Abstract
Objective
To characterize cellular infiltrates in muscle biopsies from patients with anti-3-hydroxy-3-methyl-gulatryl-CoA reductase (HMGCR)-associated myopathy.
Methods
Biopsies from 18 anti-HMGCR myopathy and 7 control dermatomyositis patients were analyzed.
Results
CD4+ and CD8+ T-cells were scattered within the endomysium in 50% of anti-HMGCR biopsies. All anti-HMGCR biopsies included increased endomysial and/or perivascular CD163+ M2 macrophages; CD11c+ M1 macrophages were present in 18.8%. CD123+ plasmacytoid dendritic (PD) cells were observed within the endomysium and perivascular spaces in 62.5% of anti-HMGCR biopsies. Membrane attack complex was deposited on endothelial cells in 50% and on the sarcolemma of non-necrotic muscle fibers in 85.7% of anti-HMGCR cases. Major histocompatibility complex class I antigen was up-regulated in 87.5% of the anti-HMGCR cases.
Conclusion
In addition to necrosis, scattered CD4+, CD8+, and PD cells are characteristic of anti-HMGCR myopathy. Predominant M2 polarization suggests infiltrating macrophages are more likely to be involved with tissue repair than destruction.
Keywords: Autoimmune myopathy, immune-mediated necrotizing myopathy, muscle histology, myositis, anti-HMGCR myopathy
Introduction
The idiopathic inflammatory myopathies (IIMs), including polymyositis (PM) and dermatomyositis (DM), are a family of autoimmune diseases characterized by proximal muscle weakness, elevated muscle enzymes, autoantibodies, and responsiveness to immunosuppressive medications. 1, 2 Histologically, PM muscle biopsies are notable for the presence of CD8+ T-cells surrounding and invading non-necrotic muscle fibers within the endomysium (i.e., primary inflammation). 3 In contrast, DM muscle biopsies typically include a larger proportion of CD4+ T-cells and plasmacytoid dendritic cells (PDCs), often in a perimysial and/or perivascular distribution. 4
While lymphocytic infiltrates within muscle tissue are found in most IIM patients, recent studies have shown that some patients with autoimmune myopathy have prominent myofiber necrosis with abundant macrophages, but only minimal lymphocytic infiltration. This unique form of myositis has been termed immune-mediated necrotizing myopathy (IMNM). 3
We recently screened a cohort of IMNM patients for novel autoantibodies and discovered that some had antibodies which recognize HMG-CoA reductase, the pharmacologic target of statin medications. 5, 6 Interestingly, the majority of anti-HMGCR positive myopathy subjects developed an autoimmune myopathy following statin exposure.
In our initial description of muscle biopsies from anti-HMGCR positive subjects, we found that while most subjects had histological profiles consistent with IMNM, 11% of statin exposed and ∼39% of statin-naïve subjects had inflammatory cell infiltrates on routine histological staining, mostly in a perivascular distribution. 5, 6 However, the cellular composition of these infiltrates has not been described previously. Here, we provide a detailed analysis of the cellular composition of infiltrates in muscle biopsy specimens from patients with anti-HMGCR positive myopathy.
Materials and Methods
Patient populations
All consecutive patients seen at the Johns Hopkins Myositis Center between 2003 and 2011 were included in this study if they had: (1) anti-HMGCR positive myopathy based on a positive immunoprecipitation as described previously, 6 or definite DM based on ENMC criteria 3 and (2) frozen muscle tissue available from a biopsy performed for clinical purposes at Johns Hopkins. We included DM as a control group in this study for 2 reasons: (1) to demonstrate that our immunohistochemical methods work in a population where the composition and distribution of inflammatory cells was already well-characterized and (2) to compare the composition and distribution of inflammatory cells in anti-HMGCR myopathy with what is seen in another autoimmune muscle disease. Muscle biopsies from DM patients fulfilled both criteria. The study was approved by the Johns Hopkins Institutional Review Board, was compliant with the Health Insurance Portability and Accountability Act, and all participants signed informed consent.
