Abstract
The aim of this meta-analysis is to examine the effects of dexmedetomidine on serum inflammatory markers when administered perioperatively. We searched multiple electronic databases for relevant research papers, and carried out meta-analyses of weighted mean differences and interpreted in the light of statistical heterogeneity (I2). Fifteen RCTs recruiting 641 patients were included. Dexmedetomidine treatment significantly decreased interleukin-6 (IL-6), IL-8 and tumor necrosis factor-alpha (TNF-α) levels with mean differences [95% CI] in the changes from baseline between dexmedetomidine treated and controls of −25.14 [−35.29, −15.00]; P < 0.00001 (for IL-6), −5.69 [−10.77, −0.60]; P < 0.04 (for IL-8), and −20.30 [−30.93, −9.67]; P < 0.0002 (for TNF-α) immediately after surgery; and −41.55 [−57.41, −25.70]; P < 0.00001 (IL-6), −6.46 [−10.83, −2.08]; P < 0.005 (IL-8), and −14.67 [−22.61, −6.73]; P < 0.0003 (TNF-α) on postoperative day 1 (random effects). IL-10 levels were found to increase significantly a day after surgery (8.33 [3.31, 13.36]; P = 0.001). Subgroup analyses did not reveal significant differences. In conclusion, perioperative adjunctive use of dexmedetomidine substantially decreases serum IL-6, IL-8 and TNF-α levels.
The postoperative period constitutes a regenerative period associated with remarkable physiological processes for wound healing. Among these, inflammation is an important process which sets a stage for the protection against infections and regenerative cell recruitment1. However, excessive inflammatory response leaves deleterious effects not only on wound healing but also on other physiological functions, therefore, a balance between inflammatory factors and timely resolution of inflammation is essential for wound healing and normalization2. Anesthetics favoring this delicate balance are thus desirable as their use may reduce postoperative complications and mortality. Among such anesthetics, dexmedetomidine has emerged as an important modulator of anxiolysis that tends to normalize arterial blood pressure and heart rate without declining respiratory function or arterial oxygen saturation3,4,5. There is considerable evidence that this α2-adrenergic receptor agonist can manifest additional protective effects on the heart, brain and kidneys6,7,8,9.
Preclinical studies on murine models have revealed that dexmedetomidine manifests multi-faceted protective effects including the inhibition of proinflammatory cytokine production10,11,12. In vitro studies with human whole blood samples have shown that dexmedetomidine significantly suppresses lipopolysaccharide induced proinflammatory mediators including tumor necrosis factor - alpha (TNF-α), interleukin (IL)-6, and IL-8 production dose-dependently13. Clinical investigations with human subjects evaluating the effects of dexmedetomidine on serum inflammatory cytokines during perioperative conditions are rather fragmentary with between-study inconsistencies in the outcomes14,15,16,17,18,19,20,21,22,23,24,25,26,27,28. In order to evaluate the effect of dexmedetomidine on inflammatory responses, we performed a meta-analysis of randomized controlled trials (RCTs) that examined one or more parameters of inflammatory markers (pro-inflammatory as well as anti-inflammatory) in patients treated with dexmedetomidine as an adjunct to general anesthesia during surgical/critical procedures.
Method
Literature search
The literature was searched for relevant peer-reviewed research papers published between 1990 and July 2014. Electronic databases including EBSCO, Embase, Google Scholar, Ovid SP, PubMed, Scopus, and Web of Science were utilized. The major MeSH terms and important keywords including dexmedetomidine, anesthesia, sedation, surgery, inflammation, anti-inflammatory, pro-inflammatory cytokine, interleukin, IL-1, IL-2, IL-4, IL-6, IL-8, IL-10, IL-12, IL-18, tumor necrotic factor-α (TNF-α), and c-reactive protein (CRP) were used. Reference lists of significant articles were scrolled and corroborations were also explored.
Inclusion and exclusion criteria
The inclusion criteria were: (a) trials of patients undergoing surgical procedures or traumatic interventions wherein the effectiveness of perioperative dexmedetomidine against inflammatory stress was compared to a control group; (b) trials investigated the effect of dexmedetomidine on inflammatory response by measuring one or more relevant pro-inflammatory and/or anti-inflammatory cytokines, and other important modulators of inflammation; and (c) trials reporting at least one outcome measure of inflammatory response after examining baseline, postoperative, and late-point samples. Exclusion criteria were: (a) trials assessing the effects of dexmedetomidine on hemodynamic indicators or injury markers other than inflammatory modulators; (b) single arm studies; (c) case reports; and (d) in vitro studies.
Primary and secondary data points
The primary endpoints of the present study were changes in the serum proinflammatory cytokines, IL-2, IL-6, IL-8, IL-12, IL-18, TNF-α, and anti-inflammatory cytokines (IL-1, IL-4, IL-10, and IL-16) levels from baseline. The secondary endpoint was the changes in serum CRP levels.
