Abstract
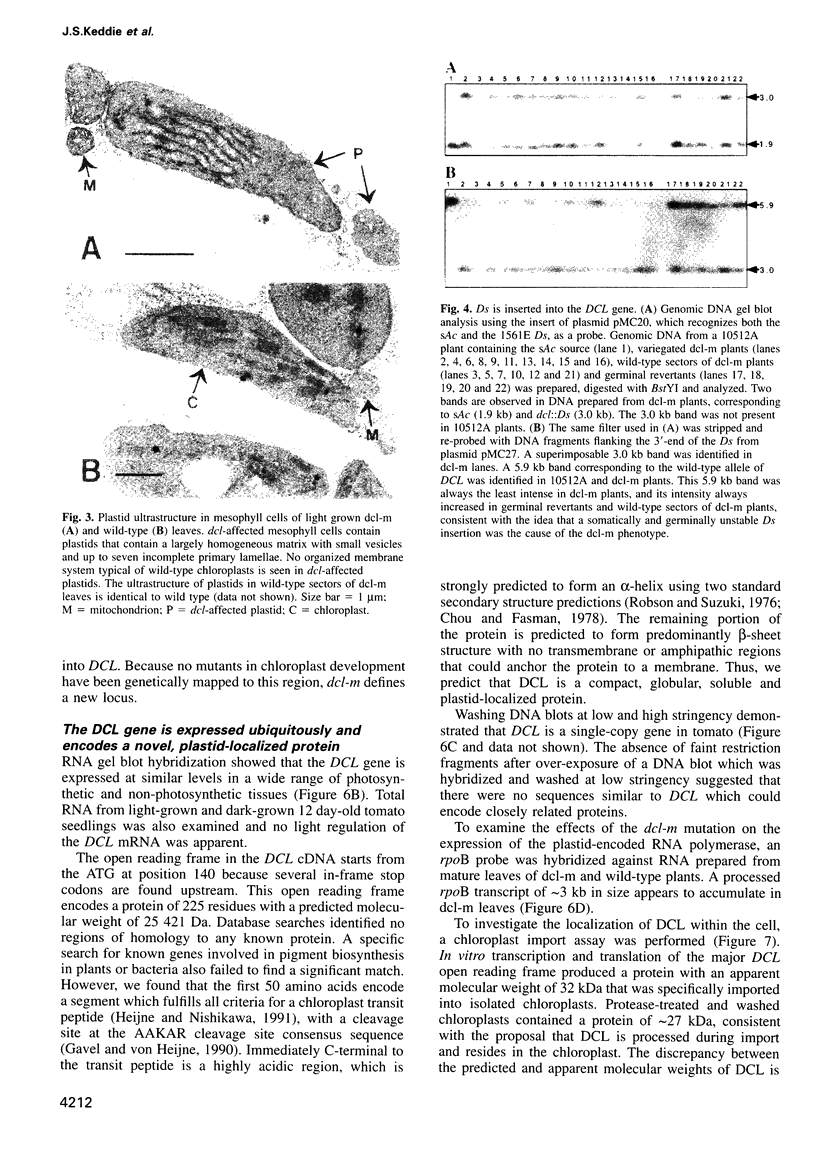
The defective chloroplasts and leaves-mutable (dcl-m) mutation of tomato was identified in a Ds mutagenesis screen. This unstable mutation affects both chloroplast development and palisade cell morphogenesis in leaves. Mutant plants are clonally variegated as a result of somatic excision of Ds and have albino leaves with green sectors. Leaf midribs and stems are light green with sectors of dark green tissue but fruit and petals are wild-type in appearance. Within dark green sectors of dcl-m leaves, palisade cells are normal, whereas in albino areas of dcl-m leaves, palisade cells do not expand to become their characteristic columnar shape. The development of chloroplasts from proplastids in albino areas is apparently blocked at an early stage. DCL was cloned using Ds as a tag and encodes a novel protein of approximately 25 kDa, containing a chloroplast transit peptide and an acidic alpha-helical region. DCL protein was imported into chloroplasts in vitro and processed to a mature form. Because of the ubiquitous expression of DCL and the proplastid-like appearance of dcl-affected plastids, the DCL protein may regulate a basic and universal function of the plastid. The novel dcl-m phenotype suggests that chloroplast development is required for correct palisade cell morphogenesis during leaf development.
