Abstract
The standard of care for unresectable lung cancer is chemoradiation. However, therapeutic options are limited and patients are rarely cured. We have previously shown that vitamin D and vitamin D analogs such as EB 1089 can enhance the response to radiation in breast cancer through the promotion of a cytotoxic form of autophagy. In A549 and H460 non-small cell lung cancer (NSCLC) cells, 1,25-D3 (the hormonally active form of vitamin D) and EB 1089 prolonged the growth arrest induced by radiation alone and suppressed proliferative recovery, which translated to a significant reduction in clonogenic survival. In H838 or H358 NSCLC cells, which lack VDR/vitamin D receptor or functional TP53, respectively, 1,25-D3 failed to modify the extent of radiation-induced growth arrest or suppress proliferative recovery post-irradiation. Sensitization to radiation in H1299 NSCLC cells was evident only when TP53 was induced in otherwise tp53-null H1299 NSCLC cells. Sensitization was not associated with increased DNA damage, decreased DNA repair or an increase in apoptosis, necrosis, or senescence. Instead sensitization appeared to be a consequence of the conversion of the cytoprotective autophagy induced by radiation alone to a novel cytostatic form of autophagy by the combination of 1,25-D3 or EB 1089 with radiation. While both pharmacological and genetic suppression of autophagy or inhibition of AMPK phosphorylation sensitized the NSCLC cells to radiation alone, inhibition of the cytostatic autophagy induced by the combination treatment reversed sensitization. Evidence for selectivity was provided by lack of radiosensitization in normal human bronchial cells and cardiomyocytes. Taken together, these studies have identified a unique cytostatic function of autophagy that appears to be mediated by VDR, TP53, and possibly AMPK in the promotion of an enhanced response to radiation by 1,25-D3 and EB 1089 in NSCLC.
Keywords: autophagy, cytostatic, cytoprotective, NSCLC, radiation
Abbreviations: ACTB, actin, β; AMPK, AMP activated protein kinase; AVO, acidic vesicular organelles; ANXA5, annexin A5; ATG5, autophagy related 5; Baf, bafilomycin A1; BECN1, Beclin 1, autophagy-related; FACS, fluorescence activating cell sorting; GLB, galactosidase; β; GFP, green fluorescent protein; H2AFX/H2AX, H2A histone family, member X; MAP1LC3/LC3, microtubule-associated protein 1 light chain 3; NSCLC, non-small cell lung cancer; PI, propidium iodide; SQSTM1, sequestosome 1; TP53, tumor protein p53; VDR, vitamin D (1, 25-dihydroxyvitamin D3) receptor; WT, wild-type; LC3 and LC3-II acronyms have been used to indicate the LC3B and LC3BII isoforms respectively
Introduction
Lung cancer is the leading cause of cancer-related death in the United States,1 accounting for 29% of male deaths and 26% of female deaths.2 Lung cancer has a poor prognosis with a median survival rate of less than 12 mo.3 Lung cancer is frequently unresectable and the current available therapies such as radiation and chemotherapy, while generally increasing patient survival, are frequently only palliative.3,4 Therefore, there is a pressing need for the development of new therapies that are more effective and selective than what is currently available for the treatment of lung cancer patients.
The standard of care for lung cancer is generally radiation in combination with chemotherapy to maximize its impact.5,6 A limitation of radiotherapy is tumor cell resistance either by overactivation of compensatory cell growth pathways or DNA repair pathways; this can often result in disease recurrence.7 One potential approach for overcoming these limitations could be to combine radiation treatment with vitamin D or vitamin D analogs (which tend to be less hypercalcemic) to enhance the response to radiation therapy.8-10 Vitamin D is a steroid hormone that plays an important role in maintaining calcium homeostasis.11 Vitamin D has also been shown to have antiproliferative and antitumorigenic effects.11,12 A number of Vitamin D analogs, some of which are being used in clinical trials, have been developed that have also shown antitumor effects.13
Vitamin D exerts its biological activity by binding to VDR (vitamin D [1,25-dihydroxyvitamin D3] receptor), which is a member of the steroid-thyroid-retinoid gene super family.14 The VDR acts as a transcriptional regulator, which is activated by binding of the ligand, 1,25-D3, which leads to formation of a heterodimer between VDR and RXR (retinoid X receptor). This complex recognizes and binds to vitamin D responsive elements, which further leads to recruitment of nuclear receptor coactivator proteins and VDR-interacting proteins. This recruitment causes an increase in VDR transcriptional activation.14,15 Clinically it has been shown that lung cancers express VDRs, indicating that they would likely be responsive to vitamin D.14-16 The literature suggests that vitamin D deficiency may contribute to cancer and high circulating levels of 25 (OH)-D3, which is an immediate precursor of calcitriol (1,25-D3), have been associated with prolonged survival of NSCLC patients.17 Studies have also shown a link between improved survival rate in lung cancer patients and high level of expression of VDR.14 Finally, previous studies in our laboratory have demonstrated the capacity of 1,25-D3 as well as the vitamin D analog, EB 1089, to enhance the response to radiation in breast tumor cells, both in cell culture and in tumor bearing mice.8-10,18,19
The current work defines a new mechanism whereby 1,25-D3 and the vitamin D analog EB 1089 enhance sensitivity to radiation in NSCLC cells. We observed that autophagy that is induced by radiation in combination with either 1,25-D3 or EB 1089 has a cytostatic function that translates into an enhanced reduction of clonogenic survival. Further, we observed the apparent conversion of the cytoprotective form of autophagy induced by radiation alone to a cytostatic form of autophagy by radiation in combination with +1,25-D3 or EB 1089.
Results
Influence of 1,25-D3 and the vitamin D analog EB 1089 on sensitivity to radiation in non-small cell lung cancer cells
Figure S1A shows expression levels for VDR, which is generally considered to be required for vitamin D to affect cell function,20 in a number of NSCLC cell lines. High levels of the receptor are evident in the A549, H460, and H358 cell lines and the receptor is also clearly expressed in the H1299 and H520 cell lines but is barely detectable in the H838 or H2030 cells.
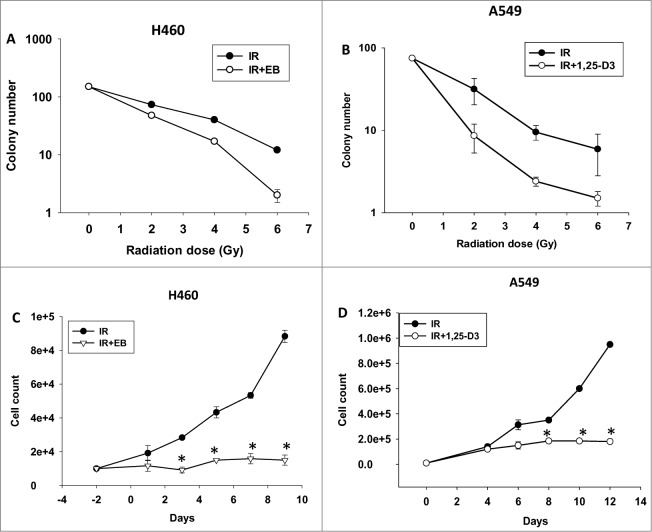
Figure 1A and B present clonogenic survival studies performed at different doses of radiation alone (varying from 2Gy to 6Gy) and in combination with either the vitamin D analog, EB 1089, or 1,25-D3 in the H460 and A549 cells, respectively. The inclusion of either 1,25-D3 or EB 1089 resulted in a significantly greater loss of clonogenicity at every radiation dose, with sensitization factors of between 1.4 and 2.0. Temporal response studies assessing cell viability in Figure 1C and D corroborated the results of the clonogenic survival study. While a single dose of 6Gy results in transient growth inhibition that is followed by proliferative recovery, cell growth inhibition is more pronounced and proliferative recovery is significantly impaired with the combination treatment of radiation + 1,25-D3 or EB 1089. These results differ from our findings in breast tumor cells where the combination treatment resulted in clear evidence of cell killing (a time-dependent decline in viable cell number).18,19,21
Figure 1.
Radiosensitization of NSCLC cells. H460 (A) and A549 (B) NSCLC cells were exposed to various doses of ionizing radiation (IR) alone or in combination with 100 nM EB 1089 or 100 nM 1,25-D3 and clonogenic survival was assessed after 14 d. H460 cells (C) or A549 cells (D) both of which are wt TP53 and VDR+, were exposed to radiation (IR, 6 Gy) alone or in combination with either 100 nM EB 1089 or 1,25-D3. Viable cell number was determined at the indicated days following radiation exposure (n = 3, mean ± SE, *P < 0.05).
Clonogenic survival studies shown in Figure S1B and C indicate that neither 1,25-D3 (the active form of vitamin D) nor EB 1089 (a synthetic analog of vitamin D) alone, at concentrations of 10 nM, 50 nM and 100 nM, significantly interfered with growth of the A549 or H460 NSCLC cells. Similarly, cell viability studies shown in Figure S1D and E indicated that neither 1,25-D3 nor EB 1089 inhibited growth or survival of H460 and A549 cells at 100 nM, the concentration used in the current work as well as our previous studies in breast cancer. The lack of effect of these agents alone establishes that the sensitization effects observed are not simply the consequence of additive antiproliferative or cytotoxic interactions. Furthermore, treatment with 1,25-D3 and EB 1089 had an essentially identical impact in terms of tumor cell sensitization (sensitization by 1,25-D3 and EB 1089 was significant for both cell lines; this becomes apparent when the data is plotted on an expanded scale). Consequently, while the bulk of studies in A549 cells were performed using 1,25-D3, studies in H460 cells were performed using EB 1089, which tends to be more stable for prolonged periods in solution. Additionally, in terms of potential clinical utility, EB 1089 has reduced calcemic effects.22
1,25-D3 and the vitamin D analog EB 1089 fail to increase the extent of DNA damage induced by IR or interfere with DNA repair in non-small cell lung cancer cells
In our previous studies with breast tumor cells, we have found no evidence for increased DNA damage or decreased DNA repair associated with sensitization to radiation by 1,25-D3/EB 1089.8,19,21 Figure S2 presents studies in which DNA damage was assessed by measuring levels of phosphorylated H2AFX/γH2AX in response to radiation and radiation + EB 1089 in the H460 cells. DNA damage was observed within 1 h after irradiation and declined thereafter, but neither the initial extent of damage nor the rate of decline, indicative of DNA repair, was affected by the presence of the EB 1089. It is also worth noting that there was no effect of EB 1089 alone, consistent with the lack of effect on cell growth.
