Abstract
Impaired social interaction is a hallmark symptom of many psychiatric disorders. In substance use disorders, impaired social interaction is triply harmful (a) because addicts increasingly prefer the drug of abuse to the natural reward of drug-free social interaction, thus worsening the progression of the disease by increasing their drug consumption, (b) because treatment adherence and, consequently, treatment success itself depends on the ability of the recovering addict to maintain social interaction and adhere to treatment, and (c) because socially interacting with an individual suffering from a substance use disorder may be harmful for others. Helping the addict reorient his/her behavior away from the drug of abuse toward social interaction would therefore be of considerable therapeutic benefit. This article reviews our work on the neural basis of such a reorientation from cocaine, as a prototypical drug of abuse, toward dyadic (i.e. one-to-one) social interaction and compares our findings with the effects of other potentially beneficial interventions, that is, environmental enrichment or paired housing, on the activation of the accumbens and other brain regions involved in behavior motivated by drugs of abuse or nondrug stimuli. Our experimental models are based on the conditioned place preference paradigm. As the therapeutically most promising finding, only four 15 min episodes of dyadic social interaction were able to inhibit both the subsequent reacquisition/re-expression of preference for cocaine and the neural activation associated with this behavior, that is, an increase in the expression of the immediate early gene Early Growth Response protein 1 (EGR1, Zif268) in the nucleus accumbens, basolateral and central amygdala, and the ventral tegmental area. The time spent in the cocaine-associated conditioning compartment was correlated with the density of EGR1-activated neurons not only in the medial core (AcbCm) and medial shell (AcbShm) of the nucleus accumbens, but was observed in all regions medial to the anterior commissure (‘accumbens corridor’), including (from medial to lateral), the vertical limb of the diagonal band and the medial septum (VDB+MS), the major island of Calleja and the intermediate nucleus of the lateral septum (ICjM+LSI), the AcbShm, and the AcbCm. All effects were limited to GABAergic projection neurons (called ‘medium spiny neurons’, in the accumbens), encompassing both dopamine D1 receptor-expressing and D2 receptor-expressing medium spiny neuron subtypes. Our EGR1 expression findings were mirrored in multielectrode array recordings. Finally, we have validated our paradigm in C57BL/6 mice to make use of the plethora of transgenic models available in this genus.
Keywords: accumbens; cocaine; conditioned place preference; dopamine D1 receptor, dopamine D2 receptor; diagonal band; island of Calleja; medium spiny neuron; septum; social interaction
Impaired social interaction in dependence syndromes/substance use disorders
Impaired social interaction is a hallmark symptom of many psychiatric disorders (World Health Organization, 1992; American Psychiatric Association, 2013), such as major depression, dysthymia, schizophrenia, autism spectrum disorders, and ‘dependence syndromes’, also called ‘substance use disorders’, which are the focus of the present review. In substance use disorders, impaired social interaction is triply harmful (a) because addicts increasingly prefer the drug of abuse to the natural reward of drug-free social interaction, thus worsening the progression of the disease by increasing their drug consumption, (b) because treatment adherence itself depends on the ability of the recovering addict to maintain social interaction, and (c) because socially interacting with an individual suffering from a substance use disorder may be harmful for others, especially if they are not trained to cope with the addict’s behavior. In the following sections, these three issues will be discussed in more detail.
Drug addicts prefer the drug of abuse to drug-free social interaction
The clinical finding that nondrug-related social interactions are increasingly reduced during the progression of substance dependence is so well acknowledged in the field that both diagnostic manuals, that is, the International Classification of Diseases, revision 10, of the World Health Organization (1992) as well as the Diagnostic and Statistical Manual, revision 5 (DSM5), of the American Psychiatric Association (2013) have made this symptom a diagnostic criterion. Among healthcare providers, it is also well known that even treatment-seeking patients with a substance use disorder regularly prefer the drug of abuse over nondrug-associated social interaction, even if such a social interaction involves a well-meaning, empathic, and self-disciplined psychotherapist who can be expected to be trained to hold his/her negative emotions in check.
Treatment adherence itself depends on the ability of the recovering addict to maintain social interaction
Dyadic social interactions (DSIs) between treatment-seeking addicts and various healthcare providers (e.g. case manager, psychiatrist, psychotherapist, or social worker) are the mainstay of a successful therapy. The considerable difficulty that the recovering addict has in maintaining social interaction even with well-meaning and trained healthcare providers can be considered a major factor in why treatment adherence (compliance) in addicts is notoriously low and dropout rates (attrition) are high. For example, of the 1700 alcohol-dependent, treatment-seeking, and surviving participants of Project MATCH, which, to our knowledge, has remained the largest clinical trial comparing different psychotherapeutic approaches to treat alcohol dependence [comparing cognitive behavioral therapy (CBT); motivational enhancement therapy; and 12-step facilitation therapy, which is based on the Alcoholics Anonymous principles], only 37% of individuals in the outpatient group and 48% of individuals in the aftercare group completed the prescribed therapy sessions (12 CBT or 12-step facilitation therapy sessions or four motivational enhancement therapy sessions) and remained in treatment only for an average of around 8 weeks during the 12-week study (Mattson et al., 1998). Similarly, in a randomized clinical trial of medications for cocaine dependence (dopamine agonists, antidepressants, mood stabilizers, or nootropic/neuroprotectives vs. placebo), during which all 357 participants received CBT, only about 58% of the individuals in the placebo group and only about 56% of the individuals in the treatment group completed the 8-week trial as estimated by Kaplan–Meier analysis (Elkashef et al., 2005). Thus, none of the interventions tested was efficacious, and only 56–58% of the treatment-seeking cocaine-dependent individuals could adhere to psychotherapy even for a period as short as 8 weeks.
Social interaction with a substance-dependent individual may be harmful for others
The impaired social interaction of a substance-dependent individual may gravely harm others. In a recent large quantitative study comparing drug harm across pharmacological classes (Nutt et al., 2010), seven of 16 evaluation criteria referred to harm to others (as opposed to the harm afflicting the drug users themselves), with four of these seven criteria directly pertaining to social interaction: physical and psychological injury, crime, family adversities, and community. In treatment settings, even health professionals trained to interact with substance-dependent patients face a number of hazards with respect to their personal mental well-being and the coherence of the therapeutic team (Zernig et al., 2000, 2007). Thus, helping the addict to show more prosocial behavior is of considerable societal interest.
For all these reasons, the reorientation of an addict’s behavior away from the drug of abuse toward social interaction would be of considerable therapeutic benefit. To emphasize, all currently defined approaches – case management, psychotherapy, pharmacotherapy, and social work – are important for the treatment of substance use disorders (see e.g. Zernig et al., 2000). However, the pharmacotherapy of most substance use disorders – and especially the pharmacotherapy of cocaine addiction – is still very limited, both with respect to its effectiveness and the available medications (Pierce et al., 2012; van den Brink, 2012). Therefore, psychotherapy – defined here as a theory-guided structured dyadic (i.e. one-to-one) social interaction with the treatment-seeking addict – remains the mainstay of dependence syndrome therapy. To emphasize, social interaction (at least in its dyadic form) is not only an alternative stimulus to drug consumption (providing the addict with a nondrug-associated goal for reorientation) but is also a prerequisite for the treatment of dependence syndromes because the recovering addict’s ability to interact socially with his/her therapist is necessary for treatment adherence and, consequently, for treatment success. It is therefore worthwhile to investigate the neural basis of the distorted preference (choice) of the addict for the drug of abuse over (drug-free) social interaction and to investigate the neurobiological changes that underlie the reorientation from the drug of abuse toward social interaction, in the hope of developing medications that can enhance this beneficial shift.
Investigating the neural basis of the reorientation away from the drug of abuse toward dyadic social interaction
This review will focus on drug-free DSI of adult rodents, the therapeutically beneficial inhibiting effect of DSI on both the reacquistion/re-expression of conditioned place preference (CPP) for cocaine and the associated broad activation of the accumbens corridor. We will also compare this inhibitory effect of DSI on the cocaine CPP reacquisition/re-expression-associated activation of the accumbens corridor to the effect that other potentially beneficial interventions, that is, environmental enrichment (EE) (Solinas et al., 2008; Thiel et al., 2010; Chauvet et al., 2011) or pair housing (PC) (Thiel et al., 2010), have on the activation of the accumbens corridor and other brain regions involved in behavior motivated by drugs of abuse or nondrug stimuli.
