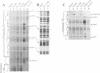Abstract
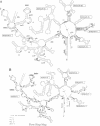
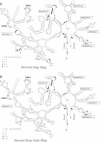
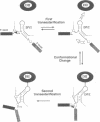
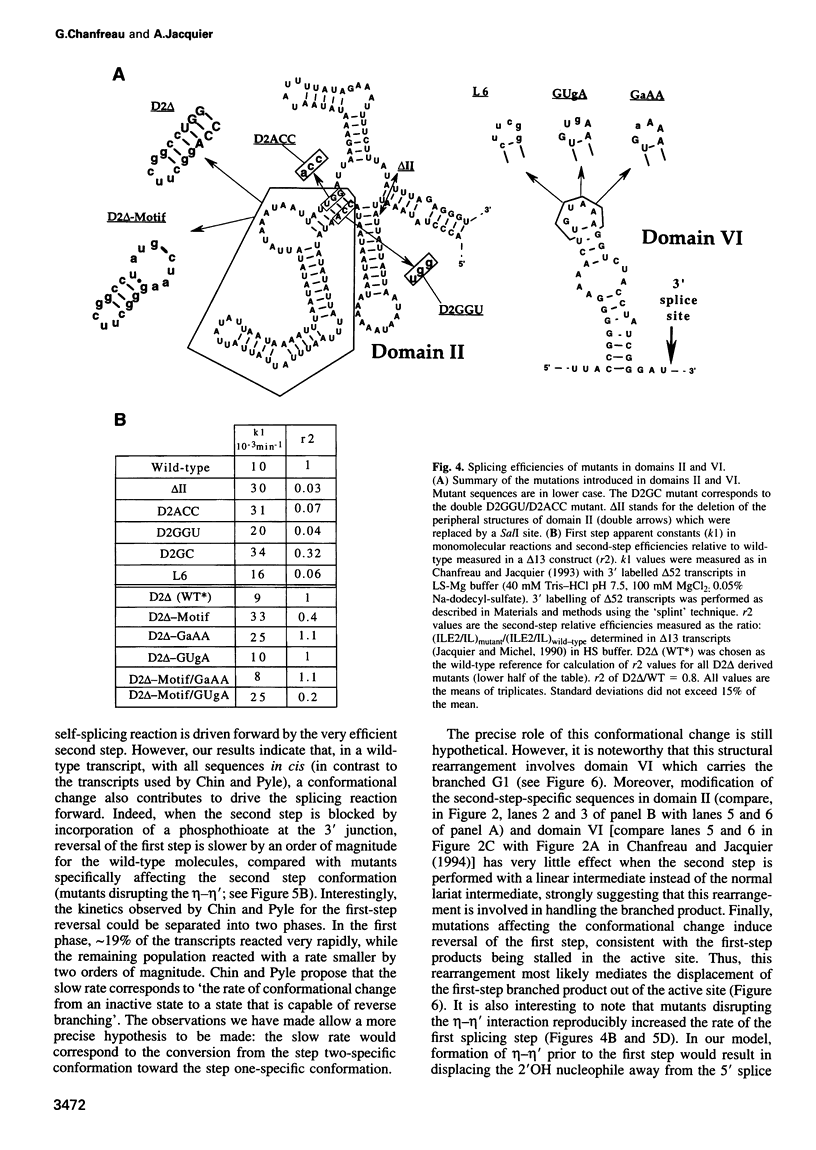
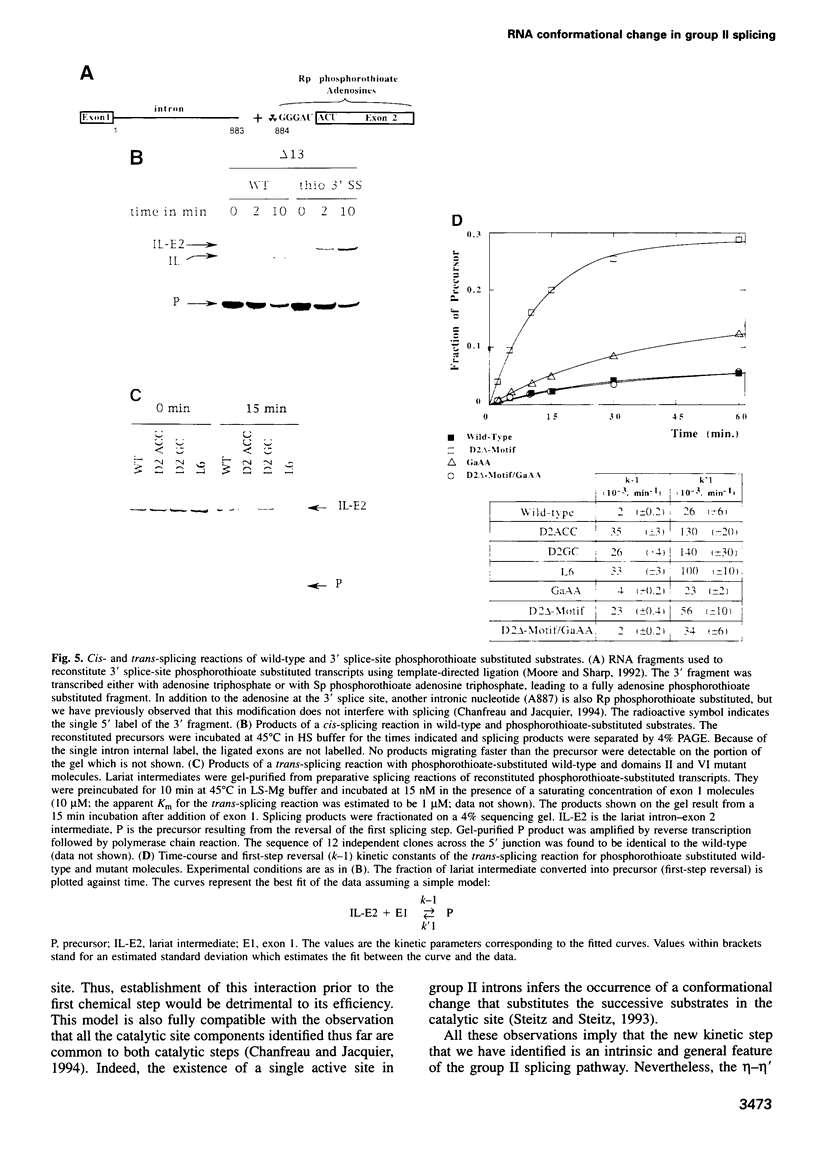
As for nuclear pre-mRNA introns, the splicing pathway of group II self-splicing introns proceeds by two successive transesterifications involving substrates with different chemical configurations. These two reactions have been proposed to be catalysed by two active sites, or alternatively by a single active site rearranging its components to accommodate the successive substrates. Here we show that the structural elements specific for the second splicing step are clustered in peripheral structures of domains II and VI. We show that these structures are not required for catalysis of the second chemical step but, instead, take part in a conformational change that occurs between the two catalytic steps. This rearrangement involves the formation of a tertiary contact between part of domain II and a GNRA tetraloop at the tip of domain VI. The fact that domain VI, which carries the branched structure, is involved in this structural rearrangement and the fact that modifications affecting the structures involved have almost no effect when splicing proceeds without branch formation, suggest that the conformational change results in the displacement of the first-step product out of the active site. These observations give further support to the existence of a single active site in group II introns.