Quality assessment of the trials
Each individual study was carefully evaluated for strengths, limitations, design, methodology, outcome dissemination, and interpretation. A formal quality assessment was made by using the Cochrane Collaboration Risk of Bias Assessment Tool for the assessment of RCTs which examines each trial’s internal validity, possibilities of risk of bias in various phases of trial conduct and outcome analyses. The tool weighs risk of a possible bias under “Low” or “High” measures and does not attempt numerical scores29.
Data collection and statistical analysis
The required data including primary and secondary endpoint outcomes, dexmedetomidine dosage, general anesthetic type and dosage, subjects’ demographics, and other relevant information was collected from the individual papers and organized in a database by two authors (BL and HW) independently with good inter-rater reliability (kappa = 0.95).
Meta-analyses were performed using the RevMan software (Version 5.2; Cochrane Collaboration) under both fixed- (FEM) and random-effects (REM) models. Changes from baseline in serum inflammatory markers were first measured and then means and standard deviations were used to calculate mean differences with 95% confidence intervals (CI). For studies reporting effect sizes as a median with range or interquartile range, the median was used instead of the mean while the standard deviation was estimated from range or interquartile range with the help of statistical tools providing reliable approximations30,31. The overall effect of each meta-analysis was a weighted average with 95% CI of the inverse variance adjusted effect size of individual studies.
Between-studies statistical heterogeneity was tested with I2 index. Sensitivity analyses were performed to examine the source of higher heterogeneity and effect size validity. Subgroup analyses were carried out in order to evaluate the impact of dose concentration, duration of surgery, duration of anesthesia, timing of dexmedetomidine administration, and type of surgery on the changes from baseline in the serum levels of IL-6 and TNF-α after surgery. For each variable, subgroup pair was first defined and then meta-analyzed. The overall effect sizes of each member of a subgroup pair were subjected to two-tailed z test for examining the significance of difference. Details of these subgroups are given in Table 1.
Table 1. Meta-analyses outcomes as mean differences and 95% confidence interval between dexmedetomidine treated and control patients in changes from baseline in inflammatory markers.
| Parameter | Studies | Patients | Mean difference [95% confidence interval] |
I2 | Results favour | |
|---|---|---|---|---|---|---|
| Fixed effects | Random effects | |||||
| IL-6 levels (pg/ml) after surgery | 14 | 564 | −12.32 [−14.19, −10.46]; P < 0.00001 | −25.14 [−35.29, −15.00]; P < 0.00001 | 95% | DEX |
| IL-6 levels (pg/ml) a day after surgery | 13 | 557 | −18.39 [−20.54, −16.25]; P < 0.00001 | −41.55 [−57.41, −25.70]; P < 0.00001 | 98% | DEX |
| IL-8 levels (pg/ml) after surgery | 5 | 233 | −7.36 [−8.70, −6.02]; P < 0.00001 | −5.69 [−10.77, −0.60]; P < 0.04 | 90% | DEX |
| IL-8 levels (pg/ml) a day after surgery | 5 | 235 | −5.73 [−7.50, −3.96]; P < 0.00001 | −6.46 [−10.83, −2.08]; P < 0.005 | 70% | DEX |
| TNF-α levels (pg/ml) after surgery | 10 | 438 | −6.00 [−7.05, −4.94]; P < 0.00001 | −20.30 [−30.93, −9.67]; P < 0.0002 | 98% | DEX |
| TNF-α levels (pg/ml) a day after surgery | 10 | 438 | −5.90 [−7.08, −4.71]; P < 0.00001 | −14.67 [−22.61, −6.73]; P < 0.0003 | 96% | DEX |
| IL-10 levels (pg/ml) after surgery | 4 | 153 | 4.44 [−0.80, 9.68]; P = 0.1 | −2.18 [−31.28, 26.92]; P = 0.88 | 89% | Indifferent |
| IL-10 levels (pg/ml) a day after surgery | 3 | 158 | 8.33 [3.31, 13.36]; P = 0.001 | 8.08 [−0.06, 16.22]; P = 0.05 | 45% | DEX |
| CRP levels (mg/l) after surgery | 2 | 94 | 1.07 [−0.74, 2.88]; P = 0.25 | −23.05 [−73.86, 27.75]; P = 0.37 | 94% | Indifferent |
| CRP levels (mg/l) a day after surgery | 2 | 94 | 8.34 [2.31, 14.37]; P < 0.008 | −49.58 [−180.90, 81.73]; P = 0.46 | 99% | Indifferent |
Results
Fifteen RCTs14,15,16,17,18,19,20,21,22,23,24,25,26,27,28 were selected for inclusion. The process of study screening and selection is summarized in Fig. S1. Multiple database literature searches led to the identification of 1149 article abstracts. At the screening level, 818 search engine-retrieved abstracts were excluded because of the irrelevancy with research question. Main reasons for the exclusions at study eligibility determination stage included: the endpoints restricted to sympatho-adrenal parameters only (n = 21); the endpoints restricted to hemodynamic, cardiovascular, respiratory, neurological, and/or other manifestations of physiological stress (n = 87); the study performed by using animal model/s (n = 46); other dexmedetomidine efficacy, safety, comparative, dose-sparing assessments and combinational studies (n = 162).
