Abstract
The antigenic variability of tumor cells leading to dynamic changes in cancer epitope landscape along with escape from immune surveillance by down-regulating tumor antigen expression/presentation and immune tolerance are major obstacles for the design of effective vaccines. We have developed a novel concept for immunogen construction based on introduction of massive mutations within the epitopes targeting antigenically variable pathogens and diseases. Previously, we showed that these immunogens carrying large combinatorial libraries of mutated epitope variants, termed as variable epitope libraries (VELs), induce potent, broad and long lasting CD8+IFN-γ+ T-cell response as well as HIV-neutralizing antibodies. In this proof-of-concept study, we tested immunogenic properties and anti-tumor effects of the VELs bearing survivin-derived CTL epitope (GWEPDDNPI) variants in an aggressive metastatic mouse 4T1 breast tumor model. The constructed VELs had complexities of 10,500 and 8,000 individual members, generated as combinatorial M13 phage display and synthetic peptide libraries, respectively, with structural composition GWXPXDXPI, where X is any of 20 natural amino acids. Statistically significant tumor growth inhibition was observed in BALB/c mice immunized with the VELs in both prophylactic and therapeutic settings. Vaccinated mice developed epitope-specific spleen cell and CD8+ IFN-γ+ T-cell responses that recognize more than 50% of the panel of 87 mutated epitope variants, as demonstrated in T-cell proliferation assays and FACS analysis. These data indicate the feasibility of the application of this new class of immunogens based on VEL concept as an alternative approach for the development of molecular vaccines against cancer.
Keywords: antigenic variability, cancer epitope vaccine, peptide vaccine, variable epitope library
Abbreviations
- CTL
cytotoxic T lymphocyte
- Ag
antigen
- APLs
altered peptide ligands
- wt
wild type
- MHC
major histocompatibility complex
- ICS
intracellular cytokine staining
- OSPs
overlapping synthetic peptides
- TILs
tumor-infiltrating lymphocytes
Introduction
The immune escape of cancer cells carrying permanently changing epitope composition due to genetic instability as well as our inability to brake immune tolerance or, in other words, to induce a destructive immune attack against the body's own ex "normal" now cancer cells, are considered as the major obstacles for cancer vaccines development.1,2 Nowadays, the cancer immunoediting theory, indicating that a permanent interaction between immune system and cancerous cells leads to the escape of certain cells from immune attack and to the selection of immune resistant or immunosuppressive cell populations, is largely accepted.3,4
Preclinical studies demonstrated that cellular immune responses, particularly cytotoxic T lymphocytes (CTLs), are major participants in elimination of tumor cells and in inhibition of tumor growth.5,6 While data from adoptive cell transfer-based immunotherapies indicate that T cells can mediate durable complete responses, even in the setting of advanced metastatic disease, it is clear that the successful cancer vaccine must confront: (i) corrupted tumor microenvironment containing regulatory T cells and aberrantly matured myeloid cells, (ii) a tumor-specific T cell repertoire prone to immunologic exhaustion and senescence, and (iii) highly mutable tumor targets capable of antigen (Ag) loss and immune evasion.7
Currently, many clinical trials are underway with the hope to show the therapeutic efficacy of vaccine candidates, previously tested in preclinical studies in animal models.5,8 Active immunogenic components of these vaccines are very diverse ranging from whole cancer cell lysates, protein fractions, cDNA expression libraries to recombinant proteins and synthetic peptides bearing defined epitopes.1,9-11 Similarly, the list of vaccine adjuvants and delivery systems along with diverse vaccination schedules is impressive: viral vectors, bacterial systems, modified/pulsed dendritic cells, plasmid DNA, RNA, nanoparticles, in vivo electroporation technology, molecular and numerous conventional adjuvants are being tested. In general, while subunit vaccines are safer, more stable and more suitable for mass production, they frequently provide lower protection compared with viral vectors or live attenuated vaccines, and typically induce humoral and not cellular immune response.12
In the field of cancer epitope vaccines, the modified, optimized or variant peptides, also known as altered peptide ligands (APLs), mimotopes, heteroclitic peptides or peptide analogs, bearing mutated versions of natural epitopes derived from tumor-associated antigens (TAAs) are considered to be promising candidates for the development of vaccines.13,14 Comprehensive screening strategies, such as testing virtually every single amino acid substitution within an epitope by genetic screen, may lead to identification of superagonist APLs capable of eliciting potent anti-tumor patient-specific CTL responses when the native or wild type (WT) tumor-associated epitope fails.15 Interestingly, central TCR-contact residues of antigenic peptides can be replaced even by non-peptidic units without loss of binding affinity to major histocompatibility complex (MHC) class-I molecules and T-cell triggering capacity.16 The direct approach to identify tumor epitopes is the analysis of surgically resected cancer tissues with respect to MHC-binding peptides and gene expression profiles.17 Recently, a novel approach that bypasses the need for epitope mapping, consisting in generation of a mixture of 34 overlapping synthetic peptides (OSPs) representing a tumor antigen, was successfully tested in a mouse TS/A breast carcinoma model.18 Another approach for identification of APLs was the generation of peptide epitopes/mimotopes through successive rounds of selection from a large (up to hundreds of billion members) positional scanning combinatorial peptide library that resulted in 2 APLs differing by 5 residues from the reference human telomerase reverse transcriptase-derived T cell epitope.19 Importantly, the selected epitopes were more effective than wild-type epitope in inducing cross-reactive CTL in HLA A2.1-transgenic mice. Also, systematic amino acid substitutions, generated using peptides simultaneously synthetized on derivatized cellulose membranes (SPOT synthesis), were shown to improve the efficiency of phage display-derived mimotope vaccination against mouse neuroblastoma.20 Vaccine immunogens bearing proteins that are highly homologous to their autologous counterparts or xenoantigens are a separate class of vaccine candidates, and were used in animal models and clinical trials.21 However, not always immune responses induced by xenoantigen are recognized by native Ag , thus imposing limitations for the development of this type of cancer vaccines.22
In order to avoid tumor escape, it is desirable to target a tumor Ag that is essential for tumor survival and expressed by tumors at high levels. One of these Ags is survivin, an oncogenic inhibitor-of-apoptosis protein, which is expressed at high levels in virtually all malignancies and is commonly referred to as a universal tumor Ag.23 Importantly, survivin-specific T-cell reactivity strongly correlates with tumor response and patient survival, as shown in a Phase II peptide vaccination trial in metastatic melanoma.24
Recently, we have reported a new concept for generation of vaccine immunogens termed as variable epitope library (VEL) that specifically targets genetically/antigenically variable pathogens.25,26 These new types of immunogens are combinatorial libraries bearing a mixtures of heavily mutated variants of immunodominant CTL epitope (30-50 % of amino acids at certain positions within the epitope are replaced by one of the 20 natural amino acids at each position). We have demonstrated that T cells induced by such immunogens recognized more than 50% of heavily mutated variants of HIV-1 gp120 V3 loop-derived cytotoxic T lymphocyte epitope, and that the sera from VEL-immunized mice were capable of neutralizing 5 out of 10 primary viral isolates from Tier 2 reference panel.26 These data indicate the feasibility of the application of immunogens based on VEL concept as an alternative and promising approach for the development of molecular vaccines against antigenically variable pathogens.
The objective of this proof-of-concept study was to test if VEL-based vaccine immunogens bearing variant epitope libraries of survivin-derived CTL epitope GWEPDDNPI may induce protective immunity against breast cancer in a highly tumorigenic, spontaneously metastasizing mouse 4T1 breast tumor model.27 We think that the targets of VEL-based vaccines may not be restricted to Ags showing high mutation rate, since the activation of a larger pool of T cells by immunization with VELs, probably carrying a number of peptide variants with improved HLA binding affinity, would also target a WT epitope.
Results
Immunogen Construction
We generated recombinant M13 phage expressing the survivin-derived H-2Dd-restricted wild-type CTL epitope23 GWEPDDNPI referred as FSWT, and SWT epitope variants bearing combinatorial VEL, designated as FSVL, using degenerate oligonucleotides encoding a library of epitope variants with structural composition GWXPXDXPI, where X is any of 20 natural amino acids. The DNA fragments corresponding to SWT and SVL, respectively, were amplified by PCR and cloned into pG8SAET phagemid vector25 that allows the expression of epitopes at high copy numbers as peptides fused to phage cpVIII. The amino acids at MHC-binding anchor positions were maintained within the epitope while mutations were introduced at positions responsible for interaction with TCR. Thus, each variant epitope has random amino acid substitutions (mutations) at 3 defined positions within the SWT and, the theoretical complexity of such library is 8 × 103 individual members. The FSVL phage library has a complexity of 10,500 original clones.
To check the correct cloning and to generate Ags for T-cell assays, 26 phage clones were randomly isolated from the FSVL, and the deduced amino acid sequences of corresponding peptides/epitopes were determined after DNA sequencing. As shown in Table 1, 12–16 different amino acids were detected at mutated particular amino acid positions in 26 epitope variants, indicating an acceptable epitope diversity of the generated library. In order to generate a panel of variant epitopes, we randomly isolated 87 phage clones from FSVL epitope library, and used them in T-cell assays. Also, a control non-related phage clone FB22 was used in immunization experiments.