Immunohistochemistry
Briefly, 7 μ-thick frozen muscle biopsy sections were washed with Tris buffer at pH7.2 and incubated with primary antibody (Table 1) for 1 hour in a humidified chamber at room temperature. After washing with Tris buffer twice, the slides were incubated with HRP-conjugated secondary antibodies (Dako, P0447) at 1:100 dilution for 30 minutes in a dark humidified chamber at room temperature. After the secondary incubation, the slides were washed 3 times with Tris buffer and incubated with DAB chromogen (DAKO, K3468) for 30 minutes. The slides were then counterstained with Gill hematoxylin for 30 seconds.
Table 1.
Primary antibodies and dilutions used in this study.
| Antigen | Primary antibody | Dilution |
|---|---|---|
| CD3 (pan T cell marker) | DAKO M7254 (F7.2.38) | 1:60 |
| CD4 (helper T cell) | DAKO M7310 (4B12) | 1:40 |
| CD8 (cytotoxic T cell) | DAKO M7103 (C8/144B) | 1:100 |
| CD20 (B cell) | DAKO M0755 (L26) | 1:200 |
| CD68 (macrophage) | DAKO M0718 (EBM11) | 1:100 |
| CD11c (M1 marker) | Abcam Ab-53441 | 1:100 |
| CD163 (M2 marker) | Santa Cruz SC-20066 | 1:100 |
| CD123 (PDC marker) | Novus NB-600-1185 | 1:100 |
Normal human lymph node, spleen, and tonsil tissues were used as positive controls for each surface marker. A case of normal human muscle tissue was used as negative control. To distinguish a non-specific primary antibody reaction, we included a “no primary” control for each case. For each primary antibody, all muscle sections were processed simultaneously under the same conditions. The details of each antibody and the dilutions used are summarized in Table 1.
Pathological definitions
Primary inflammation was defined as the presence of at least 1 non-necrotic myofiber surrounded and invaded by lymphocytes. Necrotizing myopathy was defined as the presence of myofiber necrosis with little or no lymphocytic inflammation on routine histological staining (H&E).
After staining for cell subtypes, the extent of infiltration was assessed semi-quantitatively by counting the number of positively stained cells per high power (200×) field. For each case, at least 4 fields were examined, and the average scores were used for final analysis. We defined “positive staining” of cell surface markers to be when the brownish DAB chromagen completely encircled the cell surface. The scoring was done as follows: 0 = absence of staining in the entire biopsy; 1 = 1-10 cells stained per high power field; 2 = 10-50 cells per high power field; 3 = more than 50 cells per high power field. Biopsy regions were divided into endomysial, perimysial, and perivascular areas, and each area was scored separately.
Results
Clinical characteristics of HMGCR and DM myopathy patients
Muscle biopsy samples from 18 patients with anti-HMGCR-positive myopathy and 7 patients with DM were available for immunohistological analysis. The demographics and clinical characteristics of the anti-HMGCR positive patients were similar to what we have previously reported (Supplementary table S1, available online). 5, 6 Twelve (66%) women and 6 (33%) men with an average age of 56 years were included. Sixteen of 18 (88.9%) anti-HMGCR-positive patients had a history of statin exposure prior to the onset of weakness. Although all patients had proximal muscle weakness, the severity ranged from mild in ambulatory patients to severe in a patient who was hospitalized with respiratory failure and had the highest recorded creatine kinase (CK) level of 9,551 IU/L. The median disease duration at the time of biopsy was 13 months; 6 of 18 (33%) were treated with immunosuppressive medications at the time of biopsy. Two anti-HMGCR patients had cancer; 1 was diagnosed and treated for ovarian cancer 5 years prior to the onset of weakness, and the other was diagnosed with cholangiocarcinoma 2 years after muscle biopsy and died shortly afterwards.
All DM patients were women, and their average age was 54 years (Supplementary table S1, available online). Five of 7 (71%) DM patients had anti-synthetase antibodies, and 2 had interstitial lung disease. All DM patients had proximal muscle weakness with a mean CK of 3,360 IU/L. Only 1 (14%) DM patient had a history of statin exposure prior to disease onset. The median disease duration before biopsy was 8 months, and all but 1 patient was taking immunosuppressive medications at the time of biopsy. Two cases were associated with cancer.
