Abstract
Pancreatic cystic lesions (PCLs) are being increasingly identified in recent years. They show a wide spectrum of imaging and clinical features. The diagnosis and discrimination of these lesions are very important because of the risk for concurrent or later development of malignancy. PCLs are usually first diagnosed and characterized by conventional imaging modalities such as trans-abdominal ultrasonography (US), computed tomography (CT) and magnetic resonance imaging (MRI). However, their ability to differentiate the benign and malignant lesions remains limited. Endoscopic US may be more helpful for the diagnosis and differentiation of PCLs because of its high resolution and better imaging characteristics than cross-sectional imaging modalities. It also allows for fine-needle aspiration (FNA) of cystic lesions for biochemical, cytological and DNA analysis that might be further helpful for diagnosis and differentiation. The management options of PCLs are to observe, endoscopic treatment or surgical resection. However, the decision for management is sometimes hampered by limitations in current diagnostic and tissue sampling techniques. As further diagnostic and non-invasive management options become available, clinical decision-making will become much easier for these lesions.
Keywords: Pancreas, cystic lesions, pseudocyst, mucinous cyst, intraductal papillary mucinous neoplasms (IPMNs), endoscopic ultrasonography (endoscopic US)
Introduction
Pancreatic cystic lesions (PCLs) are a broad group of pancreatic tumors that have varying demographical, morphological, histological and clinical characteristics. There has been a large increase in the number of patients with PCLs in recent years. The rising prevalence might be caused by significant improvement of imaging technologies, increased awareness of their existence and the growth of the aging population. Besides, PCLs are being discovered increasingly in patients who are otherwise asymptomatic (1). Image-based studies report prevalence of PCLs ranging from 1.2% to 19% (1-3). Among 24,039 computed tomography (CT) or magnetic resonance imaging (MRI) scans, 290 patients (1.2%) had pancreatic cysts, and a majority of the patients had no history of pancreatitis (4). In an autopsy series of 300 patients, 186 cystic lesions were found in 73 of 300 autopsy cases (24.3%) (5). The prevalence of cysts increases with age (2).
PCLs may be classified simply into two main classes such as non-neoplastic and neoplastic cysts (Box 1). Neoplastic cysts are more commonly defined as pancreatic cystic neoplasms (PCNs). It is important to distinguish non-neoplastic cysts from neoplastic or non-mucinous from mucinous cysts because the latter are considered being premalignant lesions. In general, non-neoplastic cysts account up to 80% of all PCLs. However, the rate of PCNs increases significantly with age (1,4). Diagnostic methods, management algorithms and treatment options of PCLs have been developed significantly in recent years. In this chapter, the major types of PCLs are reviewed based on the recent advances in diagnosis and management.
Box 1. Classification of pancreatic cystic lesions (PCLs).
| Non-neoplastic cysts |
| Pseudocyst |
| Simple or congenital cyst |
| Retention cyst |
| Neoplastic cysts [pancreatic cystic neoplasms (PCNs)] |
| Mucinous cystic lesions |
| Intraductal papillary mucinous neoplasm (IPMN) |
| Mucinous cystic neoplasm (MCN) |
| Non-Mucinous cystic neoplastic lesions |
| Serous cystic neoplasm (SCN) |
| Solid-pseudopapillary neoplasm (SPN) |
| Cystic neuroendocrine neoplasm |
| Acinar-cell cystic neoplasm |
| Other neoplastic lesions |
| Ductal adenocarcinoma with cystic degeneration |
Non-neoplastic cysts
Pseudocysts
Pancreatic pseudocysts are inflammatory fluid collections associated with pancreatitis and account approximately 80% of PCLs. They predominantly develop in adult men as a complication of alcoholic, biliary, or traumatic acute pancreatitis (6). The term “pseudocyst” refers to the fact that this cystic lesion has no epithelial lining and therefore is not a true cyst. Fluid collections adjacent to the pancreas are the most common complication of acute and chronic pancreatitis. In the setting of acute pancreatitis, a focal fluid collection located in or near the pancreas occurs without a wall of granulation and or fibrous tissue (7). The development of a well-defined wall composed of granulation or fibrous tissue distinguishes a pseudocyst from an acute fluid collection. The formation of a pseudocyst usually requires four or more weeks from the onset of acute pancreatitis. Without an antecedent episode of acute pancreatitis, pseudocyst may arise insidiously in patients with chronic pancreatitis (8). A pseudocyst is usually rich in pancreatic enzymes and is usually sterile. Pseudocysts are mostly single but can be multiple in 10% of cases. They are commonly round or oval, but some may be multilocular and irregular in shape (Figure 1). The size of pseudocysts varies from 2 to 20 cm (6-8).
Figure 1.
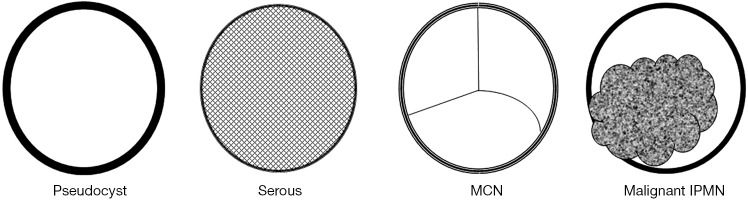
A schematization of morphologic details in common cysts of pancreas. MCN, mucinous cystic neoplasm; IPMN, intraductal papillary mucinous neoplasm.
Small pancreatic pseudocysts are usually intimately associated with the pancreas and are surrounded by a thin wall. Large pseudocysts may occupy spaces adjacent to the stomach and pancreas or remote areas, including the chest. The histologic features of pseudocyst walls are similar in all types of pseudocysts, consisting of fibrosis and inflammatory tissue. Most pancreatic pseudocysts originate from large or small leaks from the ductal system and persist because of the constant filling by pancreatic secretions (7).
The symptoms associated with chronic pancreatic pseudocysts are usually mild. The common symptoms are recurrent abdominal pain, early satiety, nausea and vomiting. In general, the size and the duration of the clinical course of the pseudocyst are the most important predictors of symptoms (9). With large pseudocysts, there may be a palpable fullness or a mass that is sensed by the patient or an examining physician. As a result of gastric compression, weight loss is observed in 20% of patients, and is a result of poor intake as well as maldigestion. Jaundice as manifest by icterus, dark urine, and pruritus, and acolic stools may be noted in 10% of patients. The onset of jaundice is usually slow, as a result of bile duct compression by the pseudocyst or the inflamed pancreas itself. Fever is unusual in chronic, uncomplicated pseudocysts and its presence should raise the suspicion of an occult infection of a pseudocyst (10).
Diagnosis
Pancreatic pseudocysts are commonly diagnosed based on clinically apparent clues or patient history, but in some instances this diagnosis can be difficult to conclude because the acute episode of pancreatitis may not be apparent or the patient may have mild chronic pancreatitis. Trans-abdominal ultrasonography (US) is usually the method of choice for the initial investigation of the pseudocysts. They usually appear as an echoic structure associated with distal acoustic enhancement. The sensitivity of US is inferior to CT which has a sensitivity of 90% to 100% for detection of pancreatic pseudocysts. A round, fluid filled structures surrounded by a thick, dense wall adjacent to pancreas on an abdominal CT in a patient with a history of pancreatitis is nearly diagnostic for pancreatic pseudocysts (6). The adjacent pancreas typically may reveal evidence of acute or chronic pancreatitis. Large pseudocysts may appear in the mediastinum or pelvis or involve the mesentery. Although pseudocysts are most commonly unilocular, fibrotic strands within the cavity may cause multiple septations, commonly encountered in patients with post pancreatitis, complex fluid collections. The pseudocyst cavity may also contain debris, blood, or infections that appear as high-attenuation areas within the fluid-filled cavity. It may be difficult to distinguish between pseudocysts and pancreatic mucinous cysts without the use of cyst fluid analysis in some cases. CT scans can also provide more detailed information regarding the surrounding anatomy and can demonstrate additional pathology. MRI and magnetic resonance cholangiopancreatography (MRCP) are also sensitive diagnostic methods but they usually do not add extra information on CT (11). Endoscopic retrograde cholangiopancreatography (ERCP) is not used for diagnosis of pseudocysts but it can be helpful for treatment in some cases.
