Abstract
Purpose
Nonsyndromic retinitis pigmentosa (RP) is the most common inherited retinal degeneration, and prevalence of the disease has been reported in populations of American and European origin with a relatively low consanguinity rate. Our aim was to determine the prevalence of nonsyndromic RP in the Jerusalem region, which has a population of about 1 million individuals with a high rate of consanguinity.
Methods
The patients’ clinical data included eye exam findings (visual acuity, anterior segment, and funduscopy) as well as electroretinographic (ERG) testing results under scotopic and photopic conditions. Mutation analysis on a subgroup of patients was performed mainly with candidate gene analysis and homozygosity mapping.
Results
We evaluated the medical records of patients with degenerative retinal diseases residing in the Jerusalem region who were examined over the past 20 years in a large tertiary medical center. A total of 453 individuals affected with nonsyndromic RP were diagnosed at our center, according to funduscopic findings and ERG testing. Based on the estimated population size of 945,000 individuals who reside in the vicinity of Jerusalem, the prevalence of nonsyndromic RP in this region is 1:2,086. The prevalence of RP was higher among Arab Muslims (1:1,798) compared to Jews (1:2,230), mainly due to consanguineous marriages that are more common in the Arab Muslim population. To identify the genetic causes of RP in our cohort, we recruited 383 patients from 183 different families for genetic analysis: 70 with autosomal recessive (AR) inheritance, 15 with autosomal dominant, 86 isolate cases, and 12 with an X-linked inheritance pattern. In 64 (35%) of the families, we identified the genetic cause of the disease, and we revised the inheritance pattern of 20 isolate cases to the AR pattern; 49% of the families in our cohort had AR inheritance. Interestingly, in 42 (66%) of the genetically identified families, the cause of disease was a founder mutation.
Conclusions
Previous studies showed an approximate prevalence of 1:5,260 on average for nonsyndromic RP in American and European populations. We show that the prevalence in the vicinity of Jerusalem is two-and-a-half times higher due to a high rate of consanguinity and highly prevalent founder mutations within the historically semi-isolated subpopulations we serve.
Introduction
Hereditary retinal diseases (HRDs) are heterogeneous disorders that cause incurable visual loss mainly due to the dysfunction or degeneration of rod and cone photoreceptor cells in the retina. Although a wide and sometimes overlapping spectrum of phenotypes exists, HRDs that mainly affect photoreceptors and/or RPE cells can be roughly divided into three main categories based on the predominant cell type affected and the pattern of disease progression: rod-dominated diseases in which pronounced, widespread rod photoreceptor loss precedes cone involvement (retinitis pigmentosa [RP], the most common form of inherited retinal degeneration, classically follows this pattern), cone-dominated diseases with more pronounced cone photoreceptor loss, and regional degeneration in which a specific retinal region (e.g., the macula) is affected. The total number of genes responsible for these heterogeneous diseases is still unknown but is estimated to be more than 200. Numerous manuscripts and many excellent reviews have been published on the genetic and clinical aspects of inherited retinal diseases [1-7].
The Israeli population contains several different ethnic groups including Jews of various origins (75% of the population), Arab Muslims (17%), Bedouins (2.8%), Arab Christians (2%), and Druze (1.6%; data are based on the Israeli Central Bureau of Statistics as of 2010). Consanguinity has been reported to be high in various ethnic groups in Israel for many generations, stemming from historic, ethnic, religious, and cultural causes [8,9]. Genetic analysis of Israeli families with various inherited diseases was and is actively pursued, and has led to the identification of the disease-causing genes (including novel ones) in many cases (data are available online in the Israeli National Genetic Database).
The Jewish population originated in the Middle East but during the course of history (spanning thousands of years) was divided into isolated ethnic groups separated geographically throughout practically the entire world. Within these semi-isolated ethnic groups, the need and desire to maintain religion and culture led to relatively high levels of intracommunity marriages. Over the past 150 years, and more prominently following the establishment of the state of Israel in 1948, many returned to their homeland, and in recent history, a lower rate of consanguinity and an elevated rate of interethnic marriages are already evident [8]. The largest Jewish ethnic group is the Ashkenazi Jewish population, which comprises about 55% of Israeli Jews, followed by North African Jews (about 25%), and Eastern Jews (from Iran, Iraq, etc., about 20%). Genetic analyses of Ashkenazi Jewish families aided in the identification and characterization of many genes in which founder mutations cause common inherited diseases in this ethnic group (see [10] for review), including Tay-Sachs [11], Gaucher [12], and Usher syndrome [13,14] and several genes that cause recessive RP [15-17]. An intense scientific effort was directed during the past 15 years using different genetic tools and markers to genetically analyze the different Israeli subpopulations. These studies led to better classification of the different ethnic groups and shed light on the common origin of most of these populations [18-23].
Individuals of Arab Muslim origin tend to live in villages that were founded a few generations ago by a small number of individuals. In accordance with the customs of the Arab population throughout the Middle East [24,25], consanguineous marriages are common among Israeli Arabs, with a preference for first-cousin marriages [25,26]. This is one of the reasons for the high rate of occurrence of autosomal recessive diseases in this population, with more than 200 diseases described thus far [27].