1,25-D3 or EB 1089 fail to increase the extent of senescence induction by radiation in NSCLC cells
In studies that were performed prior to recognition of the potential role of autophagy in the response to radiation, we demonstrated that the primary response to radiation is senescence,23,24 in the absence of significant apoptosis, in cells expressing wild-type TP53/TRP53/p53 (note that the mouse nomenclature is TRP53, but we will use TP53 for either the human and mouse genes/proteins for simplicity). Since the temporal cell viability studies in Figure 1 indicated that the cells were undergoing primarily growth arrest, we evaluated whether the extent of senescence might have been increased for the combination treatments. Figure S3A and B present GLB (galactosidase, β)-staining images in A549 and H460 NSCLC cell lines, which indicates that both radiation treatment alone and the combination of radiation with 1,25-D3 or EB 1089 leads to an increase in GLB staining and alterations in morphology indicative of senescence. Figure S3C and D provide quantification of the time-dependent increase in senescence induced by radiation and the combination treatments in A549 (left panel) and H460 (right panel) cell lines. These studies fail to demonstrate any significant differences between radiation treatment alone and the combination of radiation + 1,25-D3 or EB 1089 in terms of the extent of senescence.
Minimal induction of apoptosis or necrosis by radiation and radiation + 1,25-D3 or EB 1089 in NSCLC cells
Although the primary responses observed either with radiation alone or radiation + EB 1089 or radiation + 1,25-D3 appeared to be growth arrest, it was formally possible that a fraction of the cell population might be undergoing apoptosis (or necrosis). Apoptosis and necrosis were evaluated by ANXA5 and PtdIns staining and flow cytometry. Figures S4A and Figure S4B show that there was minimal induction of apoptosis or necrosis (data for necrosis not shown) by radiation and radiation + 1,25-D3 or EB 1089 treatments in A549 and H460 cell lines. This outcome was further confirmed by the lack of PARP (poly ADP ribose polymerase) cleavage where staurosporine was used as a positive control (data not shown).
Increased sensitivity to radiation is associated with a novel form of autophagy
We have previously reported quite extensively on the capacity of 1,25-D3 and vitamin D analogs to sensitize breast tumor cells to radiation.8-10,18,19,21 In recent work, we have confirmed that this sensitization, a decline in viable cell number, occurs through the promotion of autophagy based on reversal of sensitization through either pharmacological or genetic interference with autophagy.18,19 The studies presented below indicate that sensitization in the NSCLC cells is related to conversion of the cytoprotective autophagy induced by radiation alone to what we have termed “cytostatic autophagy” when the combination of radiation and radiation + 1,25-D3 or EB 1089 is used.
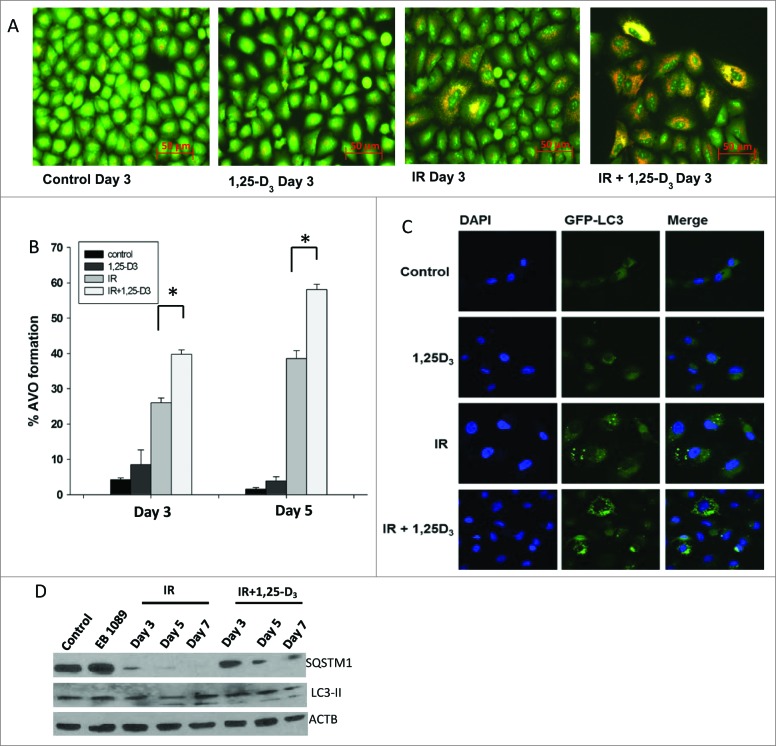
Studies in the A549 cells exposed to either radiation alone or radiation + 1,25-D3 indicated that radiation alone promotes autophagy, based on acridine orange staining and formation of autophagic puncta (Fig. 2A). Figure 2B indicates that the extent of autophagic vesicle formation (as quantified by flow cytometry) was significantly increased by the combination of radiation and 1,25-D3 in the A549 cells. GFP-LC3 puncta formation shown in Figure 2C further corroborates the flow cytometry data indicative of the promotion of autophagy.
Figure 2.
Autophagy in A549 NSCLC cells. (A) Induction of autophagy in A549 cells by radiation (IR, 6 Gy) alone or radiation+1,25-D3 by acridine orange staining. (B) Quantification of autophagy by flow cytometry in A549 cells (n = 3, mean ± SE, *P < 0.05). (C) A549 cells exposed to vehicle control, 100 nM 1,25-D3, radiation (IR, 6Gy), or 100 nM 1,25-D3 in combination with radiation. GFP-LC3 puncta indicate induction of autophagy (all images were taken at the same magnification). (D) Assessment of autophagic proteins, SQSTM1, and LC3-II in response to different treatment conditions by protein gel blots.
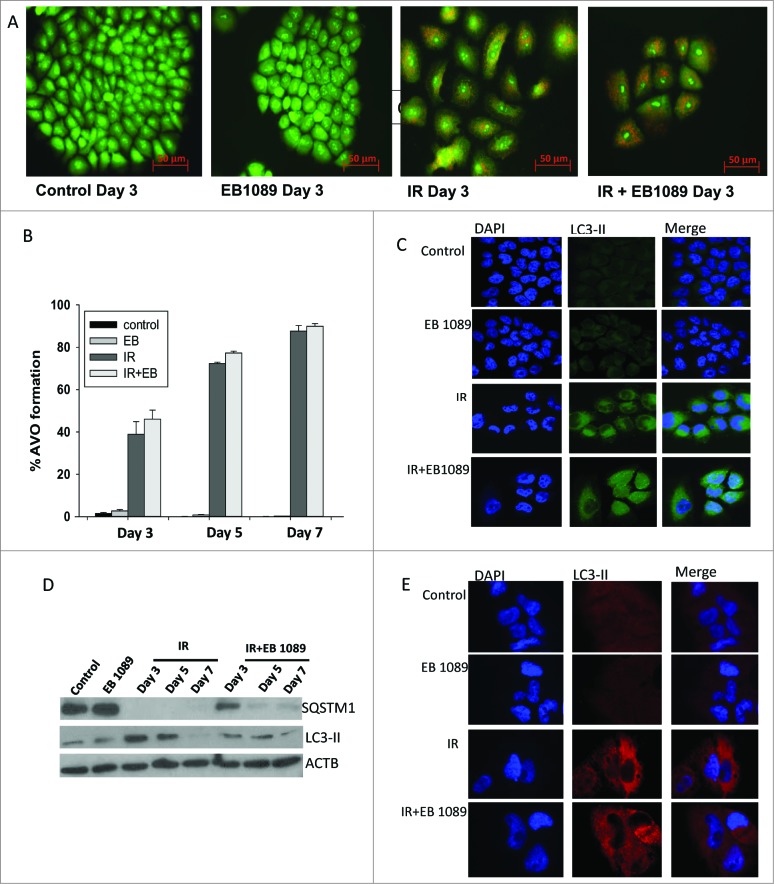
Figure 3A and B show autophagic vesicle formation by acridine orange staining and quantification by flow cytometry in the H460 cells; GFP-LC3 puncta formation further indicative of autophagy is shown in Figure 3C and endogenous LC3-II levels assessed using confocal microscopy are shown in Figure 3E. However, as shown in Figure 3B, the extent of autophagy was not significantly different for radiation alone and EB 1089 + radiation in the H460 cells. Consequently, despite the evidence for increased autophagy for the combination treatment in the A549 cells, these findings in H460 cells argue against the simple explanation that sensitization might be caused solely by an increase in the extent of autophagy.
Figure 3.
Autophagy in H460 NSCLC cells. (A) Induction of autophagy in H460 cells by radiation alone or the combination of radiation+ EB 1089. (B) Quantification of acidic vesicle formation in H460 cells by flow cytometry. (n = 3, mean ± SE) (C) GFP-LC3 puncta formation indicative of the induction of autophagy in H460 cells (all images were taken at same magnification). (D) Assessment of autophagic flux markers, SQSTM1 and LC3-II in response to different treatment conditions by protein gel blots. (E) Endogenous LC3-II foci formation was determined by confocal microscopy. Nuclei were stained with DAPI. Images were taken on d 3 at the same magnification.
