ABSTRACT
Newcastle disease virus (NDV) avirulent strain LaSota was used to coexpress gp160 Env and p55 Gag from a single vector to enhance both Env-specific and Gag-specific immune responses. The optimal transcription position for both Env and Gag genes in the NDV genome was determined by generating recombinant NDV (rNDV)-Env-Gag (gp160 located between the P and M genes and Gag between the HN and L genes), rNDV-Gag-Env (Gag located between the P and M genes and gp160 between the HN and L genes), rNDV-Env/Gag (gp160 followed by Gag located between the P and M genes), and rNDV-Gag/Env (Gag followed by gp160 located between the P and M genes). All the recombinant viruses replicated at levels similar to those seen with parental NDV in embryonated chicken eggs and in chicken fibroblast cells. Both gp160 and Gag proteins were expressed at high levels in cell culture, with gp160 found to be incorporated into the envelope of NDV. The Gag and Env proteins expressed by all the recombinants except rNDV-Env-Gag self-assembled into human immunodeficiency virus type 1 (HIV-1) virus-like particles (VLPs). Immunization of guinea pigs by the intranasal route with these rNDVs produced long-lasting Env- and Gag-specific humoral immune responses. The Env-specific humoral and mucosal immune responses and Gag-specific humoral immune responses were higher in rNDV-Gag/Env and rNDV-Env/Gag than in the other recombinants. rNDV-Gag/Env and rNDV-Env/Gag were also more efficient in inducing cellular as well as protective immune responses to challenge with vaccinia viruses expressing HIV-1 Env and Gag in mice. These results suggest that vaccination with a single rNDV coexpressing Env and Gag represents a promising strategy to enhance immunogenicity and protective efficacy against HIV.
IMPORTANCE
A safe and effective vaccine that can induce both systemic and mucosal immune responses is needed to control HIV-1. In this study, we showed that coexpression of Env and Gag proteins of HIV-1 performed using a single Newcastle disease virus (NDV) vector led to the formation of HIV-1 virus-like particles (VLPs). Immunization of guinea pigs with recombinant NDVs (rNDVs) elicited potent long-lasting systemic and mucosal immune responses to HIV. Additionally, the rNDVs were efficient in inducing cellular immune responses to HIV and protective immunity to challenge with vaccinia viruses expressing HIV Env and Gag in mice. These results suggest that the use of a single NDV expressing Env and Gag proteins simultaneously is a novel strategy to develop a safe and effective vaccine against HIV.
INTRODUCTION
Developing a safe and effective vaccine against human immunodeficiency virus type 1 (HIV-1) remains one of the most elusive goals of medicine. The encouraging results from the RV144 vaccine trial demonstrated that a preventive vaccine against HIV-1 can be developed (1). That trial and other studies have suggested that strong humoral, cellular, and mucosal immune responses are needed for complete protection against HIV (2). HIV-1 virus-like particles (VLPs) containing native forms of Env have been shown to elicit strong humoral and cellular immune responses (3, 4). Various approaches have been used to produce HIV VLPs. Most of these approaches are based on recombinant production of HIV proteins in yeast and mammalian cell expression systems (5–7). However, these strategies are expensive and require high doses of VLPs and adjuvants and repeated administration as well. Other strategies have used nonreplicating canarypox vectors or naked DNA, again requiring multiple boosts (3, 8, 9). In light of these limitations, there is a need to evaluate live replicating viral vectors that can produce cost-effective HIV VLPs and can be used as a safe vaccine in humans.
Newcastle disease virus (NDV) is a member of the family Paramyxoviridae, a family of nonsegmented, negative-sense RNA viruses (10). NDV causes severe disease in avian species but is apathogenic in nonavian species. NDV strains are categorized into three pathotypes based on severity of disease in chickens: lentogenic (avirulent), mesogenic (moderately virulent), and velogenic (highly virulent). Currently, lentogenic strains of NDV such as LaSota are used as live NDV vaccines for poultry throughout the world. NDV has a number of characteristics that make it an ideal vaccine vector for human use (11). NDV is safe in humans due to natural host range restriction. NDV has a long history of administration to humans parenterally and intranasally as well as in the form of oncolytic therapy, and it is well tolerated (12). NDV is antigenically distinct from common human and animal pathogens; therefore, there is no preexisting immunity to NDV in humans. NDV infects via the intranasal route and has been shown to induce humoral and cellular immune responses to protective antigens of human pathogens in laboratory animals and in nonhuman primates (NHP) at both the mucosal and systemic levels (13–21). Additionally, there are nine serotypes of avian paramyxoviruses (APMVs); therefore, several serologically distinct vectors can be developed to express different foreign genes for use in prime-boost immunization approaches.
In our previous studies, we evaluated recombinant NDV (rNDV) as a vaccine vector to produce humoral, mucosal, and cellular immune responses to different forms of Env protein in guinea pigs (18, 20). Recently, we evaluated the effect of mixing rNDV expressing HIV Gag with rNDVs expressing different forms of Env protein on Env-specific humoral and mucosal immune responses in guinea pigs and Env- and Gag-specific cellular immune responses in mice (22). In this study, to enhance the immunogenicity of vectored NDV vaccine and to overcome viral interference in a mixed infection, we coexpressed HIV gp160 Env protein (2.6-kb gene) and p55 Gag precursor protein (1.6-kb gene) from a single avirulent NDV. Our results showed that rNDV can accommodate an increase in genome size of at least ~22% without a significant reduction of virus growth in embryonated chicken eggs and in cell culture. Infection of embryonated chicken eggs by rNDVs coexpressing Env and Gag proteins resulted in production of HIV VLPs. Immunization of guinea pigs with rNDVs elicited potent long-lasting systemic and mucosal immune responses to HIV. Further, the rNDVs were also efficient in inducing cellular immune responses to HIV and protective immunity to challenge with vaccinia viruses expressing HIV Env and Gag in mice. These results suggest that the use of a single NDV expressing Env and Gag proteins simultaneously is a novel strategy to develop a safe and effective vaccine against HIV.
RESULTS
Generation of rNDVs expressing HIV-1 gp160 and Gag.
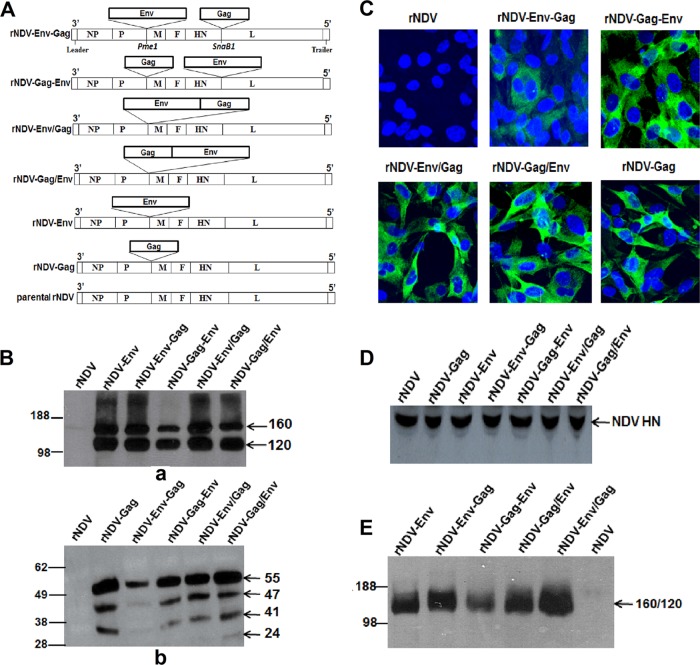
A recombinant avirulent NDV vaccine strain, LaSota, was used to construct four recombinant virus strains expressing gp160 Env glycoprotein and p55 Gag protein of HIV-1 strain BaL.1, namely, (i) rNDV-Env-Gag (gp160 located between the P and M genes and Gag between the HN and L genes), (ii) rNDV-Gag-Env (Gag located between the P and M genes and gp160 between the HN and L genes), (iii) rNDV-Env/Gag (gp160 followed by Gag located between the P and M genes), and (iv) rNDV-Gag/Env (Gag followed by gp160 located between the P and M genes) (Fig. 1A). We made these rNDVs expressing both Env and Gag from a single vector on the basis of the assumption that coexpression of HIV Env and Gag antigens from a single vector may circumvent the viral interference that could arise if two different vaccine vectors, each expressing a different antigen, were used in immunization. Additionally, expression of Env along with Gag by a single NDV vector may lead to the production of VLPs containing Env spikes on their surface. For comparison, we also used rNDV-Env, rNDV-Gag, and a mixture of rNDV-Env plus rNDV-Gag in this study. The construction and generation of rNDV-Env and rNDV-Gag were described in our previous studies (18, 22).