Social influences (of which DSI is but one) on the neurobehavioral pharmacology of abused drugs have been covered by two recent excellent comprehensive reviews (Neisewander et al., 2012; Bardo et al., 2013), including a detailed comparison of the effects of ‘prosocial’/‘agonistic’/‘friendly’ versus ‘antagonistic’ social interaction on measures of drug abuse (Bardo et al., 2013). A special form of prosocial behavior, that is, play, has been reviewed by far more competent experts (Vanderschuren et al., 1997; Trezza et al., 2010) than the authors of the present review. Finally, it is well beyond the scope of the present review to discuss nonsocial alternative (i.e. nondrug) rewards (e.g. sweet taste or wheel running) to animal experimental measures of drug abuse and substance dependence; the reader is referred to the excellent work of, for example, Ahmed and coworkers on sweet taste (Lenoir et al., 2007) or Carroll and colleagues on physical exercise (i.e. wheel running; Zlebnik et al., 2012; Zlebnik and Carroll, 2015).
Our experimental models
We made use of the fact that social interaction is rewarding in the CPP paradigm (Douglas et al., 2004; Bardo et al., 2013) and have developed several CPP-based animal experimental models (Zernig et al., 2013) to study the preference of an individual for DSI versus cocaine as a prototypical drug of abuse and to investigate an individual’s reorientation from cocaine toward DSI.
The conditioned place preference paradigm as the basis of our experimental approaches: validity, translational power, and theoretical considerations
As emphasized above, our experimental models are based on the CPP paradigm (Rossi and Reid, 1976; Bardo and Bevins, 2000; Tzschentke, 2007), which allows the experimenter to quantify the control that previously neutral contextual stimuli acquire over an individual’s behavior after they have been associated with an unconditioned stimulus of interest [see, e.g., Fig. 1 of Zernig et al. (2007) for a schematic diagram of the various psychological constructs contributing toward apparent drug reward and drug reinforcement]. The unconditioned stimuli investigated in our models are DSI or cocaine. These stimuli acquire control over the animal’s behavior in the sense that they are approached and sought out (CPP) or are avoided [conditioned place aversion (CPA)]. Of note, CPP and CPA have recently been shown in humans for widely varying appetitive and aversive stimuli, that is, amphetamine (Childs and de Wit, 2009), palatable food (i.e. M&Ms) (Astur et al., 2014) and consonant versus dissonant music (Molet et al., 2013), the two latter studies using a virtual reality setting. Thus, animal CPP experiments have predictive validity for the human situation.
Fig. 1.

Timelines of the three experimental models used by our group. Details of the three experimental models are provided in the text. Red fields: place preference conditioning with cocaine (intraperitoneal injections, cocaine dissolved in saline in a volume of 1 ml/kg for rats, 10 ml/kg for mice). Tan field: extinction of cocaine conditioning by pairing the previously cocaine-associated conditioned place preference (CPP) compartment with i.p. saline injections. Green fields: conditioning to dyadic social interaction (preceded by an i.p. saline injection). Pretests (to quantify pre-CPP compartment bias) and CPP tests are not indicated here for the sake of clarity.
For example, many of us may have observed an urge to approach and linger at a location associated with a previous reward, be that a redecorated shop window, a closed coffee shop, or the emptied cookie jar. Having become conscious of this urge, some of us may even have commented on our behavior, using terms such as ‘craving’ (e.g. ‘I crave chocolate’ is a cultural staple in English-speaking countries) or ‘urge’ or ‘wanting’. Thus, the authors of this review would suggest that CPP is a plausible measure of what humans may be able to report as ‘craving’ (Epstein et al., 2009, 2010), one of the most important determinants of drug lapse and relapse (Preston et al., 2009). To emphasize, although ‘craving’ seems to be only a colloquial term, this is exactly what scientific studies on the subject assess in the participants [‘Right now, do you crave cocaine?’, see Epstein et al. (2010), p.310].
Whether the rodent CPP paradigm indeed represents a plausible measure of craving reported by human is a matter of debate. However, we would suggest that a discussion of the CPP paradigm that remains at the strictly descriptive level risks losing any translational power with respect to the human situation; we would therefore prefer to err on the side of overestimating the translational value of CPP-based animal experimental models. Most researchers and therapists in the addiction field would agree that drug-associated conditioned stimuli (drug cues) – which the CPP paradigm quantifies – ‘have powerful motivational effects in human drug abusers, eliciting craving and engendering drug-seeking behavior’ [Everitt and Robbins, 2000, p.20, reviewed by Zernig et al. (2007)]. Accordingly, craving is listed as a diagnostic criterion of ‘dependence disorders’ as defined by the World Health Organization (1992) and ‘substance use disorders’ as defined by the American Psychiatric Association (2013). Of note, the development and expression of craving does not require dependence and withdrawal (which in turn may require a long history of drug taking). For example, in a survey of the intravenous drug-injection habits of the patients of our substitution program outpatient clinic (Zernig et al., 2003), many participants anecdotally reported that they started craving the respective drug of abuse after their first experience with it. Most of us have indeed experienced craving for a stimulus after only one exposure, even if they may not act upon this craving. Accordingly, the human participants in the virtual reality CPP study by Molet et al., (2013) were exposed only once to an attractive or an aversive stimulus that many would consider as only mildly effective, that is, consonant or dissonant music, and developed robust CPP or CPA. In animals, CPP has also been reported after only a single drug–environment pairing (see, e.g. Bardo et al., 1986). Thus, the four episodes of stimulus exposure commonly used in CPP paradigms (Bardo and Bevins, 2000; Tzschentke, 2007) – including the models developed by us – seem more than adequate to model this phenomenon.
Although ‘social interactions are crucial for survival and reproduction, and accordingly,… are powerful determinants of behavior’ (Neisewander et al., 2012, p.34), a social interaction has both appetitive and aversive aspects. As an example, alcohol (ethanol) is often used as a ‘social lubricant’ because it relieves the anxiety/inhibitions resulting from the expectation of a social interaction’s negative aspects. Accordingly, the facilitation of social interaction (i.e. play) by low doses of ethanol and other anxiolytics can even be found in rats (Vanderschuren et al., 1997). Therefore, in our experimental models, the CPP for, or aversion to, social interaction must always be considered the sum total of its appetitive and aversive aspects and the sum total of the appetitive and aversive components of the handling and intraperitoneal (i.p.) saline injection administered immediately before placing the animals together within the confines of the CPP apparatus minus the sum total of the appetitive and aversive aspects of the alternative stimulus (i.e. the handling and i.p. injection of saline alone or of cocaine dissolved in the saline vehicle). The same, that is, the simultaneous existence of attractive and aversive aspects for a stimulus or even an aspect of stimulus, also holds true for ‘novelty’ (discussed in more detail below). To re-emphasize, the simultaneous contribution of appetitive and aversive aspects to an overall preference or avoidance is not particular for the composite stimulus ‘social interaction’ or the aspect of ‘novelty’. Cocaine has also been shown to have both appetitive and aversive properties at self-administered doses (see, e.g. Guzman and Ettenberg, 2007, or, for a review, Zernig et al., 2007). Therefore, a minimum of four different factors will have contributed toward the neural changes that the experimenter chooses to observe when studying the reorientation away from cocaine toward DSI, rendering any conclusion on the contribution of each one and on the nature of the interactions of these factors difficult. Our hope is to identify one neural parameter that is differentially affected by the overall preference (choice) of the individual for either DSI or cocaine.
Details of our experimental models
To briefly summarize, in our experimental models (see Fig. 1 for simplified timelines), animals receive an i.p. injection of saline and are placed in the conditioning chamber, either alone (saline control) or with another conspecific of the same sex and weight (DSI), or receive an i.p. injection of varying doses of cocaine and are placed in the conditioning chamber alone. Thus, in our experimental models, we offer the stimulus ‘DSI’ within the confines of the CPP apparatus, rendering it directly testable – and comparable with other stimuli such as cocaine – in the CPP paradigm itself as opposed to, for example, studying the effect of pair housing on separately tested cocaine CPP (Thiel et al., 2010). Single housing (individual housing) of the animals is started 1 week before the pretest and is continued throughout the experiment.
Sex and weight matching
In our experimental models, we eliminated sexual attraction and hierarchical differences (Kummer et al., 2011) as confounding factors by investigating social interaction only between sex-matched and weight-matched rodents. With respect to sex, we have so far published data only on male rodents.
Age
We initially chose to investigate young rodents (Sprague–Dawley rats or mice of the C57BL/6 or CD1 strain) on the basis that younger individuals may find social interaction more rewarding than older ones (Bardo et al., 2013) to increase the likelihood of a large enough DSI reward signal. Rats at an age of 6–8 weeks are variously termed ‘adolescent’ (Spear, 2000), ‘adult’ (Yates et al., 2013), ‘young adult’ (Zernig et al., 2013), or ‘early adult’ (Prast et al., 2014b), confirming the tenet that “adolescence” is a period that, according to a highly regarded review in the field, cannot be defined ‘by a discrete event or events’ (Spear, 2000, p.417).