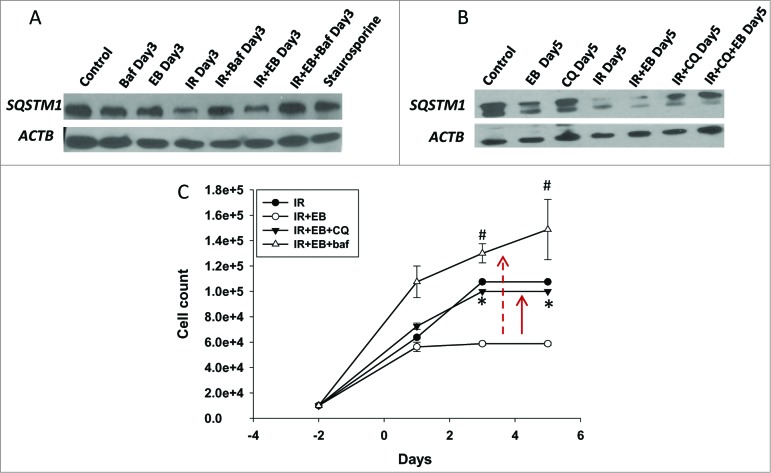
Experiments were also performed to determine whether the autophagy observed reflected autophagic flux, where autophagy proceeds to completion in terms of the degradation of select protein or organelle substrates.25 Figure 2D and Figure 3D demonstrate that autophagic flux markers SQSTM1/p62 and LC3-II were degraded both with radiation alone and the combination treatment in A549 and H460 cells. However, there was no apparent difference in the extent or time course of degradation for radiation alone versus the combination treatment with either 1,25-D3 or EB 1089. Furthermore, as shown in Figure 4A and B, respectively, the autophagy inhibitors bafilomycin A1 (Baf) and chloroquine interfered with SQSTM1 degradation by radiation alone as well as the combination of radiation with EB 1089, consistent with both treatments promoting autophagic flux.
Figure 4.
Evidence for cytostatic autophagy by pharmacological inhibition. (A, B) Western blot indicating that pharmacological inhibitors Baf (100 nM, panel A) and chloroquine (10 μM; panel B) inhibit autophagy by blocking degradation of SQSTM1 in lysates collected from H460 cells. (C) H460 NSCLC cells were exposed to EB 1089 (100 nM) in combination with 6 Gy radiation in the presence of chloroquine (CQ, 10 μM) or Baf ( 100 nM ) and cell viability was monitored for a period of 6 d (n = 3, mean ± SE).
It has been established by a number of laboratories, including our own, that radiation-induced autophagy frequently has a cytoprotective function.18,19 Recent studies by Kroemer's lab have shown this to be the case in the A549 and H460 cells by demonstrating that inhibition of radiation-induced autophagy increases sensitivity to radiation26 and this has been confirmed in our current work. Figure S5A and B show that radiation sensitivity increases (downward arrow), when autophagy is blocked by chloroquine in both A549 and H460 cell lines, respectively, consistent with the cytoprotective function of autophagy in response to radiation alone. Furthermore, Figure S5C and D demonstrate that blocking cytoprotective autophagy by either chloroquine or Baf led to a modest (but not statistically significant) increase in apoptosis. In contrast, no increase in apoptosis was observed when autophagy was inhibited for the combination treatment. We therefore hypothesized that exposure of the NSCLC cells to 1,25-D3 or EB 1089 could be altering the function of the autophagy, converting the cytoprotective autophagy induced by radiation to what we have termed cytostatic autophagy, based on the temporal responses to treatment observed in Figure 1C and D.
To investigate whether an altered function of autophagy might be responsible for the apparent increase in sensitivity to radiation, we first determined whether interference with autophagy could reverse the impact of radiation + EB 1089, using chloroquine and Baf, both well-characterized pharmacological inhibitors of the late stages of autophagy.25 As indicated above, Figure 4A and B present western blots indicating that both inhibitors by themselves and in combination with radiation and EB 1089, lead to an accumulation of SQSTM1, indicative of the inhibition of autophagic flux.25 As shown in Figure 4C, both chloroquine and Baf, when used to block autophagy, reversed the radiosensitization effects of EB 1089 (upward arrows). This indicates that what we have termed a cytostatic form of autophagy (based on the temporal response studies presented in Fig. 1C and D) is likely to be the mode of radiosensitization with the combination treatment.
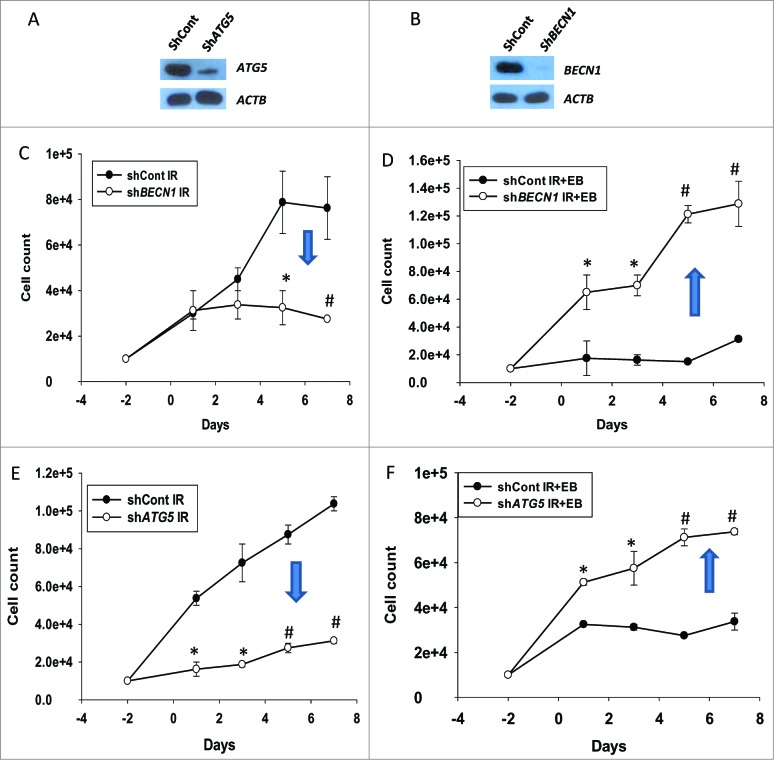
The conclusion that the cytoprotective autophagy induced by radiation has been converted to cytostatic autophagy by the inclusion of 1,25-D3 or EB 1089 is strongly supported by complementary genetic silencing studies, where the autophagy-related genes BECN1 and ATG5 were knocked down utilizing shRNA, as shown in the immunoblots in Figure 5A and B. Figure S6 confirms successful inhibition of autophagy, based on the reduction in the extent of autophagic vesicle formation, by genetic silencing of BECN1 and ATG5. (It should also be noted that the increased cell size that is a component of autophagy induction is not observed in cells where the autophagy genes have been silenced). The temporal and cell viability studies in Figure 5D and F indicate that as was the case with pharmacological inhibition of autophagy, radiosensitization by EB 1089 is reversed when cytostatic autophagy is genetically suppressed. In dramatic contrast, Figure 5C and E demonstrate that when cytoprotective autophagy is genetically suppressed, tumor cell sensitivity to radiation is increased.
Figure 5.
Cytoprotective and cytostatic autophagy (genetic silencing). (A) Silencing of ATG5 and (B) BECN1 in H460 NSCLC cells. ((C)and D) Effect of BECN1 silencing on response to radiation alone or EB 1089 + radiation was assessed by cell viability studies (n = 3, mean ± SE, *P < 0.05, #P < 0.001). ((E)and F) Effect of ATG5 silencing on response to radiation alone or EB 1089 + radiation was assessed by cell viability studies (n = 3, mean ± SE, *P < 0.05, #P < 0.001).
It is important to note that the sensitization to radiation that occurs when the cytoprotective autophagy induced by radiation is inhibited (Fig. 5C and 5E) is no longer evident in the cells exposed to EB 1089 + radiation; otherwise, it would be difficult to reverse the sensitization induced by EB 1089, a problem we encountered in our studies in breast tumor cells.18,19 This is the basis for our conclusion that the cytoprotective form of autophagy induced by radiation has been converted to a cytostatic form.
The effects of radiation and EB 1089 + radiation are associated with cell cycle arrest
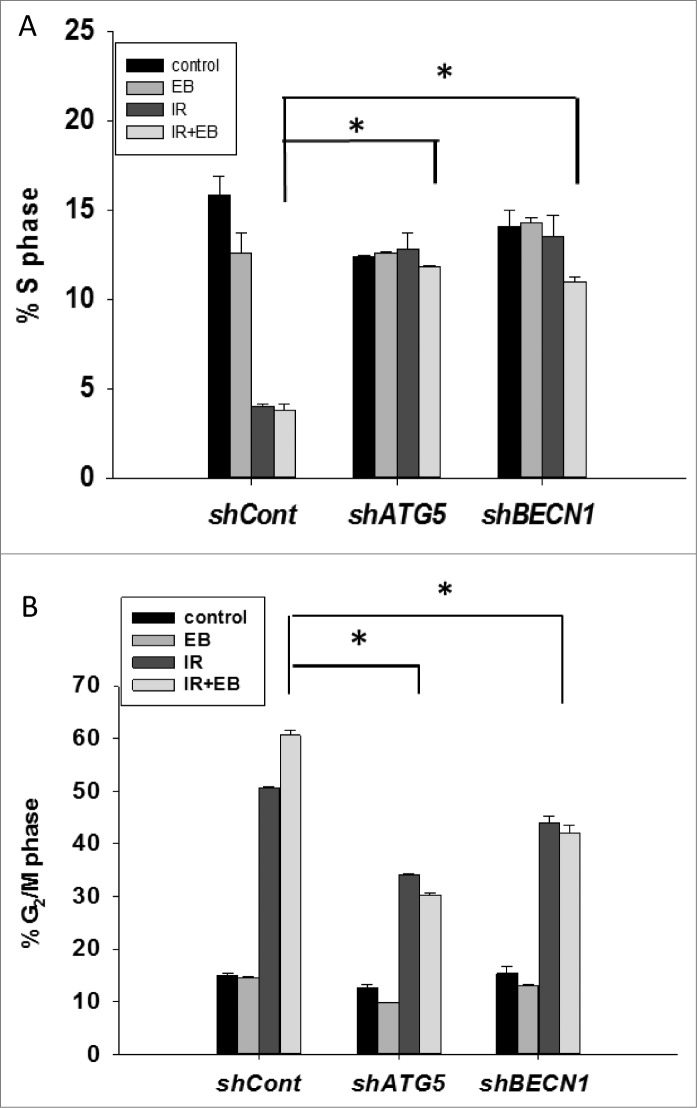
As shown in the temporal response studies presented in Figure 1, the primary effects of radiation as well as radiation in combination with either 1,25-D3 or EB 1089 were to arrest cell growth without evidence for cell killing in either condition. To confirm these observations, cell cycle analysis was performed in H460 cells exposed to radiation alone and EB 1089 + radiation with and without knockdown of the autophagy genes ATG5 and BECN1. As shown in Figure 6A and B, radiation alone as well as EB 1089 + radiation reduced the S phase population and increased the G2/M phase population. Effects of EB 1089 alone on cell cycle distribution were not significant, again consistent with its lack of effect on cell growth. As would be expected, inhibition of autophagy by genetic silencing reversed the G2/M associated growth arrest.
