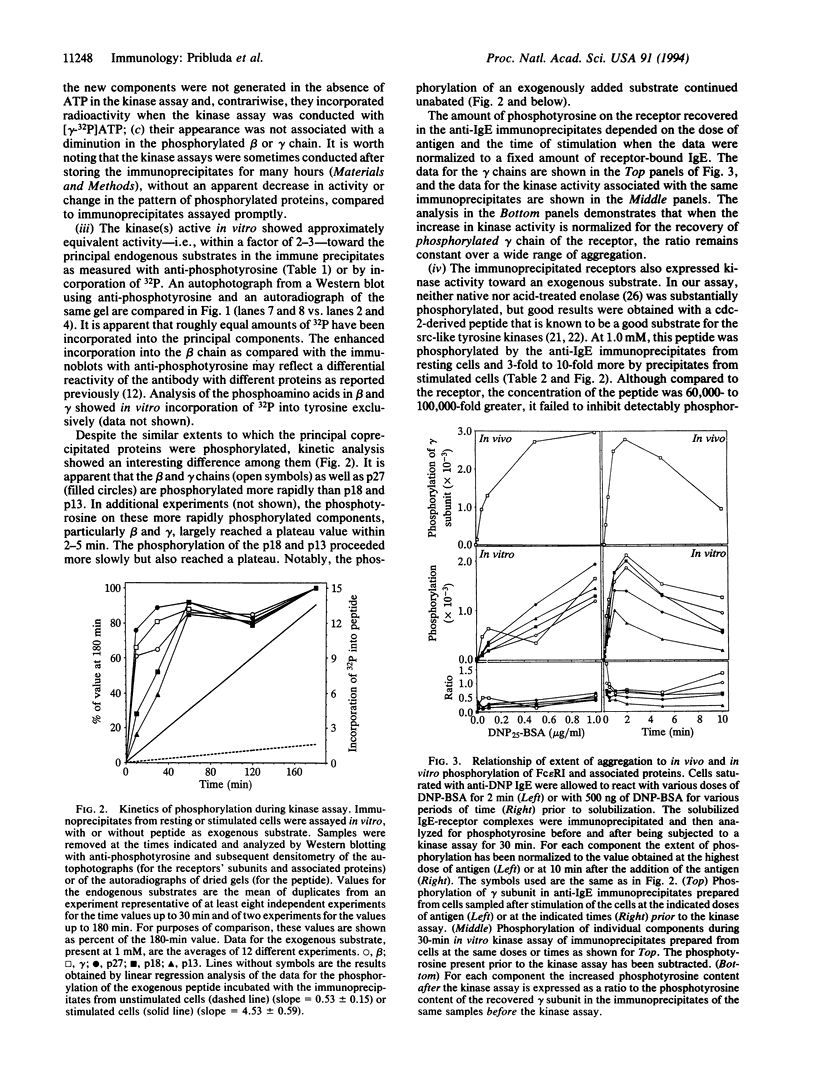
Abstract
When aggregated, the high-affinity receptors for IgE on mast cells (Fc epsilon RI) launch a series of phosphorylations, particularly of protein tyrosines. We have analyzed how aggregation initiates this cascade. We examined Fc epsilon RI from unstimulated cells and from cells exposed to a polyvalent hapten conjugate that aggregates the Fc epsilon RI via the receptor-bound anti-hapten IgE. We also examined the latter receptors after they had been disaggregated in vitro with monovalent hapten. By an in vitro kinase assay: (i) Unaggregated and disaggregated receptors are associated with a kinase that phosphorylates an exogenous (peptide) substrate but minimally, or not at all, the subunits of Fc epsilon RI or associated proteins (endogenous substrates). After aggregation, phosphorylation of the exogenous substrate is linear with time, but the modification of the endogenous substrates reaches a plateau, presumably because only those endogenous substrates that are adjacent to the kinase are phosphorylated. (ii) Aggregated receptors and disaggregated receptors have enhanced kinase activity toward exogenous substrate. The state of phosphorylation of the receptor correlates strongly with the yield of enhanced kinase activity. We propose that upon aggregation of Fc epsilon RI, a constitutively associated kinase phosphorylates endogenous substrates by transphosphorylation. As a result, additional kinase activity becomes manifest and this promotes further transphosphorylation. In view of the homology between Fc epsilon RI and other receptors central to the immune response, the latter receptors likely utilize a similar transphosphorylation mechanism.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Barsumian E. L., Isersky C., Petrino M. G., Siraganian R. P. IgE-induced histamine release from rat basophilic leukemia cell lines: isolation of releasing and nonreleasing clones. Eur J Immunol. 1981 Apr;11(4):317–323. doi: 10.1002/eji.1830110410. [DOI] [PubMed] [Google Scholar]
- Beaven M. A., Metzger H. Signal transduction by Fc receptors: the Fc epsilon RI case. Immunol Today. 1993 May;14(5):222–226. doi: 10.1016/0167-5699(93)90167-j. [DOI] [PubMed] [Google Scholar]
- Benhamou M., Gutkind J. S., Robbins K. C., Siraganian R. P. Tyrosine phosphorylation coupled to IgE receptor-mediated signal transduction and histamine release. Proc Natl Acad Sci U S A. 1990 Jul;87(14):5327–5330. doi: 10.1073/pnas.87.14.5327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benhamou M., Ryba N. J., Kihara H., Nishikata H., Siraganian R. P. Protein-tyrosine kinase p72syk in high affinity IgE receptor signaling. Identification as a component of pp72 and association with the receptor gamma chain after receptor aggregation. J Biol Chem. 1993 Nov 5;268(31):23318–23324. [PubMed] [Google Scholar]
- Benhamou M., Siraganian R. P. Protein-tyrosine phosphorylation: an essential component of Fc epsilon RI signaling. Immunol Today. 1992 Jun;13(6):195–197. doi: 10.1016/0167-5699(92)90152-w. [DOI] [PubMed] [Google Scholar]