EUS is usually used to further evaluate pancreatic cysts detected by other imaging modalities and most useful to distinguish pseudocysts from other PCLs (12). Pseudocysts appear as anechoic, fluid-filled structures adjacent to the upper GI tract and pancreas in EUS (Figure 2). Early fluid collections associated with acute pancreatitis will not be surrounded with a wall, whereas pseudocysts are often surrounded by a thick, hyperechoic rim. Calcifications in a cyst wall are highly suggestive of a mucinous cystadenoma, rather than a pseudocyst. Debris in the dependent portion of the cavity is common and may represent blood, infection, or necrotic material. Color Doppler of the wall will often reveal multiple, prominent vessels, including paragastric varices. EUS guided fine-needle aspiration (FNA) with cyst fluid analysis will differentiate between pseudocysts and neoplastic cysts in more than 90% of patients (Figure 3) (10). A high concentration of amylase in aspirated fluid is predictive of a connection with the main pancreatic duct and helps confirm the diagnosis of a pseudocyst duct and helps confirm the diagnosis of a pseudocyst. Pseudocysts should have relatively low levels of CEA and this might be helpful for differentiation from intraductal papillary mucinous neoplasms (IPMN) and mucinous cystic neoplasm (MCN) (13). The aspirated fluid is examined cytologically for degenerative debris, inflammatory cells and histiocytes. If there is cytologic evidence of epithelial cells with the cyst fluid, this should raise the suspicion of a cystic neoplasm rather than a pseudocyst (13). The presence of granulocytes in the aspirated fluid is suggestive of an acute infection.
Figure 2.
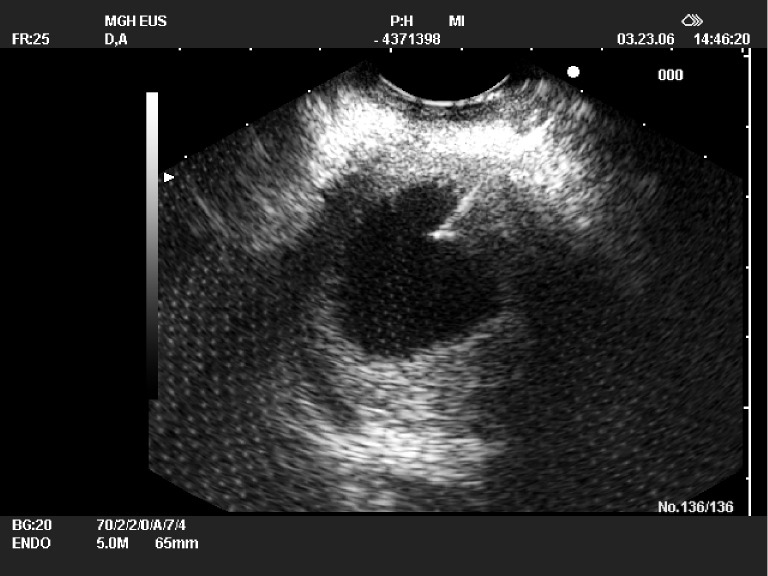
EUS-FNA of a pseudocyst with alcoholic chronic pancreatitis. Cyst fluid amylase was very high and cyst cytology was negative for malignant cells, and no definitive epithelial cells were identified. FNA, fine-needle aspiration.
Figure 3.
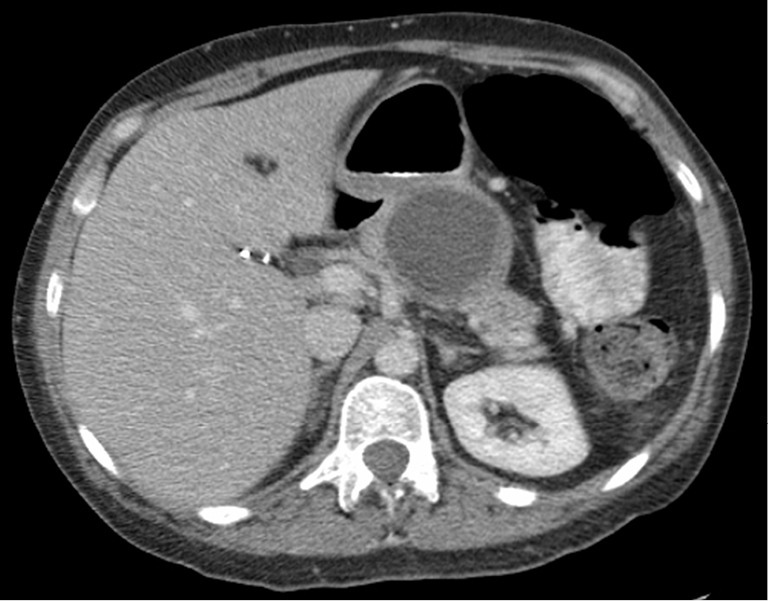
A unilocular, 7 cm in diameter pancreatic pseudocyst with debris.
Treatment
Simple, peripancreatic fluid collections that arise during acute pancreatitis usually resolve spontaneously. Without a constant source of fluid from an epithelium, pseudocysts have also the potential for spontaneous resolution. Small pseudocysts, less than 4 cm in diameter, often resolve and are rarely associated with complications, but in general, larger cysts are more likely to become symptomatic or cause complications. Spontaneous resolution of pseudocysts takes place through drainage into the GI tract or the pancreatic duct. In longterm observational studies, fewer than 10% of patients will suffer a complication. The main indications for drainage of pseudocysts are persistence or complications (infection, bleeding, gastric outlet or biliary obstruction). Forty percent of pseudocysts less than 6 cm will require drainage (14).
Drainage of pancreatic pseudocysts may be accomplished with a variety of procedures (15). A drainage catheter may be placed percutaneously into the fluid cavity under the CT/US guidance, and fluid is drained into an external collection system. The short-term success rate of this relatively simple technique is very high but it has a high risk of infections and creates significant patient discomfort (6). Surgical drainage of pseudocysts is performed by providing a large anastomosis between the pseudocyst cavity and the stomach or small bowel. Overall success rate of surgical drainage is very high but it is an invasive technique with high complication rates. It should be reserved for those patients that cannot tolerate or failed other drainage methods (16).
Drainage of pancreatic pseudocysts using endoscopic techniques is the current preferred method (17). Drainage is accomplished with either a transpapillary approach with ERCP or direct, endoscopic drainage across the stomach or duodenal wall. A transpapillary approach with drainage is used when the pseudocyst communicates with the main pancreatic duct, usually in the head of the pancreas. The transpapillary approach has also proven successful in the drainage of infected pseudocysts or pseudocysts associated with strictures or leaks of the main pancreatic duct (18). A transgastric or duodenal approach is used when the pseudocyst is directly adjacent to the gastroduodenal wall. EUS is used to determine the size, location, and thickness of the pseudocyst wall. A cyst wall thickness of more than 1 cm or the presence of large intervening vessels or varices as evident by the EUS examination are relative contraindications for endoscopic drainage. With the presence of a visible bulge in the wall of the stomach or the duodenum, endoscopic drainage is successful by the placement of transmural catheters or stents. EUS guidance is required if a bulge is not evident during the endoscopic evaluation prior to drainage. EUS-guided drainage is possible with the therapeutic linear echoendoscopes. This approach has proven highly successful and can be used for infected pseudocysts. Endoscopic drainage of necrotic pancreatic tissue through an endoscopic cyst-gastrostomy or duodenostomy is possible using balloon dilation and creating a fistulous tract (Figure 4). Overall, the complication rate of elective endoscopic drainage is about 13%, with success rates of more than 90% and recurrence rates of less than 10% (10).
Figure 4.
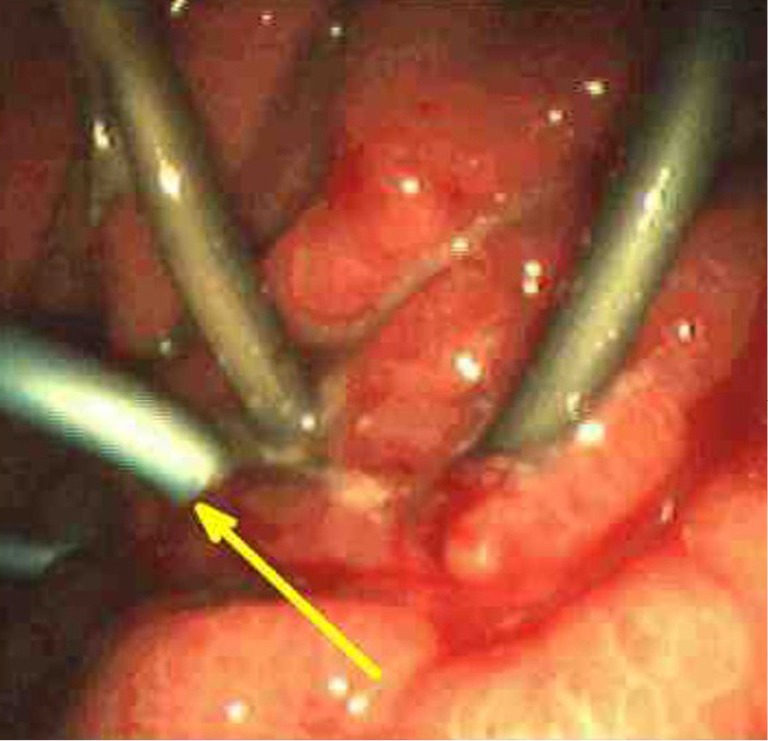
Endoscopic cystgastrostomy.
