Abstract
During the past two decades, researchers at the University of Arizona’s Center for Gamma-Ray Imaging (CGRI) have explored a variety of approaches to gamma-ray detection, including scintillation cameras, solid-state detectors, and hybrids such as the intensified Quantum Imaging Device (iQID) configuration where a scintillator is followed by optical gain and a fast CCD or CMOS camera. We have combined these detectors with a variety of collimation schemes, including single and multiple pinholes, parallel-hole collimators, synthetic apertures, and anamorphic crossed slits, to build a large number of preclinical molecular-imaging systems that perform Single-Photon Emission Computed Tomography (SPECT), Positron Emission Tomography (PET), and X-Ray Computed Tomography (CT). In this paper, we discuss the themes and methods we have developed over the years to record and fully use the information content carried by every detected gamma-ray photon.
Keywords: molecular imaging, instrumentation, detectors, collimation, estimation, reconstruction, SPECT, PET, CT
1. INTRODUCTION
Molecular imaging is a minimally invasive tomography (3D imaging) method in which the objective is to elucidate biological function via the uptake of trace amounts of radio-labeled targeting molecules. These radiotracers, known as radiopharmaceuticals when used in the clinic, can be designed to reveal many scales of phenomena in living tissues – from the uniformity of perfusion of blood in the muscles of the heart down to the expression of receptors at the cellular level. When molecular imaging is used in the clinical setting, it is also known as nuclear medicine.
There are two principal techniques in molecular imaging: Single Photon Emission Computed Tomography (SPECT), which makes use of gamma rays emitted one-by-one, and Positron Emission Tomography (PET), in which a correlated pair of gamma-ray photons, arising from the matter-antimatter annihilation of a positron with an electron, together comprise the detected signal.[1] The Center for Gamma-Ray Imaging (CGRI) was founded in 1999 with the mission of advancing the state of the art of preclinical SPECT, but many of the technological and theoretical tools developed in the center are applicable to preclinical PET, and can be translated to clinical SPECT and PET as well.
In this overview, we trace through the new detector technologies, imaging principles, and completed systems created and investigated during CGRI’s first 15 years of operation, and consider the impact that concurrent advances in computing power have had on the ability to record and fully use the information content carried by gamma-ray photons.
1.1 Origins
CGRI’s origin was inspired by two prior decades of research, dating back to the early 1970s, into technology and theory for nuclear medicine by Barrett, Barber, Patton, Woolfenden, their collaborators, and multiple generations of Optical Sciences Ph.D. and Masters students. Among the myriad fundamental accomplishments of this period, key to the founding of CGRI were the development of compact modular scintillation cameras [2] with practical maximum-likelihood event processing,[3] the demonstration of the FastSPECT concept in which a large enough number of these cameras acquire projection data that tomography can be achieved without any movement of cameras or patient,[4] and the exploration of cadmium-zinc-telluride (CZT) focal-plane arrays as next-generation gamma-ray detectors.[5–7] The latter effort had culminated in a custom readout ASIC colloquially known as the Arizona readout.[6, 8]
There is also a strong image-science component in CGRI,[9] and the theoretical foundations included objective measures of image quality and means for incorporating carefully measured camera and system calibrations into data processing and tomographic reconstruction.[10] Indeed of the original core projects in CGRI, one was dedicated to detectors, one to electronics and data processing, one to systems, and two to theory and simulations. In this paper, we are focusing on the instrumentation projects.
1.2 Initial Technology
At CGRI’s founding in 1999 there were two primary detector technologies at hand: the 2×2-photomultiplier (PMT)-array modular scintillation camera shown at left in fig. 1 and the recently prototyped 64×64-pixel-array CZT detector shown at right in fig. 1. Also present was a 48×48 precursor [11] mounted in a cryogenic cooler that was used to study the deep trap structure for holes.[12] Both CdTe and CZT suffer from relatively poor hole transport that leads to severe tailing in energy spectra. An important contribution made at this time was the formulation of the small pixel effect, the observation that the effect of poor transport of one type of charge carrier is minimized if the read-out electrodes collecting the complementary carrier are made small relative to the detector thickness.[13]
Figure 1.
Detectors at CGRI’s founding: a) the 2×2 square-end-PMT NaI(Tl) modular gamma camera is shown without its back case and connectors; and b) the 64×64 CZT pixel-array detector indium-bump bonded to an Arizona readout ASIC on a ceramic carrier board.
The primary SPECT system in operation was FastSPECT I, shown in fig. 2. This system was initially conceived as a human brain imager, but with the rising interest in small-animal (now called preclinical) imaging beginning in the mid-1990s, it was ready for outfitting with a new aperture suitable for mice and rats.
Figure 2.
CGRI’s original FastSPECT I imager incorporates 24 of the modular cameras in fig. 1a arranged in two rings, with 24 matching pinholes machined into an exchangeable lead cylinder.
1.3 Scientific Goals
The overall objective of the past two decades of instrumentation development has been to develop advanced gamma-ray detectors and imaging systems that push the limits of spatial and temporal resolution for molecular imaging. A key challenge in emission-tomography-imager design is that there are inevitable tradeoffs between resolution, sensitivity, and field of view. One of the few ways to enable all three attributes to be improved at once is by increasing the number of effective detector pixels.[14, 15]
2. CZT DETECTORS AND SYSTEMS
A general way of describing the spatial-positioning performance of a gamma-ray detector, whether it is intrinsically pixelated or continuous, is via the space-bandwidth product.[16] This attribute, which is essentially a count of the number of resolvable detector positions to which an event can be assigned, allows a comparison between systems working with different detector technologies, sizes, and magnifications. CGRI’s early detector developments were aimed at improving the space-bandwidth products of modular gamma-camera detectors by creating a new generation of camera with more PMTs and new information-preserving electronics, as well as exploiting the capabilities of the new CZT detectors in a variety of system applications.
2.1 Semiconductor detectors and the Spot Imager
The CGRI CZT detector consisted of a 1” × 1” × 2mm-thick cadmium-zinc-telluride crystal patterned with a common cathode electrode on the illumination side and a 64×64 array of 380µm × 380µm-pitch anode electrodes on the readout side, as shown in fig. 3a.[6] The anode pixels were indium-bump-bonded to input contacts on the Arizona ASIC, a gated- integrator readout that clocked out the contents of the entire pixel-array every millisecond, in row-by-column format much like a CCD. In-line processing via a digital-signal-processor (DSP) board subtracted dark-current signals for each pixel and compared the remainders against a threshold table to find events. Since the dark (or leakage) current was sensitive to even minor temperature fluctuations, very careful regulation of a thermoelectric cooler was required for stable long-term operation.
Figure 3.
a) CGRI’s original CZT detector comprised a 2mm thick crystal indium bump-bonded to a custom readout ASIC; b) a matching collimator was produced by Tecomet; and c) the first dedicated imager produced in CGRI with this detector and collimation technology was known as the Spot Imager.
