [PubMed] [Google Scholar]
- Cook W. D., Scharff M. D. Antigen-binding mutants of mouse myeloma cells. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5687–5691. doi: 10.1073/pnas.74.12.5687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Datta A., Jinks-Robertson S. Association of increased spontaneous mutation rates with high levels of transcription in yeast. Science. 1995 Jun 16;268(5217):1616–1619. doi: 10.1126/science.7777859. [DOI] [PubMed] [Google Scholar]
- French D. L., Laskov R., Scharff M. D. The role of somatic hypermutation in the generation of antibody diversity. Science. 1989 Jun 9;244(4909):1152–1157. doi: 10.1126/science.2658060. [DOI] [PubMed] [Google Scholar]
- Galfrè G., Milstein C. Preparation of monoclonal antibodies: strategies and procedures. Methods Enzymol. 1981;73(Pt B):3–46. doi: 10.1016/0076-6879(81)73054-4. [DOI] [PubMed] [Google Scholar]
- Gartel A. L. Identification of multiple B-cell transcriptional repressor elements in S mu-C mu intron of mouse IgH chain locus. Somat Cell Mol Genet. 1994 Sep;20(5):371–379. doi: 10.1007/BF02257454. [DOI] [PubMed] [Google Scholar]
- Green N. S., Rabinowitz J. L., Zhu M., Kobrin B. J., Scharff M. D. Immunoglobulin variable region hypermutation in hybrids derived from a pre-B- and a myeloma cell line. Proc Natl Acad Sci U S A. 1995 Jul 3;92(14):6304–6308. doi: 10.1073/pnas.92.14.6304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacob J., Kelsoe G., Rajewsky K., Weiss U. Intraclonal generation of antibody mutants in germinal centres. Nature. 1991 Dec 5;354(6352):389–392. doi: 10.1038/354389a0. [DOI] [PubMed] [Google Scholar]
- Jacob J., Przylepa J., Miller C., Kelsoe G. In situ studies of the primary immune response to (4-hydroxy-3-nitrophenyl)acetyl. III. The kinetics of V region mutation and selection in germinal center B cells. J Exp Med. 1993 Oct 1;178(4):1293–1307. doi: 10.1084/jem.178.4.1293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kocks C., Rajewsky K. Stable expression and somatic hypermutation of antibody V regions in B-cell developmental pathways. Annu Rev Immunol. 1989;7:537–559. doi: 10.1146/annurev.iy.07.040189.002541. [DOI] [PubMed] [Google Scholar]
- Lang R. B., Stanton L. W., Marcu K. B. On immunoglobulin heavy chain gene switching: two gamma 2b genes are rearranged via switch sequences in MPC-11 cells but only one is expressed. Nucleic Acids Res. 1982 Jan 22;10(2):611–630. doi: 10.1093/nar/10.2.611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leanderson T., Källberg E., Gray D. Expansion, selection and mutation of antigen-specific B cells in germinal centers. Immunol Rev. 1992 Apr;126:47–61. doi: 10.1111/j.1600-065x.1992.tb00630.x. [DOI] [PubMed] [Google Scholar]
- Lebecque S. G., Gearhart P. J. Boundaries of somatic mutation in rearranged immunoglobulin genes: 5' boundary is near the promoter, and 3' boundary is approximately 1 kb from V(D)J gene. J Exp Med. 1990 Dec 1;172(6):1717–1727. doi: 10.1084/jem.172.6.1717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levy N. S., Malipiero U. V., Lebecque S. G., Gearhart P. J. Early onset of somatic mutation in immunoglobulin VH genes during the primary immune response. J Exp Med. 1989 Jun 1;169(6):2007–2019. doi: 10.1084/jem.169.6.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lozano F., Rada C., Jarvis J. M., Milstein C. Affinity maturation leads to differential expression of multiple copies of a kappa light-chain transgene. Nature. 1993 May 20;363(6426):271–273. doi: 10.1038/363271a0. [DOI] [PubMed] [Google Scholar]
