Abstract
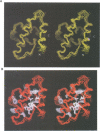
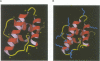
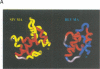
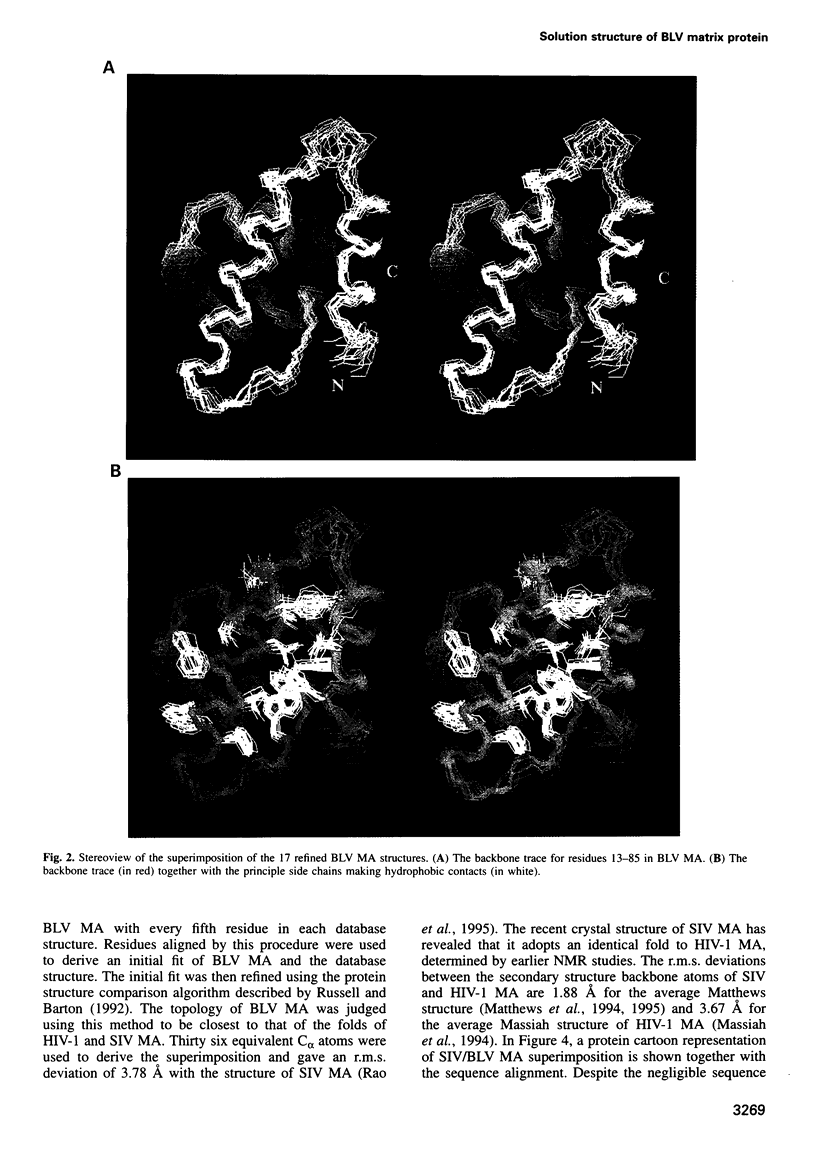
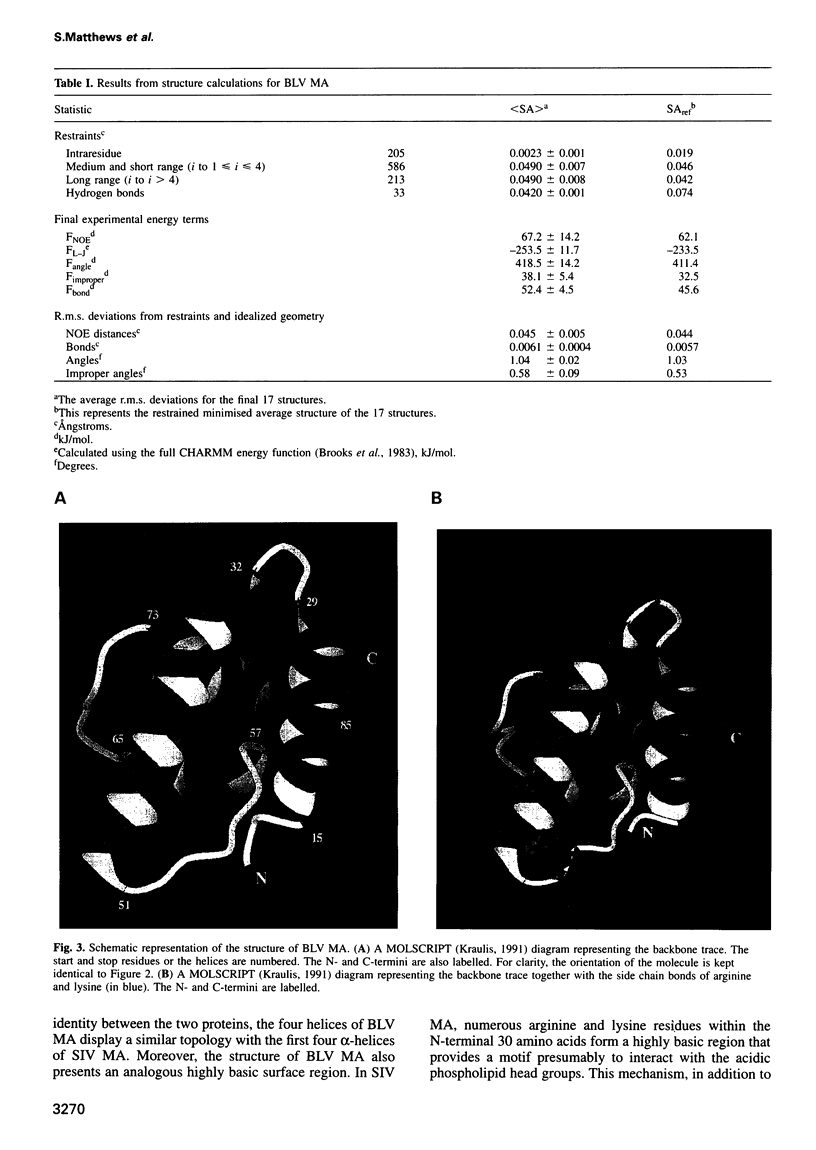
In the mature virion, retroviral matrix proteins are found in association with the inner face of the viral membrane. They play a critical role in determining the morphogenesis of virus assembly. We have determined the three-dimensional solution structure of the bovine leukaemia virus (BLV) matrix protein by heteronuclear nuclear magnetic resonance. The protein contains four principal helices that are joined by short, partially structured loops. Despite no sequence similarity with the lentiviruses, the structure shows an intriguing homology with the equivalent protein from the human and simian immunodeficiency viruses. A root-mean-square deviation of 3.78 angstrom is observed over the backbone atoms of 36 equivalent helical positions. The similarity implies a possible common assembly unit for the matrix proteins of type C retroviruses.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bukrinsky M. I., Haggerty S., Dempsey M. P., Sharova N., Adzhubel A., Spitz L., Lewis P., Goldfarb D., Emerman M., Stevenson M. A nuclear localization signal within HIV-1 matrix protein that governs infection of non-dividing cells. Nature. 1993 Oct 14;365(6447):666–669. doi: 10.1038/365666a0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bukrinsky M. I., Sharova N., McDonald T. L., Pushkarskaya T., Tarpley W. G., Stevenson M. Association of integrase, matrix, and reverse transcriptase antigens of human immunodeficiency virus type 1 with viral nucleic acids following acute infection. Proc Natl Acad Sci U S A. 1993 Jul 1;90(13):6125–6129. doi: 10.1073/pnas.90.13.6125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dorfman T., Bukovsky A., Ohagen A., Höglund S., Göttlinger H. G. Functional domains of the capsid protein of human immunodeficiency virus type 1. J Virol. 1994 Dec;68(12):8180–8187. doi: 10.1128/jvi.68.12.8180-8187.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Driscoll P. C., Clore G. M., Marion D., Wingfield P. T., Gronenborn A. M. Complete resonance assignment for the polypeptide backbone of interleukin 1 beta using three-dimensional heteronuclear NMR spectroscopy. Biochemistry. 1990 Apr 10;29(14):3542–3556. doi: 10.1021/bi00466a018. [DOI] [PubMed] [Google Scholar]