FIG 1 .
Expression analysis of gp160 and Gag in DF1 cells infected with different rNDVs. (A) Schematic representation of rNDV strain rLaSota with human codon-optimized HIV-1 p55 Gag and gp160 Env genes inserted at different positions. The restriction sites that were used to insert the Gag and the gp160 genes (PmeI and SnaB1) are indicated. Each transcriptional cassette was flanked by NDV gene start and a gene end transcription signals. (B) Expression and processing of Env and Gag proteins in DF1 cells infected with the indicated viruses. Cell lysates were analyzed 48 h postinfection by Western blotting using a pool of gp120-specific MAbs (panel a) and p24 Gag-specific MAb (panel b). (C) Immunofluorescence analysis of the intracellular expression of Gag protein in DF1 cells infected with indicated virus. The cells were probed with p24 Gag-specific MAb followed by staining with Alexa Fluor 488-conjugated goat anti-mouse IgG antibodies (green) and DAPI (4[prime],6-diamidino-2-phenylindole) (blue) and subsequently visualized under a confocal Zeiss LSM 510 fluorescence microscope. (D) Western blot analysis of NDV HN protein expression by different rNDVs in DF1 cells. (E) Analysis of incorporation of HIV-1 gp120 and 160 into purified rNDV virions using a pool of gp120-specific MAbs. The positions of the HIV gp160 precursor and its cleavage product gp120 and of the p55 Gag precursor and its cleavage products p47, p41, and p24 are indicated by arrows in the right margin in different gels.
The recombinant viruses were recovered and propagated in embryonated chicken egg cells with peak titers of 108 to 109 hemagglutinin (HA) units and in DF1 cells with peak titers of 106 to 107.5 50% tissue culture infective doses (TCID50)/ml. To determine the stability of the gp160 and Gag genes in rLaSota vector, the recovered viruses were passaged 10 times in embryonated chicken eggs and the sequences were confirmed.
Characterization of rNDVs expressing HIV-1 Env and Gag proteins.
To detect the expression of Env and Gag proteins, DF1 cells were infected with different rNDVs and cell lysates were analyzed by Western blotting. Analysis of the cell lysates using a pool of gp120-specific monoclonal antibodies (MAbs) revealed the presence of precursor protein (160 kDa) as well as the 120-kDa protein derived by cleavage of gp160 (Fig. 1B, panel a). Densitometric analysis of the gp120 protein bands revealed that the level of gp160 and gp120 produced by rNDV-Gag-Env was 2.5-fold lower than the level seen with the other recombinants. Additionally, Western blot analysis of cell lysates revealed the presence of four bands representing p55 Gag precursor protein and its cleavage products of ~47 kDa, ~41 kDa, and ~24 kDa, which was similar to our previous findings (22) (Fig. 1B, panel b). The densitometric analysis showed that the levels of p55 Gag protein and its cleavage products in the lysate of rNDV-Env-Gag were 5-fold lower than those seen with the other recombinants. The lower levels of Env and Gag proteins seen with rNDV-Gag-Env and rNDV-Env-Gag, respectively, could have been due to insertion of the second gene between the HN and L genes of the NDV genome, as the viral mRNAs are transcribed in decreasing amounts from the 3′ to 5′ end of the paramyxovirus genome. Immunofluorescence analysis using p24 Gag-specific MAbs confirmed the lower expression of Gag protein in the cytoplasm of DF1 cells infected with rNDV-Env-Gag than in those infected with the other recombinants (Fig. 1C). The insertion of HIV Env and Gag genes (combined length of 4.2 kb) into the rNDV genome did not affect expression of NDV gene, as shown by the similar levels of NDV HN protein expression by all the rNDVs (Fig. 1D).
To determine whether coexpression of Gag and Env from the same vector would allow incorporation of gp160 and gp120 into NDV virions, the recombinant viruses were purified through sucrose gradients and the viral proteins were examined by Western blotting. Our results showed that all the rNDVs incorporated mostly gp120 protein but with 2.5-fold-lower incorporation in the case of rNDV-Gag-Env virus (Fig. 1E). These results suggest that coexpression of Gag from the single NDV vector does not affect the incorporation of HIV Env protein into NDV envelope.
The multicycle growth kinetics of rNDVs showed that the presence of two transgenes retarded the growth of the viruses only slightly compared to that seen with the parental rNDV (see Fig. S1 in the supplemental material). The maximum titer of parental rNDV was 7.2 TCID50, and the titers of rNDVs containing two transgenes ranged from 5.7 to 6.6 TCID50. The pathogenicity of all the rNDVs was evaluated in 9-day-old embryonated chicken eggs by the mean death time (MDT) test. The values of MDT for rNDV, rNDV-Env, rNDV-Gag, rNDV-Env-Gag, rNDV-Gag-Env, rNDV-Env/Gag, and rNDV-Gag/Env were 105 h, 110 h, 110 h, 122 h, 116 h, 114 h, and 122 h, respectively. This test indicated that introduction of gp160 and Gag genes together into the rNDV genome did not increase the pathogenicity of the recombinant viruses in chickens. Indeed, the presence of two additional genes conferred further attenuation.
Characterization of HIV-1 VLPs produced by coexpression of Gag and Env proteins.
To determine whether the Gag and Env proteins produced by rNDV vectors self-assembled into HIV VLPs without the addition of other HIV proteins, we infected 9-day-old embryonated chicken eggs with rNDV-Gag/Env. The allantoic fluid was collected 72 h after infection and concentrated by passage through a 30% sucrose cushion followed by a 20 to 60% sucrose gradient that was subsequently analyzed by Western blotting. HIV gp160 and its gp120 cleavage product were detected in fractions 6 to 9, and p55 Gag and its cleavage products were detected in fractions 4 to 9, indicating the presence of HIV VLPs (Fig. 2A, panel a). The fractions were also analyzed for the presence of NDV HN protein in NDV virion particles (Fig. 2A, panel b). NDV HN protein was detected in fractions 6 to 9. Expression of the NDV HN protein and HIV-1 Gag and Env proteins peaked in the same fractions, indicating that VLPs and NDV virions have similar sizes and are therefore difficult to separate. To confirm the presence of HIV VLPs, we probed fraction 8 with anti-gp120 MAb followed by treatment with 6-nm-diameter-gold-nanoparticle-conjugated goat-anti-mouse IgG antibodies and performed analysis by electron microscopy (EM) (Fig. 2B). VLPs of the expected morphology (110 to 120 nm in diameter) were observed in this fraction (Fig. 2B, panel c), with numerous gold particles surrounding the Gag particles indicating the presence of HIV Env spikes anchored on the surface of the Gag particles. The density of the gold nanoparticles ranged from 25 to 35 per VLP. We did not detect any HIV VLPs in parental rNDV-infected allantoic fluid (Fig. 2B, panel a). We found NDV particles (>100 nm in diameter) in both parental rNDV-infected (Fig. 2B, panel b) and rNDV-Gag/Env-infected (Fig. 2B, panel d) allantoic fluids. The allantoic fluid of the eggs infected with rNDV-Env/Gag and rNDV-Gag-Env also produced VLPs of similar morphologies (data not shown). We were not able to detect VLPs in allantoic fluid of eggs infected with rNDV-Env-Gag. These data suggest that HIV Gag and Env expressed by NDV formed VLPs with a native cleavage-competent form of gp160 protein on their surface.
FIG 2 .