Figure 6.
Cell cycle analysis. Cell cycle analysis was performed in H460 (shcont, shATG5, and shBECN1) cells. (A) Percentage of cells undergoing S-phase is shown (n = 2, mean ± SE, *P < 0.05; shcont IR+EB 1089 compared to shATG5 IR+EB 1089 and shCont IR+EB 1089 is compared to shBECN1 IR+EB 1089). (B) Percentage of cells arresting in G2/M phase is shown. n = 2, mean ± SE, *P < 0.05; shCont IR+EB 1089 compared to shATG5 IR+EB 1089, and shCont IR+EB 1089 is compared to shBECN1 IR+EB 1089.
Requirement for functional TP53 and VDR in sensitization of NSCLC cells
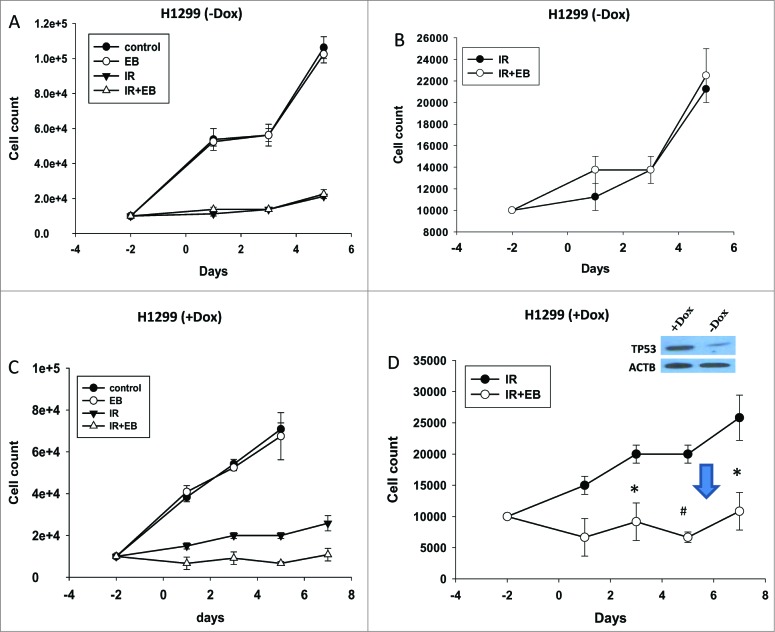
In our previous work with breast tumor cells, we identified a fundamental requirement for functional TP53 in radiosensitization by 1,25-D3 or EB 1089.8,18,19 Both A549 and H460 cells are known to express wild-type TP53.27,28 However the combination treatment of radiation +EB 1089 failed to increase sensitivity to radiation in the H358 NSCLC cell line (Fig. S7A), which has mutant TP53, despite the very high expression of the VDR (Fig. S1A). To establish the importance of the requirement for VDR in enhancement of the response to radiation, similar combination studies were performed in H838 cells, which lack VDR (Fig. S1A) but are wild type in TP53.29 Figure S7B indicates that these cells did not show any enhancement in the response to the combination treatment of radiation + 1,25-D3 compared to radiation treatment alone.
To confirm this requirement for functional TP53 in radiosensitization, we demonstrate (Fig. 7A and B, where 7B is an expanded scale for IR and IR+EB 1089) that sensitization to radiation by EB 1089 also fails to occur in the H1299 NSCLC cell line, which is tp53-null,30 but expresses the VDR (Fig. S1A). In dramatic contrast, Figure 7C and D (7D is an expanded scale for IR and IR + EB 1089) indicate that H1299 cells could be radiosensitized by EB 1089 (downward arrow) when TP53 was expressed under the influence of doxycycline (induction of TP53 shown in figure inset).
Figure 7.
Functional TP53 is required for sensitization of NSCLC cells to radiation. ((A)and B) tp53-null H1299 cells and ((C)and D) H1299 cells with TP53 induced by doxycycline (inset) were exposed to radiation (IR, 6 Gy) or EB 1089 in combination with radiation and cell viability was monitored for a period of 5 to 7 d. n = 3, mean ± SE. (Figs. 7B and 7D are expanded scales for IR and IR+EB 1089 *P < 0.05, #P < 0.001.).
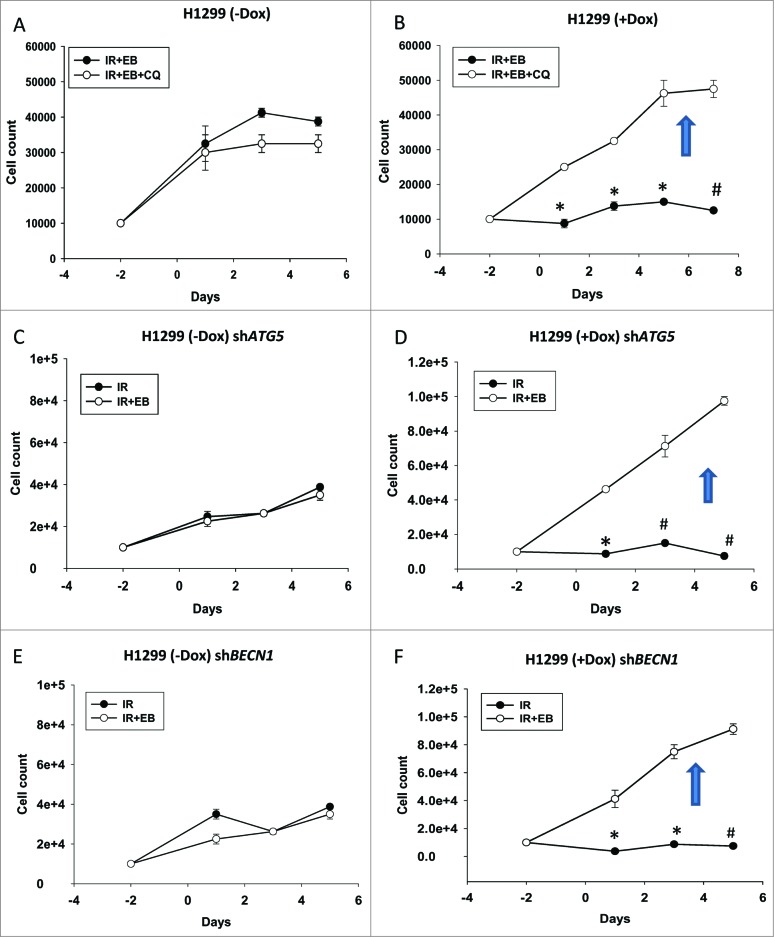
To confirm and extend these observations in the H1299 cells, we investigated the effects of both pharmacological and genetic autophagy inhibition in both the tp53-null and inducible cell lines. As shown in Figure S8A and B, both ATG5 and BECN1 were successfully silenced for tp53-null and tp53-inducible H1299 cells. Figure S8C shows the reduction in autophagic vesicle formation when ATG5 and BECN1 were knocked down while Figure S8D shows that chloroquine effectively promoted accumulation of LC3-II, which is indicative of interference with the late stage of autophagy. Figures 8B, D and F indicate that in H1299 cells with induced TP53, sensitization by the combination treatment was reversed when autophagy was blocked. In quite dramatic contrast, blocking autophagy in the tp53-null H1299 parental cells had minimal effect (chloroquine) or no effect (genetic silencing of ATG5 or BECN1) on tumor cell sensitivity to the combination treatment of EB 1089 + radiation (Fig. 8A, C and E). It should be noted that the low cell number for radiation alone in Figures 8B, D and F reflects sensitization to radiation through interference with the cytoprotective function of radiation-induced autophagy. Conversely, the high cell numbers for the EB 1089 + radiation combination reflects the fact that sensitization to radiation by EB 1089 was eliminated through interference with cytostatic autophagy.
Figure 8.
Blocking autophagy reverses the sensitization in TP53-inducible H1299 cells. Cell viability studies indicate that inhibition of autophagy in H1299 parental tp53-null cells by using either. (A) chloroquine or genetic silencing of (C) ATG5 or (E) BECN1 did not cause any change in sensitivity of the cells. However in TP53-inducible H1299 cells blocking autophagy either by using (B)chloroquine or genetically silencing (*P < 0.05, #P < 0.001) (D) ATG5 (*P < 0.05, #P < 0.001) or (F) BECN1 (*P < 0.05, #P < 0.001) reversed the sensitization that was achieved with the combination treatment of EB 1089 and radiation. n = 3, mean ± SE.