- Ehrlich L. S., Agresta B. E., Carter C. A. Assembly of recombinant human immunodeficiency virus type 1 capsid protein in vitro. J Virol. 1992 Aug;66(8):4874–4883. doi: 10.1128/jvi.66.8.4874-4883.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fäcke M., Janetzko A., Shoeman R. L., Kräusslich H. G. A large deletion in the matrix domain of the human immunodeficiency virus gag gene redirects virus particle assembly from the plasma membrane to the endoplasmic reticulum. J Virol. 1993 Aug;67(8):4972–4980. doi: 10.1128/jvi.67.8.4972-4980.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gallay P., Swingler S., Aiken C., Trono D. HIV-1 infection of nondividing cells: C-terminal tyrosine phosphorylation of the viral matrix protein is a key regulator. Cell. 1995 Feb 10;80(3):379–388. doi: 10.1016/0092-8674(95)90488-3. [DOI] [PubMed] [Google Scholar]
- Gelderblom H. R. Assembly and morphology of HIV: potential effect of structure on viral function. AIDS. 1991 Jun;5(6):617–637. [PubMed] [Google Scholar]
- Gelderblom H. R., Ozel M., Pauli G. Morphogenesis and morphology of HIV. Structure-function relations. Arch Virol. 1989;106(1-2):1–13. doi: 10.1007/BF01311033. [DOI] [PubMed] [Google Scholar]
- González S. A., Affranchino J. L., Gelderblom H. R., Burny A. Assembly of the matrix protein of simian immunodeficiency virus into virus-like particles. Virology. 1993 Jun;194(2):548–556. doi: 10.1006/viro.1993.1293. [DOI] [PubMed] [Google Scholar]
- Grimes J., Basak A. K., Roy P., Stuart D. The crystal structure of bluetongue virus VP7. Nature. 1995 Jan 12;373(6510):167–170. doi: 10.1038/373167a0. [DOI] [PubMed] [Google Scholar]
- Ina Y., Gojobori T. Molecular evolution of human T-cell leukemia virus. J Mol Evol. 1990 Dec;31(6):493–499. doi: 10.1007/BF02102076. [DOI] [PubMed] [Google Scholar]
- Marion D., Driscoll P. C., Kay L. E., Wingfield P. T., Bax A., Gronenborn A. M., Clore G. M. Overcoming the overlap problem in the assignment of 1H NMR spectra of larger proteins by use of three-dimensional heteronuclear 1H-15N Hartmann-Hahn-multiple quantum coherence and nuclear Overhauser-multiple quantum coherence spectroscopy: application to interleukin 1 beta. Biochemistry. 1989 Jul 25;28(15):6150–6156. doi: 10.1021/bi00441a004. [DOI] [PubMed] [Google Scholar]
