Abstract
Purpose
Concerns about the potential for genomic advances to increase health disparities have been raised. Thus it is important to assess referral and uptake of genetic counseling (GC) and testing in minority populations at high risk for hereditary breast and ovarian cancer (HBOC).
Methods
Black women diagnosed with invasive breast cancer ≤ age 50 in 2009-2012 were recruited through the Florida State Cancer Registry 6-18 months following diagnosis and completed a baseline questionnaire. Summary statistics, Chi-square tests, and path modeling were conducted to examine which demographic and clinical variables were associated with referral and access to genetic services.
Results
Of the 440 participants, all met national criteria for GC yet only 224 (51%) were referred for or received GC and/or HBOC testing. Variables most strongly associated with healthcare provider referral for GC included having a college education (OR=2.1), diagnosis at or below age 45 (OR=2.0), and triple negative tumor receptor status (OR=1.7). The strongest association with receipt of GC and/or HBOC testing was healthcare provider referral (OR=7.9), followed by private health insurance at diagnosis (OR=2.8), and household income greater than $35,000 in the year prior to diagnosis (OR=2.0).
Conclusions
Study findings suggest efforts are needed to improve genetic services access among a population-based sample of high-risk Black women. These results indicate that socioeconomic factors and physician referral patterns contribute to disparities in access to genetic services within this underserved minority population.
Keywords: BRCA testing, genetic counseling, Hereditary Breast Cancer, Cancer Registry, Disparities
Introduction
Of all cancers which develop in women in the United States (US), breast cancer has the highest cancer incidence regardless of race or ethnicity [1], and is the second most common cause of cancer-related death among Caucasian and Black women [2]. Approximately 5-10% of BC is due to highly penetrant inherited gene mutations [3,4], the majority of which are attributed to the BRCA1 and BRCA2 (BRCA) genes [5,6]. The disproportionate cancer burden faced by those with hereditary breast and ovarian cancer (HBOC) may be put into perspective by comparing cancer risks in BRCA carriers (~60-70%) to those in the general population (~8%) [7,8]. Identifying breast cancer survivors who are BRCA carriers, empowers them with options to substantially reduce their high risk for a second primary breast cancer [9] as well as their 20-40% lifetime risk for ovarian cancer [7,8,10,11].
Within the US, any physician may order BRCA testing; however, genetic counseling (GC) prior to testing remains the gold standard, endorsed by many professional organizations [12-15]. According to U.S. national practice guidelines, all women with breast cancer diagnosed age ≤50 years should be offered genetic risk assessment/counseling services [14]. However, national data indicate that only ~50% of high risk BC patients are referred for BRCA counseling or testing [16], with lower access to testing among Blacks and Hispanics [17,18]. In addition to socioeconomic factors that may contribute to variation in access [18,17], there is less awareness of testing among minorities [19,20].
Growing healthcare disparities in HBOC testing necessitate improving our understanding of access among minorities as was recently identified as a research gap by the US Preventive Services Task Force (USPSTF) [21]. In a population-based sample of Black women with early onset breast cancer, we sought to evaluate the prevalence of and factors associated with: 1) referral to genetic counseling (GC) and 2) access to genetic services (including GC attendance and/or BRCA testing).
Methods
Participants
Eligible participants were self-identified Black women diagnosed with invasive breast cancer at or below age 50 between the years 2009-2012 living in Florida at the time of diagnosis, alive at the time of recruitment (6-18 months after diagnosis), and English speaking. Upon approval of the institutional review boards of the University of South Florida and the Florida Department of Health (DOH), recruitment was initiated using contact information on all eligible participants released by the Florida State Cancer Registry.
Participants were recruited 6-18 months after their diagnosis using previously described state-mandated recruitment methods [22], which consisted of 2 mailings, 3 weeks apart, including a ‘telephone response card’ to give potential participants the option to either decline (i.e., indicating that they did not wish to be contacted by phone) or express interest in participation (i.e., indicating that they were interested in having the study team call them to follow-up). If no response was received within 3 weeks of the second mailing, a member of the study team attempted to contact the potential participant by telephone to explain the study and determine interest in participation. For those willing to participate, written informed consent was obtained and a baseline study questionnaire was completed.