A matching collimator was produced by Tecomet of Woburn, MA. Many thin layers of photolithographically etched tungsten were laminated together to form a very precise, high-aspect-ratio parallel-hole collimator with one bore per pixel as shown in fig. 3b. The first dedicated imaging system based on this detector/collimator combination was known as the Spot Imager.[17] The imaging components were mounted in a compact head assembly, with the supporting electronics and a circulating coolant system connected via a flexible tether arrangement. Planar test images on phantoms and small-animal models showed an unprecedented level of spatial detail.
2.2 A Compact Dual-Modality SPECT/CT System
Not long after the idea emerged that SPECT and PET would be extremely valuable as preclinical research tools, the concept of dual-modality imaging was introduced by Hasegawa et al for both clinical and preclinical applications. Addition of a nearly-simultaneously-measured anatomical image that could be co-registered with the functional image would increase the value of the study by increasing confidence in the location of molecular imaging signals. It would also allow compensation for a variety of physical effects, including attenuation, that must be included in tomographic reconstructions if quantitative accuracy is needed. This is especially true in clinical SPECT and PET.
CGRI’s first dual modality system was built as a compact desktop system with a single Spot Imager combined with a micro-focal x-ray source and a 1-megapixel x-ray camera, all in a powder-coated, shielded enclosure as shown in fig. 4.[18, 19] As a prototype SPECT/CT system, the rotation axis was vertical; in CGRI’s instruments that go into routine preclinical service, the mouse or rat is held in a more physiologically normal horizontal orientation.[20]
Figure 4.
a) CGRI’s Compact Dual-Modality System is a desktop instrument that combines a CZT spot imager for SPECT with a CGRI-designed transmission x-ray system; b) x-ray and SPECT measurements are made sequentially, with a 90-degree arrangement that minimizes the scatter of x-rays into the CZT camera.
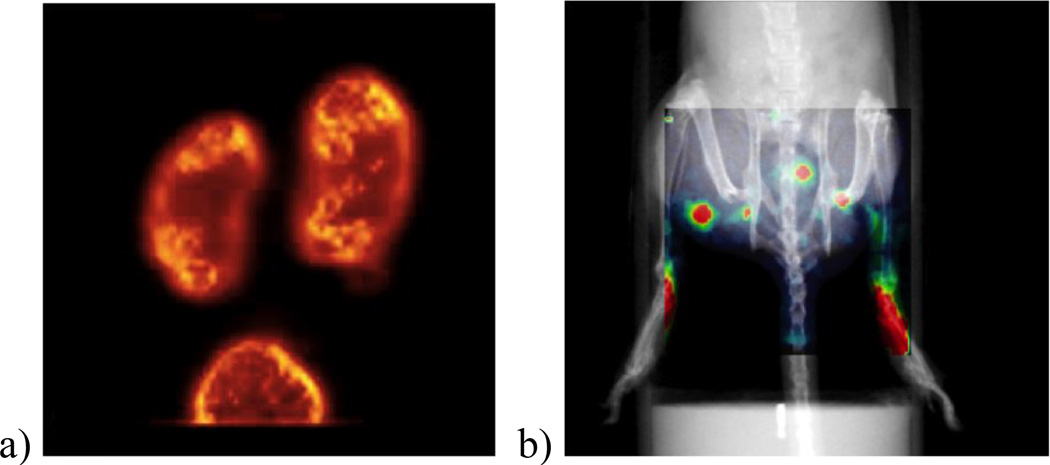
The Compact Dual-Modality System produced remarkable tomographic and planar images. The 99mTc-glucarate mouse-kidney image in fig. 5a below served as a reference standard that commercial system developers aspired to for many years. The image in fig. 5b illustrates the value of adding the x-ray information as an anatomical backdrop: the 99mTc-sulfur colloids injected in both hind limbs can be seen migrating to and concentrating in abdominal lymph nodes in a murine model of lymphedema.[21]
Figure 5.
Sample images from the Compact Dual-Modality System: a) a 99mTc-glucarate SPECT study of mouse kidney (overall image size is 25mm × 25mm); and b) an overlay of planar x-ray and SPECT measurements show the transport of 99mTc-sulfur colloid injected under the skin in both hind limbs (overall image size is 50mm × 50mm).
2.3 SemiSPECT – A CZT FastSPECT
Semiconductor detectors, which directly convert gamma-ray energy into electron-hole pairs, can in principle have energy resolutions a factor ten or better than detector technologies that use an intermediate scintillation step. But this is only achieved when charge transport and collection is relatively complete. In the CGRI CZT detectors, the small anode pixel size (relative to detector thickness) minimizes the impact of CZT’s poor hole transport via the small pixel effect, but small pixels in turn inevitably result in significant charge sharing. When accounted for, charge sharing between neighboring pixels can be turned into an advantage in that the signals in a small cluster of pixels centered on the interaction location provide data that can be processed with maximum-likelihood methods to not only recover the event energy, but also perform position estimation with sub-pixel-size accuracy. It was even found to permit estimation of the depth of interaction inside the crystal if the detector response was accurately mapped with careful calibration.[22]
In order to build a CZT version of FastSPECT concept, an efficient acquisition strategy was needed that read out not just single pixels above threshold, but the local pixel neighborhoods from which the complete event could be reassembled. This was accomplished by streaming digitized pixel data through a custom-designed FPGA board that kept enough rows in memory that dark current subtraction and triggering could operate on 3×3 pixel groups rather than one pixel at a time. When an event was found, all 9 individual pixel values were streamed back to a PCI list-mode buffer. Careful logic ensured that only one cluster of pixels was sent for each event.
Eight CGRI CZT detectors were mounted in a compact imager called SemiSPECT, designed specifically to image mice.[23] Each was provided with its own thermoelectric cooler for precise temperature control as well as complete acquisition electronics and dedicated PCI back-end list-mode buffer. A rotary stage system allowed for the collection of additional angular views and a lift stage permitted helical scanning. Two computers were used to run the imager: one for acquisition and another dedicated to implementing the eight independent software-PID temperature controllers and monitoring ASIC currents and other environmental conditions to protect these valuable detectors from damage.
SemiSPECT was designed with pinhole collimation in order to provide higher sensitivity compared to the parallel-hole collimation in the Spot Imager and also make it possible to increase the field of view via a slight demagnification. Several exchangeable pinhole carriers with different diameters and pinhole sizes, one example of which is shown in fig. 6a, were produced and used during imaging experiments. As was pioneered in the FastSPECT I system, a three-axis stage system was used to perform exhaustive calibration of the system imaging matrix (H-matrix) that is the basis for iterative tomographic reconstruction methods. Example images from studies of tumor xenografts in mice are shown in fig. 7.
Figure 6.