Routine histology
In all cases of HMGCR myopathy, H&E staining showed myofibers undergoing degeneration, necrosis, and regeneration (Figure 1A). Most cases revealed diffuse rounded and polygonal atrophic myofibers and an increased number of myofibers with internalized nuclei. In anti-HMGCR myopathy, macrophages frequently replaced necrotizing myofibers (myophagocytosis). There was no perifascicular or perimyisal preference for necrotic or atrophic fibers in anti-HMGCR myopathy. In contrast, most inflammatory cells and atrophic myofibers were found in the perifascicular and perimysial areas, respectively, in DM patients (Figure 1B). Perivascular lymphocytes were noted in 6 of 18 (33.33%) and 4 of 7 (57.14%) cases of anti-HMGCR myopathy and DM, respectively. Of note, no example of primary inflammation was found in any anti-HMGCR muscle biopsy.
Figure 1.

Representative serial muscle biopsies sections from anti-HMGCR myopathy (A, C, E, G, I, K) and DM (B, D, F, H, J, L) patients were stained with H&E (A, B), anti-CD3 (C, D), CD4 (E, F), CD8 (G, H), CD20 (I, J), and CD68 (K, L). In the anti-HMGCR case, H&E staining reveals degeneration, necrosis, and myophagocytosis of endomysial myofibers. IHC staining shows sparse endomysial CD3, CD4, and CD8 cells with no CD20 positive cells. In anti-HMGCR myopathy, most inflammatory infiltrates are CD68-positive macrophages. In the DM case, H&E staining shows myofiber atrophy, myofiber degeneration, and inflammatory infiltrates. Inflammatory infiltrates from the DM case include numerous CD68 positive macrophages with similar numbers of CD3, CD4, and CD8 cells and a few scattered CD20 cells.
Semi-quantitative analysis of cellular infiltrates
To further characterize the infiltrating inflammatory cells, we stained muscle biopsy sections for the following cell surface markers: CD3 (T-cells), CD4 (helper T-cells), CD8 (cyotoxic T-cells), CD20 (B-cells), CD68 (macrophages), CD11c (M1 macrophages), and CD163 (M2 macrophages). The degree of cellular infiltration was assessed semi-quantitatively using the scoring system described in Methods, and the results are summarized in Table 2 (Supplementary table S2, available online).
Table 2.
Distribution of inflammatory cell subsets within muscle biopsy specimens of anti-HMGCR myopathy and DM patients.
| CD3 | CD4 | CD8 | CD20 | CD68 | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| E | PV | PM | E | PV | PM | E | PV | PM | E | PV | PM | E | PV | PM | |
| HMGCR | 0.67 | 0.18 | 0 | 0.56 | 0.18 | 0 | 0.5 | 0.06 | 0 | 0.17 | 0 | 0 | 1.72 | 0.47 | 0 |
| DM | 0.57 | 1 | 1.14 | 0.43 | 0.86 | 0.71 | 0.29 | 0.43 | 0.57 | 0.14 | 0.71 | 0.57 | 1 | 0.86 | 1.29 |
The biopsy specimens were divided into endomysial (E), perivascular (PV), and perimysial (PM) areas; individual detailed scoring is described in supplementary table S1 (available online). The mean values for each area are recorded in this table.
As expected, CD68+ macrophages were the most prevalent inflammatory cells in biopsies from anti-HMGCR myopathy subjects; they were located in both endomysial and perivascular areas (Figure 1K). In contrast, macrophages preferentially infiltrated the perimysial areas in DM muscle biopsies (Figure 1L). Furthermore, we stained the macrophages with CD163, a cell surface marker for M2 macrophages, which are thought to have a predominant “anti-inflammatory” role and promote tissue recovery, and CD11C, a cell surface marker for M1 macrophages, which are thought to be “pro-inflammatory” and play a role in mediating tissue damage. 7-9 Staining of serial sections for CD163 revealed that most of the macrophages in both anti-HMGCR myopathy (as seen in Figure 2A and 2B) and DM were of the M2 subclass (Table 3, also refer supplementary table S3, available online). Small numbers of CD11c+ M1 macrophages were found in 3 of 16 (19%) anti-HMGCR-positive subjects, each of whom also had abundant infiltration with M2 macrophages. In 2 cases, M1 macrophages were present in very small numbers only. However, a single anti-HMGCR positive case showed more, but still very modest, numbers of M1 macrophages in both perivascular and endomysial areas (Figure 2F and 2H). Interestingly, this patient died from complications of cholangiocarcinoma shortly after the biopsy was obtained.
