Abstract
The accumulation of uremic toxins is involved in the progression of CKD. Various uremic toxins are derived from gut microbiota, and an imbalance of gut microbiota or dysbiosis is related to renal failure. However, the pathophysiologic mechanisms underlying the relationship between the gut microbiota and renal failure are still obscure. Using an adenine-induced renal failure mouse model, we evaluated the effects of the ClC-2 chloride channel activator lubiprostone (commonly used for the treatment of constipation) on CKD. Oral administration of lubiprostone (500 µg/kg per day) changed the fecal and intestinal properties in mice with renal failure. Additionally, lubiprostone treatment reduced the elevated BUN and protected against tubulointerstitial damage, renal fibrosis, and inflammation. Gut microbiome analysis of 16S rRNA genes in the renal failure mice showed that lubiprostone treatment altered their microbial composition, especially the recovery of the levels of the Lactobacillaceae family and Prevotella genus, which were significantly reduced in the renal failure mice. Furthermore, capillary electrophoresis–mass spectrometry-based metabolome analysis showed that lubiprostone treatment decreased the plasma level of uremic toxins, such as indoxyl sulfate and hippurate, which are derived from gut microbiota, and a more recently discovered uremic toxin, trans-aconitate. These results suggest that lubiprostone ameliorates the progression of CKD and the accumulation of uremic toxins by improving the gut microbiota and intestinal environment.
Keywords: CKD, gastrointestinal medications, uremia, intestine
CKD is a global health problem that carries a substantial risk for ESRD, cardiovascular disease, and death.1 In CKD, the accumulation of uremic toxins accelerates the progression of CKD and hence, mortality.2,3 Therefore, reducing the accumulation of uremic toxins is important for the protection against and amelioration of renal damage.
Recent evidence has suggested that alterations in the gut microbiota are linked to various diseases conditions, including obesity and diabetes as well as cardiovascular4,5 and kidney diseases.6,7 Because various uremic solutes, such as indoxyl sulfate, are derived from gut microbial metabolism, the gut microbial status affects the accumulation of uremic toxins.8 Furthermore, it has been reported that the gut microbiota composition is changed for the worse in CKD,6 and the gut environment is associated with both the etiology and progression of CKD. However, the mechanistic link between the gut and CKD remains poorly understood.
Lubiprostone is a synthetic bicyclic fatty acid derivative of prostaglandin E1 that is clinically used for the treatment of chronic constipation.9 Lubiprostone activates the ClC-2 chloride channel, resulting in an enhancement of the intestinal luminal Cl− secretion and water motility, which effects are responsible for its laxatives properties.10 In general, chronic constipation, which is frequent in patients with CKD, worsens the intestinal environment and alters the composition of the gut microbiota.11,12 We examined the beneficial effects of lubiprostone on CKD using an adenine-induced renal failure (RF) mouse model. Lubiprostone was found to exert a renoprotective effect on the progression of CKD with a reduction in the plasma concentration of uremic toxins and improvement in the gut microbiota population. These findings suggest a potential therapeutic approach to CKD on the basis of the improvement of the intestinal environment.
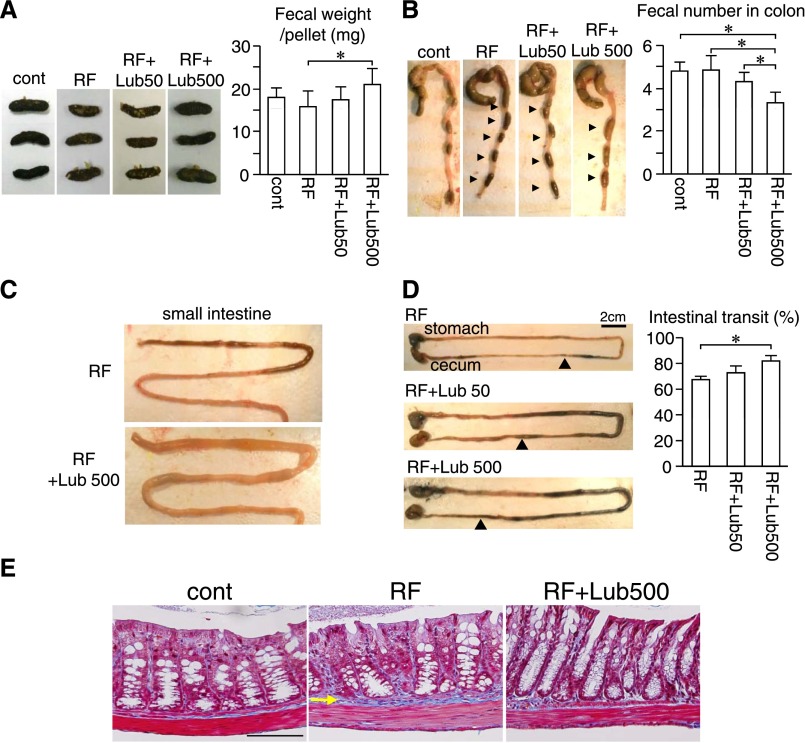
We examined the effect of lubiprostone on uremic RF using an adenine-induced RF mouse model13 (Supplemental Figure 1). RF was developed by 6-week oral administration of adenine into normal mice, which were then treated with 50 or 500 µg/kg per day lubiprostone (RF+Lub50 and RF+Lub500, respectively). RF mice exhibited reduced food intake, body weight loss, abnormal polyuria, and anemia compared with control mice (Supplemental Table 1). Body weight and food intake were comparable among the RF, RF+Lub50, and RF+Lub500 groups, but the serum sodium level was significantly reduced in both of the lubiprostone-treated RF groups (148.3 and 146.7 mEq/L in RF+Lub50 and RF+Lub500 groups, respectively) compared with the RF group (153.0 mEq/L). Concerning the fecal and intestinal effects, the RF+Lub500 group exhibited a significantly higher level of each fecal sample weight and a lower amount of residual feces in the colon than the RF group (Figure 1, A and 1B). In addition, slightly soft feces were observed in the RF+Lub500 group because of the increase in the small intestinal bowel fluid (Figure 1C) and accelerated intestinal transit (Figure 1D). However, diarrhea was not observed in either the RF+Lub50 or RF+Lu500 group. The pH of the cecal fluid was comparable among the groups (pH was 6.5±0.33, 6.6±0.34, and 6.5±0.29 in control, RF, and RF+Lub500 groups, respectively). By Masson’s trichrome staining (MTS) of the colon, dense collagen deposition in the lamina propria was detected in the RF group as reported.14 This collagen deposition in the colon was suppressed in the RF+Lub500 group compared with the RF group (Figure 1E).