The SemiSPECT CZT imager with 8 detectors is shown in a) with top and aperture removed; b) shows the detectors mounted inside the dry-nitrogen dewar; and c) shows the system together with its external chiller and electronics rack.
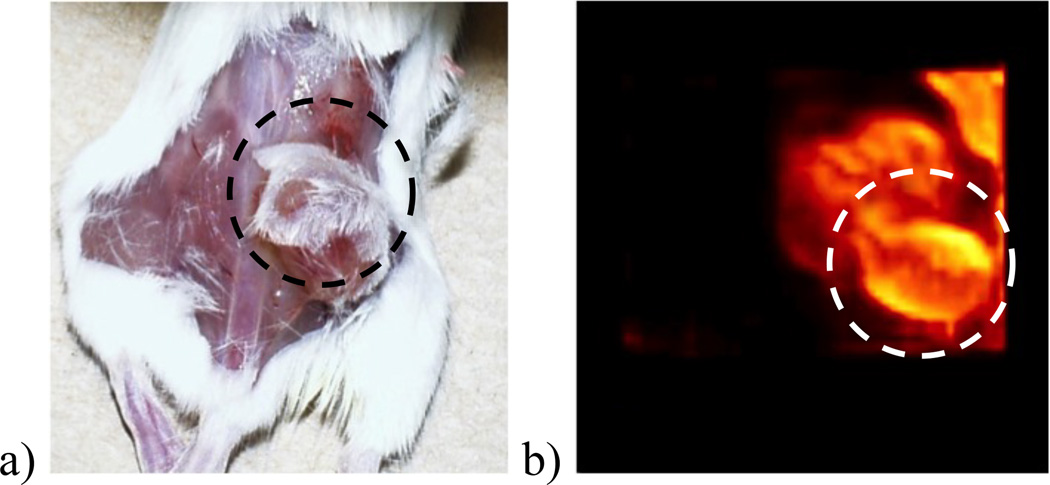
Figure 7.
A xenografted A549 lung tumor, shown inside the dashed circle in the optical image in a), was imaged in SemiSPECT with 99mTc-glucarate. A maximum-intensity projection (MIP) from the reconstruction is shown in b).
There were many challenges in the fabrication of CGRI’s CZT pixel array detectors. Two issues stood out as hard to overcome: 1) the tendency of crystal boules to grow as assemblies of smaller single crystals that made it rare to find large-area (> 1”×1”) detector-quality material and 2) the inherent mismatch in the thermal coefficients of expansion between CZT and the bump-bonded Si read-out ASIC.
3. MODULAR SCINTILLATION CAMERAS AND SYSTEMS
The exploration of CZT was just one branch of CGRI’s technology development mission; another was the design of a new generation of modular gamma cameras and associated electronics that could take full advantage of the power of maximum-likelihood (ML) estimation for deducing gamma-ray interaction parameters: deposited energy, 3D interaction location, and time.[24, 25] The rapid progress in computing power, networking, and storage was making it clear that old limitations on what was feasible in collecting and processing data no longer applied.
Modular gamma cameras (ModCAMs) are essentially small versions of Anger’s classic design, but optimized specifically to produce data that will be processed with ML methods that consider the statistical nature of the signals, rather than conventional methods that rely on inaccurate centroiding algorithms. The new ModCAM design was chosen from an array of prototypes produced by vendors to our specifications based on maximizing a performance-to-price ratio. The final version, shown in fig. 8, incorporated a 4.5” × 4.5” × 5 mm monolithic NaI(Tl) crystal, an 8–12 mm thick light guide, and a 3×3 array of PMTs.[26]
Figure 8.
A schematic of the 3×3 CGRI modular scintillation camera is shown in a), the final packaged version in b), and the camera with its super-list-mode acquisition electronics in c).
New electronics were designed that split the acquisition process into separate front-end event processors and back-end event buffers to implement a super-list-mode architecture.[15] This new approach packages all of the PMT signals and the time associated with each event into a digital data packet that streams to the back-end buffer where a photon-by-photon list accumulates during acquisition. In the ModCAM system, there are 108 bits of PMT signal and 32 bits of timing information recorded for each detected gamma-ray, but the architecture is scalable to much larger data vectors if needed. Indeed, the same back-end buffers are used in the SemiSPECT system.
ModCAMs operate independently and are relatively inexpensive, and are therefore convenient building blocks for imaging systems of arbitrary complexity.
3.1 FastSPECT II
A second-generation FastSPECT system was built that incorporated 16 copies of the new ModCAMS and a host of algorithm improvements for position estimation and image reconstruction that were made possible with the introduction of computing hardware that supported parallel processing – at first via computer clusters and later via dedicated CPUs and GPUs. Some of the innovative attributes of FastSPECT II, which has been in continuous operation for the last decade, include the arrangement of the 16 cameras into two rings of 8 to yield good angular sampling, the ability to perform recalibration of cameras and the imaging matrix in situ with a 5-axis stage system, exchangeable pinhole apertures and adjustable camera positions that allow the selection of magnification factors in a range between 2.5–18 (albeit with recalibration required), and an overall digital data generation rate of 50 GBits per second that is sorted through by FPGAs to identify gamma-ray events and create the super-list-mode packets.[26] The FastSPECT systems’ unique strengths are the inherent dynamic imaging capability provided by stationary instruments that acquire all of the projections required for tomographic reconstruction in parallel without motion of either system or imaging subject. Commercial systems based on the stationary concept have been inspired by FastSPECT I and II, but they still require translating the subject,[27] and they can’t capture the very fast tracer uptake and washout dynamics that CGRI’s systems are capable of studying. The internal and external structure of FastSPECT II is shown in fig. 9.
Figure 9.
The internal framework and camera arrangement of FastSPECT II is shown in a); the external shielding and calibration/positioning stage system is shown in b).
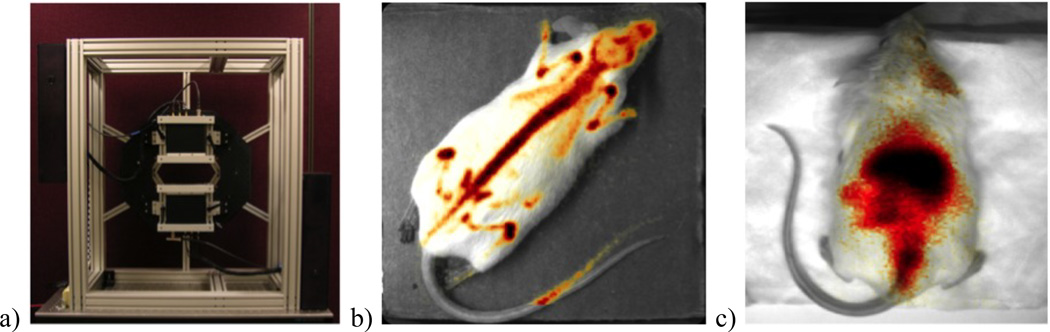
The power of magnification in visualizing fine structures is demonstrated in the FastSPECT II images shown in fig. 10.[28] In a study of the invasion of bone by neuroblastoma cancer cells implanted in the marrow of a mouse femur, the high magnification mode of FastSPECT II allows the clear visualization of the tumor’s effect on bone in the knee, while the normal magnification mode allows convenient acquisition of the whole body image. In FastSPECT II, separate imaging sessions were required to capture these images – the ability to see detail at multiple length scales in the same acquisition motivated CGRI researchers to propose a whole new class of instruments capable of changing configuration on the fly, ie to perform adaptive imaging in response to the radiotracer distribution.[29]
Figure 10.