- Benhamou M., Stephan V., Robbins K. C., Siraganian R. P. High-affinity IgE receptor-mediated stimulation of rat basophilic leukemia (RBL-2H3) cells induces early and late protein-tyrosine phosphorylations. J Biol Chem. 1992 Apr 15;267(11):7310–7314. [PubMed] [Google Scholar]
- Blank U., Ra C., Miller L., White K., Metzger H., Kinet J. P. Complete structure and expression in transfected cells of high affinity IgE receptor. Nature. 1989 Jan 12;337(6203):187–189. doi: 10.1038/337187a0. [DOI] [PubMed] [Google Scholar]
- Cheng H. C., Litwin C. M., Hwang D. M., Wang J. H. Structural basis of specific and efficient phosphorylation of peptides derived from p34cdc2 by a pp60src-related protein tyrosine kinase. J Biol Chem. 1991 Sep 25;266(27):17919–17925. [PubMed] [Google Scholar]
- Cheng H. C., Nishio H., Hatase O., Ralph S., Wang J. H. A synthetic peptide derived from p34cdc2 is a specific and efficient substrate of src-family tyrosine kinases. J Biol Chem. 1992 May 5;267(13):9248–9256. [PubMed] [Google Scholar]
- Cooper J. A., Esch F. S., Taylor S. S., Hunter T. Phosphorylation sites in enolase and lactate dehydrogenase utilized by tyrosine protein kinases in vivo and in vitro. J Biol Chem. 1984 Jun 25;259(12):7835–7841. [PubMed] [Google Scholar]
- DeFranco A. L. Structure and function of the B cell antigen receptor. Annu Rev Cell Biol. 1993;9:377–410. doi: 10.1146/annurev.cb.09.110193.002113. [DOI] [PubMed] [Google Scholar]
- Dombrowicz D., Flamand V., Brigman K. K., Koller B. H., Kinet J. P. Abolition of anaphylaxis by targeted disruption of the high affinity immunoglobulin E receptor alpha chain gene. Cell. 1993 Dec 3;75(5):969–976. doi: 10.1016/0092-8674(93)90540-7. [DOI] [PubMed] [Google Scholar]
- Duclos B., Marcandier S., Cozzone A. J. Chemical properties and separation of phosphoamino acids by thin-layer chromatography and/or electrophoresis. Methods Enzymol. 1991;201:10–21. doi: 10.1016/0076-6879(91)01004-l. [DOI] [PubMed] [Google Scholar]
- Eiseman E., Bolen J. B. Engagement of the high-affinity IgE receptor activates src protein-related tyrosine kinases. Nature. 1992 Jan 2;355(6355):78–80. doi: 10.1038/355078a0. [DOI] [PubMed] [Google Scholar]
- Eiseman E., Bolen J. B. Signal transduction by the cytoplasmic domains of Fc epsilon RI-gamma and TCR-zeta in rat basophilic leukemia cells. J Biol Chem. 1992 Oct 15;267(29):21027–21032. [PubMed] [Google Scholar]
- Hutchcroft J. E., Geahlen R. L., Deanin G. G., Oliver J. M. Fc epsilon RI-mediated tyrosine phosphorylation and activation of the 72-kDa protein-tyrosine kinase, PTK72, in RBL-2H3 rat tumor mast cells. Proc Natl Acad Sci U S A. 1992 Oct 1;89(19):9107–9111. doi: 10.1073/pnas.89.19.9107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jouvin M. H., Adamczewski M., Numerof R., Letourneur O., Vallé A., Kinet J. P. Differential control of the tyrosine kinases Lyn and Syk by the two signaling chains of the high affinity immunoglobulin E receptor. J Biol Chem. 1994 Feb 25;269(8):5918–5925. [PubMed] [Google Scholar]
- Kamps M. P. Determination of phosphoamino acid composition by acid hydrolysis of protein blotted to Immobilon. Methods Enzymol. 1991;201:21–27. doi: 10.1016/0076-6879(91)01005-m. [DOI] [PubMed] [Google Scholar]