Figure 2.

Representative serial sections from 2 typical anti-HMGCR myopathy patients (A,B and C,D) show significant staining for M2 macrophages with anti-CD163 (A,C) but no M1 macrophages staining positive for CD11c (B,D). In contrast, sections from a single patient who was later diagnosed with cholangiocarcinoma had a modestly increased number of CD11c positive M1 macrophages (F,H) in addition to large numbers of M2 macrophages which stain positively for CD163 (E,G).
Table 3.
The distribution of macrophage subsets and plasmacytoid dendritic cells in anti-HMGCR myopathy and DM patients.
| CD68 | CD163 | CD11c | CD123 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| E | PV | PM | E | PV | PM | E | PV | PM | E | PV | PM | |
| HMGCR | 1.94 | 1.69 | 0.00 | 1.94 | 1.56 | 0.00 | 0.06 | 0.19 | 0.00 | 1.23 | 0.85 | 0.00 |
| DM | 1.14 | 2.00 | 2.57 | 1.00 | 1.86 | 2.29 | 0.00 | 0.00 | 0.00 | 0.71 | 1.00 | 1.43 |
The biopsy specimens were divided into endomysial (E), perivascular (PV), and perimysial (PM) areas; the individual detailed scoring is described in supplementary table S2 (available online). The mean values for each area are recorded in this table
Staining for lymphocyte subsets revealed scattered CD4+ and CD8+ T-cells in the endomysial regions of 9 of 18 (50%) anti-HMGCR positive subjects (Figure 1E and 1G). These lymphocytes, though not easily detectable on H&E staining, were increased in numbers compared to normal controls when studied by immunohistochemistry. In a minority of anti-HMGCR myopathy patients, CD4+ and/or CD8+ cells were also present in perivascular regions. No non-necrotic muscle fibers were surrounded and invaded by CD8+ cells. A few (17%) anti-HMGCR subjects had small numbers of CD20+ cells, which were restricted to the endoymysium. In contrast, 6 of 7 (86%) DM subjects had CD20+ cells, predominantly in perivascular and/or perimysial spaces (Figure 1J).
MHC class I expression and membrane attack complex (MAC) deposition
Fourteen of 16 (88%) cases of HMGCR myopathy revealed prominent up-regulation of MHC class I protein on the sarcolemma surface of non-necrotic fibers. These fibers were scattered diffusely throughout the endomysium. In contrast, DM muscle biopsies showed MHC class I positive myofibers in perimysial areas, even when perifascicular atrophy was not evident.
In anti-HMGCR myopathy, MAC deposition was noted on endomysial capillaries in 7 of 16 cases (44%) and on the surface of non-necrotic muscle fibers in 14 of 16 (87%) cases. In DM, MAC deposition occurred on endomysial capillaries in 67%. However, only 1 of 6 (16.7%) DM cases revealed MAC deposition on non-necrotic muscle fibers (Table 4).
Table 4.
C5-9b and MHC I deposition in anti-HMGCR myopathy and DM.
| C5-9b deposition | MHC I | ||
|---|---|---|---|
| HMGCR Patient | Endothelial | Sacrolemmal | |
| 1 | Y | Y | Y |
| 2 | N | N | N |
| 3 | N | Y | Y |
| 4 | N | Y | Y |
| 5 | N | Y | Y |
| 6 | Y | Y | Y |
| 7 | N | Y | Y |
| 8 | N | Y | Y |
| 10 | N | N | Y |
| 11 | Y | Y | Y |
| 12 | N | Y | Y |
| 13 | N | N/A | N |
| 14 | Y | Y | Y |
| 15 | Y | Y | Y |
| 17 | N | N/A | Y |
| 18 | Y | Y | Y |
| DM Patient | |||
| 1 | Y | N | Y |
| 2 | N | N | Y |
| 3 | Y | N | Y |
| 4 | N | Y | Y |
| 5 | Y | N | Y |
| 6 | N/A | N/A | N/A |
| 7 | Y | N | Y |
“Y” = at least 1 endomysial capillary or myofiber sarcolemma staining positive for C5-9b. “N” = absence of such staining in the entire biopsy sample. “N/A” = staining technically inadequate for interpretation.