a) Whole-body mouse bone scan acquired on FastSPECT II using 99mTc-MDP in normal 3× magnification. The rectangle indicates the field of view of the high magnification (18×) acquisition, an image from which is shown in b). The areas of high uptake in the mouse knee show insult from invading neuroblastoma tumor.
3.2 FaCT – An adaptive CT for FastSPECT II
A companion x-ray computed tomography system called FaCT was designed and built in order to provide co-registered anatomical landmarks for FastSPECT II’s functional images.[30] This CT system was designed around a microfocal x-ray source with a 6-micron source spot and a two-megapixel commercial x-ray flat-panel detector. Since we had recognized the value of on-the-fly adjustable magnification for emission tomography, this cone-beam CT was designed with a translation stage on the rotating gantry that permits the source and detector to move relative to the object (and rotation axis) to bring that capability to transmission tomography. To compensate for the motion and prevent flex in the system, a dynamic counterweight was included to keep the center of gravity invariant.[31]
Since high-resolution x-ray tomography is associated with elevated overall radiation dose levels, a 4-blade masking system was incorporated that allows the beam to be shaped on the fly to avoid exposing areas outside a region of interest. Combined with the programmable tube voltage and current, the adjustable exposure time per image, the variable number of turns per helical acquisition, and the stage-controlled pitch of the helix, this system has complete flexibility in the data set acquired for tomographic reconstruction.
3.3 M3R and the Prototype Adaptive System
Adaptive SPECT imaging embodies a nonlinear optimization strategy in which imaging parameters are automatically adjusted in response to the object, or more specifically the data being acquired from the object.[29] As mentioned above, our interest in creating an adaptive version of the FastSPECT II system was partly motivated by the challenges we faced in the mouse model of neuroblastoma. We simultaneously needed imaging at length scales on the order of 100µm in order to find the very small lesions with high sensitivity in the mouse knee, while also being able to acquire rapid whole-body bone scans at 1-mm-scale reconstructed resolution. The quandary this presented is that no single imager configuration could achieve both results, since resolution, sensitivity, and field of view can virtually never be simultaneously optimized.
We realized that if the apertures and cameras were made movable under computer control (robotic), and adjustments were made in accordance with image-science principles, not only would imaging at multiple length scales in the same session become practical, but significant gains could be achieved in the performance of specific scientific imaging tasks. These ideas were explored in a pair of ModCAM-based instruments, shown in fig. 12, called M3R (for multi-ModCAM multi-resolution system) [32] that assessed task-based hardware optimization [33] and the Prototype Adaptive System that allowed testing of adaptation rules.[34]
Figure 12.
a) The task-based-hardware-optimization system known as M3R used 4 cameras, each with a different collimator configuration. b) The prototype adaptive system placed camera and aperture positions under computer control.
M3R allowed the simultaneous acquisition of image data from the same object with multiple choices of collimators and magnifications. Results from phantom experiments with M3R proved that imaging related tasks could often be performed quantitatively better when acquisitions were made with a combination of configurations rather than any single design choice. It also turned out to be a good test bed for multiplexed (overlapping) multi-pinhole small-animal imaging. The prototype adaptive system used only a single pinhole at a time, but mounted an array of different sizes that could be selected between in an aperture box that could be translated independently of the camera. This allowed testing of simple adaptation rules, such as maximizing the magnification of a phantom lesion to fill the camera face as angular samples were acquired throughout a complete circular trajectory.
3.4 AdaptiSPECT
The AdaptiSPECT imager is a 16-camera FastSPECT-II-inspired adaptive small-animal SPECT system currently being integrated in which apertures and camera positions are under full computer control. The system can smoothly and rapidly move through a matrix of configurations – ranging from high-magnification single-pinhole-per-camera views to multi-pinhole projections that emphasize larger fields of view. The system has initially been designed around high-quality versions of our FastSPECT-II-generation ModCAMs that achieve a ~2.25mm × 2.25mm intrinsic planar resolution.[35] The imaging aperture is made of three coaxial cylinders of increasing diameter that attach together endwise to make a single assembly that can be translated to select which aperture cylinder is imaging the object onto the cameras. The system design and internal skeleton without shielding are shown in fig. 13.
Figure 13.
a) The rendered 3D design of the AdaptiSPECT imager. As with FastSPECT II, cameras are arranged in two rings of 8, but the camera positions and aperture are under computer control. b) An image of the internal skeleton of AdaptiSPECT prior to mounting of cameras and associated electronics.
The final aperture design has a machined tungsten skeleton for strength and rapid-prototype created pinholes aimed at the center of the common field of view.[36] One of the novel aspects of AdaptiSPECT is that it has shutters that allow a choice between single and multiple pinhole projections. These shutters are pneumatically driven and controlled via small solenoid valves. Custom PC boards have been designed that drive the solenoids under computer control and their reliability has been confirmed.
AdaptiSPECT’s acquisition system is based on CGRI’s super-list-mode electronics. The back-end system has been ported to the modern PCI-E bus.[37] The control software operates all 16 cameras on a single CPU outfitted with a Tesla GPU card. We have implemented real-time GPU-powered ML estimation of event positioning based on our contracting-grid algorithm [38] such that a live display of all 16 projections are a routine part of the user interface, even though the list-mode acquisition is preserving all measured signals at full precision in accordance with the super-list-mode, or photon-processing, concept.
3.5 ModPET
As a final example of the flexibility of the ModCAM detectors, two such cameras operating with a common clock source were used to form a compact positron-emission tomography system called ModPET. It was the emergence of scientific GPU computing that made it practical, since the simple acquisition geometry requires sophisticated ML positioning and list-mode ML reconstruction. The novel aspect of ModPET is that the cameras are large relative to mice and are brought into such close proximity that nearly 75% of the 4π steradians of solid angle into which annihilation gamma ray pairs are emitted are covered. It is by no means a limited-angle tomography system, and it achieves a nearly isotropic ~1.5 mm resolution across the field of view as measured with a Rayleigh task, the ability to see two point sources as distinct entities.