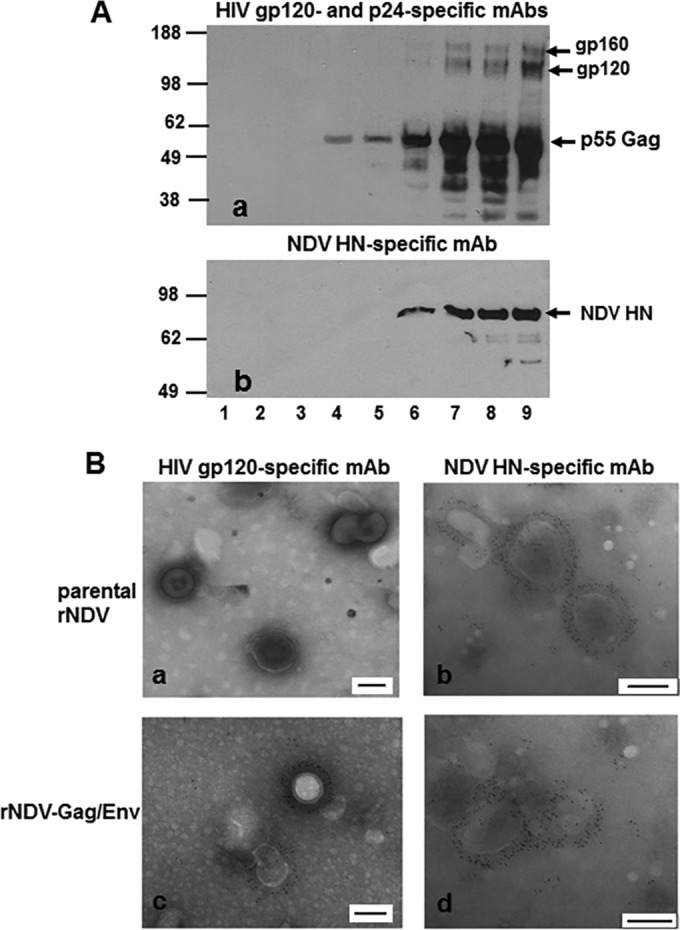
Biochemical characterization of HIV-1 VLPs produced in the allantoic fluid of 9-day-old embryonated chicken eggs infected with rNDV. (A) Western blot analysis of sucrose gradient fractions obtained after ultracentrifugation of rNDV-Gag/Env-infected allantoic fluid using HIV gp120-specific and p24-specific MAbs (panel a) or NDV HN-specific MAb (panel b). (B) Results of analysis of incorporation of gp160 (Env) protein on HIV-1 VLPs were verified by electron microscopy. Fraction 8 (shown in lane 8 in panel A) was adsorbed onto carbon-coated EM grids and stained with either anti-HIV gp120 MAb (panels a and c) or NDV HN-specific MAb (panels b and d) followed by detection with 6-nm-diameter-gold-nanoparticle-conjugated anti-mouse IgG antibodies. Each scale bar in panels a to d represents 100 nm.
Humoral immune responses in guinea pigs.
Eight groups of female Hartley guinea pigs (n = 5) were immunized on days 0 and 21 with different rNDVs as described in Materials and Methods and shown in Fig. S2 in the supplemental material. The animals in all the groups did not show any overt clinical signs of infection or any loss of body weight throughout the study (not shown), indicating that rNDVs were avirulent in guinea pigs.
The induction of NDV-specific serum antibodies was measured on days 21, 35, 56, and 160 using an NDV-specific enzyme-linked immunosorbent assay (ELISA) (Fig. 3A). All eight animal groups exhibited similar levels of NDV-specific IgG antibodies on those days, suggesting that all the viruses replicated to the same extent in the immunized animals. The boost on day 21 increased the NDV-specific IgG titer by more than 30-fold compared to the titer after the first immunization. The NDV-specific IgG antibodies peaked on day 35, decreased on day 56, and then remained stable until day 160.
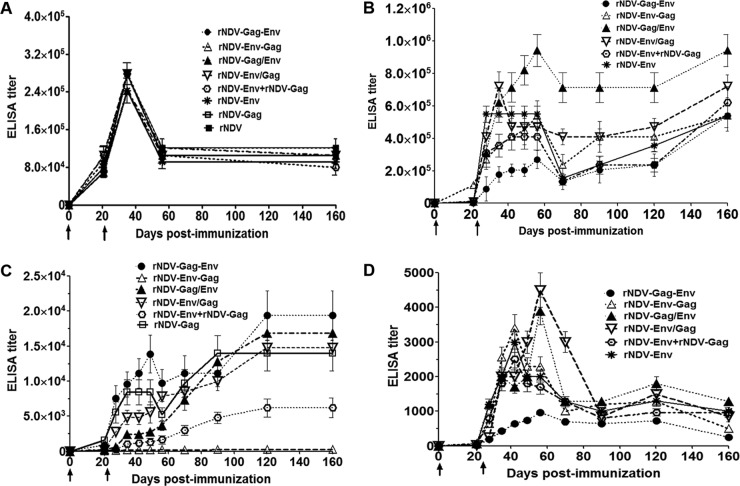
FIG 3 .
Immune responses to NDV or HIV Env and Gag proteins in guinea pigs. Guinea pigs were immunized with the indicated rNDVs either alone or in a mixture of two by the intranasal route. Mean ELISA endpoint titers are indicated for each analysis. (A) NDV-specific total IgG serum antibodies on days 21, 35, 56, and 160 analyzed by the use of commercial NDV ELISA kits (Synbiotics Corporation). (B) HIV-1 gp120-specific total IgG serum antibody responses on days 0, 21, 28, 35, 42, 49, 56, 70, 90, 120, and 160 analyzed by ELISA using purified gp120. (C) HIV-1 Gag-specific total IgG serum antibody responses on the same days as those listed in the panel B legend analyzed by ELISA using purified Gag (p24). (D) gp120-binding antibodies in vaginal washes performed on the same days as those listed in the panel B legend. Antibodies specific to gp120 and Gag p24 were not detected in any animal on any day in the parental rNDV control group. The graph shows the geometric mean values ± standard errors of the means (SEM) of the results determined for 5 animals in each group. Arrows indicate times of rNDV immunizations on days 0 and 21. P values of HIV-1 gp120-specific total IgG response differences in the serum results from different groups were <0.05. The P value of HIV-1 Gag-specific total IgG response differences in the serum results from different groups was <0.0001. The P value of HIV-1 gp120-specific total IgG response differences in the results from different groups in vaginal washes was 0.16. The P value of NDV-specific total IgG response differences in the results from different groups in serum was >0.05.
The induction of HIV-1 Env-specific serum antibodies was measured in all the groups on days 21, 28, 35, 42, 49, 56, and 70 (Fig. 3B). Env-specific antibody responses were detected on day 21 following the initial immunization and increased 30-fold to 200-fold after the boost, except in the parental rNDV and rNDV-Gag groups, where no response was detected. The total IgG response peaked between days 28 and 56 in all the groups. The highest gp120-specific IgG titer was observed with rNDV-Gag/Env followed by rNDV-Env/Gag (both groups with gp160 and Gag inserted at PmeI site but in the opposite orders), with titers even higher than those seen with the rNDV-Env group (rNDV with the Env insertion only). The responses in the other groups were lower than the rNDV-Env responses. The lowest titer was observed in the rNDV-Gag-Env group. The titer of total IgG declined on day 70 in all the groups (decreasing by 1.5-fold to 4.5-fold compared to the level seen on day 56).
In addition, we checked the durability of Env-specific serum antibodies until day 160 (the titers were measured on days 90, 120, and 160). Surprisingly, the titer of Env-specific total IgG did not decline further on those days; rather, it started increasing until day 160, reaching levels close to the peak titer or even above it in all the groups. These results suggest that anti-Env immune responses generated by rNDV vector are long lasting and persist for at least 160 days.
We measured the induction of HIV-1 Gag-specific serum antibodies in all the guinea pig groups at the same time points by ELISA (Fig. 3C). A very low titer of anti-Gag antibodies was detected on day 21 in all the groups, and boosting on day 21 increased the titers by 3-fold to 63-fold in the different groups except in the rNDV control and rNDV-Env groups, where no response was detected. Unlike the Env-specific immune response, the patterns of Gag-specific responses differed greatly among the different groups. In the rNDV-Gag-Env and rNDV-Gag groups, the Gag-specific total IgG titer increased between day 35 and day 49, decreased on day 56, and started increasing again and reached the peak value on days 120 and 90, respectively. In the other groups, the titer increased slowly after the boost and reached peak values on day 120. The titer remained stable from day 120 until day 160 in all the groups. The highest titer was observed in the rNDV-Gag-Env group followed by the rNDV-Gag/Env, rNDV-Env/Gag, rNDV-Gag, and rNDV-Env plus rNDV-Gag groups. These results indicated that Gag-specific IgG responses were significantly higher in the rNDV-Gag-Env, rNDV-Gag/Env, and rNDV-Env/Gag groups (rNDVs either with both the Gag and Env insertions at same site or with insertion of the Gag before the Env at different sites) and comparable to the immune responses induced by rNDV-Gag. These responses persisted until day 160.