Plasmacytoid dendritic cell infiltration is common in anti-HMGCR myopathy
Greenberg el al. previously reported that PDCs are found in DM muscle biopsies, suggesting a role of innate immunity in the pathogenesis of this disease. 4 In our study, we noted CD123+ cells in 10 of 16 (63%) anti-HMGCR myopathy and 7 of 7 (100%) DM subjects. Consistent with the previous finding by Greenberg et al., PDC cells were prominent in the perimysial areas of DM biopsies. In contrast, PDCs in anti-HMGCR myopathy were restricted to the endomysium and perivascular areas and were absent within the perimysium (Figure 3A and 3B).
Figure 3.
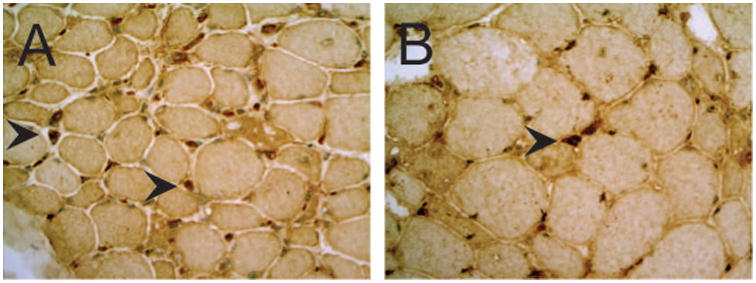
Immunostaining reveals a modest number of CD123 positive plasmacytoid dendritic cell cells (arrowheads) within the endomysium of 2 different anti-HMGCR myopathy patients (A and B). Note that both inflammatory cells (arrowheads) and myonuclei are stained with anti-CD123.
Disussion
We and others have reported that patients with anti-HMGCR-associated myopathy have a necrotizing myopathy with marked myofiber degeneration, abundant macrophages, and minimal lymphocytic infiltrate. 5, 6, 10 Although we observed perivascular inflammation in a minority of anti-HMGCR positive patients, the composition of these and other inflammatory cells has not been analyzed previously. Here we report the results of immunohistochemistry analysis of muscle biopsies from anti-HMGCR myopathy patients and a disease control group of DM subjects using a variety of cell surface markers to define inflammatory cell subtypes and their distribution.
The data confirm that macrophages are the predominant infiltrating cell type in patients with anti-HMGCR myopathy. Furthermore, we found that these macrophages are predominantly CD163+, suggesting they are mostly of the M2 phenotype; this population of macrophages is thought to play an important role in muscle regeneration and repair. 7-9 In contrast, less than 20% of anti-HMGCR cases included CD11c+ macrophages, which likely represent M1, or so-called “killer” macrophages that secrete tissue-damaging nitric oxide (NO). 7 It should be noted, however, that using cell surface markers to define M1 and M2 macrophages has certain limitations. Macrophages found in vivo may lie within a spectrum of activation states rather than at an extreme of polarization. 11 Thus, it may be inappropriate to classify these cells into narrow subgroups based on surface marker expression.
Interestingly, the patient who had relatively large numbers of M1 macrophages was the only patient studied who had cancer-associated myositis (defined as malignancy occurring 3 years before or after myositis onset). Although this patient had a negative PET CT scan within a month of the biopsy, he was diagnosed with cholangiocarcinoma 2 years later. Since M1 macrophages are thought to play a role in tumor suppression, 12 we speculate that their presence at the time of biopsy could have been related to a subclinical malignancy that was effectively, but only temporarily, kept in check by an antitumor immune response.