Full text
PDF
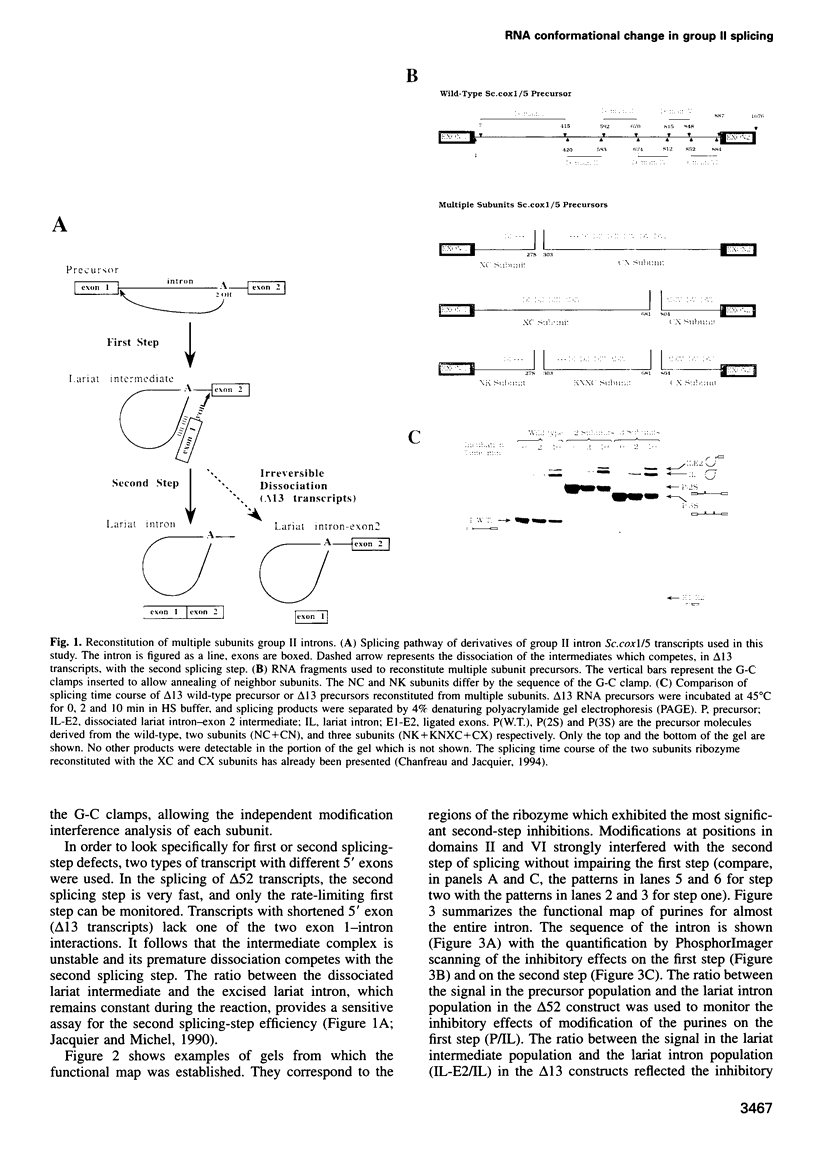
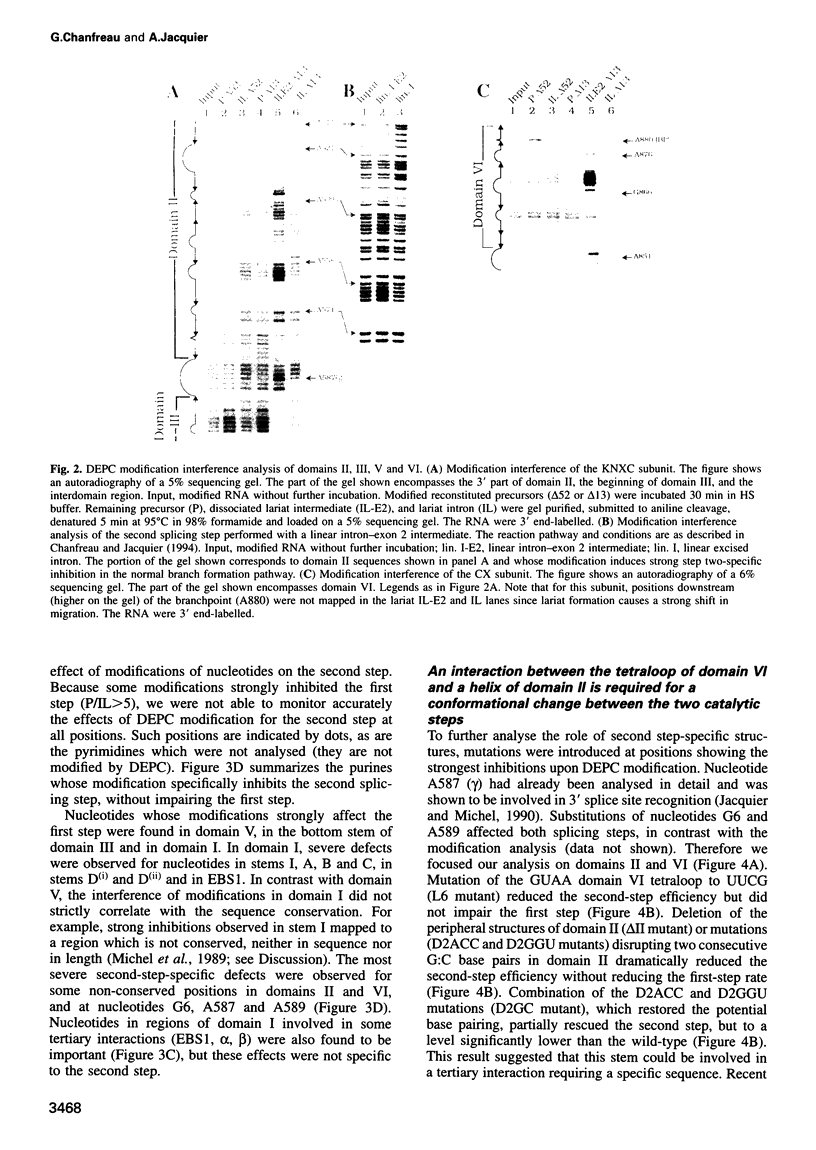

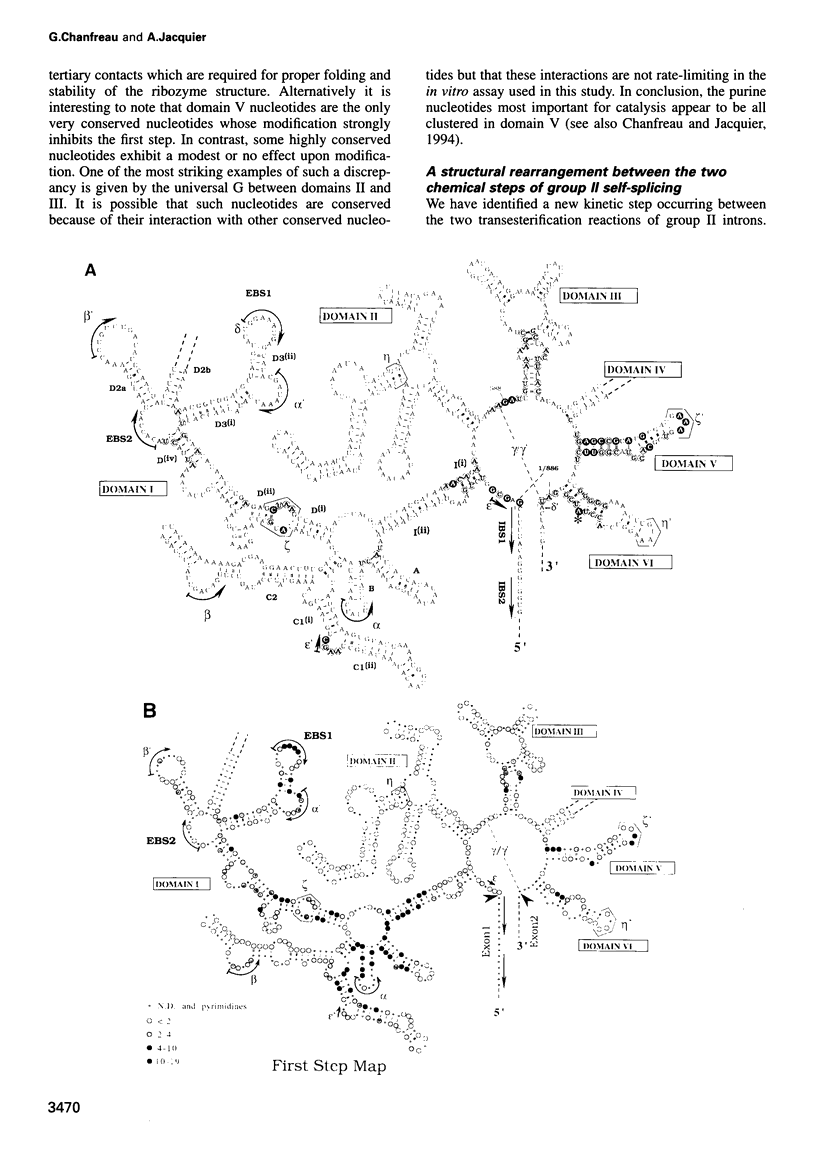
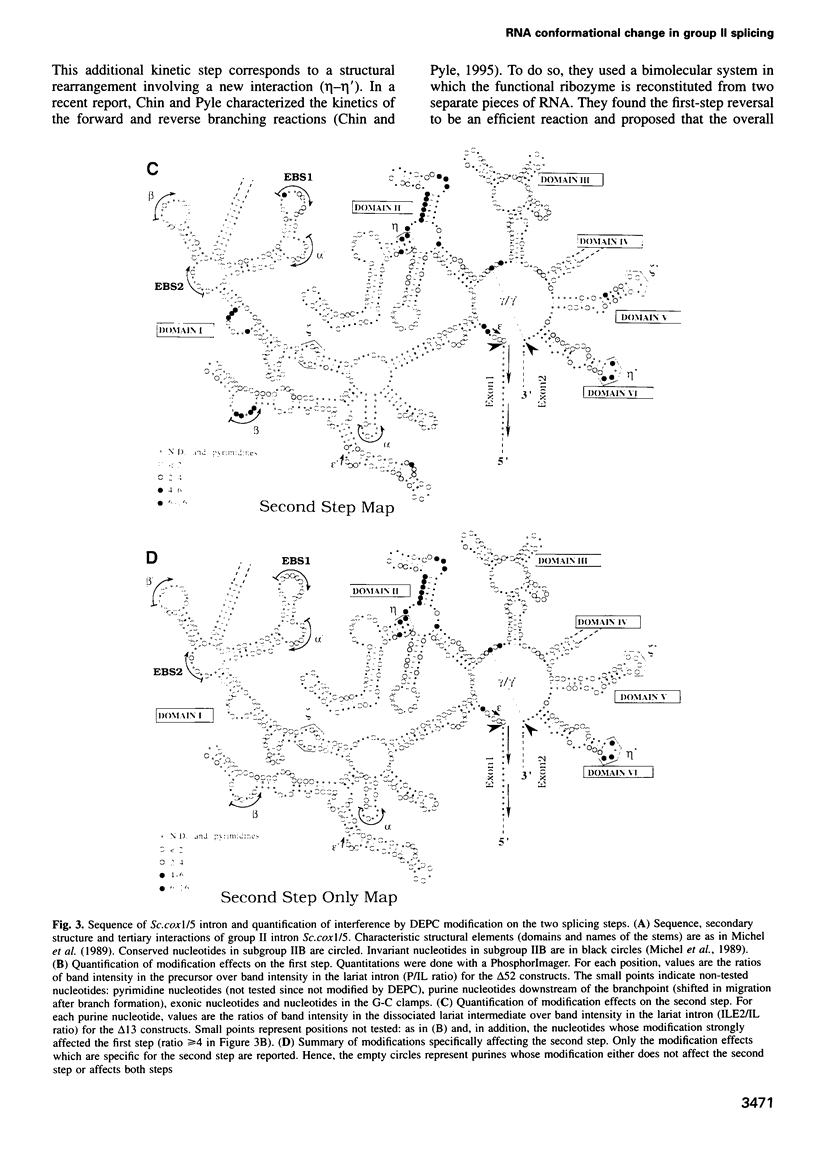



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Chanfreau G., Jacquier A. Catalytic site components common to both splicing steps of a group II intron. Science. 1994 Nov 25;266(5189):1383–1387. doi: 10.1126/science.7973729. [DOI] [PubMed] [Google Scholar]
- Chanfreau G., Jacquier A. Interaction of intronic boundaries is required for the second splicing step efficiency of a group II intron. EMBO J. 1993 Dec 15;12(13):5173–5180. doi: 10.1002/j.1460-2075.1993.tb06212.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chin K., Pyle A. M. Branch-point attack in group II introns is a highly reversible transesterification, providing a potential proofreading mechanism for 5'-splice site selection. RNA. 1995 Jun;1(4):391–406. [PMC free article] [PubMed] [Google Scholar]
- Conway L., Wickens M. Modification interference analysis of reactions using RNA substrates. Methods Enzymol. 1989;180:369–379. doi: 10.1016/0076-6879(89)80112-0. [DOI] [PubMed] [Google Scholar]
- Costa M., Michel F. Frequent use of the same tertiary motif by self-folding RNAs. EMBO J. 1995 Mar 15;14(6):1276–1285. doi: 10.1002/j.1460-2075.1995.tb07111.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferat J. L., Michel F. Group II self-splicing introns in bacteria. Nature. 1993 Jul 22;364(6435):358–361. doi: 10.1038/364358a0. [DOI] [PubMed] [Google Scholar]