Figure 1.
Effect of lubiprostone on fecal and intestinal characteristics in uremic mice. (A) Fecal shape and weight per pellet. Fecal weight per pellet was calculated as (fecal number)/(total fecal wet weight). n=6–7 for each group. (B) Residual fecal number in the colon. n=6–7. (C) Representative small intestinal images of RF and RF+Lub500 groups. (D) Intestinal transit analysis after lubiprostone administration in RF mice. The arrowheads denote the leading edge of trypan blue. *P<0.05 between the indicated groups. n=3. Scale bar, 2 cm. (E) Representative histologic images of the colon. MTS-stained cross-section of the colon containing a fecal pellet. The arrow indicates lamina propria. P<0.05 between the indicated groups. Cont, control. Scale bar, 100 µm.
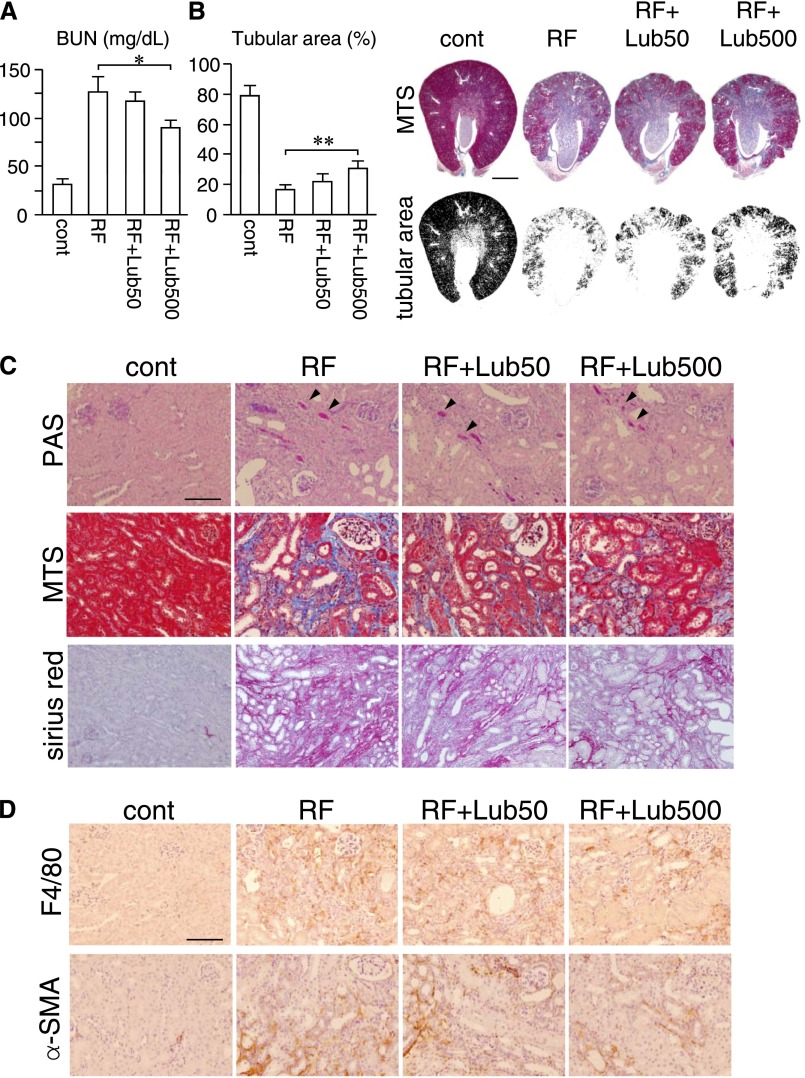
We next examined the effect of lubiprostone on renal function and histologic changes in the RF mice. The BUN level was 4.1-fold higher in the RF group than in the control group, whereas it was significantly suppressed in the RF+Lub500 group (Figure 2A). In addition, the decrease in the cortical tubular area in the RF group was slightly but significantly recovered in the RF+Lub500 group (Figure 2B), although the adenine-derived cast formation was also observed in the RF+Lub500 group in a similar manner to the RF group (Figure 2C, periodic acid–Schiff staining). In contrast, the fibrotic area and interstitial collagen deposition in the damaged kidneys were decreased in the RF+Lub500 group compared with the RF group (Figure 2C).