Table 1.
Sequences of survivin-derived SWT epitope variants
| Wild-type epitope SWT | G | W | E | P | D | D | N | P | I |
|---|---|---|---|---|---|---|---|---|---|
| Epitope Library | G | W | Xa | P | X | D | X | P | I |
| Epitope Variants | |||||||||
| 1 | — | — | F | — | L | — | A | — | — |
| 2 | — | — | L | — | N | — | Y | — | — |
| 3 | — | — | R | — | T | — | V | — | — |
| 4 | — | — | F | — | L | — | N | — | — |
| 5 | — | — | I | — | S | — | F | — | — |
| 6 | — | — | Q | — | T | — | E | — | — |
| 7b | — | — | T | — | K | — | D | — | — |
| 8 | — | — | D | — | L | — | I | — | — |
| 9 | — | — | Q | — | M | — | S | — | — |
| 10 | — | — | I | — | T | — | A | — | — |
| 12 | — | — | C | — | Y | — | T | — | — |
| 22 | — | — | N | — | S | — | L | — | — |
| 25 | — | — | V | — | T | — | L | — | — |
| 38 | — | — | H | — | L | — | N | — | — |
| 41 | — | — | N | — | F | — | G | — | — |
| 45 | — | — | D | — | L | — | Q | — | — |
| 50 | — | — | A | — | N | — | N | — | — |
| 53 | — | — | V | — | D | — | Y | — | — |
| 58 | — | — | Q | — | V | — | R | — | — |
| 59 | — | — | E | — | T | — | H | — | — |
| 65 | — | — | C | — | Q | — | L | — | — |
| 73 | — | — | W | — | Q | — | E | — | — |
| 79 | — | — | F | — | L | — | V | — | — |
| 80 | — | — | V | — | Y | — | Y | — | — |
| 82 | — | — | R | — | V | — | P | — | — |
| 88 | — | — | T | — | I | — | R | — | — |
| Amino acid frequencies | 14/26 | 12/26 | 16/26 |
aX- any of 20 natural amino acids.
bThe clones marked in grey were used as Ag in T-Cell assays.
Anti-tumor Effects in Mice Vaccinated with VELs
Although the major goal in cancer vaccinology is the development of therapeutic vaccines, the analysis of immunogenic properties of VEL-based vaccines and the influence of the stimulation of immune system before tumor challenge allows to compare directly the molecular profiles of recognition of a panel of mutated variant epitopes by T cells in various states of complex interaction between growing tumor and immune system.
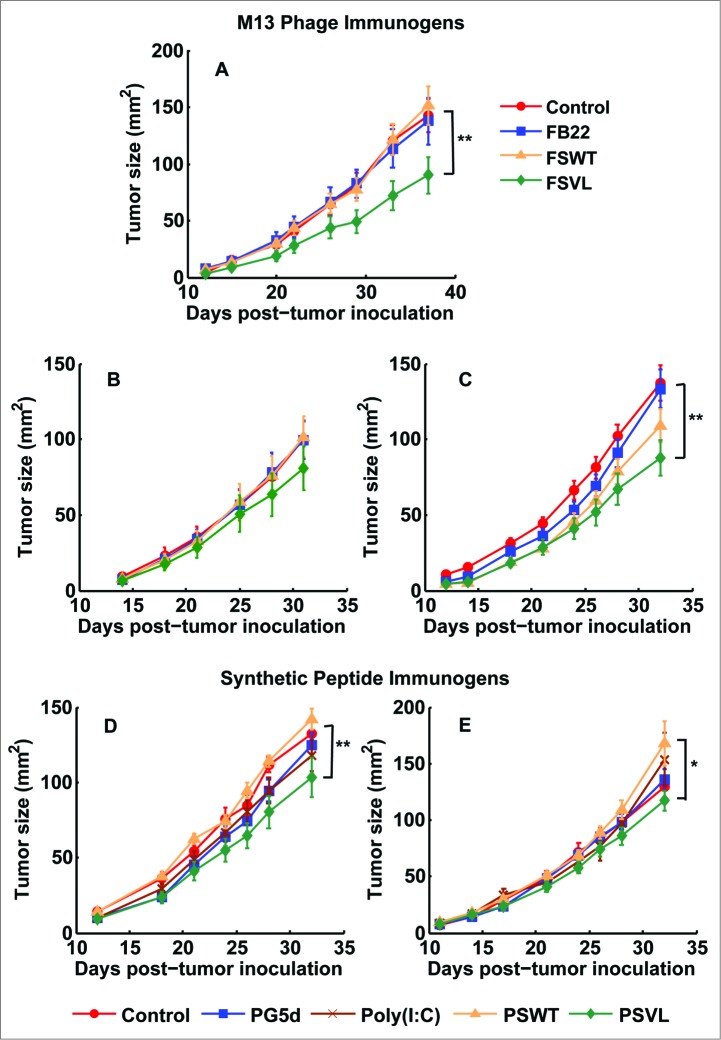
Therefore, we first examined whether prophylactic vaccination with VEL could be effective against syngeneic 4T1 tumor cell challenge in BALB/c mice. Mice were immunized with FSVL, FSWT and FB22 phages expressing VEL-bearing library, wild-type SWT epitope and the control non-related B22 epitope, respectively, by i.v. injection without any adjuvant as illustrated schematically in Figure 1. As a control, a group of non-immunized mice was included. Forty days after vaccination, all mice were inoculated with 104 4T1 tumor cells and monitored prospectively for development of tumors. As demonstrated in Figure 2 A, statistically significant (P < 0.05) tumor growth inhibition was observed in FSVL-vaccinated mice compared with mice immunized with FSWT and FB22 as well as with control non-immunized mice that received only 4T1 cell transplant.
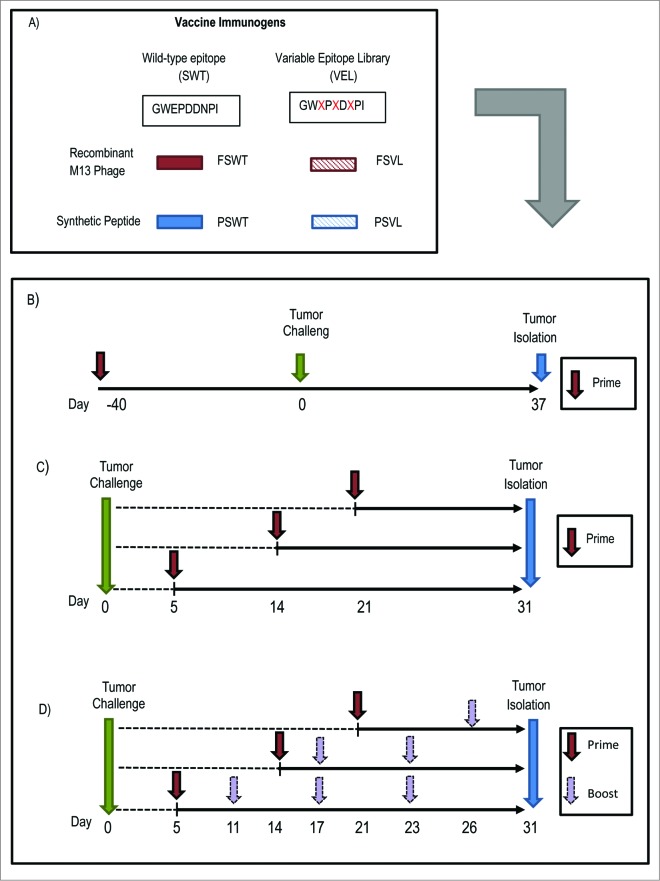
Figure 1.
Schematic diagram of generation of VEL-based immunogens and vaccination studies. (A) VELs were generated based on survivin-derived wild-type epitope (SWT) as M13 phage-displayed library (FSVL) and as synthetic peptide library (PSVL). SWT expressing phage FSWT and PSWT synthetic peptide were also used as immunogens. (B) In prophylactic vaccination setting BALB/c mice were primed by i.v. injection with FSVL, FSWT or control phage FB22, inoculated with 4T1 tumor cells on day 40 and sacrificed 37 days later. Tumors were measured every 2-3 days. (C) For therapeutic studies, after challenge mice were immunized i.v. once on day 5, 14 or 21 with above mentioned phage immunogens or subcutaneously (s.c.) with PSVL library, non-related peptide library and PSWT peptide. A separate group of untreated mice was included in this trial. (D) In separate studies, after tumor challenge and priming on day 5, 14 or 21mice received 3 (on days 11,17 and 23), 2 (on days 17 and 23) or one (on day 26) booster respectively, either with phage or synthetic peptide immunogens.
Figure 2.