Although the presence of PDCs in polymyositis and dermatomyositis muscle tissue is well-described, 4, 13 this study shows that these potent secretors of interferon are also common in an IMNM. In contrast to DM muscle, where PDCs are located preferentially in perifascicular and perimysial areas, PDCs are scattered diffusely throughout the endomysium in anti-HMGCR myopathy biopsies. This suggests the possibility that interferon may play a role in the immunopathogenesis of HMGCR myopathy. Future studies will be required to determine whether circulating levels of interferon correlate with disease activity in anti-HMGCR myopathy, as reported in patients with DM and PM. 14, 15
Dermatomyositis muscle biopsies are known to include relatively large numbers of infiltrating CD20+ B-cells, predominantly in a perivascular distribution within the perimysium. 16 We confirmed this finding in our DM control biopsy specimens. In contrast, less than 20% of anti-HMGCR biopsies had infiltrating CD20+ cells, which were found within the endomysium. This finding suggests that in most cases, infiltration of muscle tissue by B-cells is unlikely to play a primary role in anti-HMGCR disease pathology.
Infiltrating T-cells are a prominent feature of most inflammatory myopathy subtypes. For example, perimysial and perivascular accumulations of T-cells are characteristic of DM, and the presence of CD8+ T-cells surrounding and invading non-necrotic fibers has been described as hallmark of PM. 17, 18 In the anti-HMGCR myopathy patients studied here, we found very few examples of the former and no examples of CD8+ T-cells surrounding and invading non-necrotic fibers. Instead, we found sparse, predominantly endomysial, CD4+ and CD8+ cells in just over half of the anti-HMGCR biopsies. Although we cannot exclude the possibility that these T-cells play a role in mediating myofiber damage, the marked myofiber necrosis seen on biopsy appears to be out of proportion to the minimal lymphocytic infiltration observed here.
We confirmed that up-regulation of MHC class I protein on intact muscle fibers is a common feature in anti-HMGCR myopathy. This feature is commonly used to support the diagnosis of an immune-mediated myopathy. However, it should be noted that MHC I overexpression has also been reported in the non-necrotic fibers of muscle biopsies from patients with genetic muscle diseases such as Duchenne muscular dystrophy and dysferlinopathy. 19-21 Thus, MHC I overexpression is not specific for an immune-mediated myopathy.
In approximately half of anti-HMGCR and DM cases, MAC deposition was noted on endomysial capillaries. Interestingly, prior studies have reported that capillary MAC deposition occurs at an early stage in dermatomyositis and resolves following IVIG treatment. 22, 23 However, we did not find a correlation between the presence of capillary MAC deposition and treatment history or disease duration at the time of biopsy in this cohort of anti-HMGCR myopathy patients. As in our prior study, 5 we noted that MAC deposition on the sarcolemmal surface of apparently non-necrotic fibers was a common feature of anti-HMGCR myopathy. In contrast, only 1 of 6 DM biopsies had examples of sarcolemmal MAC deposition. Although this finding raises the possibility that MAC deposition could play a role in mediating myofiber damage in anti-HMGCR myopathy, we cannot exclude the possibility that MAC deposits are non-specific and occur as a consequence of early sarcolemmal damage caused by some other pathophysiologic process.
In summary, we have shown that muscle biopsies from patients with anti-HMGCR myopathy have characteristic cellular infiltrates in addition to prominent necrosis. The infiltrating cells include numerous macrophages as well as few scattered helper (CD4+) T-cells, cytolytic (CD8+) T-cells, and plasmacytoid dendritic cells. Since the infiltrating macrophages are mostly of the M2 subtype, we expect that they serve to facilitate muscle regeneration rather than mediate muscle injury. Similarly, while scattered T-cells and PDCs are also found within the endomysium, it is not clear how the relatively small number of these cells would be the cause of the widespread tissue destruction seen in many of these cases. Given our prior observation that anti-HMGCR antibody titers correlate with disease activity and that MAC deposition is frequently observed on the surface of healthy appearing myofibers, we hypothesize that binding of antibodies to the cell surface may activate the classical complement pathway, causing MAC deposition and subsequent myofiber lysis. In this regard, it is noteworthy that sera from patients with anti-SRP associated IMNM can mediate MAC-mediated cell death in cultured myoblasts. 24 Future in vitro and in vivo studies will be required to demonstrate whether anti-HMGCR antibodies are pathogenic.