Irrespective of terminology, the rodents investigated in our experimental models are clearly older than the rats that Vanderschuren and colleagues have been studying in their seminal work on social play (see, e.g. (Trezza et al., 2010; van Kerkhof et al. 2011; Achterberg et al., 2015): those animals are 3 weeks old (postnatal day 21, PND21) at intake (i.e. have just been weaned), are 4+ weeks old at the start of the behavioral experiments, and can be called ‘juvenile’ or ‘childlike’ (Dr Louk Vanderschuren, personal communication, 15 January 2015). In rats of this age, social (play) behavior can be used to induce CPP (Trezza et al., 2011).
The Sprague–Dawley rats that have been used by Bardo and coworkers as direct ‘adult’ comparators in an independent parametric study (Yates et al., 2013) of our initial findings (Fritz et al., 2011b) were 8.6 weeks old (PND60) at intake and were compared with ‘adolescent’ rats that were 3 weeks old (PND21) at intake. Neisewander and colleagues (Peartree et al., 2012) used ‘adolescent’ rats 3 weeks (PND22) at intake and 4–6 weeks (PND28–42) at the start of the experiments in an independent study that confirmed our findings on the importance of touch in social reward (Kummer et al., 2011).
Noncounterbalanced allocation to the initially nonpreferred compartment
It is our experience that reviewers often become highly concerned about a procedural detail, that is, the so-called ‘unbiased’ versus ‘biased’ design when discussing CPP compartment allocation. First, following the differentiation of Tzschentke (2007), we would like to emphasize that ‘biased’ versus ‘unbiased’ refers to the apparatus (‘CPP box’), that is, to the different visual and tactile stimuli used to differentiate the conditioning compartments. We have exercised care to keep the following stimuli as unbiased as possible: the wall pattern of the conditioning compartments has either vertical or horizontal stripes of equal total brightness and the steel plate floors have round holes or slits of a similar total fraction of the floor area. Overall, our CPP apparatus consists of three compartments of which the middle (neutral) one is smaller than the conditioning compartments, and has a plastic floor and walls painted white.
Again following the differentiation of Tzschentke (2007), we use a ‘noncounterbalanced’ design with respect to the initial compartment allocation in that the unconditioned stimulus of interest is allocated to the initially nonpreferred side. We believe that such a noncounterbalanced design is conceptually cleaner than the ‘counterbalanced’ allocation favored by some researchers. In our opinion, such a ‘counterbalanced’ design results in a mixture of two distinct groups of animals, that is, (a) those in which initial avoidance is reverted to preference and (b) those in which initial preference is strengthened. Generating such an inhomogenous starting group (which actually consists of two subgroups) subverts the statistical power of the expected findings and would in fact necessitate subjecting the two subgroups to separate statistical analyses. However, and as discussed in detail below (Fig. 2), we have found no statistical evidence that two distinct groups (i.e. ‘preferring’ and ‘avoiding’ animals) exist.
Fig. 2.

Times spent in the compartment associated with dyadic social interaction or cocaine: mouse versus rat comparison. Solid symbols show the time spent in the compartment associated with the stimulus of interest [(a), dyadic social interaction, int, green symbols; (b), 15 mg/kg i.p. cocaine, coc, red symbols] during the conditioned place preference (CPP) test (total test duration, 900 s). Each symbol represents the behavior of one individual animal. M, C57BL/6N mouse, square. R, Sprague–Dawley rat, triangle. All animals were male and weight matched (rats, 150–250 g; mice, 22–23 g). Open symbols represent transformed data, that is show the time spent in the compartment associated with the stimulus of interest minus the time spent in the saline-associated compartment. Black lines show the mean of the experimental group. The following numbers of animals and experimenters (given in parentheses) contributed toward each experimental group: M int, 42 mice (seven experimenters); R int, 27 rats (four experimenters); M coc, eight mice (two experimenters); and R coc, 26 rats (three experimenters). All groups except for the rat cocaine (R coc) and rat cocaine-saline (R coc-sal) groups passed all three normality tests used (Kolmogorov–Smirnov, D’Agostino–Pearson, Shapiro–Wilk; Prism 5; http://www.graphpad.com). After removing one visible outlier (time spent in the coc-associated compartment, 716 s) in the R coc group, the also R coc and R coc-sal group passed all normality tests too. i.p., intraperitoneally.
Translating from the situation of the human addict, it is also our opinion that a sufficiently strong conditioning of an associated stimulus by a drug of abuse should be independent of any initial preference for or avoidance of the to-be-conditioned stimulus. To explain this in more detail: it is a core criterion of substance dependence that an individual’s preference is channeled toward the drug of abuse at the cost of previously preferred alternative (i.e. nondrug) stimuli (World Health Organization, 1992; American Psychiatric Association, 2013; see also Zernig et al., 2007). This is actually the core symptom of substance dependence: irrespective of which stimuli an individual was attracted to before becoming dependent on the drug, substance dependence means that this individual’s preference has been directed almost exclusively to drug-associated stimuli. Hence, the continuous debate of the relative merits of a ‘counterbalanced’ versus an ‘noncounterbalanced’ initial compartment allocation is, in our opinion, irrelevant for the human situation. Accordingly, in our publications, we prefer to show the raw data for the time spent in each compartment (including the neutral compartment) and express CPP as the difference between the compartment associated with the stimulus of interest and the other conditioning compartment. It may be argued that if there is a strong initial bias for one of the compartments, then the effects of a treatment cannot distinguish between effects that influence loss of avoidance or gain of preference (approach). We would suggest that constructs such as ‘loss of avoidance’ or ‘gain of preference’ are very hard to show in the CPP paradigm. To give another example from our own laboratory, our CPP data (Fig. 2, discussed in detail below) do not show a dichotomous distribution of the times spent in the cocaine-associated or the social interaction-associated compartment (which would indicate two distinct populations of ‘preferring’ vs. ‘avoiding’ animals) in either rats or mice, but rather show a continuous gradient of time spent in the compartment of the stimulus of interest, even if the time spent in the comparator-associated compartment (i.e. i.p. saline injection) is subtracted from the time spent in the compartment associated with the stimulus of interest (cocaine or DSI). In practice, all these concerns may matter little, particularly as there are rarely strong initial preferences in CPP in most common arrangements. For more data on and discussion of ‘counterbalanced’ versus ‘noncounterbalanced’ compartment allocation, the reader is referred to the reviews by Tzschentke (2007) and Bardo et al. (1995).
Novelty
Novelty is a powerful reward (Bardo et al., 2013) and has to be considered an aspect of DSI. However, we always pair the same animals, thus obtaining a total of four 15 min interactions with the same individual before testing both animals for social interaction CPP. We thus take care to avoid novelty as a confounding variable – or to minimize novelty, depending on how strictly one cares to define ‘novelty’. The time course of social interaction counterconditioning (see Fritz et al., 2011b, Fig. 3) clearly shows that CPP for social interaction increases continuously over these four consecutive social interaction events. If novelty were a major determinant, CPP for these social interactions should decrease, not increase. However, novelty may also serve as an aversive stimulus, perhaps causing anxiety (‘neophobia’). In that case, CPP would increase continuously over the four subsequent social interaction episodes, as it did. A detailed behavioral analysis, however, showed that the rats spent 81% of the 15 min pairing in direct physical contact during the first encounter, 80% during the second, 76% during the third, and 78% during the fourth and final encounter (Kummer et al., 2014), that is, that the time spent in direct physical contact remained essentially the same from the first encounter onward. Thus, we have no indication that novelty plays any significant role in the social reward that we quantify in our paradigm.
Fig. 3.
