Abstract
Manipulation of the CD28/CTLA-4 pathway is at the heart of a number of immunomodulatory approaches used in both autoimmunity and cancer. Whilst it is clear that CTLA-4 is a critical regulator of T cell responses, the immunological contexts in which CTLA-4 controls immune responses are not well defined. Here we show that whilst CD80/CD86-dependent activation of resting human T cells caused extensive T cell proliferation and robust CTLA-4 expression, in this context CTLA-4 blocking antibodies had no impact on the response. In contrast, in settings where CTLA-4+ cells were present as “regulators”, inhibition of resting T cell responses was dependent on CTLA-4 expression and specifically related to the number of antigen presenting cells. At low numbers of APC or low levels of ligand, CTLA-4-dependent suppression was highly effective whereas at higher APC numbers or high levels of ligand, inhibition was lost. Accordingly, the degree of suppression correlated with the level of CD86 expression remaining on the antigen presenting cells. These data reveal clear rules for the inhibitory function of CTLA-4 on Treg which are predicted by its ability to remove ligands from antigen presenting cells.
Introduction
T cell activation takes place at the interface between T cells and antigen presenting cells (APC) in secondary lymphoid organs. Typically, APC at sites of infection, upregulate CD80 and CD86 in response to signalling by Toll-like receptors or other microbial pattern recognition receptors and migrate to lymph nodes (1), (2) (3). As a result, APC increase both in number and level of costimulatory molecule expression, resulting in the initiation of T cell responses in a CD28-dependent manner (4), (5), (6). CD28 signalling is important in the expansion, survival and helper function of T cells (7), (8), (9) (10). Against this background, the inhibitory receptor CTLA-4 shares the same ligands with CD28 but opposes T cell responses such that the absence of CTLA-4 results autoimmune T cell activation with accompanying tissue infiltration and destruction (11), (12). The expression of CTLA-4 on both regulatory T cells (Treg) as well as activated T cells raises the issue of the mechanism by which CTLA-4 acts and the immunological context where inhibition takes place. A surprisingly large number of models of CTLA-4 function have been proposed, including both cell intrinsic and extrinsic mechanisms (13), (14), (15). However, the ability of these models to predict CTLA-4 functional behaviour is variable. For example, despite popular perceptions of CTLA-4 as an inhibitory signal for T cell activation a consistent body of literature indicates that the major function of CTLA-4 in vivo is via a cell-extrinsic pathway (13), i.e. that CTLA-4 influences the cells around it rather than the cell expressing it. Therefore, whilst the role of CTLA-4 as a negative regulator is well established, the context for its effective function is not. Ultimately, understanding how to predictably measure and understand CTLA-4 function in humans has considerable implications in autoimmune settings as well as other disorders involving immune dysregulation.
We recently proposed a model for CTLA-4 function whereby the central feature was the ability of CTLA-4 to capture ligands (CD80 and CD86) from APC and degrade them inside the CTLA-4 expressing cell (16). Such a mechanism is a form of cell-extrinsic ligand competition that makes several predictions for CTLA-4 function. Most obvious is that CTLA-4 function should be evident only when it depletes ligands to below a level sufficient for CD28 costimulation. A corollary of this concept is that the amount of ligand on the APC relative to the amount of CTLA-4 on T cells should dictate whether the threshold for CD28 costimulation is achieved. Accordingly, in situations where the supply of ligand is limited then consumption by CTLA-4 should be more functionally effective and vice versa.
We therefore set out to test how parameters such as the number of APC, and their relative ratio to CTLA-4+ cells affected the ability of CTLA-4 to regulate T cell activation. Using a model system, we demonstrate that the efficacy of suppression by CTLA-4 is dictated by the total amount of costimulatory molecules in the system. Under conditions favouring CTLA-4 function there was effective depletion of costimulatory ligands, sufficient to suppress T cell responses and the degree of suppression was tightly correlated with the observed downregulation of ligands on APC. In contrast, under un-favourable conditions with high levels of ligands, CTLA-4 continued to function however its impact on T cell proliferation was minimal since sufficient ligand still remained. Based on this model system, we tested the ability of natural Treg to suppress T cell responses. We observed that in accordance with our model, CTLA-4-dependent suppression was profoundly influenced by the ratio of APC:Treg and corresponded with the level of CD86 downregulation. In contrast, no CTLA-4-dependent inhibition was observed during stimulation with CD3/28 antibodies (Abs) showing that CTLA-4 suppressive function was strictly dependent on ligand-driven T cell activation. Together these data establish immunological contexts that predict the functional capacity of CTLA-4 that are consistent with a model whereby an important function of CTLA-4 is to deplete its ligands from APC in a T cell-extrinsic manner.
Materials and Methods
Cell lines and culture
CHO (Chinese hamster ovary) cells transfected with human CD80, CD86, CTLA-4 and Jurkat cells transduced with human CTLA-4 as previously described (17) and CHO cells with CD86GFP fusion protein with GFP tagged to the C-terminus of CD86 were previously described (16). CHO transfectants were maintained in DMEM (Invitrogen) complete culture media containing 10% FBS (Biosera), 1% L-glutamine (Sigma) and 1% penicillin and streptomycin (Invitrogen). Jurkat cells were maintained in RPMI 1640 (Invitrogen) complete culture media containing 10% FBS (Biosera), 1% L-glutamine (Sigma) and 1% penicillin and streptomycin (Invitrogen). All cells were cultured for at 37°C in 5% CO2 as previously described (18).
Preparation of human T cells and DC
PBMC were isolated from fresh human blood leucocyte cones, provided by The National Blood Transfusion Services (Birmingham and Colindale, U.K.) by density centrifugation on Ficoll-Paque PLUS (GE Healthcare). Human monocytes and T cells were isolated using EasySep® kit according to the manufacturer’s instructions (Stem Cell Technologies). Briefly, to deplete CD4+CD25+ Treg, CD4+CD25− T cells were purified by negative selection using EasySep® Human CD4+CD25− T cell enrichment kit. Naïve T cells were purified by negative selection using EasySep® Human CD4+CD25− T cell and then naïve CD4+ T cell enrichment kit. Treg were purified by negative selection using EasySep® Human CD4+ T cell and then CD4+CD127lowCD49d− Treg enrichment kit. Cells were collected, washed and utilised in T cell stimulation experiments. CD3−CD11c+CD14+ monocytes were purified by negative selection using human monocyte enrichment kit. To generate DC, monocytes were differentiated by culturing in RPMI 1640 (Invitrogen) DC culture media containing GM-CSF at 20ng/ml and IL-4 at 100ng/ml (Peprotech) at 37°C for 7 days.
Flow cytometry
T cells were labeled with anti-human CD3 FITC; CD4 PE, APC or PB; CD11c APC; CD14 PerCP; CD25 PE; CD45RA PE; CD45RO FITC, CD49d FITC; CD80 PE; CD86 FITC; CD127 PE-Cy7; and CTLA-4 PE (Pharmingen) and FoxP3 APC (eBioscience) antibodies at the recommended concentrations at 4°C in RPMI 1640 buffer with 2% FBS. CTLA-4 staining was carried out on fixed using 2% paraformaldehyde and permeabilised T cells with 0.05% saponin. FoxP3 (forkhead box P3) intracellular staining was performed using a FoxP3 staining kit (eBioscience) according to manufacturer’s instructions. For intracellular cytokine staining, after 5 days of stimulation samples were re-stimulated with PMA (50ng/ml), Ionomycin (1μM) and Brefeldin A (5μg/ml) for 4 hrs to induce cytokine production, the cells were then fixed with 3% PFA(paraformaldehyde) and permeabilised in a 5% PBS-Saponin solution before being stained for IL-2 production. For cell sorting, Far Red− CHO-CD86GFP cells were purified in over 98% purity on a MoFlo™ sorter (Dako) before used for T cell stimulation. All stained cell after washes were acquired on Cyan flow cytometer (Dako) using Summit software. Data analysis was performed using FlowJo software (Tree Star).