Major characteristics of the included studies are presented in Table S1. From the included studies, data of 641 patients with average age 47.47 ± 8.47 years (range: 4 ± 5 months to 72.7 ± 7 years) were used for the meta-analyses of various parameters. Initial doses (first 10 minutes) of dexmedetomidine ranged from 0.5 to 2.5 μg/kg (mean: 1.08 ± 0.56) which followed maintenance doses of 0.05–0.5 μg/kg/h (mean ± SD: 0.36 ± 0.15) throughout anesthesia period. Dexmedetomidine was administered intraoperatively in 12 studies, postoperatively in two studies, and for sedation of trauma patients in one study. Anesthetics and anxiolytics administered during the surgical procedures included sufentanil, fentanyl, alfentanil, diazepam, propofol, midazolam, lidocaine, etomidate, bupivacaine, sevoflurane, isoflurane, vecuronium, pancuronium, rocuronium, vecuronium, and atracurium (Table S1).
Types of surgeries included elective multilevel lumbar fusion, hepatectomy, laparoscopic cholecystectomy, tetralogy of fallot, ventricular septal/atrioventricular defects, elective major abdominal/pelvic surgery, elective multilevel lumbar fusion, one lung ventilation, cardiopulmonary bypass, ileus, esophageal carcinoma resection, and cerebral surgery.
The quality of the included studies, in general, ranged from medium to high, when weighed with the Cochrane Collaboration Risk of Bias Assessment Tool (Table S2). Among the included studies, all were randomized but 7 were double blind, one single blind, and remainder not blinded interventions.
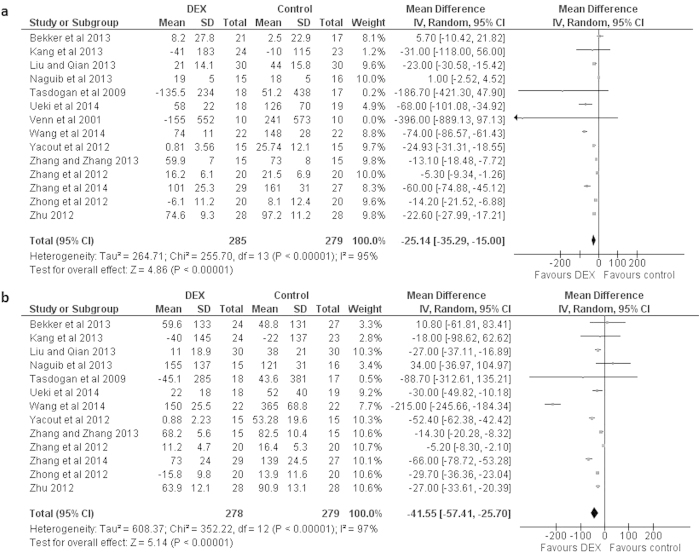
Major findings of the meta-analyses are presented in Table 1. Dexmedetomidine administration significantly decreased IL-6 levels in the samples taken from patients after surgery. Mean difference [95% CI] between dexmedetomidine treated and control patients in changes from baseline was −25.14 [−35.29, −15.00] pg/ml; P < 0.00001; REM (Fig. 1a). This statistically significant difference was also observed on the first postoperative day with a mean of −41.55 [−57.41, −25.70] pg/ml; P < 0.00001; REM (Fig. 1b).
Figure 1. Forest graph showing the effect of dexmedetomidine on the changes from baseline in serum IL-6 levels.
(a) after surgery, and (b) a day after surgery. Boxes represent mean differences and line across each box represents respective 95% CI. Diamond represents overall effect size of the meta-analysis.
In the subgroup analyses, there were no significant differences between the utility of dexmedetomidine dose of over median vs under median; or surgery duration of more than median vs less than median; or anesthesia duration of more than median vs less than median; or timing of dexmedetomidine administration (intraoperative vs postoperative); or surgery type of cardiac vs non-cardiac/gastrointestinal vs non-gastrointestinal (Table 2).