Vaccination with VEL-based immunogens delays and inhibits breast tumor growth. (A) BALB/c mice (5-7 in each group) were immunized i.v. with FSVL, FSWT and non-related FB22 phage or left untreated, as described in Methods. After 40 days, all animals were inoculated with 4T1 tumor cells and the tumor growth was monitored by measuring tumor size till day 37. Growth rates of tumors are shown. In therapeutic study, mice were immunized with phage immunogens on day 5 after tumor inoculation by single i.v. injection and the tumors were measured till day 31 (B), or the mice were primed by s.c. immunization with PSVL, PG5D peptide libraries, poly (I:C) adjuvant alone or PSWT peptide on day 5 after tumor challenge (D). In separate studies, mice primed on day 5 after tumor challenge received 3 booster immunizations either with corresponding phage immunogen (C) or with synthetic peptide (E) on days 11, 17 and 23 post-tumor challenge. The data represent the average of at least 2 experiments, all error bars show ± s.e.m., ** indicates significance (P < 0.05), *indicates significance (P < 0.1).
To test anti-tumor effect of VELs on already established tumors in therapeutic setting, mice were transplanted with 4T1 cells and vaccinated with FSVL, FSWT and FB22 phages or PSWT (synthetic peptide corresponding to SWT epitope GWEPDDNPI), PSVL (SVL (GWXPXDXPI) peptide/epitope library) and PG5D (a control peptide/epitope library (A[G/F]PXXXXX[L/M]), by single i.v. and s.c. injection, respectively, on day 5 after tumor challenge (Fig. 2B and D). We observed inhibition of tumor growth in FSVL-vaccinated mice, although without reaching statistical significance, while vaccination with PSVL led to significant inhibition (P < 0.05) of tumor growth as compared with mice immunized with PSWT peptide or with non-immunized tumor-bearing mice (Fig. 2D). Also, some degree of the reduction in tumor size was obtained in mice immunized with control PG5D peptide library. Single vaccination of mice with phage and synthetic peptide immunogens on days 14 and 21 after tumor inoculation was not inhibitory for tumor growth (data not shown). Next, we investigated whether booster immunizations may enhance anti-tumor effects by vaccinating animals 3 more times on days 11, 17 and 23. As shown in, Fig. 2C 3 booster immunizations with FSVL improved the vaccine potency as compared with FSVL-priming data (Fig. 2B), resulting in statistically significant tumor growth inhibition (Fig. 2C). However, 3 booster immunizations with the same schedule of mice primed on day 5 with synthetic peptide immunogens did not lead to the increase in vaccine efficacy (Fig. 2E) compared with single therapeutic intervention (Fig. 2D). No significant anti-tumor effect was observed in mice bearing established (>10 mm2, day 14) or large (>40 mm2, day 21) tumors that were primed on days 14 or 21, and then received 2 or 1 booster inoculations, on days 17 and 23 or on day 26, respectively, by phage or synthetic peptides (data not shown). The immunized mice undergoing necropsy were also routinely assessed for evidence of autoimmunity and no abnormal lymphocytic infiltrates into organs were observed (data not shown). Collectively, these data indicate that vaccination with VELs in the form of recombinant M13 phage particles and synthetic peptides has statistically significant anti-tumor effect when applied prophylactically and therapeutically (at early stages) in mouse 4T1 mammary carcinoma model.
Cellular Immune Responses Induced by VEL
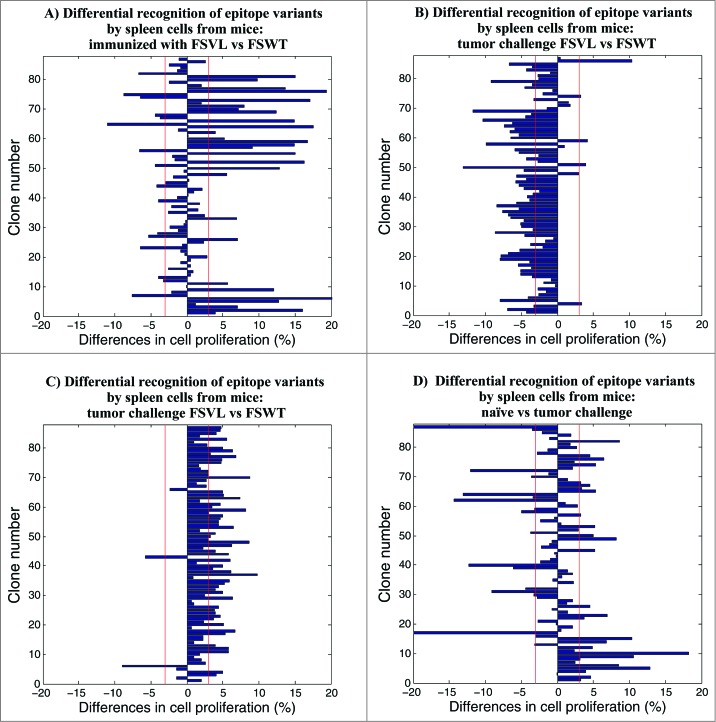
To test whether immunization with FSVL phage-displayed epitope library is able to elicit immune responses to CTL epitope variants in prophylactic setting, mice were immunized by a single injection with FSVL and FSWT, and some of these mice were challenged with 4T1 cells 40 days later while the rest of the mice were not transplanted with tumor cells. 15 days after the challenge, splenocytes were prepared from both groups, stimulated in vitro with 87 phage clones and the breadth (number of responding epitope variants) and the magnitude of total T-cell proliferative responses were measured by flow cytometry. To more clearly visualize differential recognition of epitope variants by spleen cells of mice immunized with FSVL and FSWT, instead of showing absolute numbers of cell proliferation data, we calculated differences observed between experimental groups.
As shown in Figure 3A, the immunization with FSVL library generated a pool of splenocytes with highly variable capacities of recognizing individual epitope variants. Thus, the levels of proliferation of the cells from mice immunized with FSVL and FSWT were 8.50-43.60 % and 9.61-28.41%, respectively (p = 0.006), and 29 out of 87 epitope variants (33 %) showed higher stimulatory capacity against cells from FSVL-immunized mice compared with FSWT-immunized mice (3-20% increase in percentages of cell proliferation). Only 14 epitope variants showed 3-11% increase in stimulatory capacity against cells from FSWT-immunized mice (Fig. 3A) (29/87 vs 14/87, p = 0.009). These data indicate the superior immunogenicity of FSVL over FSWT and the induction of long-lasting immune memory, which is in agreement with our previous study.25Interestingly, 18 epitope variants were better Ags than wild-type epitope-expressing FSWT phage. The FSWT phage, in turn, was more than 2-fold stronger stimulating Ag for cells from both FSVL- and FSWT-immunized mice than FB22 control phage and, the FB22 phage always led to only background levels of cell proliferation (data not shown). However, a strong inhibition of these immune responses was observed after tumor challenge and, almost all epitope variants were able to stimulate cells from mice immunized with FSWT more efficiently (more than 3 % of differences) than cells from FSVL-immunized mice (Fig. 3B) (59/87 vs 3/87 P < 0.0001). After tumor challenge, both FSWT and FB22 induced similar almost background level of cell proliferation against spleen cells from either FSVL- or FSWT-immunized mice (data not shown).
Figure 3.
Measurement of proliferative responses by CFSE dilution after immunization/challenge. The differences in cell proliferation (%) were calculated by subtracting values obtained in assays using spleen cells from FSWT-immunized mice from those obtained in assays using spleen cells from FSVL-immunized animals (A, B and C). (A) Mice were immunized with FSVL and FSWT, 40 day later the spleen cells were harvested and stimulated for 72 hours with the panel of 87 epitope variant-bearing phage clones, as well as non-related FB22 and SWT-expressing FSWT phage. (B) 40 days after priming, mice were inoculated with 4T1 tumor cells and, spleen cells collected on day 15 were stimulated as described above. (C) Mice were challenged with tumor cells and immunized with FSVL and FSWT on day 5, then on day 15 spleen cells were processed for cell proliferation. (D) Differences in cell proliferation (%) between non-immunized tumor-challenged and naïve mice. The spleen cells from untreated (tumor free) or 4T1-inoculated mice were harvested on day 15 post-tumor challenge and used in cell proliferation. The vertical red lines indicate arbitrary defined 3 % threshold for differences in cell proliferation. Data from one representative experiment out of 2 are shown.
To evaluate the ability of FSVL phage-displayed epitope library to elicit immune responses to CTL epitope variants in therapeutic setting, the groups of mice were immunized by a single injection with FSVL, FSWT or control FB22 phage, respectively, on day 5 after tumor challenge or were left naïve to any treatment. 15 days after the challenge, splenocytes were prepared, stimulated in vitro with 87 phage clones, and analyzed in lymphocyte proliferation assay. As shown in, Figure 3C the general levels of proliferation of cells from FSVL- and FSWT-immunized mice were lower (with absolute values 6.86–28.46 % and 12.14–27.64 %, respectively; P < 0.0001, not shown) than those obtained in prophylactic study. Cells from FSVL-immunized mice exhibited proliferative responses to 53 epitope variants (61 % of variants) with values of 3–10 % above those obtained with cells from FSWT-immunized mice (53/87 vs 2/87 P < 0.0001). The spleen cells from tumor-bearing mice immunized with control FB22 phage demonstrated only background levels of cell proliferation against almost all variant epitopes (data not shown) indicating the induction of epitope variant- and epitope-specific immune responses by VEL-bearing FSVL. Importantly, in this case, in the presence of established tumor, FSVL was superior as an immunogen compared with FSWT while in prophylactic vaccination we did not observe this phenomenon after tumor challenge. The recall responses against individual epitope variants were highly reproducible and, interestingly, 11 and 18 epitope variants resulted better stimulating Ags than FSWT wild-type epitope for cells from FSVL-and FSWT-immunized mice, respectively (data not shown). These data indicate that in prophylactic vaccination (prior to tumor challenge) and in therapeutic intervention, several epitope variants are more potent Ags than wild-type CTL epitope, acting as potential agonist Ags and, possibly, immunogens.