BIAcore and Ab blocking assay
Anti-CTLA-4 binding affinity was determined using surface plasmon resonance using a BIAcore 3000 instrument. CTLA-4Ig was bound through amine coupling to a CM5 chip as a target for Ab binding. Increasing concentrations of Tremelimumab, BNI3, 11G1 and 10A8 were then passed over the chip at 25°C for 30 mins and the amount of binding detected. The maximal response from each dilution was plotted as a graph and the affinity of the Ab calculated from the slope. In assays of blocking CTLA-4 binding to its ligands, various dilutions of the Abs were incubated with CTLA-4Ig that had been pre-labelled with a PE-fluorophore. To this, CHO cells expressing either CD80 or CD86 were added for 30 mins on ice. After washing, the detection of bound CTLA-4Ig was carried out by flow cytometry.
Immunofluorescence staining and confocal microscopy
For co-localisation of CD86 and CTLA-4, CHO cells, were incubated together for 16 hours on a poly-L-lysine coated coverslip in a 24 well plate. Cells were then fixed with methanol at −20°C for 20 mins. Non-specific binding sites were blocked by incubation in blocking solution which consisted of 5% donkey serum (Sigma) in PBS at room temperature. Cells were incubated with anti-CD86 primary Ab (B7-2, C19 Ab, Santa Cruz, CA) and anti-CTLA-4 (BNI3 Pharmingen), washed and then labeled with donkey anti-goat Alexa 546 and anti-mouse Alexa 488 secondary Abs (Molecular Probes). Imaging was carried out using a Zeiss LSM 510 or a Zeiss LSM 780 inverted laser scanning confocal microscope using a 100× oil immersion objective with excitation at 488nm and 543nm. Constant laser powers and acquisition parameters were maintained throughout individual experiments for analysis. Digital images were prepared using ImageJ (Wayne Rasband, NIH). All confocal images shown are representative of at least 30 micrographs.
CD86 transendocytosis assays
CD86 transfer from donor CHO cells expressing CD86-GFP fusion protein into recipient CHO cells expressing CTLA-4 was measured by flow cytometry. Donor 86-GFP cells were labeled with CellTrace™Far Red DDAO-SE labelling kit according to manufacturer’s instructions (Molecular Probes). In brief, CD86GFP+ CHO cells were incubated together with CTLA-4+ or control CHO cells in round-bottom 96-well plate at 37°C at various ratios for time stated. Cells were then disaggregated by pipetting, singlets were gated and GFP measured in both donors (Far Red+) and recipients by FACS. Anti-CD86 was also used to stain for residual CD86 expression on the donor cells in some cases.
T cell proliferation assays
Resting T cells were washed with PBS and incubated with CellTrace™Violet Cell Proliferation Kit according to manufacturer’s instructions (Molecular Probes). The reaction was quenched followed by PBS wash and cells were resuspended at 1.8 × 106 cells/ml before use as responder T cells. All T cell proliferation assays were performed in 250μl RPMI 1640 complete culture media. Responder T cells (0.9 × 105) were stimulated with soluble anti-CD3 Ab (OKT3; hybridoma from ATCC) at 0.5μg/ml or Dynabeads Human T-activator CD3/CD28 (Invitrogen) in 96-well flat bottom plates. To provide costimulation signals, monocyte-derived DCs or fixed CHO transfectants expressing human CD80, CD86 were added at indicated ratio as previously described (16). Cells were cultured for 5 days in presence or absence of 10ug/ml CTLA-4Ig (Abatacept; ORENCIA, BMS) to block CD80 and CD86. Division of responder T cells was measured by the dilution of violet dye using flow cytometry. Live proliferating T cell counts were performed using counting beads (Dako) and analysed using FlowJo software.
T cell suppression assays
In suppression assay, unlabelled CTLA-4+ Treg, Jurkat cells or T cell blasts were used as suppressors and added into violet-labeled responder T cells in presence or absence of anti-CTLA-4 blocking Abs (tremelimumab provided by Pfizer) at 40μg/ml. When using purified Treg or T cell blasts, these were negatively selected (untouched). When using Jurkat cells, their proliferation was inhibited by incubating with Mitomycin C (Sigma) 100μg/ml at 37°C for 1hr prior to adding into responder T cell culture. Activated T cells (T blasts) were generated by simulating naïve CD4 T with DC at DC:T ratio of 1:10 and soluble anti-CD3 Ab (OKT3; hybridoma from ATCC) at 0.5μg/ml for at 37°C for 7 days. To deplete DC, cells were incubated with anti-CD11c PE followed by anti-PE microbeads. CD11c+ DC were discarded by passing through a MS column (Milteni Biotech) and negative selected CTLA-4+ T blasts were then collected. T cell proliferation was measured as above.
Results
CTLA-4 expression does not intrinsically restrain primary T cell responses
In an attempt to understand the settings where CTLA-4 inhibits T cell responses that are specifically driven by the natural CD28 ligands, we established costimulation assays using cells transfected with either CD80 or CD86. In these experiments T cells were stimulated by either ligand in the presence of anti-CD3 and responses modulated by blocking with CTLA-4Ig or anti-CD28 Ab. This revealed that T cell proliferative responses were completely ligand and CD28-dependent (Fig. 1A). Strikingly, blockade with anti-CTLA-4 had no effect on ligand stimulated T cell responses (Fig. 1A). Given the lack of efficacy of anti-CTLA-4 Ab, we questioned whether T cells activated in this manner expressed CTLA-4 (Fig. 1B). The data showed that despite robust CTLA-4 expression both prior to and during cell division, blocking anti-CTLA-4 had no impact on cell division or indeed IL-2 production (Fig. 1C), both of which are proposed targets for CTLA-4 signalling. Given the reductionist nature of this experiment we further tested whether the same effect was seen using cultured dendritic cells (DC) to stimulate T cells. As shown (Fig. 1D and E), parallel data were obtained using DC, indicating little or no impact of CTLA-4 blockade in these settings. Thus, in spite of robust CTLA-4 expression prior to cell division and clear evidence that the natural ligands were effective in driving CD28 costimulation, CTLA-4 was not effective at inhibiting primary T cell responses. To study the kinetics of CD80/CD86 constimulation, we blocked CD80/CD86 by CTLA-4Ig during T cell stimulation driven by DC and anti-CD3 at different time points. Intriguingly, the inhibitory effect of T cell proliferation correlated with the earliness of CTLA-4Ig addition (Fig. 1F). This data indicate that CD80/86 constimulation is strictly required for T cell commitment to divide in the first 12 hour time window.
Figure 1. CTLA-4 blockade has little impact on resting T cell proliferation or IL-2 production.
CFSE labelled responder CD4 T cells were stimulated with 0.5μg/ml of soluble anti-CD3 and CHO-CD80 or CHO-CD86 expressing cells (A, B and C) or monocyte derived DC (D and E). A. T cell stimulations were carried out in the presence or absence anti-CTLA-4 Ab (Tremelimumab; 40μg/ml), CTLA-4 Ig (Abatacept 10μg/ml) or anti-CD28 Ab (10μg/ml) for 5 days and analysed by flow cytometry. B and C. Cells stimulated as in A were analysed for cell division and total CTLA-4 expression in the presence or absence of CTLA-4 blockade or for IL-2 production. In D and E, T cell responses were stimulated with monocyte-derived DC in the presence of anti-CD3 and analysed for CTLA-4 expression (D) and IL-2 production (E). For cytokine analysis, cultures were re-stimulated at day 5, with PMA/Ionomycin in the presence of Brefeldin A for 4 hours. The data presented is representative of five independent experiments. F. CD80/86 control commitment of CD4 T cells at the first 24-48 hours. Cell Trace Violet labeled naïve CD4 conventional T cells were stimulated by soluble anti-CD3 (0.5ug/ml) and human monocyte-derived DC in the presence of CTLA-4Ig (or Abatacept 10ug/ml) at indicated time after culture setup. Data are representative of three separate experiments.
