Abstract
We used a spatial light modulator to project an optical micropattern of 473 nm light with a quartic intensity gradient on a single lung cancer cell. We observed that the intracellular amounts of reactive oxygen species (ROS) of the cancer cells were proportional to the intensity of the blue light, and the blue light intensity gradients could drive directional cell migration. This optically induced directional cell migration was inhibited by a ROS scavenger in the culture medium in a dose-dependent manner. In contrast, the ROS levels in fibroblasts were saturated by the blue light at low intensity and therefore the fibroblasts did not exhibit directional migration in the intensity gradient.
OCIS codes: (170.4520) Optical confinement and manipulation, (170.0170) Medical optics and biotechnology
1. Introduction
Metastasis is the major cause of mortality of most patients suffering from cancers. In order to reduce the possibility of metastasis, it is crucial to investigate the factors correlated with cell motility or invasiveness. Among the cellular mechanisms related to cancer metastasis, chemotaxis, directional cell migration induced by particular extracellular chemoattractants, could be the most well-studied one [1]. In addition to growth factors and other chemoattractants leading to chemotaxis, more and more physical stimulations that could influence the cell migration directions, such as durotaxis [2] or electrotaxis [3], have been intensively investigated in the past decade because of their potentials in therapeutic applications.
Light–cell interactions have been demonstrated to influence the migration of various types of cells. Ehrlicher et al. demonstrated that near-infrared light could guide the growth of neuronal cells more than ten years ago [4]. Biener et al. found that the polarization of light could apply torques on the actin filaments in cells and hence influenced the motility of neuroblastoma cells, which tended to align along the direction of polarization of the electric field [5]. Using a two-wavelength setup, Xiao et al. verified that focused laser light spots can be used to influence the growth and retraction of cellular lamellipodia [6]. Recently, spatial light modulators (SLMs) have been employed to facilitate a variety of cellular guidance. For example, 473 nm blue light patterns generated by a SLM had been used to conduct patterned cell proliferation [7] and guided migration [8] of adherent cells. Apparently, optical illumination is an effective way to control cell migration. However, the mechanisms of optical cell guidance could be different for different types of cells at different wavelengths of light. Therefore, detailed investigations about the mechanisms affecting cell migration under optical illumination are very desirable for further applications of optical cell guidance.
In addition to its abundant biological effects in plant cells, blue light has recently attracted much attention in biology of animal cells. For example, it has been shown that blue light (wavelength ~470 nm) could induce the production of intracellular reactive oxygen species (ROS) in retinal pigment epithelial cells [9]. However, it was also reported that blue light of wavelengths 453 nm and 480 nm showed low cellular toxicity in dermal fibroblasts [10]. It seems that the degrees of blue light-induced ROS production are cell-type dependent. In fact, ROS also play important roles in cell migration [11]. Although the exact mechanisms for ROS to influence cell migration are not clear now, intracellular productions of ROS enhance cell migration in various types of cells. It is therefore interesting to investigate how the blue light-produced ROS are used to induce directional migration of specific types of cells, especially cancer cells.
In the present work, we employed a SLM to generate a quartic optical intensity gradient near an individual lung cancer cell. Because the intracellular ROS levels of the lung cancer cell were proportional to the blue light intensity, the intensity gradient could lead to an inhomogeneous distribution of ROS in the illuminated cell. As a result, the cell exhibited directional migration away from the gradient of optical intensity. In order to further verify that this optically induced directional migration is induced by the intracellular ROS production, we used a ROS scavenger to adjust the amount of ROS. The migration distance away from the intensity gradient showed an inverse dose-dependent behavior to the concentration of the ROS scavenger. In comparison, we also tested the intensity gradient of blue light on the migration of lung fibroblast. We found that the intracellular ROS levels in fibroblasts were saturated at low intensity. Therefore the fibroblasts did not exhibit directional migration in the optical intensity gradient.