Figure 2.
Effect of lubiprostone on renal fibrosis and inflammation in the uremic mouse kidney. (A) BUN concentration in mice. n=6–7 for each group. (B) Morphometric analysis of the percentage of the cortical tubular area and whole-kidney images of MTS. The cortical tubular area was calculated on the basis of the MTS image. n=6–7. Scale bar, 1 cm. *P<0.05 compared with the RF group; **P<0.01 compared with the RF group. (C) Representative histologic images of periodic acid–Schiff- (PAS-), MTS-, and picrosirius red-stained kidney. The arrowheads denote adenine-derived casts. Scale bar, 100 µm. (D) Immunostaining of α-smooth muscle actin (α-SMA) and F4/80 in kidney sections. Cont, control. Scale bar, 100 µm.
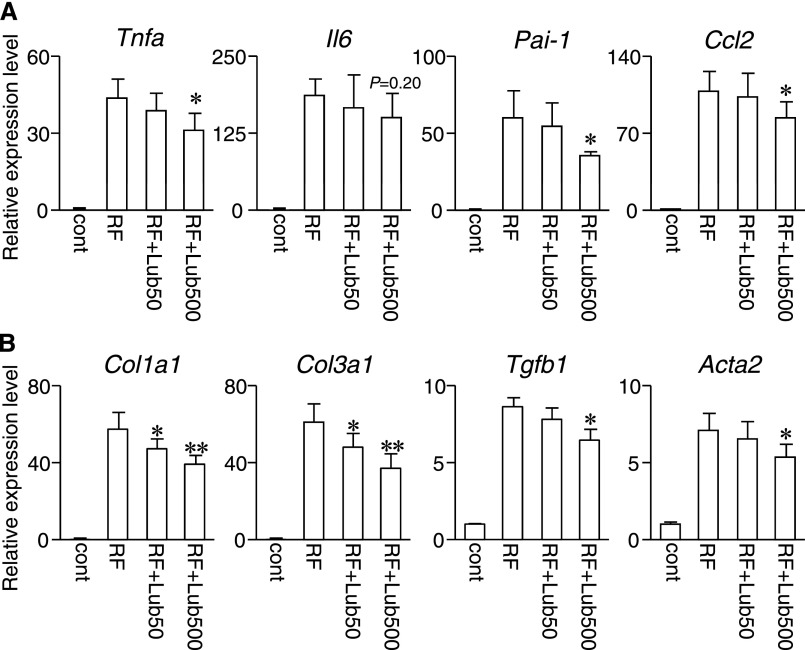
We further evaluated renal fibrosis and inflammation by immunohistochemistry and quantitative PCR. By immunohistochemical analysis, the RF+Lub500 group exhibited a decreased number of macrophages and myofibroblasts compared with the RF group, which was shown by anti-F4/80 and anti–α-smooth muscle actin staining (Figure 2D). Quantitative PCR indicated a significant reduction in the expression of renal fibrosis-related genes (Acta2, Tgfb1, Col1a1, and Col3a1) and inflammatory cytokines (Tnfa, Pai-1, and Ccl2) in the kidney of the RF+Lub500 group compared with the RF group (Figure 3). The expression of Il-6 mRNA was not significant but tended to be reduced in the RF+Lub500 group. These results suggested that the treatment with lubiprostone ameliorated the progression of renal fibrosis and local inflammation in the RF mouse model.
Figure 3.
Effects of lubiprostone on renal gene expression in uremic mice. Relative transcript levels of genes in (A) renal inflammation and (B) fibrosis were measured using real-time PCR. The names in parentheses indicate the coding protein: Tnfa (TNF-α), Il6 (IL-6), Pai-1 (PAI-1), Ccl2 (MCP-1), Col1a1 (type I collagen-α1), Col3a1 (type III collagen-α1), Tgfb1 (TGF-β), and Acta2 (α-smooth muscle actin). The expression levels were first normalized to those of Gapdh and then further normalized to the levels in the kidney from the control mice. n=6–7 for each group. Cont, control. *P<0.05 versus the RF group; **P<0.01 versus the RF group.
