Abstract
Blastic plasmacytoid dendritic cell neoplasm (BPDCN) is a rare tumor that usually arises in the skin. Most patients develop skin lesions, which may be isolated and subsequently spread to affect the whole body. The prognosis is poor. Although BPDCN is usually treated by chemotherapy, radiation therapy is used in some cases (e.g., isolated lesions, elderly patients, or patients with comorbidities). The overall therapeutic efficacy and dose of radiation therapy remain unknown. We herein present a case of successful radiation treatment for BPDCN in a 77-year-old Japanese patient and describe the results of the first literature review on BPDCN of the skin initially treated with radiation therapy. The patient developed a raised, tender, pruritic skin lesion on his left forearm and similar multiple disseminated lesions on his right shoulder and precordial region. Histological and immunohistochemical examination of biopsy specimens of the skin lesions on the left forearm and right shoulder established a diagnosis of BPDCN. The other multiple skin lesions were considered to be disseminated BPDCN. The patient declined chemotherapy and instead elected radiation therapy for the lesion on his left forearm (total of 30 Gy). The skin lesion regressed until only pigmentation was present on the forearm, but the untreated lesions rapidly expanded. Although BPDCN of the skin may respond favorably to initial radiation therapy, it tends to recur in the short term. The optimal dose of radiation therapy remains unclear. Our findings provide a baseline for future research.
Keywords: Dendritic cells, radiation, 18F-fluorodeoxyglucose-positron emission tomography/computed tomography (FDG-PET/CT), skin neoplasms
Introduction
Blastic plasmacytoid dendritic cell neoplasm (BPDCN) is a hematopoietic precursor cell tumor that is derived from plasmacytoid dendritic cells (PDCs) and develops mainly in the skin. The BPDCN disease concept was established by the revised World Health Organization classification in 2008 [1]. BPDCN is very rare and reportedly occurs at an incidence of 0.76% vs. acute myelogenous leukemia and 0.27% vs. non-Hodgkin’s lymphoma [2]. Early symptoms include skin lesions, which are present in more than 90% of patients, along with nodules, erythematous plaques, and bruise-like areas. The skin lesions may begin as isolated lesions, which then multiply and spread to the entire body as in disseminated disease. BPDCN is diagnosed by its blastic or plasmacytoid morphology and PDC-restricted expression of CD4, CD56, and CD123. No standard therapy has yet been established. BPDCN is considered to be a hematologic disorder and is often treated by chemotherapy. However, radiation therapy is used in patients with isolated skin lesions, those who cannot be treated with chemotherapy because of advanced age or complications, and those with recurrent disease after chemotherapy. We recently administered radiation therapy to relieve the symptoms associated with a skin lesion in an elderly patient with BPDCN who refused chemotherapy. The radiation therapy achieved a therapeutic response. Overall, however, the therapeutic efficacy of radiation therapy and its optimal dose remain unclear in patients with BPDCN. Therefore, we reviewed the relevant literature and summarized previous case reports in which radiation therapy was administered as the initial treatment for skin lesions in patients with BPDCN to assess the therapeutic efficacy of radiation therapy in such patients.