Mucosal immune responses in guinea pigs.
Vaginal washes were collected from each animal at different time points and evaluated by ELISA using BaL.1 gp120-coated plates (Fig. 3D). Very low responses were detected on day 21 following the initial immunization, but the boost on day 21 increased immune responses significantly in all the groups (titers increased 30-fold to 150-fold), except in the rNDV control and rNDV-Gag groups, where no responses were detected. The titer of total IgG peaked between days 42 and 56, decreased between days 70 and 90, and had a tendency to increase between days 90 and 120 before decreasing on day 160. The total IgG response was highest in the rNDV-Env/Gag group followed by the rNDV-Gag/Env, rNDV-Env-Gag, rNDV-Env, rNDV-Env plus rNDV-Gag, and rNDV-Gag-Env groups. These results indicated that, similarly to humoral immune responses to Env, mucosal immune responses were higher in the rNDV-Env/Gag and rNDV-Gag/Env groups than in the other groups. We assayed the total IgG response in fecal samples in all the groups but were unable to detect any titer (not shown).
NAb responses in guinea pigs.
Sera collected on days 21, 28, 56, 70, 90, and 160 from animals immunized with the different rNDVs were evaluated by the TZM.bl assay for their ability to neutralize homologous clade B tier 1B HIV-1 strain BaL.26 (Fig. 4A) and heterologous clade B tier 1A HIV-1 strain MN.3 (Fig. 4B). Neutralizing antibody (NAb) activity (expressed as a 50% inhibitory concentration [IC50] value) against BaL.26 was detected in sera from all groups of animals, with the highest titer observed in the rNDV-Gag/Env group followed by the rNDV-Env, rNDV-Env plus rNDV-Gag, rNDV-Env-Gag, rNDV-Env/Gag, and rNDV-Gag-Env groups. The NAb response to HIV-1 strain MN.3 in immunized guinea pigs was significantly higher than the response to BaL.26 in the different groups. A comparison of the mean IC50 values for MN.3 showed the strongest NAb response in the rNDV-Env/Gag group followed by the rNDV-Gag/Env, rNDV-Env plus rNDV-Gag, rNDV-Env-Gag, rNDV-Env, and rNDV-Gag-Env groups. The patterns of response differed among different groups. In the rNDV-Env/Gag group, the titer peaked on day 28, decreased on day 56, and again peaked on day 70, whereas the peak titer was observed between days 70 and 90 in the other groups. A very low response to BaL.26 and MN.3 that was close to parental rNDV group response was detected in the rNDV-Gag group. Similarly to the anti-Env and anti-Gag immune responses, the NAb responses generated by all the rNDV vectors were long lasting and persisted for at least 160 days. In contrast to neutralization of the HIV-1 MN.3 strain, neutralization of the BaL.26 isolate by all the rNDVs was weak in this study. That isolate is more neutralization resistant than MN.3, perhaps due to a more closed conformation of major variable loops that decreases the accessibility of major gp120 epitopes to antibodies (23).
FIG 4 .
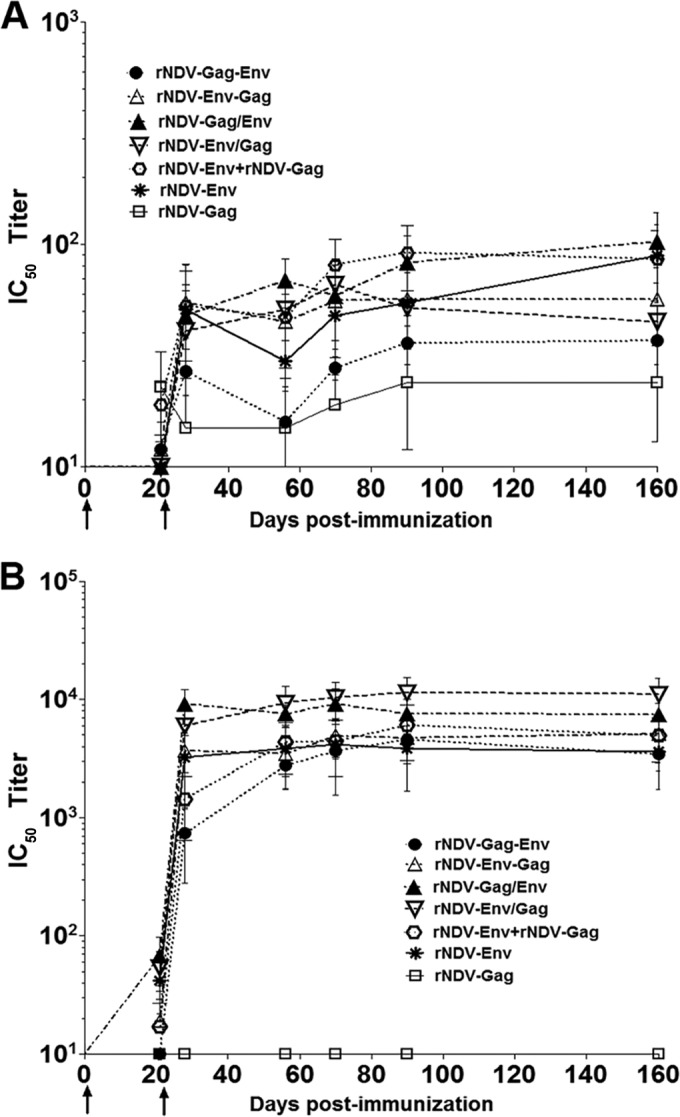
Virus-neutralizing antibody activity (50% inhibitory concentration [IC50] titers) against homologous HIV-1 clade B tier 1B strains (BaL.26) and heterologous clade B tier 1A strain MN.3 in sera from guinea pigs immunized with the indicated rNDVs. (A) Guinea pig sera obtained on days 21, 28, 56, 70, 90, and 160 were tested against BaL.26 pseudovirus by the TZM-bl assay. (B) Guinea pig sera obtained on days 21, 28, 56, 70, 90, and 160 were tested against MN.3 pseudovirus by the TZM-bl assay. Preimmune sera were used to establish values representing baseline-neutralizing activity in each individual guinea pig, and these values were subtracted from the values shown. The values for neutralizing antibody activity against each virus in sera obtained from guinea pigs immunized with parental rNDV control virus were <20 and are not shown in the graphs. The samples that did not neutralize the target virus (titers of <20) were given a value of 10 for plotting purposes; therefore, all results from samples registering an IC50 titer of 10 are negative. Statistically significant differences in data from serological analysis of different immunized guinea pig groups were evaluated by one-way analysis of variance (ANOVA). P values of BaL.26-specific neutralizing antibody titer differences in comparisons of serum between different groups were <0.05. P values of MN.3-specific neutralizing antibody titer differences in comparisons of serum between different groups were <0.0001.
Cellular immune responses in mice.
Due to the lack of availability of antibody reagents for analyzing cellular immune responses in guinea pigs, the abilities of rNDVs to stimulate cellular immune responses to HIV-1 Env and Gag were evaluated in female BALB/c mice (n = 6/group). We used an enzyme-linked immunosorbent spot (ELISPOT) assay to evaluate the capacity of splenocytes from mice to secrete gamma interferon (IFN-γ) in response to stimulation with Gag peptide and Env peptide pools (Fig. 5). In response to Gag peptide stimulation (Fig. 5A), levels of IFN-γ induced from the rNDV-Gag/Env (385 spot-forming cells [SFC]/106 cells), rNDV-Env/Gag (335 SFC/106), and rNDV-Gag-Env (310 SFC/106) groups were higher than the level of IFN-γ from the rNDV-Gag group (210 SFC/106). The values of SFC/106 splenocytes for the rNDV-Env plus rNDV-Gag and rNDV-Env-Gag groups were 145 and 35, respectively. Responses close to the base level were detected in the rNDV-Env and rNDV control groups. In response to Env peptide stimulation (Fig. 5B), the strongest T cell response was observed in the rNDV-Env/Gag group (325 SFC/106) followed by the rNDV-Gag/Env (290 SFC/106), rNDV-Env-Gag (250 SFC/106) and rNDV-Env (155 SFC/106), rNDV-Env plus rNDV-Gag (140 SFC/106), and rNDV-Gag-Env (115 SFC/106) groups. Very low responses close to the base level were detected in the rNDV control and rNDV-Gag groups. These results indicated that rNDV-Env/Gag and rNDV-Gag/Env with insertions of both Gag and Env at the PmeI site in the NDV genome are more efficient in induction of Gag- and Env-specific cellular immune responses than the other rNDVs.