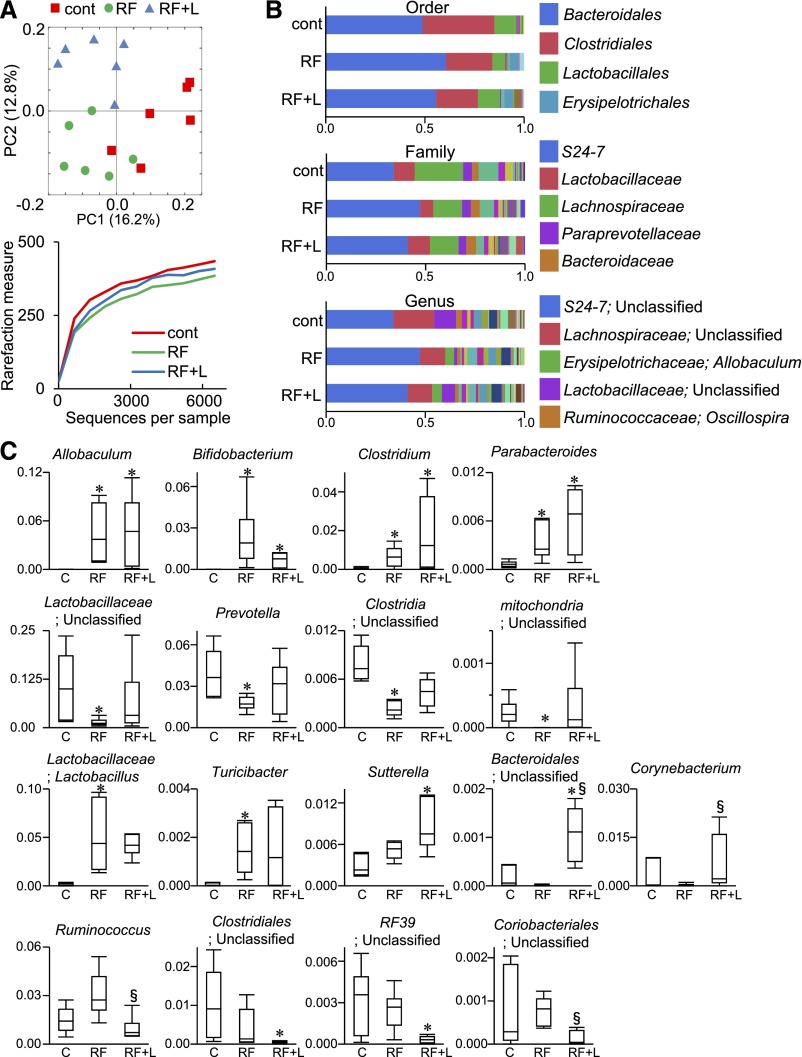
To elucidate the mechanism of the renoprotective effect induced by lubiprostone treatment, we investigated the effect of lubiprostone on the gut microbiota in RF mice. Gut microbiome analysis of bacterial 16S rRNA genes followed by operational taxonomic unit and unweighted Unifrac distance analysis indicated that the gut microbial composition could be roughly categorized into three groups (i.e., the control, RF, and RF+Lub500 groups) (Figure 4A). In addition, the diversity analysis showed that the decreased microbial diversity in the RF group became more similar to the control group by lubiprostone treatment (Figure 4A). Consistent with this, microbial population analysis showed that a minor population of gut microbiota in each group was different (Figure 4B). At the genus level, quantitative analysis confirmed a significant increase in Allobaculum, Bfidobacterium, Clostridium, and Parabacteroides in both the RF and RF+Lub500 groups (Figure 4C). Interestingly, an unclassified Lactobacillaceae family, a Prevotella genus, an unclassified Clostridia class, and an unclassified mitochondria family were significantly decreased in the RF group, but this decrease was not found in the RF+Lub500 group (Figure 4C). Furthermore, the significant increase in the Lactobacillus and Turicibacter genuses in the RF group was also abrogated by lubiprostone treatment, suggesting that lubiprostone is able to ameliorate gut microbiota imbalances in RF mice.
Figure 4.
Effects of lubiprostone on gut microbiota in uremic mice. (A) Unweighted unifrac distance analysis (upper panel) and diversity of bacterial species as indicated by Chao1 rarefaction measure (lower panel) of gut microbiota in the control, RF, and RF+Lub500 groups. n=6. (B) Relative abundance of microbiota on the basis of the average number of each subfamily at the order, family, and genus levels. The major subfamilies are indicated on the right. n=6. Cont, control. (C) The proportional change of each subfamily at the genus level. Genera displaying a significant change among the three groups are shown (Steel–Dwass). The y axis indicates the abundance of each microbe (percentage). C, control; RF+L, RF treated with 500 µg/kg per day lubiprostone. *P<0.05 versus the control group; §P<0.05 versus the RF group.
To exclude the possibility that the adenine diet per se induced the alteration of gut microbiota, we performed a fecal microbiome analysis during adenine feeding (Supplemental Figure 2A). The composition of the microbiota did not obviously change 1 and 2 weeks after adenine feeding. BUN began to significantly increase after the 2-week adenine diet (Supplemental Figure 2B). A slight alteration of the microbiota was observed after 4 weeks and evident after 6 weeks of the adenine diet. Simultaneously, BUN was markedly increased after 4 and 6 weeks of the adenine diet.
These results suggest that the adenine diet per se had a little influence on the gut microbiota and that the alteration of the gut microbiota in adenine-induced RF was mainly dependent on the RF state.