The Bedouin population is unique in its structure, which is tribal, and exhibits an extremely high level of consanguinity. Studying the genetics of Bedouin Israeli families was a key factor in the discovery of many disease-causing genes, including those causing Bardet-Biedl syndrome, such as BBS1 (GeneID 582,OMIM: 209901), [28], BBS2 (GeneID 583, OMIM: 606151) [29], ARL6 (GeneID 84100, MIM: 608845) [30], and TRIM32 (GeneID 22954, OMIM: 602290) [31].
To date, the prevalence of inherited retinal diseases has been estimated in studies that were performed mainly in North America or Europe. Nonsyndromic RP [MIM # 268000] is the most common inherited retinal degeneration with an estimated prevalence of 1:5,193 in Maine [32], 1:6,134 in Denmark [33], and 1:4,440 in Norway [34]. Similarly, the total prevalence of RP (including syndromic as well as nonsyndromic forms) was estimated at 1:4,756 in Maine [32], 1:4,869 in Birmingham [35], 1:3,937 in Denmark [33], and 1:3,784 in China [36].
No accurate data are currently available on the prevalence of RP in the Israeli and Palestinian population. Merin and Auerbach [37] estimated that the prevalence of RP in Israel, and autosomal recessive RP in particular, is higher than that reported in other countries because of the high rate of consanguineous marriages. In a survey that was performed to roughly estimate the prevalence of RP in the Israeli Jewish population, 341,175 files of individuals (ages 17–20 years) were evaluated [38]. In this survey, the best-corrected visual acuity (VA) was determined, and those who had acuity of less than 20/25 were further examined by an ophthalmologist. The prevalence in this study was estimated at 1:4,610, which was predicted to be an underestimation since many patients with RP in this age group have relatively preserved visual acuities and would not have been identified with the VA screening parameter used. The purpose of the current study was to determine the prevalence of nonsyndromic RP in the Jerusalem region and to compare prevalence data of the two major subpopulations residing in this region, namely, Jews and Arab Muslims.
Methods
Subjects
Ethical approval for this study was obtained from the Hadassah Medical Center Institutional Review Board (IRB). All participants in the genetic research signed an informed consent that adhered to the tenets of the Declaration of Helsinki. Subsequently, venous blood samples were collected and processed immediately. Genomic DNA was extracted using FlexiGene DNA kit (Qiagen, Venlo, The Netherlands).
Clinical evaluation
The ocular diagnosis was determined mainly using a full ophthalmologic exam and full-field electroretinography (FFERG). In many cases, additional supportive functional and structural data was available, including combinations of electro-oculography (EOG), color vision testing using the Panel D-15 test, Humphrey and/or Goldmann perimetry, optical coherence tomography (OCT), color, infrared and fundus autofluorescence (FAF) imaging, and fluorescein angiography (FA).
Genetic analyses
Various genetic analyses were used to identify the cause of disease, including homozygosity mapping followed by Sanger sequencing of candidate genes, mutation analysis of founder mutations, and whole exome sequencing as detailed elsewhere [16,39,40].
Results
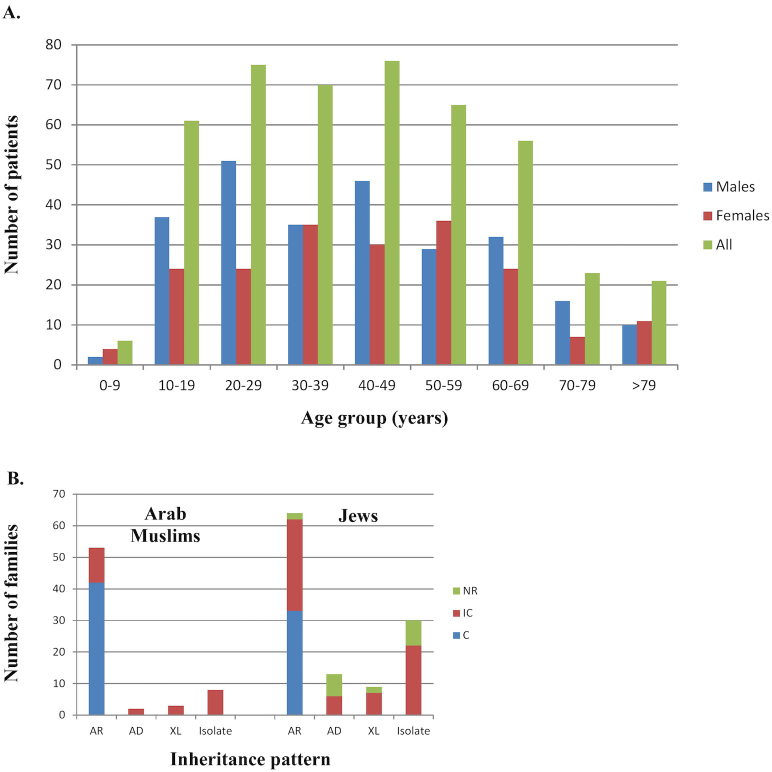
Aiming to estimate the prevalence of nonsyndromic RP in the vicinity of Jerusalem, we created a database of patients who were examined with ERG at our unit, were clinically examined at our ophthalmology clinic, or participated in the ophthalmic genetic study. For the prevalence calculation, we included only individuals who were diagnosed with nonsyndromic RP and reside in the vicinity of Jerusalem. We identified 453 patients with RP in a region that is estimated to be populated by 945,000 individuals (data collected from the Israeli Bureau of Statistics), resulting in an estimated prevalence of 1:2,086 individuals. There was a higher number of affected men (258, 57%), probably due to the contribution of the X-linked (XL) RP cases. We then estimated the prevalence of nonsyndromic RP in the two major subpopulations residing in the Jerusalem area: Jews (about 65% of the population) and Arab Muslims (35%). The prevalence of RP was higher among Arab Muslims (1:1,798) compared to Jews (1:2,230). The distribution of each disease by age group is shown in Figure 1A. As expected, RP is mainly prevalent from age 10 and on, as it is often not diagnosed earlier.