FIG 5 .
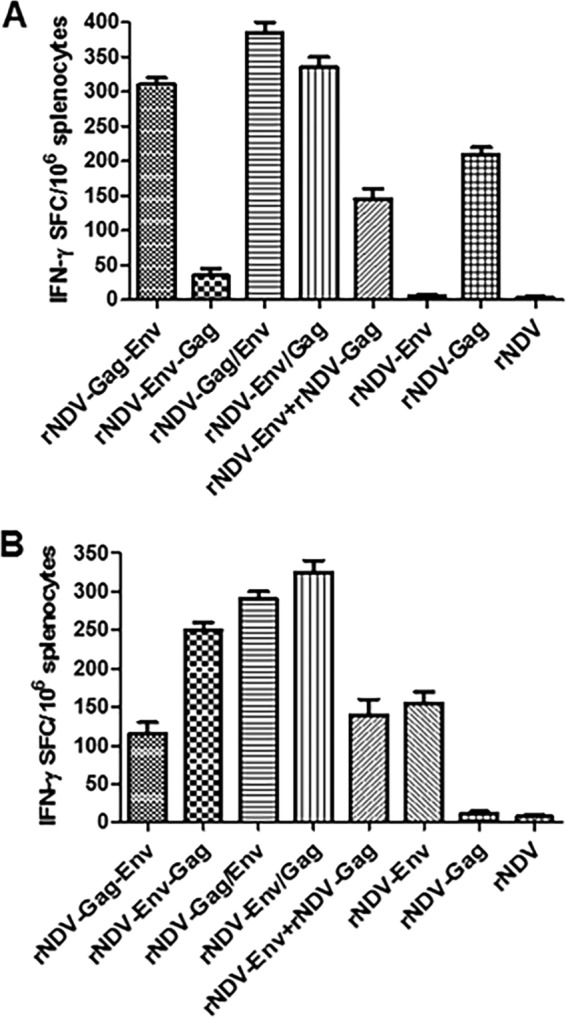
HIV-1 Gag- and Env-specific T cell response. Mice in groups of 6 each were immunized with 105 PFU/ml of the indicated rNDV either alone or in a mixture of two rNDVs by the intranasal route on days 0 and 21. On day 26, splenocytes were isolated, stimulated with either Gag-specific peptide (A) or a pool of overlapping Env-specific peptides (B), and analyzed for production of IFN-γ by ELISPOT assay. Results are presented as mean values for spot-forming cells (SFC)/106 cells ± standard deviations (SD) obtained from three experiments in each group where spleens from two mice were pooled in each experiment. P values of SFC/106 for comparisons between different groups were <0.0001.
Protection of mice from vaccinia virus-Env and vaccinia virus-Gag challenge.
Finally, we determined the ability of rNDVs to protect against virus challenge via a mucosal route in different groups of mice as shown in Fig. S2 in the supplemental material. In order to ascertain which rNDV induces the best protective immune response, each rNDV expressing Env and Gag was used to immunize BALB/c mice intranasally. We challenged the immunized mice 6 weeks after the boost via intranasal route with 107.2 TCID50/ml of virulent vaccinia virus expressing HIV-1 Gag (vaccinia virus-Gag). Mice were sacrificed at day 5 postchallenge, and the lung tissues were harvested. As shown in Fig. 6A, animals challenged with the rNDV control had high titers of vaccinia virus-Gag at close to 106 PFU/ml in their lungs. In marked contrast to the virus titers in control mice, the mice in all groups except the rNDV-Env and rNDV-Env-Gag groups showed a significant (>2 log10) reduction in virus titer. The order of greatest to least inhibition was rNDV-Gag/Env (2.8 log10), rNDV-Env/Gag (2.5 log10), rNDV-Gag-Env (2.4 log10), rNDV-Gag (2.12 log10), and rNDV-Env plus rNDV-Gag (1.98 log10). There was a marginal decrease in the vaccinia virus-Gag titer in the rNDV-Env-Gag group (5-fold), while the vaccinia virus-Gag titer was similar to the level seen with the control in the rNDV-Env group. We also checked the ability of rNDVs to protect against vaccinia virus expressing Env of the homologous BaL strain (vaccinia virus-BaL Env). After 6 weeks of boost, immunized mice were challenged with 107.6 TCID50/ml vaccinia virus-BaL Env via the intranasal route. Five days after the challenge, lungs were harvested and the challenge virus titer was determined (Fig. 6B). Although the overall level of replication of vaccinia virus-BaL Env was lower in lungs of parental rNDV mice than the level of replication seen in lungs of mice challenged with vaccinia virus-Gag, there was a significant reduction in the challenge virus titer in the different groups. The order from highest to lowest replication was rNDV-Env/Gag (650-fold), rNDV-Gag/Env (580-fold), rNDV-Env-Gag (300-fold), and rNDV-Env and rNDV-Env plus rNDV-Gag (both 207-fold). The mice in rNDV-Gag-Env group showed a reduction in replication of >70-fold, whereas those in the rNDV-Gag group did not show a significant reduction compared to the control group results. These results demonstrate that intranasal administration of rNDVs expressing Env and Gag from the same vector induces both Gag-specific and Env-specific protective immune responses. Furthermore, we showed that, similarly to the induction of cellular immune responses, the rNDVs having insertion of HIV Env and Gag together at PmeI site of NDV induced the strongest protective immune responses.
FIG 6 .
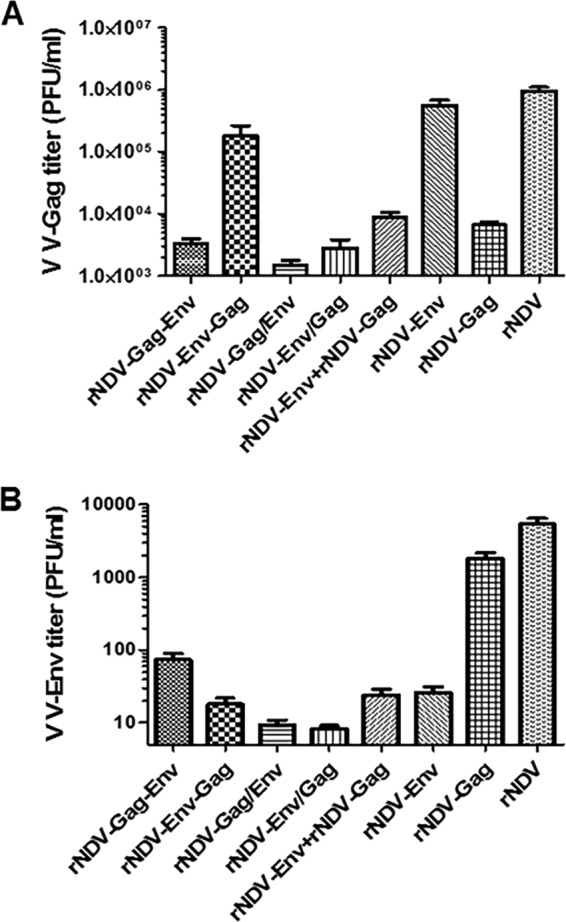
Protective efficacy of rNDVs against challenge with vaccinia virus-Gag (V V-Gag) and vaccinia virus-BaL Env. Mice in groups of 5 each were immunized with 105 PFU/ml of the indicated rNDV either alone or in a mixture of two rNDVs by the intranasal route on days 0 and 21. On day 63, mice were challenged by the intranasal route with either 107.2 TCID50/ml of vaccinia virus-Gag (A) or 107.6 TCID50/ml of vaccinia virus-BaL Env (B). Five days after the challenge, the mice were sacrificed and lungs were collected. Lungs were homogenized, and viral titers, represented as PFU/ml, were determined by standard plaque assay on Vero cells. The data represent three similar experiments. P values of PFU/ml for comparisons between different groups were <0.0001.