Case report
A 77-year-old Japanese man developed a skin lesion on his left forearm. The lesion rapidly expanded for 2 months, at which point he visited a doctor. The initial examination revealed a dome-like, tender, dark purple, firm mass measuring 50 × 45 mm on the left forearm (Figure 1). Additionally, multiple pale dark skin eruptions with infiltration were present on the right shoulder and precordial region, and the lesions were accompanied by pruritus. Superficial lymph nodes were not palpable. Biopsy specimens were obtained from the skin lesions on the left forearm and right shoulder. Histological examination of both biopsied specimens showed proliferation of medium to large atypical tumor cells with lymphocytic infiltration (Figure 2A and 2C). Immunohistochemical staining showed that the atypical cells were diffusely and strongly positive for CD4, CD56, and CD123 and negative for CD20, MPO, CD1a, and S-100 protein (Figure 2B). In situ hybridization for the Epstein-Barr encoding region was negative. Based on these findings, both lesions were diagnosed as BPDCN. 18F-Fluorodeoxyglucose-positron emission tomography/computed tomography (FDG-PET/CT) revealed significant FDG accumulation in the skin lesion on the left forearm at a maximum standardized uptake value (SUVmax) of 3.5 (Figure 3). No abnormal accumulation was observed in the other skin lesions or lymph nodes. Hematologic testing revealed a normal level of soluble interleukin-2 receptor (sIL-2R) and no abnormalities in any other parameters. Bone marrow biopsy showed no atypical cells, and chromosome testing showed no abnormalities. Because BPDCN was identified in two separate skin lesions, the other multiple skin eruptions were considered to be disseminated BPDCN. Therefore, chemotherapy was recommended to the patient, but he declined because of its unknown efficacy. However, the skin lesion on the left forearm was large enough to significantly deteriorate the patient’s quality of life, and radiation therapy was chosen for local containment. The skin lesion was a tumorous mass arising from the skin surface and extended only 8 mm below the skin surface as demonstrated by FDG-PET/CT. Thus, the skin surface was irradiated with electron beams with energy of 5 MeV and bolus thickness of 5 mm. The total radiation dose was 30 Gy at 3 Gy per fraction. The lesion was flattened and the tenderness resolved during irradiation, and the therapeutic response after completion of irradiation was determined to be a partial response. No adverse reactions associated with the radiation therapy occurred. However, the other skin lesions in untreated areas, such as in the precordial region, rapidly expanded even during the radiation therapy. Three months after completion of the radiation therapy, the irradiated site exhibited only pigmentation.
Figure 1.
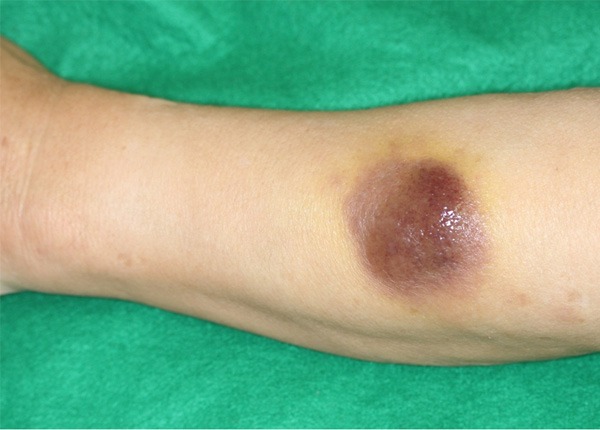
Initial examination of firm mass on left forearm. A large, violet-colored, tender nodule was present on the left forearm.
Figure 2.
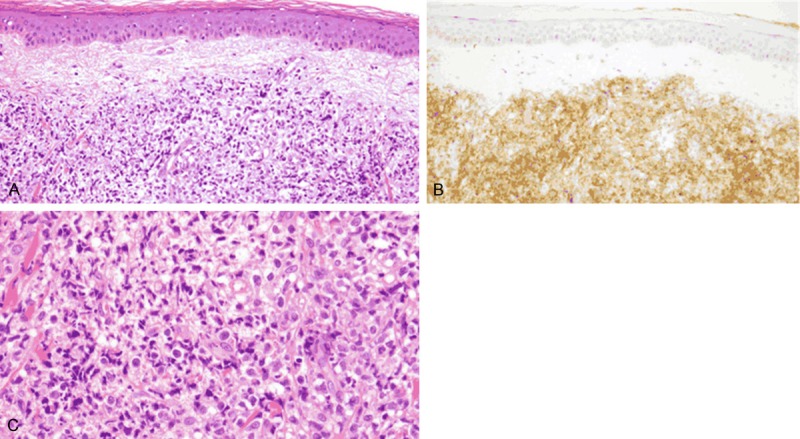
Histological examination of biopsied specimens from left forearm and right shoulder. A, B. Hematoxylin and eosin staining and immunostaining using anti-CD123 antibody revealed proliferation of atypical cells within the dermis of the skin lesion (× 200, both). Positive staining of CD123 was found in the tumor. C. High-power view of tumor area (× 400, hematoxylin and eosin).
Figure 3.