Figure 1.
Distribution of patients with RP by age, origin, consanguinity, and inheritance type. A: The distribution of the studied set of patients by age. Each bar represents the number of patients within each specific age group, divided into 10-year intervals. Data are presented for all patients (green), men only (blue), and women only (red). B: The distribution of the cohort of recruited families by origin, inheritance type, and parental relatedness. For each origin (Arab Muslims and Jews), families are presented by inheritance pattern and relatedness. AR=autosomal recessive and consanguineous isolate cases; AD=autosomal dominant; XL=X-linked; NR=parents are not related; IC=intracommunity marriages; C=consanguineous marriages.
To identify the genetic causes of RP in our cohort, we recruited for genetic analysis 383 patients from 183 different families: Based on the family tree structure, 70 families showed an autosomal recessive (AR) inheritance pattern, 15 were suggestive of autosomal dominant (AD) inheritance, 32 cases were isolate with consanguinity (indicative of AR inheritance), 54 were non-consanguineous isolate cases, and 12 families showed an X-linked inheritance pattern (Table 1). A distribution analysis by mode of inheritance and parental relatedness (Figure 1B) shows an extremely high contribution of consanguinity (blue bar) and intracommunity (brown bar) marriages to AR inheritance in Arab Muslims and Jews. This is likely to be the major reason for the relatively high proportion of families with AR in our cohort and the relatively high disease prevalence.
Table 1. Mode of inheritance patterns in the studied set of patients.
| Analyzed cohort | No. of recruited families (No. of patients)** | AR | Isolate cases, from consanguineous families | AD | XL | Isolate cases |
|---|---|---|---|---|---|---|
| Based on family history* |
183 (383) |
70 (38%) |
32 (17%) |
15 (8%) |
12 (7%) |
54 (30%) |
| Updated following genetic evaluation* |
183 (383) |
90 (49%) |
26 (14%) |
15 (8%) |
12 (7%) |
40 (22%) |
| Arab Muslims |
66 (151) |
36 (55%) |
17 (26%) |
2 (3%) |
3 (4%) |
8 (12%) |
| Jews | 116 (227) | 54 (46%) | 10 (9%) | 13 (11%) | 8 (7%) | 31 (27%) |
*The inheritance pattern in each family was initially determined by the family history (first row- “Based on family history”). Following the genetic diagnosis (in 35% of cases), the inheritance pattern was revised based on the identified gene and mutation (second row- “Updated following genetic evaluation“), resulting in more accurate values. **- The total number of patients (383) includes mainly patients of Arab-Muslim origin (151) and Jewish origin (227), as well as 5 patients (who belong to the same family) of Bedouin origin.
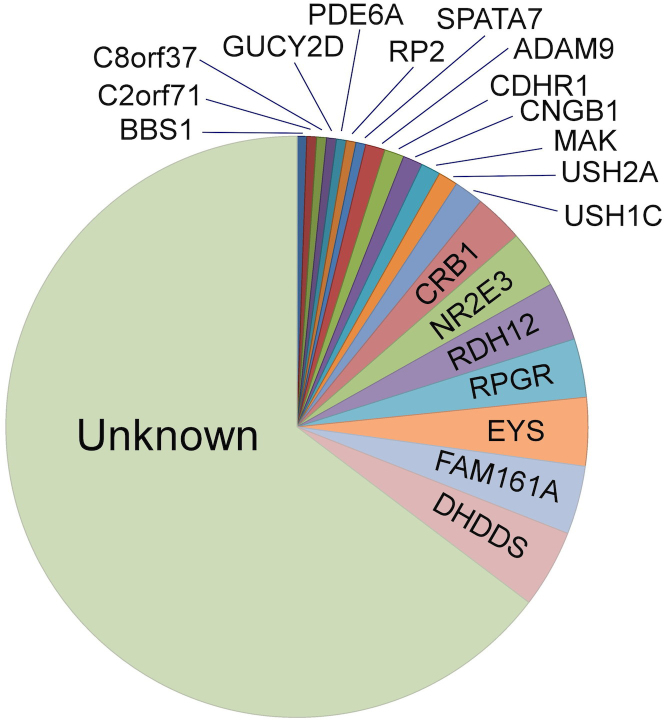
In 64 (35%) of the families, we identified the genetic cause of disease and thus revised the inheritance pattern of 14 nonconsanguineous and six consanguineous isolate cases to an AR pattern; at least 49% of the families in our cohort had AR inheritance (Table 1). Moreover, since almost all consanguineous isolate cases are expected to be due to AR genes, our cohort is likely to contain at least 63% families with AR inheritance pattern. This high level of AR cases is more prominent in the Arab Muslim cohort, which has a combined AR inheritance of at least 81% (Table 1, clear AR inheritance in 55% of cases and consanguineous isolate in 26% of cases). Interestingly, in 42 (66%) of the genetically identified families, the cause of disease was a founder mutation within the studied population, identified in the following genes: ADAM9 (GeneID 8754, OMIM: 602713), CNGB1 (GeneID 1258, OMIM: 600724), CRB1 (GeneID 23418, OMIM: 604210), DHDDS (GeneID 79947, OMIM: 608172), EYS (GeneID 346007, OMIM: 612424), FAM161A (GeneID 84140, OMIM: 613596), MAK (GeneID 4117, OMIM: 154235), NR2E3 (GeneID 10002, OMIM: 604485), RDH12 (GeneID 145226, OMIM: 608830), RPGR (GeneID 6103, OMIM: 312610), and USH1C (GeneID 10083, OMIM: 605242; Table 2 and Figure 2). The most common disease-causing allele in the vicinity of Jerusalem is the p.K42E missense mutation in DHDDS found in this study in 13.8% of the identified alleles (Table 2), followed by the c.1355_6delCA frameshift mutation in the FAM161A gene (10.3% of identified alleles). Similarly, the most common RP-causing genes were DHDDS (13.8% of identified alleles), FAM161A (12%), and EYS (11.9%).