Measures
Clinical (i.e., age at diagnosis, stage of diagnosis, histologic subtype, tumor receptor status) and demographic (i.e., primary insurance at diagnosis and employment at diagnosis) data were obtained from the cancer registry for all potential participants in the sampling frame. Additional information was obtained from consented participants who completed a risk-factor questionnaire, including: partner status (dichotomized as married or living together versus other), income in the year prior to diagnosis (dichotomized as ≥$35,000 versus less than $35,000 to approximate the median household income for Blacks in the US), history of breast cancer in one or more 1st-3rd degree relatives (dichotomized as present versus absent), history of ovarian cancer in one or more 1st-3rd degree relatives (dichotomized as present versus absent), and educational attainment (dichotomized as college graduate versus no college degree). Age at first breast cancer diagnosis was dichotomized as ≤45 vs. >45 based on the 2014 NCCN guideline that all women diagnosed at or under age 45 be tested for HBOC [14]. Complete tumor receptor status information was available from the registry for 291 participants. An additional 77 were positive for at least one of the three receptors (ER, PR, or HER2) and classified as non-triple negative. Among the remaining 72 cases in which triple negative (TN) tumor status could not be determined from cancer registry data, this information was obtained from medical records in 19 cases, through self-report in 14 cases, and remained unknown in 39 cases. Insurance status was dichotomized according to cancer registry data as those having private insurance at the time of diagnosis versus the uninsured and those with any type of government insurance (i.e., Medicare, Medicaid, Indian health services, Military) in the other group. There were 38 individuals reported as having “insurance not otherwise specified”, which was considered missing data. Risk for a BRCA mutation was estimated using 3-generation pedigrees obtained as part of the study in conjunction with Myriad risk assessment tables.
Additional survey items assessed whether participants had been: 1) referred to genetic counseling by a healthcare provider to learn about hereditary cancer risks; 2) saw a genetic counselor to learn about cancer risks; and 3) had BRCA testing. Results were verified through collection of the BRCA test report in 86% of individuals who indicated that they had undergone testing.
Data Analysis
Demographic and clinical characteristics available through the cancer registry were summarized using descriptive statistics. To assess for differences between consented participants versus all others in the sampling frame within the registry, comparisons were made using Pearson's chi-squared test.
Response frequencies were assessed to create a Venn diagram showing the numbers and proportions of those who were referred to and/or accessed genetic counseling and/or testing. Bivariate analyses were conducted using Pearson's chi-squared test to determine significant differences between those who were referred to or accessed genetic services (i.e., GC and/or BRCA testing) and those who were neither referred nor accessed services. A two-sided p-value of ≤0.05 was considered statistically significant. Bivariate analyses were conducted using SPSS Statistics for Windows, Version 20.
To further evaluate factors associated with referral to and receipt of genetic services, a multivariable path model was conducted using Mplus version 6.12. To simultaneously adjust for influential factors, those demographic and clinical variables with a p-value ≤0.2 in bivariate analyses were included in the model. Specifically, direct paths were modeled between referred for GC and the following independent variables: diagnosed at age ≤45; annual household income ≥$35,000; college graduate; private insurance; triple negative tumor status; ovarian cancer family history; and breast cancer family history. Paths were also specified between receipt of genetic services and these same independent variables with the exception of ovarian cancer family history. An additional direct path was added to evaluate the relationship between referral for GC and the final outcome (i.e., receipt of genetic services). Relative strengths of associations between variables in the model were determined using adjusted odds ratios (OR) with 95% confidence intervals.
Results
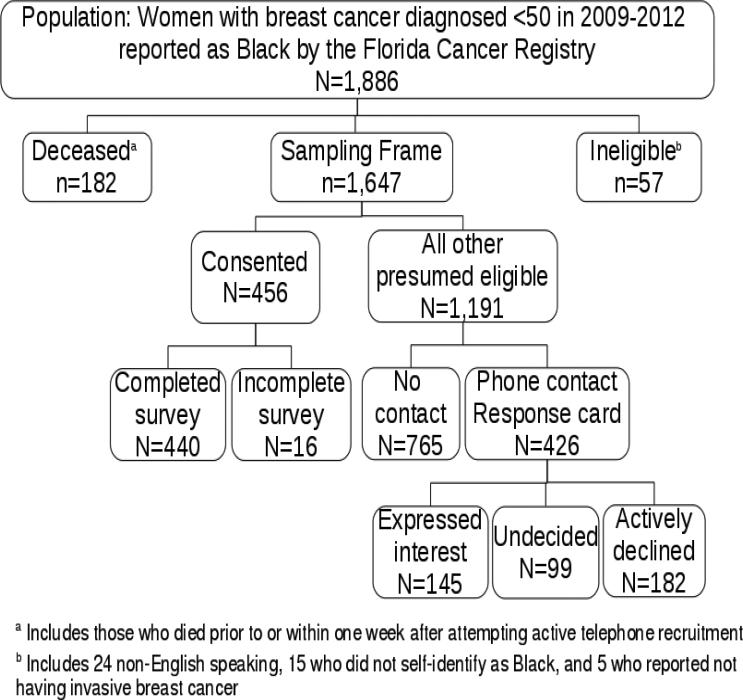
A total of 1,886 women with breast cancer diagnosed ≤ age 50 in 2009-2012 were reported as Black by the Florida Cancer Registry. After excluding 182 who were deceased and 57 deemed ineligible for other reasons, the total number in the sampling frame was 1,647 (Figure 1). Participants included all 440 women meeting inclusion criteria who consented to the study and completed the baseline questionnaire. Demographic and clinical comparisons between the 440 participants and all others in the sampling frame revealed no statistically significant differences (Table 1).
Figure 1.
Breakdown of the population, sampling frame, and study participants
Table 1.