- Luria S. E., Delbrück M. Mutations of Bacteria from Virus Sensitivity to Virus Resistance. Genetics. 1943 Nov;28(6):491–511. doi: 10.1093/genetics/28.6.491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Margulies D. H., Kuehl W. M., Scharff M. D. Somatic cell hybridization of mouse myeloma cells. Cell. 1976 Jul;8(3):405–415. doi: 10.1016/0092-8674(76)90153-7. [DOI] [PubMed] [Google Scholar]
- Neuberger M. S., Milstein C. Somatic hypermutation. Curr Opin Immunol. 1995 Apr;7(2):248–254. doi: 10.1016/0952-7915(95)80010-7. [DOI] [PubMed] [Google Scholar]
- Oi V. T., Morrison S. L., Herzenberg L. A., Berg P. Immunoglobulin gene expression in transformed lymphoid cells. Proc Natl Acad Sci U S A. 1983 Feb;80(3):825–829. doi: 10.1073/pnas.80.3.825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oi V. T., Morrison S. L., Herzenberg L. A., Berg P. Immunoglobulin gene expression in transformed lymphoid cells. Proc Natl Acad Sci U S A. 1983 Feb;80(3):825–829. doi: 10.1073/pnas.80.3.825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Piechaczyk M., Blanchard J. M., Marty L., Dani C., Panabieres F., El Sabouty S., Fort P., Jeanteur P. Post-transcriptional regulation of glyceraldehyde-3-phosphate-dehydrogenase gene expression in rat tissues. Nucleic Acids Res. 1984 Sep 25;12(18):6951–6963. doi: 10.1093/nar/12.18.6951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reynaud C. A., Garcia C., Hein W. R., Weill J. C. Hypermutation generating the sheep immunoglobulin repertoire is an antigen-independent process. Cell. 1995 Jan 13;80(1):115–125. doi: 10.1016/0092-8674(95)90456-5. [DOI] [PubMed] [Google Scholar]
- Rogozin I. B., Kolchanov N. A. Somatic hypermutagenesis in immunoglobulin genes. II. Influence of neighbouring base sequences on mutagenesis. Biochim Biophys Acta. 1992 Nov 15;1171(1):11–18. doi: 10.1016/0167-4781(92)90134-l. [DOI] [PubMed] [Google Scholar]
- Shan H., Shlomchik M., Weigert M. Heavy-chain class switch does not terminate somatic mutation. J Exp Med. 1990 Aug 1;172(2):531–536. doi: 10.1084/jem.172.2.531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siden E. J., Baltimore D., Clark D., Rosenberg N. E. Immunoglobulin synthesis by lymphoid cells transformed in vitro by Abelson murine leukemia virus. Cell. 1979 Feb;16(2):389–396. doi: 10.1016/0092-8674(79)90014-x. [DOI] [PubMed] [Google Scholar]
- Sohn J., Gerstein R. M., Hsieh C. L., Lemer M., Selsing E. Somatic hypermutation of an immunoglobulin mu heavy chain transgene. J Exp Med. 1993 Feb 1;177(2):493–504. doi: 10.1084/jem.177.2.493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tonegawa S. Somatic generation of antibody diversity. Nature. 1983 Apr 14;302(5909):575–581. doi: 10.1038/302575a0. [DOI] [PubMed] [Google Scholar]
- Wabl M., Burrows P. D., von Gabain A., Steinberg C. Hypermutation at the immunoglobulin heavy chain locus in a pre-B-cell line. Proc Natl Acad Sci U S A. 1985 Jan;82(2):479–482. doi: 10.1073/pnas.82.2.479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weigert M. G., Cesari I. M., Yonkovich S. J., Cohn M. Variability in the lambda light chain sequences of mouse antibody. Nature. 1970 Dec 12;228(5276):1045–1047. doi: 10.1038/2281045a0. [DOI] [PubMed] [Google Scholar]
- Zhu M., Rabinowitz J. L., Green N. S., Kobrin B. J., Scharff M. D. A well-differentiated B-cell line is permissive for somatic mutation of a transfected immunoglobulin heavy-chain gene. Proc Natl Acad Sci U S A. 1995 Mar 28;92(7):2810–2814. doi: 10.1073/pnas.92.7.2810. [DOI] [PMC free article] [PubMed] [Google Scholar]