Table 2. Disease-causing mutations identified in RP patients from the vicinity of Jerusalem.
| Gene Name | Mutation name | # of Families (# of alleles) | % of the total number of alleles | Origin |
|---|---|---|---|---|
|
ADAM9 |
c.1087T>A (p.C363S) |
2 (4) |
3.4% |
Iraqi Jewish |
|
BBS1 |
c.479G>A (p.R160Q) |
1 (2) |
1.7% |
Arab-Muslim |
|
CDHR1 |
c.1381C>T (p.Gln461*)
c.2087_2090del4 (p.D696Afs*3) |
1 (2)
1 (2) |
1.7%
1.7% |
Arab-Muslim
Arab-Muslim |
|
CNGB1 |
c.2284C>T (p.R762C) |
2 (4) |
3.4% |
Buchara Jewish |
|
CRB1 |
c.1148G>A (p.C383Y)
c.2498G>A (p.G833D)
c.3306G>A (p.G1103R) |
1 (2)
2 (4)
2 (4) |
1.7%
3.4%
3.4% |
Kurdish Jewish
Iraqi Jewish
Arab-Muslim |
|
c2orf71 |
c.2950C>T (p.R984*)
c.3289C>T (p.Q1097*) |
1 (1)
1 (1) |
0.8%
0.8% |
Turkey Jewish
Iranian Jewish |
|
C8orf37 |
c.529C>T (p.R177W) |
1 (2) |
1.7% |
Arab-Muslim |
|
DHDDS |
c.124A>G (p.K42E) |
8 (16) |
13.8% |
Ashkenazi Jewish |
|
EYS |
c.403delA,c.406G>T,c.del410_424 (p.T135Lfs*25)
c.1211_1212insA (p.N404Kfs*2)
c.8218_8219delCA (p.H2740Yfs*27)
c.9286_95del10 (p.V3096Lfs*28) |
5 (9)
1 (2)
1 (1)
1 (2) |
7.7%
1.7%
0.8%
1.7% |
North-African Jewish
North-African Jewish
Iraqi Jewish
Ashkenazi Jewish |
|
FAM161A |
c.1355_6delCA (p.T452Sfs*3)
c.1567C>T (p.R523*) |
6 (12)
1 (2) |
10.3%
1.7% |
North-African Jewish
Syrian Jewish |
|
MAK |
c.1296_7ins353 (Alu insertion) |
2 (4) |
3.4% |
Ashkenazi Jewish |
|
NR2E3 |
c.119–2A>C (IVS1–2A>C)
c.932G>A (p.R311Q)
c.747+1G>C (IVS5+1G>C)
c.194–202del9bp (p.N65-C67del) |
2 (2)
2 (2)
1 (1)
1 (1) |
1.7%
1.7%
0.8%
0.8% |
Arab-Muslim
Ashkenazi Jewish
Ashkenazi Jewish
Ashkenazi Jewish |
|
PDE6A |
c.1960C>T (p.Q654*) |
1 (2) |
1.7% |
Arab-Muslim |
|
RDH12 |
c.295C>A (p.L99I)
c.377C>T (p.A126V)
c.658G>A (IVS5+1G>A)
c.740T>C (p.L274P) |
2 (4)
2 (4)
1 (2)
1 (2) |
3.4%
1.7%
1.7%
3.4% |
North-African Jewish
Arab-Muslim
Arab-Muslim
Arab-Muslim |
|
RPGR |
c.259G>T (p.E87*)
c.592G>A (p.G198R)
c.2964_5delGG (p.G998Gfs*88)
c.2797delG (p.E943Kfs*155)
c.2405_6delAG (p.E802Gfs*31) |
1 (1)
2 (2)
1 (1)
1 (1)
1 (1) |
0.8%
1.7%
0.8%
0.8%
0.8% |
Bedouin
Arab-Muslim
Ashkenazi Jewish
Ashkenazi Jewish
Ashkenazi Jewish |
|
RP2 |
c.530_531delTT (p.F177Yfs*40) |
1 (1) |
0.8% |
Ashkenazi Jewish |
|
SPATA7 |
c.288T>A (p.C96*) |
1 (2) |
1.7% |
Arab-Muslim |
|
USH1C |
c.1220delG (p.G407Efs*56) |
3 (6) |
5.2% |
Yemenite Jewish |
| USH2A | c.377delG (p.S126Ifs*18) c.3959C>T (p.P1320L) c.6937G>T (p.G2313C) c.12052G>A (p.A4018T) | 1 (1) 1 (1) 1 (1) 1 (1) | 0.8% 0.8% 0.8% 0.8% | Arab-Muslim Arab-Muslim Arab-Muslim Arab-Muslim |
Figure 2.