- Fontaine J. M., Rousvoal S., Leblanc C., Kloareg B., Loiseaux-de Goër S. The mitochondrial LSU rDNA of the brown alga Pylaiella littoralis reveals alpha-proteobacterial features and is split by four group IIB introns with an atypical phylogeny. J Mol Biol. 1995 Aug 18;251(3):378–389. doi: 10.1006/jmbi.1995.0441. [DOI] [PubMed] [Google Scholar]
- Franzen J. S., Zhang M., Peebles C. L. Kinetic analysis of the 5' splice junction hydrolysis of a group II intron promoted by domain 5. Nucleic Acids Res. 1993 Feb 11;21(3):627–634. doi: 10.1093/nar/21.3.627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harris-Kerr C. L., Zhang M., Peebles C. L. The phylogenetically predicted base-pairing interaction between alpha and alpha' is required for group II splicing in vitro. Proc Natl Acad Sci U S A. 1993 Nov 15;90(22):10658–10662. doi: 10.1073/pnas.90.22.10658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hausner T. P., Giglio L. M., Weiner A. M. Evidence for base-pairing between mammalian U2 and U6 small nuclear ribonucleoprotein particles. Genes Dev. 1990 Dec;4(12A):2146–2156. doi: 10.1101/gad.4.12a.2146. [DOI] [PubMed] [Google Scholar]