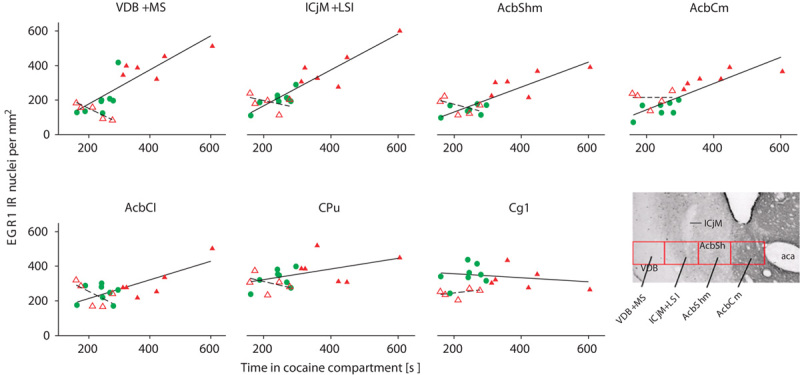
Accumbens corridor activation associated with the reacquisition/re-expression of cocaine CPP and its inhibition by dyadic social interaction. The correlations of EGR1 expression per mm2 versus the time spent in the cocaine compartment during the cocaine CPP reacquisition/re-expression test are shown for animals that had undergone cocaine conditioning, followed by extinction with saline (red triangles, filled) and for animals that were counterconditioned with social interaction after an initial cocaine conditioning (green circles, filled) before being subjected to a final cocaine challenge and tested for the reacquisition/re-expression of cocaine 24 h later. The diagram in the lower right corner was taken from the rat brain atlas of Paxinos and Watson (2007) and shows the following subregions of the accumbens corridor: aca, anterior commissure; AcbCm, medial accumbens core; AcbShm, medial accumbens shell; ICjM+LSI, major island of Calleja and intermediate nucleus of the lateral septum; VDB+MS, vertical limb of the diagonal band and medial septum. Solid lines show the correlation of data pooled from saline-extinguished (filled red triangles) and DSI-counterconditioned (filled green circles) animals. The respective correlation coefficients, r, and P values were for the VDB+MS: r=0.86, P<0.0001; ICjM+LSI: r=0.89, P<0.0001; AcbShm: r=0.87, P<0.0001; AcbCm: r=0.9, P<0.0001; AcbCl: r=0.33, P=0.12; CPu: r=0.41, P=0.07; and Cg1: r=−0.16, P=0. 29. There was no statistically significant correlation (shown as a dashed line) between the EGR1 expression per mm2 and the time spent in the cocaine compartment for animals injected noncontingently with cocaine (unfilled red triangles). This figure is an extraction and combination of the data presented in figures 6 and 8 of Prast et al. (2014b).
We have developed three different experimental approaches, ranked in the following according to decreasing experimenter work requirement (Zernig et al., 2013). These three approaches test different aspects of cocaine versus social interaction preference. Figure 1 presents simplified timelines for the three models.
The models
Model 1: social interaction counterconditioning effect on the reacquisition/re-expression of cocaine CPP
This is the experimental approach that in our opinion has the highest face validity and translational therapeutic promise for the situation of the treatment-seeking human addict. It is also by far the most time consuming, requiring 24 days for completion (see Fritz et al., 2011b, Fig. 1 or Prast et al., 2014b, Fig. 1). Animals are first trained to acquire CPP for cocaine. Then, the preference for cocaine is extinguished by pairing the previously cocaine-associated compartment with saline. (The other conditioning chamber has always been paired with an i.p. saline injection.) Extinction is obtained and tested in four cycles, each cycle being a sequence of saline conditioning - saline conditioning - CPP test. After the fourth extinction cycle, the animal is exposed to one more cocaine training session (arguably modeling a ‘freebie’ in the human situation) and tested for reacquisition/re-expression of cocaine CPP 24 h later, that is, in a cocaine-free state.
In the social interaction counterconditioning condition, after CPP for cocaine is established, the previously cocaine-paired compartment is paired with saline and the previously saline-paired compartment is now paired with the usual i.p. saline injection, followed by a DSI with a sex-matched and weight-matched male conspecific, through all training cycles. As social interaction serves as a reward in the CPP paradigm (Bardo et al., 2013), the animals usually reverse their CPP from cocaine to social interaction. The final cocaine challenge is performed 24 h after the last CPP test of the four cycles. The reacquisition/re-expression of cocaine CPP is tested 24 h after the last cocaine challenge (training), that is, in a cocaine-free state (Fritz et al., 2011b; Zernig et al., 2013).
Model 2: concurrent CPP for social interaction versus cocaine
In a much less time-consuming experiment (total experiment time, 10 days; see Fig. 1 of the present review or Fritz et al., 2011b, Fig. 1), CPP for DSI and cocaine is acquired concurrently in an alternate-day stimulus exposure paradigm (Fritz et al., 2011a) in the very same CPP apparatus (‘CPP box’) by pairing one compartment with an i.p. cocaine injection and, on alternate days, pairing the other compartment with an i.p. saline injection, followed by DSI within the confines of the CPP box. This concurrent CPP paradigm directly pitches the cocaine reward against the social interaction reward and should prove most useful when investigating the neural basis of the reorientation (preference shift) away from cocaine toward social interaction in a time-efficient experimental procedure. By manipulating the cocaine training dose, the concurrent CPP paradigm also allows for a fully quantitative analysis of the preference shift (Kummer et al., 2014). Our findings using this concurrent drug versus social interaction paradigm have been confirmed and further validated with amphetamine by Bardo and colleagues (Yates et al., 2013).
Model 3: CPP for either social interaction or cocaine
The least complex experimental approach with respect to the neural investigation of the CPP induced by cocaine versus DSI is to train and test different animals for either cocaine CPP or DSI CPP (Fritz et al., 2011b; El Rawas et al., 2012a). The time requirement is 10 days (see Fig. 1 of the present review or Fritz et al., 2011b, Fig. 1), that is, the same as for the concurrent CPP paradigm described as model 2 (above). Bardo and coworkers have further validated our paradigm and have found that the length of exposure to DSI and the age of the animals are of great importance for successfully establishing DSI as a reward (Yates et al., 2013).
Interestingly, the Sprague–Dawley rats tested by Bardo and colleagues were of an only slightly older age, that is, 8.6 weeks (PND60), than the 6–8-week-old Sprague–Dawley rats that we tested (Fritz et al., 2011b; Kummer et al., 2014). However, Bardo and colleagues found that their 8.6-week-old animals did not, on average, develop any preference or avoidance for DSI (irrespective of whether they were singly housed or pair housed), whereas our animals (which were singly housed) did, on average, develop a preference for DSI (see Fig. 2 for individual animals’ behavior). In a thorough discussion of this apparent discrepancy, Dr Yates and Dr Bardo (personal communication 31 May 2015) proposed that the most salient difference in the respective experimental protocols was the length of the DSI conditioning sessions, which lasted 15 min in our laboratory and 30 min in the Bardo laboratory. We all agree that social interaction may lose much of its attractiveness after the first 15 min [during which our rats spent ≥76% of their time in direct physical contact and engaged in a number of prosocial behaviors (Kummer et al., 2014)], although none of the rats investigated by Yates et al. (2013) seemed to have fallen asleep after the first 15 min. The importance of the 15 versus 30 min exposure time is underscored by the findings of Spear, who reported that social behavior declined during the last 5 min of a 15 min session (Douglas et al., 2004). Thus, experiencing DSI for 30 min may induce no preference for DSI in individually housed rats, whereas a 15 min DSI may induce a preference. Most likely, the combination of (a) the younger age of animals tested by us (with younger rats being more eager to engage in prosocial/play behavior) and (b) the more attractive DSI during the conditioning sessions induced DSI CPP in our rats may have resulted in the net DSI CPP that was not observed by Bardo and colleagues.
Implementing our models in mice
To make use of the plethora of transgenic mouse models for the identification of neural determinants of the reorientation from cocaine toward DSI, we moved our experimental paradigm from Sprague–Dawley rats to C57BL/6 mice (Kummer et al., 2014). Our findings suggest that a higher percentage of C57BL/6 mice than Sprague–Dawley rats find DSI, if tested against the i.p. saline injection alone (model 3, above), aversive. This would confer our mouse model with even greater translational power for the situation of the human addict, who often finds drug-free social interaction aversive and a considerable challenge to cope with.
Figure 2 compares the CPP induced by the strikingly different stimuli that DSI and cocaine represent. In Fig. 2, behavior is expressed both as the raw experimental value ‘time spent in the stimulus-associated compartment’ and as the difference between the time spent in the stimulus-associated compartment minus the time spent in the comparator compartment, with the comparator stimulus, that is, an i.p. saline injection, designed to be the same when investigating cocaine CPP or DSI CPP. The values are given for both mice and rats. Interestingly, group data for the raw experimental value ‘time spent in the stimulus-associated compartment’ were always normally distributed (with the exception of a single outlier animal in the cocaine group, the elimination of which led to normal distribution of the cocaine data as well; Fig. 2). Even when expressing the time spent in the respective compartments as the difference between the compartment associated with the stimulus of interest minus the time spent in the saline-associated compartment, that is, by quantifying preference or avoidance at the time of the CPP test, the data still showed a normal distribution. To emphasize, we did not find a dichotomous distribution, that is, did not identify two different populations that could be separated according to their preference or avoidance for either the cocaine or the DSI stimulus. On the contrary, the individual rodent’s CPP/avoidance behavior can be better described along a continuum of time spent in the compartment associated with the stimulus of interest. Thus, declaring CPP or aversion was simply based on the numerical difference between the time spent in the stimulus-associated compartment and the time spent in the saline-associated compartment.