This simple imaging system, which embodies the photon-processing concept from detection through reconstruction,[40] produces images easily comparable to the commercial offerings, evident in fig. 14. We also elected to provide companion modalities in the system. An anatomical reference is provided via the use of an optical camera. Furthermore, parallel hole collimators can be attached to the two camera faces and the PMT voltage reduced under computer control, creating a conjugate-counting scintigraphy system that yields dynamic images of SPECT tracers.
Figure 14.
a) ModPET system with opposing ModCAM gamma cameras. b) Single .75-mm-thick slice of 18F-NaF ModPET mouse bone scan overlaid on optical image. c) SPECT scintigraphy mode in ModPET using 99mTc- IL-1ra-Fc to image inflammation caused by topical irritant applied to right ear.[39]
4. INTENSIFIED DETECTORS AND SYSTEMS
4.1 The iQID Camera
One of CGRI’s most innovative gamma-ray-detector developments is iQID,[41] which uses CCD or CMOS sensors with moderately fast frame rates (~200 frames per second) to image scintillation light that has been amplified with an image intensifier.[42] Compared to conventional approaches to gamma-ray detection, the intrinsic spatial resolution is improved by more than one order of magnitude.[43] A key enabling technology is the on-the-fly GPU-based parsing of each frame to extract regions of CMOS pixels, each containing the image of one gamma-ray event. 100 or more gamma-ray events can be resolved on a single 5 msec frame, easily enough for event rates encountered in preclinical and clinical SPECT.
The iQID detector approach is not restricted to gamma rays, and we have been using this approach to image beta-particle emission in a dorsal-skin-flap window-chamber mouse model that is especially useful in cancer research.[44] Using the positrons emitted from 18F to form images, rather than detecting the annihilation gamma rays, allows much higher-resolution, nearly microscopic, imaging of tumor properties such as pattern of FDG uptake. Since alpha particles are an important radio-decay product for targeted radio-nuclide therapy purposes, iQID is being used to image alpha particles as well.[41]
4.2 FastSPECT III
We have used iQID cameras to develop a number of CGRI systems. FastSPECT III is the third generation in the FastSPECT series of stationary SPECT imagers.[45, 46] It uses 20 iQID cameras to image a field of view designed to be appropriate for dynamic evaluation of tracer uptake in rodent brains. It achieves an outstanding reconstructed resolution as evident in fig. 16c, where even the 0.35 mm features are visibly distinct. A key advance was the development of a GPU-enabled reconstruction code for FSIII in which the elements of the H-matrix are interpolated on the fly as needed.[47] This is a fundamental contribution to progress in high-resolution molecular imaging where the number of resolvable detector elements (the system space-bandwidth product) and the number of reconstructed voxels are so large that storage of an H matrix is impossible, even in sparse form.
Figure 16.
a) The FastSPECT III imaging system with associated electronics. b) The imager comprises 20 iQID cameras arranged in rings of 5, 10, and 5 with nearly spherical sampling. c) Resolution phantom images show excellent performance down to the smallest .35mm features.
FastSPECT III is now being refitted with a new, thicker set of scintillation crystals designed to increase the sensitivity for imaging with higher energy isotopes such as 99mTc and 123I.
4.3 The Synthetic Collimator System
Another CGRI system that made use of the exquisite spatial resolution of iQID detectors consists of a movable single detector outfitted with an aperture with ~50 focused pinholes,[48] experimentally demonstrating the synthetic aperture concept suggested by Wilson and Clarkson.[49, 50] This system achieves the remarkable feat of good tomography even though it views the object from only a single direction. It is the translation of the camera behind the aperture, thereby changing the magnification in a depth-dependent way, that samples the z-dimension in the object.
5. CROSSED STRIP SEMICONDUCTOR DETECTORS
Double-sided crossed strip detectors are an appealing alternative to semiconductor pixel-arrays, since they require readout only at two edges. This means that a one-megapixel system requires only 2000 connections between strips and front-end electronics. These connections can be made with wire bonds, making differential thermal expansion a much smaller technical issue than when detector crystals must be bump bonded to their readouts.
5.1 The Silicon Crossed-Strip, Crossed-Slit Prototype System
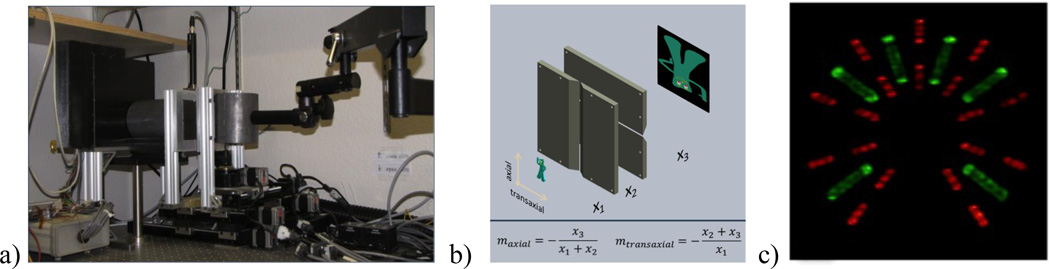
CGRI’s anamorphic Si double-sided strip detector (DSSD) system is a unique SPECT instrument that uses independent crossed slits in conjunction with a mega-pixel silicon crossed-strip gamma-ray detector [51, 52] to yield an imager with an unprecedented collimation flexibility and intrinsic detector resolution.[53] Using the ability of crossed-slit collimators to separately define the magnification in the axial and transaxial directions, this systems offers outstanding opportunities for adaptive imaging and the ability to always use the full detector area.[54]
As with the FastSPECT III system, an OSEM reconstruction based on the H-matrix-on-the-fly approach discussed above was required due to the large number of detector pixels and object voxels.[55] Unlike the earlier system, the H-matrix could not be measured or stored in any form due to the continuously adjustable magnifications and slit openings. Instead, matrix elements were computed from first principles with a GPU-enabled forward-model code that rapidly calculates the point-spread function (PSF) created by the crossed-slit aperture, including radiometry and finite detector-thickness effects. Reconstructed 3D images from the system were exceptional in their ability to easily resolve the internal structure of a phantom made up of Brachytherapy seeds that contained either 125I or 123Pd. The images, shown in fig. 18c immediately reveal that each iodine seed comprises a thin coating on the exterior of a silver-plated rod while the palladium seed is in the form of solid beads separated by cold spacers. The estimated resolution is 70 µm in each direction.
Figure 18.
a) The SiliSPECT crossed slit prototype system. b) It makes use of independent blades to create an effective pinhole that has different magnification in the two directions, allowing an object to be scaled to always make use of the entire detectorarea. c) The excellent energy and spatial resolution of SiliSPECT allowed complete separation of 125I (green) and 123Pd (red) sources in a test phantom.