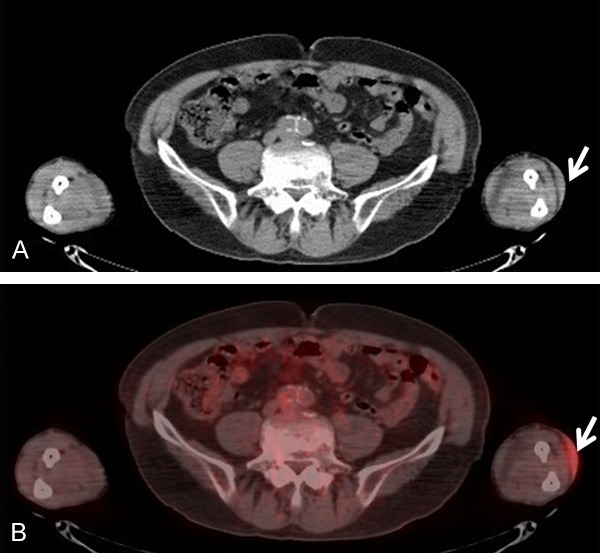
18F-Fluorodeoxyglucose-positron emission tomography/computed tomography. Accumulation was present in the skin of the left forearm (arrows). The maximum standardized uptake value was 3.5.
Discussion
BPDCN has a poor prognosis. The mean survival time of patients with BPDCN is reportedly 14.0 to 16.7 months, and the 2- and 5-year overall survival rates are 17% to 33% and 2% to 6%, respectively [3,4]. Bone marrow infiltration is absent in 70% of patients at the initial examination, but lymph node and bone marrow infiltration occurs during the disease course in 72% of patients, resulting in leukemic change and eventual death in many cases [2,3,6]. As evidenced by the fact that BPDCN was historically classified as blastic NK-cell lymphoma [7], this disease is often treated with chemotherapy; i.e., CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisone) and DeVIC (carboplatin, etoposide, ifosfamide, and dexamethasone) as in the treatment of malignant lymphomas, or with chemotherapy as in the treatment of acute myeloid leukemia and acute lymphocytic leukemia [4,8,9]. Furthermore, Dalle et al. [4] reported that bone marrow transplantation was the only option that could improve the outcome. BPDCN blasts overexpress the interleukin-3 receptor, and a new targeted therapy directed against this receptor was reported in a recent prospective study [5].
However, patients with BPDCN are often at an advanced age and are not suitable candidates for chemotherapy or bone marrow transplantation. In these patients, radiation therapy is often used for local containment. Because lymphocytes are highly sensitive to radiation, BPDCN is expected to be contained locally by radiation therapy, as is the case with malignant lymphoma. However, the therapeutic efficacy of radiation therapy and its optimal dose for BPDCN remain unclear because no standard therapy has been established for this disease.
We performed the first review of the relevant literature with a focus on radiation therapy for BPDCN. We identified 9 articles including 22 patients with BPDCN-related skin lesions for which radiation therapy was administered as the initial treatment. Our own is the 23rd case (Table 1). The patients’ median age was 72 years (range, 36-90 years); 16 patients were male and 7 were female. In general, the mean age of patients with BPDCN is 61 to 67 years, and the male: female ratio is 3.3:1.0 [1]. A similar distribution was seen in the 23 patients in the present study. Fourteen patients (60.9%), including ours, were treated with radiation therapy alone, and the remaining nine patients received radiation therapy in combination with various chemotherapies, although the specific regimen was not reported in some cases. The radiation dose was reported for only three patients (27, 40, and 51 Gy, respectively). In our case, a total of 30 Gy was irradiated at 3 Gy per fraction, and the patient exhibited a partial response to the initial treatment. In past reports, the initial response was characterized by a complete response in 13 patients (56.5%) and both complete and partial responses in 19 patients (82.6%). However, although BPDCN tends to temporarily respond well to treatment, it tends to recur within a short period of time. This was also the case for most of the 23 patients reported in this study, and disease-free survival was reported in only 4 (19%) of 21 patients who were under follow-up observation. In one report, a patient underwent CHOP followed by total body irradiation rather than local radiation therapy and developed recurrence. He subsequently underwent bone marrow transplantation from a related donor. This patient survived for 30 months, but the report did not provide the radiation dose used in