Abstract
Purpose
To investigate associations between serum thyroid stimulating hormone (TSH) receptor antibody (TRAb) levels and Graves' orbitopathy (GO) activity/severity in chronic-stage GO and compare the performance of two newly-developed TRAb assays (third-generation TSH-binding inhibition immunoglobulin [TBII] assay versus Mc4 thyroid-stimulating immunoglobulin [TSI] bioassay).
Methods
This study is a retrospective review of medical charts and blood tests from Korean GO patients who first visited the departments of ophthalmology and endocrinology, Yonsei University College of Medicine from January 2008 to December 2011, were diagnosed with GO and Graves' hyperthyroidism, and were followed up for ≥18 months. Third-generation M22-TBII and Mc4-TSI assays were performed in the chronic-inactive GO patients in whom euthyroidism status was restored. Patients' GO activity/severity clinical activity scores (CAS), and modified NOSPECS scores were examined for a correlation with TRAb assays.
Results
Fifty patients (mean age, 41.3 years; 41 females) were analyzed. The mean duration of Graves' hyperthyroidism symptom was 63 months (range, 18 to 401 months) and that of GO was 46 months (range, 18 to 240 months). All patients had been treated previously with anti-thyroid drugs for a median period of 52.3 months, and two patients underwent either radioiodine therapy or total thyroidectomy. Mean CAS and NOSPECS scores were 0.5 ± 0.9 (standard deviation) and 4.8 ± 3.1, respectively. Mean M22-TBII and Mc4-TSI values were 7.5 ± 10.2 IL/L and 325.9 ± 210.1 specimen-to-reference control ratio. TSI was significantly correlated with NOSPECS score (R = 0.479, p < 0.001); however, TBII was not associated with NOSPECS score (p = 0.097). Neither TSI nor TBII correlated with CAS (p > 0.05), because GO inflammatory activity subsided in the chronic stages of GO.
Conclusions
In chronic-inactive GO after euthyroid restoration, GO activity score did not associate with serum levels of TRAb or TBII. However, levels of the functional antibody Mc4-TSI did correlate with GO severity. Therefore, the TSI bioassay is a clinically relevant measure of disease severity even in chronic inactive GO.
Keywords: Biological assay, Chronic inactive Graves' orbitopathy, Mc4-TSI, Thyroid-stimulating immunoglobulin, Thyrotropin-binding inhibitory immunoglobulin
Graves' orbitopathy (GO) is a component of autoimmune Graves' hyperthyroidism, in which thyroid stimulating hormone (TSH) receptor antibody (TRAb) stimulates orbital and periorbital tissues [1]. The natural history of GO is not well understood. It is generally thought that GO has an inflammatory, active phase that subsides after 1 to 2 years. The average active inflammatory phase duration is approximately 18 months (range, 3 to 36 months) [2]. After the inflammation subsides, patients may suffer permanent structural changes around the eyes that require surgical repair.
Currently, there are two established assays to measure TRAb: the competitive TSH-binding inhibition immunoglobulin (TBII) assay and the functional thyroid-stimulating immunoglobulin (TSI) bioassay [3,4]. The TBII assay utilizes the ability of TRAb to inhibit the binding of radiolabeled TSH to TSH-receptors. The newly-developed, third-generation TBII assay measures the inhibition in the binding of a labelled monoclonal antibody clone M22 to the TSH-receptor rather than the traditional radiolabeled TSH-TSH receptor binding [4,5]. This assay enhanced the sensitivity and specificity of earlier assays using radiolabeled TSH [6,7,8]. The TSI bioassay measures cyclic adenosine monophosphate production after TRAb binds to the TSH-receptor, thus enabling identification of functional TRAb [9,10]. The development of the Mc4-CHO cell line simplified the cell culture protocols for the TRAb bioassays. The Mc4-CHO TSI bioassay has superior diagnostic potential for differentiating Graves' disease (GD) from painless thyroiditis [11].
Recently, several reports have focused on the relevance of TRAb, especially TSI, in untreated early stage GO [12]. Ponto et al. [13] reported that TSI levels correlated with disease activity (R = 0.89) and severity (R = 0.81) in untreated GO. Lytton et al. [12] showed a similar correlation between TSI and GO activity/severity. In previous studies we investigated whether serum TRAb in newly-diagnosed, untreated GO patients were predictive of the disease course beyond the first year after the initial diagnosis [14]. The results showed that patients with higher initial TRAb levels had a greater risk of severe disease outcomes. Likewise, we hypothesized that serum TRAb levels could provide important prognostic information to clinicians regarding early stage GO patients [14].