Pie chart showing the distribution of disease-causing genes in the vicinity of Jerusalem. Please note that most families (65%) have not been genetically diagnosed (“Unknown”).
Discussion
Autosomal recessive retinal diseases are predicted to be more common in populations in which consanguineous and intracommunity marriages are common. Consanguinity levels in different Israeli subpopulations have been reported to be relatively high [8,9,41], and this fact was appreciated even earlier by others who predicted a high prevalence of AR diseases, including nonsyndromic RP [37].
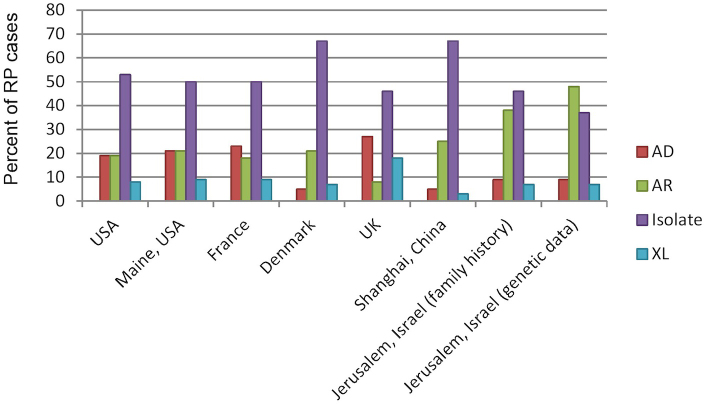
To date, few studies have estimated the prevalence of nonsyndromic RP, and these studies were performed in populations with a relatively low level of consanguinity including the state of Maine (USA), Denmark, and Norway [32-34]. Taken together, these studies suggested a prevalence rate of 1:5,256 individuals on average. We provide evidence that substantiates for the first time Merin and Auerbach’s hypothesis regarding the prevalence of nonsyndromic RP in our population, which in the vicinity of Jerusalem is two-and-a-half to three times higher than that previously reported in other populations. This high prevalence is likely to be due to the high frequency of consanguineous marriages in the studied population in association with founder mutations. We showed that the proportion of families with the AR inheritance pattern is much higher than that reported in other populations (Figure 3). The rate of AR inheritance presented is probably an underestimate in all populations, as the yet-unidentified isolate cases, which constitute a large group in all the studies, are likely to represent mainly AR as well as XL cases. Once the causative gene is identified in these cases, the proportion is likely to shift toward an even higher rate of AR cases (Figure 3, family tree versus genetic data bars).
Figure 3.
Distribution of RP inheritance patterns in different countries. The data are based on the following publications: the United States [45], Maine (the United States) [32], France [46], Denmark [33], the United Kingdom [47], and Shanghai (China) [48]. In some reports, the inheritance pattern could not be clearly established for some of the families, and we therefore corrected the percentage data accordingly. The data of the current study are presented based on family history as well as a revised distribution following results obtained in the genetic analysis.
Founder mutations identified in the Israeli Jewish population, and mainly in Ashkenazi Jews, were also reported to be prevalent among non-Israeli Jews residing in North America [17,42-44]. This can greatly facilitate mutation analysis of Ashkenazi Jewish patients with RP. The most common disease-causing alleles found in this study are DHDDS p.K42E and FAM161A c.1355_6delCA, and the most common RP-causing genes are DHDDS, FAM161A, and EYS. This distribution is different from that reported in other populations in which other genes such as USH2A (GeneID 7399, OMIM: 608400), ABCA4 (GeneID 24, OMIM: 601691), and RHO (GeneID 6010, OMIM: 180380) are the major RP-causing genes when mutated [4]. In summary, we present evidence for the relatively high prevalence of nonsyndromic RP in the vicinity of Jerusalem, and we predict that this high prevalence reflects similar rates throughout the country and many other countries in the Middle East in which consanguineous marriages and founder mutations are not rare.
Acknowledgments
The study was financially supported by the Foundation Fighting Blindness USA (BR-GE-0510–0490-HUJ to DS) and the Yedidut 1 research grant (to EB).