Finally, we investigated antigenic properties of the generated panel of epitope variants against spleen cells obtained from non-immunized control mice to measure a background level of immune recognition and to determine the changes induced in epitope reactivity profile after tumor challenge. While general level of proliferation of splenocytes from both groups of mice were similar (10.49–37.79 % and 11.73–37.73 %, respectively; p = 0.309, data not shown), the spontaneous cell-stimulating capacities of 27 epitope variants (31 % of total number) were diminished after tumor challenge according to decreased percentages of cell proliferation (differences between groups were 3–18 %) as shown in Figure 3D. On the other hand, 17 epitopes (19 % of total number) were preferentially activating cells from tumor-bearing mice as compared with control mice (3–21 % of differences) (27/87 vs 17/87 p = 0.083). Again, as in the case of tumor challenge of immunized mice described above, both FSWT and FB22 did not stimulate splenocytes from naïve or tumor-inoculated mice.
Together, these data demonstrated that vaccination with VELs induces specific anti-tumor cellular immune responses and suggested the superiority of VEL-based immunogens over immunogens carrying defined wild-type epitope in their capacity of inducing broad and potent immune responses without inducing harmful autoimmune reactions.
Phenotypic Analysis of Activated lymphocytes
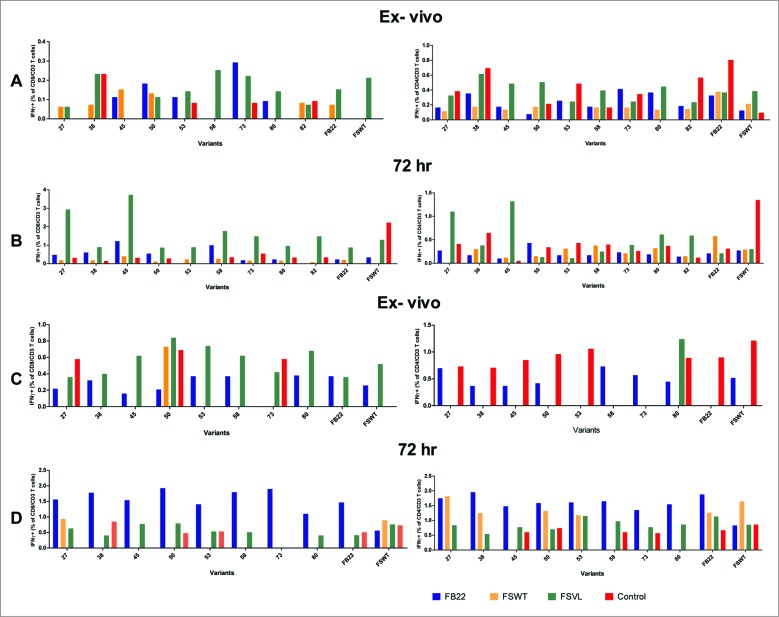
To determine the subpopulations of proliferating T cells and to test whether the immunization with VELs induces epitope-specific activation of both CD4+ IFN-γ+ and CD8+ IFN-γ+ cells, we performed flow cytometry and intracellular cytokine staining (ICS) assays (Fig. 4). The pooled splenocytes from each group of mice, used in prophylactic and therapeutic vaccine settings, were analyzed by FACS either after 6 hours of stimulation (ex vivo cells) or upon 3 days of incubation with 9 phage-displayed variant epitopes selected at random among 29 clones showing superior antigenic properties in a cell proliferation assay described above (Fig. 3A), as well as with FSWT wild-type epitope and non-related FB22 epitope.
Figure 4.
Flow cytometric analysis of spleen T cells. The splenocytes were isolated from mice and stimulated with Ags as described in Figure 3. CD4+ and CD8+ T-cell responses were evaluated by using intracellular staining (ICS) for IFN-γ both ex vivo and in vitro by stimulating fresh lymphocytes for 6 h or 72 hours, respectively. Spleen cells from mice of therapeutic vaccination groups challenged with tumor cells and immunized with FSVL and FSWT on day 5, were used for ex vivo (A) as well as for 72 hours (B) ICS assays. Splenocytes from mice used in prophylactic study (as described in Figure 3 B) were assessed for the presence of IFN-γ producing T cells ex vivo (C) or upon stimulation with variant epitopes (D). Data from one representative experiment out of 2 are shown.
In ICS assay with ex vivo cells in therapeutic vaccine setting, while the spleen cells collected from mice immunized with FSVL contained only small proportion of IFN-γ-producing CD8+ T cells (0.05-0.25 %), a strong, several-fold increase in percentages of epitope variant-specific CD8+IFN-γ+ cells was observed upon stimulation with several clones during 72 hr (48-, 14-, 7-, 6.7- and 21-fold increase for clones # 27, 45, 58, 73 and 82, respectively) (Fig. 4 A). The incubation of spleen cells with the rest of the variants as well as with the control FB22 resulted in less than 1 % of IFN-γ-producing cells (Fig. 4 B). In the case of CD4+IFN-γ+ cells, only 2 clones (27 and 45) were stimulatory for cells from FSVL-immunized mice with 2-3 fold increase in cell percentages.
A different pattern was observed when we analized cells from prophylactic vaccine groups. Thus, although the presence of epitope variant-specific CD8+IFN-γ+ ex vivo cells in FSVL-immunized mice was detected (clones 45, 53, 58 and 80 showed cell percentages above those obtained with the cells from mice immunized with FSWT, FB22 and control untreated mice) (Fig. 4 C), 72 hr incubation led only to slight increase in cell proliferation and, unexpectedly, the cell numbers were decreased with several epitopes (clones 53, 58, 73 and 80) (Fig. 4 D). The analysis of CD4+IFN-γ+ cells showed that despite the detection of this type of cells after stimulation with epitope variants (Fig. 4D), we could not conclude that these responses are truly epitope-specific due to the high level of background values obtained with the cells from control FB22-immunized mice as well as when FB22 was used as control Ag (Fig. 4 C and D).
Together, these results demonstrate that vaccination with the VEL induces both CD4+ and CD8+ effector cytokine-producing T cells and that several variant epitopes are better cell stimulators than FSWT epitope.
Immunohistochemical Analysis
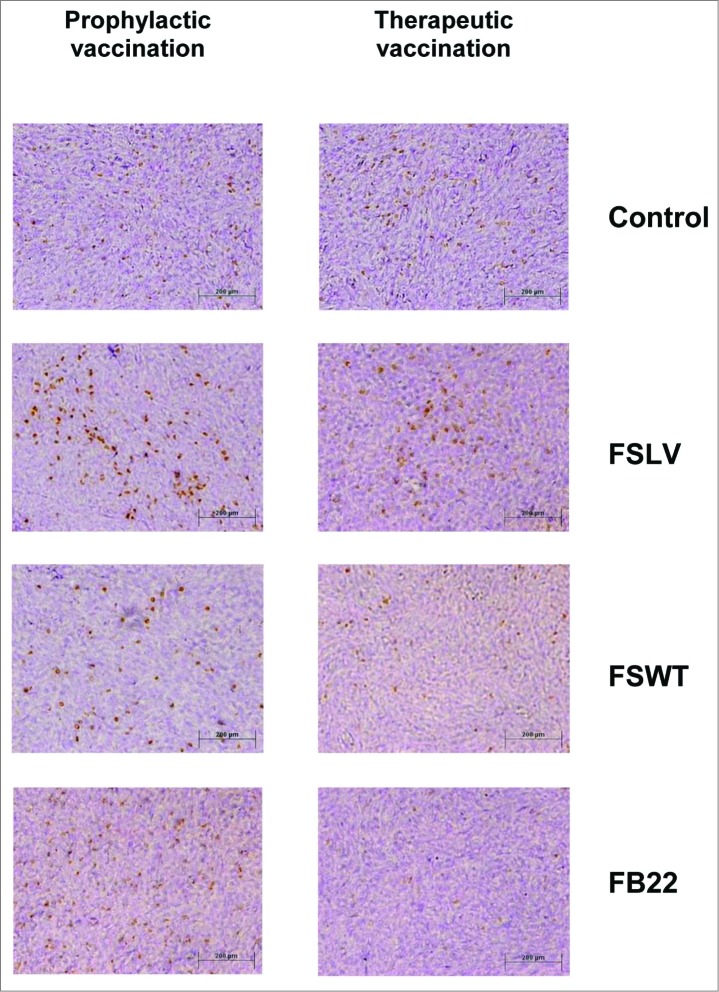
Next, we decided to test whether VEL-immunized mice had more tumor-infiltrating lymphocytes (TILs) compared to control mice. Tumor tissue sections obtained from both prophylactic and therapeutic vaccination studies were stained using anti-CD3 antibody. More CD3+ cells were observed in the tumors from mice vaccinated by a single inoculation with FSVL 40 days before tumor challenge compared with mice immunized with FSWT, FB22 or with mice transplanted with 4T1 cells without any treatment (Fig. 5, left panel). A similar pattern of lymphocytic infiltration was observed in mice vaccinated by prime/boost regimen using the same immunogens in therapeutic study (Fig. 5, right panel). Also, the presence of TILs was observed in mice vaccinated with corresponding synthetic peptide immunogens, although the general number of CD3+ T cells was lower compared with phage-vaccinated mice (data not shown). These data correlated well with tumor challenge studies, indicating possible involvement of these TILs in observed anti-tumor effects induced by vaccination with VELs.