2. Method and material
The setup used in the present work is the same as that we described in our previous publications [7, 8]. In brief, the light source was a 473 nm diode-pumped solid state laser. Optical patterns produced by a liquid-crystal SLM (HEO 6001-SC-II, Holoeye, Berlin, Germany) were projected onto the focal plane of a 10 × , 0.30 numerical aperture (NA) objective or a 20 × , 0.45 NA objective. In order to reduce the potential cellular damage caused by the blue light, we adopted intermittent illumination during the experiments. The illumination period was five minutes, in which the laser was turned on for three minutes, and the cells had a two-minute recovery time before the next illumination. In this way most of the tested cells kept alive for more than eight hours of the quartic gradient-pattern illuminaiton without blebbing or being detached from the bottom of the culture dish. We took one image of the cell at the end of every five minutes. For measuring the cell migration speed, the boundary of the cell in the image was marked by hand, and then the geometric center of this cell was determined by using ImageJ. Then we plotted the cell migration paths with Chemotaxis and Migration Tool Ver. 2.0 (ibidi GmbH, Martinsried, Germany). The system was controlled by a LabVIEWTM computer program and the temperature surrounded the culture dish was maintained at 37 ± 0.5°C. For quantifying the cell motility, we conducted the experiments for 500 minutes.
We cultured the human lung carcinoma cells A549 in Ham’s F12K (Kaighn’s) medium supplemented with 10% fetal bovine serum (FBS) and 1% antibiotic pen-strep-ampho (PSA) in an incubator at 37°C. Before the cells were moved into the temperature-controlled chamber on the microscope, the F12K medium was replaced with the Leibovitz L-15 medium for a CO2-independent culture environment. For the comparison study, we also cultured the human lung fibroblast MRC-5 in Dulbecco’s Modified Eagle’s Medium supplemented with 10% FBS and 1% antibiotic PSA. The following experimental procedures on the MRC-5 fibroblasts were the same as those on the A549 cells.
In order to ascertain the involvement of ROS in the optical intensity-gradient induced cell migration, we used Trolox (Sigma-Aldrich, St. Louis, MO, USA) to scavenge the ROS in the culture medium. This scavenger was dissolved in the L-15 medium with the desired concentration. For the treatment experiments we replaced the original culture medium with the medium with Trolox.
3. Results and discussion
3.1 Intracellular ROS levels of the cancer cells increased with blue light intensity
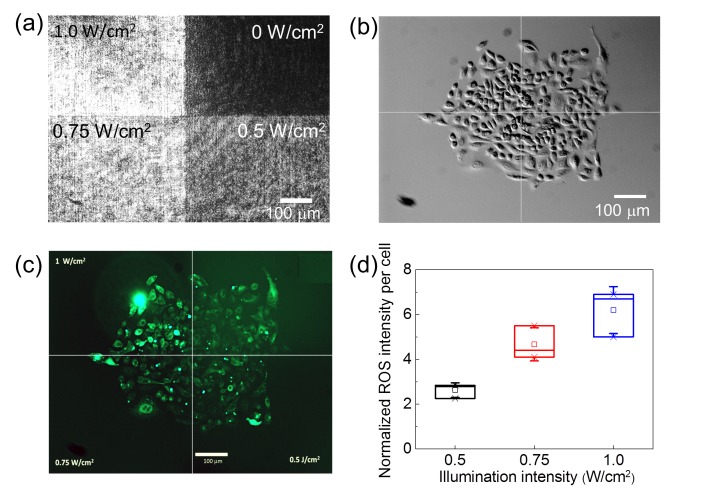
The intracellular ROS levels were indicated by a fluorescent ROS indicator CM-H2DCFDA (Molecular Probes, Life Technologies Inc., Taiwan). This ROS indicator was excited with a mercury lamp through a filter set of excitation wavelengths centered at 470 nm and emission wavelengths centered at 525 nm. During the observation of the ROS levels the 473 nm laser light was blocked. Figure 1(a) shows the optical pattern for quantifying the intracellular levels of ROS. We calibrated the intensity levels on the bottom of a culture dish by using a homogeneous circular pattern with a single value of intensity. Then we projected the pattern in Fig. 1(a) onto a group of A549 cells. The intensity of the ROS indicator was measured after one hour of illumination by the blue light. Under the homogenous illumination of 1.0 W/cm2 intensity, we found that some of the cells started shrinking or became rounded after eight hours. We thus set the illumination time as short as one hour to ensure that the cellular damage was not serious, while the production of ROS reached sufficiently high levels for comparison. To obtain the signal of the ROS indicator per cell, we measured the total fluorescence intensity of the cells illuminated by one intensity level, subtracted the background, and then divided the total fluorescence intensity by the cell number. Figure 1(d) shows the normalized intensity of the ROS indicator per cell measured in three individual experiments. The amounts of the ROS indicator per cell are nearly proportional to the illumination intensity. Therefore we postulated that an intensity gradient of blue light could produce an intracellular ROS gradient.