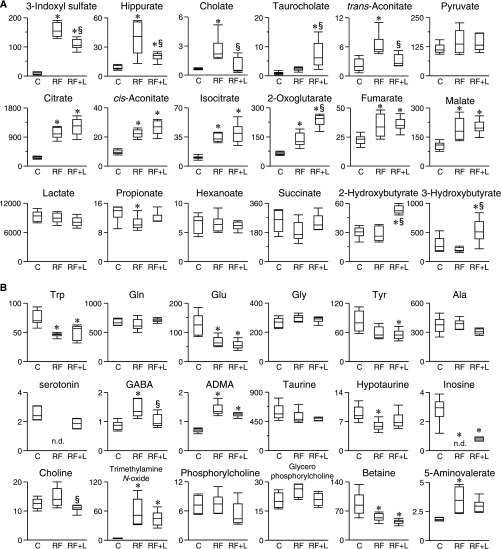
We next performed capillary electrophoresis with electrospray ionization time-of-flight mass spectrometry (CE-TOFMS)-based metabolome analysis of the plasma in the control, RF, and RF+Lub500 groups. The increased levels of the well known uremic solutes indoxyl sulfate and hippurate in the RF group were significantly suppressed in the RF+Lub500 group (Figure 5A). In addition to these uremic solutes, higher plasma concentrations of certain anionic metabolites in the RF group, such as trans-aconitate, propionate, and cholate, were significantly suppressed by lubiprostone treatment (Figure 5A). Furthermore, most of the tricarboxylic acid cycle-related components (citrate, cis-aconitate, isocitrate, fumarate, and malate) were significantly increased in both the RF and RF+Lub500 groups compared with the control group. Among the cationic solutes, the amino acids tryptophan, glutamate, inosine, and betaine were significantly decreased in both RF and RF+Lub500 groups, whereas the elevated concentration of γ-aminobutyric acid in RF was significantly decreased in the RF+Lub500 group compared with the RF group. (Figure 5B). The plasma acetate and p-cresyl sulfate concentrations were also measured (Supplemental Figure 3). The plasma acetate concentration was comparable among these groups. The plasma p-cresyl sulfate level was significantly increased in both the RF and RF+Lub500 groups compared with the control group but not changed between the RF and RF+Lub500 groups.
Figure 5.
Effects of lubiprostone on the plasma uremic solutes analyzed by CE-TOFMS–based metabolome analysis in uremic mice. Each of the plasma concentrations was measured by CE-TOFMS. (A) Anionic and (B) cationic solutes. The y axis indicates the micromolar concentration. n=6. C, control; GABA, γ-aminobutyric acid; RF+L, RF treated with 500 µg/kg per day lubiprostone. *P<0.05 versus the control group; §P<0.05 versus the RF group (ANOVA).
In CKD, the intestinal environment is reported to be adversely altered.15,16 The potential exacerbating factors include the retention of uremic toxins, intestinal ischemia, intestinal transit time prolonged by constipation, decreased intestinal fluid secretion, and malnutrition of the gut lining with evident atrophy.8,16 Such a poor condition of the gut is suggested to be involved in the etiology and progression of CKD because of an imbalance in the gut microbiota, leading to an accumulation of uremic toxins and bacterial translocation.16
In this study, we showed that lubiprostone treatment ameliorated the accumulation of uremic toxins and mitigated the renal fibrosis and inflammation that play a major role in the progression of renal damage.17 Lubiprostone actively induced bowel fluid secretion by activating Cl− channels, leading to a reduction in the intestinal transit time and changes in the fecal characteristics through altering the gut environment. This gut microbiome analysis showed that the relative abundance of the Lactobacillaceae family and Prevotella genus was significantly decreased in the RF group, which is consistent with a previous report.6 Furthermore, our findings show that lubiprostone treatment of RF mice ameliorated the decrease in the Lactobacillaceae family and Prevotella genus. This increased abundance of the Lactobacillaceae family by lubiprostone treatment has also been reported in a previous study using a cystic fibrosis model mice.18
It has also been reported that the gut microbiota largely contributes to the production of various uremic solutes, such as indole derivatives (e.g., indoxyl sulfate) and glycine conjugates (e.g., hippurate)19; therefore, dysbiosis might be related to the accumulation of uremic toxins. In this study, oral administration of lubiprostone reduced the plasma concentration of indoxyl sulfate and hippurate as well as trans-aconitate. Indoxyl sulfate is associated with the progression of RF through the exacerbation of oxidative stress, inflammation, and fibrosis.20 Trans-aconitate is also known as a uremic toxin and a recently identified biomarker for predicting the onset of renal damage.21 Therefore, the alteration of the gut microbiota induced by treatment with lubiprostone in RF mice is suggested to have caused the improvement of the plasma uremic solute levels, which in turn, led to the amelioration of CKD.
Lactobacillaceae and Prevotella, which were increased by the lubiprostone treatment, are saccharolytic bacteria. Indoles, a precursor of indole derivatives, are produced as a result of protein fermentation by colonic microbiota.8 Therefore, we suppose that lubiprostone may promote the growth of saccharolytic bacteria in RF at the expense of the other proteolytic bacteria, resulting in the reduction of some uremic toxins (e.g., indoxyl sulfate).
In addition, it has been reported that a prolonged intestinal transit time promotes the conversion of amino acids into uremic wastes through microbial fermentation.11 Thus, a reduction of the intestinal transit time by lubiprostone treatment may also be involved in the reduction in uremic solutes and the amelioration of CKD symptoms.
Intestinal mucosal barrier functions are impaired in CKD.22 This impairment leads to a high intestinal epithelial permeability, resulting in bacterial translocations from the gut lumen into the blood stream, and this increased bacterial translocation is reported to be a source of microinflammation in CKD.23 Recently, Moeser et al.24 reported that lubiprostone exerts a reparative effect on the function of the intestinal mucosal barrier against ischemic intestinal damage. In addition, lubiprostone treatment stimulated the intestinal secretion of mucin,25 which works as the first line of defense of the gastrointestinal mucosal barrier. Therefore, the preventive effect of lubiprostone treatment in RF mice might be through upregulation of the mucosal barrier function.