References
- 1.Roosing S, Thiadens AA, Hoyng CB, Klaver CC, den Hollander AI, Cremers FP. Causes and consequences of inherited cone disorders. Prog Retin Eye Res. 2014;42:1–26. doi: 10.1016/j.preteyeres.2014.05.001. [DOI] [PubMed] [Google Scholar]
- 2.Berger W, Kloeckener-Gruissem B, Neidhardt J. The molecular basis of human retinal and vitreoretinal diseases. Prog Retin Eye Res. 2010;29:335–75. doi: 10.1016/j.preteyeres.2010.03.004. [DOI] [PubMed] [Google Scholar]
- 3.Rivolta C, Sharon D, DeAngelis MM, Dryja TP. Retinitis pigmentosa and allied diseases: numerous diseases, genes, and inheritance patterns. Hum Mol Genet. 2002;11:1219–27. doi: 10.1093/hmg/11.10.1219. [DOI] [PubMed] [Google Scholar]
- 4.Hartong DT, Berson EL, Dryja TP. Retinitis pigmentosa. Lancet. 2006;368:1795–809. doi: 10.1016/S0140-6736(06)69740-7. [DOI] [PubMed] [Google Scholar]
- 5.den Hollander AI, Roepman R, Koenekoop RK, Cremers FP. Leber congenital amaurosis: genes, proteins and disease mechanisms. Prog Retin Eye Res. 2008;27:391–419. doi: 10.1016/j.preteyeres.2008.05.003. [DOI] [PubMed] [Google Scholar]
- 6.Kennan A, Aherne A, Humphries P. Light in retinitis pigmentosa. Trends Genet. 2005;21:103–10. doi: 10.1016/j.tig.2004.12.001. [DOI] [PubMed] [Google Scholar]
- 7.Rosenberg T. Epidemiology of hereditary ocular disorders. Dev Ophthalmol. 2003;37:16–33. doi: 10.1159/000072036. [DOI] [PubMed] [Google Scholar]
- 8.Cohen T, Vardi-Saliternik R, Friedlander Y. Consanguinity, intracommunity and intercommunity marriages in a population sample of Israeli Jews. Ann Hum Biol. 2004;31:38–48. doi: 10.1080/0301446032000159255. [DOI] [PubMed] [Google Scholar]
- 9.Vardi-Saliternik R, Friedlander Y, Cohen T. Consanguinity in a population sample of Israeli Muslim Arabs, Christian Arabs and Druze. Ann Hum Biol. 2002;29:422–31. doi: 10.1080/03014460110100928. [DOI] [PubMed] [Google Scholar]
- 10.Charrow J. Ashkenazi Jewish genetic disorders. Fam Cancer. 2004;3:201–6. doi: 10.1007/s10689-004-9545-z. [DOI] [PubMed] [Google Scholar]
- 11.Myerowitz R, Costigan FC. The major defect in Ashkenazi Jews with Tay-Sachs disease is an insertion in the gene for the alpha-chain of beta-hexosaminidase. J Biol Chem. 1988;263:18587–9. [PubMed] [Google Scholar]
- 12.Zimran A, Gelbart T, Beutler E. Linkage of the PvuII polymorphism with the common Jewish mutation for Gaucher disease. Am J Hum Genet. 1990;46:902–5. [PMC free article] [PubMed] [Google Scholar]
- 13.Ben-Yosef T, Ness SL, Madeo AC, Bar-Lev A, Wolfman JH, Ahmed ZM, Desnick RJ, Willner JP, Avraham KB, Ostrer H, Oddoux C, Griffith AJ, Friedman TB. A mutation of PCDH15 among Ashkenazi Jews with the type 1 Usher syndrome. N Engl J Med. 2003;348:1664–70. doi: 10.1056/NEJMoa021502. [DOI] [PubMed] [Google Scholar]
- 14.Ness SL, Ben-Yosef T, Bar-Lev A, Madeo AC, Brewer CC, Avraham KB, Kornreich R, Desnick RJ, Willner JP, Friedman TB, Griffith AJ. Genetic homogeneity and phenotypic variability among Ashkenazi Jews with Usher syndrome type III. J Med Genet. 2003;40:767–72. doi: 10.1136/jmg.40.10.767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Stone EM, Luo X, Heon E, Lam BL, Weleber RG, Halder JA, Affatigato LM, Goldberg JB, Sumaroka A, Schwartz SB, Cideciyan AV, Jacobson SG. Autosomal Recessive Retinitis Pigmentosa caused by Mutations in the MAK Gene. Invest Ophthalmol Vis Sci. 2011;52:9665–73. doi: 10.1167/iovs.11-8527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zelinger L, Banin E, Obolensky A, Mizrahi-Meissonnier L, Beryozkin A, Bandah-Rozenfeld D, Frenkel S, Ben-Yosef T, Merin S, Schwartz SB, Cideciyan AV, Jacobson SG, Sharon D. A missense mutation in DHDDS, encoding dehydrodolichyl diphosphate synthase, is associated with autosomal-recessive retinitis pigmentosa in Ashkenazi Jews. Am J Hum Genet. 2011;88:207–15. doi: 10.1016/j.ajhg.2011.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Züchner S, Dallman J, Wen R, Beecham G, Naj A, Farooq A, Kohli MA, Whitehead PL, Hulme W, Konidari I, Edwards YJ, Cai G, Peter I, Seo D, Buxbaum JD, Haines JL, Blanton S, Young J, Alfonso E, Vance JM, Lam BL, Pericak-Vance MA. Whole-exome sequencing links a variant in DHDDS to retinitis pigmentosa. Am J Hum Genet. 2011;88:201–6. doi: 10.1016/j.ajhg.2011.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Behar DM, Yunusbayev B, Metspalu M, Metspalu E, Rosset S, Parik J, Rootsi S, Chaubey G, Kutuev I, Yudkovsky G, Khusnutdinova EK, Balanovsky O, Semino O, Pereira L, Comas D, Gurwitz D, Bonne-Tamir B, Parfitt T, Hammer MF, Skorecki K, Villems R. The genome-wide structure of the Jewish people. Nature. 2010;466:238–42. doi: 10.1038/nature09103. [DOI] [PubMed] [Google Scholar]