Table 2. Subgroup analyses for the assessment of impact of dexmedetomidine (DEX) dose, operative time, duration of anesthesia, timing of DEX administration and surgery type on serum IL-6 and TNF-α levels after surgery.
| Subgroups | Studies with | No. of studies | Mean difference [95% CI] between DEX treated and control subjects | Between subgroup significance |
|---|---|---|---|---|
| IL-6 | ||||
| D1 | DEX dose* equal & over median (0.4 μg/kg/h) | 8 | −17.54 [−28.57, −6.52]; P = 0.002; REM | Z = 0.656; P = 0.512 |
| D2 | DEX dose* under median (0.4 μg/kg/h) | 6 | −91.66 [−126.28, −57.04]; P < 0.00001; REM | |
| ST1 | Surgery duration over median (184 min) | 5 | −31.31 [−54.82, −7.81]; P = 0.009; REM | Z = 0.041; P = 0.967 |
| ST2 | Surgery duration under median (184 min) | 4 | −30.01 [−46.69, −13.33]; P = 0.0004; REM | |
| AT1 | Anesthesia duration over median (266 min) | 5 | −108.29 [−171.25, −45.32]; P = 0.0008; REM | Z = 0.919; P = 0.358 |
| AT2 | Anesthesia duration under median (266 min) | 4 | −39.16 [−60.86, −17.46]; P = 0.0004; REM | |
| IO | Intraoperative DEX administration | 11 | −25.86 [−36.70, −15.02]; P < 0.00001; REM | Z = 0.635; P = 0.525 |
| PO | Postoperative DEX administration | 3 | −199.00 [−506.41, 108.41]; P = 0.20; REM | |
| C | Cardiac surgery patients | 4 | −18.50 [−33.48, −3.52]; P = 0.02; REM | Z = 1; P = 0.318 |
| NC | Non-cardiac surgery patients | 10 | −54.08 [−74.05, −34.11]; P < 0.00001; REM | |
| GIT | Gastrointestinal surgery patients | 5 | −113.58 [−167.78, −59.38]; P < 0.0001; REM | Z = 1.75; P = 0.079 |
| NGIT | Non-gastrointestinal surgery patients | 9 | −13.64 [−21.95, −5.33]; P = 0.001; REM | |
| TNF-α | ||||
| D1 | DEX dose* equal & over median (0.4 μg/kg/h) | 5 | −8.88 [−19.96, 2.20]; P = 0.12; REM | Z = 0.983; P = 0.325 |
| D2 | DEX dose* under median (0.4 μg/kg/h) | 4 | −43.26 [−69.92, −16.60];p = 0.001; REM | |
| ST1 | Surgery duration over median (167 min) | 4 | −26.08 [−45.08, −7.09]; P = 0.007; REM | Z = 0.923; P = 0.35 |
| ST2 | Surgery duration under median 167 min) | 3 | −134.65 [−265.76, −3.54]; P = 0.04; REM | |
| AT1 | Anesthesia duration over median (226 min) | 4 | −23.75 [−41.44, −6.07];P = 0.008; REM | Z = 0.642; P = 0.52 |
| AT2 | Anesthesia duration under median (226 min) | 2 | −171.94 [−490.11, 146.24]; P = 0.29; REM | |
| C | Cardiac surgery patients | 3 | −25.76 [−62.05, 10.52]; P = 0.16; REM | Z = 0.095; P = 0.924 |
| NC | Non-cardiac surgery patients | 7 | −22.32 [−34.65, −9.98]; P = 0.0004; REM | |
| GIT | Gastrointestinal surgery patients | 3 | −72.86 [−131.99, −13.74]; P = 0.02; REM | Z = 1.177; P = 0.239 |
| NGIT | Non-gastrointestinal surgery patients | 7 | −9.79 [−18.79, −0.80]; P = 0.03; REM | |
*Maintenance dose
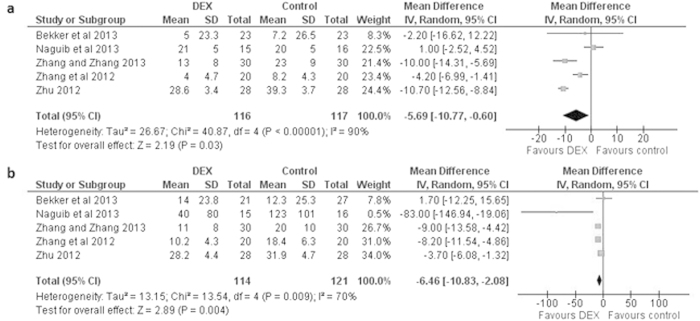
Among the other proinflammatory markers, IL-8 levels also decreased significantly in the postoperative period as well as a day after surgery in dexmedetomidine administered patients with a mean difference of −5.69 [−10.77, −0.60] pg/ml (P < 0.04) immediately postoperative and −6.46 [−10.83, −2.08] pg/ml (P < 0.005) at postoperative day 1 (Fig. 2a,b).
Figure 2. Forest graph showing the effect of dexmedetomidine on the changes from baseline in serum IL-8 levels.
(a) after surgery, and (b) a day after surgery. Boxes represent mean differences and line across each box represents respective 95% CI. Diamond represents overall effect size of the meta-analysis.