The major pancreatic cystic neoplasms (PCNs)
PCNs are classified at Box 1. The four major types of PCNs are IPMN, MCN, serous cystic neoplasm (SCN) and solid-pseudopapillary neoplasm (SPN). The proportion of PCNs varies with population. In the Western Hemisphere, SCNs account for 32% to 39%, MCNs for 10% to 45%, IPMNs for 21% to 33%, and SPNs for less than 10% of all PCNs. A nationwide survey from Korea reports the proportions of PCNs which are composed of IPMNs (41.0%), MCNs (25.2%), SPNs (18.3%), SCNs (15.2%), and others (0.3%) (1,19). Distinguishing among the four most common types of cysts is important, since the diagnosis and management varies with each type of cyst (Table 1).
Table 1. Characteristics of common pancreatic cysts.
| Parameters | Pseudocyst | IPMN (MD and BD) | MCN | SCN |
|---|---|---|---|---|
| Demographic | Alcohol abuse, the history of pancreatitis, middle-aged men | Middle aged and older individuals | Middle-aged women | Usually in older women |
| Location | Common in tail, solitary small to very large size | Common in pancreatic head, may be incidental and multifocal | Body and tail, incidental, single lesion | Entire pancreas, many small cysts or oligo/macrocystic |
| CT/MRI | Usually unilocular cyst, paranchimal inflammatory changes | MD: diffuse or focal involvement of MPD; BD: cyst or cluster of cysts, may be multifocal, ductal communication | Large cysts with thick septae, peripheral calcification, wall thickening | Microcystic multiple small cyst, central fibrous scar with calcification, sometimes oligocytic |
| EUS findings | Thick-walled, anechoic, unilocular cystic lesion, chronic pancreatitis | MD: dilation of MPD, hyperechoic nodules arising ductal wall; BD: small-cluster of grape-like dilations of BD, mural nodule | Macrocystic lesion with few septations. Sometimes focal, peripheral, calcification, no ductal dilation. Atypical papillary projections may seen | Multiple, small, anechoic cystic areas and ‘honeycamp’ appearance, sometimes central fibrosis or calcification |
| Cytology | Degenerative debris, inflammatory cells, histiocytes, no epithelial cells | Colloid-like mucin, mucin stains positive, mucinous epithelial cells with varying degrees of atypia, sparsely cellular | Mucinous epithelial cells with varying degrees of atypia, colloid-like mucin, mucin stains positive | Usually acellular and non-diagnostic, small cluster of cells with bland cuboidal morphology, glycogen stain positive, mucin negative |
| Cyst fluid analyses | Thin, clear or brown to green, non-mucinous, sometimes hemorrhagic, CEA concentration very low, amylase and lipase concentrations usually high | Thick, viscous mucus, CEA concentration usually high, amylase concentration may be high c (60%), KRAS mutation (+) (80%) | Thick, viscous mucus, CEA concentration usually high, KRAS mutation (+) (14%), GNAS mutation (−) | Clear and thin, may be hemorrhagic, CEA and amylase concentrations very low |
| Confocal endomicroscopy | No description yet | Epithelial villous structures; no vascular networking | Epithelial villous structures; no vascular networking | Thickened cyst wall; unilocular vascular networking; fibrous bands |
MD, main duct; BD, branch duct; MPD, main pancreatic duct; IPMN, intraductal papillary mucinous neoplasm; MCN, mucinous cystic neoplasm; SCN, serous cystic neoplasm; CT, computed tomography; MRI, magnetic resonance imaging; CEA, carcinoembryonic antigen.
Intraductal papillary mucinous neoplasms (IPMNs)
IPMNs are mucinous cystic lesions of the pancreas that are characterized by neoplastic, mucin-secreting, papillary cells projecting from the pancreatic ductal surface (20). They arise from the epithelial lining of the main pancreatic duct or its side branches. Intraductal proliferation of mucin-producing columnar cells is the main histologic characteristics of IPMNs and intraluminal growth cause dilatation of the involved duct and its proximal segment. They are usually found in the head of the pancreas as a solitary cystic lesion, but in 20% to 30% of the cases they may be multifocal. In 5% to 10% of cases they may involve the pancreas diffusely (20,21). IPMNs have become a major clinical focus as a result of their increased identification in recent years. This may be due to a true increase in the incidence by aging of the population, improvement in the understanding of IPMN, and/or increased use of cross-sectional imaging in clinical practice. In fact, the true incidence of IPMN is unknown; however, they are reported to be the one of the most common among the PCNs which accounts 20% to 50% of all PCNs (1,20,21).
IPMNs may range from premalignant lesions with low-grade dysplasia to invasive malignancy and they have a clear tendency to become invasive carcinoma (22,23). The World Health Organization (WHO) classified IPMNs into three subgroups according to degree of dysplasia: (I) IPMN with low- or intermediate-grade dysplasia; (II) IPMN with high-grade dysplasia (carcinoma in situ); and (III) IPMN with an associated invasive carcinoma. According to the involvement of pancreatic ductal system, IPMNs are classified as either main-duct IPMN (MD-IPMN) or branch-duct IPMN (BD-IPMN). If both main and branch ducts are involved together, then defined as combined-type IPMN. The clinicopathologic behavior of combined-type IPMN is similar to that of MD-IPMN. The neoplastic epithelium may show diverse architecture and cytology. Four subtypes of IPMNs have been characterized: gastric, intestinal, pancreatobiliary, and oncocytic. Most of BD-IPMNs are composed of gastric-type epithelium. However, intestinal type is more common in MD-IPMN. In a recent report, the four subtypes of IPMNs were associated with significant differences in survival (24). Patients with gastric-type IPMN had the best prognosis, whereas those with intestinal and pancreatobiliary type had a bad prognosis.
Diagnosis
IPMNs are most commonly asymptomatic and discovered incidentally on routine imaging. Some patients may present with recurrent non-specific or pancreatitis-like symptoms such as abdominal discomfort, abdominal pain, malaise, nausea and vomiting (20). Weight loss, diabetes mellitus, and jaundice may be detected especially in patients with an associated invasive carcinoma. IPMNs are usually detected in the elderly, mostly diagnosed after the fifth decade of life with a slight male dominance. Routine blood tests, such as complete blood count, liver function test, amylase, and lipase, are usually within normal limits or show nonspecific changes in patients with IPMNs. Serum CA19-9 and carcinoembryonic antigen (CEA) are generally not of diagnostic value (1).
Imaging plays a crucial role for detecting IPMNs (25). The aim of imaging for IPMN are: (I) to detect IPMN and exclude other cystic lesions of the pancreas; (II) to differentiate the MD-IPMN and BD-IPMN; (III) to determine the risk of malignancy and to evaluate the resectability. Different imaging modalities are used to reach these goals.
ERCP was the standard diagnostic tool for IPMN in the past (25). In MD-IPMN, the hallmark finding is a diffusely dilated main pancreatic duct with filling defects correlating to mucinous filling or papillary tumors. For BD-IPMN, the affected branch ducts are cystically dilated and communicate with the main pancreatic duct. In some occasions, the cystic side branch ducts do not fill with contrast due to mucus plugging. In some cases, duodenoscopy during ERCP reveals a patulous duodenal papilla and mucin extrusion through the orifice. The use of ERCP for the diagnosis of IPMN is limited by its invasiveness and risk of complications. In some cases, visualization of the entire pancreatic duct system is not possible because of copious amount of mucin.