- Massiah M. A., Starich M. R., Paschall C., Summers M. F., Christensen A. M., Sundquist W. I. Three-dimensional structure of the human immunodeficiency virus type 1 matrix protein. J Mol Biol. 1994 Nov 25;244(2):198–223. doi: 10.1006/jmbi.1994.1719. [DOI] [PubMed] [Google Scholar]
- Matthews S., Barlow P., Boyd J., Barton G., Russell R., Mills H., Cunningham M., Meyers N., Burns N., Clark N. Structural similarity between the p17 matrix protein of HIV-1 and interferon-gamma. Nature. 1994 Aug 25;370(6491):666–668. doi: 10.1038/370666a0. [DOI] [PubMed] [Google Scholar]
- Matthews S., Barlow P., Clark N., Kingsman S., Kingsman A., Campbell I. Refined solution structure of p17, the HIV matrix protein. Biochem Soc Trans. 1995 Nov;23(4):725–729. doi: 10.1042/bst0230725. [DOI] [PubMed] [Google Scholar]
- McInnes C., Kay C. M., Hodges R. S., Sykes B. D. Conformational differences between cis and trans proline isomers of a peptide antigen representing the receptor binding domain of Pseudomonas aeruginosa as studied by 1H-NMR. Biopolymers. 1994 Sep;34(9):1221–1230. doi: 10.1002/bip.360340910. [DOI] [PubMed] [Google Scholar]
- Nermut M. V., Hockley D. J., Jowett J. B., Jones I. M., Garreau M., Thomas D. Fullerene-like organization of HIV gag-protein shell in virus-like particles produced by recombinant baculovirus. Virology. 1994 Jan;198(1):288–296. doi: 10.1006/viro.1994.1032. [DOI] [PubMed] [Google Scholar]
- Nicholls A., Sharp K. A., Honig B. Protein folding and association: insights from the interfacial and thermodynamic properties of hydrocarbons. Proteins. 1991;11(4):281–296. doi: 10.1002/prot.340110407. [DOI] [PubMed] [Google Scholar]
- Nilges M., Gronenborn A. M., Brünger A. T., Clore G. M. Determination of three-dimensional structures of proteins by simulated annealing with interproton distance restraints. Application to crambin, potato carboxypeptidase inhibitor and barley serine proteinase inhibitor 2. Protein Eng. 1988 Apr;2(1):27–38. doi: 10.1093/protein/2.1.27. [DOI] [PubMed] [Google Scholar]
- Poiesz B. J., Ruscetti F. W., Gazdar A. F., Bunn P. A., Minna J. D., Gallo R. C. Detection and isolation of type C retrovirus particles from fresh and cultured lymphocytes of a patient with cutaneous T-cell lymphoma. Proc Natl Acad Sci U S A. 1980 Dec;77(12):7415–7419. doi: 10.1073/pnas.77.12.7415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poznański J., Ejchart A., Wierzchowski K. L., Ciurak M. 1H- and 13C-NMR investigations on cis-trans isomerization of proline peptide bonds and conformation of aromatic side chains in H-Trp-(Pro)n-Tyr-OH peptides. Biopolymers. 1993 May;33(5):781–795. doi: 10.1002/bip.360330507. [DOI] [PubMed] [Google Scholar]