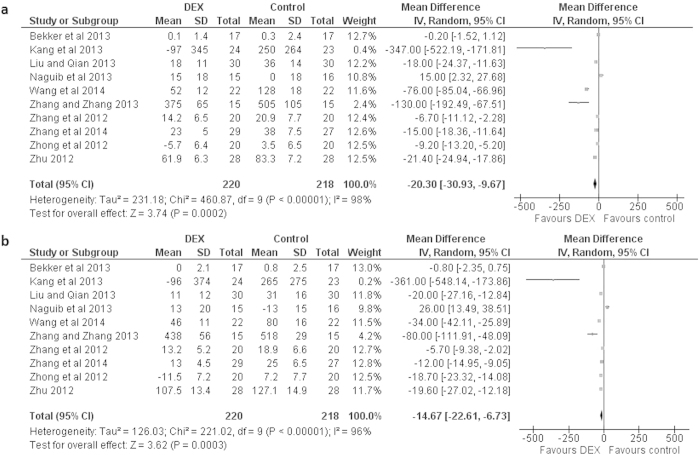
Dexmedetomidine administration was also associated with a significant reductions in the serum concentrations of TNF-α by a mean difference (95% CI) between dexmedetomidine treated and control patients in changes from baseline of −20.30 [−30.93, −9.67] pg/ml (P < 0.0002; REM) immediately after surgery and −14.67 [−22.61, −6.73] pg/ml (P < 0.0003; REM) one day after surgery (Fig. 3a,b).In the subgroup analyses, there were no significant differences between the utility of dexmedetomidine dose of over median vs under median; or surgery duration of more than median vs less than median; or anesthesia duration of more than median vs less than median; or surgery type of cardiac vs non-cardiac/gastrointestinal vs non-gastrointestinal (Table 2).
Figure 3. Forest graph showing the effect of dexmedetomidine on the changes from baseline in serum TNF-α levels.
(a) after surgery, and (b) a day after surgery. Boxes represent mean differences and line across each box represents respective 95% CI. Diamond represents overall effect size of the meta-analysis.
There was no significant difference in the changes from baseline in the serum levels of IL-10 between dexmedetomidine treated and control patients soon after surgery (mean difference −2.18 [−31.28, 26.92]; P = 0.88; REM) but IL-10 levels were significantly higher in dexmedetomidine treated patients a day after surgery (mean difference 8.33 [3.31, 13.36]; P = 0.001; FEM). No significant differences were noted with regards to CRP levels (Table 1).
For the evaluation of other inflammatory cytokines, data were either not available or insufficient for the meta-analyses. Among these, no significant differences were noted in the changes from baseline either immediately after surgery or a day after surgery between dexmedetomidine and control patients in the levels of IL-1a14, IL-1b15, and IL-119,25. However, postoperative IL-4 levels were found to decrease significantly in the dexmedetomidine treated subjects compared to saline-treated controls in one study (mean difference in the change from baseline: −260.00 [−420.39, −99.61]; P < 0.002)16.
Discussion
The surgical stress response involves metabolic, inflammatory, and immune reactions. Proinflammatory cytokines and chemokines attract leukocytes to the inflammatory sites and recruit neutrophils for the phagocytosis of bacteria. Increased secretion of cytokines as a result of stimulation of immunological and inflammatory reactions disturbs normal proinflammatory and anti-inflammatory cytokine balance which can increase morbidity and mortality32. Sedatives can reduce the severity of this surgical stress response and improve the tolerance of patients for critical procedures33, and of the modern sedatives, dexmedetomidine can provide multiple benefits1,2,3,4,5,6,7.
This meta-analysis was performed to synthesize the fragmentary data pertaining to the effects of perioperative use of dexmedetomidine on inflammatory modulators. We have found that when used as an adjunct to general anesthesia, dexmedetomidine significantly reduced postoperative serum IL-6, IL-8, and TNF-α levels postoperatively as well as at postoperative day 1, and significantly increased IL-10 levels a day after surgery.
Interleukin-6 is a proinflammatory cytokine that exerts both local and systemic effects for reducing tissue injury and combating infections while promoting biochemical mechanisms of healing and repair34. It is the principal interleukin released after surgery and acts as a reliable indicator of the inflammatory response to surgical trauma35. Tumor necrosis factor-α is also a proinflammatory cytokine that regulates several other cytokines36. Increased production of TNF-α is associated with several chronic inflammatory conditions37. Interleukin-8, another pro-inflammatory cytokine, has also been implicated in several pathological conditions including some cancers and may act as a prognostic or predictive biomarker38,39. In the present study, we find a significant decrease in IL-6, IL-8 and TNF-α levels in dexmedetomidine treated patients which is indicative of the anti-inflammatory potentials of dexmedetomidine when used as perioperative adjunctive drug.