In clinical practice, PCLs including IPMN are usually first diagnosed by conventional imaging modalities such as transabdominal US, CT and MRI (26,27). These tests are usually performed for unrelated conditions. The anatomic location, size, number, locularity, septation, calcification, pancreatic duct dilation and appearance of cysts on the conventional imaging might be helpful to differentiate the type of the cysts (Figure 5). MRCP can show communication between the duct and cyst more clearly and might be better than CT for the diagnosis of IPMN. However, with advances in multidetector CT, imaging details of CT including visualization of ductal communication have improved similar to those of MRI/MRCP (1,25). Both CT and MRI can also detect metastasis in case of invasive carcinoma associated with IPMN.
Figure 5.
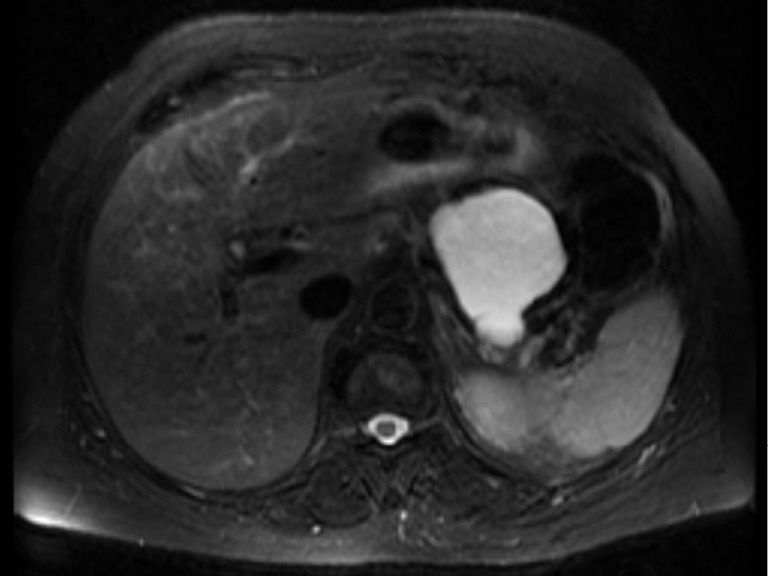
MRI finding of a branch-duct IPMN (BD-IPMN) at the tail of the pancreas. Note the fine septations. MRI, magnetic resonance imaging; IPMN, intraductal papillary mucinous neoplasm; BD-IPMN, branch-duct IPMN.
EUS may be more helpful for the diagnosis and differentiation of IPMNs because of its high resolution and better imaging characteristics than cross-sectional imaging modalities (28). It is particularly useful when the diagnosis is uncertain at cross-sectional imaging methods, for cysts with worrisome features in CT/MRI and for verification of malignancy before surgery in high risk patients with comorbidities or older age. EUS findings of IPMN include segmental or diffuse, moderate to marked dilatation of the main pancreatic duct, often associated with intraductal nodules in MD-IPMN. Obstruction of the main pancreatic duct with mucus can result in parenchymal changes. The pancreas may appear to be enlarged and may show signs of pancreatitis, or pancreatic parenchymal atrophy. Because of these changes, it is sometimes difficult to differentiate IPMN from chronic pancreatitis. BD-IPMN consists of multiple 5 to 20 mm cysts that have the appearance of a “cluster of grapes.” The main duct is mildly dilated or not dilated in BD-IPMN. The internal septation, debris, cyst wall thickening, papillary projections and mural nodule of cysts can be visualized effectively (Figure 6). Vascular invasion and lymph node metastases can also be detected successfully (1,12,20,28).
Figure 6.
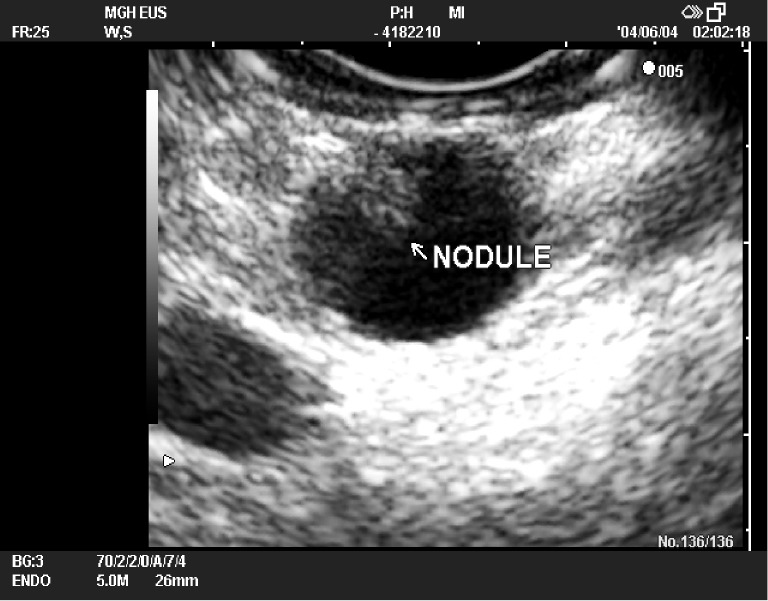
EUS finding of a branch-duct IPMN (BD-IPMN) with a mural nodule (arrow). IPMN, intraductal papillary mucinous neoplasm; BD-IPMN, branch-duct IPMN.
EUS criteria associated with malignancy in IPMN patients include marked dilatation of the main pancreatic duct (>10 mm) in MD-IPMN and large tumors (>40 mm) with irregular septa in BD-IPMN; mural nodule greater than 10 mm in height was associated with malignancy in both MD-IPMN and BD-IPMN (29). Large unilocular cystic component, focal hypoechoic mass, thick septations and thickening of cyst wall are also features of malignant or potentially malignant lesions. Based on these criteria, the accuracy of EUS to discriminate between benign and malignant IPMN varies from 40% to 90% in different studies (30). EUS has been found more accurate than transabdominal US, ERCP and cross-sectional imaging methods for the diagnosis of malignancy in patients with IPMN. The limitations of EUS include operator dependence and the inability to differentiate between malignancy and areas of focal inflammation that infiltrate pancreatic parenchyma and mimic malignancy.
EUS also allows for FNA of cystic lesions for biochemical, cytological and DNA analysis that might be further helpful for diagnosis and differentiation (31,32). Macroscopically, highly viscous fluid is the first clue that the cyst is likely IPMN or MCN. High concentration of CEA reflects the presence of a mucinous epithelium and it is elevated in both IPMNs and MCNs. Thus, it is mainly beneficial to distinguish mucinous cysts from non-mucinous. It does not differentiate IPMNs from MCNs or benign IPMNs from malignant IPMNs. A cut-off CEA level of 192 ng/mL has the sensitivity of 73%, specificity of 84%, and accuracy of 79% for differentiating mucinous from non-mucinous PCLs (33). Among all the cyst fluid diagnostic parameters, CEA concentration alone is the most accurate test for the diagnosis of cystic mucinous neoplasms. Due to connectivity to the pancreatic ductal system, amylase level may be elevated in IPMNs. However, the utility to differentiate IPMNs from other PCLs is not clear.
A recent study identified glucose and kynurenine to be differentially expressed between non-mucinous and mucinous pancreatic cysts (34). Metabolomic abundances for both were significantly lower in mucinous cysts compared with non-mucinous cysts. The clinical utility of these biomarkers will be addressed in future studies. Cytological examination alone is often non-diagnostic due to the low cellularity of the aspirated fluid. Cytology is the most accurate test for the detection of malignancy in patients with mucinous cysts and a “positive” or “malignant” diagnosis is generally 100% specific (35). In addition, the presence of high grade epithelial atypia in the cyst fluid analysis has a high accuracy of 80% to predict malignancy (36).
DNA analysis of pancreatic cyst fluid demonstrated that KRAS mutation is highly specific (96%) for mucinous cysts but the sensitivity is only 45%. KRAS is an early oncogenic mutation in the adenoma-carcinoma sequence but cannot distinguish a benign from malignant mucinous cyst. A recent study demonstrated that the GNAS mutation detected in cyst fluid can separate IPMN from MCN but, similar to KRAS mutations, do not predict malignancy (37). The absence of a GNAS mutation also does not correlate with a diagnosis of MCN because not all IPMNs will demonstrate a GNAS mutation. A GNAS mutation was present in 66% of IPMNs and either KRAS or GNAS mutations were identified in 96% of IPMNs.
Confocal laser endomicroscopy (CLE) is a novel imaging technology that uses low-power laser to obtain in vivo histology of the gastrointestinal mucosa. Recently, a CLE miniprobe has been developed to use during EUS-FNA to visualize cyst wall and epithelium directly through a 19-gauge FNA needle (Figure 7). Technical feasibility of this probe was shown and the preliminary studies of PCLs revealed that the presence of epithelial villous structures was associated with IPMNs, with 59% sensitivity and 100% specificity (38).