We have recently been working with CdTe crossed-strip detectors supplied by collaborators at JAXA (Japan Aerospace Exploration Agency).[56] These offer much higher stopping power for the isotopes commonly used in preclinical and clinical studies and will be used in CGRI’s next-generation systems.[57]
6. CONCLUSION
Over the past nearly two decades CGRI has explored a variety of approaches to gamma-ray detection, including scintillation cameras, solid-state detectors, and hybrids such as the intensified Quantum Imaging Device (iQID) configuration where a scintillator is followed by optical gain and a fast CCD or CMOS camera. In every case, the detectors use rigorous maximum-likelihood (ML) methods to estimate multiple attributes of each gamma interaction event, including its 2D or 3D location and the energy deposited, and/or perform tomographic reconstruction. Most systems produce super-list-mode data, in which sensor values without any binning or windowing are accumulated in a large multidimensional list, with information content far exceeding that of conventional digital data.
Building on this photon-processing concept, we have developed a large number of preclinical imaging systems for SPECT and PET. We have made use of a variety of collimation schemes, including single and multiple pinholes, parallel-hole collimators, synthetic apertures, and anamorphic crossed slits. CGRI’s FastSPECT systems are characterized by their unique dynamic capabilities (no motion of the subject, aperture or detector); large number of intrinsic resolution elements (space-bandwidth products ~2×106 for FastSPECT III and SiliSPECT); unprecedented reconstructed spatial resolution in some cases (~70 µm for SiliSPECT); extraordinary energy resolution in other systems (~1% FWHM with the JAXA CdTe detector); and highly accurate system calibration (all systems). We have developed methods and algorithms for in-line photon processing to keep pace with these advances. Moreover, we have pioneered the concept of task-based adaptive imaging and are looking forward to implementing it in the AdaptiSPECT system currently being completed.
Figure 11.
a) The internal structure of the FaCT adaptive CT system that serves as a companion to the FastSPECT II SPECT system. b) A co-registered FastSPECT II/FaCT SPECT/CT image with the CT in grayscale and 99mTc-MDP bonescan SPECT in color.
Figure 15.
a) An iQID camera consisting (from right to left) of a scintillator, image intensifier, lens coupling and CMOS camera. b) The spatial resolution is sufficient to separately image the primary interaction location and the reabsorption of the k x ray that is often emitted following photoelectric interaction.
Figure 17.
The 50 pinhole aperture of the synthetic collimator system is shown along with a representation of the common field of view. This system utilizes camera motion behind the aperture to achieve depth sensitivity.
ACKNOWLEDGMENTS
The work on detectors and systems was largely carried out by graduate students in the College of Optical Sciences. The early CZT detector work was led by Josh Eskin, Kevin Matherson, and Dan Marks. The first spot imager was built by Steven Balzer. George Kastis integrated the compact SPECT/CT system. Hyunki Kim developed SemiSPECT, with contributions from Mick Crawford, William Hunter, and post-doc Todd Peterson. John Sain helped with the design of the new ModCAMS. FastSPECT II was integrated by Yi-Chun Chen with contributions from a senior design team led by Josh Butters and assembly by Noura Eleid and Lance Fesler. Jacob Hesterman built M3R and Melanie Freed developed the prototype adaptive system. Brian Miller led the iQID development effort and also built FastSPECT III with contributions from BME student Stephen Moore. Stephen Moore also developed ModPET. Jared Moore designed and integrated FaCT. Heather Durko developed the crossed-slit Silicon system. Ronan Havelin built the synthetic collimator imager. Cecile Chaix is leading the final design, fabrication and integration of AdaptiSPECT, with assistance from Steven Kovalsky. Jared Moore and visiting scholar Roel Van Holen contributed early design work. Jin Park worked on PET applications of iQID and Liying Chen developed beta imaging. Ling Han is refining FastSPECT III. Esen Salcin is completing studies of the CdTe crossed strip detectors. Todd Bonham developed reconstruction code for FaCT. Luca Caucci, now a faculty member, has led the work on parallel computing, photon processing, and advanced reconstruction methods. Research tech Christy Barber contributed to all small animal studies. Many other students have contributed to CGRI projects not mentioned in this instrumentation overview.
The Center for Gamma-Ray Imaging is supported by NIH/NIBIB grant P41EB002035. Parallel computing is supported by NIH/NIBIB grant R37 EB000803.
REFERENCES
- 1.Wernick MN, Aarsvold JN. Emission tomography: the fundamentals of PET and SPECT. Academic Press; 2004. [Google Scholar]
- 2.Milster TD, Aarsvold JN, Barrett HH, Landesman AL, Mar LS, Patton DD, Roney TJ, Rowe RK, R HS., III A Full-Field Modular Gamma Camera. J. Nucl. Med. 1990;31(5):632–639. [PubMed] [Google Scholar]
- 3.Milster TD, Selberg LA, Barrett HH, Landesman AL, Seacat RH. Digital position estimation for the modular scintillation camera. IEEE Trans. Nucl. Sci. 1985;32(1):748–752. [Google Scholar]
- 4.Klein WP, Barrett HH, Pang IW, Patton DD, Rogulski MM, Sain JD, Smith WE. FASTSPECT: Electrical and mechanical design of a high-resolution dynamic SPECT imager. IEEE Nucl. Sci. Symp. Med. Imag. Conf. Rec. 1995;2:931–933. [Google Scholar]
- 5.Barber HB, Barrett HH, Augustine FL, Hamilton WJ, Apotovsky BA, Dereniak EL, Doty FP, Eskin JD, Garcia JP, Marks DG, Matherson KJ, Woolfenden JM, Young ET. Development of a 64×64 CdZnTe array and associated readout integrated circuit for use in nuclear medicine. J. Elect. Mat. 1997;26:765–772. [Google Scholar]
- 6.Barber HB, Marks DG, Apotovsky BA, Augustine FL, Barrett HH, Butler JF, Dereniak EL, Doty FP, Eskin JD, Hamilton WJ, Matherson KJ, Woolfenden JM, Young ET. Progress in developing focal-plane-multiplexer readout for large CdZnTe arrays for nuclear medicine applications. Nucl. Inst. Meth. Phys. Res. A. 1996;380:262–265. [Google Scholar]
