Abstract
Mutations in APP, PSEN1 and PSEN2 as the genetic causes of familial Alzheimer's disease (FAD) have been found in various ethnic populations. A substantial number of FAD pedigrees with mutations have been reported in the Japanese population; however, it remains unclear whether the genetic and clinical features of FAD in the Japanese population differ from those in other populations. To address this issue, we conducted a systematic review and meta-analysis of Japanese FAD and frontotemporal dementia with parkinsonism linked to chromosome 17 (FTDP-17) by literature search. Using this analysis, we identified 39 different PSEN1 mutations in 140 patients, 5 APP mutations in 35 patients and 16 MAPT mutations in 84 patients. There was no PSEN2 mutation among Japanese patients. The age at onset in Japanese FAD patients with PSEN1 mutations was significantly younger than that in patients with APP mutations. Kaplan–Meier analysis revealed that patients with MAPT mutations showed a shorter survival than patients with PSEN1 or APP mutations. Patients with mutations in different genes exhibit characteristic clinical presentations, suggesting that mutations in causative genes may modify the clinical presentations. By collecting and cataloging genetic and clinical information on Japanese FAD and FTDP-17, we developed an original database designated as Japanese Familial Alzheimer's Disease Database, which is accessible at http://alzdb.bri.niigata-u.ac.jp/.
Mutations in APP, PSEN1 and PSEN2 as the genetic causes of familial Alzheimer's disease (FAD) have been found in various ethnic populations.1, 2 In addition, patients with mutations in MAPT associated with frontotemporal dementia with parkinsonism linked to chromosome 17 (FTDP-17) have been shown to exhibit Alzheimer's disease (AD)-like phenotypes.3, 4 Although a substantial number of FAD pedigrees have been reported in Japan, it is not yet clear whether the genetic and clinical features of FAD in the Japanese population differ from those in other ethnic populations. To characterize the genetic and clinical features of Japanese FAD and FTDP-17, we here performed a systematic review and meta-analysis, and developed an original database of Japanese FAD and FTDP-17.
To comprehensively review the previously reported Japanese FAD and FTDP-17 cases, we performed a systematic search for publications in PubMed and Ichushi, a bibliographic database of medical literature in Japanese. The terms ‘familial Alzheimer', ‘familial AD', ‘FTDP-17', ‘presenilin', ‘PSEN1', ‘PSEN2', ‘APP' and ‘MAPT' were used to search in PubMed, and the equivalent terms in Japanese were used to search in Ichushi. From the literature searches we found 60 English and 29 Japanese articles and/or abstracts that reported on Japanese FAD and FTDP-17 pedigrees bearing the causative mutations (Supplementary Table 1). Using the information obtained by the systematic literature review, we developed an original database for Japanese FAD and FTDP-17 designated as Japanese Familial Alzheimer's Disease database (JFADdb). In the database, each of the mutations in APP, PSEN1/2, MAPT and GRN was described in accordance with the reference sequences.5 Information on age at onset, clinical manifestations, age at death and APOE genotype were included in the database (Supplementary Figure 1).
We identified 39 different PSEN1 mutations in 140 patients, 5 APP mutations in 35 patients and 16 MAPT mutations in 84 patients (Table 1). Among them, 10 PSEN1 mutations, 5 APP mutations and 11 MAPT mutations were not included in the well-known Alzheimer Disease and Frontotemporal Dementia Mutation database (http://www.molgen.ua.ac.be/ADMutations/).6 No PSEN2 mutation has been found in Japanese FAD. The frequency of mutated genes in FAD patients in the Japanese population was not significantly different from those in other populations (χ2, P=0.99).6 Most FAD pedigrees show autosomal dominant inheritance; however, an APP ΔE693 mutation was responsible for a recessively inherited FAD.7 Sporadic occurrences of mutations were observed: five patients with PSEN1 mutations, one patient with APP mutation and two patients with MAPT mutations. In our analysis, the majority of mutations (73%) was observed in a single small pedigree. Considering that novel mutations in FAD tend to be reported rapidly, note that there may be publication bias in the frequency of mutations in the database. Although rare, there were two GRN mutations in patients with primary progressive aphasia and frontotemporal lobar degeneration (FTLD).8 Because the number of GRN mutations was too small, the patients with GRN mutations were excluded from further meta-analysis.