DISCUSSION
It is known that both humoral and cellular immune responses are needed for protection against HIV (2). Therefore, in this study we used rNDV to express both Env and Gag proteins to induce humoral and cellular immune responses. To overcome interference that might have arisen during immunization if the Env and Gag proteins were expressed by separate rNDV vectors, we coexpressed HIV-1 Env and Gag proteins using a single NDV vector. Previous studies performed with other negative-stranded RNA viruses such as vesicular stomatitis virus (VSV) and measles virus have demonstrated that these viruses can accommodate foreign genes at levels of up to 40% and 30%, respectively, of genomic RNA (24, 25). Although the upper limit of the size of a foreign gene that NDV can accommodate is not known, in this study we generated recombinant NDVs containing HIV Env and Gag genes together (totaling 4.2 kb in size) in the same genome, representing an approximately 22% increase in the size of the NDV genome. The rNDVs expressing HIV Env and Gag were highly stable for at least 10 passages in embryonated eggs, and the presence of Env and Gag insertions did not significantly affect the growth of the rNDVs in embryonated eggs or in DF1 cells. The remarkable stability of expression of the transgene by rNDV is due to the modular nature of the genome, the pleomorphic morphology, and the presence of helical nucleocapsids.
We have investigated the optimal location of Env and Gag genes in the NDV genome for higher levels of expression of these genes. The NDV genome contains six transcriptional units, NP-P-M-F-HN-L. We chose to use the intergenic regions between the P and M genes and between the HN and L genes of NDV because these sites were found previously to accommodate foreign genes without significantly affecting the growth of the virus (18, 26, 27). The choice of insertion of Gag between the P and M genes and of Env between the HN and L genes of NDV in rNDV-Gag-Env was based on the fact that the mature HIV-1 particles contain more Gag protein than Env protein (28). We also generated three additional recombinants. In rNDV-Env-Gag, Env was inserted between the P and M genes and Gag between the HN and L genes in the expectation of a possible increase in the spike density of Env on the surface of the virions. In rNDV-Gag/Env and rNDV-Env/Gag, Gag and Env were inserted together between the P and M genes in the opposite orders. We found that the level of expression correlated with the proximity of the foreign gene to the 3′ end of the NDV genome, as the expression levels of Gag and Env were high in rNDV-Gag-Env and in rNDV-Env-Gag, respectively. The expression level of either Env or Gag was not affected in rNDV-Env/Gag and rNDVGag/Env, as Env and Gag genes were inserted together between the P and M positions. We also examined the incorporation of Env proteins (gp160 and gp120) into the NDV virions purified from allantoic fluid of rNDV-infected eggs. A large amount of gp120 and a small amount of gp160 were incorporated in all the recombinants except for rNDV-Gag-Env, which showed lower expression of Env protein due to the distant location of the Env gene from the 3′ end of the NDV genome.
We previously showed the ability of NDV vector to induce Env-specific humoral and mucosal immune and Gag-specific humoral responses in guinea pigs when the animals were boosted after 2 weeks of priming (18, 20, 22). In the present study, the guinea pigs were boosted after 3 weeks of priming. The increase in the time between priming and boosting enhanced the Env-specific humoral and mucosal responses and Gag-specific humoral responses significantly. The Env-specific total IgG titers in serum and in vaginal washes were increased by 5- and 10-fold, respectively. The Gag-specific total IgG titer in serum was increased by 10-fold. These results suggest that the use of NDV vector coexpressing Env and Gag is a better strategy to produce higher immune responses, particularly against Env, than the use of a mixture of rNDVs expressing Env and Gag separately. We showed that rNDV-Gag/Env and rNDV-Env/Gag exhibited higher Env-specific humoral and mucosal immune responses than rNDV-Env and rNDV-Env plus rNDV-Gag, possibly due to the augmentation of immune responses by production of HIV VLPs. The lowest Env-specific total IgG titer in serum and vaginal washes in guinea pigs was recorded in rNDV-Gag-Env, which could have been due to the lower level of expression of Env protein by this recombinant.
Unlike the Env-specific immune response, the magnitude of the Gag-specific immune response was quite low among all the groups. These findings are similar to those in our previous study in guinea pigs immunized with rNDV expressing Gag either alone or in combination with rNDVs expressing Env (22). The lower serum IgG response to Gag could have been due to the localization of this protein intracellularly, which reduced the availability of Gag for B cell presentation. Among the different rNDVs coexpressing Env and Gag, the lowest Gag-specific total serum IgG response was observed in rNDV-Env-Gag. Also, VLPs were not produced by this recombinant. This might have been due to synthesis of a low level of Gag protein by this construct. Immunization with a mixture of rNDV-Env and rNDV-Gag did not affect the Env-specific immune response compared to immunization with rNDV-Env alone, but the Gag-specific immune response was significantly reduced after administration of this mixture compared to the immune response induced by rNDV-Gag alone. This could have been due to the competition of expression resulting in immunodominance of Env over Gag as reported previously (29–31).
With some experimental vaccines, the antibody response to HIV antigens has been short lived and has lacked memory B cells; such responses are considered to be among the major obstacles to HIV vaccine development (32, 33). In this study, HIV Env expressed by live NDV vector elicited long-lasting Env-specific IgG responses in serum, as the antibody titers, after a marginal decrease on day 70, continued to rise again until day 160. In general, persistent antibody titers are believed to depend on the presence of long-lived plasma cells and memory B cell responses and both are signs of germinal center function during immunization (34). Compared to the Env-specific immune response, the NDV-specific immune response in serum peaked on day 35, decreased on day 56, and remained stable thereafter until day 160. One possibility is that, in the case of NDV, the host immune response, probably a cellular immune response, suppressed NDV replication, resulting in a lower antibody response to NDV on day 56, whereas HIV Env protein expressed by rNDV might have remained in the body and continued to mount an immune response independently of NDV replication. In addition, HIV Env protein is heavily glycosylated and may be stable in the body for a long time.
Several studies have indicated that T cells, particularly CD8+ T lymphocytes, can control viremia in HIV-1-infected humans (35, 36). Previously, we showed that vaccination using rNDV expressing gp160 stimulated Th1-biased immune responses (18). Stimulation of the Th1-type response is mainly associated with induction of IFN-γ. In this study, we analyzed the Gag- and Env-specific IFN-γ secretion profiles in mice immunized with eight rNDVs expressing Env and Gag proteins either together or separately. Our results indicated that all the rNDVs stimulated cellular responses as shown by the results of ELISPOT assay for IFN-γ stimulation. The rNDV-Env/Gag and rNDV-Gag/Env viruses were again the most immunogenic among all the recombinants for stimulation of Env-specific and Gag-specific IFN-γ-secreting T cells, indicating augmentation of the immune response due to production of VLPs. It has been reported that NDV induces activation of dendritic cells (DCs), which is partially related to the ability of NDVs to induce secretion of IFN (37). There is a possibility that the presence of Env on VLPs produced by these rNDVs could increase uptake by DCs and induce more cross-presentation and hence increase class I presentation, resulting in efficient CD8+ T cell activation. A similar observation has been reported previously from a study of HIV-1 VLPs produced by measles virus (24). We also observed that rNDV-Gag-Env and rNDV-Env-Gag were the least immunogenic for stimulation of Env-specific and Gag-specific IFN-γ-expressing T cells, respectively, in mice due to the low expression level of Env or Gag protein. Stimulation of Env-specific IFN-γ-producing T cells by a mixture of rNDV-Env and rNDV-Gag was as efficient as that seen with rNDV-Env alone, but stimulation of Gag-specific IFN-γ-producing T cells by a mixture of these rNDVs was lower than the level seen with rNDV-Gag alone. This observation is consistent with data from our previous studies indicating that inclusion of Gag with gp160 enhanced the level of Env-specific IFN-γ-producing CD4+ and CD8+ T cells in mice but that there was some interference with enhancement of Gag-specific CD4+ and CD8+ T cell responses (22). Unfortunately, we were not able to detect significant Env-specific IgG responses by the use of ELISA in sera of mice due to cross-reactivity of serum from NDV negative-control mice with BaL-gp120.