Supplementary Material
Acknowledgments
Grant Support: This work was supported by the Huayi and Siuling Zhang Discovery Fund. This work was supported [in part] by the Intramural Research Program of the National Institute of Arthritis and Musculoskeletal and Skin Diseases of the National Institutes of Health.
Abbreviations
- CT
computed tomography
- DM
dermatomyositis
- H&E
hematoxylin and eosin
- HIPAA
Health Insurance Portability and Accountability Act
- HMGCR
3-hydroxy-3-methyl-gulatryl-CoA reductase
- IIM
idiopathic inflammatory myopathy
- IMNM
immune-mediated necrotizing myopathy
- MAC
membrane attack complex
- MHC I
Major histocompatibility complex class I
- PDC
plasmacytoid dendritic cell
- PET
positron emission tomography
- PM
polymyositis
- SRP
signal recognition particle
Footnotes
Conflicts of interest: ALM and LC-S have patented the anti-HMGCR antibody test.
References
- 1.Rider LG, Miller FW. Deciphering the clinical presentations, pathogenesis, and treatment of the idiopathic inflammatory myopathies. JAMA. 2011;305:183–190. doi: 10.1001/jama.2010.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Mammen AL. Dermatomyositis and polymyositis: Clinical presentation, autoantibodies, and pathogenesis. Ann N Y Acad Sci. 2010;1184:134–153. doi: 10.1111/j.1749-6632.2009.05119.x. [DOI] [PubMed] [Google Scholar]
- 3.Hoogendijk JE, Amato AA, Lecky BR, Choy EH, Lundberg IE, Rose MR, et al. 119th ENMC international workshop: trial design in adult idiopathic inflammatory myopathies, with the exception of inclusion body myositis, 10-12 October 2003, Naarden, The Netherlands. Neuromuscul Disord. 2004;14:337–345. doi: 10.1016/j.nmd.2004.02.006. [DOI] [PubMed] [Google Scholar]
- 4.Greenberg SA, Pinkus JL, Pinkus GS, Burleson T, Sanoudou D, Tawil R, et al. Interferon-alpha/beta-mediated innate immune mechanisms in dermatomyositis. Ann Neurol. 2005;57:664–678. doi: 10.1002/ana.20464. [DOI] [PubMed] [Google Scholar]
- 5.Christopher-Stine L, Casciola-Rosen LA, Hong G, Chung T, Corse AM, Mammen AL. A novel autoantibody recognizing 200-kd and 100-kd proteins is associated with an immune-mediated necrotizing myopathy. Arthritis Rheum. 2010;62:2757–2766. doi: 10.1002/art.27572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Mammen AL, Chung T, Christopher-Stine L, Rosen P, Rosen A, Doering KR, et al. Autoantibodies against 3-hydroxy-3-methylglutaryl-coenzyme A reductase in patients with statin-associated autoimmune myopathy. Arthritis Rheum. 2011;63:713–721. doi: 10.1002/art.30156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Tidball JG. Inflammatory processes in muscle injury and repair. Am J Physiol Regul Integr Comp Physiol. 2005;288:R345–53. doi: 10.1152/ajpregu.00454.2004. [DOI] [PubMed] [Google Scholar]
- 8.Tidball JG, Villalta SA. Regulatory interactions between muscle and the immune system during muscle regeneration. Am J Physiol Regul Integr Comp Physiol. 2010;298:R1173–87. doi: 10.1152/ajpregu.00735.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Tidball JG. Mechanisms of muscle injury, repair, and regeneration. Compr Physiol. 2011;1:2029–2062. doi: 10.1002/cphy.c100092. [DOI] [PubMed] [Google Scholar]
- 10.Allenbach Y, Drouot L, Rigolet A, Charuel JL, Jouen F, Romero NB, et al. Anti-HMGCR autoantibodies in European patients with autoimmune necrotizing myopathies: inconstant exposure to statin. Medicine (Baltimore) 2014;93:150–157. doi: 10.1097/MD.0000000000000028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kharraz Y, Guerra J, Mann CJ, Serrano AL, Munoz-Canoves P. Macrophage plasticity and the role of inflammation in skeletal muscle repair. Mediators Inflamm. 2013;2013:491497. doi: 10.1155/2013/491497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Biswas SK, Mantovani A. Macrophage plasticity and interaction with lymphocyte subsets: cancer as a paradigm. Nat Immunol. 2010;11:889–896. doi: 10.1038/ni.1937. [DOI] [PubMed] [Google Scholar]