the total body irradiation [15]. FDG-PET/CT findings were reported in only four patients with BPDCN [5,8,16,17]. Additionally, the SUVmax of the skin lesion was available in only one case in which slight FDG accumulation was seen in a skin lesion on the left shoulder, but was considered to be only an inflammatory finding after resection with an SUVmax of 2.4 [8]. In our case, FDG accumulation was observed in the skin lesion on the left forearm and was significant with a SUVmax of 3.5. FDG accumulation was not seen in the other skin lesions on the right shoulder and precordial region, probably because of the small size of these lesions. Nizza et al. [16] reported that FDG-PET/CT was useful to assess the extent of nodal involvement in patients with BPDCN, but FDG accumulation was not noted in bone marrow lesions. Further studies with larger numbers of cases are needed to clarify the optimal dose of radiation for local containment of BPDCN. Pileri et al. [18] described a patient > 75 years of age with an isolated skin lesion that achieved an initial complete response after radiation therapy alone, indicating that radiation therapy may be an option for the treatment of BPDCN in patients older than 70 years. The more precise characterization of BPDCN with new algorithms [19] may identify patients with localized cutaneous disease that has less tendency to expand to other regions of the body and that may respond well to radiation therapy. Our patient should continue to undergo strict observation because he did not undergo whole-body therapy, such as chemotherapy, and began to show worsening of other skin lesions in untreated areas.
Table 1.
Patients’ clinical features and treatment outcomes
| Patient No. | Age | Sex | Skin location | Initial treatment | Radiation therapy total dose | Response | RFS (months) | OS (months) | References |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 82 | M | Forehead | RT + PC | PR or CR | 12 | 24 | [6] | |
| 2 | 62 | M | Adbomen | RT + PC | NR | NA | 13 | [6] | |
| 3 | 60 | M | Scalp | RT | PR or CR | 13 | 22 | [6] | |
| 4 | 43 | F | Generalized | RT + PC | NR | NA | 6 | [6] | |
| 5 | 64 | M | Arm | RT | PR or CR | 31 | 32 | [6] | |
| 6 | 73 | M | Chest | RT | PR or CR | 2 | Alive | [6] | |
| 7 | 60 | F | Face | MTX, L-ASPDEX + RT | 40Gy | CR | 30 | NED | [10] |
| 8 | 36 | M | Trunk and extremities | PSL+RT | CR | 4 | NED | [11] | |
| 9 | 73 | M | Shoulder | RT | PD | NA | 36 | [4] | |
| 10 | 63 | F | Face | RT | CR | 2 | Alive | [4] | |
| 11 | 81 | F | Trunk and leg | RT | CR | 3 | 11 | [4] | |
| 12 | 83 | M | Face | RT | CR | 8 | Alive | [4] | |
| 13 | 82 | M | Scalp | RT | CR | 9 | 19 | [4] | |
| 14 | 60 | F | Face | MTX, L-ASP + RT | CR | 11 | LFU | [4] | |
| 15 | 77 | M | Face | RT | 51Gy | CR | 10 | 25 | [12] |
| 16 | 59 | F | Neck | RT | CR | Relapse | 6 | [9] | |
| 17 | 72 | M | Not written | PSL + RT | PD | NA | 3 | [9] | |
| 18 | 60 | M | Back | RT | CR | 7 | NED | [9] | |
| 19 | 90 | F | Trunk and arms | VP-16 + RT | CR | Relapse | Alive | [9] | |
| 20 | 74 | M | Post auricular | RT | 27Gy | CR | 2 | Dead | [13] |
| 21 | 74 | M | Shoulder | TR + DeVIC + RT | CR | 12 | NED | [8] | |
| 22 | 40 | M | Face and thigh | RT | PR | Unknown | Unknown | [14] | |
| 23 | 77 | M | Arm | RT | 30Gy | PR | Our case |
Abbreviations: RFS = relapse-free survival; OS = overall survival; RT = radiation therapy; PC = polychemotherapy; PR = partial response; CR = complete response; NR = no remission; NA = not available; MTX = methotrexate; L-ASP = l-asparaginase; DEX = dexamethasone; NED = no evidence of disease; PSL = prednisone; PD = progressive disease; LFU = lost to follow-up; VP-16 = etoposide; TR = total resection; DeVIC = carboplatin, etoposide, ifosfamide, and dexamethasone.