At the time of our first across-genus comparison (Kummer et al., 2014), the percentages of animals that showed CPA to DSI were 15% (i.e. four of 27) for rats and 29% (i.e. 12 of 42) for mice (see Fig. 1 for the individual animals’ behavior), with both mice and rats starting the experiment at an age of 6–8 weeks and both conspecifics being of the male sex. In a subsequent study (Kummer et al., 2015), the percentage of C57BL/6N mice developing aversion to social interaction increased to 42% (five of 12). As our experimental database for C57BL/6 mice grows and the number of different experimenters increases, the overall percentage of mice that develop CPA to DSI is shifting more toward 50%. We are currently investigating this phenomenon systematically as we are aware of an experimenter effect, that is, of the experimenter–mouse interaction, on DSI CPP (Kummer et al., 2014), which we would like to control better.
The indication that a higher percentage of mice than rats find DSI aversive (rather than preferable), compared with an i.p. saline injection alone, is supported by a host of laboratory observations that suggest that rats in general, and the Sprague–Dawley strain in particular, are ‘social’ or ‘tame’ or ‘friendly,’ whereas mice in general have been observed to show considerable male–male fighting when held in animal housing facilities. With respect to the aggressiveness of different mouse strains, the picture seems less clear. For example, C57BL/6J mice have been described as a ‘prosocial’ and a ‘more prosocial’ strain compared with the ‘relatively asocial’ BALB/cJ strain (Kennedy et al., 2012). A detailed mouse interstrain comparison, however, is beyond the scope of the present review.
In their wild habitats, individuals of both the rat (Barnett, 1975) and the mouse (Dixon, 2004) genus mark and defend their territory. Both genera form hierarchies. Hierarchies formed by mice are more rudimentary than hierarchies formed by rats, and consist of a despot, a subdominant, and subordinates, both in artificial enclosures and in the wild at high population densities (Dixon, 2004). Rats are known to form colonies ‘which may number many hundreds’ (Barnett, 1975). As mentioned before, our Sprague–Dawley rats spent at least 76% of their 15 min social interaction time in direct physical contact, whereas gross observation of the C57BL/6 mice indicates that they spend much less than 10% of their social interaction time in direct physical contact, plausibly also finding it less preferable (to i.p. saline injections alone). We are currently studying this behavioral aspect in more detail.
Finally, despite all the shortcomings and caveats and open questions discussed above, we could successfully use our experimental models to answer a number of questions on DSI versus cocaine reward (Fritz et al., 2011a, 2011b, 2011c; Kummer et al., 2011, 2014, 2015; Zernig et al., 2011; El Rawas et al., 2012a, 2012b; Prast et al., 2012, 2014b) and to investigate the neural basis for the increased cocaine CPP shown by animals bred for high anxiety (Prast et al., 2014a).
Findings obtained with our experimental models
Our therapeutically most promising finding was that both the reacquisition/re-expression of cocaine conditioning and the associated EGR1 expression in the whole accumbens corridor were inhibited by a previous history of only four 15 min episodes of DSI (Fritz et al., 2011b; Prast et al., 2014b). Thus, DSI was able to inhibit both the subsequent reacquisition/re-expression of preference for cocaine and the neural activation associated with this behavior. Figure 3, which is an extraction and combination of previously published findings (Prast et al., 2014b), shows this. Our findings will now be described in detail.
Correlation between cocaine-conditioned place preference and neural activation
The time spent in the cocaine-associated conditioning compartment was correlated with the density of EGR1-activated neurons in both the core and the shell of the accumbens, provided that these accumbal subregions were located medial to the anterior commissure (Prast et al., 2014a, 2014b; see also Fig. 3). Figure 3 also shows that there was no correlation if cocaine was administered noncontingently, that is, outside the context of the CPP experiment. To phrase this differently, the direct pharmacological effects of cocaine did not show any correlation with the degree of the accumbens activation (Fig. 3).
This correlation generalized across four dimensions: (a) rodent genus, that is, Sprague–Dawley rats and CD1 mice, (b) initial acquisition/expression versus reacquistion/re-expression of cocaine CPP, (c) mice bred for normal versus high anxiety, and (d) immediate early genes, that is, EGR1 and c-Fos. Strikingly, the correlation between time spent in the cocaine-associated compartment and the density of EGR1-immunopositive or C-Fos-immunopositive neurons was not limited to the medial core (AcbCm) and medial shell (AcbShm) subregions of the nucleus accumbens, but extended to all regions medial to the anterior commissure (‘accumbens corridor’, Fig. 3), that is, included the major island of Calleja and the intermediate nucleus of the lateral septum (ICjM+LSI) as well as the vertical limb of the diagonal band and the medial septum (VDB+MS) (Prast et al., 2014a, 2014b). Table 1 summarizes our findings on the activation of the individual subregions of the accubens corridor and compares them with the findings of other groups in the field.
Table 1.
Effects of social interaction, pair housing, or environmental enrichment on cocaine conditioning-induced activation of motivational neuronetworks

The accumbens corridor cell type involved: D1-medium and D2-medium spiny neurons
As discussed in detail in Prast et al., 2014b, the immunohistochemically (Paxinos et al., 2009) and cytoarchitectonically (Franklin and Paxinos, 2007; Paxinos and Watson, 2007) diverse subregions (Zahm and Heimer, 1993) of the accumbens corridor share an important commonality: their major neuron type is a GABAergic projection neuron that predominantly expresses either dopamine D1 or D2 receptors (see Zahm et al., 2013, and other references quoted in Prast et al., 2014b). These GABAergic projection neurons have dendritic and axonal fields of 200 μm (Gerfen, 2004; Humphries et al., 2010). Therefore, their input and output fields most likely extend far beyond the subregional borders of the accumbens corridor. In the accumbens, these GABAergic projection neurons are called ‘medium spiny neurons’ (D1-MSNs and D2-MSNs); in the ICjM, they are called ‘granule cells’ (Ribak and Fallon, 1982).
The cocaine CPP-associated neuronal activation was limited to these D1-MSNs and D2-MSNs irrespective of genus (i.e. mice and rats) and irrespective of the immediate early gene (IEG) used to quantify neuronal activation (i.e. EGR1 and c-Fos) (Prast et al., 2014a, 2014b). For reasons that are not obvious to us, c-Fos expression is much more frequently used by researchers in the field than EGR1 expression to quantify neural activation. We therefore took care to quantify both EGR1 and c-Fos expression in the same brains (Prast et al., 2014a) for a direct comparison of their respective detection qualities in the hands of the same experimenters. Glial cells or GABAergic or cholinergic interneurons were not involved in the cocaine CPP-associated response (details in Prast et al., 2014a, 2014b). We are currently investigating the likely differential involvement of the presynaptic partners of D1-MSNs in mediating preference for the strikingly different rewards that cocaine and DSI represent, and aim to characterize the associated synaptic plasticity.
Resolving apparent discrepancies in the role of the nucleus accumbens core in cocaine versus dyadic social interaction preference
To re-emphasize, when we quantified neural activation by the expression of the IEGs EGR1 or c-Fos, the most striking finding was that cocaine CPP resulted in a broad activation of the accumbens corridor, that is, across a variety of seemingly diverse brain regions that, however, are all located medial to the anterior commissure (see Fig. 3 for EGR1 expression). In contrast, the lateral core (AcbCl) showed an activation/deactivation pattern that was much more reminiscent of the dorsal striatum than of the accumbens corridor (Prast et al., 2014a, 2014b). Thus, the EGR1 and c-Fos activation patterns observed immunohistochemically (Prast et al. 2014a, 2014b) strongly support the concept of a ventromedial to dorsolateral striatal gradient with respect to the expression of behavior motivated by drugs of abuse, particularly cocaine (Voorn et al., 2004; Ikemoto, 2007; Haber and Knutson, 2010). In contrast, our electrophysiologic [multielectrode array (MEA)] findings (Kummer et al., 2015, discussed below) and our lesion study (Fritz et al., 2011a) are more difficult to interpret according to this gradient concept.
Although we did find activation of the medial accumbens shell (AcbShm) after the acquisition/expression (El Rawas et al., 2012a) and after the reacquisition/re-expression of cocaine (Fritz et al., 2011b; Prast et al., 2014b), paralleling our MEA findings (see below), our findings on the contribution of the nucleus accumbens core show apparent discrepancies that, however, can be resolved if one clearly distinguishes between the nucleus accumbens core subregions that lie medial and lateral to the anterior commissure.