To ensure that these results were not related to lack of effective blockade by anti-CTLA-4 we compared a number of different Abs for their binding to CTLA-4 using surface plasmon resonance and their capacity to block CTLA-4 interaction with its ligands. All Abs tested were effective at binding to CTLA-4 with affinities in the low nanomolar range (Fig. 2A and Table I). To confirm the capacity of anti-CTLA-4 Ab to block CTLA-4-ligand interactions, CTLA-4Ig was used to bind cells expressing CD80 or CD86 and detected by flow cytometry. The ability of the various CTLA-4 Abs to disrupt CTLA-4Ig binding to ligands was then tested. All Abs tested were capable of blocking CTLA-4 binding to both CD80 and CD86 and completely ablate ligand binding at a concentration of around 7nM (Fig. 2B and C). Furthermore at 10 fold lower concentrations nearly 80% binding to both CD80 and CD86 was blocked by tremelimumab and BNI3 (Fig. 2B and C). Given that we used a concentration of ~270nM (40μg/ml) in our functional assays this suggests that CTLA-4 was fully blocked in our experiments. Since tremelimumab was the highest affinity and most effective blocking Ab (albeit broadly similar to other anti-CTLA-4 Abs) this was used in subsequent functional experiments to try and identify contexts where CTLA-4 function was evident.
Figure 2. Characterization of blocking anti-CTLA-4 Abs.
A. Analysis of anti-CTLA-4 Abs binding to CTLA-4-Ig using surface plasmon resonance. The maximum response unit for each concentration of anti-CTLA-4 was plotted for four different anti-CTLA-4 clones. B and C. Anti-CTLA-4 Abs were tested for their ability to block CTLA-4-Ig binding to CD80 or CD86. CHO Cells expressing either CD80 (B) or CD86 (C) were incubated with 0.1μg/ml of CTLA-4Ig in the presence of increasing concentrations (nM) of Tremelimumab, BNI3, 11G1 and 10A8 anti-CTLA-4 Abs. Percentage blocking is calculated based on the MFI of unblocked CTLA-4-Ig staining.
Table I.
Calculated antibody binding affinity to CTLA-4
| Antibody | kD (nM) |
|---|---|
| Tremelimumab | 1.051 |
| BN13 | 6.908 |
| 11G1 | 13.89 |
| 10A8 | 6.891 |
KD value is determined using the non-linear regression one site binding model in Prism.
Determining the parameters that affect transendocytosis of ligands into CTLA-4 expressing cells
Since well characterised blocking Abs for CTLA-4 did not affect T cell responses in standard costimulation assays, we tested their ability to block ligand downregulation by CTLA-4 which occurs during the process of transendocytosis. Here, CTLA-4 expressing cells acquire CD80 and CD86 from the surface of donor cells resulting in ligand depletion (16). We initially tested the ability of anti-CTLA-4 to block ligand transfer as assessed by confocal microscopy. In these experiments anti-CTLA-4 potently inhibited transendocytosis as shown by the lack of CD86-GFP transfer to CTLA-4 expressing cells (red) as well as the retention of CD86-GFP on the donor cell surface (Fig. 3A). We also used flow cytometry to measure transendocytosis allowing us to simultaneously monitor downregulation of GFP-ligand from labelled donor cells (far-red) and track GFP uptake by the CTLA-4+ recipient CHO cells. This analysis showed that co-incubation of CD86-expressing cells with CTLA-4-expressing cells resulted in the loss of GFP from the CD86 donors and uptake of GFP by CTLA-4-expressing but not control recipients (Fig. 3B). To further explore the rate of depletion of CD86-GFP by CTLA-4 over time, we measured the expression of remaining GFP on the CD86 donors at various time points. At 4:1 of donor to recipient ratio, about 2-3% of GFP was depleted per hour. At 4:16 with increased amount of CTLA-4 present, the GFP depletion rate is approximately 2-4 times faster than that of at 4:1 (Fig. 3C). This suggests that the more CTLA-4 there were, the faster they depleted their ligand CD86.
Figure 3. Characterisation and blocking of CD86 transendocytosis.
A. CTLA-4 mediated acquisition of CD86 is blocked by anti-CTLA-4. Confocal micrographs show adherent CHO-CD86 cells and CHO-CTLA-4 cells after overnight incubation in presence or absence of anti-CTLA-4 blockade. CD86 (green) and CTLA-4 (red) were detected by Ab staining. Colocalization of CD86 and CTLA-4 is shown in yellow. B Depletion of CD86 from CD86+ cells is associated with concomitant CD86 acquisition by CTLA-4+ cells detected by flow cytometry. Far red labeled donor CHO-CD86-GFP cells were co-cultured with recipient CHO-CTLA-4 (RH panel) or CHO-Control (LH Panel) at 37°C for 20h. After coculture, singlet CTLA-4+ cells were analyzed for CD86-GFP acquisition based on far-red negative cells and loss of GFP from donors was determined by gating on far-red positive cells. C. Depletion of CD86 by CTLA-4 overtime. CHO-CD86-GFP cells were cultured with CHO-CTLA-4 as in B. at 4:1 (upper panel) and 4:16 (lower panel) of donor to recipient ratio for 3, 10 and 21 hours. The MFI of GFP remaining on donor cells were used to calculate relative expression level. Bar graph shows mean ± SEM of 3 experiments. P values were determined by unpaired, 2-tailed, t-test. *P < 0.05, **P < 0.01, ***P < 0.001. D and E. Mean fluorescence intensity (MFI) of GFP on donors or recipients are shown for both high and low densities of ligand expression at 3h(D) and 24h(E). F and G. CD86 remaining on donors is inversely correlated with CD86 acquired by CTLA-4 on recipients. Conditions in D-G are numbered as: 1. CHO-Control only; 2. CHO-CD86GFP only; 3. CHO-CTLA-4 only; 4. 4:0.25 (CHO-CD86GFP: CHO-CTLA-4); 5. 4:1 (CHO-CD86GFP: CHO-CTLA-4); 6. 4:4 (CHO-CD86GFP: CHO-CTLA-4); 7. 4:16 (CHO-CD86GFP: CHO-CTLA-4); 8. 4:16 (CHO-CD86GFP: CHO-Control). Total fluorescent intensity (TFI) of GFP was calculated as cell number × GFP MFI on both donors and recipients following transendocytosis as shown in C and D. G. Plots show the relationship between total GFP acquired by CTLA-4 expressing cells vs GFP remaining on the donors. Data are presented at 3h and 24h using both high and low density donors.
A number of interesting characteristics were evident from these experiments when we varied parameters predicted to dictate CTLA-4 function, such as the level of ligand expression, the relative ratios of CTLA-4:CD86-expressing cells and the contact time between cells. As shown in Fig. 3D-E, downregulation of ligand from the CD86+ donors (left panels) was compared with acquisition of GFP by the recipient CTLA-4+ cells (right panels) at 3h and 24h. In addition, donor CD86-GFP cells expressing either high and low levels of ligand were also compared (top vs bottom rows). In co-cultures with higher numbers of CD86-GFP donors relative to the number of CTLA-4 “regulators” (4:0.25 ratio - lane 4) downregulation of CD86 was limited especially at shorter co-culture times (Fig. 3D left panels). However, as we increased the number of CTLA-4+ cells (4:16 ratio - lane 7), or increased the co-culture time (Fig. 3E- 24 hrs) more substantial removal of CD86 was achieved. In control experiments with untransfected recipients (no CTLA-4) (lane 8) no GFP downregulation was observed. Interestingly, when we analysed GFP uptake into the CTLA-4 recipients (Fig. 3D and E right panels) we found a greater capture of CD86-GFP per CTLA-4+ cell at low CTLA-4 cell numbers (lane 4) compared with higher numbers (lane 7). Thus where CD86-bearing donor cells were abundant, each CTLA-4 recipient acquired more ligand per cell despite relatively ineffective downregulation of the donor. In contrast at low numbers of ligand bearing cells (and thereby relatively increased CTLA-4) each CTLA-4-recipient acquired less CD86-GFP per cell but achieved much more significant ligand downregulation overall.