To the best of our knowledge, no prior reports of TRAb levels in chronic stage GO patients have been published. Chronic stage GO frequently imposes severe psychological, social, and economic burdens on patients because patients often experience substantial facial disfigurement including proptosis, puffy eyelids, and strabismus [1,15,16]. Although GO usually spontaneously resolves, patients often show heterogeneous clinical courses with complications such as restrictive strabismus and severe proptosis. We observed that patients who were current heavy smokers or of old age often exhibited high serum TSI levels even after prolonged anti-thyroid drug (ATD) treatment. TRAb level, TSI and TBII was usually high during the early disease period, and decreased gradually during ATD treatment. TBII typically decreased after a few months of ATD treatment, however, TSI levels generally remained high, especially in patients with complicated GO.
In the present study, we investigated the associations between serum TRAb levels and GO activity/severity in chronic-stage GO and compared the performance of two newly-developed TRAb assays: the third-generation TBII assay and the Mc4-TSI bioassay.
Materials and Methods
Patients
A retrospective review was conducted of the medical charts and the blood test results of all Korean GO patients who first visited both the departments of ophthalmology and endocrinology, Yonsei University College of Medicine from January 2008 to December 2011, were diagnosed with GO and Graves' hyperthyroidism and had been followed up for ≥18 months. The patients were seen by both the endocrinologist and ophthalmologist in a combined clinic system for thyroid diseases. Thus the diagnoses of GD and GO were frequently made simultaneously. Initial diagnosis of GD was made by the department of endocrinology based on peripheral thyroid function, TRAb values, thyroid ultrasound pattern and/or thyroid scan, and clinical features such as diffuse goiter or hyperthyroidism symptoms. Initial diagnosis of GO was made based on clinical ophthalmic examination by one examiner (JSY) using the following assessments: history taking, slit-lamp examination, visual acuity, intraocular pressure, exophthalmometry measured with a Hertel exophthalmometer, Hess screen test, binocular single vision test, and a computed tomography scan.
Among the 73 patients who visited both the department of ophthalmology and the department of endocrinology from January 2008 to December 2011, only patients with chronic inactive GO, restored euthyroidism, and simultaneous Mc4-TSI and third-generation M22-TBII assay measurements were selected for analysis. One oculoplasty specialist (JSY) determined whether each patient was in the chronic inactive-stage of GO, through a thorough examination of each patient's medial chart and photographs. In accordance with previous literature which reported that the average active inflammatory phase duration was approximately 18 months [2], we included only patients who had been diagnosed with GD or GO ≥18 months before enrollment. Patients with previous rehabilitative surgery such as orbital decompression and/or eyelid or muscle surgery were excluded.
Laboratory data from the time point when the patient was determined to be in the chronic inactive-stage of GO was used for analysis. Thyroid function test (TSH and free T4 levels), which were measured with the TRAb assays were also analyzed. Only euthyroid patients with normal free T4 and TSH were included in the study. Past medical history, smoking history, GD and GO duration, current GD treatment type, family history of thyroid disease, and steroid use history were all recorded.
Clinical Graves' orbitopathy evaluation
GO was diagnosed clinically using seven points of the clinical activity scores (CAS), as described by Mourits et al. [17] GO severity was assessed using the modified NOSPECS classification [18,19]. The modified NOSPECS score included the following: eyelid retraction (class 1), soft-tissue involvement (class 2), proptosis (class 3), extraocular muscle enlargement (class 4), and corneal (class 5) or optic nerve (class 6) involvement. The individual class scores ranged from 0 to 3 (except for classes 1 and 2, which score a maximum of 2 points combined), according to severity, and the sum of the scores constituted the modified NOSPECS score (range, 0 to 14 points) [20].
Thyroid stimulating hormone receptor autoantibody assays
The M22-TBII (third-generation TRAb) assay was performed using the automated Cobas electrochemiluminescence immunoassay system (Elecsys; Roche Diagnostics, Penzberg, Germany). The cut-off value for positive results was 1.75 IU/L. Mc4-TSI was measured with the Thyretain TSI reporter BioAssay (Diagnostic Hybrids, Athens, OH, USA) according to manufacturer instructions. Results were considered positive when the specimen-to-reference control ratio (SRR) was >140%. The antibody assays were performed at the same time as the physical examination and determination of CAS and NOPECS scores.
Statistical analysis
PASW Statistics ver. 18.0 (SPSS Inc., Chicago, IL, USA) was used to conduct data analyses. Spearman's correlation coefficient was calculated to compare MC4-TSI and M22-TBII to CAS scores, NOSPECS scores, and individual NOSPECS parameter scores. Regression analysis was used to evaluate the relationships between CAS, NOSPECS score, MC4-TSI level and M22-TBII levels, to reveal which of the laboratory parameters might correlate with disease activity or severity scores. Data were presented as means ± standard deviation, unless otherwise indicated.
Results
Fifty patients were included in the study. Their mean age was 41.3 ± 14.2 years, and 41 (82%) patients were women. The median GD duration was 62.7 months (range, 18 to 401 months) and the median GO duration was 46.3 months (range, 18 to 240 months).