Table 1. Summary of genetic features of Japanese FAD and FTDP-17.
| Disease | Genes | Number of mutations | Number of pedigrees | Number of patients |
|---|---|---|---|---|
| FAD | PSEN1 | 39 | 40 | 140 |
| PSEN2 | 0 | 0 | 0 | |
| APP | 5a | 13 | 35 | |
| FTDP-17 | MAPT | 16 | 29 | 84 |
| GRN | 2 | 2 | 2 |
Abbreviations: FAD, familial Alzheimer's disease; FTDP-17, frontotemporal dementia with parkinsonism linked to chromosome 17.
APP duplication was included.
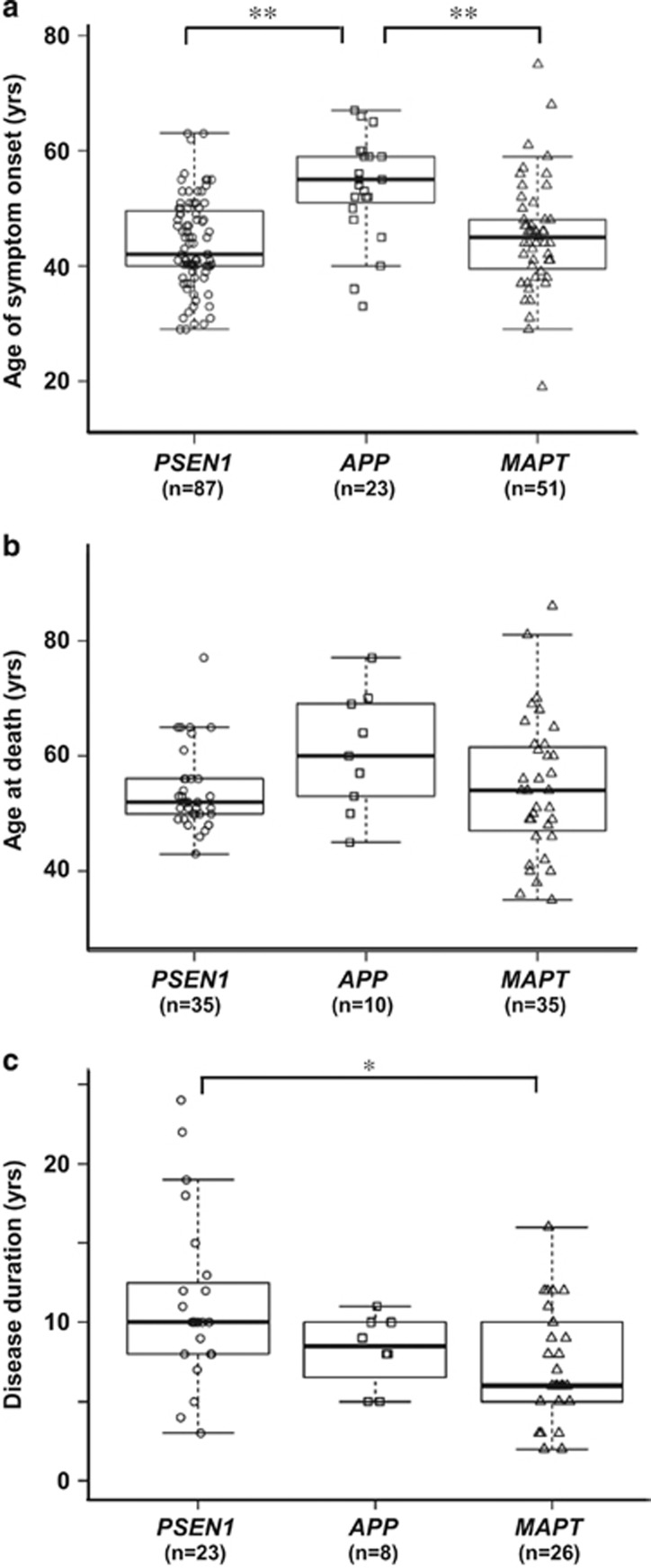
The ages at onset were 44±8 years (mean±s.d.) in patients with PSEN1 mutations (n=87), 54±9 years in APP mutations (n=23) and 45±10 years in MAPT mutations (n=51). These ages at onset of FAD in our analysis are consistent with those reported in other ethnic populations.6, 9 The age at onset in patients with APP mutations was significantly older than those in patients with PSEN1 or MAPT mutations (Figure 1a). The clinical phenotypes of patients with MAPT mutations were classified into three subgroups: FTLD,10 AD-like3, 4 and progressive supranuclear palsy (PSP) phenotypes.8 The age at onset in patients with the FTLD or PSP phenotype was significantly younger than that with the AD-like phenotype (Supplementary Figure 2). APOE genotypes did not significantly modify the age at onset in patients with causative mutations (Supplementary Figure 3). There was no significant difference in age at death among the patients with mutations in the three genes (Figure 1b). The disease duration from age at onset to death in patients with MAPT mutations was significantly shorter than that with PSEN1 mutations (Figure 1c). The survival of patients after the onset was analyzed by Kaplan–Meier estimation, which revealed that patients with MAPT mutations showed a shorter survival than patients with PSEN1 or APP mutations (Supplementary Figure 4).