Figure 5.
Tumor infiltrating T cells are increased in VEL-immunized mice. Extensive infiltration of CD3+ T cells was observed in tumors from FSVL-immunized mice in both prophylactic (left panel) and therapeutic (right panel) studies compared with tumors from mice immunized with FSWT, FB22 and treatment-naïve control mice. Scale bar: 200 μm.
Discussion
In this proof-of-principle study, we tested the prophylactic and therapeutic efficacy of vaccine immunogens bearing VELs of survivin-derived CTL epitope in an aggressive mouse 4T1 breast tumor model. The original hypothesis and the rationale for the generation of this new class of vaccines was that the immunization with VELs carrying a large number of epitope variants will lead to simultaneous presentation of altered/mutated epitopes to immune system and, consequently, to the activation of a broad pool of T cells capable of exerting anti-tumor effects. This aggressive cancer model was used, as we believe that the true measure of any candidate cancer vaccine's potency in preclinical studies should be their testing in this type of models and not in less aggressive and immunogenic tumor models, such as Ag-transfected cancer cells or genetically modified/engineered animals, that are nevertheless providing important information about general aspects of tumor biology, tumor microenvironment or particular genes involved in cancer development.
The anti-tumor effect observed in this study after a single inoculation with VELs is important taking into account that vaccine delivery context used was not the most potent one among currently existing approaches. Thus, we believe that the application of other modern vaccine delivery systems to VEL-based immunogens, such as in vivo DNA electroporation, Ag loaded dendritic cells, new viral vectors or molecular adjuvants, may produce more robust and protective anti-tumor immune responses.
Interestingly, we observed that booster vaccinations enhanced anti-tumor effect in the case of phage but not synthetic peptide immunogen (Fig. 2 C and E). We speculate that this may be a result of a relatively faster activation of T cells by peptide vaccines compared to phage immunogens. This supposition could also explain the better performance of PSVL synthetic peptide library compared to FSVL phage library in therapeutic studies after single vaccination of mice on day 5 post-tumor inoculation (Fig. 2B and D). Although PG5D peptide library was used as a control immunogen, the observed slight anti-tumor effect was not totally unexpected due to the high complexity of the library and the presence of H-2d haplotype-specific anchor amino acids within the epitope, probably leading to immune interference upon immunization. The failure of our phage and peptide immunogens to inhibit established tumors by priming on days 14 or 21 after challenge may be attributed to the larger size of these tumors. The recognition of a larger number of mutated epitope variants by spleen cells from FSVL-immunized mice compared with wild-tipe FSWT epitope-immunized mice (29 vs 14; Fig. 3 A), is a clear indication of the superior immunogenicity of FSVL library over FSWT epitope. Worth to mention that 18 epitope variants were better Ag stimulators than FSWT epitope itself for the cells from FSVL-immunized mice, suggesting that these epitope mutants are agonist Ags and, possibly, more potent immunogens. These data, along with our previous report with the VEL based on HIV-1 gp-120-derived CTL epitope 25 are reflecting, probably, a general feature for VEL-based immunogens. However, the observed pattern of epitope recognition was changed after tumor challenge leading to general inhibition of cell proliferation and to preferential recognition by variant epitopes of spleen cells from FSWT-immunized mice compared with FSVL-immunized animals (Fig. 3 B). We hypothesize that the observed inhibition of epitope variant-specific cell proliferation by tumor may be an indication of the presence of truly protective, but unfavorable for tumor, T-cell responses in PSVL-immunized mice prior to challenge. However, such inhibition did not substantially interfere with anti-tumor effect induced by immunization with the VEL bearing phage.
On the other hand, the cell proliferation assays carried out in therapeutic vaccination studies revealed another picture. The 61 % of epitope variants preferentially stimulated spleen cells from FSVL-immunized mice (Fig.3 C). In addition, 11 and 18 variant epitopes were better Ags for splenocytes from FSVL- and FSWT-immunized mice, respectively, compared to FSWT wild-type epitope (Fig. 3 C). These differences between prophylactic and therapeutic vaccine study groups may be the result of the presence of high number of cross-reactivity between tumor-specific and epitope variant-specific T cells in already tumor-bearing vs tumor naïve vaccinated mice. Importantly, despite the observed inhibition of T cell responses upon tumor challenge in prophylactic vaccination group, the induced cellular responses were protective in both experimental settings. Detailed vaccine studies using separate groups of T cell-stimulating epitope variants identified in this study, may allow the generation of novel potent vaccine immunogens, and experiments are underway.
Also, the phenotypic ICS analysis of T cells carried out using 9 variant epitope, selected at random, revealed that at least 5 of these variants were stimulating CD8+ IFN-γ+ cells and only 2 clones were stimulating CD4+ IFN-γ+ cells from therapeutically FSVL-immunized mice (Fig. 4 B). By contrast, when using cells of mice from prophylactic vaccination assays, 4 epitopes detected variant-specific CD8+ IFN-γ+ cells ex vivo, but in vitro proliferation led to only slight increase in cell proliferation and 4 other clones even induced inhibition of cell proliferation (Fig. 4 C and D). Although we have no explanation for these phenomena at present, these data reflect somehow the observed differences in variant epitope recognition patterns obtained in cell proliferation assays using spleen cells from 2 modes of vaccination (Fig. 3 A and B). Finally, we expect that the anti-tumor effects induced by VELs are mediated by SWT epitope- and epitope variant-specific T cells, including IFN-γ-producing lymphocytes, which might be among TILs detected in tumors from mice vaccinated with VEL immunogens (Fig. 5). We also believe, that the novel approach for epitope variant screening in a T-cell proliferation assay described in the present study, is opening several intriguing opportunities for further practical applications. For example, along with animal studies, it is attractive to test whether similarly generated panels of CTL epitope variants may permit the generation of molecular profiles (patterns) of epitope recognition using cancer patient-derived T cells. Undoubtedly, such studies may lead to the generation of novel Ags, involved in complex interplay between growing tumor and immune system, with diagnostic, prognostic and/or therapeutic value.
Despite the clear evidence that all tumors have mutator phenotype due to genomic/genetic instability, this issue was not addressed adequately and in sufficient detail in cancer vaccine research.28,29 Hence, considering that both the TCR repertoire and cancer-derived epitopes are of random origin, the VELs carrying random mutations are perfectly suited as immunogens capable of dealing with antigenic variability. The presence of thousands of random mutations within individual cell indicates that there are billions of mutations within any tumor and, even though the most highly malignant tumors may fail to exhibit a high mutation rate in the late stages of cancer, the imprints of the mutator phenotype should still be imbedded in the DNA of these tumors and should be evidenced by their elevated mutation frequency.28 On the other hand, there are data indicating that accelerated mutagenesis prevails late into tumor progression, and it is suggested that elevation of random mutation frequency in tumors might serve as a prognostic indicator.28 Moreover, a cDNA expression screening using cancer patient-derived lymphocyte-tumor cell cultures revealed that T cells against mutated epitopes clearly predominate, documenting a high degree of individuality for the cellular anti-tumor response.30 Individual cancers contain many mutant genes coding for unique Ags not present in normal tissues. Interestingly, epitope landscapes in breast and colorectal cancer were identified suggesting that with appropriate manipulation of the immune system, tumor cell destruction in situ may provide a polyvalent tumor vaccine, carrying a mixture of all mutated epitopes, without a requirement for knowledge of the targeted Ags.31 Currently, the use of deep sequencing technology to systematically analyze immunogenicity mutations at genome scale, as in the case of the mutanome of mouse melanoma cells, may lead to individualized immunotherapy for cancer patients.32
Since all thymically selected T cells are inherently auto- and cross-reactive, T lymphocytes recognizing self Ags expressed by cancer cells are prevalent in the immune repertoire. However, the activation of these cells is limited by weak signals that are incapable of fully priming naïve T cells.33 To overcome an immune tolerance, specific point mutations to create APLs were introduced into the gene encoding a non-immunogenic tissue self Ag expressed by melanoma, tyrosinase-related protein-1 (Tyrp1). Immunization of mice with mutated Tyrp1 DNA carrying 10 epitopes elicited cross-reactive T cell responses against multiple non-mutated epitopes leading to rejection of poorly immunogenic melanoma and prolonged survival in prophylactic and therapeutic studies.33 Also, the same authors introduced widespread random mutations into coding sequences of Tyrp1 and dopachrome tautomerase (Dct) cDNA and demonstrated that the immunization of melanoma bearing mice with these DNA libraries by gene gun induces autoimmune hypopigmentation and tumor immunity through cross-recognition of non-mutated gene products.34 These results confirm that the presentation of a vast repertoire of Ag variants to the immune system can enhance immune responses to self-epitope. We are convinced that despite the complex nature of the interplay between autoimmunity and protective anti-tumor immune responses described in these studies, they are reflecting a general rule: to be protective, anti-tumor immune responses should activate a large repertoire of T cells that should overlap with naturally activated T cell's repertoire. Indeed, recent reports from other groups confirmed that autoimmune-mediated activation of T cells may lead to efficient prophylaxis in mouse models of breast cancer and to growth control of established ovarian tumors, respectively.35,36The intriguing issue is the observed frequent targeting of ribosomal proteins by both autoimmune and anti-tumor T-cell responses by yet unclear mechanism.37
There is also a huge number of potential epitopes expressed in tumors and model systems that are encoded by non-primary open reading frame (ORF) sequences (frame-shift mutations) or derived from other non-traditional sources, such as transcriptional/translational mechanisms or splicing events, collectively referred as epitopes derived from defective ribosomal products (DRiPs).38 Thus, the pool of epitopes and corresponding TCRs in cancer is huge and of random origin, although many of these phenomena were not studied systematically and are not addressed sufficiently in current vaccine efforts. For this reason, the VELs carrying random mutations are perfectly suited as immunogens capable of dealing with antigenic variability. In other words, the new strategy proposed here attacks the diseases using their own weapons: rapid and random mutation rates.