Clinical and demographic variables of participants and non-participants
| Participants N=440 | Non-participantsa N= 1207 | P-value | |
|---|---|---|---|
| Mean age at cancer diagnosis (SD) | 42.10 (6.09) | 42.61 (6.37) | 0.15 |
| Stage (n (%)) | |||
| Localized | 230 (52.3) | 580 (48.1) | 0.08 |
| Regional | 182 (41.4) | 502 (41.6) | |
| Distant | 21 (4.8) | 95 (7.9) | |
| Unstaged | 7 (1.6) | 30 (2.5) | |
| Histologic subtype (n (%)) | |||
| Ductal | 347 (78.9) | 976 (80.9) | 0.47 |
| Lobular | 25 (5.7) | 48 (4.0) | |
| Mixed | 29 (6.6) | 84 (7.0) | |
| Other/Unknown | 39 (8.9) | 99 (8.2) | |
| ER/PR/HER2 receptor status known (n (%)) | 291 (66.1) | 776 (64.3) | 0.49 |
| Receptor status (n (%))b | |||
| Triple negative | 78 (26.8) | 188 (24.2) | 0.39 |
| Non-triple negative | 213 (73.2) | 588 (75.8) | |
| Married or cohabiting (n (%)) | |||
| Yes | 173 (39.3) | 524 (43.4) | 0.26 |
| no | 260 (59.1) | 659 (54.6) | |
| Unknown | 7 (1.6) | 24 (2.0) | |
| Insurance at diagnosis (n (%)) | |||
| Not Insured | 47 (10.7) | 114 (9.4) | 0.79 |
| Private Insurance | 245 (55.7) | 709 (58.7) | |
| Medicaid | 73 (16.6) | 192 (15.9) | |
| Medicare | 21 (4.8) | 45 (3.7) | |
| Military or Indian public health services | 16 (3.6) | 34 (2.8) | |
| Other Insurance | 33 (7.5) | 94 (7.8) | |
| Unknown | 5 (1.1) | 19 (1.6) | |
| Employment at diagnosis (n (%)) | |||
| Unemployed | 51 (11.6) | 133 (11.0) | 0.62 |
| Employed | 275 (62.5) | 732 (60.6) | |
| Unknown | 114 (25.9) | 342 (28.3) | |
| Metropolitan (n (%)) | 422 (95.9) | 1159 (96.5) | 0.92 |
access to genetic services
There were 16 individuals included with “non-participants” who consented but did not complete the baseline survey.
Valid percentages are shown based on those individuals where information for all three receptors was known.
Given the study inclusion criteria of participants diagnosed with invasive breast cancer ≤50, all met national guidelines for GC referral [14]. However, 216 (49%) were neither referred for GC by a healthcare provider nor accessed genetic services (including GC or BRCA testing) (Figure 2). Only 152 (35%) were referred for GC, with 63% of referrals being made by oncologists. Of the 91 who saw a GC, 70 underwent BRCA testing. An additional 89 underwent BRCA testing (including 63 who were not referred for genetic counseling and 26 who were referred but did not see a GC).
Figure 2.
Referral and access to genetic services among young Black breast cancer survivors (N=440)
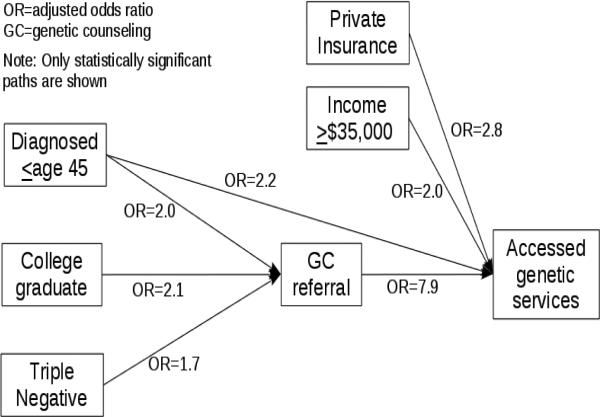
Bivariate comparisons based on referral and access to genetic services revealed several significant associations (Table 2). Of the 440 participants, 97 were excluded from the multivariable path model due to missing data. When controlling for all other variables in the model, significant positive associations with referral for GC included: college education (OR=2.1, 95% CI [1.3, 3.3]); diagnosed ≤age 45 (OR=2.0, 95% CI= [1.3, 3.0]; and triple negative tumor (OR=1.7, 95% CI [1.1, 2.6]. Referral for GC was also strongly associated with access to genetic services, including GC or BRCA testing (OR=7.9 ; 95% CI= [5.0, 12.7]). Other variables demonstrating direct associations with access to genetic services included: private health insurance (OR=2.8, 95% CI [1.7, 4.6]); diagnosed ≤age 45 (OR=2.2, 95% CI [1.4, 3.6]); and household income >$35,000 (OR=2.0, 95% CI [1.2, 3.4]). A simplified version of the model is shown in Figure 3 and illustrates the statistically significant paths. Although bivariate analyses found significant associations between receipt of genetic services and two other variables (i.e., being a college graduate and triple negative cancer), their lack of statistical significance suggests that their influence is primarily mediated through referral for GC; thus, these paths are not shown in the final model (Figure 3).