Finally, in order to correlate the enhancement of IFN-γ-producing T cell responses with protective immunity, we challenged the mice with live vaccinia virus expressing HIV-1 Gag (vaccinia virus-Gag) and also with vaccinia virus expressing homologous HIV-BaL Env (vaccinia virus-BaL Env). The mice were completely protected against both the challenge viruses. The protective immune responses strongly supported the cellular immune responses, and the overall virus titers were again lowest in the mice immunized with rNDV-Gag/Env and rNDV-Env/Gag compared to those immunized with rNDVs with a single gene insertion. Although we demonstrated a role of Gag-specific as well as Env-specific cellular immune responses in protection against challenge virus in this study, it will be interesting to test the protective role of antibodies and antibody-dependent cell-mediated cytotoxicity (ADCC). Although the efficacy of NDV-vectored vaccine in mice challenged with vaccinia virus expressing homologous HIV Env seen in this study was encouraging, the efficacy of a similar vaccine regimen against defined mucosal simian-human immunodeficiency virus (SHIV) challenge in a NHP model needs to be evaluated.
In summary, these results support the idea of the use of NDV as a vaccine vector for expression of HIV immunogens capable of inducing neutralizing antibodies against diverse HIV-1 strains, thus providing a platform for ultimately testing the NDV-based vaccines in nonhuman primates and humans.
MATERIALS AND METHODS
Ethics statement.
All of the guinea pigs and mice used in this study were housed in isolator cages in our biosafety level 2+ facility and cared for in accordance with established guidelines, and the experimental procedures were performed with approval from the Institutional Animal Care and Use Committee (IACUC) of the University of Maryland.
Cells, viruses, antibodies, and protein.
Cells, viruses, antibodies, and proteins used in the study are described in Text S1 in the supplemental material.
Construction and characterization of rNDVs expressing HIV-1 gp160 and Gag.
Various versions of NDV antigenomic cDNAs were made expressing HIV gp160 and Gag from a single NDV vector, namely, NDV-Env-Gag (gp160 and Gag cDNAs were inserted at the PmeI site between the P and M genes and at the SnaBI site between the HN and L genes in the NDV antigenome, respectively), NDV-Gag-Env (Gag and gp160 cDNAs were inserted at the PmeI site and at the SnaBI site, respectively), and NDV-Env/Gag and NDV-Gag/Env (gp160 and Gag cDNAs were inserted together at PmeI). In NDV-Env/Gag, gp160 cDNA was inserted first followed by Gag cDNA, whereas in NDV-Gag/Env, Gag cDNA was inserted first followed by gp160 cDNA (Fig. 1). Recombinant viruses (rNDV-Env-Gag, rNDV-Gag-Env, rNDV-Env/Gag, and rNDV-Gag/Env) were recovered as described previously (18). In addition, for comparison purposes, rNDV-Env (containing the gp160 gene between the P and M genes), rNDV-Gag (containing the Gag gene between the P and M genes), a mixture of rNDV-Env plus rNDV-Gag, and parental rNDV as a negative control were used in this study. The details of construction and growth of rNDVs, analysis of expression of HIV-1 gp160 and Gag proteins, and characterization of pathogenicity can be found in Text S1 in the supplemental material.
Analysis of VLP production.
The ability of rNDVs to produce VLPs in embryonated chicken eggs was evaluated by using a procedure described previously (21) with some modifications. Further details can be found in Text S1 in the supplemental material.
Immunization of guinea pigs and mice, characterization of humoral and cellular immune responses, neutralization assays, and challenge studies.
Details of animals, immunization, different assays such as ELISA and ELISPOT, virus neutralization, and challenge with vaccinia viruses expressing HIV Gag and BaL Env can be found in Text S1 in the supplemental material.
Statistical analysis.
Statistical differences between the groups were calculated by one-way analysis of variance (ANOVA) and an unpaired (two-tailed) t test with the use of Prism 5.0 (Graph Pad Software Inc., San Diego, CA) with a significance level of P of <0.05.
SUPPLEMENTAL MATERIAL
Supplemental Materials and Methods. Download
Comparison of multicycle growth kinetics of parental rNDV and rNDVs expressing either Env or Gag or both in DF1 cells. Download
Immunization schedules. Download
ACKNOWLEDGMENTS
We thank Yunsheng Wang, Laura Sanglas Crespi, and other laboratory members for their technical assistance and help. We thank Girmay Gebreluul and Yonas Araya for their help with handling of guinea pigs.
This research was supported by NIH R21 grant AI-093198 awarded to S.K.S. and by NIAID Primate Central Immunology Laboratory contract HHSN27201100016C awarded to D.C.M.
The views expressed here do not necessarily reflect the official policies of the Department of Health and Human Services, and mention of trade names, commercial practices, or organizations does not imply endorsement by the United States government.
Footnotes
Citation Khattar SK, Manoharan V, Bhattarai B, LaBranche CC, Montefiori DC, Samal SK. 2015. Mucosal immunization with Newcastle disease virus vector coexpressing HIV-1 Env and Gag proteins elicits potent serum, mucosal, and cellular immune responses that protect against vaccinia virus Env and Gag challenges. mBio 6(4):e01005-15. doi:10.1128/mBio.01005-15.
REFERENCES
- 1.Rerks-Ngarm S, Pitisuttithum P, Nitayaphan S, Kaewkungwal J, Chiu J, Paris R, Premsri N, Namwat C, de Souza M, Adams E, Benenson M, Gurunathan S, Tartaglia J, McNeil JG, Francis DP, Stablein D, Birx DL, Chunsuttiwat S, Khamboonruang C, Thongcharoen P, Robb ML, Michael NL, Kunasol P, Kim JH, MOPH-TAVEG Investigators. 2009. Vaccination with ALVAC and AIDSVAX to prevent HIV-1 infection in Thailand. N Engl J Med 361:2209–2220. doi: 10.1056/NEJMoa0908492. [DOI] [PubMed] [Google Scholar]
- 2.McElrath MJ, Haynes BF. 2010. Induction of immunity to human immunodeficiency virus type-1 by vaccination. Immunity 33:542–554. doi: 10.1016/j.immuni.2010.09.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hammonds J, Chen X, Zhang X, Lee F, Spearman P. 2007. Advances in methods for the production, purification, and characterization of HIV-1 Gag-Env pseudovirion vaccines. Vaccine 25:8036–8048. doi: 10.1016/j.vaccine.2007.09.016. [DOI] [PubMed] [Google Scholar]
- 4.Young KR, McBurney SP, Karkhanis LU, Ross TM. 2006. Virus-like particles: designing an effective AIDS vaccine. Methods 40:98–117. doi: 10.1016/j.ymeth.2006.05.024. [DOI] [PubMed] [Google Scholar]
- 5.Sakuragi S, Goto T, Sano K, Morikawa Y. 2002. HIV type 1 Gag virus-like particle budding from spheroplasts of Saccharomyces cerevisiae. Proc Natl Acad Sci U S A 99:7956–7961. doi: 10.1073/pnas.082281199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hammonds J, Chen X, Fouts T, DeVico A, Montefiori D, Spearman P. 2005. Induction of neutralizing antibodies against human immunodeficiency virus type 1 primary isolates by Gag-Env pseudovirion immunization. J Virol 79:14804–14814. doi: 10.1128/JVI.79.23.14804-14814.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.McBurney SP, Ross TM. 2009. Human immunodeficiency virus-like particles with consensus envelopes elicited broader cell-mediated peripheral and mucosal immune responses than polyvalent and monovalent Env vaccines. Vaccine 27:4337–4349. doi: 10.1016/j.vaccine.2009.04.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Fang ZY, Kuli-Zade I, Spearman P. 1999. Efficient human immunodeficiency virus (HIV)-1 Gag-Env pseudovirion formation elicited from mammalian cells by a canarypox HIV vaccine candidate. J Infect Dis 180:1122–1132. doi: 10.1086/315028. [DOI] [PubMed] [Google Scholar]
- 9.Young KR, Smith JM, Ross TM. 2004. Characterization of a DNA vaccine expressing a human immunodeficiency virus-like particle. Virology 327:262–272. doi: 10.1016/j.virol.2004.07.009. [DOI] [PubMed] [Google Scholar]
- 10.Lamb RA, Park GD. 2007. Paramyxoviruses: the viruses and their replication, p 144–196. In Knipe DM, Lamb RA, Martin MA, Rioizman B, Strauss SE (ed), Field’s virology, 5th ed. Lippincott Williams and Wilkins, Philadelphia, PA. [Google Scholar]