- 19.Ostrer H. A genetic profile of contemporary Jewish populations. Nat Rev Genet. 2001;2:891–8. doi: 10.1038/35098506. [DOI] [PubMed] [Google Scholar]
- 20.Behar DM, Hammer MF, Garrigan D, Villems R, Bonne-Tamir B, Richards M, Gurwitz D, Rosengarten D, Kaplan M, Della Pergola S, Quintana-Murci L, Skorecki K. MtDNA evidence for a genetic bottleneck in the early history of the Ashkenazi Jewish population. Eur J Hum Genet. 2004;12:355–64. doi: 10.1038/sj.ejhg.5201156. [DOI] [PubMed] [Google Scholar]
- 21.Rodríguez-Ezpeleta N, Alvarez-Busto J, Imaz L, Regueiro M, Azcarate MN, Bilbao R, Iriondo M, Gil A, Estonba A, Aransay AM. High-density SNP genotyping detects homogeneity of Spanish and French Basques, and confirms their genomic distinctiveness from other European populations. Hum Genet. 2010;128:113–7. doi: 10.1007/s00439-010-0833-4. [DOI] [PubMed] [Google Scholar]
- 22.Atzmon G, Hao L, Pe'er I, Velez C, Pearlman A, Palamara PF, Morrow B, Friedman E, Oddoux C, Burns E, Ostrer H. Abraham's children in the genome era: major Jewish diaspora populations comprise distinct genetic clusters with shared Middle Eastern Ancestry. Am J Hum Genet. 2010;86:850–9. doi: 10.1016/j.ajhg.2010.04.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kopelman NM, Stone L, Wang C, Gefel D, Feldman MW, Hillel J, Rosenberg NA. Genomic microsatellites identify shared Jewish ancestry intermediate between Middle Eastern and European populations. BMC Genet. 2009;10:80. doi: 10.1186/1471-2156-10-80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Al-Gazali L, Hamamy H, Al-Arrayad S. Genetic disorders in the Arab world. BMJ. 2006;333:831–4. doi: 10.1136/bmj.38982.704931.AE. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Bittles AH, Black ML. Evolution in health and medicine Sackler colloquium: Consanguinity, human evolution, and complex diseases. Proc Natl Acad Sci USA. 2010;107(Suppl 1):1779–86. doi: 10.1073/pnas.0906079106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Jaber L, Bailey-Wilson JE, Haj-Yehia M, Hernandez J, Shohat M. Consanguineous matings in an Israeli-Arab community. Arch Pediatr Adolesc Med. 1994;148:412–5. doi: 10.1001/archpedi.1994.02170040078013. [DOI] [PubMed] [Google Scholar]
- 27.Zlotogora J. The molecular basis of autosomal recessive diseases among the Arabs and Druze in Israel. Hum Genet. 2010;128:473–9. doi: 10.1007/s00439-010-0890-8. [DOI] [PubMed] [Google Scholar]
- 28.Carmi R, Rokhlina T, Kwitek-Black AE, Elbedour K, Nishimura D, Stone EM, Sheffield VC. Use of a DNA pooling strategy to identify a human obesity syndrome locus on chromosome 15. Hum Mol Genet. 1995;4:9–13. doi: 10.1093/hmg/4.1.9. [DOI] [PubMed] [Google Scholar]
- 29.Nishimura DY, Searby CC, Carmi R, Elbedour K, Van Maldergem L, Fulton AB, Lam BL, Powell BR, Swiderski RE, Bugge KE, Haider NB, Kwitek-Black AE, Ying L, Duhl DM, Gorman SW, Heon E, Iannaccone A, Bonneau D, Biesecker LG, Jacobson SG, Stone EM, Sheffield VC. Positional cloning of a novel gene on chromosome 16q causing Bardet- Biedl syndrome (BBS2). Hum Mol Genet. 2001;10:865–74. doi: 10.1093/hmg/10.8.865. [DOI] [PubMed] [Google Scholar]
- 30.Chiang AP, Nishimura D, Searby C, Elbedour K, Carmi R, Ferguson AL, Secrist J, Braun T, Casavant T, Stone EM, Sheffield VC. Comparative genomic analysis identifies an ADP-ribosylation factor-like gene as the cause of Bardet-Biedl syndrome (BBS3). Am J Hum Genet. 2004;75:475–84. doi: 10.1086/423903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Chiang AP, Beck JS, Yen HJ, Tayeh MK, Scheetz TE, Swiderski RE, Nishimura DY, Braun TA, Kim KY, Huang J, Elbedour K, Carmi R, Slusarski DC, Casavant TL, Stone EM, Sheffield VC. Homozygosity mapping with SNP arrays identifies TRIM32, an E3 ubiquitin ligase, as a Bardet-Biedl syndrome gene (BBS11). Proc Natl Acad Sci USA. 2006;103:6287–92. doi: 10.1073/pnas.0600158103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Bunker CH, Berson EL, Bromley WC, Hayes RP, Roderick TH. Prevalence of retinitis pigmentosa in Maine. Am J Ophthalmol. 1984;97:357–65. doi: 10.1016/0002-9394(84)90636-6. [DOI] [PubMed] [Google Scholar]