Table 2.
Comparisons of demographics and clinical characteristics based on referral for genetic counseling (GC) and whether genetic services were accessed
| Referred for GC N=152 | Not referred N=288 | P-value | Accessed services N=180 | Not accessed N=260 | P-value | |
|---|---|---|---|---|---|---|
| Age at first breast cancer (n (%)) | ||||||
| ≤40 years | 76 (50) | 90 (31) | <0.001* | 95 (53) | 71 (27) | <0.001* |
| 41-45 years | 37 (24) | 83 (29) | 44 (24) | 76 (29) | ||
| 46-50 years | 39 (26) | 115 (40) | 41 (23) | 113 (43) | ||
| Insurance at diagnosis (n (%)) | ||||||
| Private | 94 (62) | 144 (50) | 0.009* | 128 (71) | 110 (42) | <0.001* |
| Governmenta | 44 (29) | 120 (42) | 39 (22) | 125 (48) | ||
| Unknownb | 14 (9) | 24 (8) | 13 (7) | 25 (10) | ||
| Annual household income (n (%)) | ||||||
| <$35,000 | 61 (40) | 155 (54) | 0.003* | 62 (34) | 154 (59) | <0.001* |
| ≥$35000 | 82 (54) | 112 (39) | 109 (61) | 65 (25) | ||
| Unknownb | 9 (6) | 21 (7) | 9 (5) | 21 (8) | ||
| Education (n (%)) | ||||||
| College graduate | 80 (53) | 95 (33) | <0.001* | 97 (54) | 78 (30) | <0.001* |
| Some college or vocational training | 47 (31) | 108 (38) | 60 (33) | 95 (36) | ||
| <High school/GED | 25 (16) | 83 (29) | 23 (13) | 85 (33) | ||
| Unknownb | 0 | 2 (1) | 0 | 2 (1) | ||
| Receptor status (n (%))c | ||||||
| Triple negative | 43 (28) | 58 (20) | 0.107 | 53 (29) | 48 (18) | 0.009* |
| Non-triple negative | 101 (66) | 199 (69) | 113 (63) | 187 (72) | ||
| Unknownb | 8 (5) | 31 (11) | 14 (8) | 25 (10) | ||
| Family history of ovarian cancer (n (%))d | ||||||
| Yes | 26 (17) | 33 (11) | 0.098 | 27 (15) | 32 (12) | 0.415 |
| No | 126 (83) | 255 (89) | 153 (85) | 228 (88) | ||
| Family history of breast cancer (n (%))d | ||||||
| Yes | 92 (61) | 156 (54) | 0.201 | 111 (62) | 137 (53) | 0.062 |
| No | 60 (39) | 132 (46) | 69 (38) | 123 (47) | ||
| Risk for carrying a BRCA mutation (n (%))e | ||||||
| >10% risk | 53 (35) | 95 (33) | 0.642 | 62 (34) | 86 (33) | 0.747 |
| ≤ 10% risk | 95 (62) | 188 (65) | 114 (63) | 169 (65) | ||
| missing | 4 (3) | 5 (2) | 4 (2) | 5 (2) | ||
| Married or cohabiting (n (%))e | ||||||
| Yes | 62 (41) | 115 (40) | 0.906 | 76 (42) | 101 (39) | 0.420 |
| No | 90 (59) | 171 (59) | 102 (57) | 159 (61) | ||
| Unknown | 0 | 2 (1) | 2 (1) | 0 | ||
| Children (n (%)) | ||||||
| Yes | 134 (88) | 252 (88) | 0.841 | 156 (87) | 230 (90) | 0.573 |
| No | 18 (12) | 36 (12) | 24 (13) | 30 (11) |
Statistically significant at p<0.05
Government insurance includes Medicare, Medicaid, Indian health services, and Military.
Unknown were not included in analyses.
Numbers for whom receptor status is known differ from Table 1 because study participants who were positive for any one of the three receptors (ER, PR, or HER2) were coded as non-triple negative and receptor status was determined through medical records or self-report for others.
One or more first, second, or third degree relatives affected.
Calculated using pedigree data and Myriad risk assessment prevalence table.
f Number of married participants differs from Table 1 given that marital status from baseline surveys are reported here.
Figure 3.
Demographic and clinical associations between genetic counseling referral
Discussion
Through a population-based study of young Black women with breast cancer, our findings indicate that approximately half were either referred for or accessed genetic services, despite national practice guidelines which indicate all should have been referred. Our findings highlight the need to improve identification of high risk patients and address existing disparities in access.