Fig. 1.
(a) The optical pattern for quantifying the intracellular levels of ROS. (b) DIC image of a group of A549 cells under the illumination of the pattern in (a). (c) Fluorescence image of a ROS indicator CM-H2DCFDA in the cells in (b). (d) Intensity of the ROS indicator per cell measured in three individual experiments. The data were normalized to that obtained from cells without the blue-light illumination.
3.2 Directional cell migration induced by an intensity gradient of blue light
We tried to use an intensity gradient to induce directional cell migration. Because the diameters of individual A549 cells were ~50 μm, we set the full width of the optical pattern tobe 50 μm for a potentially asymmetric production of intracellular ROS. We compared the effects of linear, quadratic, and quartic gradient profiles of the same width on the directional migration of cancer cells. The linear gradient pattern did not induce obvious cell migration. Instead, most of the tested cells stayed at their original positions and became rounded after eight hours of blue light illumination. The quadratic gradient pattern sometimes introduced directional cell migration; and the cells kept alive longer than those under the illumination of the linear gradient pattern. In the following experiments, we therefore adopted the quartic gradient pattern (as shown in Fig. 2) that could cause obvious directional cell migration without observable cellular damage.
Fig. 2.
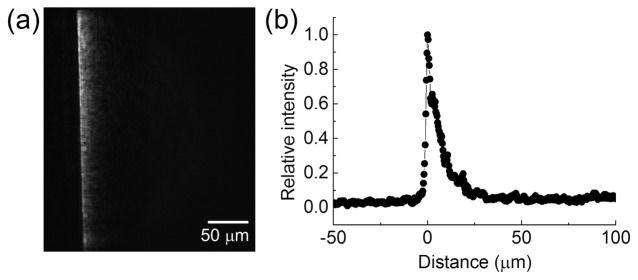
The pattern of a quartic intensity gradient used for the experiments of directional cell migration.
Figure 3 shows the migration of the A549 cell under the quartic intensity gradient. In Figs. 3(a) and 3(b), the cell exhibited directional migration reverse to the intensity gradient. The motion of the nucleus is more evident than that of the whole cell (also see the Supplementary Visualization 1 (1.1MB, MOV) ). We repeated the observation on 7 cells and show their migration trajectories of the geometric centers in Fig. 3(c). In 500 minutes of the observation period, the cells moved in the region with sufficiently bright illumination by the gradient pattern [~25 μm wide as shown in Fig. 2(b)]. Outside the illumination the cells showed relatively low motility or even ceased to move. Therefore we confirmed that this directional cell migration was driven by the optical intensity gradient.
Fig. 3.