- Jacquier A., Jacquesson-Breuleux N. Splice site selection and role of the lariat in a group II intron. J Mol Biol. 1991 Jun 5;219(3):415–428. doi: 10.1016/0022-2836(91)90183-7. [DOI] [PubMed] [Google Scholar]
- Jacquier A., Michel F. Base-pairing interactions involving the 5' and 3'-terminal nucleotides of group II self-splicing introns. J Mol Biol. 1990 Jun 5;213(3):437–447. doi: 10.1016/S0022-2836(05)80206-2. [DOI] [PubMed] [Google Scholar]
- Jacquier A., Michel F. Multiple exon-binding sites in class II self-splicing introns. Cell. 1987 Jul 3;50(1):17–29. doi: 10.1016/0092-8674(87)90658-1. [DOI] [PubMed] [Google Scholar]
- Jaeger L., Michel F., Westhof E. Involvement of a GNRA tetraloop in long-range RNA tertiary interactions. J Mol Biol. 1994 Mar 11;236(5):1271–1276. doi: 10.1016/0022-2836(94)90055-8. [DOI] [PubMed] [Google Scholar]
- Jarrell K. A., Dietrich R. C., Perlman P. S. Group II intron domain 5 facilitates a trans-splicing reaction. Mol Cell Biol. 1988 Jun;8(6):2361–2366. doi: 10.1128/mcb.8.6.2361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kunkel T. A. Rapid and efficient site-specific mutagenesis without phenotypic selection. Proc Natl Acad Sci U S A. 1985 Jan;82(2):488–492. doi: 10.1073/pnas.82.2.488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kück U., Godehardt I., Schmidt U. A self-splicing group II intron in the mitochondrial large subunit rRNA (LSUrRNA) gene of the eukaryotic alga Scenedesmus obliquus. Nucleic Acids Res. 1990 May 11;18(9):2691–2697. doi: 10.1093/nar/18.9.2691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MacMillan A. M., Query C. C., Allerson C. R., Chen S., Verdine G. L., Sharp P. A. Dynamic association of proteins with the pre-mRNA branch region. Genes Dev. 1994 Dec 15;8(24):3008–3020. doi: 10.1101/gad.8.24.3008. [DOI] [PubMed] [Google Scholar]
- Michel F., Ferat J. L. Structure and activities of group II introns. Annu Rev Biochem. 1995;64:435–461. doi: 10.1146/annurev.bi.64.070195.002251. [DOI] [PubMed] [Google Scholar]
- Michel F., Umesono K., Ozeki H. Comparative and functional anatomy of group II catalytic introns--a review. Gene. 1989 Oct 15;82(1):5–30. doi: 10.1016/0378-1119(89)90026-7. [DOI] [PubMed] [Google Scholar]
- Moore M. J., Sharp P. A. Evidence for two active sites in the spliceosome provided by stereochemistry of pre-mRNA splicing. Nature. 1993 Sep 23;365(6444):364–368. doi: 10.1038/365364a0. [DOI] [PubMed] [Google Scholar]
- Moore M. J., Sharp P. A. Site-specific modification of pre-mRNA: the 2'-hydroxyl groups at the splice sites. Science. 1992 May 15;256(5059):992–997. doi: 10.1126/science.1589782. [DOI] [PubMed] [Google Scholar]
- Padgett R. A., Podar M., Boulanger S. C., Perlman P. S. The stereochemical course of group II intron self-splicing. Science. 1994 Dec 9;266(5191):1685–1688. doi: 10.1126/science.7527587. [DOI] [PubMed] [Google Scholar]