Studies in normal lung epithelial cells and cardiomyocytes
Drug selectivity is a critical consideration in studies related to cancer therapies; treatments should ideally be highly targeted to minimize the damage to normal cells and tissue. This is particularly true for the utilization of radiation in diseases such as non-small cell lung cancer. Consequently, studies were conducted to test for selectivity of the combination treatment of 1,25-D3 with radiation in normal human bronchial epithelial cells and cardiomyocytes in culture. As with the experiments in H460 cells, the HBEC3-KT bronchial epithelial cells were pretreated with 100 nM EB 1089 24 h before irradiation with a single dose of 6 Gy while the H9C2 cardiomyocytes were exposed to 100 nM 1,25-D3 for 72 h followed by a single dose of radiation (5 Gy). Figure S9A and B indicate that there were no significant differences between the impact of radiation alone and the combination of radiation + 1,25-D3 or radiation + EB 1089 treatments in the epithelial cells or the cardiomyocytes, respectively.
Additional evidence for differences in the functional forms of autophagy induced by radiation alone and by radiation + EB 1089.
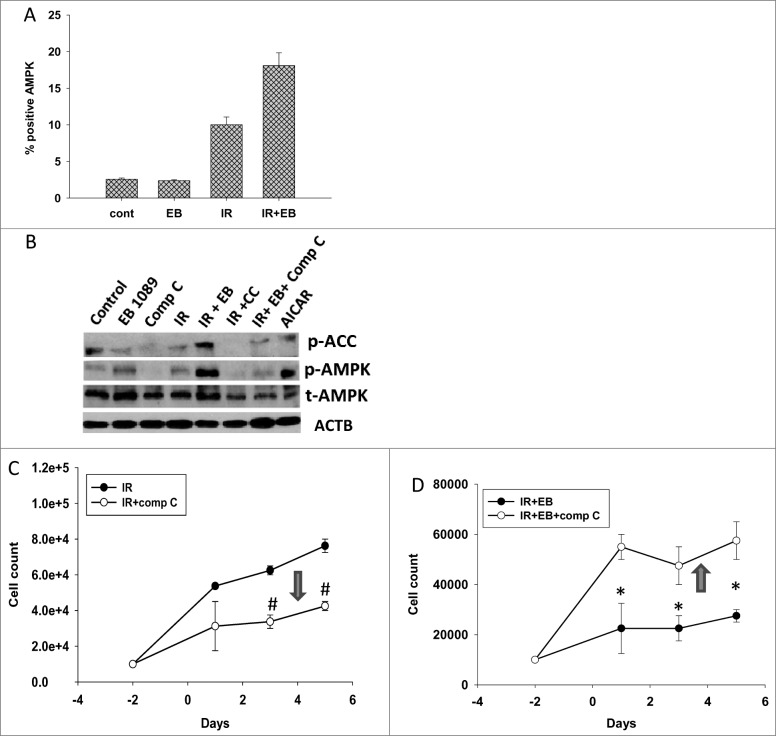
AMPK, an energy-sensing protein kinase, has been shown to be involved in regulation of autophagy.31,32 Studies were performed to investigate changes in the phosphorylated (active) form of AMPK levels in response to radiation alone (cytoprotective autophagy) and radiation + EB 1089 (putatively cytostatic autophagy). Figure 9A indicates that increased phospho-AMPK levels were observed with radiation alone as well as with the combination of radiation + EB 1089 as measured by flow cytometry. After 48 h, AMPK activation by the combination treatment significantly exceeded that for radiation alone. This was confirmed by the protein gel blotting data presented in Figure 9B where p-AMPK levels are clearly higher for the combination treatment. A recent study conducted by Singh K et al. indicates that AMPK is activated upon irradiation of H1299 cells,33 consistent with our own findings.
Figure 9.
Dual roles of AMPK in cytoprotective and cytostatic autophagy. (A) H460 cells were exposed to 6 Gy of radiation (IR) or EB 1089 (100 nM) + radiation and phosphorylation of AMPK (Thr172) was monitored by flow cytometry at 24 h post-radiation. (B) Western blotting was performed to confirm successful inhibition of phosphorylation of AMPK and its downstream target ACC (acetyl-CoA carboxylase) by using compound C (inhibitor of AMPK). (C/D) Effect of compound C on the temporal response to radiation (IR, 6 Gy) alone (C)or EB 1089 + radiation (D) was assessed by monitoring cell viability. n = 3, mean ± SE, *P < 0.05, #P < 0.001.
To potentially distinguish between the involvement of AMPK in autophagy signaling by radiation alone and radiation + EB 1089, compound C was used to inhibit AMPK. Figure 9B confirms that compound C interfered with the phosphorylation of AMPK and its downstream target ACC (acetyl-CoA carboxylase) induced by radiation alone as well as EB 1089 + radiation. Figure 9D shows that inhibition of AMPK resulted in reversal of the radiosensitizing effects of EB 1089. In dramatic contrast, treatment with compound C enhanced the response to radiation alone as shown in Figure 9C. Consequently, these findings closely parallel the outcome of the experiments presented in Figures 4, 5 and 7, where autophagy inhibition enhanced sensitivity to radiation but reversed the enhancement of radiation sensitivity by EB 1089. These studies support the premise that autophagy has diametrically opposite functions in the case of radiation alone (cytoprotective) and radiation + EB 1089 (cytostatic radiosensitizing).
Discussion
The treatment of lung cancer continues to represent a clinical challenge. Current treatments do prolong patient life but often become primarily palliative.3,4 Chemo-radiation is routinely used to treat lung cancer;5-7 however, the doses that can be administered are limited by toxicity to normal adjoining tissue.34 Consequently, increasing the selectivity of radiation therapy would be of great benefit to lung cancer patients.
Vitamin D has been shown to have antiproliferative effects in various experimental models of cancer.11,12 However, in our hands, vitamin D or EB 1089 alone, at 100 nM, showed no antiproliferative or cytotoxic effects against the NSCLC cell lines (H460, A549, H1299 and H358) used in the current work despite high expression of VDR: this highlights that fact that the effects of the combination treatments are not merely additive but reflect a mechanistic interaction that is likely to be related to the induction of autophagy. While the combination treatment of radiation + 1,25-D3 or EB 1089 resulted in cell death in breast tumor cells,18,19 in the current studies we observe primarily extended growth inhibition and suppression of proliferative recovery. The combination treatment of radiation + 1,25-D3 or EB 1089 did not increase either apoptosis or necrosis, similar to what we have reported previously for breast tumor cells.18,19 In contrast to the studies in breast tumor cells, there was no evidence of cell death (i.e., a decline in viable cell number over time). Nevertheless, we observed unequivocal evidence for radiosensitization in clonogenic survival assays, which is consistent with a reduction in the capacity for cellular self-renewal. The observed impact on self-renewal capacity and proliferative recovery could prove to be relevant to the problem of disease recurrence in lung cancer patients.
While our initial experiments in A549 cells suggested that sensitization might be related to an increase in the extent of autophagy, subsequent experiments in H460 cells tended to rule out this possibility. Instead, our data support the relatively novel premise that the cytoprotective function of autophagy that is evident with radiation alone is converted to a cytostatic function (pronounced growth inhibition, suppression of proliferative recovery, and reduced clonogenic survival) with the combination treatment. This conclusion is supported by the observations that inhibition of autophagy, whether by pharmacological agents or genetic silencing, sensitizes the cells to radiation but protects the cells from the combination treatment. In complementary studies, inhibition of AMPK by compound C, a relatively selective inhibitor of AMPK mediated signaling,35 also sensitizes the cells to radiation while protecting the cells from the combination treatment. Our argument for the conversion of one form of autophagy to the other is supported by the fact that sensitization to radiation through autophagy inhibition appears to have been entirely eliminated from the experimental system; otherwise, we might have expected sensitization to radiation by inhibition of the protective autophagy induced by radiation alone to have masked the reversal of sensitization to EB 1089 or 1,25-D3 when cytostatic autophagy was inhibited.
Recent literature has shown that TP53 is involved in regulating autophagy via the MTOR-AMPK pathway in lung cancer cells.36 Our previous work established that TP53 is required for sensitization to radiation in breast tumor cells18,19 and this appears to be the case for NSCLC cells as well, since H358 cells with mutant TP53 did not demonstrate any radio-sensitization by EB 1089. Furthermore, sensitization to radiation by EB 1089 could not be shown in the tp53-null, VDR-expressing H1299 cell line unless TP53 expression was induced under the influence of doxycycline. The requirement for functional TP53 is consistent with the apparent involvement of AMPK in autophagy as TP53 has been shown to act upstream of AMPK;36,37 Additionally, Feng et al. show activation of AMPK in a TP53-dependent manner and further indicate that TP53 can bind to the β subunit of AMPK and lead to its activation.38,39 This upregulation of AMPK has been shown to further suppress MTOR, leading to autophagy induction.38,39 Future studies will be designed to address the putative relationship between TP53 and AMPK in the induction of autophagy in this experimental model as well as other potential components of the putative signaling pathway(s) such as those modulated by the ULK1/2/3/4 family and MTORC.40,41 Further, as would be expected, the presence of the VDR is critical since studies conducted with TP53 wt H838 NSCLC cells, which lack VDR, also showed lack of radiosensitization with the combination treatment.
Drug selectivity and specificity are critical elements in efforts to avoid damage to normal tissue. Off-target actions of drugs can cause severe and undesired side effects. We showed that EB 1089 and 1,25-D3, respectively, do not increase radiation sensitivity in normal bronchial epithelial cells in spite of their expression of VDR42 or in H9C2 cardiomyocytes which have wt TP53,43 the latter being particularly relevant in cases where the heart might fall within the radiation field during the course of treatment.
The current work builds upon and extends our previous findings in breast cancer in demonstrating that 1,25-D3 and the vitamin D analog EB 1089 enhance sensitivity to radiation in NSCLC cells. Again, as in previous work, sensitization is limited to NSCLC cells that express VDR and functional TP53. Furthermore, there is no apparent sensitization in normal lung epithelial cells or cardiomyocytes, consistent with previous work that indicated that sensitization did not occur in untransformed cells.44 The current work also is similar to our previous findings in implicating autophagy as the mechanism underlying the radiosensitization. However, in contrast to the promotion of a cytotoxic form of autophagy in breast tumor cells,18,19,21 we present the novel observation that the autophagy induced by radiation in combination with either 1,25-D3 or EB 1089 has a cytostatic function that nevertheless translates into an enhanced reduction of clonogenic survival. Finally, in these studies, we observe the apparent conversion of the cytoprotective form of autophagy induced by radiation alone to the cytostatic form by radiation in combination with +1,25-D3 or EB 1089. While we have not directly ruled out other modes of “cell death” such as autosis or necroptosis, or methuosis,45-47 in fact we do not actually observe cell death, but extensive and prolonged growth arrest. This fact, accompanied by the reversal of sensitization with autophagy inhibition, appears to support our contention that the nature of autophagy has been altered (i.e., an autophagic switch) rather than that the growth arrest induced by radiation alone has been converted to cell death.