Other registry-based efforts to determine patient referral and/or access to genetic services among breast cancer survivors are limited to two prior studies [23,24]. In a study of 289 breast cancer survivors diagnosed <50, 42.2% reported receiving GC [23]. Blacks were underrepresented among the study sample, comprising 8.3% of respondents, yet 21.3% of non-respondents. Nevertheless, among the 24 Black participants, only 5 (21%) reported having received GC, which is similar to the proportion who reported having seen a genetic counselor in our study (i.e., 22.5%). In another registry-based study which included 546 high risk breast cancer survivors, McCarthy et al reported that 53% of participants reported that a physician had recommended BRCA testing to them. Given that 90% of participants in this study were White, no data were reported according to race, precluding race-based comparisons with our results.
Nonetheless, our finding that nearly half of the Black women were referred to or accessed genetic services was higher than expected based on reports of racial disparities in physician referral [25] and access [19,18,25,17]. Furthermore, the proportion of young Black breast cancer survivors who had BRCA testing in our study (36%) was substantially higher than expected given that only 12% of 82 Black women had testing in a national study of insured breast cancer survivors diagnosed between age 20-40 years [17]. However, this earlier study was based on records from 2004-2007 and reported that women diagnosed in 2007 were 3.7 times as likely to be tested as those diagnosed in 2004. Thus, our higher rates of access to GC and/or BRCA testing may be explained by our more recent years of recruitment.
Another potential factor contributing to the relatively high rate of BRCA testing observed in our study could be the influence of multiple direct-to-consumer marketing campaigns in Florida [26-28]. Additionally, marketing to community-based physicians by commercial laboratories may contribute to increasing numbers of clinicians across disciplines who order genetic tests [29,30]. Thus, such marketing efforts may have contributed to the higher than expected rates of testing observed in young Black breast cancer survivors in our study.
Although the proportion of Black women who accessed clinical BRCA testing in our study is encouraging, substantial disparities in genetic services referral and access exist even within this minority population. Given that receipt of a referral from a healthcare provider was the strongest predictor of accessing genetic services in both our study and the recent McCarthy et al study [24], improving appropriate referral rates is critical. Our model indicates that diagnosis at or below age 45 and triple negative disease are both associated with referral, which suggests that healthcare providers recognize that these risk factors confer an increased likelihood of hereditary breast cancer, consistent with other research [31]. On the other hand, in contrast to a recent study [31], family history of cancer does not appear to strongly influence referral practices of healthcare providers in our study. Despite this conflicting finding, our results are consistent with studies that suggest inadequate elicitation and documentation of cancer family history in medical records [16,32,33]. Our finding that those patients with a college education were more likely than those without a college education to report receiving a referral for genetic counseling is consistent with findings from other studies [24,34] and could be the result of several possibilities including: 1) healthcare providers may be more likely to refer college educated patients than those with lower educational attainment, 2) healthcare providers who treat more highly educated patients may be more likely to refer their patients compared to those healthcare providers who treat patients with lower educational attainment, and 3) patients with higher education may be more aware of HBOC and/or more likely to bring this up with their healthcare provider and request a referral even though all of the women in this study would have been eligible for GC based on national guidelines. Congruent with results of other studies [23,17], our results highlight financial and insurance barriers associated with disparities in access to genetic services.
Notably, women in our study diagnosed at or below age 45 were more likely to be both referred to and access genetic services. Other studies have also reported an association between younger age at breast cancer diagnosis and receipt of genetic services [23,35]. This may be the result of NCCN guidelines which indicate that breast cancer diagnosis ≤ age 45 is sufficient to warrant BRCA testing regardless of family history. Furthermore, many insurers cover BRCA testing in women ≤ age 45 regardless of family history whereas those over age 45 usually require additional personal and/or family history of cancer [36].
The current study has a number of strengths including the population-based design resulting in a large representative sample of young Black breast cancer survivors in the state of Florida based on available clinical and demographic variables. As a result, findings are likely to provide a more accurate and updated estimate of GC referral and receipt of genetic services within this underserved minority population compared to prior studies that had limited representation of Black women, used non-population based sampling, or were conducted earlier. Furthermore, we were able to confirm access to BRCA testing through medical record verification in most cases. Other strengths include the use of path modeling which examined referral and access to genetic services simultaneously while controlling for the effects of other demographic and clinical variables.
Despite these strengths, there remain some limitations such as our inability to confirm that observed associations were causal. However, given that variables hypothesized to contribute to genetic services access occurred prior to accessing services, the logic of the path model is supported. Furthermore, there was missing data; however, findings from sensitivity analyses conducted for variables that had missing data for more than 5% of cases (i.e., tumor receptor status, private insurance, and income) determined that bivariate relationships were robust regardless of which dichotomous category the missing data were included (data not shown). Other considerations include reliance on self-report with regard to GC referral and receipt of genetic counseling. However, receipt of genetic testing was verified in most cases. Finally, our findings were based on access in young Black women, thus we were unable to compare across populations. However another funded effort is currently underway to recruit non-Blacks within the same sampling frame so that access can be compared across populations.