- Pley H. W., Flaherty K. M., McKay D. B. Model for an RNA tertiary interaction from the structure of an intermolecular complex between a GAAA tetraloop and an RNA helix. Nature. 1994 Nov 3;372(6501):111–113. doi: 10.1038/372111a0. [DOI] [PubMed] [Google Scholar]
- Pyle A. M., Green J. B. Building a kinetic framework for group II intron ribozyme activity: quantitation of interdomain binding and reaction rate. Biochemistry. 1994 Mar 8;33(9):2716–2725. doi: 10.1021/bi00175a047. [DOI] [PubMed] [Google Scholar]
- Schwer B., Guthrie C. A conformational rearrangement in the spliceosome is dependent on PRP16 and ATP hydrolysis. EMBO J. 1992 Dec;11(13):5033–5039. doi: 10.1002/j.1460-2075.1992.tb05610.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steitz T. A., Steitz J. A. A general two-metal-ion mechanism for catalytic RNA. Proc Natl Acad Sci U S A. 1993 Jul 15;90(14):6498–6502. doi: 10.1073/pnas.90.14.6498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Waring R. B. Identification of phosphate groups important to self-splicing of the Tetrahymena rRNA intron as determined by phosphorothioate substitution. Nucleic Acids Res. 1989 Dec 25;17(24):10281–10293. doi: 10.1093/nar/17.24.10281. [DOI] [PMC free article] [PubMed] [Google Scholar]