In conclusion, a therapeutic response was achieved after radiation therapy of a skin lesion in an elderly patient with BPDCN who refused chemotherapy. Overall, however, the therapeutic efficacy of radiation therapy and its optimal dose remain unclear in patients with BPDCN. Although BPDCN may respond well to radiation therapy, it tends to quickly recur. Further study is required to determine the optimal radiation dose for localized therapy.
Disclosure of conflict of interest
None.
References
- 1.Facchetti F, Jones DM, Petrella T. Blastic plasmacytoid dendritic cell neoplasms. In: Swerdlow SH, Campo E, Harris NL, Pileri SA, Stein H, Thiele J, Vardiman JW, editors. WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues. 4th edition. Lyon: IARC; 2008. pp. 145–7. [Google Scholar]
- 2.Leitenberger JJ, Berthelot CN, Polder KD, Pro B, McLaughlin P, Jones D, Duvic M. CD4+ CD56+ hematodermic/plasmacytoid dendritic cell tumor with response to pralatrexate. J Am Acad Dermatol. 2008;58:480–4. doi: 10.1016/j.jaad.2007.12.012. [DOI] [PubMed] [Google Scholar]
- 3.Bekkenk MW, Jansen PM, Meijer CJ, Willemze R. CD56+ hematological neoplasms presenting in the skin: a retrospective analysis of 23 new cases and 130 cases from the literature. Ann Oncol. 2004;15:1097–108. doi: 10.1093/annonc/mdh268. [DOI] [PubMed] [Google Scholar]
- 4.Dalle S, Beylot-Barry M, Bagot M, Lipsker D, Machet L, Joly P, Dompmartin A, d’Incan M, Maubec E, Grange F, Dereure O, Prey S, Barete S, Wetterwald M, Fraitag S, Petrella T. Blastic plasmacytoid dendritic cell neoplasm: is transplantation the treatment of choice? Br J Dermatol. 2010;162:74–9. doi: 10.1111/j.1365-2133.2009.09373.x. [DOI] [PubMed] [Google Scholar]
- 5.Frankel AE, Woo JH, Ahn C, Pemmaraju N, Medeiros BC, Carraway HE, Frankfurt O, Forman SJ, Yang XA, Konopleva M, Garnache-Ottou F, Angelot-Delettre F, Brooks C, Szarek M, Rowinsky E. Activity of SL-401, a targeted therapy directed to interleukin-3 receptor, in blastic plasmacytoid dendritic cell neoplasm patients. Blood. 2014;124:385–92. doi: 10.1182/blood-2014-04-566737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Petrella T, Bagot M, Willemze R, Beylot-Barry M, Vergier B, Delaunay M, Meijer CJ, Courville P, Joly P, Grange F, De Muret A, Machet L, Dompmartin A, Bosq J, Durlach A, Bernard P, Dalac S, Dechelotte P, D’Incan M, Wechsler J, Teitell MA. Blastic NK-cell lymphomas (agranular CD4+ CD56+ hematodermic neoplasms): a review. Am J Clin Pathol. 2005;123:662–75. [PubMed] [Google Scholar]
- 7.Chan JKC, Jaffe ES, Ralfkiaer E. Blastic NK-cell lymphoma. In: Jaffe ES, Harris NL, Stein H, Vardiman JW, editors. WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues. 3rd edition. Lyon: IARC; 2001. pp. 214–5. [Google Scholar]
- 8.Sugimoto KJ, Shimada A, Yamaguchi N, Imai H, Wakabayashi M, Sekiguchi Y, Izumi H, Ota Y, Komatsu N, Noguchi M. Sustained complete remission of a limited-stage blastic plasmacytoid dendritic cell neoplasm followed by a simultaneous combination of low-dose DeVIC therapy and radiation therapy: a case report and review of the literature. Int J Clin Exp Pathol. 2013;6:2603–8. [PMC free article] [PubMed] [Google Scholar]