Figure 7.
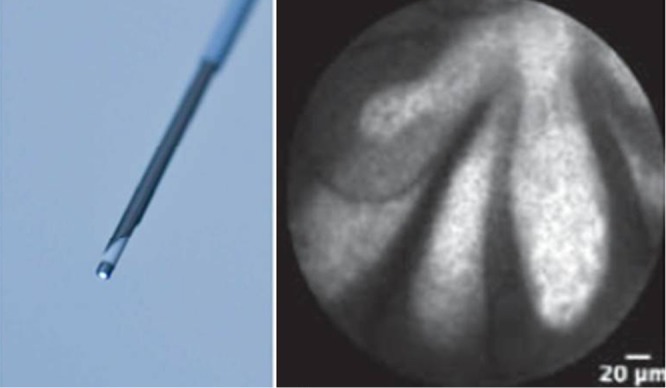
nCLE probe and papillary structures in an intraductal papillary mucinous neoplasms (IPMN) case.
Management
The mean frequency of malignancy in MD-IPMN is 61.6% and the mean frequency of invasive IPMN is 43.1%. Considering these high incidences of malignant/ invasive lesions and the low 5-year survival rates (31-54%), international consensus guidelines recommend resection for all surgically fit patients with MD-IPMN (29). If the margin is positive for high-grade dysplasia, additional resection should be attempted to obtain at least moderate-grade dysplasia at the surgical margin. The same guideline recommended evaluation but no immediate resection for patients with a MPD diameter of 5-9 mm as a “worrisome feature”.
The mean frequency of malignancy in resected BD-IPMN is 25.5% and the mean frequency of invasive cancer is 17.7%. BD-IPMN mostly occurs in elderly patients, and the annual malignancy rate is only 2-3%. These factors support conservative management with follow-up in patients who do not have any symptoms or risk factors predicting malignancy such as mural nodule, rapidly increasing cyst size and high grade atypia in cytology. There is insufficient data to support immediate resection for all BD-IPMNs >3 cm without “high-risk stigmata” and “worrisome features” (29) (Table 2).
Table 2. “High-risk stigmata” and “worrisome features” of IPMN on cross-sectional imaging.
| High-risk stigmata |
| Obstructive jaundice in a patient with cystic lesion of the head of the pancreas |
| Enhancing solid component within cyst |
| Main pancreatic duct size of 5-9 mm |
| Main pancreatic duct >10 mm in size |
| Worrisome features |
| Cyst >3 cm |
| Thickened/enhancing cyst walls |
| Non-enhancing mural nodule |
| Lymphadenopathy |
| Worrisome features |
IPMN, intraductal papillary mucinous neoplasm.
According to international guidelines, there is still an important group of patients who surgical treatment is controversial. Particularly younger patients (<65 years) with BD-IPMN need long-term follow-up which increase the cumulative risk of malignancy and cost of management. The resection is also not clear for BD-IPMNs >3 cm without “high-risk stigmata” and “worrisome features”. In addition, there are patients who refuse surgery or high-risk surgical candidates. As a result, these kinds of patients warrant a more conservative management for IPMNs and EUS-guided cyst ablation therapies has been introduced as an alternative treatment (39). Injection of a cytotoxic agent into a PCL will result in ablation of the cyst epithelium. The first cytotoxic agent used was ethanol and it was found to provide greater rates of complete ablation as compared with saline lavage. Ethanol lavage has been coupled with paclitaxel injection in a large series with a variety of PCLs (39,40). The combination of ethanol and paclitaxel injection resulted in elimination of the cysts, as determined by CT scanning, in 29/47 (62%) of patients, in a median follow-up period of 21.7 months. These studies were not specifically for IPMNs alone, the participating subjects were heterogeneous and contained IPMNs and other PCLs. Radiofrequency ablation (RFA) of PCNs has been recently described in a pilot study of six patients (41). The post procedure imaging at 3-6 months showed complete resolution of the cysts in two patients, whilst in three patients there was 48.4% reduction in size. These initial results suggest that the procedure is technically easy and safe. However, more studies are needed to show especially the effectiveness of the method.
After resection, the overall recurrence rate of IPMN varies from 7% to 30% and regular follow-up and monitoring of disease for recurrence is needed. A regimen consisting of yearly CT or MRI/MRCP for non-invasive, and every 6 months for invasive IPMNs have been mostly suggested during follow-up (29).
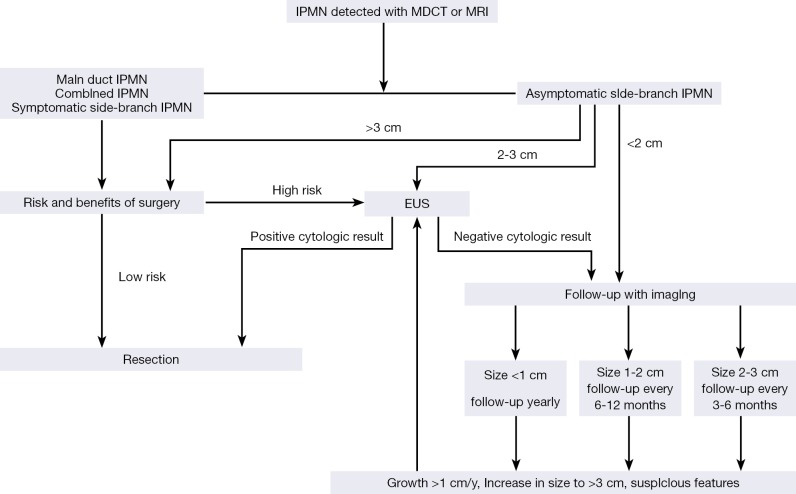
The aims of long term follow-up for unresected IPMNs are to detect a possible malignant transformation from originally benign lesion, and a concomitant ductal adenocarcinoma of the pancreas (42). The international guidelines have suggested follow-up of patients with BD-IPMNs <2 cm and without any “worrisome features” by cross-sectional imaging modalities (<1 cm in 2-3 years, 1-2 cm in yearly then lengthen interval if no change). For BD-IPMNs >2 cm and without any “worrisome features”, EUS follow-up for 3-6 months, then lengthen the interval if there is no change and alternating MRI have been recommended.
Mucinous cystic neoplasms (MCNs)
MCNs are defined as cyst-forming epithelial neoplasms that are usually without communication with the pancreatic duct and composed of columnar, mucin-producing ductal epithelium with an underlying ovarian-type stroma (1,43). Nearly all MCNs are surrounded by a thick layer of spindle cells containing receptors for progesterone and estrogen. The dense ovarian-like tissue simulates an ovarian hamartoma and, at times, a sarcoma. The possible derivation of the stromal component of MCNs from ovarian tissue is supported by morphology and the tendency to undergo luteinization. It has been hypothesized that ectopic ovarian stroma incorporated during embryogenesis in the pancreas may release hormones and growth factors, causing nearby epithelium to proliferate and form cystic tumors. The mucinous transitional epithelium is the source of nearly all malignancies arising from MCNs. Similar to IPMNs, MCNs are classified according to the grade of dysplasia: (I) MCN with low or intermediate-grade dysplasia; (II) MCN with high-grade dysplasia; and (III) MCN with an associated invasive carcinoma (44,45).
Macroscopically, MCNs present as single spherical masses. The lesions may be unilocular or multilocular. The cysts contain thick mucin or a mixture of mucin and hemorrhagic-necrotic material. There is no communication between the tumor and the pancreatic duct, unless there is fistula formation. The frequency of the lesion communicating with the pancreatic duct system may be high. In a Japanese multi-institutional report, 18.1% (25 of 138 patients) of MCNs demonstrated communication with the pancreatic duct (46).
MCNs almost exclusively occur in women, with a peak incidence in the fifth decade. The body and the tail of the pancreas are predominantly affected. Up to one-third of MCNs are reported to harbor an invasive carcinoma. Risk factors for the presence of malignancy include large tumor size, associated mass or mural nodules, and advanced age. Around 30% of the patients may be without symptoms or signs (47). Symptomatic patients may complain of abdominal pain, palpable mass, weight loss, anorexia, fatigue, or jaundice. Some patients may present with pancreatitis. The results of routine laboratory testing are usually nonspecific. Patients with bile duct obstruction display a cholestatic liver function abnormality (48).