- 13.Page G, Chevrel G, Miossec P. Anatomic localization of immature and mature dendritic cell subsets in dermatomyositis and polymyositis: Interaction with chemokines and Th1 cytokine-producing cells. Arthritis Rheum. 2004;50:199–208. doi: 10.1002/art.11428. [DOI] [PubMed] [Google Scholar]
- 14.Walsh RJ, Kong SW, Yao Y, Jallal B, Kiener PA, Pinkus JL, et al. Type I interferon-inducible gene expression in blood is present and reflects disease activity in dermatomyositis and polymyositis. Arthritis Rheum. 2007;56:3784–3792. doi: 10.1002/art.22928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Baechler EC, Bauer JW, Slattery CA, Ortmann WA, Espe KJ, Novitzke J, et al. An interferon signature in the peripheral blood of dermatomyositis patients is associated with disease activity. Mol Med. 2007;13:59–68. doi: 10.2119/2006-00085.Baechler. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Brunn A, Hans VJ, Vogelgesang S, Deckert M. Inflammatory myopathy with abundant macrophages and dermatomyositis: two stages of one disorder or two distinct entities? Acta Neuropathol. 2009;118:793–801. doi: 10.1007/s00401-009-0570-8. [DOI] [PubMed] [Google Scholar]
- 17.Arahata K, Engel AG. Monoclonal antibody analysis of mononuclear cells in myopathies. I: Quantitation of subsets according to diagnosis and sites of accumulation and demonstration and counts of muscle fibers invaded by T-cells. Ann Neurol. 1984;16:193–208. doi: 10.1002/ana.410160206. [DOI] [PubMed] [Google Scholar]
- 18.Giorno R, Ringel SP. Analysis of macrophages, activated cells and T cell subsets in inflammatory myopathies using monoclonal antibodies. Pathol Immunopathol Res. 1986;5:491–499. doi: 10.1159/000157034. [DOI] [PubMed] [Google Scholar]
- 19.Nagappa M, Nalini A, Narayanappa G. Major histocompatibility complex and inflammatory cell subtype expression in inflammatory myopathies and muscular dystrophies. Neurol India. 2013;61:614–621. doi: 10.4103/0028-3886.125264. [DOI] [PubMed] [Google Scholar]
- 20.Confalonieri P, Oliva L, Andreetta F, Lorenzoni R, Dassi P, Mariani E, et al. Muscle inflammation and MHC class I up-regulation in muscular dystrophy with lack of dysferlin: an immunopathological study. J Neuroimmunol. 2003;142:130–136. doi: 10.1016/s0165-5728(03)00255-8. [DOI] [PubMed] [Google Scholar]
- 21.Dai TJ, Li W, Zhao QW, Zhao YY, Liu SP, Yan CZ. CD8/MHC-I complex is specific but not sensitive for the diagnosis of polymyositis. J Int Med Res. 2010;38:1049–1059. doi: 10.1177/147323001003800332. [DOI] [PubMed] [Google Scholar]
- 22.Basta M, Dalakas MC. High-dose intravenous immunoglobulin exerts its beneficial effect in patients with dermatomyositis by blocking endomysial deposition of activated complement fragments. J Clin Invest. 1994;94:1729–1735. doi: 10.1172/JCI117520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Sakuta R, Murakami N, Jin Y, Nagai T, Nonaka I, Nishino I. Diagnostic significance of membrane attack complex and vitronectin in childhood dermatomyositis. J Child Neurol. 2005;20:597–602. doi: 10.1177/08830738050200071201. [DOI] [PubMed] [Google Scholar]
- 24.Rojana-udomsart A, Mitrpant C, Bundell C, Price L, Luo YB, Fabian V, et al. Complement-mediated muscle cell lysis: a possible mechanism of myonecrosis in anti-SRP associated necrotizing myopathy (ASANM) J Neuroimmunol. 2013;264:65–70. doi: 10.1016/j.jneuroim.2013.08.008. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