Figure 1.
Age at onset and death, and disease duration in Japanese FAD and FTDP-17 patients. (a) Age at onset for patients grouped on the basis of PSEN1, APP and MAPT mutations. The horizontal line in the box indicates the median, the lower and upper boundaries of the box represent the lower and upper quartile boundaries, respectively, and whiskers are 1.5 times the interquartile range. Patients with PSEN1 and MAPT mutation showed significantly younger age at onset than patients with APP mutations (**P<0.01, ANOVA with post hoc Tukey's test). (b) Age at death of three groups with gene mutations. There was no significant difference in age at death among the groups. (c) Disease duration was defined as the period from age at onset to death. The disease courses of patients with MAPT mutations (7±4 years, mean±s.d.) were significantly shorter than those with PSEN1 mutations (11±5) (*P<0.05, ANOVA with post hoc Tukey's test). ANOVA, analysis of variance; FAD, familial Alzheimer's disease; FTDP-17, frontotemporal dementia with parkinsonism linked to chromosome 17.
The clinical diagnosis of AD before the genetic testing was performed in 96% of patients with PSEN1 mutations and 97% of patients with APP mutations (Supplementary Table 2). Notably, only 57% of patients with MAPT mutations were clinically diagnosed as having FTLD; 19% and 12% of patients with MAPT mutations were clinically diagnosed as having AD and PSP, respectively (Supplementary Table 2). This finding suggests that mutational screening of clinically diagnosed FAD patients should not only include APP and PSEN1/2 mutations but also include MAPT mutations.