Interleukin-10 is an anti-inflammatory cytokine. In the present study, whereas, IL-10 levels were non-significantly higher in the dexmedetomidine treated patients soon after surgery, but a day after surgery, there was a significant increase in IL-10 levels in dexmedetomidine treated than in control patients. However, this meta-analytical outcome gained almost all weight from 2 studies14,25 and therefore, this outcome is subject to the availability of more data in future.
Interleukin-6, IL-8 and TNF-α are implicated in the exhibition of sickness behavior40,41. Dexmedetomidine is reported to attenuate sepsis-induced lung and kidney injuries in a rodent model42. Keeping in view these observations and in the light of results of several studies including meta-analyses that report significantly shorter ICU stay of dexmedetomidine treated patients in perioperative conditions43,44,45, it seems reasonable to ascribe the anti-inflammatory effects of perioperative dexmedetomidine observed in this meta-analysis as beneficial. Indeed, a pre-emptive administration of dexmedetomidine has been advocated by some authors keeping in view its anti-inflammatory effects15,46.
Effects of anesthetics on immunomodulation of inflammation follow a delicate pattern as the immunosuppression can leave positive as well as negative effects and therefore it is necessary to apply an anesthetic or anesthetics in the light of the immunological status of patients. Hazards associated with perioperative immunosuppression can be the increased risk of tumor metastasis and infection whereas the anti-inflammatory effects of anesthetics may provide more benefit in conditions such as ischemia, reperfusion injury and systemic inflammatory response syndrome47.
Although a number of mechanisms of dexmedetomidine action have been postulated including the modulation of cytokine production by macrophages and monocytes during the stress response which may also be stimulated via the α2-adrenoceptors, inhibition of apoptosis, central sympatholytic effects including the stimulation of cholinergic anti-inflammatory pathway, and antinociceptive action involving interactions between pain and immune factors such as proinflammatory cytokines15, however, so far, it is not clear what mechanisms are involved in the perioperative effectiveness of dexmedetomidine rather further research is needed.
The main limitation of this study was the limited number of papers that could be included in some meta-analytical comparisons, and therefore, evidence regarding the effects of dexmedetomidine on IL-1, IL-2, IL-4, and CRP levels remains inconclusive. Methodological heterogeneity may also have some impact on overall outcomes; eleven of the included studies administered dexmedetomidine intraoperatively, 2 studies administered postoperatively, and one study administered the drug to severe trauma patients. One study recruited pediatric patients which was included in the meta-analysis keeping in view that the patterns of cytokine secretion in children remain similar to adults48,49. Use of different anesthetics in different studies may also have slight interference in manifesting the effects of dexmedetomidine. However, sensitivity analyses did not exhibit any significant difference in the overall results.
Conclusion
Perioperative use of dexmedetomidine as an adjunct to general anesthesia leads to significant decreases in serum levels of IL-6, IL-8, and TNF-α within a period of 24 hours postoperatively. High level of meta-analytical heterogeneity may have some impact on the overall results achieved herein. More studies will be required to further refine these results and to evaluate the effect of perioperative dexmedetomidine on IL-1, IL-2, IL-4, IL-10, IL-18, and CRP.
Additional Information
How to cite this article: Li, B. et al. Anti-inflammatory Effects of Perioperative Dexmedetomidine Administered as an Adjunct to General Anesthesia: A Meta-analysis. Sci. Rep. 5, 12342; doi: 10.1038/srep12342 (2015).
Supplementary Material
Footnotes
Author Contributions B.L., Y.L.L., A.H.Z. and H.X.W. carried out the literature search, participated in data analysis and drafted the manuscript; H.X.W., S.S.T. and C.J.G. participated in the design of the study and coordination and helped to draft the manuscript; A.H.Z. and H.W. contributed greatly in the revision of the manuscript. All authors read and approved the final manuscript.