On CT, MCNs appear as large cysts with thin septae; the septae are best shown after the administration of intravenous contrast. Calcifications may be seen, which are lamellated and located on the periphery of the lesion, in contrast to the central, stellate calcifications of the SCN. On MRI, the cysts have high signal intensity (bright) on T2-weighted images. On T1-weighted images with intravenous gadolinium administration, the wall and the septae are more conspicuously demonstrated. The presence of peripheral calcification, wall thickening, and thick septations can be suggestive of a malignant MCN. In a study of 52 patients with MCNs, the presence of these three findings predicted a 95% risk of malignancy (49).
EUS findings of MCN are thin-walled, septated fluid-filled cavities with diameter greater than 1 to 2 cm (3). Duct communication is rarely seen. Increased size, cyst-wall irregularity and thickening, intracystic solid regions, or an adjacent solid mass are findings suggestive of malignancy. Cyst CEA levels are high as a result of secretion by the mucinous epithelium. As mentioned, it is difficult to distinguish MCN from IPMN on the basis of cyst fluid cytology. Since MCNs rarely communicate with the pancreatic duct, ERCP is not routinely performed in the evaluation of MCNs.
Current consensus guideline advocates that all MCNs should be resected, unless there are contraindications for operation (29). For MCNs of <4 cm without mural nodules, laparoscopic resection as well as parenchyma-sparing resections and distal pancreatectomy with spleen preservation is recommended. Surgical resection is curative in nearly all patients with noninvasive MCN. Non-invasive MCNs require no surveillance after resection. For MCNs with an associated invasive carcinoma, prognosis depends on the extent of the invasive component, tumor stage, and resectability. The 2-year survival rate and 5-year survival rate of patients with resected MCN with an associated invasive carcinoma are about 67% and 50%, respectively (1). EUS-guided cyst ablation therapies may be considered for patients who are not a good candidate for surgery or refused the surgery.
Serous cystic neoplasms (SCNs)
SCNs are cystic neoplasms arise from centroacinar cells and composed of cuboidal, glycogen-rich epithelial cells. The lesions are filled with serous fluid. According to the degree of dysplasia, they are classified as either serous cystadenoma or serous cystadenocarcinoma. SCNs occur more frequently in women. Patients are usually diagnosed with SCN in their late 50s or early 60s. They occur more frequently in the body or the tail of the pancreas. Despite their benign nature, these lesions tend to grow slowly and may achieve large diameters (50).
Nearly 90% of von Hippel-Lindau (VHL) syndrome patients are reported to develop SCNs and 70% of serous cystadenomas has a mutation in the VHL gene (51). K-ras mutations are rarely seen in SCNs. SCNs are rarely malignant; only about 25 malignant cases have been reported to this date (1). SCNs are usually single, round lesions, with diameters that can be greater than 20 cm. On cross section, the cysts are composed of numerous microcysts filled with serous fluid (Figure 1). SCNs do not communicate with the pancreatic duct. A dense fibronodular scar is often located in the center of the lesion. A single layer of cuboidal epithelial cells lines the cysts. The central scar is composed of acellular hyalinized tissue and a few clusters of tiny cysts. The lesions are rich in vascular epithelial growth factor receptors, and a complex vascular structure supports the lesion. Four variants of serous cystadenoma are known. The serous epithelial components of these variants are identical to those of serous cystadenoma. They are macrocystic serous cystadenoma, solid serous adenoma, VHL-associated SCN, and mixed serous neuroendocrine neoplasm. Macrocystic serous cystadenomas include previous serous oligocystic and ill-demarcated serous adenoma. Solid serous adenomas are well-circumscribed neoplasms that have a solid gross appearance; they share the cytologic and immunohistologic features of classic SCN. VHL-associated SCN describes multiple serous cystadenomas and macrocystic variants that occur in VHL syndrome patients. In patients with VHL, SCNs typically involve the pancreas diffusely or in a patchy fashion (52). The mixed serous neuroendocrine neoplasm is the rare entity of serous cystadenomas associated with pancreatic neuroendocrine neoplasms. This is highly suggestive of VHL syndrome.
Most patients are without symptoms or signs on diagnosis. Symptomatic patients may present with abdominal pain, palpable mass, anorexia, jaundice, fatigue/malaise, or weight loss (45).
On CT and MRI, SCNs may have the classic microcystic appearance or the less common oligocystic appearance (Figure 8). Microcystic-type lesions comprise multiple small cysts. A central fibrous scar with calcification, which occurs up to 30% in SCNs, is considered pathognomonic. The dense tissue is arranged in a stellate form. In some cases, the small cysts and dense fibrous component may make the lesions appear solid on CT. The oligocystic pattern is often difficult to differentiate from MCN on CT/MRI because of the morphologic similarities (53).
Figure 8.
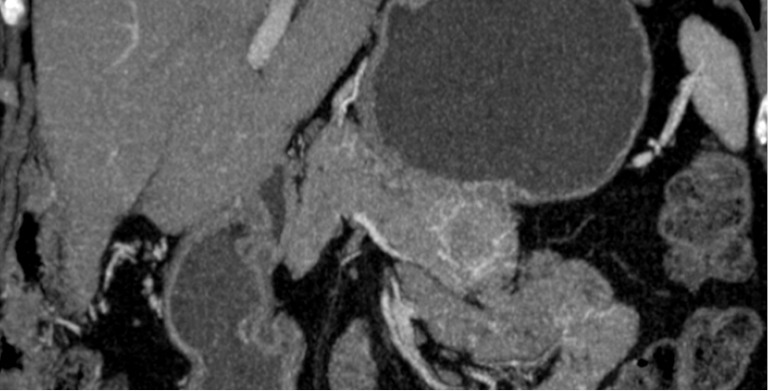
CT findings of SCN. (A) Axial image. Note the septa coming from the central scar; (B) sagittal image. Note the focal high-intensity lesion within a cyst representing hemorrhage (arrow). CT, computed tomography; SCN, serous cystic neoplasm.
Oligocystic SCNs should be suspected when a unilocular cystic lesion with lobulated contour without wall enhancement is located in the pancreatic head (45). On T1-weighted fat-suppressed MRI, the fluid component shows lower signal intensity compared to the fibrous matrix. On T2-weighted images, the fluid becomes bright. On EUS, the typical SCN has multiple small, anechoic cystic areas and thin septations. Because of the vascular nature of the SCN, aspirants from EUS-FNA may be bloody or contain hemosiderin-laden macrophages. Aspirated cyst fluid is low in CEA concentration. The yield of cytology with EUS-FNA is poor (54). A superficial vascular network sign, corresponding to a dense and subepithelial capillary vascularization, has been visualized in SCNs by nCLE of with 63% sensitivity and 100% specificity in a recent study of 18 cases (Figure 9).
Figure 9.

Demonstration of vascular network on cyst wall by confocal laser endomicroscopy (CLE) in a patient with serous cystadenoma.
The prognosis for patients with SCN is excellent. Even in the rare cases of serous cystadenocarcinoma, there are reports of a long-term survival after resection. Currently, proposed indications for surgical resection are presence of symptoms, size of greater than 4 cm, and uncertainty about the nature of the cystic neoplasm. Although increased size does not predict malignancy, large SCNs are reported to grow at a faster rate and are more likely to cause symptoms (50,52).
Solid-pseudopapillary neoplasms (SPNs)
SPNs are low-grade malignant neoplasms composed of monomorphic epithelial cells that form solid and pseudopapillary structures. Microscopically, they are a combination of solid pseudopapillary component and hemorrhagic-necrotic pseudocystic components. The solid portion is formed with poorly cohesive monomorphic cells and myxoid stromal bands containing thin-walled blood vessels. When the poorly cohesive neoplastic cells fall out, the remaining neoplastic cells and the stroma form the pseudopapillae. Mucin is absent, and glycogen is not conspicuous. Macroscopically, SPNs are large, round, single masses (average size, 8-10 cm). They are well demarcated and often fluctuant. The cut section discloses lobulated solid areas and zones with a mixture of hemorrhage, necrosis, and cystic degeneration. SPNs frequently undergo hemorrhagic cystic degeneration (55).
SPNs without histologic criteria of malignant behavior, such as perineural invasion, angioinvasion, or infiltration of the surrounding parenchyma, may metastasize. Therefore, all SPNs are classified as low-grade malignant neoplasms (56). SPNs occur predominantly in young women. The mean age at diagnosis is in the patient’s 20s or 30s. Symptomatic patients may present with pain, mass, anorexia, nausea/vomiting, jaundice, or weight loss. SPNs are reported to occur evenly throughout the pancreas.