In summary, our current studies for the first time demonstrate an enhanced responsiveness to radiation in NSCLC by 1,25-D3 and EB 1089. Furthermore, this work identifies the conversion of cytoprotective autophagy induced by radiation to a novel cytostatic form of autophagy, which appears to mediate the effectiveness of this treatment combination.
Materials and Methods
Cell lines
The wild-type (WT) TP53 A549 cell line, VDR null H838 cell line, TP53 mutant H358 cell line, and normal bronchial epithelial cell line HBEC3-KT originally generated by the Minna laboratory48 were generously provided by Dr. Charles Chalfant (Virginia Commonwealth University). H460 (WT TP53) NSCLC cells were a generous gift from Dr. Richard Moran at Virginia Commonwealth University. H460 shBECN1, shATG5 cells were generated as described below. H1299 tp53-null and TP53-inducible cells were originally developed by Dr. Constantinos Koumenis49 and provided through our collaborators, Drs. Frank and Suzy Torti, (University of Connecticut).
Mission shRNA bacterial stocks for ATG5 and BECN1 were purchased from Sigma Aldrich (TRCN00151963 and TRCN0000299864, respectively). Lentiviruses were produced in HEK 293TN cells cotransfected using Lipofectamine (Invitrogen, 11668–019) with psPAX2 and pMD2.G packaging constructs (Addgene, 12260, 12259 ). Viruses shed into the media were then used to infect H460 cells. Puromycin (1 ug/ml) was used as the selection marker to enrich the infected cells.
Cell culture and treatment
H460, A549, and H1299 cells were maintained in DMEM media supplemented with 10% (v/v) fetal bovine serum (Thermo Scientific, SH30066.03), 100 U/ml penicillin G sodium (Invitrogen, 15140–122), and 100 μg/ml streptomycin sulfate (Invitrogen, 15140–122). H838 cells were maintained in RPMI 1640 media with the same supplements. HBEC3-KT cells were maintained in Keratinocyte-SFM media (Life Technologies, 12568–010) supplemented with fetal bovine serum (provided with the kit), 100 U/ml penicillin G sodium, and 100 μg/ml streptomycin sulfate. H460 shATG5 and shBECN1 and H1299 shATG5 and shBECN1 cells were maintained with puromycin (1 μg/ml; Sigma, P8833) for selection. All cells were maintained at 37°C under a humidified, 5% CO2 atmosphere. Except where otherwise indicated, after equilibration post-seeding, cells were treated with single dose of 6 Gy radiation alone (using a 137Cs irradiator), 100 nM 1,25-D3 alone, or 100 nM EB 1089 (Tocris Bioscience, 2551, 3993) alone, or the combination of EB 1089 or 1,25-D3 with 6 Gy radiation. Cells were washed free of the 1,25-D3 or EB 1089 after 48 h (i.e., exposure for 24 h prior to and 24 h post irradiation). To induce TP53 expression, H1299 cells were continuously incubated with doxycycline (1 μg/ml; Sigma, 09891) throughout the experiment. Media was replenished once all the treatments were completed. H1299 shATG5 and shBECN1 cell lines were maintained in doxycycline post transfection.
Cell viability and clonogenic survival
Cell viability was assessed by trypan blue exclusion at various time points after the treatment. Cells were harvested using trypsin, stained with 0.4% trypan blue (Sigma, T8154), and counted using a hemocytometer under phase contrast microscopy. For clonogenic studies, cells were plated in 6-well dishes at an appropriate density. After 2 wk, the cells were washed with phosphate-buffered saline (PBS; Life Technologies, 10010023), fixed with 100% methanol, air-dried and stained with 0.1% crystal violet (Sigma, C3886). Cells in groups of 50 or more were counted as colonies and data were normalized relative to untreated controls.
Western blotting
After the indicated treatments, cells were scraped from the culture dishes, collected, and lysed using M-PER mammalian protein extraction reagent (Thermo Scientific, 78505) containing protease and phosphatase inhibitors (Sigma Aldrich, P8340, P5726). Protein concentrations were determined by the Bradford assay (Bio-Rad Laboratories, 500–0205r). Total protein was then diluted in SDS sample buffer and dry boiled for 10 min. Protein samples were subjected to SDS-polyacrylamide gel electrophoresis, transferred to polyvinylidene difluoride membrane, and blocked in 5% milk, 1x PBS, 0.1% Tween 20 (Fisher, BP337) for 1 h. Primary antibodies used at a 1:1000 dilution with overnight incubation were SQSTM1/p62 (BD Biosciences, 610497), ATG5 (Cell Signaling Technology, 2630), BECN1 (BD Biosciences, 612112), VDR (Santa Cruz Biotechnology, sc1008), LC3B-I/LC3B-II (Cell Signaling Technology, 3868), phospho-AMPK (Cell Signaling Technology, 2531), total-AMPK (Cell Signaling Technology, 2795s), phospho-ACAC/acetyl-CoA carboxylase (p-ACC; Cell signaling Technology, 3661s), TP53 (BD Biosciences, 554157), ACTB/β-actin (Sigma, A5316). The membrane was then incubated with secondary antibody of either horseradish peroxidase-conjugated goat anti-rabbit IgG antibody (1:10000; GE Healthcare, NA434) or goat anti-mouse (1:10000; GE Healthcare, NA931) for 1 h, followed by extensive washing with Tween-PBS (PBS with 0.1% Tween 20). Blots were developed using Pierce enhanced chemiluminescence reagents and Bio-Max film (Phenix Reseach Products, F-BX57).
Detection and quantification of acidic vesicles with acridine orange staining
Cells were plated in 6-well culture dishes, stained with acridine orange (Sigma, A6014) at a final dilution of 1:10,000 in respective culture media and allowed to incubate at 37°C for 10 min. After 2 washes with PBS, cells were imaged under an inverted fluorescence microscope (Olympus, Tokyo, Japan). All images were taken at the same magnification. For quantification of autophagic vesicles (AVOs), cells were trypsinized, harvested, and washed with PBS at approximately 1 × 108 cells/ml. Pellet fractions were resuspended in PBS, stained with a 1:10,000 dilution of acridine orange for 10 min and analyzed by BD FACSCanto II (Virginia Commonwealth University) using BD FACSDiva software at the Virginia Commonwealth University Flow Cytometry Core Facility. A minimum of 10,000 cells within the gated region was analyzed. Data collected was further analyzed using FCS Express 4 Flow research edition.
Detection and quantification of senescent cells
The method described by Dimri et al.50 was used for detection of GLB-positive cells. After treatment, cells were washed with PBS and fixed with 2 % formaldehyde/0.2% glutaraldehyde/PBS for 5 to 8 min. Cells were again washed with PBS and stained with 5-bromo-4-chloro-3-inolyl-β-D-galactopyranoside (Fisher, R0404) in dimethylformamide (20 mg/ml), 40 mM citric acid/sodium phosphate, pH 6.0, 5 mM potassium ferrocyanide, 5 mM potassium ferricyanide, 150 mM NaCl, 2 mM MgCl2 and incubated at 37°C for 24 h. After incubation, the cells were washed twice with PBS and visualized using an Olympus inverted microscope. All images were taken at the same magnification. For quantification of SA-GLB/β-galactosidase staining, cells were treated as above and analyzed using a fluorescent GLB activity marker C12FDG (Life Technologies, D2893). The protocol was adapted from Debacq-Chainiaux et al.51
RFP-LC3 and GFP-LC3 redistribution
The RFP-LC3 construct was a generous gift from Dr. Keith Miskimins, Sanford Research, University of South Dakota. H460 cells were transfected with RFP-LC3 using lipofectamine. A549 cells were transfected with GFP-LC3 (Addgene, 22405) using lopofectamine (Invitrogen, 11668–019). Cells were treated as described above, fixed with 3% paraformaldehyde, and visualized using a Leica Confocal laser-scanning microscope (Virginia Commonwealth University). Cells were counterstained with DAPI to stain and visualize the nucleus.
FACS analysis for ANXA5 and propidium Iodide (PI)-positive cells to assess apoptosis
After treatments, cells were collected, washed with PBS, and resuspended in 500 μl of 1x Binding Buffer (ANXA5-FITC apoptosis detection kit; BD Biosciences, 556547) with 5 μl of ANXA5-FITC, and 5 μl of propidium iodide. Samples were gently mixed and incubated at room temperature in the dark for 15 min. The number of cells with increased ANXA5 and PtdIns staining was assessed by flow cytometry and analyzed by BD FACSCanto II using FACSDiva software. A minimum of 10,000 cells within the gates region was analyzed.
FACS analysis for cell cycle distribution
The protocol described by Menon VR et al.52 was used to assess cell cycle distribution. H460 control, H460 shATG5 and shBECN1 cells were seeded in 100-mm tissue culture plates at different densities based on the time points for collection. After 24 h, cells were exposed either to radiation alone, EB 1089 alone, or the combination of EB 1089 and radiation. At indicated time points, 1 x 106 cells were suspended in 1 ml PI solution (Sigma, P4864) with added RNase A (Sigma, R5125) at a final concentration of 0.2 mg/ml. Cells were maintained in the dark at 4°C overnight and analyzed the next day to measure the DNA content by flow cytometry.