- Rao Z., Belyaev A. S., Fry E., Roy P., Jones I. M., Stuart D. I. Crystal structure of SIV matrix antigen and implications for virus assembly. Nature. 1995 Dec 14;378(6558):743–747. doi: 10.1038/378743a0. [DOI] [PubMed] [Google Scholar]
- Rhee S. S., Hunter E. A single amino acid substitution within the matrix protein of a type D retrovirus converts its morphogenesis to that of a type C retrovirus. Cell. 1990 Oct 5;63(1):77–86. doi: 10.1016/0092-8674(90)90289-q. [DOI] [PubMed] [Google Scholar]
- Russell R. B., Barton G. J. Multiple protein sequence alignment from tertiary structure comparison: assignment of global and residue confidence levels. Proteins. 1992 Oct;14(2):309–323. doi: 10.1002/prot.340140216. [DOI] [PubMed] [Google Scholar]
- Smith D. B., Johnson K. S. Single-step purification of polypeptides expressed in Escherichia coli as fusions with glutathione S-transferase. Gene. 1988 Jul 15;67(1):31–40. doi: 10.1016/0378-1119(88)90005-4. [DOI] [PubMed] [Google Scholar]
- Spearman P., Wang J. J., Vander Heyden N., Ratner L. Identification of human immunodeficiency virus type 1 Gag protein domains essential to membrane binding and particle assembly. J Virol. 1994 May;68(5):3232–3242. doi: 10.1128/jvi.68.5.3232-3242.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wills J. W., Craven R. C. Form, function, and use of retroviral gag proteins. AIDS. 1991 Jun;5(6):639–654. doi: 10.1097/00002030-199106000-00002. [DOI] [PubMed] [Google Scholar]
- Yuan X., Yu X., Lee T. H., Essex M. Mutations in the N-terminal region of human immunodeficiency virus type 1 matrix protein block intracellular transport of the Gag precursor. J Virol. 1993 Nov;67(11):6387–6394. doi: 10.1128/jvi.67.11.6387-6394.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou W., Parent L. J., Wills J. W., Resh M. D. Identification of a membrane-binding domain within the amino-terminal region of human immunodeficiency virus type 1 Gag protein which interacts with acidic phospholipids. J Virol. 1994 Apr;68(4):2556–2569. doi: 10.1128/jvi.68.4.2556-2569.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]