Our vaccine concept closely resembles the natural course of tumor development in respect to interactions between immune system and rapidly evolving epitopes. The reason of failure of cancer vaccines is not the general lack of immune responses,but the progressive deficits in T-cell repertoires caused by evolving antigenic diversity, leading to activation of T cells that recognize non-protective epitopes and/or are unable to recognize new epitope variants. Our hypothesis is that simultaneous presentation of thousands of epitope variants to immune system will induce the activation of a broad range of T cells, both CTL and Th, including moderately auto-reactive T cells, representing natural repertoire of TCRs that overlaps with tumor-specific immune responses, thus offering an alternative way for breaking immune tolerance. Furthermore, due to the down regulation of MHC molecules in tumor cells, the immunization with VELs may lead to the selection of more "fit" TCRs as a result of competition between peptides for binding to MHC in tumors and the presence of unusually diverse and large pool of newly activated T cells.The VELs, particularly representing immunodominant epitopes, may serve as powerful vaccine components, since having the capacity to alter and activate diverse TCR pools, they will maintain T cell immunodominance hierarchies which is fundamental for the development of vaccines based on cellular responses. Indeed, the primary determinants of immunodominance are independent of TCR repertoire composition and diversity, as was recently shown in a mouse influenza infection model39 and, the number of effector T cells in the mammalian host adapts to immunological experience. Thus, the vaccines that abundantly introduce new memory CD8 T cells, like VEL-based vaccines, should not necessarily ablate pre-existing immunity to other infections or diseases.40
In conclusion, we have shown the feasibility of the generation of immunogens, based on incorporation of a large number of epitope variants in a single composition, capable of inducing a broad range of epitope variant-specific T-cell responses leading to statistically significant protection and therapeutic effect in an aggressive and non-immunogenic mouse 4T1 breast tumor model. To the best of our knowledge, the reported anti-tumor effects are the best observed to date, since there are no reports describing the application of epitope/peptide vaccines in this cancer model with comparable results. Importantly, the VELs can be easily constructed based on epitopes and/or on epitope-rich regions from any cancer-related antigen. Finally, we believe that the clear demonstration of the superiority of VEL-based vaccines over wild-type epitope counterparts in this study along with the relative simplicity of the VEL´s design and generation are pointing to the possibility of immediate practical use of this novel vaccine concept. The application of VEL epitope/peptide libraries in cancer vaccine trials, if not instead of, but at least in parallel with candidate native peptide vaccines, is worth to be considered.
Materials and Methods
Immunogens
To generate the immunogens, molecular biology procedures were carried out using standard protocols. Restriction enzymes, Taq DNA polymerase, DNA isolation/purification kits, T4 DNA ligase and M13KO7 helper phage were obtained from, Promega (Madison, WI, USA), Invitrogen (Carlsbad, CA, USA), IDT (San Diego, CA, USA), Sigma-Aldrich (St. Louis, MO, USA) or Qiagen (Valencia, CA, USA).
In order to express the survivin-derived wild-type CTL peptide epitope GWEPDDNPI (aa 66-74, SWT) and SWT epitope variants (GWXPXDXPI) bearing VEL (SVL), on M13 phage surface as fusions with major phage coat protein (cpVIII), the corresponding DNA fragments have been generated by PCR and cloned in pG8SAET phagemid vector as previously described.25 Briefly, 2 oligonucleotides (oligos):
5′-gtatattactgtgcgggttgggaaccagatgataatccaatatggggccagggaacc-3′ and degenerate 5′-gtatattactgtgcgggttggNNKccaNNKgatNNKccaatatggggccagggaacc-3′, (N is g, a, t or c and, K is g or c nucleotide) were used in 2 separate PCRs with pair of primers carrying Nco I and Bam HI restriction sites (underlined in oligos), 5DAMP: 5-′tgatattcgtactcgagccatggtgtatattactgtgcg-3′ and 3DAMP: 5′-atgattgacaaagcttggatccctaggttccctggcccca-3′ to generate corresponding DNA fragments for their cloning in phagemid vector using electroporation procedure.
We verified the correct PCR and cloning by DNA sequencing performed on a Genetic Analyzer ABI PRISM 3100 (Applied Biosystems, Foster City, CA, USA) using miniprep-purified (Qiagen Inc.., Santa Clarita, CA, USA) double-stranded DNA and the primers 5DAMP and 3DAMP.
The resulting recombinant phage clone FSWT expressing SWT epitope and phage library carrying SWT-based VEL, referred as FSVL, were rescued/amplified using M13KO7 helper phage by infection of Escherichia coli TG1 cells and purified by double precipitation with polyethylene glycol (20 % PEG/2.5 M NaCl) as described.25 Similarly, 87 phage clones randomly selected from FSVL library and expressing different epitope variant, were rescued/amplified from 0.8 mL of bacterial cultures using 96 well 1 mL round bottom blocks (Corning, Inc., Corning, NY, USA). The typical phage yields were 1010 to 1011 colony forming units (CFU) per milliliter of culture medium. As a control phage immunogen, a non-related phage FB22 expressing HIV-1 Gag-derived epitope/peptide ALQRLFETC was used.25 The DNA inserts of 27 phage clones from FSVL library were sequenced and the amino acid sequences of the peptides were deduced (Table 1).
The synthetic peptides corresponding to SWT epitope (GWEPDDNPI) and SVL (GWXPXDXPI) peptide/epitope library, bearing the complexity of 8 × 103 individual members, designated as PSWT and PSVL, respectively, as well as a control peptide/epitope library PG5D (A[G/F]PXXXXX[L/M]) with theoretical complexity of 3.2 × 106 individual members wer prepared at GenScript Corporation (Piscataway, NJ, USA).
Ethics Statement
Experiments were conducted following approved protocols from Animal Care and Use Committee of the Instituto de Investigaciones Biomedicas, Universidad Nacional Autonoma de Mexico and in accordance with the recommendations in the Guide for the Care and Use of laboratory Animals of the NIH. Mice were euthanized with CO2. All efforts were made to minimize suffering during experiments.
Animal Studies
4T1 mouse mammary carcinoma cells (American Type Culture Collection) were maintained for a limited time in vitro by passage in RPMI-1640 medium (Gibco®, Grand Island, NY, USA) supplemented with 10% FBS (Gemini Bio-Products, Woodland, CA, USA) containing penicillin (100 U/ml), streptomycin (100 μg/ml) and fungizone (0.75 μg/ml). Groups of 5–7 female 4 to 6 weeks old BALB/c mice were used. To generate breast tumors, mice were injected subcutaneously (s.c.) with 104 viable 4T1 cells in 50 μL of PBS into the right mammary fat pad. Primary tumors were detected by palpation within 1-2 weeks, the mice were observed every 3 day to monitor tumor growth, tumor area was calculated as length x breadth using Vernier calipers and mice were euthanized with CO2 31 days after 4T1 inoculation. For the prevention study, mice were immunized with 5 × 1012 FSWT, FB22 and FSVL recombinant M13 phage particles (5 × 1012 CFU) in 200 μL of PBS by i.v. injection into tail vain, and then mice were inoculated with 4T1 cells on day 40 post-vaccination. The tumor growth was monitored as described above. For the established disease study, mice were immunized once with above mentioned phage particles by i.v. injection or with 150 μg of synthetic peptides PSWT, PSVL and PG5D plus 150 μg of polynosine-polycytidylic acid (Poly (I:C), Sigma-Aldrich) in 100 μl of phosphate-buffered saline (PBS) by s.c injection on days 5, 14 and 21 of tumor challenge. In separate studies, the mice primed as described above received 3 (on days 11, 17 and 23 post-tumor cell injection), 2 (on days 17 and 23) or one (on day 26) booster vaccinations with phage or peptide immunogens (5 × 1011 CFU and 100 μg of synthetic peptides, respectively).