In our two latest immunohistochemical studies, we quantified the neural (i.e. EGR1 and c-Fos) activation of the accumbens corridor – including the medial shell and core of the accumbens – in a spatially well-defined manner, that is, in regular bins of 250 μm in the rat (Prast et al., 2014b) or 190 μm in the mouse (Prast et al., 2014a), covering the whole accumbens corridor from the medial border of the anterior commissure to the interhemispheric border at the dorsoventral level of the anterior commissure (Fig. 3). In contrast, both our previous immunohistochemical studies (Fritz et al., 2011b; El Rawas et al., 2012a, 2012b) and our previous lesion study (Fritz et al., 2011a) compared the medial accumbens shell with all core subregions around the anterior commissure, which comprise both the medial and the lateral aspects of the core (Franklin and Paxinos, 2007; Paxinos and Watson, 2007). Our MEA study, by the nature of its methodological approach, could not obtain the same degree of spatial resolution (Kummer et al., 2015).
The fact that the lateral accumbens core (AcbCl) can be considered more similar in function to the dorsal striatum than to the accumbens corridor in our behavioral paradigms may explain an apparent discrepancy in our results: although we had shown a differential effect of lesioning the medial shell (AcbShm) versus both the medial and lateral core (AcbCm+AcbCl) on the relative rewarding strength of social interaction versus cocaine, with the AcbShm mediating CPP for social interaction and the AcbCm+AcbCl cocaine CPP (Fritz et al., 2011a), we found a uniform effect on EGR1 and c-Fos activation by cocaine CPP throughout the whole accumbens corridor (Fig. 3), which comprises both the medial shell and the medial core (Prast et al., 2014a, 2014b). It is conceivable that in our previous study (Fritz et al., 2011a), lesioning the lateral core had contributed more toward the overall core lesion effect than lesioning the medial core close to the anterior commissure, creating an overall differential effect, that is, shifting the balance between CPP for social interaction versus cocaine toward social interaction, whereas lesioning of the medial shell had shifted the CPP balance toward cocaine (Fritz et al., 2011a).
Differential involvement of the accumbens corridor versus the lateral nucleus accumbens core in cocaine-oriented versus prosocially oriented behavior
In partial support of our hypothesis that the lateral nucleus accumbens core (AcbCl) may support behavior that is competing with prosocial behavior (Fritz et al., 2011a), baclofen+muscimol inactivation of the nucleus accumbens core located dorsal and dorsolateral to the anterior commissure was found to increase the duration of social play by Vanderschuren and colleagues (van Kerkhof et al., 2013). In both cases, that is, play between 5-week-male-old Wistar rats (van Kerkhof et al., 2013) or prosocial interaction between 6–8-week-old male Sprague–Dawley rats (Fritz et al., 2011a), lesioning the dorsolateral accumbens core (AcbCdl) seems to have increased the attractiveness of prosocial/agonistic social interaction. Interestingly, duration was the only aspect of play behavior that was affected by the AcbCdl inactivation. The frequency of play behavior was not affected by inactivating the AcbCdl (van Kerkhof et al., 2013), suggesting that the increased attractiveness of play (resulting in a statistically significant increase in play duration and a nonsignificant increase in nonplayful social interaction) did not translate into a more frequent emission of play behavior. This apparent discrepancy may be resolved by considering that the core is also important for invigorating many aspects of motivated behavior. For example, the Pavlovian approach and Pavlovian-to-instrumental transfer are inhibited by lesions to the core, but not the shell (see Cardinal et al., 2002 for a review), and the activity of the core may be controlled less by actual action–outcome contingencies (see Salamone and Correa, 2002 for a review, but see also Ito et al., 2004). Using an elegant experimental design that separated the effect of ‘pure’ conditioning to the stimuli of the cocaine self-administration-associated environment on c-Fos expression from the effects of lever pressing on c-Fos expression, in Sprague–Dawley rats, Neisewander et al. (2000) found that c-Fos expression in the dorsomedial shell could be accounted for by cocaine conditioning only, whereas c-Fos expression in the medial core immediately adjacent to the anterior commissure was significantly different from unconditioned rats only if both conditioning and lever pressing (motivated behavior) were considered. This finding also suggests that direct locomotor effects engage the pericommissural core more than the medial shell, the c-Fos activation of which was actually decreased when introducing the lever pressing component (see Neisewander et al., 2000, Table 1). Accordingly, cocaine-induced reinstatement of responding was inhibited by baclofen+muscimol inactivation of the pericommissural core, whereas inactivation of the ventromedial shell enhanced it (Peters et al., 2008).
Finally, although both inactivation of the AcbCdl (van Kerkhof et al., 2013) and lesions of the pericommissural AcbC (Fritz et al., 2011a) led to an increase in prosocial interaction, a yet unexplained discrepancy remains: no aspect of play behavior was affected by inactivation of the medial accumbens shell (van Kerkhof et al., 2013), whereas CPP for social interaction was decreased by AcbShm lesioning, shifting the balance in our concurrent CPP paradigm toward cocaine (Fritz et al., 2011a). In support of a functional dichotomy between the dorsolateral/circumcommissural core (mediating cocaine reward and seeking at the cost of prosocial interaction) versus the medial shell (mediating prosocial interaction at the cost of drug seeking), lesions of the medial shell (with the lesions reaching well into the medial aspects of the accumbens corridor) but not the circumcommissural/dorsal core were shown to disrupt maternal behavior, that is, pup retrieval (Li and Fleming, 2003).
Activation of the acumbens corridor versus the dorsal septum by social interaction versus cocaine CPP: immunohistochemical versus electrophysiological (MEA) results
As detailed above, our immunohistochemical experiments showed a correlation between time spent in the cocaine-associated compartment and the density of EGR1-immunopositive or C-Fos-immunopositive neurons in all regions medial to the anterior commissure, that is, in a long corridor of seemingly diverse brain regions that we termed ‘accumbens corridor’ (Prast et al. 2014a, 2014b). To remind the reader, the accumbens corridor comprises (from medial to lateral; see Fig. 3) the vertical limb of the diagonal band and the medial septum (VDB+MS), the major island of Calleja and the intermediate nucleus of the lateral septum (ICjM+LSI), the medial accumbens shell (AcbShm), and the medial core (AcbCm).
We went on to test these immunohistochemical results with an electrophysiological approach that would enable us to simultaneously quantify neuron firing in broad stretches of brain tissue by using MEA recordings of 400-μm thick coronal brain slices obtained from C57BL/6N mice roughly around the same time (i.e. 30–45 min) after the start of the CPP test session as in our EGR1 (Sprague–Dawley rats, 2 h; CD1 mice, 1 h) and c-Fos (CD1 mice, 1 h) (Prast et al., 2014a, 2014b) expression studies. Place conditioning indeed increased firing of accumbal and septal neurons irrespective of the valence, that is, after both CPP and avoidance, and irrespective of reward type, that is, both for cocaine and for social interaction. Thus, both our immunohistochemical and electrophysiological experiments broadly suggest that conditioning per se – irrespective of the direction of the motivated behavior, that is, approach or avoidance, and irrespective of the nature of the underlying reward – leads to an activation of the whole accumbens corridor. Thus, both appetitive and aversive conditioning to such strikingly different stimuli as cocaine and social interaction engage the same multitude of brain regions within the accumbens corridor, necessitating a focus for future research on individual neuron types and/or microcircuits within this broad stretch of the brain.
In our MEA study, recordings were obtained from both the ventral and the dorsal septal areas (see Kummer et al., 2015, Fig. 2), encompassing, dorsally, the dorsal, ventral, and intermediate parts of the lateral septal nucleus (LSD+LSV+LSI) and, ventrally, the medial septum, the vertical limb of the diagonal band, the ventral aspect of the intermediate part of the lateral septum, and the major island of Calleja (MS+VDB+LSI+ICjM). To complicate the interpretation of our MEA findings further, the ICjM is adjacent to the medial accumbens shell (see, e.g. Prast et al., 2014b, Fig. 6). Thus, our septal MEA recordings also sampled from septal regions dorsal of the accumbens corridor (Prast et al., 2014a, 2014b). Statistically, the increase in the firing frequency by cocaine CPP was significant only in the septal nuclei, whereas the increase in firing frequency by social interaction CPP was significant only in the accumbens (i.e. the medial and lateral core and shell regions and perhaps also the ICjM) (Kummer et al., 2015). As stated above, our MEA recordings captured the spontaneous activity not only in the ICjM and the LSI, that is, accumbens corridor regions proper, but also in the LSD+LSV+LSI, regions that are located dorsal to the accumbens corridor (Fig. 3). An increase in firing frequency, however, was found for both stimuli (social interaction and cocaine) in both the septal and the accumbal region (Kummer et al., 2015). In neither the septal nor the accumbal region was there a significant difference between the firing frequency induced by the acquisition/expression cocaine CPP versus social interaction CPP, in accordance with our previous immunohistochemical EGR1 findings in the AcbShm (El Rawas et al., 2012a). A better spatial resolution, which seems out of reach with the current MEA equipment, may have yielded a more uniform pattern of neuronal firing activity in the accumbens corridor proper – or perhaps a much more diverse activation pattern than the uniform pattern that we found with our immunohistochemical approach.