In summary, our findings demonstrate that among young Black breast cancer survivors, socioeconomic factors and physician referral patterns contribute to disparities in access to BRCA testing. However, it remains important to better understand the etiology of these disparities in access through assessing other potentially modifiable facilitators and barriers to genetic service access such as psychological, cultural and geographic factors. In addition to patient-specific factors, it is also important to examine factors associated with genetic referral practices of clinical providers, and develop solutions by which to facilitate identification and referral of high risk patients. Ultimately, concerted efforts at multiple levels are needed to address differential access and utilization of GC and testing among young Black women in order reduce the growing healthcare disparities in clinical cancer genetics.
Acknowledgements
This work was supported by grants through the Bankhead Coley Granting agency (IBG10-34199) and the American Cancer Society (RSG-11-268-01-CPPB). Support for Deborah Cragun's time was provided by a NCI R25T training grant awarded to Moffitt Cancer Center (5R25CA147832-04).
This work has been supported in part by the Biostatistics Core (5P30CA076292-16) and Survey Core at the Moffitt Cancer Center, a National Cancer Institute Comprehensive Cancer Center (P30-CA076292).
Cancer data for this study (analysis) was provided by the Florida Department of Health, Florida Cancer Data System (FCDS). Cancer data are made available to aid public health surveillance and research to advance cancer control and prevention activities to better serve the population at risk for developing cancer and improve treatment for cancer patients. The contents of this study are solely the responsibility of the authors and do not necessarily reflect the official view of the Florida Department of Health, Florida Cancer Data System.
We thank the following members of our Community Advisory Panel for their valuable input: Joyce Austin, Sue Friedman, Benita Hayes, Evora Pimento, Peggie Sherry, Cheryl Clinton, Gwendolyn Dawson, Gloria Wood, Linda Paige, Deneen Wyman, Khaliah Fleming, and Valerie Poindexter.
Footnotes
Conflicts of interest:
The authors report no conflicts of interest
References
- 1.Society AC . Cancer Facts & Figures 2013 & 2014. American Cancer Society, Inc.; 2014. [February 10 2014]. http://www.cancer.org/acs/groups/content/@research/documents/document/acspc-042725.pdf. [Google Scholar]
- 2.Centers for Disease Control and Prevention [Jan 10 2014];Breast Cancer Statistics. 2013 http://www.cdc.gov/cancer/breast/statistics/.
- 3.Filippini SE, Vega A. Breast cancer genes: beyond BRCA1 and BRCA2. Frontiers in bioscience (Landmark edition) 2013;18:1358–1372. doi: 10.2741/4185. [DOI] [PubMed] [Google Scholar]
- 4.Beggs AD, Hodgson SV. Genomics and breast cancer: the different levels of inherited susceptibility. European journal of human genetics : EJHG. 2009;17(7):855–856. doi: 10.1038/ejhg.2008.235. doi:10.1038/ejhg.2008.235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Miki Y, Swensen J, Shattuck-Eidens D, Futreal PA, Harshman K, Tavtigian S, Liu Q, Cochran C, Bennett LM, Ding W, et al. A strong candidate for the breast and ovarian cancer susceptibility gene BRCA1. Science. 1994;266(5182):66–71. doi: 10.1126/science.7545954. [DOI] [PubMed] [Google Scholar]
- 6.Wooster R, Bignell G, Lancaster Jea. Identification of the breast cancer susceptibility gene BRCA2. Nature. 1995;378(6559):789–792. doi: 10.1038/378789a0. [DOI] [PubMed] [Google Scholar]
- 7.Mavaddat N, Peock S, Frost D, Ellis S, Platte R, Fineberg E, Evans DG, Izatt L, Eeles RA, Adlard J, Davidson R, Eccles D, Cole T, Cook J, Brewer C, Tischkowitz M, Douglas F, Hodgson S, Walker L, Porteous ME, Morrison PJ, Side LE, Kennedy MJ, Houghton C, Donaldson A, Rogers MT, Dorkins H, Miedzybrodzka Z, Gregory H, Eason J, Barwell J, McCann E, Murray A, Antoniou AC, Easton DF. Cancer risks for BRCA1 and BRCA2 mutation carriers: results from prospective analysis of EMBRACE. Journal of the National Cancer Institute. 2013;105(11):812–822. doi: 10.1093/jnci/djt095. doi:10.1093/jnci/djt095. [DOI] [PubMed] [Google Scholar]