- 33.Haim M. Epidemiology of retinitis pigmentosa in Denmark. Acta Ophthalmol Scand Suppl. 2002;233:1–34. doi: 10.1046/j.1395-3907.2002.00001.x. [DOI] [PubMed] [Google Scholar]
- 34.Grøndahl J. Estimation of prognosis and prevalence of retinitis pigmentosa and Usher syndrome in Norway. Clin Genet. 1987;31:255–64. doi: 10.1111/j.1399-0004.1987.tb02804.x. [DOI] [PubMed] [Google Scholar]
- 35.Bundey S, Crews SJ. A study of retinitis pigmentosa in the city of Birmingham. II Clinical and genetic heterogeneity. J Med Genet. 1984;21:421–8. doi: 10.1136/jmg.21.6.421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Hu DN. Prevalence and mode of inheritance of major genetic eye diseases in China. J Med Genet. 1987;24:584–8. doi: 10.1136/jmg.24.10.584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Merin S, Auerbach E. Retinitis pigmentosa. Surv Ophthalmol. 1976;20:303–46. doi: 10.1016/s0039-6257(96)90001-6. [DOI] [PubMed] [Google Scholar]
- 38.Rosner M, Hefetz L, Abraham FA. The prevalence of retinitis pigmentosa and congenital night blindness in Israel. Am J Ophthalmol. 1993;116:373–4. doi: 10.1016/s0002-9394(14)71358-3. [DOI] [PubMed] [Google Scholar]
- 39.Bandah-Rozenfeld D, Mizrahi-Meissonnier L, Farhy C, Obolensky A, Chowers I, Pe'er J, Merin S, Ben-Yosef T, Ashery-Padan R, Banin E, Sharon D. Homozygosity mapping reveals null mutations in FAM161A as a cause of autosomal-recessive retinitis pigmentosa. Am J Hum Genet. 2010;87:382–91. doi: 10.1016/j.ajhg.2010.07.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Khateb S, Zelinger L, Ben-Yosef T, Merin S, Crystal-Shalit O, Gross M, Banin E, Sharon D. Exome sequencing identifies a founder frameshift mutation in an alternative exon of USH1C as the cause of autosomal recessive retinitis pigmentosa with late-onset hearing loss. PLoS ONE. 2012;7:e51566. doi: 10.1371/journal.pone.0051566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Zlotogora J. Autosomal recessive diseases among Palestinian Arabs. J Med Genet. 1997;34:765–6. doi: 10.1136/jmg.34.9.765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Venturini G, Di Gioia SA, Harper S, Weigel-DiFranco C, Rivolta C, Berson EL. Molecular genetics of FAM161A in North American patients with early-onset retinitis pigmentosa. PLoS ONE. 2015;9:e92479. doi: 10.1371/journal.pone.0092479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Venturini G, Koskiniemi-Kuendig H, Harper S, Berson EL, Rivolta C. Two specific mutations are prevalent causes of recessive retinitis pigmentosa in North American patients of Jewish ancestry. Genet Med. 2015;17:285–90. doi: 10.1038/gim.2014.132. [DOI] [PubMed] [Google Scholar]
- 44.Tucker BA, Scheetz TE, Mullins RF, DeLuca AP, Hoffmann JM, Johnston RM, Jacobson SG, Sheffield VC, Stone EM. Exome sequencing and analysis of induced pluripotent stem cells identify the cilia-related gene male germ cell-associated kinase (MAK) as a cause of retinitis pigmentosa. Proc Natl Acad Sci USA. 2011;108:E569–76. doi: 10.1073/pnas.1108918108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Fishman GA. Retinitis pigmentosa. Genetic percentages. Arch Ophthalmol. 1978;96:822–6. doi: 10.1001/archopht.1978.03910050428005. [DOI] [PubMed] [Google Scholar]
- 46.Bocquet B, Lacroux A, Surget MO, Baudoin C, Marquette V, Manes G, Hebrard M, Senechal A, Delettre C, Roux AF, Claustres M, Dhaenens CM, Rozet JM, Perrault I, Bonnefont JP, Kaplan J, Dollfus H, Amati-Bonneau P, Bonneau D, Reynier P, Audo I, Zeitz C, Sahel JA, Paquis-Flucklinger V, Calvas P, Arveiler B, Kohl S, Wissinger B, Blanchet C, Meunier I, Hamel CP. Relative frequencies of inherited retinal dystrophies and optic neuropathies in Southern France: assessment of 21-year data management. Ophthalmic Epidemiol. 2013;20:13–25. doi: 10.3109/09286586.2012.737890. [DOI] [PubMed] [Google Scholar]
- 47.Jay M. On the heredity of retinitis pigmentosa. Br J Ophthalmol. 1982;66:405–16. doi: 10.1136/bjo.66.7.405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Hu DN. Genetic aspects of retinitis pigmentosa in China. Am J Med Genet. 1982;12:51–6. doi: 10.1002/ajmg.1320120107. [DOI] [PubMed] [Google Scholar]