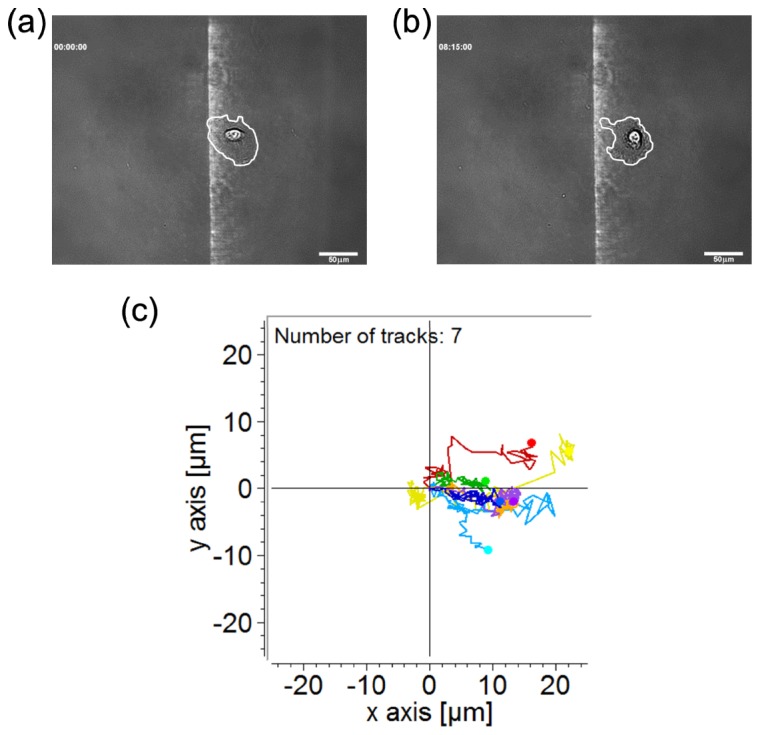
(a) Image of an A549 cell illuminated by blue light of a quartic intensity gradient at the starting point. The white line marks the cell boundary. (b) The same cell in (a) after 495 minutes of illumination. Scale bar, 50 μm. (c) Migration trajectories of 7 cells under the quartic optical intensity gradient. The recording duration is 500 minutes.
3.3 Effects of a ROS scavenger on the directional cell migration induced by the intensity gradient
An asymmetric physical or chemical migratory cue in the cell or the neighboring microenvironment is a necessary condition for inducing directional cell migration. As we show in Fig. 1(d), the intracellular amounts of ROS were nearly proportional to the illumination intensity. Therefore an intensity gradient could lead to an asymmetric distribution of intracellular ROS. In order to ascertain the implication of ROS in the intensity gradient-induced directional migration, we used Trolox as a scavenger of the ROS to intervene in the optical stimulation. Trolox is a water-soluble form of vitamin E, which has been known to reduce the oxidative stress-induced apoptosis [12]. Recently, Sung et al. verified that Trolox could reduce the phorbol 12-myristate 13-acetate (PMA)-induced migration of A549 and HeLa cells, and the PMA-induced ROS generation was suppressed by Trolox [13]. Therefore, if the intracellular ROS production is the mechanism of the intensity gradient-induced directional cell migration, Trolox could also play a suppressing role.
For the A549 cell, we found a Trolox concentration of 300 μM caused cell rounding anddetachment within one hour. Therefore we used the Trolox concentration as high as 150 μM in the following experiments. We compare the blue light-induced intracellular ROS levels of A549 cells without and with the treatment of Trolox in Fig. 4. The left-half regions of Figs. 4(b) and 4(c) show obvious cellular damage as well as ROS production caused by the blue light illumination without Trolox. The morphology of cells in the left-half region of Fig. 4(d) did not show significant blebbing or being rounded in comparison with the cells in the right-half region of Fig. 4(b), suggesting that the simultaneous blue light and Trolox treatments did not result in serious cellular damage. Under the illumination of 1.0 W/cm2 blue light for one hour, the treatment of 150 μM Trolox suppressed the intracellular production of ROS in most of the illuminated cells, as shown in Fig. 4(e). Therefore we confirmed that Trolox can reduce the blue-light induced ROS production.
Fig. 4.
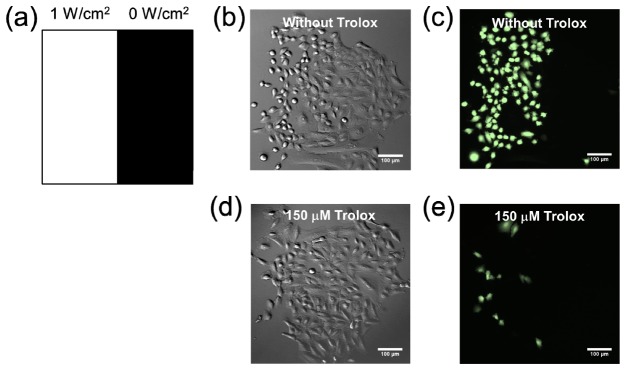
(a) The optical pattern for testing the effect of Trolox on the production of ROS induced by blue light. (b) DIC image of the A549 cells under the pattern illumination for one hour. (c) Intracellular ROS levels indicated by a ROS indicator CM-H2DCFDA of the cells in (b). (d) DIC image of the A549 cells under the pattern illumination and the treatment of 150 μM Trolox. (e) Intracellular ROS levels of the cells in (d). Scale bar, 100 μm.