Effects of social interaction, pair housing, or environmental enrichment on cocaine conditioning-induced activation of motivational neural networks
We now compare the findings of other groups on the effects of interventions of potential therapeutic benefit in the treatment of substance use disorders, that is, EE or PC, on the activation of motivational neural networks (reward pathways) by cocaine conditioning, with our own findings on the effects of DSI on the cocaine CPP-induced activation of these brain regions.
Table 1 summarizes the evidence, first by comparing the data obtained by different groups on the neural activation produced by cocaine conditioning alone in various brain regions involved in motivated behavior (Neisewander et al., 2000; Zavala et al., 2008; El Rawas et al., 2012a) and then by comparing the modulation of this cocaine conditioning-induced activation by DSI (Fritz et al., 2011b; Prast et al., 2014b), PC (Thiel et al., 2010), or EE (Solinas et al., 2008; Thiel et al., 2010; Chauvet et al., 2011). Markers for neuronal activation were the protein products of the IEGs c-Fos or EGR1, or in one study (Prast et al., 2014a), both c-Fos and EGR1 – or the mRNA of the IEG arc.
As this review focuses on the involvement of the accumbens corridor in cocaine conditioning and its modulation by potentially beneficial therapeutic interventions such as DSI, PC, or EE, we also considered the effect of all these experimental interventions on the activation/deactivation of regions providing inputs into the accumbens corridor (Table 1). For example, all accumbens corridor regions receive input from the hippocampus and – with the possible exception of the medial septum (Oades and Halliday, 1987) – the ventral tegmental area (Prast et al., 2014b). The abundance of experimental evidence renders Table 1 rather busy; it, does, however, provide an immediate overview with respect to which brain regions are activated or deactivated by conditioning to cocaine or DSI, which are activated by a variety of interventions that are of likely benefit in cocaine use disorders, which brain areas are not affected by these interventions, and which brain areas are underinvestigated.
With respect to the neural activation by cocaine conditioning alone versus the activation by motivated behavior, Neisewander and colleagues have succeeded in separating the effects of cocaine conditioning (i.e. exposure to the self-administration environment without the availability of a lever operandum) from that of lever pressing (locomotor) behavior (exposure to the self-administration environment with the opportunity to engage in lever pressing) (Neisewander et al., 2000). The same group has also obtained data on cocaine conditioning as measured in the CPP paradigm (Zavala et al., 2008). Our group could provide a direct comparison of the neural activation by CPP for cocaine versus CPP for DSI (El Rawas et al., 2012a).
For the comparison of the effects of interventions of likely therapeutic benefit – DSI, EE, or PC – on brain excitation by cocaine conditioning, we have added the effect of a detrimental personality trait, that is, anxiety, on cocaine CPP (Prast et al., 2014a): male CD1 mice bred for high anxiety-related behavior showed a more pronounced CPP for a high dose of cocaine (i.e. 15 mg/kg i.p.), as well as a higher activation of the accumbens corridor, than their normal anxiety behavior-emitting counterparts. In summary, the cocaine CPP-induced activation of the accumbens corridor has been shown to be modulated in either direction: interventions of likely therapeutic benefit decrease (inhibit) the activation, whereas anxiety, a risk factor for substance dependence (see evidence discussed in Prast et al., 2014a), increases it.
The comparison shown in Table 1 also highlights a difference between the initial acquisition/expression of CPP versus its modulation upon reacquisition/re-expression shown by our group: the initial acquisition/expression of CPP for cocaine or DSI led to the activation of a number of input regions projecting – directly or indirectly – to the accumbens corridor (El Rawas et al., 2012a), whereas these regions upstream of the accumbens corridor were not detectably activated upon reacquisition/re-expression of cocaine CPP (Fritz et al., 2011b). The other groups reviewed in the present paper tested the effects of PC or EE on the initial acquisition/expression of cocaine CPP only. Finally, we also identified the neuron types that were activated by the reacquisition/re-expression of cocaine CPP and the reversal of this activation by a previous history of DSI, that is, D1-MSNs and D2-MSNs (Prast et al., 2014b).
Conclusion
We have been able to show that drug-free DSI of young adult (6–8 week old) rodents is able to inhibit both the reacquistion/re-expression of CPP for cocaine and the associated broad activation of D1-medium and D2-medium spiny neurons in the whole accumbens corridor. We have also compared this beneficial inhibitory effect of social interaction with the effect of other potentially beneficial interventions, that is, EE or PC, on the activation of the accumbens corridor and of other brain regions involved in behavior motivated by drugs of abuse or nondrug stimuli. We thus hope to have contributed toward the neural targeting of future pharmacotherapies aimed at helping recovering addicts to reorient their behavior away from the drug of abuse toward alternative, that is, nondrug, stimuli.
Acknowledgements
The authors thank Dr Michael T. Bardo (University of Kentucky, Lexington, USA), Dr Justin R. Yates (Northern Kentucky University, Highland Heights, USA), and Dr Hermann Dietrich (Medical University of Innsbruck, Austria) for valuable information and discussions, the editors Dr Louk Vanderschuren and Dr Paul Willner for discussing a first draft of the manuscript and suggesting a number of improvements, and the anonymous reviewers of this paper for insightful comments and suggestions.
The study was funded by Austrian Science Fund grants W1206-B18 (PhD program ‘Signal Processing in Neurons’, http://www.neurospin.at, B.S.P. and G.Z.) and P26248-B24 (G.Z.).
Conflicts of interest
There are no conflicts of interest.
References
- Achterberg EJ, van Kerkhof LW, Damsteegt R, Trezza V, Vanderschuren LJ. (2015). Methylphenidate and atomoxetine inhibit social play behavior through prefrontal and subcortical limbic mechanisms in rats. J Neurosci 35:161–169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- American Psychiatric Association (2013). Diagnostic and statistical manual of mental disorders (DSM-5(R). Washington, DC: American Psychiatric Association. [Google Scholar]
- Astur RS, Carew AW, Deaton BE. (2014). Conditioned place preferences in humans using virtual reality. Behav Brain Res 267:173–177. [DOI] [PubMed] [Google Scholar]
- Bardo MT, Bevins RA. (2000). Conditioned place preference: what does it add to our preclinical understanding of drug reward? Psychopharmacology (Berl) 153:31–43. [DOI] [PubMed] [Google Scholar]
- Bardo MT, Neisewander JL, Miller JS. (1986). Repeated testing attenuates conditioned place preference with cocaine. Psychopharmacology (Berl) 89:239–243. [DOI] [PubMed] [Google Scholar]
- Bardo MT, Rowlett JK, Harris MJ. (1995). Conditioned place preference using opiate and stimulant drugs: a meta-analysis. Neurosci Biobehav Rev 19:39–51. [DOI] [PubMed] [Google Scholar]
- Bardo MT, Neisewander JL, Kelly TH. (2013). Individual differences and social influences on the neurobehavioral pharmacology of abused drugs. Pharmacol Rev 65:255–290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barnett SA. (1975). The rat: a study in behavior. Chicago: The University of Chicago Press. [Google Scholar]
- Cardinal RN, Parkinson JA, Hall J, Everitt BJ. (2002). Emotion and motivation: the role of the amygdala, ventral striatum, and prefrontal cortex. Neurosci Biobehav Rev 26:321–352. [DOI] [PubMed] [Google Scholar]
- Chauvet C, Lardeux V, Jaber M, Solinas M. (2011). Brain regions associated with the reversal of cocaine conditioned place preference by environmental enrichment. Neuroscience 184:88–96. [DOI] [PubMed] [Google Scholar]
- Childs E, de Wit H. (2009). Amphetamine-induced place preference in humans. Biol Psychiatry 65:900–904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dixon AK.Hedrich HJ, Bullock G. (2004). The social behaviour of mice and its sensory control. . The laboratory mouse. Amsterdam: Elsevier; 287–300. [Google Scholar]
- Douglas LA, Varlinskaya EI, Spear LP. (2004). Rewarding properties of social interactions in adolescent and adult male and female rats: impact of social versus isolate housing of subjects and partners. Dev Psychobiol 45:153–162. [DOI] [PubMed] [Google Scholar]