Cell Proliferation Assays
Splenocytes were pooled from 3 animals from each treatment group on day 15 after immunization or tumor challenge and tested using standard flow cytometry protocols. Cells were resuspended in RPMI-1640 medium supplemented as described above plus 1% sodium pyruvate, 1% nonessential amino acids and 1% 2-mercaptoethanol, washed twice with PBS and re-suspended at 5 × 107 cells ml−1 in 5 μM CFSE (Sigma-Aldrich) for 10 min at room temperature. After washing again 2 times with 10 mL of PBS+5 % FBS at 4°C, the cells were stimulated by culturing in a 96-well flat-bottom plate (2.5 × 105 cells/well) with 1 × 1010 phage particles/well corresponding to particular epitope variant for 72 h at 37°C in CO2 incubator. The gating strategy involved exclusion of doublets and dead cells and , 10,000 lymphocytes (R1) were gated for a CD4+ versus CD8+ dot-plot graph to measure CD4+ IFN-γ +, CD8+ IFN-γ + and proliferation percentages of CD4+CD8- and CD4-CD8+ cells.
Total cell proliferation and CD4+ and CD8+ T-cell responses were evaluated by using intracellular staining (ICS) for IFN-γ both ex vivo and in vitro by stimulating fresh lymphocytes for 6 h or 72 h, respectively. During the last 4 h, 1 μl/well Monensin (2 μM) (Sigma-Aldrich) was added to the culture. The cells were stained with fluorescence-labeled monoclonal antibodies against CD4 and CD8 (BioLegend, San Diego, CA, USA ) for 30 min at room temperature, fixed with fixation buffer and, after washing, the cells were permeabilized with permeabilization wash buffer (BioLegend), then labeled for 30 min with anti-IFN-γ antibody (BioLegend) in the dark. The cells were analyzed on FACS Calibur Cytometer using CellQuest software (BD Biosciences, San Jose, CA, USA); at least 10,000 events were collected.
Immunohistochemical (IHC) Studies
The tumors were removed at day 31 post-tumor injection. The excised tumors were fixed in 4 % buffered formalin for 12 hr at 4°C. Twenty micrometer-thick free-floating sections were processed using standard protocols. Hydrogen peroxide-quenched and blocked sections were incubated overnight at 4°C with anti-CD3 primary antibody (Clone 17A2, dilution 1:500, BioLegend). Sections were washed and incubated with HRP Goat anti-rat IgG antibody (dilution 1:800, Invitrogen) for 1 h at room temperature. After multiple washes, color development was performed using liquid DAB + substrate chromogen System (Dako, Carpinteria, CA, USA). Samples were placed onto glass slides (Corning), stained with hematoxylin (Biocare, Concord, CA. USA), dehydrated with xylene, and covered with Entellan mounting medium (Merk, Darmastad, Germany). Samples were viewed on Olympus BX51 microscope equipped with Olympus DP71 digital camera.
Statistical Analysis
All results are expressed as the means ± s.e.m. Mouse sample group sizes were at least n = 5. All experiments were repeated at least once with comparable results. Tumor size data were analyzed using repeated measurements analysis with Duncan´s "post-hoc" test for multiple comparisons. Differences were considered significant at P < 0.05. Cell proliferation (%) data were analyzed with 2 tailed t-test. SAS 9.0 software was used for statistical analysis. Proportions of epitope variants were compared using 'z' 2 tailed test; STATISTICA 8.0 was used in this analysis.
Acknowledgments
Authors thank Patricia de la Torre, Alejandro Marmolejo and Carlos Castellanos for technical assistance and Georgina Diaz Herrera for animal care.
Disclosure of Potential Conflicts of Interest
No potential conflicts of interest were disclosed.
Funding
Funding provided by DGAPA-UNAM (216110) and CON-ACyT (166600), MEXICO, to KM. ANDR is a recipient of a scholarship from CONACyT and Posgrado en Ciencias Biologicas, UNAM.
References
- 1.Koos D, Josephs SF, Alexandrescu DT, Chan RC, Ramos F, Bogin V, Gammil V, Dasanu CA, De Necochea-Campion R, Riordan NH, et al. Tumor vaccines in 2010: need for integration. Cell Immunol 2010; 263(2):138-47; PMID:20434139; http://dx.doi.org/ 10.1016/j.cellimm.2010.03.019 [DOI] [PubMed] [Google Scholar]
- 2.Baitsch L, Fuertes-Marraco SA, Legat A, Meyer C, Speiser DE. The three main stumbling blocks for anticancer T cells. Trends Immunol 2012; 33:364-72; PMID:22445288; http://dx.doi.org/ 10.1016/j.it.2012.02.006 [DOI] [PubMed] [Google Scholar]
- 3.Koebel CM, Vermi W, Swann JB, Zerafa N, Rodig SJ, Old LJ, Smyth MJ, Schreiber RD. Adaptive immunity maintains occult cancer in an equilibrium state. Nature 2007; 450:903-7; PMID:18026089; http://dx.doi.org/ 10.1038/nature06309 [DOI] [PubMed] [Google Scholar]
- 4.O'Sullivan T, Saddawi-Konefka R, Vermi W, Koebel CM, Arthur C, White JM, Uppaluri R, Andrews DM, Ngiow SF, Teng MW, et al. Cancer immunoediting by the innate immune system in the absence of adaptive immunity. J Exp Med 2012; 209:1869-82; PMID:22927549; http://dx.doi.org/ 10.1084/jem.20112738 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Dranoff G. Experimental mouse tumour models: what can be learnt about human cancer immunology?. Nat Rev Immunol 2011; 12:61-6; PMID:22134155 [DOI] [PubMed] [Google Scholar]
- 6.Mahmoud SM, Paish EC, Powe DG, Macmillan RD, Grainge MJ, Lee AH, Ellis IO, Green AL. Tumor-infiltrating CD8+ lymphocytes predict clinical outcome in breast cancer. J Clin Oncol 2011; 29:1949-55; PMID:21483002; http://dx.doi.org/ 10.1200/JCO.2010.30.5037 [DOI] [PubMed] [Google Scholar]
- 7.Klebanoff CA, Acquavella N, Yu Z, Restifo NP. Therapeutic cancer vaccines: are we there yet?. Immunol Rev 2011; 239:27-44; PMID:21198663; http://dx.doi.org/ 10.1111/j.1600-065X.2010.00979.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Topalian SL, Weiner GJ, Pardoll DM. Cancer immunotherapy comes of age. J Clin Oncol 2011; 29:4828-36; PMID:22042955; http://dx.doi.org/ 10.1200/JCO.2011.38.0899 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kottke T, Errington F, Pulido J, Galivo F, Thompson J, Wongthida P, Diaz RM, Chong H, Ilett E, Chester J, et al. Broad antigenic coverage induced by vaccination with virus-based cDNA libraries cures established tumors. Nat Med 2011; 17:854-9; PMID:21685898; http://dx.doi.org/ 10.1038/nm.2390 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Willers J, Capone G, Lucchese A. Peptides: an arrival point in cancer vaccinology. Front Biosci 2012; 4:1381-92; PMID:22652880; http://dx.doi.org/ 10.2741/S340 [DOI] [PubMed] [Google Scholar]
- 11.Yamada A, Sasada T, Noguchi M, Itoh K. Next-generation peptide vaccines for advanced cancer. Cancer Sci 2013; 104:15-21; PMID:23107418; http://dx.doi.org/ 10.1111/cas.12050 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Burchill MA, Tamburini BA, Pennock ND, White JT, Kurche JS, Kedl RM. T cell vaccinology: exploring the known unknowns. Vaccine 2013; 31:297-305; PMID:23137843; http://dx.doi.org/ 10.1016/j.vaccine.2012.10.096 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bernatchez C, Zhu K, Li Y, Andersson H, Ionnides C, Fernandez-Viña M, Cano P, Copper L, Abbruzzese J, Hwu P, et al. Altered decamer and nonamer from an HLA-A0201-restricted epitope of surviving differentially stimulate T-cell responses in different individuals. Vaccine 2011; 29:3021-30; PMID:21320548; http://dx.doi.org/ 10.1016/j.vaccine.2011.01.115 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Jordan KR, Buhrman JD, Sprague J, Moore BL, Gao D, Kappler JW, Slansky JE. TCR hypervariable regions expressed by T cells that respond to effective tumor vaccines. Cancer Immunol Immunother 2012; 61:1627-38; PMID:22350070; http://dx.doi.org/ 10.1007/s00262-012-1217-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Abdul-Alim CS, Li Y, Yee C. Conditional superagonist CTL ligands for the promotion of tumor-specific CTL responses. J Immunol 2010; 184:6514-21; PMID:20483791; http://dx.doi.org/ 10.4049/jimmunol.0900448 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Douat-Casassus C, Marchand-Geneste N, Diez E, Gervois N, Jotereau F, Quideau S. Synthetic anticancer vaccine candidates: rational design of antigenic peptide mimetics that activate tumor-specific T-cells. J Med Chem. 2007; 50:1598-609; PMID:17328535; http://dx.doi.org/ 10.1021/jm0613368 [DOI] [PubMed] [Google Scholar]