Figure 5 shows the migration of the A549 cell under the treatments of the blue light intensity gradient and 150 μM Trolox. In Figs. 5(a) and 5(b), the cell did not move away from the intensity gradient within 495 minutes of illumination (also see the Supplementary Visualization 2 (1.1MB, MOV) ). We repeated the observation on 7 cells and show the migration trajectories of cellular geometric centers in Fig. 5(c). In a 500-minute observation period, the cells did not move for longer than 15 μm along the horizontal axis (X-axis).
Fig. 5.
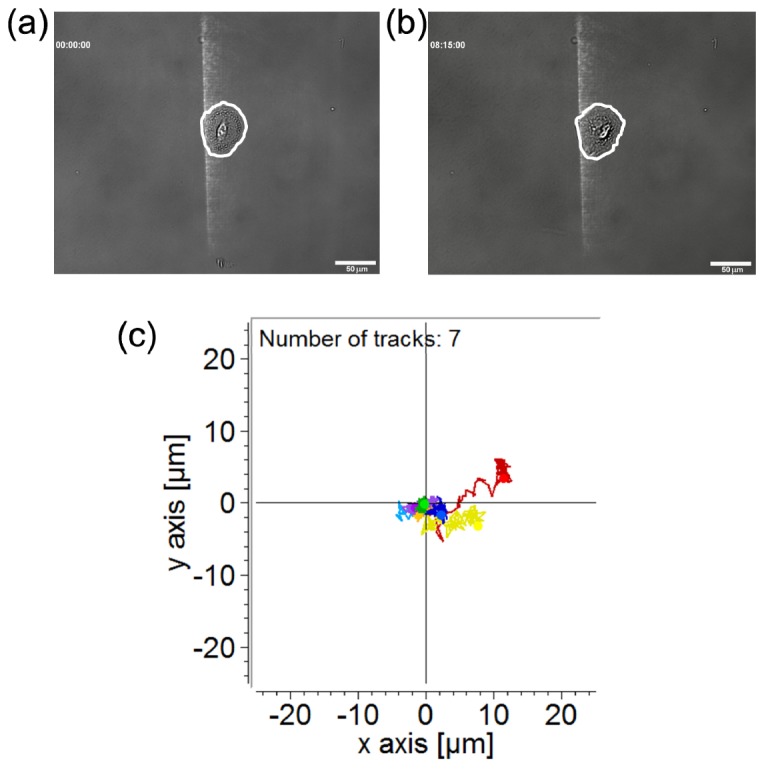
(a) Image of an A549 cell illuminated by blue light of a quartic intensity gradient with 150 μM Trolox in the culture medium at the starting point. The white line marks the cell boundary. (b) The same cell in (a) after 495 minutes of illumination. Scale bar, 50 μm. (c) Migration trajectories of 7 cells under the quartic optical intensity gradient. The recording duration is 500 minutes.
As a quantification of the degrees of directional cell migration, we measured the displacements of the geometric center of each cell along the X-axis under various treatments. As shown in Fig. 6, the control group exhibited no directional migration along the horizontal axis. The optical intensity gradient raised obvious directional migration; while the treatment of Trolox reduced this effect in a dose-dependent manner. A Student’s t-test was conducted tocompare the displacements along the X-axis between the cells treated with the intensity gradient only and those with the intensity gradient and 150 μM Trolox. A p-value smaller than 0.005 confirmed that Trolox indeed suppressed the directional cell migration caused by the blue light intensity gradient.
Fig. 6.