- Keegan A. D., Paul W. E. Multichain immune recognition receptors: similarities in structure and signaling pathways. Immunol Today. 1992 Feb;13(2):63–68. doi: 10.1016/0167-5699(92)90136-U. [DOI] [PubMed] [Google Scholar]
- Kent U. M., Mao S. Y., Wofsy C., Goldstein B., Ross S., Metzger H. Dynamics of signal transduction after aggregation of cell-surface receptors: studies on the type I receptor for IgE. Proc Natl Acad Sci U S A. 1994 Apr 12;91(8):3087–3091. doi: 10.1073/pnas.91.8.3087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kinet J. P., Alcaraz G., Leonard A., Wank S., Metzger H. Dissociation of the receptor for immunoglobulin E in mild detergents. Biochemistry. 1985 Jul 16;24(15):4117–4124. doi: 10.1021/bi00336a046. [DOI] [PubMed] [Google Scholar]
- Li W., Deanin G. G., Margolis B., Schlessinger J., Oliver J. M. Fc epsilon R1-mediated tyrosine phosphorylation of multiple proteins, including phospholipase C gamma 1 and the receptor beta gamma 2 complex, in RBL-2H3 rat basophilic leukemia cells. Mol Cell Biol. 1992 Jul;12(7):3176–3182. doi: 10.1128/mcb.12.7.3176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mao S. Y., Alber G., Rivera J., Kochan J., Metzger H. Interaction of aggregated native and mutant IgE receptors with the cellular skeleton. Proc Natl Acad Sci U S A. 1992 Jan 1;89(1):222–226. doi: 10.1073/pnas.89.1.222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paolini R., Jouvin M. H., Kinet J. P. Phosphorylation and dephosphorylation of the high-affinity receptor for immunoglobulin E immediately after receptor engagement and disengagement. Nature. 1991 Oct 31;353(6347):855–858. doi: 10.1038/353855a0. [DOI] [PubMed] [Google Scholar]
- Paolini R., Numerof R., Kinet J. P. Phosphorylation/dephosphorylation of high-affinity IgE receptors: a mechanism for coupling/uncoupling a large signaling complex. Proc Natl Acad Sci U S A. 1992 Nov 15;89(22):10733–10737. doi: 10.1073/pnas.89.22.10733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pribluda V. S., Metzger H. Transmembrane signaling by the high-affinity IgE receptor on membrane preparations. Proc Natl Acad Sci U S A. 1992 Dec 1;89(23):11446–11450. doi: 10.1073/pnas.89.23.11446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rivnay B., Wank S. A., Poy G., Metzger H. Phospholipids stabilize the interaction between the alpha and beta subunits of the solubilized receptor for immunoglobulin E. Biochemistry. 1982 Dec 21;21(26):6922–6927. doi: 10.1021/bi00269a047. [DOI] [PubMed] [Google Scholar]
- Takai T., Li M., Sylvestre D., Clynes R., Ravetch J. V. FcR gamma chain deletion results in pleiotrophic effector cell defects. Cell. 1994 Feb 11;76(3):519–529. doi: 10.1016/0092-8674(94)90115-5. [DOI] [PubMed] [Google Scholar]
- Yamashita T., Mao S. Y., Metzger H. Aggregation of the high-affinity IgE receptor and enhanced activity of p53/56lyn protein-tyrosine kinase. Proc Natl Acad Sci U S A. 1994 Nov 8;91(23):11251–11255. doi: 10.1073/pnas.91.23.11251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu K. T., Lyall R., Jariwala N., Zilberstein A., Haimovich J. Antigen- and ionophore-induced signal transduction in rat basophilic leukemia cells involves protein tyrosine phosphorylation. J Biol Chem. 1991 Nov 25;266(33):22564–22568. [PubMed] [Google Scholar]