Vaziri et al.26 have reported that the uremia-induced disruption of the intestinal barrier is mediated by urea influx into the gastrointestinal tract. Influx urea is converted by microbial urease to ammonia and ammonium hydroxide. These caustic compounds can disrupt the intestinal barrier function. Furthermore, the uremia-induced alteration of the intestinal microbiota is driven by the influx of urea into the gastrointestinal tract.14 Therefore, some effects of lubiprostone on the urea influx into the gastrointestinal tract may be postulated. Additional experiments are necessary to clarify this issue.
A recent study has shown expansion of bacteria families possessing urease, uricase, and indole- and p-cresol–forming enzymes and reduction of butyrate-producing bacteria in patients with ESRD. Wong et al.27 have shown such dysbiosis in patients with ESRD using in silico tests. In accordance with the in silico results,27 these data show the reduction of bacteria families possessing butyrate-producing enzymes (Lactobaillaceae and Prevotellaceae) in the RF mice, and the reduction was recovered by lubiprostone treatment (Supplemental Figure 4). However, the reported bacterial families that possess urease, uricase, and indole- and p-cresol–forming enzymes27 enriched in patients with ESRD were not significantly changed in the RF mice or not detected in any groups (Supplemental Figure 4). Additionally, Bifidobacterium was scarce in the normal mice and increased in the RF mice, although previous reports showed that Bifidobacterium is present under normal conditions and decreases under RF states in rat and human.28,29 We suppose that such discrepancies result from the differences among mammalian species. The bacterial families that contributed to the urease or indole-producing activities in RF mice may differ from those of human ESRD. Also, the contribution of Bifidobacterium in the normal microbiota of mice is presumably low, and the increase of the Bifidobacterium population in RF is possibly related to the uremic dysbiosis in mice. In support of this idea, Bifidobacterium was also undetectable among gut microbiota of another mouse strain, BALB/c mice (S.F. et al., unpublished data).
In conclusion, lubiprostone treatment is a potentially useful therapeutic intervention against the progression of CKD by improving the gut environment and reducing uremic toxins. These findings provide supporting evidence for the gut–kidney axis in CKD.
Concise Methods
Animal Studies
All animal experiments were approved by the Animal Committee of the Tohoku University School of Medicine. Male C57BL/6 mice were fed a normal diet (type CE-2; Clea Japan). At age 7 weeks, mice were divided randomly into the control and adenine groups. For the control group, the normal diet was continued. For the adenine group, a CE-2 diet containing 0.2% adenine (Wako) was given for 6 weeks. After 6 weeks of adenine feeding, the food was switched to the normal diet, and then, lubiprostone (50 or 500 µg/kg; Abbott, Japan) or saline in a volume of 100 μl was administered to the mice by daily gavage at night for 12 days. At the end of study, the mice were euthanized, after which blood, cecal fluid, and tissues were obtained. One day before euthanasia, mice were placed in an individual metabolic cage to measure the water and food intake and collect urine and feces. BUN and biochemical parameters were assessed using a blood analyzer (i-STAT; Fuso Pharmaceutical Industries). BP was measured by the tail-cuff method using model MK-2000 (Muromachi Kikai). The cecal fluid pH was measured by a pH meter (LAQUAtwin; HORIBA).
Intestinal Transit Analyses
The measurement of gastrointestinal transit was performed as described previously.30 Briefly, adenine-induced RF mice were fasted for 20 hours with free access to water. Mice were treated with either vehicle or lubiprostone (50 or 500 µg/kg) orally, and 1 hour later, they were given an oral gavage of 150 µl 0.5% trypan blue solution. The mice were euthanized 30 minutes after administration of the solution, and the small intestine from the jejunum to the cecum was dissected. The distance traveled by the leading edge of the small intestine and the percentage of intestinal transit for each animal were calculated as the percentage of transit (trypan blue distance)/(small intestinal length)×100.
Histology and Morphometric Analyses
Tissues were fixed in 10% neutral buffered formalin and embedded in paraffin. Kidney sections were stained with hematoxylin and eosin, periodic acid–Schiff, MTS, and picrosirius red. For analysis of the tubular area, the percentage of the tubular area in the cortex was evaluated with MTS-stained kidney sections using the National Institutes of Health ImageJ analysis software. For immunohistochemistry, sections were immunolabeled using anti–α-smooth muscle actin (DAKO) and anti-F4/80 (Serotec) antibodies.
Quantitative PCR Analyses
Whole kidneys were homogenized in TRIzol reagent (Invitrogen) and extracted according to the manufacturer’s directions. cDNA synthesis was performed using a transcriptor first-strand cDNA synthesis kit (Roche). The primers purchased from Applied Biosystems are shown in Supplemental Table 2.
Microbiome Analyses
The genomic DNA of gut microbiota was extracted from murine feces, and 454-barcode pyrosequencing of microbial 16S rRNA genes was performed as described previously.31,32 Briefly, microbial genomic DNA was extracted using a phenol–chloroform standard protocol with vigorous shaking with 0.1-mm zirconia/silica beads. The V1–V2 region of the 16S rRNA gene was amplified and subjected to 454 GS JUNIOR pyrosequencing (Roche). The 16s rRNA reads were analyzed using QIIME and the RDP classifier. The microbiome data have been deposited in the DDBJ database (http://getentry.ddbj.nig.ac.jp/) under accession number DRA002254. Detailed methods are described in Supplemental Material.