- 11.Bukreyev A, Collins PL. 2008. Newcastle disease virus as a vaccine vector for humans. Curr Opin Mol Ther 10:46–55. [PubMed] [Google Scholar]
- 12.Fournier P, Schirrmacher V. 2013. Oncolytic Newcastle disease virus as cutting edge between tumor and host. Biology (Basel) 2:936–975. doi: 10.3390/biology2030936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bukreyev A, Huang Z, Yang L, Elankumaran S, St Claire M, Murphy BR, Samal SK, Collins PL. 2005. Recombinant Newcastle disease virus expressing a foreign viral antigen is attenuated and highly immunogenic in primates. J Virol 79:13275–13284. doi: 10.1128/JVI.79.21.13275-13284.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.DiNapoli JM, Kotelkin A, Yang L, Elankumaran S, Murphy BR, Samal SK, Collins PL, Bukreyev A. 2007. Newcastle disease virus, a host range-restricted virus, as a vaccine vector for intranasal immunization against emerging pathogens. Proc Natl Acad Sci U S A 104:9788–9793. doi: 10.1073/pnas.0703584104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.DiNapoli JM, Yang L, Suguitan A Jr, Elankumaran S, Dorward DW, Murphy BR, Samal SK, Collins PL, Bukreyev A. 2007. Immunization of primates with a Newcastle disease virus-vectored vaccine via the respiratory tract induces a high titer of serum neutralizing antibodies against highly pathogenic avian influenza virus. J Virol 81:11560–11568. doi: 10.1128/JVI.00713-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.DiNapoli JM, Nayak B, Yang L, Finneyfrock BW, Cook A, Andersen H, Torres-Velez F, Murphy BR, Samal SK, Collins PL, Bukreyev A. 2010. Newcastle disease virus-vectored vaccines expressing the hemagglutinin or neuraminidase protein of H5N1 highly pathogenic avian influenza virus protect against virus challenge in monkeys. J Virol 84:1489–1503. doi: 10.1128/JVI.01946-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.DiNapoli JM, Yang L, Samal SK, Murphy BR, Collins PL, Bukreyev A. 2010. Respiratory tract immunization of non-human primates with a Newcastle disease virus-vectored vaccine candidate against Ebola virus elicits a neutralizing antibody response. Vaccine 29:17–25. doi: 10.1016/j.vaccine.2010.10.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Khattar SK, Samal S, Devico AL, Collins PL, Samal SK. 2011. Newcastle disease virus expressing human immunodeficiency virus type 1 envelope glycoprotein induces strong mucosal and serum antibody responses in Guinea pigs. J Virol 85:10529–10541. doi: 10.1128/JVI.05050-11. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 19.Xiao S, Kumar M, Yang X, Akkoyunlu M, Collins PL, Samal SK, Pal U. 2011. A host-restricted viral vector for antigen-specific immunization against Lyme disease pathogen. Vaccine 29:5294–5303. doi: 10.1016/j.vaccine.2011.05.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Khattar SK, Samal S, LaBranche CC, Montefiori DC, Collins PL, Samal SK. 2013. Comparative immunogenicity of HIV-1 gp160, gp140 and gp120 expressed by live attenuated Newcastle disease virus vector. PLoS One 8:e78521. doi: 10.1371/journal.pone.0078521. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 21.Kim SH, Chen S, Jiang X, Green KY, Samal SK. 2014. Newcastle disease virus vector producing human norovirus-like particles induces serum, cellular, and mucosal immune responses in mice. J Virol 88:9718–9727. doi: 10.1128/JVI.01570-14. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 22.Khattar SK, Palaniyandi S, Samal S, LaBranche CC, Montefiori DC, Zhu X, Samal SK. 2015. Evaluation of humoral, mucosal, and cellular immune responses following co-immunization of HIV-1 Gag and Env proteins expressed by Newcastle disease virus. Hum Vaccin Immunother 11:504–515. doi: 10.4161/21645515.2014.987006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wyatt R, Sodroski J. 1998. The HIV-1 envelope glycoproteins: fusogens, antigens, and immunogens. Science 280:1884–1888. doi: 10.1126/science.280.5371.1884. [DOI] [PubMed] [Google Scholar]
- 24.Guerbois M, Moris A, Combredet C, Najburg V, Ruffié C, Février M, Cayet N, Brandler S, Schwartz O, Tangy F. 2009. Live attenuated measles vaccine expressing HIV-1 Gag virus like particles covered with gp160DeltaV1V2 is strongly immunogenic. Virology 388:191–203. doi: 10.1016/j.virol.2009.02.047. [DOI] [PubMed] [Google Scholar]
- 25.Haglund K, Forman J, Kräusslich HG, Rose JK. 2000. Expression of human immunodeficiency virus type 1 Gag protein precursor and envelope proteins from a vesicular stomatitis virus recombinant: high-level production of virus-like particles containing HIV envelope. Virology 268:112–121. doi: 10.1006/viro.1999.0120. [DOI] [PubMed] [Google Scholar]
- 26.Zhao H, Peeters BP. 2003. Recombinant Newcastle disease virus as a viral vector: effect of genomic location of foreign gene on gene expression and virus replication. J Gen Virol 84:781–788. doi: 10.1099/vir.0.18884-0. [DOI] [PubMed] [Google Scholar]
- 27.Carnero E, Li W, Borderia AV, Moltedo B, Moran T, García-Sastre A. 2009. Optimization of human immunodeficiency virus gag expression by Newcastle disease virus vectors for the induction of potent immune responses. J Virol 83:584–597. doi: 10.1128/JVI.01443-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Zhu P, Liu J, Bess J Jr, Chertova E, Lifson JD, Grisé H, Ofek GA, Taylor KA, Roux KH. 2006. Distribution and three-dimensional structure of AIDS virus envelope spikes. Nature 441:847–852. doi: 10.1038/nature04817. [DOI] [PubMed] [Google Scholar]
- 29.Toapanta FR, Craigo JK, Montelaro RC, Ross TM. 2007. Reduction of anti-HIV-1 Gag immune responses during co-immunization: immune interference by the HIV-1 envelope. Curr HIV Res 5:199–209. doi: 10.2174/157016207780077057. [DOI] [PubMed] [Google Scholar]
- 30.Van Montfort T, Sanders RW. 2012. Optimizing cellular immunity against HIV-1 Gag and preventing suppression by HIV-1 gp120. Expert Rev Vaccines 11:1175–1177. doi: 10.1586/erv.12.102. [DOI] [PubMed] [Google Scholar]
- 31.Böckl K, Wild J, Bredl S, Kindsmüller K, Köstler J, Wagner R. 2012. Altering an artificial Gagpolnef polyprotein and mode of ENV co-administration affects the immunogenicity of a clade C HIV DNA vaccine. PLoS One 7:e34723. doi: 10.1371/journal.pone.0034723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Bonsignori M, Moody MA, Parks RJ, Holl TM, Kelsoe G, Hicks CB, Vandergrift N, Tomaras GD, Haynes BF. 2009. HIV-1 envelope induces memory B cell responses that correlate with plasma antibody levels after envelope gp120 protein vaccination or HIV-1 infection. J Immunol 183:2708–2717. doi: 10.4049/jimmunol.0901068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Gallo RC. 2005. The end or the beginning of the drive to an HIV-preventive vaccine: a view from over 20 years. Lancet 366:1894–1898. doi: 10.1016/S0140-6736(05)67395-3. [DOI] [PubMed] [Google Scholar]
- 34.Pulendran B, Ahmed R. 2011. Immunological mechanisms of vaccination. Nat Immunol 12:509–517. doi: 10.1038/ni.2039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.McDermott AB, Koup RA. 2012. CD8(+) T cells in preventing HIV infection and disease. AIDS 26:1281–1292. doi: 10.1097/QAD.0b013e328353bcaf. [DOI] [PubMed] [Google Scholar]
- 36.Hersperger AR, Migueles SA, Betts MR, Connors M. 2011. Qualitative features of the HIV-specific CD8+ T-cell response associated with immunologic control. Curr Opin HIV AIDS 6:169–173. doi: 10.1097/COH.0b013e3283454c39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Maamary J, Array F, Gao Q, García-Sastre A, Steinman RM, Palese P, Nchinda G. 2011. Newcastle disease virus expressing a dendritic cell-targeted HIV gag protein induces a potent gag-specific immune response in mice. J Virol 85:2235–2246. doi: 10.1128/JVI.02036-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplemental Materials and Methods. Download
Comparison of multicycle growth kinetics of parental rNDV and rNDVs expressing either Env or Gag or both in DF1 cells. Download
Immunization schedules. Download