From a perspective of immune regulation, the most relevant parameter is removal of ligand from the APC and therefore the total amount of CD86 costimulation available for T cell stimulation. We calculated this available costimulatory potential as the product of cell number × number of ligand molecules per cell. When viewed in this way, decreases in total CD86 in the donor population were matched by proportional increases of GFP acquired within the recipient cells (Fig 3F, G and H). This reinforces the conclusion that ligand loss from the CD86+ donor is a direct consequence of transfer to the CTLA-4 recipient via transendocytosis. Moreover, ratios at which ligand depletion was relatively ineffective at early timepoints (Fig 3F lane 6, 3h) nevertheless lead to substantial downregulation at later timepoints (Fig 3G lane 6, 3h v 24h) indicating that transendocytosis is an active time-dependent process.
With low levels of ligand, at later times downregulation occurred to such a marked extent that saturation was reached. i.e. increasing the number of CTLA4+ recipients did not reduce ligand levels further (compare E and F lower left panels lanes 4-7). Therefore, as shown in Fig. 3H at early time points a robust correlation exists between the amount of ligand removed from the donors and the total amount detected in recipients. However at later times and particularly at lower starting amounts this correlation breaks down as the ligand supply becomes exhausted. Overall, measuring total ligand downregulation revealed that control by CTLA-4 is most effective under conditions where removal is favoured by low amounts of initial ligand expression, higher numbers of CTLA-4 expressing cells, or by longer contact times.
T cell responses are directly proportional to level of ligand downregulation
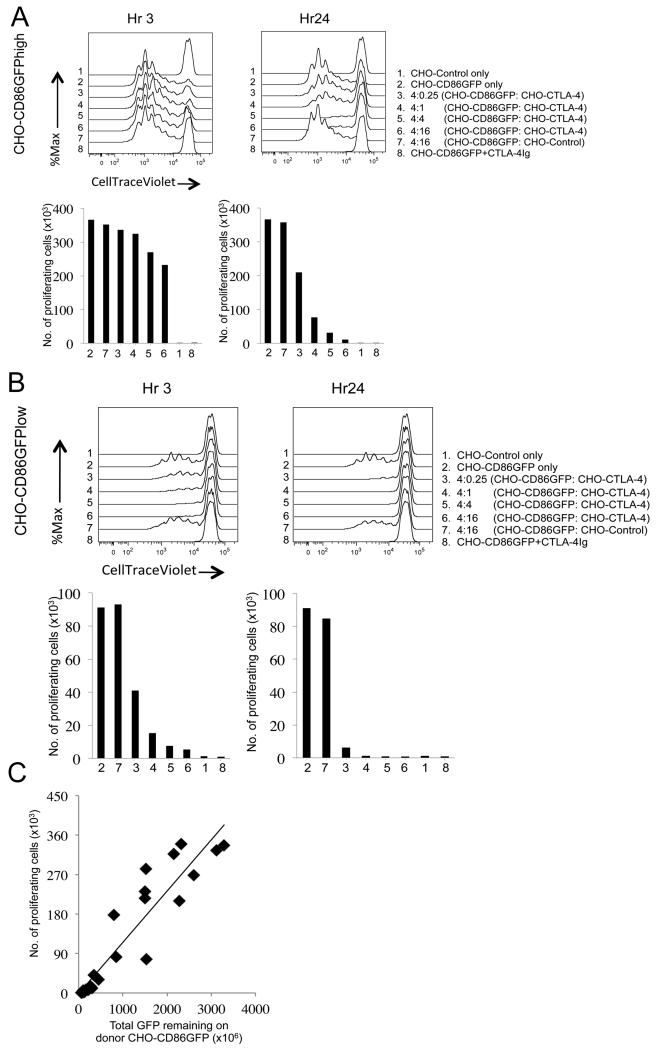
To determine the functional significance of different efficiencies of downregulation by CTLA-4, we recovered and purified the CD86-GFP expressing cells after contact with CTLA-4+ cells. The ligand expressing cells were then fixed to preserve their level of costimulatory molecule expression. These cells were then used to stimulate purified naïve human CD4+CD25− T cells in a typical CD28-costimulation assay. As shown in Fig. 4A, cells that were efficiently depleted of CD86 whether by increasing CTLA-4 ratios or by increasing contact time, were much less able to costimulate T cell responses. In contrast, those exposed to insufficient CTLA-4 “regulation”, e.g. having high level of CD86 initially or shorter contact times retained the ability to stimulate responses in a CD86-dependent manner. For example, conditions 5 and 6 resulted in marked depletion of CD86 at 24h and yielded cells that were extremely poor at costimulating T cell proliferation in contrast to the same ratio tested at 3h (Fig. 4A). Moreover, as predicted, the control of T cell responses by CTLA-4 was also substantially more marked when starting with lower levels of ligand on the APC. As seen in figure Fig. 4B, very modest ratios of CTLA-4:CD86 could suppress T cell responses especially at later time points where initial ligand levels were low. Furthermore when total CD86 level remaining was plotted against the number of proliferating T cells, a striking linear relationship was observed (Fig. 4C). Together, these data indicate that the overall reduction of costimulatory ligand determines the functional outcome of CTLA-4 transendocytosis. This in turn is controlled by the relative amounts of CD86 and CTLA-4 present and their contact time. Given that we observed a direct relationship between the amount of costimulation available and the magnitude of the T cell response this suggests that reducing ligand expression is a sensitive way and propotional way of controlling the commitment of T cells to a response.
Figure 4. CTLA-4 mediated down-regulation of CD86 is functionally relevant for T cell activation.
A and B. Cell trace violet-labelled human naïve CD4+ T cells were stimulated with CHO-CD86GFP cells which were FACS sorted following transendocytosis as performed in figure 1 in the presence of 0.5μg/ml soluble anti-CD3. Cell division was monitored using cell trace violet dilution at day 5 using flow cytometry. Live proliferating T cell counts were determined using counting beads and the total cell numbers shown in bar charts. Correlation between total CD86 remaining on the APC and total proliferating T cell number is shown in C. The data presented are representative of 3 independent experiments.
Suppression by CTLA-4 does not require a specialised cell type
Given the above data we wished to test the extent to which different CTLA-4 expressing cells could affect T cell costimulation. We therefore tested the ability of different CTLA-4 expressing cell types to act as “regulators” of T cell responses stimulated by DC. As shown in Fig. 5A CTLA-4+ Jurkat cells but not control Jurkats were able to suppress T cell responses in a manner that could be abrogated by anti-CTLA-4 Ab. Moreover, when T cell responses were stimulated by Ab coated beads rather than natural ligands, CTLA-4+ Jurkat cells were unable to inhibit the response, consistent with the ability of anti-CD28 Ab to bypass the requirement for ligand-driven costimulation (Fig. 5B). Similarly, we also investigated whether CTLA-4 on activated conventional CD4 T cells (T blasts) showed any suppressive function. CTLA-4+ blasts were also able to restrict T cell division in a CTLA-4 dependent manner particularly at lower DC:T blast ratios (Fig. 5C). Taken together these data suggest that, independent of T cell type, CTLA-4 can act extrinsically to reduce CD86 levels from DC and this is sufficient to mediate suppression. Moreover as predicted by our model, the efficiency of CTLA-4 dependent downregulation and its suppressive effect on T cell responses is highly sensitive to the number of DC (and consequently the overall amount of ligand) present.
Figure 5. CTLA-4 suppression via DC doesn’t require specialised cell type.