- 17.Neumann A, Hörzer H, Hillen N, Klingel K, Schmid-Horch B, Bühring HJ, Rammensee HG, Aebert H, Stevanovic S. Identification of HLA ligands and T-cell epitopes for immunotherapy of lung cancer. Cancer Immunol Immunother 2013; 62:1485-97; PMID:23817722; http://dx.doi.org/ 10.1007/s00262-013-1454-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Mirshahidi S, Kramer VG, Whitney JB, Essono S, Lee S, Dranoff G, Anderson KS, Ruprecht RM. Overlapping synthetic peptides encoding TPD52 as breast cancer vaccine in mice: prolonged survival. Vaccine 2009; 27:1825-33; PMID:19201387; http://dx.doi.org/ 10.1016/j.vaccine.2009.01.089 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hernández J, Schoeder K, Blondelle SE, Pons FG, Lone YC, Simora A, Langlade-Demoyen P, Wilson DB, Zaneti M. Antigenicity and immunogenicity of peptide analogues of a low affinity peptide of the human telomerase reverse transcriptase tumor antigen. Eur J Immunol 2004; 34:2331-41; PMID:15259031; http://dx.doi.org/ 10.1002/eji.200425134 [DOI] [PubMed] [Google Scholar]
- 20.Bleeke M, Fest S, Huebener N, Landgraf C, Schraven B, Gaedicke G, Volkmer R, Lode HN. Systematic amino acid substitutions improved efficiency of GD2-peptide mimotope vaccination against neuroblastoma. Eur J Cancer 2009; 45:2915-21; PMID:19695868; http://dx.doi.org/ 10.1016/j.ejca.2009.07.026 [DOI] [PubMed] [Google Scholar]
- 21.Suresh K, Scheid E, Klotz L, Venkateswaran V, Gauldie J, Foley R. Induction of specific human cytotoxic T cells using dendritic cells transduced with an adenovector encoding rat epidermal growth factor receptor 2. Int J Oncol 2011; 39:907-13; PMID:21769423 [DOI] [PubMed] [Google Scholar]
- 22.Johnson LE, Frye TP, McNeel DG. Immunization with a prostate cancer xenoantigen elicits a xenoantigen epitope-specific T-cell response. Oncoimmunology 2012; 1:1546-56; PMID:23264901; http://dx.doi.org/ 10.4161/onci.22564 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Siegel S, Wagner A, Schmitz N, Zeis M. Induction of anti-tumour immunity using survivin peptide-pulsed dendritic cells in a murine lymphoma model. Br J Haematol 2003; 122:911-4; PMID:12956760; http://dx.doi.org/ 10.1046/j.1365-2141.2003.04535.x [DOI] [PubMed] [Google Scholar]
- 24.Becker JC, Andersen MH, Hofmeister-Müller V, Wobser M, Frey L, Sandig C, Walter S, Singh-Jasuja H, Kampgen E, Opitz A, et al. Survivin-specific T-cell reactivity correlates with tumor response and patient survival: a phase-II peptide vaccination trial in metastatic melanoma. Cancer Immunol Immunother 2012; 61:2091-103; PMID:22565484; http://dx.doi.org/ 10.1007/s00262-012-1266-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Pedroza-Roldan C, Charles-Niño C, Saavedra R, Govezensky T, Vaca L, Avaniss-Aghajani E, Gevorkian G, Manoutcharian K. Variable epitope library-based vaccines: shooting moving targets. Mol Immunol 2009; 47:270-82; PMID:19853920; http://dx.doi.org/ 10.1016/j.molimm.2009.09.024 [DOI] [PubMed] [Google Scholar]
- 26.Charles-Niño C, Pedroza-Roldan C, Viveros M, Gevorkian G, Manoutcharian K. Variable epitope libraries: new vaccine immunogens capable of inducing broad human immunodeficiency virus type 1-neutralizing antibody response. Vaccine 2011; 29:5313-21; PMID:21600948; http://dx.doi.org/ 10.1016/j.vaccine.2011.05.007 [DOI] [PubMed] [Google Scholar]
- 27.Pulaski, BA and Ostrand-Rosenberg, S. 2001. Mouse 4T1 Breast Tumor Model. Current Protocols in Immunology. 39:20.2:20.2.1–20.2.16. [DOI] [PubMed] [Google Scholar]
- 28.Bielas JH, Loeb KR, Rubin BP, True LD, Loeb LA. Human cancers express a mutator phenotype. Proc Natl Acad Sci U S A 2006; 103:18238-42; PMID:17108085; http://dx.doi.org/ 10.1073/pnas.0607057103 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Fox EJ, Prindle MJ, Loeb LA. Do mutator mutations fuel tumorigenesis? Cancer Metastasis Rev 2013; 32:353-61; PMID:23592419; http://dx.doi.org/ 10.1007/s10555-013-9426-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Lennerz V, Fatho M, Gentilini C, Frye RA, Lifke A, Ferel D, Wolfel C, Huber C, Wolfel T. The response of autologous T cells to a human melanoma is dominated by mutated neoantigens. Proc Natl Acad Sci U S A 2005; 102:16013-8; PMID:16247014; http://dx.doi.org/ 10.1073/pnas.0500090102 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Segal NH, Parsons DW, Peggs KS, Velculescu V, Kinzler KW, Vogelstein B, Allison JP. Epitope landscape in breast and colorectal cancer. Cancer Res 2008; 68:889-92; PMID:18245491; http://dx.doi.org/ 10.1158/0008-5472.CAN-07-3095 [DOI] [PubMed] [Google Scholar]
- 33.Castle JC, Kreiter S, Diekmann J, Löwer M, van de Roemer N, de Graaf J, Selmi A, Diken M, Boegel S, Paret C, et al. Exploiting the mutanome for tumor vaccination. Cancer Res 2012; 72:1081-91; PMID:22237626; http://dx.doi.org/ 10.1158/0008-5472.CAN-11-3722 [DOI] [PubMed] [Google Scholar]
- 33.Guevara-Patiño JA, Engelhorn ME, Turk MJ, Liu C, Duan F, Rizzuto G, Cohen AD, Merghoub T, Wolchok JD, Houghton AN. Optimization of a self antigen for presentation of multiple epitopes in cancer immunity. J Clin Invest 2006; 116:1382-90; PMID:16614758; http://dx.doi.org/ 10.1172/JCI25591 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Engelhorn ME, Guevara-Patiño JA, Noffz G, Hooper AT, Lou O, Gold JS, Kappel BJ, Houghton AN. Autoimmunity and tumor immunity induced by immune responses to mutations in self. Nat Med 2006; 12:198-206; PMID:16444264; http://dx.doi.org/ 10.1038/nm1363 [DOI] [PubMed] [Google Scholar]
- 35.Jaini R, Kesaraju P, Johnson JM, Altuntas CZ, Jane-Wit D, Tuohy VK. An autoimmune-mediated strategy for prophylactic breast cancer vaccination. Nat Med 2010; 16:799-803; PMID:20512124; http://dx.doi.org/ 10.1038/nm.2161 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Altuntas CZ, Jaini R, Kesaraju P, Jane-wit D, Johnson JM, Covey K, Flask CA, Dutertre M, Picard JY, Tuohy VK. Autoimmune mediated regulation of ovarian tumor growth. Gynecol Oncol 2012; 124:98-104; PMID:22004903; http://dx.doi.org/ 10.1016/j.ygyno.2011.09.023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Philip M, Schietinger A, Schreiber H. Ribosomal vs. non-ribosomal cellular antigens: factors determining efficiency of indirect presentation to CD4+ T cells. Immunology 2010; 130:494-503; PMID:20331471; http://dx.doi.org/ 10.1111/j.1365-2567.2010.03258.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Li Y, Wang LX, Pang P, Cui Z, Aung S, Haley D, Fox BA, Urba WJ, Hu HM. Tumor-derived autophagosome vaccine: mechanism of cross-presentation and therapeutic efficacy. Clin Cancer Res 2011; 17:7047-57; PMID:22068657; http://dx.doi.org/ 10.1158/1078-0432.CCR-11-0951 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Kedzierska K, Guillonneau C, Gras S, Hatton LA, Webby R, Purcell AW, Rossjohn J, Doherty PC, Turner SJ. Complete modification of TCR specificity and repertoire selection does not perturb a CD8+ T cell immunodominance hierarchy. Proc Natl Acad Sci U S A 2008; 105:19408-13; PMID:19047637; http://dx.doi.org/ 10.1073/pnas.0810274105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Vezys V, Yates A, Casey KA, Lanier G, Ahmed R, Antia R, Masopust D. Memory CD8 T-cell compartment grows in size with immunological experience. Nature 2009; 457:196-9; PMID:19005468; http://dx.doi.org/ 10.1038/nature07486 [DOI] [PubMed] [Google Scholar]