Table 1 shows patient demographics and basic examination parameter data. The average exophthalmometery measurement was 18.1 ± 3.0 mm and the difference between right and left eyes was 0.7 ± 0.9 mm. The average TSH level was 2.27 ± 9.52 µIU/mL, free T4 was 1.57 ± 0.99 ng/mL, and T3 was 4.2 ± 18.8 ng/mL. The average NOSPECS score was 4.8 ± 3.1 points and the average CAS score was 0.5 ± 0.9 points. Among the participants, 33 patients had a CAS score of 0 points, 9 patients had a CAS score of 1 point, 6 patients had a CAS score of 2 points, and 2 patients had a CAS score of 3 points.
Table 1. Patient demographics.
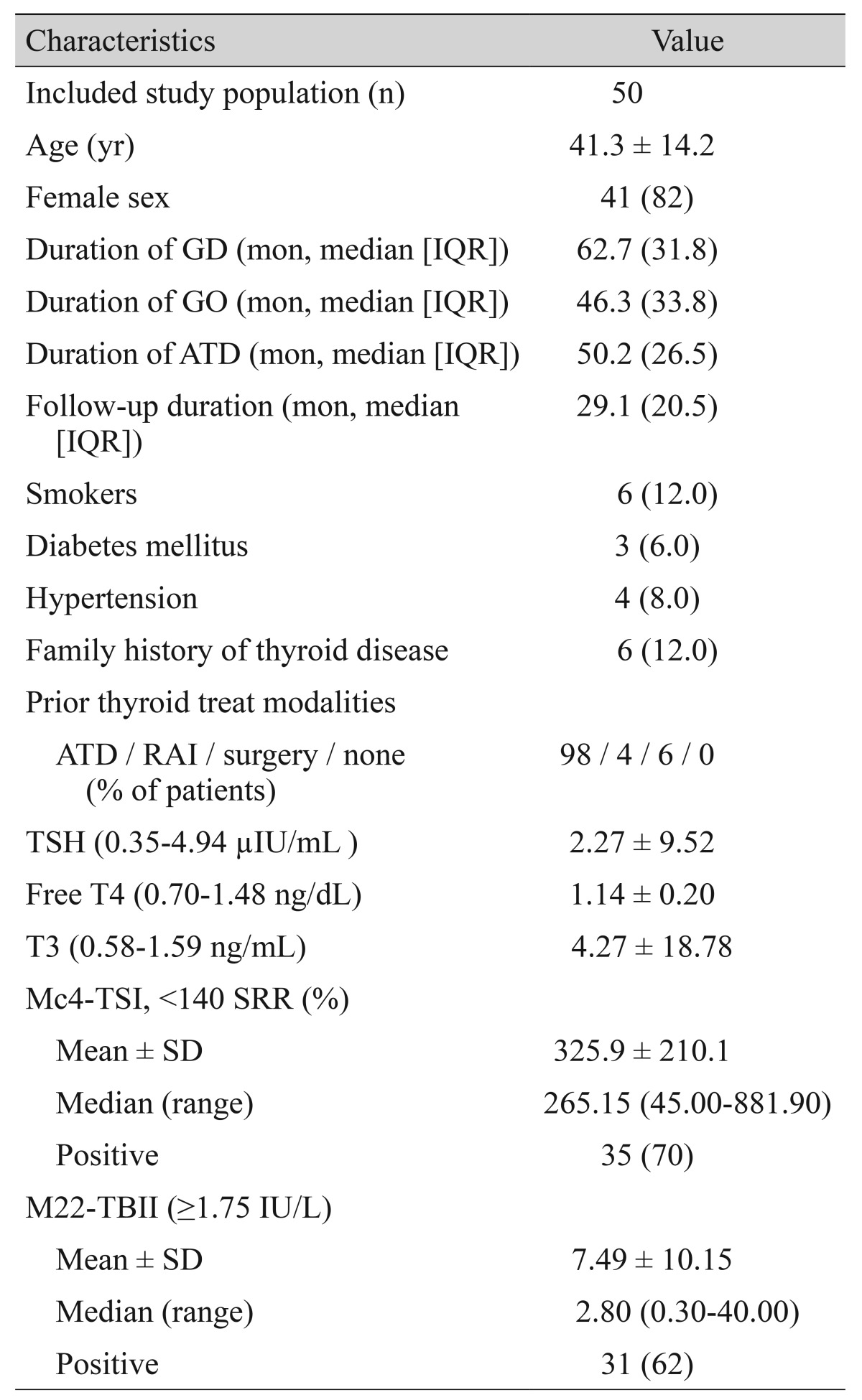
Values are presented as mean ± SD or number (%) unless otherwise indicated.