- 9.Hashikawa K, Niino D, Yasumoto S, Nakama T, Kiyasu J, Sato K, Kimura Y, Takeuchi M, Sugita Y, Hashimoto T, Ohshima K. Clinicopathological features and prognostic significance of CXCL12 in blastic plasmacytoid dendritic cell neoplasm. J Am Acad Dermatol. 2012;66:278–91. doi: 10.1016/j.jaad.2010.12.043. [DOI] [PubMed] [Google Scholar]
- 10.Fontaine J, Thomas L, Balme B, Ronger-Savle S, Traullé C, Petrella T, Dalle S. Haematodermic CD4+ CD56+ neoplasm: complete remission after methotrexate-asparaginase treatment. Clin Exp Dermatol. 2009;34:e43–5. doi: 10.1111/j.1365-2230.2008.03100.x. [DOI] [PubMed] [Google Scholar]
- 11.Xue R, Wu T, Pan H, Gu Y, Yang B, Chen Y, Qiu J. A case of cutaneous blastic plasmacytoid dendritic cell neoplasm. Acta Derm Venereol. 2010;90:645–6. doi: 10.2340/00015555-0922. [DOI] [PubMed] [Google Scholar]
- 12.Miyashita A, Fujisawa A, Inoue Y, Ihn H. A case of CD4+/CD56+ hematodermic neoplasm treated with electron beam irradiation. Skin Cancer. 2011;26:31–5. [Google Scholar]
- 13.Tsunoda K, Satoh T, Akasaka K, Ishikawa Y, Ishida Y, Masuda T, Akasaka T. Blastic plasmacytoid dendritic cell neoplasm: report of two cases. J Clin Exp Hematop. 2012;52:23–9. doi: 10.3960/jslrt.52.23. [DOI] [PubMed] [Google Scholar]
- 14.An HJ, Yoon DH, Kim S Shin SJ, Huh J, Lee KH, Suh C. Blastic plasmacytoid dendritic cell neoplasm: a single-center experience. Ann Hematol. 2013;92:351–6. doi: 10.1007/s00277-012-1614-z. [DOI] [PubMed] [Google Scholar]
- 15.Pilichowska ME, Fleming MD, Pinkus JL, Pinkus GS. CD4+/CD56+ hematodermic neoplasm (“blastic natural killer cell lymphoma”): neoplastic cells express the immature dendritic cell marker BDCA-2 and produce interferon. Am J Clin Pathol. 2007;128:445–53. doi: 10.1309/W9Q5AGYDE5LANN39. [DOI] [PubMed] [Google Scholar]
- 16.Nizza D, Simoneaux SF. Blastic plasmacytoid dendritic cell neoplasm presenting as a subcutaneous mass in an 8-year-old boy. Pediatr Radiol. 2010;40:S40–2. doi: 10.1007/s00247-010-1731-6. [DOI] [PubMed] [Google Scholar]
- 17.Matsuo T, Ichimura K, Tanaka T, Morizane S, Iwatsuki K, Eguchi M, Yoshino T. Bilateral conjunctival lesions in blastic plasmacytoid dendritic cell neoplasm. J Clin Exp Hematop. 2011;51:49–55. doi: 10.3960/jslrt.51.49. [DOI] [PubMed] [Google Scholar]
- 18.Pileri A, Delfino C, Grandi V, Agostinelli C, Pileri SA, Pimpinelli N. Blastic plasmacytoid dendritic cell neoplasm (BPDCN): the cutaneous sanctuary. G Ital Dermatol Venereol. 2012;147:603–8. [PubMed] [Google Scholar]
- 19.Tsagarakis NJ, Kentrou NA, Papadimitriou KA, Pagoni M, Kokkini G, Papadaki H, Pappa V, Marinakis T, Anagnostopoulos NI, Vadikolia C, Anagnostopoulos A, Angelopoulou MK, Terpos E, Poziopoulos C, Anargyrou K, Rontogianni D, Papadaki T, Psarra A, Kontopidou FN, Skoumi D, Papadhimitriou SI, Paterakis G Hellenic Dendritic Cell Leukemia Study Group. Acute lymphoplasmacytoid dendritic cell (DC2) leukemia: results from the Hellenic Dendritic Cell Leukemia Study Group. Leuk Res. 2010;34:438–46. doi: 10.1016/j.leukres.2009.09.006. [DOI] [PubMed] [Google Scholar]