A. Violet-labelled human naïve T cells were stimulated using DC in the presence of anti-CD3 and suppression measured in response to CTLA-4 negative (control) or CTLA-4 transduced Jurkat cells. Assays were carried out in the presence or absence of anti-CTLA-4 or in the presence of CTLA-4Ig (10μg/ml) to establish costimulation dependence. Monocyte derived DC were used for costimulation at the indicated ratio. B. Violet-labelled naïve T cells were stimulated with anti-CD3/CD28 coated beads to provide a ligand independent stimulus and suppression of CTLA-4+ Jurkat cells monitored as in A. Cell division was monitored by dye dilution at day 5 using flow cytometry and the absolute number of dividing T cells shown in bar chart. Expression levels of CD86 on CD11c+ DC are shown in mean fluorescence intensity (MFI) by flow cytometric analysis using anti-CD86 Abs. C. Activated T cells expressing CTLA-4 were used as suppressor cells in the same manner as in A. Bar chart depicts mean ± SEM. n = 3. P values were determined by unpaired, 2-tailed, t-test. ns, not significant, P > 0.05, **P < 0.01, ***P < 0.001. Data are representative of 3 independent experiments.
Transendocytosis predicts CTLA-4–dependent Treg suppressive function
To determine whether CTLA-4-dependent suppression by natural Treg was consistent with data from our models we tested the ability of purified human CD4+CD25+ T cells to regulate T cells in the presence of variable numbers of DC. As shown in Fig. 6A high numbers of DC (8:1) resulted in the majority of T cells entering division, although the number of cell divisions was rather low. At these ratios, CTLA-4Ig also had limited impact indicating that when driven by abundant APC T cell responses are less CD28-dependent. However, as DC numbers were reduced, responses became increasingly CD28-dependent as judged by the impact of CTLA-4Ig. Moreover, at these reduced numbers of DC, Treg suppression was clearly evident and CTLA-4 blockade substantially, reversed the impact of Treg suppression. Again it was notable that anti-CTLA-4 had no effect on control T cell responses in the absence of Treg or when T cells were stimulated using CD3/CD28 beads (Fig. 6B). To exclude the possibility that CTLA-4-dependent Treg suppression at low DC numbers was simply due to lower overall magnitude of T cell stimulation we repeated these experiments keeping DC numbers fixed but this time increasing Treg. Again, increasing the ratio of Treg:DC resulted in more effective Treg suppression. This indicates that the relative ratio of Treg : DC is the critical factor influencing CTLA-4-based suppression rather than the DC number per se or indeed the Treg:responder ratio. As predicted, the degree of suppression was also correlated with the extent of downregulation of CD86 expression on the DC (Fig. 6C). Overall these data show that CTLA-4 function on Treg is effective at controlling T cell responses in a manner that relates to the extent of ligand downregulation on DC. In turn, ligand downregulation is a function of Treg:DC ratio and level of ligand depletion as predicted by transendocytosis as a mechanism.
Figure 6. Transendocytosis predicts the behavior of CTLA-4 dependent suppression by Treg.
A. Cell trace violet-labelled human naïve T cells were stimulated with soluble anti-CD3 (0.5μg/ml) and human monocyte-derived DC for costimulation at the indicated ratios in the absence or presence of Treg. Anti-CTLA-4 (40μg/ml) or CTLA-4-Ig (10ug/ml) were used to block CTLA-4 or ligands respectively. Cell division was monitored by violet dilution at day 5 using flow cytometry. Absolute number of dividing T cells were calculated and shown in bar charts. B. Naïve T cells were stimulated with CD3/CD28 beads and responses measured as in A. C. Suppression assays were carried out as in (A) with the exception that the DC numbers remained fixed and the Treg numbers increased thereby maintaining equivalent stimulation of responder T cells. Number of dividing T cells were calculated and shown in bar charts along with expression levels of CD86 on CD11c+ DC are shown as MFI. Bar graph shows mean ± SEM. n =3-5. P values were determined by unpaired, 2-tailed, t-test. ns, not significant, P > 0.05, **P < 0.01, ***P < 0.001. Data are representative of 5 experiments performed.
Discussion
CTLA-4 is an essential regulator of T cell function that in combination with the CD28 pathway represents a critical decision point in T cell activation. Manipulation of this checkpoint has led to the successful development of new therapeutics which are approved for use in autoimmunity (19) and cancer settings (20). Despite this success, the mechanistic basis for how the CTLA-4 checkpoint works has been uncertain and this has hampered the design of assays which can predictably measure CTLA-4 function in vitro. Consequently, at present there are no widely adopted assays with which measure CTLA-4 function available for human use. Here we report a set of robust basic principles underpinning CTLA-4 function which can both aid assay design as well as interpretation of the many experiments where CTLA-4, CD28 and their ligands are manipulated. The data presented here suggest that control of T cell responses by CTLA-4 is highly compatible with a model based upon the quantitative control of ligand expression.
A number of potential mechanisms of CTLA-4 action have been proposed (13), (14), (15) raising the issue of how much each mechanism of action contributes to CTLA-4 physiological function. This issue is not easily resolved by standard experimental approaches, for example mutagenesis frequently affects more than one postulated mechanism. Accordingly, mutations in the CTLA-4 cytoplasmic domain that might affect signalling, can also impact on protein expression levels at the cell surface (21) as well as transendocytosis (16). Thus, most experiments to date are not decisive, and the issue of how CTLA-4 functions still remains unresolved. An alternative approach is to take each model at face value and determine the extent to which it provides useful predictions of CTLA-4 behaviour. For example, intrinsic functions, such as the delivery of inhibitory signals by CTLA-4, would indicate that upon interaction with ligands, CTLA-4 will trigger inhibitory signals such that T cell activation or IL-2 production is impaired (22) (23). However, as shown here, although CTLA-4 is robustly observed during T cell activation it is difficult to demonstrate by CTLA-4 blockade that its expression intrinsically influences T cell responses. Accordingly, in our experiments despite T cell activation occurring in a ligand-driven and CD28-dependent manner, T cells proceed through cell division expressing high levels of CTLA-4, but show no evidence for inhibition as determined by blocking CTLA-4 Abs. These data suggest that, at the very least, there are significant contextual constraints in order to observe CTLA-4 ligands delivering T cell-intrinsic inhibition.
Considerable data also exist that a cell-extrinsic function of CTLA-4 is required to prevent autoimmune disease in vivo (24), (25), (26), (27), (28), (29), suggesting that CTLA-4 expressed by T cells works by regulating other cells. Consistent with this, a number of studies have suggested that CTLA-4 can alter the level of CD80 and CD86 expression on APC (30), (31), (29), (16), (32). We recently reported that such observations could be explained by transendocytosis, a process that results in the physical capture of ligands by CTLA-4 expressing cells (16). In the experiments described here we tested whether transendocytosis was a useful model on which to design assays capable of detecting CTLA-4 function. Data using cells transfected with CD86 or CTLA-4 provided initial insights into how such a mechanism might operate to regulate T cell responses. Several features were apparent from this model: specifically the efficacy of CTLA-4 suppression correlates extremely well with its ability to deplete sufficient ligand from the ligand bearing cells thereby inhibiting CD28 costimulation. This ability is in turn influenced by the ratio of CTLA-4:ligand expression. In practical terms this suggests that the number of ligand bearing APC and their level of ligand expression relative to the number of CTLA-4 expressing “regulatory” cells can broadly dictate whether or not CTLA-4 is effective at controlling the T cell response. Using such model systems we could demonstrate that the amount of ligand removed was highly correlated with the amount of ligand acquired by the CTLA-4 expressing cells, suggesting transendocytosis was the likely mechanism of downregulation. These concepts allowed us to conduct quantitative analysis of ligand uptake and suppression to reveal functional defects of Treg from human patients with heterozygous CTLA-4 mutations. This reveals clinically significant functional defects occur in the setting of ~50% loss of CTLA-4 expression, a concept entirely in keeping with the models proposed here (33). Importantly, the data from model transendocytosis experiments were highly consistent with the data obtained using DC and Treg expressing ligand and receptor at natural levels. Downregulation of CD86 by CTLA-4 was observed in the presence of Treg and reversed by CTLA-4 blockade and again the level of downregulation of CD86 was dependent on the relative ratios of DC:Treg correlating with the level of suppression. Therefore we propose that CTLA-4 effectively sets an activation threshold by controlling the amount of CD28 ligation available via cell-extrinsic ligand depletion.