CE-TOFMS Measurement
A quantitative analysis of charged metabolites by CE-TOFMS was performed as described previously.33,34 CE-TOFMS experiments were performed using the Agilent CE System (Agilent Technologies), the Agilent G3250AA LC/MSD TOF System (Agilent Technologies), the Agilent 1100 Series Binary HPLC Pump, the G1603A Agilent CE-MS adapter (Agilent Technologies), and the G1607A Agilent CE-ESI-MS Sprayer Kit. Detailed methods are described in Supplemental Material.
Statistical Analyses
Data are presented as the means±SD. Statistical analysis was evaluated by the unpaired t test as well as ANOVA or Steel–Dwass multiple comparison procedures. Values of P<0.05 were considered to be statistically significant.
Disclosures
None.
Supplementary Material
Acknowledgments
This work was supported, in part, by JSPS Grants-in-Aid for Scientific Research (KAKENHI) Grants 26860624 (to E.M.), 24380072 (to S.F.), and 23390033 (to T.A.), the Japan Kidney Foundation, and the Miyagi Kidney Foundation.
Footnotes
Published online ahead of print. Publication date available at www.jasn.org.
This article contains supplemental material online at http://jasn.asnjournals.org/lookup/suppl/doi:10.1681/ASN.2014060530/-/DCSupplemental.
References
- 1.Jha V, Garcia-Garcia G, Iseki K, Li Z, Naicker S, Plattner B, Saran R, Wang AY, Yang CW: Chronic kidney disease: Global dimension and perspectives. Lancet 382: 260–272, 2013 [DOI] [PubMed] [Google Scholar]
- 2.Vanholder R, Baurmeister U, Brunet P, Cohen G, Glorieux G, Jankowski J, European Uremic Toxin Work Group : A bench to bedside view of uremic toxins. J Am Soc Nephrol 19: 863–870, 2008 [DOI] [PubMed] [Google Scholar]
- 3.Niwa T, Nomura T, Sugiyama S, Miyazaki T, Tsukushi S, Tsutsui S: The protein metabolite hypothesis, a model for the progression of renal failure: An oral adsorbent lowers indoxyl sulfate levels in undialyzed uremic patients. Kidney Int Suppl 62: S23–S28, 1997 [PubMed] [Google Scholar]
- 4.Tremaroli V, Bäckhed F: Functional interactions between the gut microbiota and host metabolism. Nature 489: 242–249, 2012 [DOI] [PubMed] [Google Scholar]
- 5.Fukuda S, Ohno H: Gut microbiome and metabolic diseases. Semin Immunopathol 36: 103–114, 2014 [DOI] [PubMed] [Google Scholar]
- 6.Vaziri ND, Wong J, Pahl M, Piceno YM, Yuan J, DeSantis TZ, Ni Z, Nguyen TH, Andersen GL: Chronic kidney disease alters intestinal microbial flora. Kidney Int 83: 308–315, 2013 [DOI] [PubMed] [Google Scholar]
- 7.Ramezani A, Raj DS: The gut microbiome, kidney disease, and targeted interventions. J Am Soc Nephrol 25: 657–670, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Meyer TW, Hostetter TH: Uremic solutes from colon microbes. Kidney Int 81: 949–954, 2012 [DOI] [PubMed] [Google Scholar]
- 9.Lacy BE, Levy LC: Lubiprostone: A chloride channel activator. J Clin Gastroenterol 41: 345–351, 2007 [DOI] [PubMed] [Google Scholar]
- 10.Fei G, Wang YZ, Liu S, Hu HZ, Wang GD, Qu MH, Wang XY, Xia Y, Sun X, Bohn LM, Cooke HJ, Wood JD: Stimulation of mucosal secretion by lubiprostone (SPI-0211) in guinea pig small intestine and colon. Am J Physiol Gastrointest Liver Physiol 296: G823–G832, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Stephen AM, Wiggins HS, Cummings JH: Effect of changing transit time on colonic microbial metabolism in man. Gut 28: 601–609, 1987 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Khalif IL, Quigley EM, Konovitch EA, Maximova ID: Alterations in the colonic flora and intestinal permeability and evidence of immune activation in chronic constipation. Dig Liver Dis 37: 838–849, 2005 [DOI] [PubMed] [Google Scholar]
- 13.Tanaka T, Doi K, Maeda-Mamiya R, Negishi K, Portilla D, Sugaya T, Fujita T, Noiri E: Urinary L-type fatty acid-binding protein can reflect renal tubulointerstitial injury. Am J Pathol 174: 1203–1211, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Vaziri ND: CKD impairs barrier function and alters microbial flora of the intestine: A major link to inflammation and uremic toxicity. Curr Opin Nephrol Hypertens 21: 587–592, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Evenepoel P, Meijers BK, Bammens BR, Verbeke K: Uremic toxins originating from colonic microbial metabolism. Kidney Int Suppl 114: S12–S19, 2009 [DOI] [PubMed] [Google Scholar]
- 16.Anders HJ, Andersen K, Stecher B: The intestinal microbiota, a leaky gut, and abnormal immunity in kidney disease. Kidney Int 83: 1010–1016, 2013 [DOI] [PubMed] [Google Scholar]