Full text
PDF







Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Carroll B. J., Klimyuk V. I., Thomas C. M., Bishop G. J., Harrison K., Scofield S. R., Jones J. D. Germinal transpositions of the maize element Dissociation from T-DNA loci in tomato. Genetics. 1995 Jan;139(1):407–420. doi: 10.1093/genetics/139.1.407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chory J., Peto C., Feinbaum R., Pratt L., Ausubel F. Arabidopsis thaliana mutant that develops as a light-grown plant in the absence of light. Cell. 1989 Sep 8;58(5):991–999. doi: 10.1016/0092-8674(89)90950-1. [DOI] [PubMed] [Google Scholar]
- Chou P. Y., Fasman G. D. Empirical predictions of protein conformation. Annu Rev Biochem. 1978;47:251–276. doi: 10.1146/annurev.bi.47.070178.001343. [DOI] [PubMed] [Google Scholar]
- Deng X. W., Matsui M., Wei N., Wagner D., Chu A. M., Feldmann K. A., Quail P. H. COP1, an Arabidopsis regulatory gene, encodes a protein with both a zinc-binding motif and a G beta homologous domain. Cell. 1992 Nov 27;71(5):791–801. doi: 10.1016/0092-8674(92)90555-q. [DOI] [PubMed] [Google Scholar]
- Gavel Y., von Heijne G. A conserved cleavage-site motif in chloroplast transit peptides. FEBS Lett. 1990 Feb 26;261(2):455–458. doi: 10.1016/0014-5793(90)80614-o. [DOI] [PubMed] [Google Scholar]
- Gruissem W. Chloroplast gene expression: how plants turn their plastids on. Cell. 1989 Jan 27;56(2):161–170. doi: 10.1016/0092-8674(89)90889-1. [DOI] [PubMed] [Google Scholar]
- Han C. D., Coe E. H., Jr, Martienssen R. A. Molecular cloning and characterization of iojap (ij), a pattern striping gene of maize. EMBO J. 1992 Nov;11(11):4037–4046. doi: 10.1002/j.1460-2075.1992.tb05497.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hess W. R., Prombona A., Fieder B., Subramanian A. R., Börner T. Chloroplast rps15 and the rpoB/C1/C2 gene cluster are strongly transcribed in ribosome-deficient plastids: evidence for a functioning non-chloroplast-encoded RNA polymerase. EMBO J. 1993 Feb;12(2):563–571. doi: 10.1002/j.1460-2075.1993.tb05688.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hudson A., Carpenter R., Doyle S., Coen E. S. Olive: a key gene required for chlorophyll biosynthesis in Antirrhinum majus. EMBO J. 1993 Oct;12(10):3711–3719. doi: 10.1002/j.1460-2075.1993.tb06048.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones J. D., Carland F. M., Maliga P., Dooner H. K. Visual detection of transposition of the maize element activator (ac) in tobacco seedlings. Science. 1989 Apr 14;244(4901):204–207. doi: 10.1126/science.244.4901.204. [DOI] [PubMed] [Google Scholar]
- Luna E. J., Hitt A. L. Cytoskeleton--plasma membrane interactions. Science. 1992 Nov 6;258(5084):955–964. doi: 10.1126/science.1439807. [DOI] [PubMed] [Google Scholar]
- Manly K. F. A Macintosh program for storage and analysis of experimental genetic mapping data. Mamm Genome. 1993;4(6):303–313. doi: 10.1007/BF00357089. [DOI] [PubMed] [Google Scholar]