The data presented here clearly emphasise the importance of ligand binding to CTLA-4. Indeed our studies highlight that in order for CTLA-4 to regulate a T cell response, the response needs to be both ligand and CD28-dependent. One simple test of such a property is whether a T cell response is sensitive to blockade by CTLA-4Ig. Accordingly, if the response is not suppressed by CTLA-4-Ig, then in our experience, CTLA-4 dependent Treg function is inevitably ineffective. Such a situation is seen in our control experiments where T cells are stimulated using CD3/CD28 beads, and are neither suppressed by abatacept nor by Treg. Accordingly, in our view supression assays that utilise anti-CD28 to stimulate T cell responses are unsuitable for measuring CTLA-4 function. These concepts are consistent with the fact that disease caused by CTLA-4 deficiency is dependent on both the presence of ligands and on CD28 (34), (35), (36) suggesting that CTLA-4 controls ligand driven stimulation in vivo.
One attractive feature of transendocytosis as a model of CTLA-4 function is that it presents a cogent argument for why CD28 and CTLA-4 share ligands. Indeed if this were not the case then this mechanism could not control CD28 function. This point is highlighted by the fact that CTLA-4 on Treg cannot control Ab-stimulated proliferation of T cells where ligands are not use to stimulate responses. Moreover, this also suggests that CTLA-4 dependent mechanisms are not able to control CD28-independent responses which may be relevant when considering the loss of CD28 which occurs during extensive or chronic T cell stimulation observed in autoimmune diseases (37), (38). In addition, our observation of transendocytosis by non Treg fits well with recent data showing that CTLA-4 does not have to be expressed by Foxp3-expressing Treg in order for it to function in a suppressive manner (25), (39). Indeed, recent data from the Sakaguchi lab provide further support for this concept by showing that CTLA-4 is a key component in generating Treg behaviour (32).
Our results also suggest that the functional impact of CTLA-4 on controlling T cell responses is likely to be highly variable depending on the degree of ligand downregulation achieved. Accordingly, in the steady state, ligand levels may be relatively low and sufficiently controlled by CTLA-4 expressed by Treg that activation of self-reactive T cells is precluded. Removal of CTLA-4 entirely, or selectively on Treg is sufficient to reveal underlying self-reactivity (29), (40). Conversely, our data would suggest that during robust priming of the immune system it is possible for CTLA-4 to be present on Treg but ineffective in restraining a T cell response due to overwhelming levels of ligand expression both due to increased APC and increased levels of ligand expression. Under such situations loss of CTLA-4 function is predicted to be of limited impact as has been observed experimentally (41). Additionally, control of ligand levels by CTLA-4 may also shape the magnitude and differentiation of the T cell response by potentially restricting cytokine production (42). Thus one interpretation of CTLA-4 function is that it can act as a rheostat, sensitive to both the number and activation state of DC and therefore the “context” for immune regulation.
Despite our emphasis of ligand binding in order for CTLA-4 to function, a number of reports suggest that CTLA-4 may work in a ligand independent manner (43), (44). It is reported ectopic overexpression of a splice variant CTLA-4 which lacks ligand binding region appears to protect mice from autoimmune disease models (45). Also transduction of cytoplasmic domain of CTLA-4 was shown to prevent animals from developing inflammatory conditions (46), (47). Whilst there may be distinct ligand-independent effects of CTLA-4, as pointed out above, the disease phenotype caused by CTLA-4 deficiency is largely ligand-dependent. In addition, it is notable that the cytoplasmic domain of CTLA-4 is efficiently recruited to clathrin coated pits via AP-2 (48), (49), (50). One possibility therefore is that expression of the cytoplasmic domain may have effects on the trafficking of a number of receptors, for example, transferrin receptor which is highly upregulated during T cell activation and is also internalised via AP-2. Thus ligand independent CTLA-4 may mediate effects either via impact on non-CTLA-4 related pathways such as iron transport or alternatively by influencing expression of full length CTLA-4 itself. Indeed, recent studies (51) have indicated that the function of the ligand independent form requires co-expression of the full-length molecule in some settings. Moreover, it is clear that expression of the ligand-independent form is generally insufficient to prevent disease caused by loss of full length CTLA-4, suggesting that the function of the ligand-dependent molecule is dominant (44), (51). Taken together whilst ligand-independent forms of CTLA-4 can alter disease outcomes in some settings their contribution to the overall function of CTLA-4 in vivo may be limited.
Ultimately, a precise understanding of the key mechanisms of CTLA-4 function may reveal new opportunities for further targeting of this key immune regulator. The success of anti-CTLA-4 in clinical trials has again focused attention on how such effects may be mediated. Recent evidence that Treg provide a key mechanism of anti-CTLA-4 efficacy is supported by the demonstration in tumour therapy that effectiveness required Treg depletion dependent on the Fc region of the anti-CTLA-4 Ab (52). An improved understanding of CTLA-4 function also impacts our use of related strategies such as ligand targeting therapies, eg. Abatacept and its high-affinity variant, Belatacept. Whether or not CTLA-4 provides distinct inhibitory signals based on its ligation or whether its effects are achieved predominately via limiting CD28 function clearly affects such approaches. The data presented here provide support for the view that a clear function of CTLA-4 is the cell-extrinsic control of CD28 ligands on APC. Moreover, this function of CTLA-4 appears to conform to precise and predictable rules which govern its function on Treg.
Acknowledgements
We are grateful to Pfizer for the generous gift of anti-CTLA-4 (tremelimumab).
This work was supported by the Wellcome Trust (T.Z.H.), UK Biotechnology and Biological Sciences Research Council (O.S.Q.), MRC (L.S.K.W. and C.W.). L.S.K.W. is an MRC Senior Fellow.
Abbreviation used in this article
- CTLA-4
cytolytic T lymphocyte-associated antigen-4
- Treg
regulatory T cell
- CHO
Chinese hamster ovary
- DC
dendritic cell
- FoxP3
forkhead box P3
Footnotes
The authors have no financial conflicts of interest.