On CT, SPNs appear as well-circumscribed and encapsulated masses with varying areas of soft tissue and necrotic foci. The capsule is usually thick and enhancing. Peripheral calcification has been reported up to 30% of patients. No septations are visualized. On MRI, the neoplasm is shown as a well-defined lesion with a mix of high and low signal intensity on T1- and T2-weighted images, which reflects the complex nature of the mass. Areas filled with blood products demonstrate high signal intensity on T1-weighted images and low or inhomogeneous signal intensity on T2-weighted images (57).
On EUS, SPNs are usually well-defined, hypoechoic masses. They may be solid, mixed solid and cystic, or cystic. Internal calcifications can be seen in some patients. The reported diagnostic accuracy of EUS-FNA for SPN based on cytology and immunohistochemistry is 65%. Aspirated cyst fluid may display necrotic debris. The cyst fluid CEA is low, reflecting the presence of nonmucinous epithelium (58).
The mainstay of treatment is surgery. After complete surgical resection, 85% to 95% of patients are cured (1). Even in cases with local invasion, recurrences, or metastases, long-term survival have been documented (59). No definite biological or morphologic predictors of outcome have been documented. Suggested indicators of poor outcome include old age and SPNs with an aneuploidy DNA content.
General approach to pancreatic cystic lesion (PCL)
There are many suggested algorithms on the management of PCLs (Figure 10) (14,60). Much emphasis is placed on the size and the morphology of the PCLs. Once confronted with a PCL, the first step is to differentiate PCNs from pseudocysts. The diagnosis of pseudocysts is primarily based on a patient history compatible with pancreatitis, with additional information from biochemical and imaging features. However, patients with PCNs may present with pancreatitis; patients with pseudocysts may have no apparent history suggestive of pancreatitis. Once pseudocysts have been excluded, the type of PCN should be determined. The primary focus should be on differentiating between mucinous (IPMN and MCN) and serous (SCN) cysts. Once a mucinous cyst has been diagnosed, patients with MD-IPMN, combined-type IPMN, and MCN should undergo a surgical consultation. Patients with BD-IPMN should be managed using the algorithm of the consensus guideline. SCNs should be observed, unless they are symptomatic or large (>4 cm).
Figure 10.
Suggested algorithm for pancreatic cyst management.
There are no strict published guidelines on the indication for EUS-FNA of PCLs. In general, there is no need for EUS-FNA of all cystic lesions with a clear diagnosis by cross-sectional imaging unless the results will impact patient management. IPMN lesions measuring more than 2 cm should be aspirated if the findings of a benign cytology will indicate the need for continued surveillance. If there is diagnostic uncertainty, the cyst fluid should be analyzed for CEA, KRAS and GNAS. Each analysis can be performed with less than 0.3 mL of fluid. If the primary question is whether the cyst is malignant or benign, the fluid should be sent for cytology. Cyst fluid for DNA mutations may supplement the results of cytology, particularly when a small volume of cyst fluid is available.
Acknowledgements
Disclosure: The authors declare no conflict of interest.
References
- 1.Yoon WJ, Brugge WR. Pancreatic cystic neoplasms: diagnosis and management. Gastroenterol Clin North Am 2012;41:103-18. [DOI] [PubMed] [Google Scholar]
- 2.Laffan TA, Horton KM, Klein AP, et al. Prevalence of unsuspected pancreatic cysts on MDCT. AJR Am J Roentgenol 2008;191:802-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Moparty B, Brugge WR. Approach to pancreatic cystic lesions. Curr Gastroenterol Rep 2007;9:130-5. [DOI] [PubMed] [Google Scholar]
- 4.Spinelli KS, Fromwiller TE, Daniel RA, et al. Cystic pancreatic neoplasms: observe or operate. Ann Surg 2004;239:651-7; discussion 657-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kimura W, Nagai H, Kuroda A, et al. Analysis of small cystic lesions of the pancreas. Int J Pancreatol 1995;18:197-206. [DOI] [PubMed] [Google Scholar]
- 6.Habashi S, Draganov PV. Pancreatic pseudocyst. World J Gastroenterol 2009;15:38-47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Brun A, Agarwal N, Pitchumoni CS. Fluid collections in and around the pancreas in acute pancreatitis. J Clin Gastroenterol 2011;45:614-25. [DOI] [PubMed] [Google Scholar]
- 8.Aghdassi A, Mayerle J, Kraft M, et al. Diagnosis and treatment of pancreatic pseudocysts in chronic pancreatitis. Pancreas 2008;36:105-12. [DOI] [PubMed] [Google Scholar]
- 9.Cannon JW, Callery MP, Vollmer CM, Jr. Diagnosis and management of pancreatic pseudocysts: what is the evidence? J Am Coll Surg 2009;209:385-93. [DOI] [PubMed] [Google Scholar]
- 10.Brugge WR. Approaches to the drainage of pancreatic pseudocysts. Curr Opin Gastroenterol 2004;20:488-92. [DOI] [PubMed] [Google Scholar]
- 11.Garcea G, Ong SL, Rajesh A, et al. Cystic lesions of the pancreas. A diagnostic and management dilemma. Pancreatology 2008;8:236-51. [DOI] [PubMed] [Google Scholar]
- 12.Brugge WR. The use of EUS to diagnose cystic neoplasms of the pancreas. Gastrointest Endosc 2009;69:S203-9. [DOI] [PubMed] [Google Scholar]
- 13.Pitman MB, Lewandrowski K, Shen J, et al. Pancreatic cysts: preoperative diagnosis and clinical management. Cancer Cytopathol 2010;118:1-13. [DOI] [PubMed] [Google Scholar]
- 14.Turner BG, Brugge WR. Pancreatic cystic lesions: when to watch, when to operate, and when to ignore. Curr Gastroenterol Rep 2010;12:98-105. [DOI] [PubMed] [Google Scholar]
- 15.Bennett S, Lorenz JM. The role of imaging-guided percutaneous procedures in the multidisciplinary approach to treatment of pancreatic fluid collections. Semin Intervent Radiol 2012;29:314-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lerch MM, Stier A, Wahnschaffe U, et al. Pancreatic pseudocysts: observation, endoscopic drainage, or resection? Dtsch Arztebl Int 2009;106:614-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Giovannini M. Endoscopic ultrasonography-guided pancreatic drainage. Gastrointest Endosc Clin N Am 2012;22:221-30, viii. [DOI] [PubMed] [Google Scholar]
- 18.Samuelson AL, Shah RJ. Endoscopic management of pancreatic pseudocysts. Gastroenterol Clin North Am 2012;41:47-62. [DOI] [PubMed] [Google Scholar]
- 19.Brugge WR, Lauwers GY, Sahani D, et al. Cystic neoplasms of the pancreas. N Engl J Med 2004;351:1218-26. [DOI] [PubMed] [Google Scholar]
- 20.Farrell JJ, Brugge WR. Intraductal papillary mucinous tumor of the pancreas. Gastrointest Endosc 2002;55:701-14. [DOI] [PubMed] [Google Scholar]
- 21.Sahani DV, Lin DJ, Venkatesan AM, et al. Multidisciplinary approach to diagnosis and management of intraductal papillary mucinous neoplasms of the pancreas. Clin Gastroenterol Hepatol 2009;7:259-69. [DOI] [PubMed] [Google Scholar]
- 22.Kang MJ, Lee KB, Jang JY, et al. Disease spectrum of intraductal papillary mucinous neoplasm with an associated invasive carcinoma invasive IPMN versus pancreatic ductal adenocarcinoma-associated IPMN. Pancreas 2013;42:1267-74. [DOI] [PubMed] [Google Scholar]
- 23.Sakorafas GH, Smyrniotis V, Reid-Lombardo KM, et al. Primary pancreatic cystic neoplasms revisited. Part III. Intraductal papillary mucinous neoplasms. Surg Oncol 2011;20:e109-18. [DOI] [PubMed] [Google Scholar]
- 24.Furukawa T, Hatori T, Fujita I, et al. Prognostic relevance of morphological types of intraductal papillary mucinous neoplasms of the pancreas. Gut 2011;60:509-16. [DOI] [PubMed] [Google Scholar]