GD = Graves' disease; IQR = interquartile range; GO = Graves' orbitopathy; ATD = anti-thyroid drug; RAI = radioiodine therapy; TSH = thyroid-stimulating hormone; TSI = thyroid-stimulating immunoglobulin; SRR = specimen-to-reference ratio; SD = standard deviation; TBII = thyrotropin-binding inhibitory immunoglobulin.
The mean Mc4-TSI level was 325.9 ± 210.1 SRR%, and the mean level of M22-TBII was 7.5 ± 10.2 IU/L. Seventy per cent of patients showed positive Mc4-TSI values (>140 SRR%) and 62% of patients showed positive M22-TBII values (>1.75 IU/L). Fifty-four percent of patients showed positive results in both TRAb assays (Table 1).
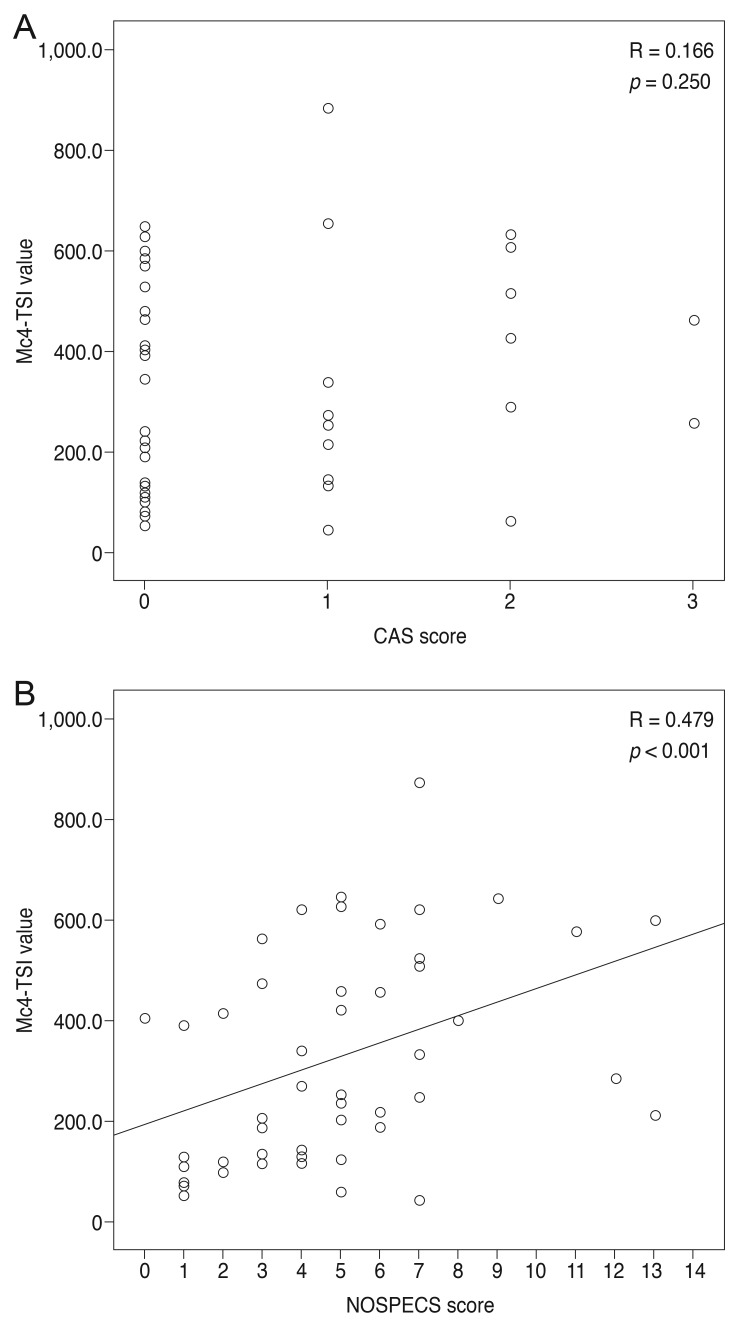
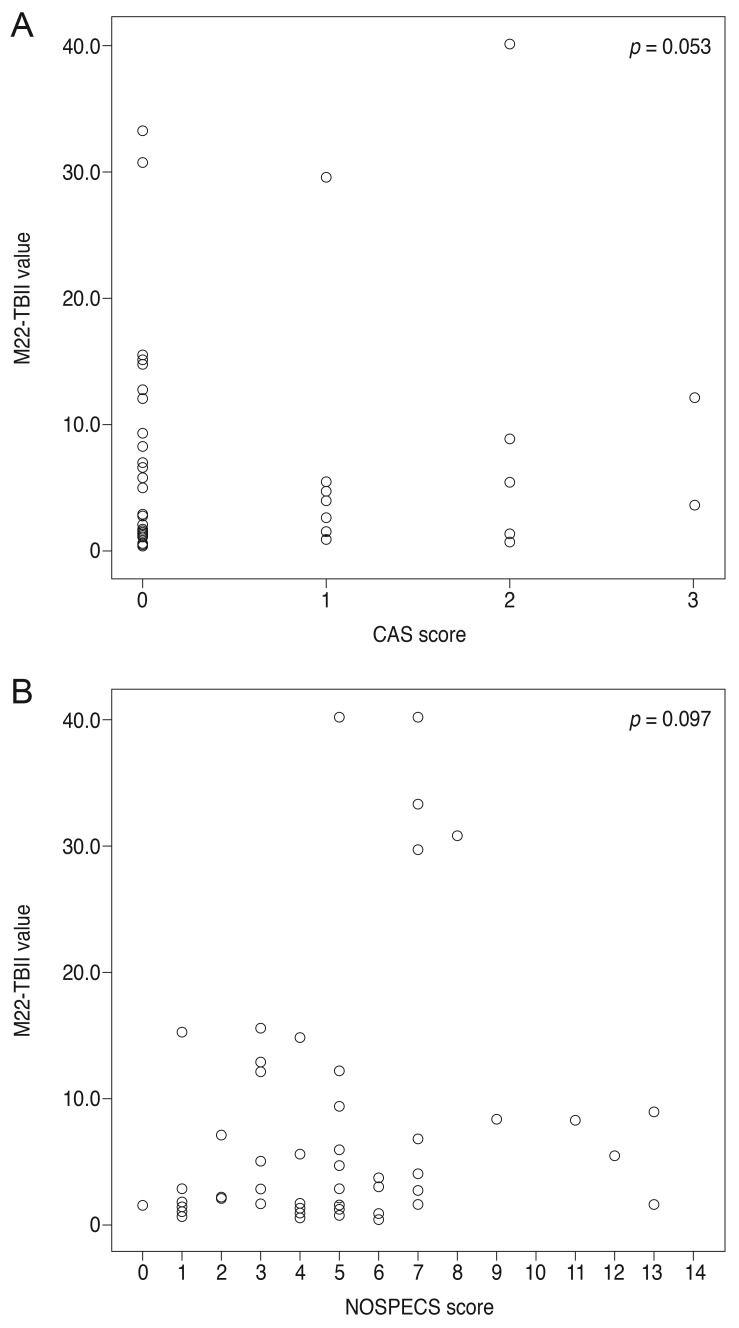
When MC4-TSI and M22-TBII results were compared with disease activity and severity scores, MC4-TSI levels correlated with NOSPECS score (R = 0.479, p < 0.001), but not with CAS (p = 0.250) (Fig. 1). On the other hand, M22-TBII results did not correlate with either CAS (p = 0.053) or NOSPECS scores (p = 0.097) (Fig. 2). MC4-TSI levels did correlate with two individual NOSPECS parameters: extraocular muscle involvement (R = 0.412, p = 0.003) and soft tissue involvement (R = 0.325, p = 0.021). However, M22-TBII did not correlate with any of the NOSPECS parameters. With respect to the exophthalmometry value, neither MC4-TSI nor M22-TBII were significantly associated with NOSPECS parameters (p > 0.05 for both comparisons, Pearson's correlation) (Table 2).
Fig. 1. Comparison of Mc4 thyroid-stimulating immunoglobulin (TSI) value with disease activity and severity scores. Comparison with (A) clinical activity score (CAS) score and with (B) NOSPECS score. In these graphs, Mc4-TSI value showed no definite correlation with CAS score (p = 0.250), however Mc4-TSI value was correlated with NOSEPCS score (p < 0.001).
Fig. 2. Comparison of M22 thyrotropin-binding inhibitory immunoglobulin (TBII) value with (A) clinical activity score (CAS) score and (B) NOSPECS score. Unlike Mc4 thyroid-stimulating immunoglobulin, M22-TBII was not correlated with any of disease activity (p = 0.053) or severity score (p = 0.097).
Table 2. Correlation of NOPECS* score parameters with M22-TBII and Mc4-TSI.