References
- 1.Banchereau J, Steinman RM. Dendritic cells and the control of immunity. Nature. 1998;392:245–252. doi: 10.1038/32588. [DOI] [PubMed] [Google Scholar]
- 2.Reis e S. Dendritic cells as sensors of infection. Immunity. 2001;14:495–498. doi: 10.1016/s1074-7613(01)00136-4. [DOI] [PubMed] [Google Scholar]
- 3.Akira S, Takeda K, Kaisho T. Toll-like receptors: critical proteins linking innate and acquired immunity. Nat.Immunol. 2001;2:675–680. doi: 10.1038/90609. [DOI] [PubMed] [Google Scholar]
- 4.Linsley PS, Clark EA, Ledbetter JA. T-cell antigen CD28 mediates adhesion with B cells by interacting with activation antigen B7/BB-1. Proc.Natl.Acad.Sci.U.S.A. 1990;87:5031–5035. doi: 10.1073/pnas.87.13.5031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Azuma M, Ito D, Yagita H, Okumura K, Phillips JH, Lanier LL, Somoza C. B70 antigen is a second ligand for CTLA-4 and CD28. Nature. 1993;366:76–79. doi: 10.1038/366076a0. [DOI] [PubMed] [Google Scholar]
- 6.Hathcock KS, Laszlo G, Pucillo C, Linsley P, Hodes RJ. Comparative analysis of B7-1 and B7-2 costimulatory ligands: expression and function. J.Exp.Med. 1994;180:631–640. doi: 10.1084/jem.180.2.631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Linsley PS, Brady W, Grosmaire L, Aruffo A, Damle NK, Ledbetter JA. Binding of the B cell activation antigen B7 to CD28 costimulates T cell proliferation and interleukin 2 mRNA accumulation. J.Exp.Med. 1991;173:721–730. doi: 10.1084/jem.173.3.721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Boise LH, Minn AJ, Noel PJ, June CH, Accavitti MA, Lindsten T, Thompson CB. CD28 costimulation can promote T cell survival by enhancing the expression of Bcl-XL. Immunity. 1995;3:87–98. doi: 10.1016/1074-7613(95)90161-2. [DOI] [PubMed] [Google Scholar]
- 9.Walker LS, Gulbranson-Judge A, Flynn S, Brocker T, Lane PJ. Co-stimulation and selection for T-cell help for germinal centres: the role of CD28 and OX40. Immunol.Today. 2000;21:333–337. doi: 10.1016/s0167-5699(00)01636-4. [DOI] [PubMed] [Google Scholar]
- 10.Greenwald RJ, Freeman GJ, Sharpe AH. The B7 family revisited. Annual review of immunology. 2005;23:515–548. doi: 10.1146/annurev.immunol.23.021704.115611. [DOI] [PubMed] [Google Scholar]
- 11.Waterhouse P, Penninger JM, Timms E, Wakeham A, Shahinian A, Lee KP, Thompson CB, Griesser H, Mak TW. Lymphoproliferative disorders with early lethality in mice deficient in Ctla-4. Science. 1995;270:985–988. doi: 10.1126/science.270.5238.985. [DOI] [PubMed] [Google Scholar]
- 12.Tivol EA, Borriello F, Schweitzer AN, Lynch WP, Bluestone JA, Sharpe AH. Loss of CTLA-4 leads to massive lymphoproliferation and fatal multiorgan tissue destruction, revealing a critical negative regulatory role of CTLA-4. Immunity. 1995;3:541–547. doi: 10.1016/1074-7613(95)90125-6. [DOI] [PubMed] [Google Scholar]
- 13.Walker LS, Sansom DM. The emerging role of CTLA4 as a cell-extrinsic regulator of T cell responses. Nat.Rev.Immunol. 2011;11:852–863. doi: 10.1038/nri3108. [DOI] [PubMed] [Google Scholar]
- 14.Rudd CE. The reverse stop-signal model for CTLA4 function. Nat.Rev.Immunol. 2008;8:153–160. doi: 10.1038/nri2253. [DOI] [PubMed] [Google Scholar]
- 15.Wing K, Yamaguchi T, Sakaguchi S. Cell-autonomous and -non-autonomous roles of CTLA-4 in immune regulation. Trends Immunol. 2011;32:428–433. doi: 10.1016/j.it.2011.06.002. [DOI] [PubMed] [Google Scholar]
- 16.Qureshi OS, Zheng Y, Nakamura K, Attridge K, Manzotti C, Schmidt EM, Baker J, Jeffery LE, Kaur S, Briggs Z, Hou TZ, Futter CE, Anderson G, Walker LS, Sansom DM. Trans-endocytosis of CD80 and CD86: a molecular basis for the cell-extrinsic function of CTLA-4. Science. 2011;332:600–603. doi: 10.1126/science.1202947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Mead KI, Zheng Y, Manzotti CN, Perry LC, Liu MK, Burke F, Powner DJ, Wakelam MJ, Sansom DM. Exocytosis of CTLA-4 is dependent on phospholipase D and ADP ribosylation factor-1 and stimulated during activation of regulatory T cells. J.Immunol. 2005;174:4803–4811. doi: 10.4049/jimmunol.174.8.4803. [DOI] [PubMed] [Google Scholar]
- 18.Sansom DM, Wilson A, Boshell M, Lewis J, Hall ND. B7/CD28 but not LFA-3/CD2 interactions can provide ‘third-party’ co-stimulation for human T-cell activation. Immunology. 1993;80:242–247. [PMC free article] [PubMed] [Google Scholar]
- 19.Bluestone JA, St Clair EW, Turka LA. CTLA4Ig: bridging the basic immunology with clinical application. Immunity. 2006;24:233–238. doi: 10.1016/j.immuni.2006.03.001. [DOI] [PubMed] [Google Scholar]
- 20.Sharma P, Wagner K, Wolchok JD, Allison JP. Novel cancer immunotherapy agents with survival benefit: recent successes and next steps. Nat.Rev.Cancer. 2011;11:805–812. doi: 10.1038/nrc3153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Masteller EL, Chuang E, Mullen AC, Reiner SL, Thompson CB. Structural analysis of CTLA-4 function in vivo. J.Immunol. 2000;164:5319–5327. doi: 10.4049/jimmunol.164.10.5319. [DOI] [PubMed] [Google Scholar]
- 22.Krummel MF, Allison JP. CD28 and CTLA-4 have opposing effects on the response of T cells to stimulation. J.Exp.Med. 1995;182:459–465. doi: 10.1084/jem.182.2.459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Walunas TL, Bakker CY, Bluestone JA. CTLA-4 ligation blocks CD28-dependent T cell activation. J.Exp.Med. 1996;183:2541–2550. doi: 10.1084/jem.183.6.2541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Bachmann MF, Kohler G, Ecabert B, Mak TW, Kopf M. Cutting edge: lymphoproliferative disease in the absence of CTLA-4 is not T cell autonomous. J.Immunol. 1999;163:1128–1131. [PubMed] [Google Scholar]
- 25.Wang CJ, Kenefeck R, Wardzinski L, Attridge K, Manzotti C, Schmidt EM, Qureshi OS, Sansom DM, Walker LS. Cutting edge: cell-extrinsic immune regulation by CTLA-4 expressed on conventional T cells. J.Immunol. 2012;189:1118–1122. doi: 10.4049/jimmunol.1200972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Schmidt EM, Wang CJ, Ryan GA, Clough LE, Qureshi OS, Goodall M, Abbas AK, Sharpe AH, Sansom DM, Walker LS. Ctla-4 controls regulatory T cell peripheral homeostasis and is required for suppression of pancreatic islet autoimmunity. J.Immunol. 2009;182:274–282. doi: 10.4049/jimmunol.182.1.274. [DOI] [PubMed] [Google Scholar]
- 27.Homann D, Dummer W, Wolfe T, Rodrigo E, Theofilopoulos AN, Oldstone MB, von Herrath MG. Lack of intrinsic CTLA-4 expression has minimal effect on regulation of antiviral T-cell immunity. J.Virol. 2006;80:270–280. doi: 10.1128/JVI.80.1.270-280.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Friedline RH, Brown DS, Nguyen H, Kornfeld H, Lee J, Zhang Y, Appleby M, Der SD, Kang J, Chambers CA. CD4+ regulatory T cells require CTLA-4 for the maintenance of systemic tolerance. J.Exp.Med. 2009;206:421–434. doi: 10.1084/jem.20081811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Wing K, Onishi Y, Prieto-Martin P, Yamaguchi T, Miyara M, Fehervari Z, Nomura T, Sakaguchi S. CTLA-4 control over Foxp3+ regulatory T cell function. Science. 2008;322:271–275. doi: 10.1126/science.1160062. [DOI] [PubMed] [Google Scholar]