- 25.Konstantinou F, Syrigos KN, Saif MW. Intraductal papillary mucinous neoplasms of the pancreas (IPMNs): epidemiology, diagnosis and future aspects. JOP 2013;14:141-4. [DOI] [PubMed] [Google Scholar]
- 26.Clores MJ, Thosani A, Buscaglia JM. Multidisciplinary diagnostic and therapeutic approaches to pancreatic cystic lesions. J Multidiscip Healthc 2014;7:81-91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Jones MJ, Buchanan AS, Neal CP, et al. Imaging of indeterminate pancreatic cystic lesions: a systematic review. Pancreatology 2013;13:436-42. [DOI] [PubMed] [Google Scholar]
- 28.Brugge WR. Endoscopic approach to the diagnosis and treatment of pancreatic disease. Curr Opin Gastroenterol 2013;29:559-65. [DOI] [PubMed] [Google Scholar]
- 29.Tanaka M, Fernández-del Castillo C, Adsay V, et al. International consensus guidelines 2012 for the management of IPMN and MCN of the pancreas. Pancreatology 2012;12:183-97. [DOI] [PubMed] [Google Scholar]
- 30.Grützmann R, Niedergethmann M, Pilarsky C, et al. Intraductal papillary mucinous tumors of the pancreas: biology, diagnosis, and treatment. Oncologist 2010;15:1294-309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kadayifci A, Brugge WR. Endoscopic ultrasound-guided fine-needle aspiration for the differential diagnosis of intraductal papillary mucinous neoplasms and size stratification for surveillance. Endoscopy 2014;46:357. [DOI] [PubMed] [Google Scholar]
- 32.Lee LS, Saltzman JR, Bounds BC, et al. EUS-guided fine needle aspiration of pancreatic cysts: a retrospective analysis of complications and their predictors. Clin Gastroenterol Hepatol 2005;3:231-6. [DOI] [PubMed] [Google Scholar]
- 33.Brugge WR, Lewandrowski K, Lee-Lewandrowski E, et al. Diagnosis of pancreatic cystic neoplasms: a report of the cooperative pancreatic cyst study. Gastroenterology 2004;126:1330-6. [DOI] [PubMed] [Google Scholar]
- 34.Park WG, Wu M, Bowen R, et al. Metabolomic-derived novel cyst fluid biomarkers for pancreatic cysts: glucose and kynurenine. Gastrointest Endosc 2013;78:295-302.e2. [DOI] [PMC free article] [PubMed]
- 35.Michaels PJ, Brachtel EF, Bounds BC, et al. Intraductal papillary mucinous neoplasm of the pancreas: cytologic features predict histologic grade. Cancer 2006;108:163-73. [DOI] [PubMed] [Google Scholar]
- 36.Pitman MB, Centeno BA, Daglilar ES, et al. Cytological criteria of high-grade epithelial atypia in the cyst fluid of pancreatic intraductal papillary mucinous neoplasms. Cancer Cytopathol 2014;122:40-7. [DOI] [PubMed] [Google Scholar]
- 37.Dal Molin M, Matthaei H, Wu J, et al. Clinicopathological correlates of activating GNAS mutations in intraductal papillary mucinous neoplasm (IPMN) of the pancreas. Ann Surg Oncol 2013;20:3802-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Konda VJ, Meining A, Jamil LH, et al. A pilot study of in vivo identification of pancreatic cystic neoplasms with needle-based confocal laser endomicroscopy under endosonographic guidance. Endoscopy 2013;45:1006-13. [DOI] [PubMed] [Google Scholar]
- 39.Brugge WR. Management and outcomes of pancreatic cystic lesions. Dig Liver Dis 2008;40:854-9. [DOI] [PubMed] [Google Scholar]
- 40.Matthes K, Mino-Kenudson M, Sahani DV, et al. EUS-guided injection of paclitaxel (OncoGel) provides therapeutic drug concentrations in the porcine pancreas (with video). Gastrointest Endosc 2007;65:448-53. [DOI] [PubMed] [Google Scholar]
- 41.Pai M, Senturk H, Lakhtakia S, et al. 351 Endoscopic Ultrasound Guided Radiofrequency Ablation (EUS-RFA) for Cystic Neoplasms and neuroen-docrine Tumors of the Pancreas. Gastrointest Endosc 2013; 77: AB143-AB144. [Google Scholar]
- 42.Kamata K, Kitano M, Kudo M, et al. Value of EUS in early detection of pancreatic ductal adenocarcinomas in patients with intraductal papillary mucinous neoplasms. Endoscopy 2014;46:22-9. [DOI] [PubMed] [Google Scholar]
- 43.Goh BK, Tan YM, Chung YF, et al. A review of mucinous cystic neoplasms of the pancreas defined by ovarian-type stroma: clinicopathological features of 344 patients. World J Surg 2006;30:2236-45. [DOI] [PubMed] [Google Scholar]
- 44.Sakorafas GH, Smyrniotis V, Reid-Lombardo KM, et al. Primary pancreatic cystic neoplasms revisited: part II. Mucinous cystic neoplasms. Surg Oncol 2011;20:e93-101. [DOI] [PubMed] [Google Scholar]
- 45.Bai XL, Zhang Q, Masood N, et al. Pancreatic cystic neoplasms: a review of preoperative diagnosis and management. J Zhejiang Univ Sci B 2013;14:185-94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Yamao K, Yanagisawa A, Takahashi K, et al. Clinicopathological features and prognosis of mucinous cystic neoplasm with ovarian-type stroma: a multi-institutional study of the Japan pancreas society. Pancreas 2011;40:67-71. [DOI] [PubMed] [Google Scholar]
- 47.Crippa S, Salvia R, Warshaw AL, et al. Mucinous cystic neoplasm of the pancreas is not an aggressive entity: lessons from 163 resected patients. Ann Surg 2008;247:571-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Crippa S, Fernández-Del Castillo C, Salvia R, et al. Mucin-producing neoplasms of the pancreas: an analysis of distinguishing clinical and epidemiologic characteristics. Clin Gastroenterol Hepatol 2010;8:213-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Procacci C, Carbognin G, Accordini S, et al. CT features of malignant mucinous cystic tumors of the pancreas. Eur Radiol 2001;11:1626-30. [DOI] [PubMed] [Google Scholar]
- 50.Sakorafas GH, Smyrniotis V, Reid-Lombardo KM, et al. Primary pancreatic cystic neoplasms revisited. Part I: serous cystic neoplasms. Surg Oncol 2011;20:e84-92. [DOI] [PubMed] [Google Scholar]
- 51.Moore PS, Zamboni G, Brighenti A, et al. Molecular characterization of pancreatic serous microcystic adenomas: evidence for a tumor suppressor gene on chromosome 10q. Am J Pathol 2001;158:317-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Farrell JJ, Fernández-del Castillo C. Pancreatic cystic neoplasms: management and unanswered questions. Gastroenterology 2013;144:1303-15. [DOI] [PubMed] [Google Scholar]
- 53.Procacci C, Graziani R, Bicego E, et al. Serous cystadenoma of the pancreas: report of 30 cases with emphasis on the imaging findings. J Comput Assist Tomogr 1997;21:373-82. [DOI] [PubMed] [Google Scholar]
- 54.Belsley NA, Pitman MB, Lauwers GY, et al. Serous cystadenoma of the pancreas: limitations and pitfalls of endoscopic ultrasound-guided fine-needle aspiration biopsy. Cancer 2008;114:102-10. [DOI] [PubMed] [Google Scholar]
- 55.Papavramidis T, Papavramidis S. Solid pseudopapillary tumors of the pancreas: review of 718 patients reported in English literature. J Am Coll Surg 2005;200:965-72. [DOI] [PubMed] [Google Scholar]
- 56.Tipton SG, Smyrk TC, Sarr MG, et al. Malignant potential of solid pseudopapillary neoplasm of the pancreas. Br J Surg 2006;93:733-7. [DOI] [PubMed] [Google Scholar]
- 57.Choi JY, Kim MJ, Kim JH, et al. Solid pseudopapillary tumor of the pancreas: typical and atypical manifestations. AJR Am J Roentgenol 2006;187:W178-86. [DOI] [PubMed] [Google Scholar]
- 58.Jani N, Dewitt J, Eloubeidi M, et al. Endoscopic ultrasound-guided fine-needle aspiration for diagnosis of solid pseudopapillary tumors of the pancreas: a multicenter experience. Endoscopy 2008;40:200-3. [DOI] [PubMed] [Google Scholar]
- 59.Lee SE, Jang JY, Hwang DW, et al. Clinical features and outcome of solid pseudopapillary neoplasm: differences between adults and children. Arch Surg 2008;143:1218-21. [DOI] [PubMed] [Google Scholar]
- 60.Scheiman JM. Management of cystic lesions of the pancreas. J Gastrointest Surg 2008;12:405-7. [DOI] [PubMed] [Google Scholar]