- 7.Matherson KJ, Barber HB, Barrett HH, Eskin JD, Dereniak EL, Marks DG, Woolfenden JM, Young ET, Augustine FL. Progress in the development of large-area modular 64×64 CdZnTe imaging arrays for nuclear medicine. IEEE Trans. Nucl. Sci. 1998;45:354–358. [Google Scholar]
- 8.Barber HB, Apotovsky BA, Augustine FL, Barrett HH, Dereniak EL, Doty FP, Eskin JD, Hamilton WJ, Marks DG, Matherson KJ, Venzon JE, Woolfenden JM, Young ET. Semiconductor pixel detectors for gamma ray imaging in nuclear medicine. Nucl. Inst. Meth. Phys. Res. A. 1997;389:421–428. [Google Scholar]
- 9.Barrett HH, Myers KJ. Foundations of image science. Wiley-VCH; 2003. [Google Scholar]
- 10.Barrett HH, Denny JL, Wagner RF, Myers KJ. Objective assessment of image quality. II. Fisher information, Fourier crosstalk, and figures of merit for task performance. J. Opt. Soc. Am. A. 1995;12(5):834–852. doi: 10.1364/josaa.12.000834. [DOI] [PubMed] [Google Scholar]
- 11.Marks DG, Barber HB, Barrett HH, Dereniak EL, Eskin JD, Matherson KJ, Woolfenden JM, Young ET, Augustine FL, Hamilton WJ, Venzon JE, Apotovsky BA, Doty FP. A 48×48 CdZnTe array with multiplexer readout. IEEE Trans. Nucl. Sci. 1996;43:1253–1259. [Google Scholar]
- 12.Hilton NR. Ph.D. Dissertation. University of Arizona; 2002. Material uniformity of cadmium zinc telluride in gamma-ray imaging detectors. [Google Scholar]
- 13.Barrett HH, Eskin JD, Barber H. Charge transport in arrays of semiconductor gamma-ray detectors. Physical Review Letters. 1995;75(1):156. doi: 10.1103/PhysRevLett.75.156. [DOI] [PubMed] [Google Scholar]
- 14.Rogulski MM, Barber HB, Barrett HH, Shoemaker RL, Woolfenden JM. Ultra-high-resolution brain SPECT imaging: simulation results. IEEE Trans. Nucl. Sci. 1993;40(4):1123–1129. [Google Scholar]
- 15.Peterson TE, Furenlid LR. SPECT detectors: the anger camera and beyond. Phys. Med. Biol. 2011;56(17):R145–R182. doi: 10.1088/0031-9155/56/17/R01. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Barrett HH, Hunter WCJ. Detectors for small-animal SPECT: I. Overview of detector technologies. In: Kupinski MA, Barrett HH, editors. Chap. 2 in [Small-Animal SPECT Imaging] Springer; 2007. [Google Scholar]
- 17.Kastis GA, Barber HB, Barrett HH, Balzer SJ, Lu D, Marks DG, Stevenson G, Woolfenden JM, Appleby M, Tueller J. Gamma-ray imaging using a CdZnTepixel array and a high-resolution parallel-hole collimator. IEEE Trans. Nucl. Sci. 2000;47:1923–1927. [Google Scholar]
- 18.Kastis GA, Wu MC, Balzer SJ, Wilson DW, Furenlid LR, Stevenson G, Barber HB, Barrett HH, Woolfenden JM, Kelly P, Appleby M. Tomographic small-animal imaging using a high-resolution semiconductor camera. IEEE Trans. Nucl. Sci. 2002;49:172–175. doi: 10.1109/TNS.2002.998747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kastis GA, Furenlid LR, Wilson DW, Peterson TE, Barber HB, Barrett HH. Compact CT/SPECT small-animal imaging system. IEEE Trans. Nucl. Sci. 2004;51(1):63–67. doi: 10.1109/TNS.2004.823337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Stevenson GD. The Animal in Small-Animal Imaging. In: Kupinski MA, Barrett HH, editors. Chap. 4 in [Small Animal SPECT] Springer; 2007. [Google Scholar]
- 21.Kriederman BM, Myloyde TL, Witte MH, Dagenais SL, Witte CL, Rennels M, Bernas MJ, Lynch MT, Erickson RP, Caulder MS. FOXC2 haploinsufficient mice are a model for human autosomal dominant lymphedema-distichiasis syndrome. Human Mol. Gen. 2003;12(10):1179–1185. doi: 10.1093/hmg/ddg123. [DOI] [PubMed] [Google Scholar]
- 22.Marks DG, Barber HB, Barrett HH, Tueller J, Woolfenden JM. Improving performance of a CdZnTe imaging array by mapping the detector with gamma rays. Nucl. Inst. Meth. Phys. Res. A. 1999;428:102–112. [Google Scholar]
- 23.Kim H, Furenlid LR, Crawford MJ, Wilson DW, Barber HB, Peterson TE, Hunter WCJ, Liu Z, Woolfenden JM, Barrett HH. SemiSPECT: A small-animal single-photon emission computed tomography (SPECT) imager based on eight cadmium zinc telluride (CZT) arrays. Med. Phys. 2006;33(2):465–475. doi: 10.1118/1.2164070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Barrett HH, Hunter WCJ, Miller BW, Moore SK, Chen Y, Furenlid LR. Maximum-likelihood methods for processing signals from gamma-ray detectors. IEEE Trans. Nucl. Sci. 2009;56(3):725–735. doi: 10.1109/tns.2009.2015308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hunter WCJ, Barrett HH, Furenlid LR. Calibration method for ML estimation of 3D Interaction Position in a thick gamma-ray detector. IEEE Trans. Nucl. Sci. 2009;56:189–196. doi: 10.1109/TNS.2008.2010704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Furenlid LR, Wilson DW, Chen YC, Kim H, Pietraski PJ, Crawford MJ, Barrett HH. FastSPECT II: a second-generation high-resolution dynamic SPECT imager. IEEE Trans. Nucl. Sci. 2004;51(3):631–635. doi: 10.1109/TNS.2004.830975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Have Fvd, Vastenhouw B, Ramakers RM, Branderhorst W, Krah JO, Ji C, Staelens SG, Beekman FJ. U-SPECT-II: an ultra-high-resolution device for molecular small-animal imaging. J. Nucl. Med. 2009;50(4):599–605. doi: 10.2967/jnumed.108.056606. [DOI] [PubMed] [Google Scholar]
- 28.Chen YC. Ph.D. Dissertation. University of Arizona; 2006. System calibration and image reconstruction for a new small-animal SPECT system. [Google Scholar]
- 29.Barrett HH, Furenlid LR, Freed M, Hesterman JY, Kupinski MA, Clarkson E, Whitaker MK. Adaptive SPECT. IEEE Trans. Med. Imag. 2008;27(6):775–788. doi: 10.1109/TMI.2007.913241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Moore JW, Furenlid LR, Barrett HH. Instrumentation Design for Adaptive SPECT/CT; IEEE Nucl. Sci. Symp. Med. Imag. Conf. Rec; 2008. pp. 5585–5587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Moore JW. Ph.D. Dissertation. University of Arizona; 2011. Adaptive X-ray Computed Tomography. [Google Scholar]