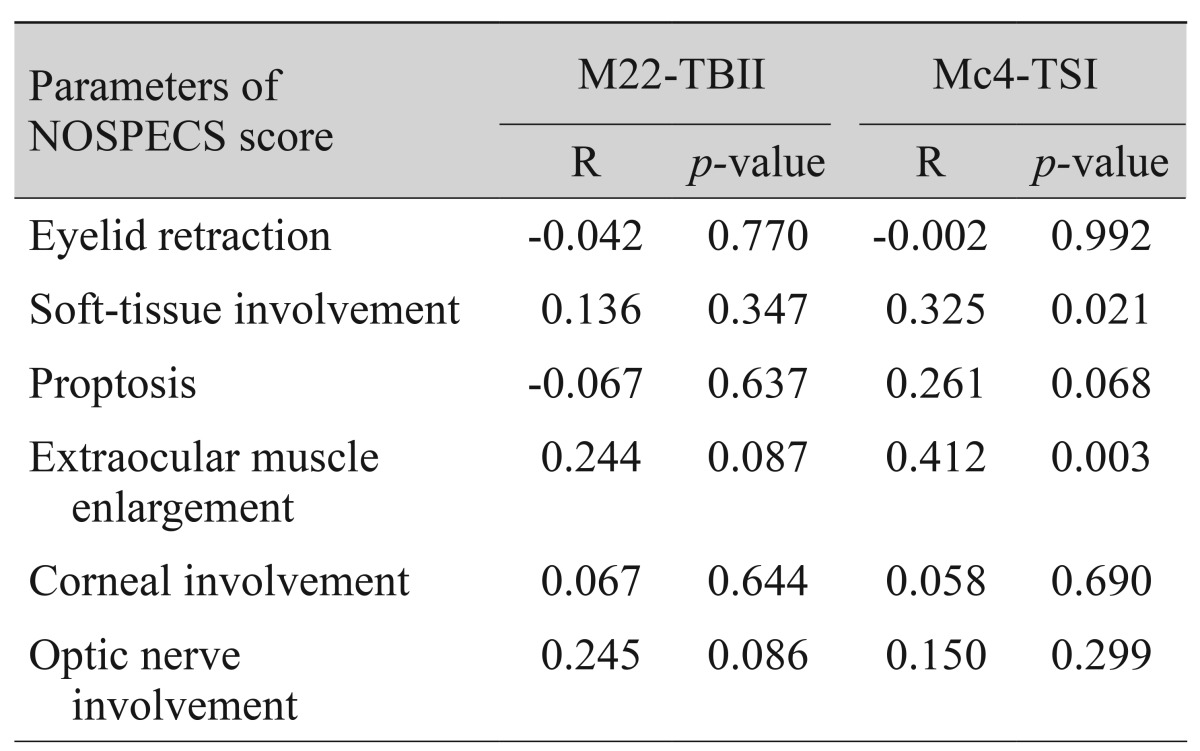
Spearman's correlation statistical method was used to compare each group.
TBII = thyrotropin-binding inhibitory immunoglobulin; TSI = thyroid-stimulating immunoglobulin.
*Endocrine ophthalmology grading scheme proposed in 1969 by SC Werner.
Univariate analysis of CAS values and NOSPECS scores with respect to both MC4-TSI and M22-TBII levels was conducted using a linear regression model, and only MC4-TSI levels were significantly predictive of NOSPECS score (R2 = 0.164, p = 0.007).
Discussion
TRAb measurement is widely accepted as a routine method for diagnosing and monitoring Graves' hyperthyroidism [18]. The TRAb assay technique has improved in diagnostic sensitivity and specificity for evaluating Graves' hyperthyroidism. In the ophthalmology field, TSI has provided greater performance power for assessing GO clinical manifestations [10]. In recent years, Mc4-TSI has been correlated with the activity and severity of untreated GO in the early disease period [12,13]. In the present study, we focused on the relevance of TRAb in chronic-stage GO. Interestingly, data from 50 patients with chronic inactive-stage GO showed that only Mc4-TSI, but not TBII, significantly correlated with the NOSPECS score. Neither TRAb levels correlated with CAS.
The GO activity score, CAS, did not associate with the two TRAb assays in chronic inactive-stage GO. One possible explanation might be that the majority of included subjects had low CAS scores. In this study, the mean CAS was 0.5 and 84% of patients had CAS values of 0 to 1 (42 out of 50 patients). These results were significantly different than a previous report that investigated the relationship between TRAb and disease activity in early untreated GO patients. Ponto et al. [13] emphasized a strikingly high correlation between Mc4-TSI levels and chemosis, which is a clinically important sign of the degree of acute inflammation.
Mc4-TSI levels associated with NOSPECS scores, especially the myopathy parameter score. The high TSI level in some patients with severe clinical courses, even after inflammatory activity had completely subsided, was an unexpected finding. Higher levels of TSI in the early stages of GO indicated that patients had a more active and severe disease, and therefore it follows that they would also have higher antibody levels and a more severe chronic inactive stage of the disease. It is well known that TRAb levels decrease with ATD use and/or over the natural disease course. Laurberg et al. [21] reported that the largest decrease in TRAb levels occurred within the first 6 months of ATD treatment. In the present study, the mean duration of Graves' hyperthyroidism was 63 months and the mean duration of GO was 46 months. All patients had been treated previously with ATD for a median of 52.3 months. However, TRAb levels from the Mc4-TSI bioassay, but not the third-generation TBII, was still clinically relevant in reflecting disease severity even in chronic inactive GO.