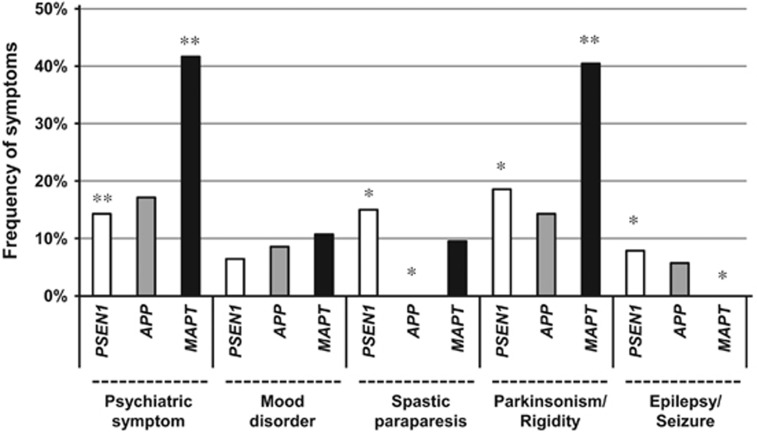
We next analyzed the frequency of each of the clinical manifestations including psychiatric symptoms, mood disorders, spastic paraparesis, parkinsonism and epilepsy/seizure (Figure 2). As expected, the frequencies of psychiatric symptoms and parkinsonism were significantly higher in patients with MAPT mutations. Spastic paraparesis, which is a characteristic symptom of ‘variant AD with cotton-wool plaque pathology'11, 12 was observed in 15% of patients with PSEN1 mutations, whereas none of the patients with APP mutations exhibited spastic paraplegia. Epilepsy/seizure was described in 8% of patients with PSEN1 and 6% of patients with APP mutations, whereas none of the patients with MAPT mutations exhibited epilepsy/seizure. Previous studies showed that the frequency of seizure was relatively high in patients with early onset of AD,13, 14 and low in patients with MAPT mutations.15 These findings suggest that epilepsy/seizure is closely associated with amyloid pathology, and that tauopathy alone may not be sufficient to cause epilepsy. Taken together, mutations in causative genes may modify the clinical presentations in patients with familial dementia.
Figure 2.
Frequency of each clinical manifestation in patients with Japanese FAD and FTDP-17. We investigated the presence or absence of psychiatric symptoms, mood disorders, spastic paraplegia, parkinsonism and epilepsy/seizure by careful reading of the original papers. We determined the frequency of each of the clinical manifestations by counting the number of patients for whom the presence of the manifestation was described in literature. In case there was no description of the manifestation, the patient was not counted as manifesting the manifestation. The observed frequencies of causative gene mutation were significantly different from the expected frequencies determined by residual analyses for χ2 statistical analysis (*P<0.05, **P<0.01). FAD, familial Alzheimer's disease; FTDP-17, frontotemporal dementia with parkinsonism linked to chromosome 17.
In summary, we have comprehensively collected, cataloged and systematically meta-analyzed the data from currently available data on Japanese FAD and FTDP-17. We made all the results publicly available on the online database ‘JFADdb'. The database may provide information useful for estimating the age at onset and the natural course of disease in future preventive or therapeutic trials of Japanese FAD.
Acknowledgments
This work is supported in part by Grants-in-Aid for scientific research from Japan Society of Promotion of Science, Japan (26870209 to KK, 23591234 and 20372469 to TI) and Grant-in-Aid from the Ministry of Health, Labour and Welfare of Japan. We are grateful to Dr Hasegawa and Ms Fukaumi for their technical assistance.
The authors declare no conflict of interest.