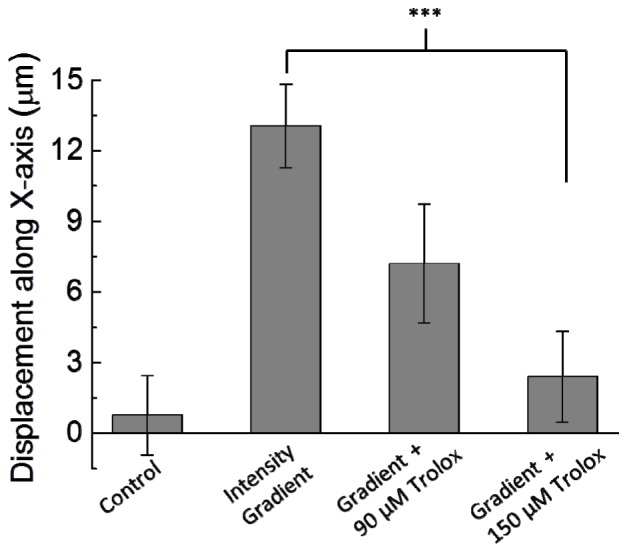
Cell displacements along the horizontal direction (X-axis) under the treatments of the optical intensity gradient and Trolox of 90 μM and 150 μM. The horizontal displacements are shortened by the Trolox in a dose-dependent manner. In each condition, migration trajectories of 7 cells in 500 minutes were recorded. Error bar, standard error of the mean. ***, p < 0.005.
3.4 Intracellular ROS levels of fibroblasts and blue light intensity
Because the amounts of intracellular ROS induced by blue light are cell-type dependent, we also used MRC-5 fibroblast as a comparison group for the blue light-induced ROS production. The experiments were conducted using the four-quadrant pattern as shown in Fig. 1(a). The data in Fig. 7(a) show that the ROS levels inside the fibroblasts were saturated at 0.75 W/cm2, and the increase of ROS amounts was much smaller than that observed in the A549 lung cancer cells at the same intensity levels [Fig. 1(d)]. This trend is similar to that reported inRef. [10], in which the blue light of 453 nm and 480 nm wavelengths caused relatively low intracellular ROS in dermal fibroblasts. In the present study, we also tracked the migration of MRC-5 fibroblasts under the stimulation of the blue light intensity gradient. The results in Fig. 7(b) showed no obvious directional migration under the irradiation of the intensity gradient. Comparing the results of A549 cancer cells and those of MRC-5 fibroblasts, we propose that the intracellular ROS production by blue light is closely related to the intensity gradient-driven directional cell migration.
Fig. 7.
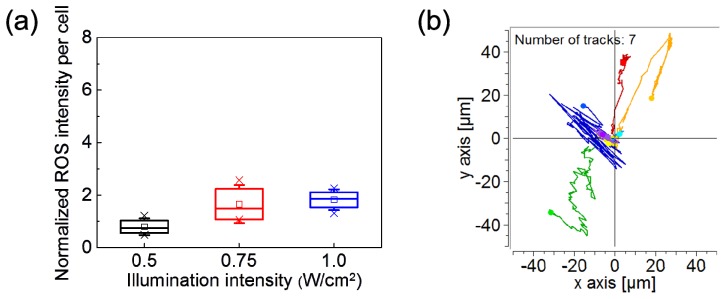
(a) Intensity of the ROS indicator per MRC-5 cell measured in three individual experiments. The data were normalized to that obtained from the cells without blue-light illumination. (b) Migration trajectories of 7 MRC-5 cells under the quartic optical intensity gradient. The recording duration is 500 minutes.
4. Conclusion
In the present work we demonstrated the directional migration of lung cancer cell A549 induced by an intensity gradient of 473 nm blue light. With the optical patterns of the same width, a quartic intensity gradient caused directional cell migration more effectively than the linear and quadratic gradient patterns. This optically induced directional cell migration could be suppressed by a ROS scavenger Trolox dissolved in the culture medium. Therefore, we conjecture that the blue light intensity gradient-driven cell migration is related to the intracellular ROS production. In contrast, the ROS levels in MRC-5 fibroblasts were not increased by the blue light illumination as much as those in the A549 cells. Therefore the intensity gradient-driven directional migration of the MRC-5 cells was not evident. However, the exact migration-relevant proteins that are activated by the blue light-generated ROS in the cancer cells still remain to be identified.
Because of the simplicity on the control and delivery of light, the phototactic behaviors demonstrated in the present work could lead to real applications, such as restricted migration of tumor cells before or during surgical excision. The light-induced ROS production could also be used as a novel way to force the migration of specific cells, such as neurons, in animal bodies.