Since the pathogenesis of GO is not fully understood, it is difficult to explain why the TSI bioassay was superior over the TBII assay in reflecting GO severity scores, even in the chronic inactive disease stage. We recently investigated the clinical characteristics of GO patients who showed discrepancies between TSI and TBII levels [22]. A certain patient group presenting with relatively high TSI but low TBII were found to also have high CAS and NOSPECS scores; however, patients with hyperthyroidism were more likely to present with high TBII but low TSI levels. Another study that compared the performance of first- and third-generation TBII and TSI bioassays showed that the Mc4-TSI bioassay was superior to the two TBIIs in assessing active inflammation and muscle restriction due to GO [22]. Additionally, ethnic difference should be considered because Chng et al. [23] mentioned in their review article that the TSI seemed to reflect GO status better than TBII, especially in Asian patients.
In some cases, we found that the CAS did not accurately reflect the status of a patient's orbital inflammation, and high TSI levels were often associated with on-going autoimmune disease processes. However, patients with a high CAS sometimes did not have active inflammation, but showed congestive symptoms caused by interruption of venous outflow caused by elevated intraorbital pressure. Differentiating congestion and actual inflammation would help improve the medical management of GO. However, CAS is currently the only clear standard to define orbital inf lammation in GO. Generally, in a clinically setting, inflammatory activity during GO is evaluated by CAS, and anti-inflammatory treatment is initiated in patients with a high CAS. Our finding that TSI levels remained elevated in some patients with a severe clinical GO course, even after inflammatory activity determined by CAS had completely disappeared, was unexpected. When evaluating and treating GO patients, both TSI and clinical parameters should be equally considered. The results of this study indicate that rehabilitation surgery should not be delayed until negative conversion of TSI, because TSI is not a good marker for the clinically inactive disease state.
Although the present study has a limitation inherent to its retrospective nature and small sample size, it is worth noting that Mc4-TSI was high in patients with high NOSPECS score, especially those with high myopathy parameter subscores, even in the chronic inactive stage of GO. We believe that serial follow-up of TSI levels might provide important clinical information to better evaluate and properly treat GO.
Acknowledgements
This research was supported by the Basic Science Research Program through the National Research Foundation of Korea, which is funded by the Ministry of Education, Science, and Technology (2013R1A1A2007190).
Footnotes
This study was presented at the 109th annual meeting of the Korean Ophthalmological Society at Exco, Daegu, Korea, April 20, 2013.
Conflict of Interest: No potential conflict of interest relevant to this article was reported.
References
- 1.Bahn RS. Graves' ophthalmopathy. N Engl J Med. 2010;362:726–738. doi: 10.1056/NEJMra0905750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hales IB, Rundle FF. Ocular changes in Graves' disease: a long-term follow-up study. Q J Med. 1960;29:113–126. [PubMed] [Google Scholar]
- 3.Eckstein AK, Plicht M, Lax H, et al. Clinical results of anti-inflammatory therapy in Graves' ophthalmopathy and association with thyroidal autoantibodies. Clin Endocrinol (Oxf) 2004;61:612–618. doi: 10.1111/j.1365-2265.2004.02143.x. [DOI] [PubMed] [Google Scholar]
- 4.Sanders J, Evans M, Premawardhana LD, et al. Human monoclonal thyroid stimulating autoantibody. Lancet. 2003;362:126–128. doi: 10.1016/s0140-6736(03)13866-4. [DOI] [PubMed] [Google Scholar]
- 5.Sanders J, Jeffreys J, Depraetere H, et al. Characteristics of a human monoclonal autoantibody to the thyrotropin receptor: sequence structure and function. Thyroid. 2004;14:560–570. doi: 10.1089/1050725041692918. [DOI] [PubMed] [Google Scholar]
- 6.Massart C, Sapin R, Gibassier J, et al. Intermethod variability in TSH-receptor antibody measurement: implication for the diagnosis of Graves disease and for the follow-up of Graves ophthalmopathy. Clin Chem. 2009;55:183–186. doi: 10.1373/clinchem.2008.115162. [DOI] [PubMed] [Google Scholar]