- 32.Hesterman JY, Kupinski MA, Furenlid LR, Wilson DW, Barrett HH. The multi-module, multi-resolution system - A novel small-animal SPECT system. Med. Phys. 2007;34(3):987–993. doi: 10.1118/1.2432071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Hesterman JY, Kupinski MA, Clarkson E, Barrett HH. Hardware assessment using the multi-module, multi-resolution system (M^3R) - A signal-detection study. Med. Phys. 2004;34:3034–3044. doi: 10.1118/1.2745920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Freed M, Kupinski MA, Furenlid LR, Wilson DW, Barrett HH. A Prototype Instrument for Single-Pinhole Small-Animal Adaptive SPECT Imaging. Med. Phys. 2008;35(5):1912–1925. doi: 10.1118/1.2896072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Furenlid LR, Moore JW, Freed M, Kupinski MA, Clarkson E, Liu Z, Wilson DW, Woolfenden JM, Barrett HH. Adaptive small-animal SPECT/CT. ISBI Biomedical Imaging: From Nano to Macro. 2008:1407–1410. doi: 10.1109/ISBI.2008.4541269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Chaix C, Kovalsky S, Kupinski MA, Barrett HH, Furenlid LR. Fabrication of the pinhole aperture for AdaptiSPECT. Proc. SPIE. 2014:9214. doi: 10.1117/12.2065907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Chaix C, Kovalsky S, Kosmider M, Barrett HH, Furenlid LR. Integration of AdaptiSPECT: a small-animal adaptive SPECT imaging system. Proc. SPIE. 2013:8853. doi: 10.1117/12.2029768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Hesterman JY, Caucci L, Kupinski MA, Barrett HH, Furenlid LR. Maximum-likelihood estimation with a contracting-grid search algorithm. IEEE Trans. Nucl. Sci. 2010;57(3):1077–1084. doi: 10.1109/TNS.2010.2045898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Liu Z, Barber C, Wan L, Xu H, Hui MM, Furenlid LR, Woolfenden JM. Characterization of 99m Tc-labeled cytokine ligands for inflammation imaging via TNF and IL-1 pathways. Nucl. Med. Biol. 2012;39(7):905–915. doi: 10.1016/j.nucmedbio.2012.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Moore SK. Ph.D. Dissertation. University of Arizona; 2011. ModPET: Novel applications of scintillation cameras to preclinical PET. [Google Scholar]
- 41.Miller BW, Gregory SJ, Fuller ES, Barrett HH, Barber HB, Furenlid LR. The iQID camera: An ionizing-radiation quantum imaging detector. Nucl. Inst. Meth. Phys. Res. A. 2014;767:146–152. doi: 10.1016/j.nima.2014.05.070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Miller BW. Ph.D. Dissertation. University of Arizona; 2011. High-Resolution Gamma-Ray Imaging with Columnar Scintillators and CCD/CMOS Sensors, and FastSPECT III: A Third-Generation Stationary SPECT Imager. [Google Scholar]
- 43.Miller BW, Barrett HH, Furenlid LR, Barber HB, Hunter RJ. Recent advances in BazookaSPECT: Real-time data acquisition and the development of a gamma-ray microscope. Nucl. Inst. Meth. Phys. Res. A. 2008;591(1):272–275. doi: 10.1016/j.nima.2008.03.072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Chen L, Gobar LS, Knowles NG, Wilson DW, Barrett HH. Direct charged-particle imaging system using an ultra-thin phosphor: physical characterization and dynamic applications. IEEE Trans. Nucl. Sci. 2009;56:2628–2635. doi: 10.1109/TNS.2009.2023519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Miller BW, Furenlid LR, Moore SK, Barber HB, Nagarkar VV, Barrett HH. System Integration of FashSPECT III, a Dedicated SPECT Rodent-Brain Imager Based on BazookaSPECT Detector Technology; IEEE Nucl. Sci. Symp. Med. Imag. Conf. Rec; 2009. pp. 4004–4008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Miller BW, Furenlid LR, Moore SK, Barber HB, Nagarkar VV, Barrett HH. FastSPECT III: A third-generation high-resolution dynamic SPECT imager. J. Nucl. Med. 2010;51(Supplement 2):83. [Google Scholar]
- 47.Miller BW, Holen RV, Barrett HH, Furenlid LR. A system calibration and fast iterative reconstruction method for next-generation SPECT imagers. IEEE Trans. Nucl. Sci. 2012;59(5):1990–1996. doi: 10.1109/TNS.2012.2198243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Havelin RJ, Miller BW, Barrett HH, Furenlid LR, Murphy JM, Dwyer RM, Foley MJ. Design and performance of a small-animal imaging system using synthetic collimation. Phys. Med. Biol. 2013;58(10):3397. doi: 10.1088/0031-9155/58/10/3397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Wilson DW, Barrett HH, Clarkson EW. Reconstruction of two-and three-dimensional images from synthetic-collimator data. IEEE Trans. Med. Imag. 2000;19(5):412–422. doi: 10.1109/42.870252. [DOI] [PubMed] [Google Scholar]
- 50.Clarkson E, Wilson DW, Barrett HH. Synthetic collimator for 2D and 3D imaging. Proc. SPIE. 1999;3659:107–117. [Google Scholar]
- 51.Shokouhi S, McDonald BS, Durko HL, Fritz MA, Furenlid LR, Peterson TE. Thick Silicon Double-Sided Strip Detectors for Low-Energy Small-Animal SPECT. IEEE Trans. Nucl. Sci. 2009;56(3):557–564. doi: 10.1109/TNS.2009.2019106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Peterson TE, Shokouhi S, Furenlid LR, Wilson DW. Multi-pinhole SPECT imaging with silicon strip detectors. IEEE Trans. Nucl. Sci. 2009;56(3):646–652. doi: 10.1109/TNS.2009.2012514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Durko HL, Barrett HH, Furenlid LR. High-Resolution Anamorphic SPECT Imaging. IEEE Trans. Nucl. Sci. 2014;61(3):1126–1135. doi: 10.1109/TNS.2014.2304853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Durko HL, Furenlid LR. Adaptive SPECT imaging with crossed-slit apertures. Proc. SPIE. 2014;9214 doi: 10.1117/12.2066188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Durko HL. Ph.D. Dissertation. University of Arizona; 2014. Anamorphic preclinical SPECT imaging with high-resolution silicon double-sided strip detectors. [Google Scholar]
- 56.Takahashi T, Watanabe S, Kouda M, Sato G, Okada Y, Kubo S, Kuroda Y, Onishi M, Ohno R. High-resolution CdTe detector and applications to imaging devices. IEEE Trans. Nucl. Sci. 2001;48(3):287–291. [Google Scholar]
- 57.Salcin E, Barrett HH, Barber HB, Takeda S, Watanabe S, Takahashi T, Furenlid LR. Fisher information analysis of depth-of-interaction estimation in double-sided strip detectors. IEEE Trans. Nucl. Sci. 2014;61(3):1243–1251. doi: 10.1109/TNS.2014.2317454. [DOI] [PMC free article] [PubMed] [Google Scholar]