Assessment of AMPK phosphorylation and H2AFX/γ-H2AFX formation
After the indicated treatments, cells were washed twice with PBS, fixed with 4% paraformaldehyde for 10 min, permeabilized with 100% cold methanol for 30 min and incubated with blocking buffer (4% BSA [Fisher, NC9857891] in PBS) for 10 min. Cells were incubated with a 1:50 dilution of primary antibody for 1 h, washed with PBS and incubated with a fluorochrome-conjugated secondary antibody (1:100 dilution) for 30 min in the dark. After the second incubation, cells were washed with blocking buffer and resuspended in 400 μl PBS. Cells were analyzed using FACS with a minimum of 10,000 cells for each sample.53 Primary antibodies used were p-PRKAA1/PRKAA2 (AMPK α1/α2 Thr172; Cell signaling Technology, 2531), and anti-p- γ-H2AFX (γ-H2AX Ser139) (EMD Millipore, 07–627).
Statistical analysis
Statistical differences were determined by StatView statistical software (SAS Institute, Cary, NC). The data were expressed as means ± SE (as standard error of the mean). Comparisons were made using one-way analysis of variance (ANOVA) followed by Turkey-Kramer post hoc test, and P values ≤ 0.05 were taken as statistically significant.
Disclosure of Potential Conflicts of Interest
No potential conflicts of interest were disclosed.
Funding
Quantification of PtdIns staining for cell cycle, ANXA5 and PI staining, GLB staining, and acridine orange staining were conducted at the Virginia Commonwealth University Flow Cytometry and Imaging Shared Resource Facility supported in part by NIH Grant P30 CA16059. Confocal microscopy was performed at the VCU Department of Anatomy and Neurobiology Microscopy Facility, supported, in part, with funding from the NIH-NINDS Center core grant 5P30NS047463. These studies were supported in part by R01 CA171101 (SVT, FMT), 5R21 CA171974 (DAG) and CTSA award No. UL1TR000058 from the National Center for Advancing Translational Sciences (DAG). David A. Gewirtz, Ph.D., was supported by an Institutional Research Grant, IRG-14–192–40 from the American Cancer Society.
Supplemental Material
Supplemental data for this article can be accessed on the publisher's website.
References
- 1. Langer CJ. Individualized therapy for patients with non-small cell lung cancer: Emerging trends and challenges. Oncology/Hematology 2012; 83:130–44. [DOI] [PubMed] [Google Scholar]
- 2. Siegel R, Naishadham D, Jemal A. Cancer statistics. CA Cancer J Clin 2012; 62(1):10-29; PMID:22237781 [DOI] [PubMed] [Google Scholar]
- 3. Saintigny P, Burger JA, Recent advances in non-small cell lung cancer biology and clinical management. Discov Med 2012; 13(71):287-97; PMID:22541616 [PubMed] [Google Scholar]
- 4. Wintner LM, Giesinger JM, Zabernigg A, Sztankay M, Meraner V, Pall G, Hilbe W, Holzner B. Quality of life during chemotherapy in lung cancer patients: results across different treatment lines. Br J Cancer 2013; 109(9):2301-8; PMID:24091620; http://dx.doi.org/ 10.1038/bjc.2013.585 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Cai S, Shi A, Yu R, Zhu G. Feasibility of omitting clinical target volume for limited-disease small cell lung cancer treated with chemotherapy and intensity-modulated radiotherapy. Radiat Oncol 2014; 9:17; PMID:24410971; http://dx.doi.org/ 10.1186/1748-717X-9-17 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Bayman N, Blackhall F, McCloskey P, Taylor P, Faivre-Finn C. How can we optimize concurrent cheoradiotherapy for inoperable stage III non-small cell lung cancer? Lung Cancer 2014; n83(2): 117-25; PMID:24373738; http://dx.doi.org/ 10.1016/j.lungcan.2013.11.017 [DOI] [PubMed] [Google Scholar]
- 7. Hamamoto Y, Kataoka M, Nogami N, Kozuki T, Kato Y, Shinohara S, and Shinkai T. Factors affecting survival time after recurrence of non-small-cell lung cancer treated with concurrent chemotherapy. Jpn J Radiol 2012; 30(3):249-54; PMID:22311092; http://dx.doi.org/ 10.1007/s11604-011-0040-9 [DOI] [PubMed] [Google Scholar]
- 8. Sundaram S, Gewirtz DA. The vitamin D3 analog EB 1089 enhances the response of human breast tumor cells to radiation. Radiat Res 1999; 152(5):479-86; PMID:10521924 [PubMed] [Google Scholar]
- 9. Gewirtz DA, Gupta MS, Sundaram S. Vitamin D3 and vitamin D3 analogues as an adjunct to cancer chemo-therapy and radiotherapy. Curr Med Chem Anticancer Agents 2002; 2(6):683-90; PMID:12678720; http://dx.doi.org/ 10.2174/1568011023353697 [DOI] [PubMed] [Google Scholar]
- 10. Sundaram S, Sea A, Feldman S, Strawbridge R, Hoopes PJ, Demidenko E, Binderup L, Gewirtz DA. The combination of a potent vitamin D3 analog, EB 1089, with ionizing radiation reduces tumor growth and induces apoptosis of MCF-7 breast tumor xenografts in nude mice. Clin Cancer Res 2003; 9(6):2350-6; PMID:12796405 [PubMed] [Google Scholar]
- 11. Krishnan AV, Trump DL, Johnson CS, Feldman D. The role of vitamin D in cancer prevention and treatment. Rheum Dis Clin North Am 2012; 38(1):161-78; PMID:22525850; http://dx.doi.org/ 10.1016/j.rdc.2012.03.014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Ramnath N, Kim S, Christenden PJ. Vitamin D and lung cancer. Expert Rev Respir Med 2011; 5(3):305-9; PMID:21702651; http://dx.doi.org/ 10.1586/ers.11.31 [DOI] [PubMed] [Google Scholar]
- 13. Trump DL, Muindi J, Faikh M, Yu WD, Johnson CS. Vitamin D compounds: clinical development as cancer therapy and prevention agents. Anticancer Res 2006; 26(4A):2551-6; PMID:16886663 [PubMed] [Google Scholar]
- 14. Srinivasan M, Parwani VA, Hershberger AP, Lenzner ED, Weissfeld LJ. Nuclear vitamin D receptor expression is associated with improved survival in non-small cell lung cancer. J Steroid Biochem Mol Biol 2011; 123(1-2):30-6; PMID:20955794 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Kim HS, Chen G, King NA, Jeon KC, Christensen JP, Zhao L, Simpson RU, Thomas DG, Giordano TJ, Brenner DE, et al. . Characterization of vitamin D receptor (VDR) in lung adenocarcinoma. Lung cancer 2012; 77(2):265-71; PMID:22564539; http://dx.doi.org/ 10.1016/j.lungcan.2012.04.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Menezes RJ, Cheney RT, Husain A, Tretiakova M, Loewen G, Johnson CS, Jayaprakash V, Moysich KB, Salgia R, Reid ME. Vitamin D receptor expression in normal, premalignant, and malignant human lung tissue. Cancer Epidemiol Biomarkers Prev 2008; 17(5):1104-10; PMID:18483332; http://dx.doi.org/ 10.1158/1055-9965.EPI-07-2713 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Zhou W, Heist SR, Liu G, Asomaning K, Neuberg SD, Hollis BW, Wain JC, Lynch TJ, Giovannucci E, Su L, et al. Circulating 25-Hydroxyvitamin D levels predict survival in early-stage non-small-cell lung cancer patients. J Clin Oncol 2007; 25(5):479-85; PMID:17290055; http://dx.doi.org/ 10.1200/JCO.2006.07.5358 [DOI] [PubMed] [Google Scholar]
- 18. Bristol ML, Di X, Beckman MJ, Wilson EN, Henderson SC, Maiti A, Fan Z, Gewirtz DA. Dual functions of autophagy in the response of breast tumor cells to radiation: cytoprtective autophagy with radiation alone and cytotoxic autophagy in radiosensitization by vitamin D3. Autophagy 2012; 8(5):739-53; PMID:22498493; http://dx.doi.org/ 10.4161/auto.19313 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Wilson EN, Bristol ML, Di X, Maltese WA, Koterba K, Beckman MJ, Gewirtz DA. A switch between cytoprotective and cytotoxic autophagy in the radiosensitization of breast tumor cells by chloroquine and vitamin D. Horm Cancer 2011; 2(5):272-85; PMID:21887591; http://dx.doi.org/ 10.1007/s12672-011-0081-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Haussler MR, Jurutka PW, Mizwicki M, Norman AW. Vitamin D receptor (VDR)- mediated actions of 1α, 25(OH)2vitamin D3: genomic and non-genomic mechanisms. Best Pract Res Clin Endocrinol Metab 2011; 25(4): 543-59; PMID:21872797 [DOI] [PubMed] [Google Scholar]
- 21. Demasters G, Di X, Newsham I, Shiu R, Gewirtz DA. Potentiation of radiation sensitivity in breast tumor cells by the vitamin D3 analogue, EB 1089, through promotion of autophagy and interference with proliferative recovery. Mol Cancer Ther 2006; 5(11):2786-97; PMID:17121925 [DOI] [PubMed] [Google Scholar]
- 22. Prudencio J, Akutsu N, Benlimame N, Wang T, Bastien Y, Lin R, Black MJ, Alaoui-Jamali MA, White JH. Action of low calcemic 1alpha,25-dihydroxyvitamin D3 analogue EB1089 in head and neck squamous cell carcinoma. J Natl Cancer inst 2001; 93(10):745-53; PMID:11353784 [DOI] [PubMed] [Google Scholar]
- 23. Jones KR, Elmore LW, Jackson-Cook C, Demasters G, Povirk LF, Holt SE, Gewirtz DA. p53-dependent accelerated senescence induced by ionizing radiation in breast tumor cells. Int J Radiat Biol 2005; 81(6):1686-700; http://dx.doi.org/ 10.1080/09553000500168549 [DOI] [PubMed] [Google Scholar]