- 30.Cederbom L, Hall H, Ivars F. CD4+CD25+ regulatory T cells down-regulate co-stimulatory molecules on antigen-presenting cells. Eur.J.Immunol. 2000;30:1538–1543. doi: 10.1002/1521-4141(200006)30:6<1538::AID-IMMU1538>3.0.CO;2-X. [DOI] [PubMed] [Google Scholar]
- 31.Oderup C, Cederbom L, Makowska A, Cilio CM, Ivars F. Cytotoxic T lymphocyte antigen-4-dependent down-modulation of costimulatory molecules on dendritic cells in CD4+ CD25+ regulatory T-cell-mediated suppression. Immunology. 2006;118:240–249. doi: 10.1111/j.1365-2567.2006.02362.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Yamaguchi T, Kishi A, Osaki M, Morikawa H, Prieto-Martin P, Wing K, Saito T, Sakaguchi S. Construction of self-recognizing regulatory T cells from conventional T cells by controlling CTLA-4 and IL-2 expression. Proc.Natl.Acad.Sci.U.S.A. 2013;110:E2116–E2125. doi: 10.1073/pnas.1307185110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Schubert D, Bode C, Kenefeck R, Hou TZ, Wing JB, Kennedy A, Bulashevska A, Petersen BS, Schaffer AA, Gruning BA, Unger S, Frede N, Baumann U, Witte T, Schmidt RE, Dueckers G, Niehues T, Seneviratne S, Kanariou M, Speckmann C, Ehl S, Rensing-Ehl A, Warnatz K, Rakhmanov M, Thimme R, Hasselblatt P, Emmerich F, Cathomen T, Backofen R, Fisch P, Seidl M, May A, Schmitt-Graeff A, Ikemizu S, Salzer U, Franke A, Sakaguchi S, Walker LS, Sansom DM, Grimbacher B. Autosomal dominant immune dysregulation syndrome in humans with CTLA4 mutations. Nature medicine. 2014 doi: 10.1038/nm.3746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Tai X, Van LF, Sharpe AH, Singer A. Induction of autoimmune disease in CTLA-4−/− mice depends on a specific CD28 motif that is required for in vivo costimulation. Proc.Natl.Acad.Sci.U.S.A. 2007;104:13756–13761. doi: 10.1073/pnas.0706509104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Tivol EA, Boyd SD, McKeon S, Borriello F, Nickerson P, Strom TB, Sharpe AH. CTLA4Ig prevents lymphoproliferation and fatal multiorgan tissue destruction in CTLA-4-deficient mice. J.Immunol. 1997;158:5091–5094. [PubMed] [Google Scholar]
- 36.Mandelbrot DA, McAdam AJ, Sharpe AH. B7-1 or B7-2 is required to produce the lymphoproliferative phenotype in mice lacking cytotoxic T lymphocyte-associated antigen 4 (CTLA-4) J.Exp.Med. 1999;189:435–440. doi: 10.1084/jem.189.2.435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Schmidt D, Goronzy JJ, Weyand CM. CD4+ CD7− CD28− T cells are expanded in rheumatoid arthritis and are characterized by autoreactivity. The Journal of clinical investigation. 1996;97:2027–2037. doi: 10.1172/JCI118638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Markovic-Plese S, Cortese I, Wandinger KP, McFarland HF, Martin R. CD4+CD28− costimulation-independent T cells in multiple sclerosis. The Journal of clinical investigation. 2001;108:1185–1194. doi: 10.1172/JCI12516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Corse E, Allison JP. Cutting edge: CTLA-4 on effector T cells inhibits in trans. J.Immunol. 2012;189:1123–1127. doi: 10.4049/jimmunol.1200695. [DOI] [PubMed] [Google Scholar]
- 40.Ise W, Kohyama M, Nutsch KM, Lee HM, Suri A, Unanue ER, Murphy TL, Murphy KM. CTLA-4 suppresses the pathogenicity of self antigen-specific T cells by cell-intrinsic and cell-extrinsic mechanisms. Nature immunology. 2010;11:129–135. doi: 10.1038/ni.1835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Walker LS, Ausubel LJ, Chodos A, Bekarian N, Abbas AK. CTLA-4 differentially regulates T cell responses to endogenous tissue protein versus exogenous immunogen. J.Immunol. 2002;169:6202–6209. doi: 10.4049/jimmunol.169.11.6202. [DOI] [PubMed] [Google Scholar]
- 42.Kastenmuller W, Gasteiger G, Subramanian N, Sparwasser T, Busch DH, Belkaid Y, Drexler I, Germain RN. Regulatory T cells selectively control CD8+ T cell effector pool size via IL-2 restriction. J.Immunol. 2011;187:3186–3197. doi: 10.4049/jimmunol.1101649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Chikuma S, Abbas AK, Bluestone JA. B7-independent inhibition of T cells by CTLA-4. J.Immunol. 2005;175:177–181. doi: 10.4049/jimmunol.175.1.177. [DOI] [PubMed] [Google Scholar]
- 44.Araki M, Chung D, Liu S, Rainbow DB, Chamberlain G, Garner V, Hunter KM, Vijayakrishnan L, Peterson LB, Oukka M, Sharpe AH, Sobel R, Kuchroo VK, Wicker LS. Genetic evidence that the differential expression of the ligand-independent isoform of CTLA-4 is the molecular basis of the Idd5.1 type 1 diabetes region in nonobese diabetic mice. J.Immunol. 2009;183:5146–5157. doi: 10.4049/jimmunol.0802610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Vijayakrishnan L, Slavik JM, Illes Z, Greenwald RJ, Rainbow D, Greve B, Peterson LB, Hafler DA, Freeman GJ, Sharpe AH, Wicker LS, Kuchroo VK. An autoimmune disease-associated CTLA-4 splice variant lacking the B7 binding domain signals negatively in T cells. Immunity. 2004;20:563–575. doi: 10.1016/s1074-7613(04)00110-4. [DOI] [PubMed] [Google Scholar]
- 46.Choi JM, Ahn MH, Chae WJ, Jung YG, Park JC, Song HM, Kim YE, Shin JA, Park CS, Park JW, Park TK, Lee JH, Seo BF, Kim KD, Kim ES, Lee DH, Lee SK, Lee SK. Intranasal delivery of the cytoplasmic domain of CTLA-4 using a novel protein transduction domain prevents allergic inflammation. Nat.Med. 2006;12:574–579. doi: 10.1038/nm1385. [DOI] [PubMed] [Google Scholar]
- 47.Choi JM, Kim SH, Shin JH, Gibson T, Yoon BS, Lee DH, Lee SK, Bothwell AL, Lim JS, Lee SK. Transduction of the cytoplasmic domain of CTLA-4 inhibits TcR-specific activation signals and prevents collagen-induced arthritis. Proc.Natl.Acad.Sci.U.S.A. 2008;105:19875–19880. doi: 10.1073/pnas.0805198105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Chuang E, Alegre ML, Duckett CS, Noel PJ, Vander Heiden MG, Thompson CB. Interaction of CTLA-4 with the clathrin-associated protein AP50 results in ligand-independent endocytosis that limits cell surface expression. J.Immunol. 1997;159:144–151. [PubMed] [Google Scholar]
- 49.Shiratori T, Miyatake S, Ohno H, Nakaseko C, Isono K, Bonifacino JS, Saito T. Tyrosine phosphorylation controls internalization of CTLA-4 by regulating its interaction with clathrin-associated adaptor complex AP-2. Immunity. 1997;6:583–589. doi: 10.1016/s1074-7613(00)80346-5. [DOI] [PubMed] [Google Scholar]
- 50.Qureshi OS, Kaur S, Hou TZ, Jeffery LE, Poulter NS, Briggs Z, Kenefeck R, Willox AK, Royle SJ, Rappoport JZ, Sansom DM. Constitutive clathrin-mediated endocytosis of CTLA-4 persists during T cell activation. J.Biol.Chem. 2012;287:9429–9440. doi: 10.1074/jbc.M111.304329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Stumpf M, Zhou X, Bluestone JA. The B7-independent isoform of CTLA-4 functions to regulate autoimmune diabetes. J.Immunol. 2013;190:961–969. doi: 10.4049/jimmunol.1201362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Simpson TR, Li F, Montalvo-Ortiz W, Sepulveda MA, Bergerhoff K, Arce F, Roddie C, Henry JY, Yagita H, Wolchok JD, Peggs KS, Ravetch JV, Allison JP, Quezada SA. Fc-dependent depletion of tumor-infiltrating regulatory T cells co-defines the efficacy of anti-CTLA-4 therapy against melanoma. J.Exp.Med. 2013;210:1695–1710. doi: 10.1084/jem.20130579. [DOI] [PMC free article] [PubMed] [Google Scholar]