References
- Gouin J. P. & Kiecolt-Glaser J. K. The impact of psychological stress on wound healing: methods and mechanisms. Immunol Allergy Clin North Am. 31, 81–93 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eming S. A., Krieg T. & Davidson J. M. Inflammation in wound repair: molecular and cellular mechanisms. J Invest Dermatol. 127, 514–25 (2007). [DOI] [PubMed] [Google Scholar]
- Venn R. M., Karol M. D. & Grounds R. M. Pharmacokinetics of dexmedetomidine infusions for sedation of postoperative patients requiring intensive care. Br J Anaesth. 88, 669–75 (2002). [DOI] [PubMed] [Google Scholar]
- Bekker A. et al. The effect of dexmedetomidine on perioperative hemodynamics in patients undergoing craniotomy. Anesth Analg. 107, 1340–7 (2008). [DOI] [PubMed] [Google Scholar]
- Piao G. & Wu J. Systematic assessment of dexmedetomidine as an anesthetic agent: a meta-analysis of randomized controlled trials. Arch Med Sci. 10, 19–24 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Biccard B. M., Goga S. & de Beurs J. Dexmedetomidine and cardiac protection for non-cardiac surgery: a meta-analysis of randomised controlled trials. Anaesth . 63, 4–14 (2008). [DOI] [PubMed] [Google Scholar]
- Ji F. et al. Perioperative dexmedetomidine improves outcomes of cardiac surgery. Circulation. 127, 1576–84 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ji F., Li Z., Young J. N., Yeranossian A. & Liu H. Post-bypass dexmedetomidine use and postoperative acute kidney injury in patients undergoing cardiac surgery with cardiopulmonary bypass. PLoS One. 8, e77446 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ji F., Li Z., Young N., Moore P. & Liu H. Perioperative dexmedetomidine improves mortality in patients undergoing coronary artery bypass surgery. J Cardiothorac Vasc Anesth. 28, 267–73 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taniguchi T., Kidani Y., Kanakura H., Takemoto Y. & Yamamoto K. Effects of dexmedetomidine on mortality rate and inflammatory responses to endotoxin-induced shock in rats. Critical Care Med . 32, 1322–6 (2004). [DOI] [PubMed] [Google Scholar]
- Yagmurdur H. et al. Dexmedetomidine reduces the ischemia-reperfusion injury markers during upper extremity surgery with tourniquet. J Hand Surgery . 33, 941–7 (2008). [DOI] [PubMed] [Google Scholar]
- Sukegawa S. et al. Locally injected dexmedetomidine inhibits carrageenin-induced inflammatory responses in the injected region. Anesth Analg. 118, 473–80 (2014). [DOI] [PubMed] [Google Scholar]
- Kawasaki T. et al. Dexmedetomidine suppresses proinflammatory mediator production in human whole blood in vitro. J Trauma Acute Care Surg. 74, 1370–5 (2013). [DOI] [PubMed] [Google Scholar]
- Bekker A. et al. The effect of intraoperative infusion of dexmedetomidine on the quality of recovery after major spinal surgery. J Neurosurg Anesthesiol. 25, 16–24 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kang S. H. et al. Effects of dexmedetomidine on inflammatory responses in patients undergoing laparoscopic cholecystectomy. Acta Anaesthesiol Scand. 57, 480–7 (2013). [DOI] [PubMed] [Google Scholar]
- Kim Y. et al. Effects of dexmedetomidine on the ratio of T helper 1 to T helper 2 cytokines in patients undergoing laparoscopic cholecystectomy. J Clin Anesth. 26, 281–5 (2014). [DOI] [PubMed] [Google Scholar]
- Liu H. L. & Qian Y. N. Effects of dexmedetomidine on perioperative inflammatory response in patients undergoing valve replacement. Zhongguo Ying Yong Sheng Li Xue Za Zhi. 29, 316–7 (2013). [PubMed] [Google Scholar]
- Naguib A. N. et al. The role of different anesthetic techniques in altering the stress response during cardiac surgery in children: a prospective, double-blinded, and randomized study. Pediatr Crit Care Med. 14, 481–90 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tasdogan M., Memis D., Sut N. & Yuksel M. Results of a pilot study on the effects of propofol and dexmedetomidine on inflammatory responses and intraabdominal pressure in severe sepsis. J Clin Anesth. 21, 394–400 (2009). [DOI] [PubMed] [Google Scholar]
- Ueki M. et al. The effects of dexmedetomidine on inflammatory mediators after cardiopulmonary bypass. Anaesthesia. 69, 693–700 (2014). [DOI] [PubMed] [Google Scholar]
- Venn R. M. et al. Effects of dexmedetomidine on adrenocortical function, and the cardiovascular, endocrine and inflammatory responses in post-operative patients needing sedation in the intensive care unit. Br J Anaesth. 86, 650–656 (2001). [DOI] [PubMed] [Google Scholar]
- Wang Z. X. et al. Dexmedetomidine reduces intestinal and hepatic injury after hepatectomy with inflow occlusion under general anaesthesia: a randomized controlled trial. Br J Anaesth. 112, 1055–64 (2014). [DOI] [PubMed] [Google Scholar]
- Yacout A. G., Osman H. A., Abdel-Daem M. H., Hammouda S. A. & Elsawy M. M. Effect of intravenous dexmedetomidine infusion on some proinflammatory cytokines, stress hormones and recovery profile in major abdominal surgery. Alexandria J Med. 48, 3–8 (2012). [Google Scholar]