- 24. Gewirtz DA, Holt SE, Elmore LW. Accelerated senescence: an emerging role in tumor cell response to chemotherapy and radiation. Biochem Pharmacol 2008; 76(8):947-57; PMID:18657518; http://dx.doi.org/ 10.1016/j.bcp.2008.06.024 [DOI] [PubMed] [Google Scholar]
- 25. Klionsky DJ, Abdalla FC, Abeliovich H, Abraham RT, Acevedo-Arozena A, Adeli K, Agholme L, Agnello M, Agostinis P, Aguirre-Ghiso JA, et al. . Guidelines for the use and interpretation of assays for monitoring autophagy. Autophagy 2012; 8(4):445-544; PMID:22966490; http://dx.doi.org/ 10.4161/auto.19496 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Ko A, Kanehisa A, Martins I, Senovilla L, Chargari C, Dugue D, Marino G, Kepp O, Michaud M, Perfettini JL, et al. . Autophagy inhibition radiosensitizes in vitro, yet reduces radioresponses in vivo due to deficient immunogenic signaling. Cell Death Differ 2014; 21(1):92-9; PMID:24037090; http://dx.doi.org/ 10.1038/cdd.2013.124 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Lai SL, Pernq RP, Hwang J. p53 gene status modulates the chemosensitivity of non-small cell lung cancer cells. J Biomed Sci 2000; 7(1):64-70; PMID:10644891; http://dx.doi.org/ 10.1007/BF02255920 [DOI] [PubMed] [Google Scholar]
- 28. Luo ZG, Tang H, Li B, Zhu Z, Ni CR, Zhu MH. Genetic alterations of tumor suppressor ING1 in human non-small cell lung cancer. Oncol Rep 2011; 25(4):1073-81; PMID:21286670 [DOI] [PubMed] [Google Scholar]
- 29. You L, Yang CT, Jablons DM. ONYX-015 works synergistically with chemotherapy in lung cancer cell lines and primary cultures freshly made from lung cancer patients. Cancer Res 2000; 60(4):1009-13; PMID:10706117 [PubMed] [Google Scholar]
- 30. Zhang F, Wang W, Tsuji Y, Torti SV, Torti FM. Post-transcriptional modulation of iron homeostasis during p53-dependent growth arrest. J Biol Chem 2008; 283(49):33911-8; PMID:18819919; http://dx.doi.org/ 10.1074/jbc.M806432200 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Hardie DG, Ross FA, Hawley SA. AMPK: a nutrient and energy sensor that maintains energy homeostasis. Nat Rev Mol Cell Biol 2012; 13(4):251-62; PMID:22436748; http://dx.doi.org/ 10.1038/nrm3311 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Inoki K, Kim J, Guan KL. AMPK and mTOR in cellular energy homeostasis and drug targets. Annu Rev Pharmacol Toxicol 2012; 52:381-400; PMID:22017684; http://dx.doi.org/ 10.1146/annurev-pharmtox-010611-134537 [DOI] [PubMed] [Google Scholar]
- 33. Singh K, Matsuyama S, Drazba JA, Almasan A. Autophagy-dependent senescence in response to DNA damage and chronic apoptosis stress. Autophagy 2012; 8(2):236-51; PMID:22240589; http://dx.doi.org/ 10.4161/auto.8.2.18600 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Van Kaick G, Delorme S. Therapy induced effects in normal tissue. Radiologe 2008; 48(9):871-80; PMID:18709346; http://dx.doi.org/ 10.1007/s00117-008-1729-3 [DOI] [PubMed] [Google Scholar]
- 35. Yang WL, Perillo W, Liou D, Marambaud P, Wang P. AMPK inhibitor compound C suppresses cell proliferation by induction of apoptosis and autophagy in human colorectal cancer cells. J Surg Oncol 2012; 106(6):680-8; PMID:22674626; http://dx.doi.org/ 10.1002/jso.23184 [DOI] [PubMed] [Google Scholar]
- 36. Cheng G, Kong D, Hou X, Liang B, He M, Liang N, Ma S, Liu X. The tumor suppressor, p53, contributes to radiosensitivity of lung cancer cells by regulating autophagy and apoptosis. Cancer Biother Radiopharm 2013; 28(2):153-9; PMID:23268708; http://dx.doi.org/ 10.1089/cbr.2012.1297 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Lorin S, Hamaï A, Mehrpour M, and Codogno P. Autophagy regulation and its role in cancer. Semin Cancer Biol 2013; 28(2):152-9; PMID:23811268; http://dx.doi.org/ 10.1016/j.semcancer.2013.06.007 [DOI] [PubMed] [Google Scholar]
- 38. Feng Z, Zhang H, Levine AJ, Jin S. The coordinate regulation of the p53 and mTOR pathways in cells. Proc Natl Acad Sci USA 2005; 102(23):8204-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Feng Z, Hu W, de Stanchina E, Teresky AK, Jin S, Lowe S, Levine AJ. The regulation of AMPK beta1, TSC2 and PTEN expression by p53: stress, cell and tissue specificity, and the role of these gene products in modulating the IGF-1-AKT-mTOR pathways. Cancer Res 2007; 67(7):3043-53; PMID:17409411; http://dx.doi.org/ 10.1158/0008-5472.CAN-06-4149 [DOI] [PubMed] [Google Scholar]
- 40. Jung CH, Jun CB, Ro SH, Kim YM, Otto NM, Cao J, Kundu M, Kim DH. ULK-Atg 13-FIP200 complexes mediate mTOR signaling to autophagy machinery. Mol Biol Cell 2009; 20(7):1992-2003; PMID:19225151; http://dx.doi.org/ 10.1091/mbc.E08-12-1249 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Gao W, Shen Z, Shang L, and Wang X. Up-regulation of human autophagy-initiation kinase ULK1 by tumor suppressor p53 contributes to DNA-damage-induced cell death. Cell Death Differ 2011; 18(10):1598-607; PMID:21475306; http://dx.doi.org/ 10.1038/cdd.2011.33 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Jeong Y, Xie Y, Lee W, Bookout AL, Girard L, Raso G, Behrens C, Wistuba II, Gadzar AF, Minna JD, et al. Research Resource: Diagnostic and Therapeutic potential of nuclear receptor expression in lung cancer. Mol Endocrinol 2012; 26(8):1443-1454; PMID:22700587; http://dx.doi.org/ 10.1210/me.2011-1382 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Yang YF, Wu CC, Chen WP, Chen YL, Su MJ. Prazosin induces p53-mediated autophagic cell death in H9C2 cells. Naunyn Schmiedebergs Arch Pharmacol 2011; 384(2):209-16; PMID:21614555; http://dx.doi.org/ 10.1007/s00210-011-0657-3 [DOI] [PubMed] [Google Scholar]
- 44. Polar MK, Gennings C, Park M, Gupta MS, Gewirtz DA. Effect of vitamin D3 analog ILX 23-7553 on apoptosis and sensitivity to fractionated radiation in breast tumor cells and normal fibroblasts. Cancer Chemother Pharmacol 2003; 51(5):415-21; PMID:12690516 [DOI] [PubMed] [Google Scholar]
- 45. Liu Y, Shoji-Kawata S, Sumpter RM, Wei Y, Ginet V, Zhang L, Posner B, Tran KA, Green DR, Xavier RJ, et al. Autosis is a Na+, K+-ATPase-regulated form of cell death triggered by autophagy-inducing peptides, starvation, and hypoxia-ischemia. Proc Natl Acad Sci U S A 2013; 110(51): 20364-71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Overmeyer JH, Maltese WA. Death pathways triggered by activated Ras in cancer cells. Front Biosci 2011; 16:1693-713; PMID:21196257; http://dx.doi.org/ 10.2741/3814 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Fulda S. The mechanism of necroptosis in normal and cancer cells. Cancer Biol Ther 2013; 14(11):999-1004; PMID:24025353; http://dx.doi.org/ 10.4161/cbt.26428 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Sato M, Vaughan MB, Girard L, Peyton M, Lee W, Shames DS, Ramirez RD, Sunaga N, Gazdar AE, Shaw JW, et al. Multiple oncogenic changes (K-RAS(V12), p53 knockdown, mutant EGFRs. P16 bypass, telomerase are not sufficient to confer a full malignant phenotype on human bronchial epithelial cells. Cancer Res 2006; 66(4):2116-28; PMID:16489012 [DOI] [PubMed] [Google Scholar]
- 49. Maecker HL, Koumenis C, Giaccia AJ. P53 promotes selection for Fas-mediated apoptotic resistance. Cancer Res 2000; 60(16):4638-44; PMID:10969818 [PubMed] [Google Scholar]
- 50. Dimri GP, Lee X, Basile G, Acosta M, Scott G, Roskelley C, Medrano EE, Linskens M, Rubelj I, Pereira-smith O, et al. . A biomarker that identifies senescent human cells in culture and in aging skin in vivo. Proc Natl Acad Sci USA 1995; 92(20):9363-7; PMID:7568133 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Debacq-Chainiaux F, Erusalimsky JD, Campisi J, Toussaint O. Protocols to detect senescence-associated β-galactosidase (SA-betagal) activity, a biomarker of senescent cells in culture and in vivo. Nat Protoc 2009; 4(12):1798-806; PMID:20010931; http://dx.doi.org/ 10.1038/nprot.2009.191 [DOI] [PubMed] [Google Scholar]
- 52. Menon VR, Peterson EJ, Valerie K, Farrell NP, Povirk LF. Ligand modulation of a dinuclear platinum compound leads to mechanistic differences in cell cycle progression and arrest. Biochem Pharmacol 2013; 86(12): 1708-20; PMID:24161784; http://dx.doi.org/ 10.1016/j.bcp.2013.10.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Gagou EM, Zuazua-Villar P, Meuth M. Enhanced H2Ax phosphorylation, DNA replication fork arrest, and cell death in the absence of Chk1. Mol Bio Cell 2010; 21(5):739-52; PMID:20053681; http://dx.doi.org/ 10.1016/10.1091/mbc.E09-07-0618 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.