Footnotes
Supplementary Information accompanies the paper on Journal of Human Genetics website (http://www.nature.com/jhg)
Supplementary Material
References
- Karch C. M., Cruchaga C., Goate A. M. Alzheimer's disease genetics: from the bench to the clinic. Neuron. 2014;83:11–26. doi: 10.1016/j.neuron.2014.05.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guerreiro R. J., Gustafson D. R., Hardy J. The genetic architecture of Alzheimer's disease: beyond APP, PSENs and APOE. Neurobiol. Aging. 2012;33:437–456. doi: 10.1016/j.neurobiolaging.2010.03.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rademakers R., Dermaut B., Peeters K., Cruts M., Heutink P., Goate A., et al. Tau (MAPT) mutation Arg406Trp presenting clinically with Alzheimer disease does not share a common founder in Western Europe. Hum. Mutat. 2003;22:409–411. doi: 10.1002/humu.10269. [DOI] [PubMed] [Google Scholar]
- Ikeuchi T., Kaneko H., Miyashita A., Nozaki H., Kasuga K., Tsukie T., et al. Mutational analysis in early-onset familial dementia in the Japanese population. The role of PSEN1 and MAPT R406W mutations. Dement. Geriatr. Cogn. Disord. 2008;26:43–49. doi: 10.1159/000141483. [DOI] [PubMed] [Google Scholar]
- Pruitt K. D., Brown G. R., Hiatt S. M., Thibaud-Nissen F., Astashyn A., Ermolaeva O., et al. RefSeq: an update on mammalian reference sequences. Nucleic Acids Res. 2014;42:D756–D763. doi: 10.1093/nar/gkt1114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cruts M., Theuns J., Van Broeckhoven C. Locus-specific mutation databases for neurodegenerative brain diseases. Hum. Mutat. 2012;33:1340–1344. doi: 10.1002/humu.22117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tomiyama T., Nagata T., Shimada H., Teraoka R., Fukushima A., Kanemitsu H., et al. A new amyloid beta variant favoring oligomerization in Alzheimer's-type dementia. Ann. Neurol. 2008;63:377–387. doi: 10.1002/ana.21321. [DOI] [PubMed] [Google Scholar]
- Ogaki K., Li Y., Takanashi M., Ishikawa K., Kobayashi T., Nonaka T., et al. Analyses of the MAPT, PGRN, and C9orf72 mutations in Japanese patients with FTLD, PSP, and CBS. Parkinsonism Relat. Disord. 2013;19:15–20. doi: 10.1016/j.parkreldis.2012.06.019. [DOI] [PubMed] [Google Scholar]
- Ryman D. C., Acosta-Baena N., Aisen P. S., Bird T., Danek A., Fox N. C., et al. Symptom onset in autosomal dominant Alzheimer disease: a systematic review and meta-analysis. Neurology. 2014;83:253–260. doi: 10.1212/WNL.0000000000000596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Foster N. L., Wilhelmsen K., Sima A. A. F., Jones M. Z., D'Amato C. J., et al. Frontotemporal dementia and parkinsonism linked to chromosome 17: a consensus conference. Ann. Neurol. 1997;41:706–715. doi: 10.1002/ana.410410606. [DOI] [PubMed] [Google Scholar]
- Larner A. J., Doran M. Clinical phenotypic heterogeneity of Alzheimer's disease associated with mutations of the presenilin-1 gene. J. Neurol. 2006;253:139–158. doi: 10.1007/s00415-005-0019-5. [DOI] [PubMed] [Google Scholar]
- Tabira T., Chui D. H., Nakayama H., Kuroda S., Shibuya M. Alzheimer's disease with spastic paresis and cotton wool type plaques. J. Neurosci. Res. 2002;70:367–372. doi: 10.1002/jnr.10392. [DOI] [PubMed] [Google Scholar]
- Noebels, J. A perfect storm: converging paths of epilepsy and Alzheimer's dementia intersect in the hippocampal formation. Epilepsia. 2011;52:39–46. doi: 10.1111/j.1528-1167.2010.02909.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vossel K. A., Beagle A. J., Rabinovici G. D., Shu H., Lee S. E., Naasan G., et al. Seizures and epileptiform activity in the early stages of Alzheimer disease. JAMA Neurol. 2013;70:1158–1166. doi: 10.1001/jamaneurol.2013.136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sperfeld A. D., Collatz M. B., Baier H., Palmbach M., Storch A., Schwarz J., et al. FTDP-17: an early-onset phenotype with parkinsonism and epileptic seizures caused by a novel mutation. Ann. Neurol. 1999;46:708–715. doi: 10.1002/1531-8249(199911)46:5<708::aid-ana5>3.0.co;2-k. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.