- 7.Kamijo K, Ishikawa K, Tanaka M. Clinical evaluation of 3rd generation assay for thyrotropin receptor antibodies: the M22-biotin-based ELISA initiated by Smith. Endocr J. 2005;52:525–529. doi: 10.1507/endocrj.52.525. [DOI] [PubMed] [Google Scholar]
- 8.Smith BR, Bolton J, Young S, et al. A new assay for thyrotropin receptor autoantibodies. Thyroid. 2004;14:830–835. doi: 10.1089/thy.2004.14.830. [DOI] [PubMed] [Google Scholar]
- 9.Gerding MN, van der Meer JW, Broenink M, et al. Association of thyrotrophin receptor antibodies with the clinical features of Graves' ophthalmopathy. Clin Endocrinol (Oxf) 2000;52:267–271. doi: 10.1046/j.1365-2265.2000.00959.x. [DOI] [PubMed] [Google Scholar]
- 10.Dragan LR, Seiff SR, Lee DC. Longitudinal correlation of thyroid-stimulating immunoglobulin with clinical activity of disease in thyroid-associated orbitopathy. Ophthal Plast Reconstr Surg. 2006;22:13–19. doi: 10.1097/01.iop.0000192649.23508.f7. [DOI] [PubMed] [Google Scholar]
- 11.Lytton SD, Kahaly GJ. Bioassays for TSH-receptor autoantibodies: an update. Autoimmun Rev. 2010;10:116–122. doi: 10.1016/j.autrev.2010.08.018. [DOI] [PubMed] [Google Scholar]
- 12.Lytton SD, Ponto KA, Kanitz M, et al. A novel thyroid stimulating immunoglobulin bioassay is a functional indicator of activity and severity of Graves' orbitopathy. J Clin Endocrinol Metab. 2010;95:2123–2131. doi: 10.1210/jc.2009-2470. [DOI] [PubMed] [Google Scholar]
- 13.Ponto KA, Kanitz M, Olivo PD, et al. Clinical relevance of thyroid-stimulating immunoglobulins in graves' ophthalmopathy. Ophthalmology. 2011;118:2279–2285. doi: 10.1016/j.ophtha.2011.03.030. [DOI] [PubMed] [Google Scholar]
- 14.Jang SY, Shin DY, Lee EJ, et al. Relevance of TSH-receptor antibody levels in predicting disease course in Graves' orbitopathy: comparison of the third-generation TBII assay and Mc4-TSI bioassay. Eye (Lond) 2013;27:964–971. doi: 10.1038/eye.2013.120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Finamor FE, Martins JR, Nakanami D, et al. Pentoxifylline (PTX): an alternative treatment in Graves' ophthalmopathy (inactive phase): assessment by a disease specific quality of life questionnaire and by exophthalmometry in a prospective randomized trial. Eur J Ophthalmol. 2004;14:277–283. [PubMed] [Google Scholar]
- 16.Khoo TK, Bahn RS. Pathogenesis of Graves' ophthalmopathy: the role of autoantibodies. Thyroid. 2007;17:1013–1018. doi: 10.1089/thy.2007.0185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Mourits MP, Prummel MF, Wiersinga WM, Koornneef L. Clinical activity score as a guide in the management of patients with Graves' ophthalmopathy. Clin Endocrinol (Oxf) 1997;47:9–14. doi: 10.1046/j.1365-2265.1997.2331047.x. [DOI] [PubMed] [Google Scholar]
- 18.Eckstein AK, Plicht M, Lax H, et al. Thyrotropin receptor autoantibodies are independent risk factors for Graves' ophthalmopathy and help to predict severity and outcome of the disease. J Clin Endocrinol Metab. 2006;91:3464–3470. doi: 10.1210/jc.2005-2813. [DOI] [PubMed] [Google Scholar]
- 19.Eckstein AK, Lax H, Losch C, et al. Patients with severe Graves' ophthalmopathy have a higher risk of relapsing hyperthyroidism and are unlikely to remain in remission. Clin Endocrinol (Oxf) 2007;67:607–612. doi: 10.1111/j.1365-2265.2007.02933.x. [DOI] [PubMed] [Google Scholar]
- 20.Werner SC. Modification of the classification of the eye changes of Graves' disease. Am J Ophthalmol. 1977;83:725–727. doi: 10.1016/0002-9394(77)90140-4. [DOI] [PubMed] [Google Scholar]
- 21.Laurberg P, Wallin G, Tallstedt L, et al. TSH-receptor autoimmunity in Graves' disease after therapy with anti-thyroid drugs, surgery, or radioiodine: a 5-year prospective randomized study. Eur J Endocrinol. 2008;158:69–75. doi: 10.1530/EJE-07-0450. [DOI] [PubMed] [Google Scholar]
- 22.Jang SY, Shin DY, Lee EJ, Yoon JS. Clinical characteristics of Graves' orbitopathy in patients showing discrepancy between levels from TBII assays and TSI bioassay. Clin Endocrinol (Oxf) 2014;80:591–597. doi: 10.1111/cen.12318. [DOI] [PubMed] [Google Scholar]
- 23.Chng CL, Seah LL, Khoo DH. Ethnic differences in the clinical presentation of Graves' ophthalmopathy. Best Pract Res Clin Endocrinol Metab. 2012;26:249–258. doi: 10.1016/j.beem.2011.10.004. [DOI] [PubMed] [Google Scholar]