Acknowledgments
This work was supported by the Ministry of Science and Technology, Taiwan (contract No. MOST 103-2112-M-001-019-MY3) and the program of Fostering the Next Generation Interdisciplinary Scientists (contract No. MOST 103-2633-H-010-001).
References and links
- 1.Roussos E. T., Condeelis J. S., Patsialou A., “Chemotaxis in cancer,” Nat. Rev. Cancer 11(8), 573–587 (2011). 10.1038/nrc3078 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Discher D. E., Janmey P., Wang Y.-L., “Tissue cells feel and respond to the stiffness of their substrate,” Science 310(5751), 1139–1143 (2005). 10.1126/science.1116995 [DOI] [PubMed] [Google Scholar]
- 3.Cortese B., Palamà I. E., D’Amone S., Gigli G., “Influence of electrotaxis on cell behaviour,” Integr Biol (Camb) 6(9), 817–830 (2014). 10.1039/C4IB00142G [DOI] [PubMed] [Google Scholar]
- 4.Ehrlicher A., Betz T., Stuhrmann B., Koch D., Milner V., Raizen M. G., Kas J., “Guiding neuronal growth with light,” Proc. Natl. Acad. Sci. U.S.A. 99(25), 16024–16028 (2002). 10.1073/pnas.252631899 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Biener G., Vrotsos E., Sugaya K., Dogariu A., “Optical torques guiding cell motility,” Opt. Express 17(12), 9724–9732 (2009). 10.1364/OE.17.009724 [DOI] [PubMed] [Google Scholar]
- 6.Xiao J.-L., Hsu T.-H., Hsu P.-Y., Yang W.-J., Kuo P.-L., Lee C.-H., “Motion of cancer-cell lamellipodia perturbed by laser light of two wavelengths,” Appl. Phys. Lett. 97(20), 203702 (2010). 10.1063/1.3517448 [DOI] [Google Scholar]
- 7.Xiao J.-L., Pan H.-J., Lee C.-H., “Optically micropatterned culture of adherent cells,” J. Biomed. Opt. 17(7), 075004 (2012). 10.1117/1.JBO.17.7.075004 [DOI] [PubMed] [Google Scholar]
- 8.Xiao J.-L., Lu D.-H., Lee C.-H., “Guiding the migration of adherent cells by using optical micropatterns,” Appl. Phys. Lett. 102(12), 123703 (2013). 10.1063/1.4798494 [DOI] [Google Scholar]
- 9.Nakanishi-Ueda T., Majima H. J., Watanabe K., Ueda T., Indo H. P., Suenaga S., Hisamitsu T., Ozawa T., Yasuhara H., Koide R., “Blue LED light exposure develops intracellular reactive oxygen species, lipid peroxidation, and subsequent cellular injuries in cultured bovine retinal pigment epithelial cells,” Free Radic. Res. 47(10), 774–780 (2013). 10.3109/10715762.2013.829570 [DOI] [PubMed] [Google Scholar]
- 10.Opländer C., Hidding S., Werners F. B., Born M., Pallua N., Suschek C. V., “Effects of blue light irradiation on human dermal fibroblasts,” J. Photochem. Photobiol. B 103(2), 118–125 (2011). 10.1016/j.jphotobiol.2011.02.018 [DOI] [PubMed] [Google Scholar]
- 11.Hurd T. R., DeGennaro M., Lehmann R., “Redox regulation of cell migration and adhesion,” Trends Cell Biol. 22(2), 107–115 (2012). 10.1016/j.tcb.2011.11.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Forrest V. J., Kang Y. H., McClain D. E., Robinson D. H., Ramakrishnan N., “Oxidative stress-induced apoptosis prevented by Trolox,” Free Radic. Biol. Med. 16(6), 675–684 (1994). 10.1016/0891-5849(94)90182-1 [DOI] [PubMed] [Google Scholar]
- 13.Sung H. J., Kim Y., Kang H., Sull J. W., Kim Y. S., Jang S.-W., Ko J., “Inhibitory effect of Trolox on the migration and invasion of human lung and cervical cancer cells,” Int. J. Mol. Med. 29(2), 245–251 (2012). [DOI] [PubMed] [Google Scholar]