- 8.Antoniou A, Pharoah PD, Narod S, Risch HA, Eyfjord JE, Hopper JL, Loman N, Olsson H, Johannsson O, Borg A, Pasini B, Radice P, Manoukian S, Eccles DM, Tang N, Olah E, Anton-Culver H, Warner E, Lubinski J, Gronwald J, Gorski B, Tulinius H, Thorlacius S, Eerola H, Nevanlinna H, Syrjakoski K, Kallioniemi OP, Thompson D, Evans C, Peto J, Lalloo F, Evans DG, Easton DF. Average risks of breast and ovarian cancer associated with BRCA1 or BRCA2 mutations detected in case Series unselected for family history: a combined analysis of 22 studies. American journal of human genetics. 2003;72(5):1117–1130. doi: 10.1086/375033. doi:10.1086/375033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Metcalfe K, Gershman S, Lynch HT, Ghadirian P, Tung N, Kim-Sing C, Olopade OI, Domchek S, McLennan J, Eisen A, Foulkes WD, Rosen B, Sun P, Narod SA. Predictors of contralateral breast cancer in BRCA1 and BRCA2 mutation carriers. British journal of cancer. 2011;104(9):1384–1392. doi: 10.1038/bjc.2011.120. doi:10.1038/bjc.2011.120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Roukos DH, Briasoulis E. Individualized preventive and therapeutic management of hereditary breast ovarian cancer syndrome. Nat Clin Pract Oncol. 2007;4(10):578–590. doi: 10.1038/ncponc0930. [DOI] [PubMed] [Google Scholar]
- 11.Narod SA, Offit K. Prevention and management of hereditary breast cancer. J Clin Oncol. 2005;23(8):1656–1663. doi: 10.1200/JCO.2005.10.035. [DOI] [PubMed] [Google Scholar]
- 12.American College of O, Gynecologists, Bulletins--Gynecology ACoP, Genetics ACo, Society of Gynecologic O ACOG Practice Bulletin No. 103: Hereditary breast and ovarian cancer syndrome. Obstetrics and gynecology. 2009;113(4):957–966. doi: 10.1097/AOG.0b013e3181a106d4. doi:10.1097/AOG.0b013e3181a106d4. [DOI] [PubMed] [Google Scholar]
- 13.Riley BD, Culver JO, Skrzynia C, Senter LA, Peters JA, Costalas JW, Callif-Daley F, Grumet SC, Hunt KS, Nagy RS, McKinnon WC, Petrucelli NM, Bennett RL, Trepanier AM. Essential elements of genetic cancer risk assessment, counseling, and testing: updated recommendations of the National Society of Genetic Counselors. Journal of genetic counseling. 2012;21(2):151–161. doi: 10.1007/s10897-011-9462-x. doi:10.1007/s10897-011-9462-x. [DOI] [PubMed] [Google Scholar]
- 14.National Comprehensive Cancer Network [July 10 2014];Genetic/Familial High-risk Assessment: Breast and Ovarian. 2014 http://www.nccn.org/professionals/physician_gls/PDF/genetics_screening.pdf.
- 15.Robson ME, Storm CD, Weitzel J, Wollins DS, Offit K. American Society of Clinical Oncology Policy Statement Update: Genetic and Genomic Testing for Cancer Susceptibility. J Clin Oncol. 2010 doi: 10.1200/JCO.2009.27.0660. [DOI] [PubMed] [Google Scholar]
- 16.Wood ME, Kadlubek P, Pham TH, Wollins DS, Lu KH, Weitzel JN, Neuss MN, Hughes KS. Quality of cancer family history and referral for genetic counseling and testing among oncology practices: a pilot test of quality measures as part of the American Society of Clinical Oncology Quality Oncology Practice Initiative. J Clin Oncol. 2014;32(8):824–829. doi: 10.1200/JCO.2013.51.4661. doi:10.1200/jco.2013.51.4661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Levy DE, Byfield SD, Comstock CB, Garber JE, Syngal S, Crown WH, Shields AE. Underutilization of BRCA1/2 testing to guide breast cancer treatment: black and Hispanic women particularly at risk. Genet Med. 2011;13(4):349–355. doi: 10.1097/GIM.0b013e3182091ba4. doi:10.1097/GIM.0b013e3182091ba4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Armstrong K, Micco E, Carney A, Stopfer J, Putt M. Racial differences in the use of BRCA1/2 testing among women with a family history of breast or ovarian cancer. Jama. 2005;293(14):1729–1736. doi: 10.1001/jama.293.14.1729. [DOI] [PubMed] [Google Scholar]
- 19.Mai PL, Vadaparampil ST, Breen N, McNeel TS, Wideroff L, Graubard BI. Awareness of cancer susceptibility genetic testing: the 2000, 2005, and 2010 National Health Interview Surveys. American journal of preventive medicine. 2014;46(5):440–448. doi: 10.1016/j.amepre.2014.01.002. doi:10.1016/j.amepre.2014.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Pagan JA, Su D, Li L, Armstrong K, Asch DA. Racial and ethnic disparities in awareness of genetic testing for cancer risk. American journal of preventive medicine. 2009;37(6):524–530. doi: 10.1016/j.amepre.2009.07.021. doi:10.1016/j.amepre.2009.07.021. [DOI] [PubMed] [Google Scholar]
- 21.Moyer VA. Risk Assessment, Genetic Counseling, and Genetic Testing for BRCA-Related Cancer in Women: U.S. Preventive Services Task Force Recommendation Statement. Annals of internal medicine. 2013 doi: 10.7326/M13-2747. doi:10.7326/m13-2747. [DOI] [PubMed] [Google Scholar]