- Martienssen R. A., Barkan A., Freeling M., Taylor W. C. Molecular cloning of a maize gene involved in photosynthetic membrane organization that is regulated by Robertson's Mutator. EMBO J. 1989 Jun;8(6):1633–1639. doi: 10.1002/j.1460-2075.1989.tb03553.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martínez-Zapater J. M., Gil P., Capel J., Somerville C. R. Mutations at the Arabidopsis CHM locus promote rearrangements of the mitochondrial genome. Plant Cell. 1992 Aug;4(8):889–899. doi: 10.1105/tpc.4.8.889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newton K. J., Coe E. H. Mitochondrial DNA changes in abnormal growth (nonchromosomal stripe) mutants of maize. Proc Natl Acad Sci U S A. 1986 Oct;83(19):7363–7366. doi: 10.1073/pnas.83.19.7363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poethig S. Genetic mosaics and cell lineage analysis in plants. Trends Genet. 1989 Aug;5(8):273–277. doi: 10.1016/0168-9525(89)90101-7. [DOI] [PubMed] [Google Scholar]
- Pyke K. A., Leech R. M. Chloroplast Division and Expansion Is Radically Altered by Nuclear Mutations in Arabidopsis thaliana. Plant Physiol. 1992 Jul;99(3):1005–1008. doi: 10.1104/pp.99.3.1005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reiter R. S., Coomber S. A., Bourett T. M., Bartley G. E., Scolnik P. A. Control of leaf and chloroplast development by the Arabidopsis gene pale cress. Plant Cell. 1994 Sep;6(9):1253–1264. doi: 10.1105/tpc.6.9.1253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robson B., Suzuki E. Conformational properties of amino acid residues in globular proteins. J Mol Biol. 1976 Nov 5;107(3):327–356. doi: 10.1016/s0022-2836(76)80008-3. [DOI] [PubMed] [Google Scholar]
- Saedler H., Nevers P. Transposition in plants: a molecular model. EMBO J. 1985 Mar;4(3):585–590. doi: 10.1002/j.1460-2075.1985.tb03670.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Susek R. E., Ausubel F. M., Chory J. Signal transduction mutants of Arabidopsis uncouple nuclear CAB and RBCS gene expression from chloroplast development. Cell. 1993 Sep 10;74(5):787–799. doi: 10.1016/0092-8674(93)90459-4. [DOI] [PubMed] [Google Scholar]
- Tanksley S. D., Ganal M. W., Prince J. P., de Vicente M. C., Bonierbale M. W., Broun P., Fulton T. M., Giovannoni J. J., Grandillo S., Martin G. B. High density molecular linkage maps of the tomato and potato genomes. Genetics. 1992 Dec;132(4):1141–1160. doi: 10.1093/genetics/132.4.1141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomas C. M., Jones D. A., English J. J., Carroll B. J., Bennetzen J. L., Harrison K., Burbidge A., Bishop G. J., Jones J. D. Analysis of the chromosomal distribution of transposon-carrying T-DNAs in tomato using the inverse polymerase chain reaction. Mol Gen Genet. 1994 Mar;242(5):573–585. doi: 10.1007/BF00285281. [DOI] [PubMed] [Google Scholar]
- Tonkyn J. C., Deng X. W., Gruissem W. Regulation of Plastid Gene Expression during Photooxidative Stress. Plant Physiol. 1992 Aug;99(4):1406–1415. doi: 10.1104/pp.99.4.1406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Voelker R., Barkan A. Two nuclear mutations disrupt distinct pathways for targeting proteins to the chloroplast thylakoid. EMBO J. 1995 Aug 15;14(16):3905–3914. doi: 10.1002/j.1460-2075.1995.tb00062.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walbot V., Coe E. H. Nuclear gene iojap conditions a programmed change to ribosome-less plastids in Zea mays. Proc Natl Acad Sci U S A. 1979 Jun;76(6):2760–2764. doi: 10.1073/pnas.76.6.2760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wallace D. C. Mitochondrial DNA mutations and neuromuscular disease. Trends Genet. 1989 Jan;5(1):9–13. doi: 10.1016/0168-9525(89)90005-x. [DOI] [PubMed] [Google Scholar]
- von Heijne G., Nishikawa K. Chloroplast transit peptides. The perfect random coil? FEBS Lett. 1991 Jan 14;278(1):1–3. doi: 10.1016/0014-5793(91)80069-f. [DOI] [PubMed] [Google Scholar]