- Zhang J. & Zhang W. Effects of dexmedetomidine on inflammatory responses in patients undergoing cardiac valve replacement with cardiopulmonary bypass. Chin J Anesthesiol . 33, 1188–91 (2013). [Google Scholar]
- Zhang L. et al. Effect of dexmedetomidine on inflammatory cytokines of patients undergoing cerebral surgery during perioperative period. Chin J Nosocomiol . 22, 2752–54 (2012). [Google Scholar]
- Zhang W., Zhang J. & Meng F. Effect of dexmedetomidine combined with continuous positive airway pressure on oxidative stress and inflammatory responses during one-lung ventilation in elderly patients undergoing radical surgery for esophageal carcinoma. Chin J Anesthesiol . 34, 5–8 (2014). [Google Scholar]
- Zhong Z., He D., Zhang L., Min S. & Shen J. Effect of sedation by infusion of dexmedetomidine on inflammatory factors in patients with severe trauma. Chin J Pathophysiol. 28, 1981–8 (2012). [Google Scholar]
- Zhu H. Effects of dexmedetomidine on intrapulmonary shunt and inflammatory response in patients. Chin J Postgrad Med. 35, 29–32 (2012). [Google Scholar]
- Higgins J. P. et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ . 343, d5928 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Higgins J. P. & Green S. editors. Cochrane Handbook for Systematic Reviews of Interventions Version 5.0.2. London: : Cochrane Collaboration, (2009). [Google Scholar]
- Hozo S. P. et al. Estimating mean and variance from median, range and the size of a sample. BMC Res Methodol . 5, 13 (2005). [DOI] [PMC free article] [PubMed] [Google Scholar]
- McBride W. T., Armstrong M. A. & McBride S. J. Immunomodulation: an important concept in modern anaesthesia. Anaesthesia. 51, 465–73 (1996). [DOI] [PubMed] [Google Scholar]
- Cohen D., Horiuchi K., Kemper M. & Weisman C. Modulating effects of propofol on metabolic and cardiopulmonary responses to stressful intensive care unit procedures. Crit Care Med. 24, 612–7 (1996). [DOI] [PubMed] [Google Scholar]
- Sheeran P. & Hall G. M. Cytokines in anesthesia. BJA . 78, 201–19 (1997). [DOI] [PubMed] [Google Scholar]
- Jameson P., Desborough J. P., Bryant A. E. & Hall G. M. The effect of cortisol suppression on IL-6 and white blood cell responses to surgery. Acta Anaesthiol Scand . 41, 304–8 (1997). [DOI] [PubMed] [Google Scholar]
- Clark I. A. How TNF was recognized as a key mechanism of disease. Cytokine Growth Factor Rev. 18, 335–43 (2007). [DOI] [PubMed] [Google Scholar]
- Olmos G. & Llado J. Tumor necrosis factor alpha: A link between neuroinflammation and excitotoxicity. Mediators Inflamm. 2014, 861231 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lotti F. & Maggi M. Interleukin 8 and the male genital tract. J Reprod Immunol. 100, 54–65 (2013). [DOI] [PubMed] [Google Scholar]
- Todorovic-Rakovic N. & Milovanovic J. Interleukin-8 in breast cancer progression. J Interferon Cytokine Res. 33, 563–70 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- McAfoose J. & Baune B. T. Evidence for a cytokine model of cognitive function. Neurosci Biobehav Rev. 33, 355–66 (2009). [DOI] [PubMed] [Google Scholar]
- Dantzer R., O’Connor J. C., Freund G. G., Johnson R. W. & Kelley K. W. From inflammation to sickness and depression: when the immune system subjugates the brain. NatRev Neurosci . 9, 46–56 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koca U. et al. The effects of dexmedetomidine on secondary acute lung and kidney injuries in the rat model of intra-abdominal sepsis. Sci World J . 2013, 292687 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tan J. A. & Ho K. M. Use of dexmedetomidine as a sedative and analgesic agent in critically ill adult patients: a meta-analysis. Intensive Care Med. 36, 926–39 (2010). [DOI] [PubMed] [Google Scholar]
- Xia Z. Q. et al. Clinical benefits of dexmedetomidine versus propofol in adult intensive care unit patients: a meta-analysis of randomized clinical trials. J Surg Res. 185, 833–43 (2013). [DOI] [PubMed] [Google Scholar]
- Pasin L. et al. Dexmedetomidine as a sedative agent in critically ill patients: a meta-analysis of randomized controlled trials. PLoS One. 8, e82913 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hofer S. et al., Central sympatholytics prolong survival in experimental sepsis. Crit Care. 13, R11 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kurosawa S. & Kato M. Anesthetics, immune cells, and immune responses. J Anesth. 22, 263–77 (2008). [DOI] [PubMed] [Google Scholar]
- Najera-Medina O. et al. Cytokine patterns in paediatric patients presenting serious gastrointestinal and respiratory bacterial infections. Centr Eur J Immunol. 39, 223–227 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaack M. B., Harrison R. M. & Roberts J. A. Effect of age and hormonal state on cytokine synthesis in the monkey. Cytokine. 10, 236–239 (1998). [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.