- 22.Pal T, Rocchio E, Garcia A, Rivers D, Vadaparampil S. Recruitment of black women for a study of inherited breast cancer using a cancer registry-based approach. Genetic testing and molecular biomarkers. 2011;15(1-2):69–77. doi: 10.1089/gtmb.2010.0098. doi:10.1089/gtmb.2010.0098. [DOI] [PubMed] [Google Scholar]
- 23.Anderson B, McLosky J, Wasilevich E, Lyon-Callo S, Duquette D, Copeland G. Barriers and facilitators for utilization of genetic counseling and risk assessment services in young female breast cancer survivors. Journal of cancer epidemiology. 2012;2012:298745. doi: 10.1155/2012/298745. doi:10.1155/2012/298745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.McCarthy AM, Bristol M, Fredricks T, Wilkins L, Roelfsema I, Liao K, Shea JA, Groeneveld P, Domchek SM, Armstrong K. Are physician recommendations for BRCA1/2 testing in patients with breast cancer appropriate? A population-based study. Cancer. 2013 doi: 10.1002/cncr.28268. doi:10.1002/cncr.28268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Shields AE, Burke W, Levy DE. Differential use of available genetic tests among primary care physicians in the United States: results of a national survey. Genet Med. 2008;10(6):404–414. doi: 10.1097/GIM.0b013e3181770184. doi:10.1097/GIM.0b013e3181770184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Matloff E, Caplan A. Direct to confusion: lessons learned from marketing BRCA testing. Am J Bioeth. 2008;8(6):5–8. doi: 10.1080/15265160802248179. [DOI] [PubMed] [Google Scholar]
- 27.Hull SC, Prasad K. Reading between the lines: direct-to-consumer advertising of genetic testing in the USA. Reproductive health matters. 2001;9(18):44–48. doi: 10.1016/s0968-8080(01)90089-8. [DOI] [PubMed] [Google Scholar]
- 28.Radford C, Prince A, Lewis K, Pal T. Factors Which Impact the Delivery of Genetic Risk Assessment Services Focused on Inherited Cancer Genomics: Expanding the Role and Reach of Certified Genetics Professionals. Journal of genetic counseling. 2013 doi: 10.1007/s10897-013-9668-1. doi:10.1007/s10897-013-9668-1. [DOI] [PubMed] [Google Scholar]
- 29.Bowen DJ, Battuello KM, Raats M. Marketing genetic tests: empowerment or snake oil? Health Educ Behav. 2005;32(5):676–685. doi: 10.1177/1090198105278825. [DOI] [PubMed] [Google Scholar]
- 30.Hogarth S, Javitt G, Melzer D. The current landscape for direct-to-consumer genetic testing: legal, ethical, and policy issues. Annu Rev Genomics Hum Genet. 2008;9:161–182. doi: 10.1146/annurev.genom.9.081307.164319. [DOI] [PubMed] [Google Scholar]
- 31.Stuckey A, Febbraro T, Laprise J, Wilbur JS, Lopes V, Robison K. Adherence Patterns to National Comprehensive Cancer Network Guidelines for Referral of Women With Breast Cancer to Genetics Professionals. American journal of clinical oncology. 2014 doi: 10.1097/COC.0000000000000073. doi:10.1097/COC.0000000000000073. [DOI] [PubMed] [Google Scholar]
- 32.Murff HJ, Greevy RA, Syngal S. The comprehensiveness of family cancer history assessments in primary care. Community Genet. 2007;10(3):174–180. doi: 10.1159/000101759. doi:10.1159/000101759. [DOI] [PubMed] [Google Scholar]
- 33.Butel-Simoes GI, Spigelman AD. Analysis of patient reports on the referral process to two NSW cancer genetic services. Familial cancer. 2014;13(3):333–343. doi: 10.1007/s10689-014-9710-y. doi:10.1007/s10689-014-9710-y. [DOI] [PubMed] [Google Scholar]
- 34.Bellcross CA, Leadbetter S, Alford SH, Peipins LA. Prevalence and healthcare actions of women in a large health system with a family history meeting the 2005 USPSTF recommendation for BRCA genetic counseling referral. Cancer epidemiology, biomarkers & prevention : a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive Oncology. 2013;22(4):728–735. doi: 10.1158/1055-9965.EPI-12-1280. doi:10.1158/1055-9965.EPI-12-1280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Thompson HS, Sussner K, Schwartz MD, Edwards T, Forman A, Jandorf L, Brown K, Bovbjerg DH, Valdimarsdottir HB. Receipt of genetic counseling recommendations among black women at high risk for BRCA mutations. Genetic testing and molecular biomarkers. 2012;16(11):1257–1262. doi: 10.1089/gtmb.2012.0114. doi:10.1089/gtmb.2012.0114. [DOI] [PubMed] [Google Scholar]
- 36.Wang G, Beattie MS, Ponce NA, Phillips KA. Eligibility criteria in private and public coverage policies for BRCA genetic testing and genetic counseling. Genet Med. 2011;13(12):1045–1050. doi: 10.1097/GIM.0b013e31822a8113. doi:10.1097/GIM.0b013e31822a8113. [DOI] [PMC free article] [PubMed] [Google Scholar]