- 17.Declèves AE, Sharma K: Novel targets of antifibrotic and anti-inflammatory treatment in CKD. Nat Rev Nephrol 10: 257–267, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Musch MW, Wang Y, Claud EC, Chang EB: Lubiprostone decreases mouse colonic inner mucus layer thickness and alters intestinal microbiota. Dig Dis Sci 58: 668–677, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wikoff WR, Anfora AT, Liu J, Schultz PG, Lesley SA, Peters EC, Siuzdak G: Metabolomics analysis reveals large effects of gut microflora on mammalian blood metabolites. Proc Natl Acad Sci U S A 106: 3698–3703, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Niwa T: Indoxyl sulfate is a nephro-vascular toxin. J Ren Nutr 20[Suppl]: S2–S6, 2010 [DOI] [PubMed] [Google Scholar]
- 21.Toyohara T, Suzuki T, Morimoto R, Akiyama Y, Souma T, Shiwaku HO, Takeuchi Y, Mishima E, Abe M, Tanemoto M, Masuda S, Kawano H, Maemura K, Nakayama M, Sato H, Mikkaichi T, Yamaguchi H, Fukui S, Fukumoto Y, Shimokawa H, Inui K, Terasaki T, Goto J, Ito S, Hishinuma T, Rubera I, Tauc M, Fujii-Kuriyama Y, Yabuuchi H, Moriyama Y, Soga T, Abe T: SLCO4C1 transporter eliminates uremic toxins and attenuates hypertension and renal inflammation. J Am Soc Nephrol 20: 2546–2555, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Vaziri ND, Yuan J, Nazertehrani S, Ni Z, Liu S: Chronic kidney disease causes disruption of gastric and small intestinal epithelial tight junction. Am J Nephrol 38: 99–103, 2013 [DOI] [PubMed] [Google Scholar]
- 23.Wang F, Jiang H, Shi K, Ren Y, Zhang P, Cheng S: Gut bacterial translocation is associated with microinflammation in end-stage renal disease patients. Nephrology (Carlton) 17: 733–738, 2012 [DOI] [PubMed] [Google Scholar]
- 24.Moeser AJ, Nighot PK, Roerig B, Ueno R, Blikslager AT: Comparison of the chloride channel activator lubiprostone and the oral laxative Polyethylene Glycol 3350 on mucosal barrier repair in ischemic-injured porcine intestine. World J Gastroenterol 14: 6012–6017, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.De Lisle RC: Lubiprostone stimulates small intestinal mucin release. BMC Gastroenterol 12: 156, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Vaziri ND, Yuan J, Norris K: Role of urea in intestinal barrier dysfunction and disruption of epithelial tight junction in chronic kidney disease. Am J Nephrol 37: 1–6, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Wong J, Piceno YM, Desantis TZ, Pahl M, Andersen GL, Vaziri ND: Expansion of urease- and uricase-containing, indole- and p-cresol-forming and contraction of short-chain fatty acid-producing intestinal microbiota in ESRD. Am J Nephrol 39: 230–237, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Wang F, Zhang P, Jiang H, Cheng S: Gut bacterial translocation contributes to microinflammation in experimental uremia. Dig Dis Sci 57: 2856–2862, 2012 [DOI] [PubMed] [Google Scholar]
- 29.Wang IK, Lai HC, Yu CJ, Liang CC, Chang CT, Kuo HL, Yang YF, Lin CC, Lin HH, Liu YL, Chang YC, Wu YY, Chen CH, Li CY, Chuang FR, Huang CC, Lin CH, Lin HC: Real-time PCR analysis of the intestinal microbiotas in peritoneal dialysis patients. Appl Environ Microbiol 78: 1107–1112, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kakino M, Izuta H, Ito T, Tsuruma K, Araki Y, Shimazawa M, Oyama M, Iinuma M, Hara H: Agarwood induced laxative effects via acetylcholine receptors on loperamide-induced constipation in mice. Biosci Biotechnol Biochem 74: 1550–1555, 2010 [DOI] [PubMed] [Google Scholar]
- 31.Kim SW, Suda W, Kim S, Oshima K, Fukuda S, Ohno H, Morita H, Hattori M: Robustness of gut microbiota of healthy adults in response to probiotic intervention revealed by high-throughput pyrosequencing. DNA Res 20: 241–253, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Furusawa Y, Obata Y, Fukuda S, Endo TA, Nakato G, Takahashi D, Nakanishi Y, Uetake C, Kato K, Kato T, Takahashi M, Fukuda NN, Murakami S, Miyauchi E, Hino S, Atarashi K, Onawa S, Fujimura Y, Lockett T, Clarke JM, Topping DL, Tomita M, Hori S, Ohara O, Morita T, Koseki H, Kikuchi J, Honda K, Hase K, Ohno H: Commensal microbe-derived butyrate induces the differentiation of colonic regulatory T cells. Nature 504: 446–450, 2013 [DOI] [PubMed] [Google Scholar]
- 33.Soga T, Ohashi Y, Ueno Y, Naraoka H, Tomita M, Nishioka T: Quantitative metabolome analysis using capillary electrophoresis mass spectrometry. J Proteome Res 2: 488–494, 2003 [DOI] [PubMed] [Google Scholar]
- 34.Akiyama Y, Takeuchi Y, Kikuchi K, Mishima E, Yamamoto Y, Suzuki C, Toyohara T, Suzuki T, Hozawa A, Ito S, Soga T, Abe T: A metabolomic approach to clarifying the effect of AST-120 on 5/6 nephrectomized rats by capillary electrophoresis with mass spectrometry (CE-MS). Toxins (Basel) 4: 1309–1322, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.