- El Rawas R, Klement S, Kummer KK, Fritz M, Dechant G, Saria A, Zernig G. (2012a). Brain regions associated with the acquisition of conditioned place preference for cocaine versus social interaction. Front Behav Neurosci 6:63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- El Rawas R, Klement S, Salti A, Fritz M, Dechant G, Saria A, Zernig G. (2012b). Preventive role of social interaction for cocaine conditioned place preference: correlation with FosB/DeltaFosB and pCREB expression in rat mesocorticolimbic areas. Front Behav Neurosci 6:8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elkashef A, Holmes TH, Bloch DA, Shoptaw S, Kampman K, Reid MS, et al. (2005). Retrospective analyses of pooled data from CREST I and CREST II trials for treatment of cocaine dependence. Addiction 100 (Suppl 1):91–101. [DOI] [PubMed] [Google Scholar]
- Epstein DH, Marrone GF, Heishman SJ, Schmittner J, Preston KL. (2010). Tobacco, cocaine, and heroin: craving and use during daily life. Addict Behav 35:318–324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Epstein DH, Willner-Reid J, Vahabzadeh M, Mezghanni M, Lin JL, Preston KL. (2009). Real-time electronic diary reports of cue exposure and mood in the hours before cocaine and heroin craving and use. Arch Gen Psychiatry 66:88–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Everitt BJ, Robbins TW. (2000). Second-order schedules of drug reinforcement in rats and monkeys: measurement of reinforcing efficacy and drug-seeking behaviour. Psychopharmacology (Berl) 153:17–30. [DOI] [PubMed] [Google Scholar]
- Franklin KBJ, Paxinos G. (2007). The mouse brain in stereotaxic coordinates. New York: Academic Press. [Google Scholar]
- Fritz M, El Rawas R, Klement S, Kummer K, Mayr MJ, Eggart V, et al. (2011a). Differential effects of accumbens core vs shell lesion in a rat concurrent conditioned place preference paradigm for cocaine vs social interaction. PloS One 6:e26761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fritz M, El Rawas R, Salti A, Klement S, Bardo MT, Kemmler G, et al. (2011b). Reversal of cocaine-conditioned place preference and mesocorticolimbic Zif268 expression by social interaction in rats. Addict Biol 16:273–284. [DOI] [PubMed] [Google Scholar]
- Fritz M, Klement S, El Rawas R, Saria A, Zernig G. (2011c). Sigma1 receptor antagonist BD1047 enhances reversal of conditioned place preference from cocaine to social interaction. Pharmacology 87:45–48. [DOI] [PubMed] [Google Scholar]
- Gerfen GR.Paxinos G. (2004). Basal ganglia. . The rat nervous system. Amsterdam: Elsevier; 455–508. [Google Scholar]
- Guzman D, Ettenberg A. (2007). Runway self-administration of intracerebroventricular cocaine: evidence of mixed positive and negative drug actions. Behav Pharmacol 18:53–60. [DOI] [PubMed] [Google Scholar]
- Haber SN, Knutson B. (2010). The reward circuit: linking primate anatomy and human imaging. Neuropsychopharmacology 35:4–26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Humphries MD, Wood R, Gurney K. (2010). Reconstructing the three-dimensional GABAergic microcircuit of the striatum. PLoS Comput Biol 6:e1001011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ikemoto S. (2007). Dopamine reward circuitry: two projection systems from the ventral midbrain to the nucleus accumbens-olfactory tubercle complex. Brain Res Rev 56:27–78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ito R, Robbins TW, Everitt BJ. (2004). Differential control over cocaine-seeking behavior by nucleus accumbens core and shell. Nat Neurosci 7:389–397. [DOI] [PubMed] [Google Scholar]
- Kennedy BC, Panksepp JB, Runckel PA, Lahvis GP. (2012). Social influences on morphine-conditioned place preference in adolescent BALB/cJ and C57BL/6J mice. Psychopharmacology (Berl) 219:923–932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kummer K, Klement S, Eggart V, Mayr MJ, Saria A, Zernig G. (2011). Conditioned place preference for social interaction in rats: contribution of sensory components. Front Behav Neurosci 5:80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kummer KK, Hofhansel L, Barwitz CM, Schardl A, Prast JM, Salti A, et al. (2014). Differences in social interaction- vs. cocaine reward in mouse vs. rat. Front Behav Neurosci 8:363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kummer KK, El Rawas R, Kress M, Saria A, Zernig G. (2015). Social interaction and cocaine conditioning in mice increase spontaneous spike frequency in the nucleus accumbens or septal nuclei as revealed by multielectrode array recordings. Pharmacology 95:42–49. [DOI] [PubMed] [Google Scholar]
- Lenoir M, Serre F, Cantin L, Ahmed SH. (2007). Intense sweetness surpasses cocaine reward. PLoS One 2:e698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li M, Fleming AS. (2003). The nucleus accumbens shell is critical for normal expression of pup-retrieval in postpartum female rats. Behav Brain Res 145:99–111. [DOI] [PubMed] [Google Scholar]
- Mattson ME, Del Boca FK, Carroll KM, Cooney NL, DiClemente CC, Donovan D, et al. (1998). Compliance with treatment and follow-up protocols in project MATCH: predictors and relationship to outcome. Alcohol Clin Exp Res 22:1328–1339. [PubMed] [Google Scholar]
- Molet M, Billiet G, Bardo MT. (2013). Conditioned place preference and aversion for music in a virtual reality environment. Behav Processes 92:31–35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neisewander JL, Baker DA, Fuchs RA, Tran-Nguyen LT, Palmer A, Marshall JF. (2000). Fos protein expression and cocaine-seeking behavior in rats after exposure to a cocaine self-administration environment. J Neurosci 20:798–805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neisewander JL, Peartree NA, Pentkowski NS. (2012). Emotional valence and context of social influences on drug abuse-related behavior in animal models of social stress and prosocial interaction. Psychopharmacology (Berl) 224:33–56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nutt DJ, King LA, Phillips LD, Independent Scientific Committee on Drugs (2010). Drug harms in the UK: a multicriteria decision analysis. Lancet 376:1558–1565. [DOI] [PubMed] [Google Scholar]
- Oades RD, Halliday GM. (1987). Ventral tegmental (A10) system: neurobiology. 1. Anatomy and connectivity. Brain Res 434:117–165. [DOI] [PubMed] [Google Scholar]
- Paxinos G, Watson C. (2007). The rat brain in stereotaxic coordinates. New York: Academic Press. [DOI] [PubMed] [Google Scholar]
- Paxinos G, Watson C, Carrive P, Kirkcaldie MTK, Ashwell K. (2009). Chemoarchitectonic atlas of the rat brain. Amsterdam: Elsevier. [Google Scholar]
- Peartree NA, Hood LE, Thiel KJ, Sanabria F, Pentkowski NS, Chandler KN, Neisewander JL. (2012). Limited physical contact through a mesh barrier is sufficient for social reward-conditioned place preference in adolescent male rats. Physiol Behav 105:749–756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peters J, LaLumiere RT, Kalivas PW. (2008). Activity in infralimbic cortex suppresses cocaine relapse. J Neurosci 28:6064–6053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pierce RC, O’Brien CP, Kenny PJ, Vanderschuren LJ. (2012). Rational development of addiction pharmacotherapies: successes, failures, and prospects. Cold Spring Harb Perspect Med 2:a012880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prast JM, Kummer KK, Barwitz CM, Humpel C, Dechant G, Zernig G. (2012). Acetylcholine, drug reward and substance use disorder treatment: intra- and interindividual striatal and accumbal neuron ensemble heterogeneity may explain apparent discrepant findings. Pharmacology 90:264–273. [DOI] [PubMed] [Google Scholar]
- Prast JM, Schardl A, Sartori SB, Singewald N, Saria A, Zernig G. (2014a). Increased conditioned place preference for cocaine in high anxiety related behavior (HAB) mice is associated with an increased activation in the accumbens corridor. Front Behav Neurosci 8:441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prast JM, Schardl A, Schwarzer C, Dechant G, Saria A, Zernig G. (2014b). Reacquisition of cocaine condtioned place preference and its inhibition by previous social interaction preferentially affect D1-medium spiny neurons in the accumbens corridor. Front Behav Neurosci 8:317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Preston KL, Vahabzadeh M, Schmittner J, Lin JL, Gorelick DA, Epstein DH. (2009). Cocaine craving and use during daily life. Psychopharmacology (Berl) 207:291–301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ribak CE, Fallon JH. (1982). The island of Calleja complex of rat basal forebrain. I. Light and electron microscopic observations. J Comp Neurol 205:207–218. [DOI] [PubMed] [Google Scholar]
- Rossi NA, Reid LD. (1976). Affective states associated with morphine injections. Physiol Psychol 4:269–274. [Google Scholar]
- Salamone JD, Correa M. (2002). Motivational views of reinforcement: implications for understanding the behavioral functions of nucleus accumbens dopamine. Behav Brain Res 137:3–25. [DOI] [PubMed] [Google Scholar]
- Solinas M, Chauvet C, Thiriet N, El Rawas R, Jaber M. (2008). Reversal of cocaine addiction by environmental enrichment. Proc Natl Acad Sci USA 105:17145–17150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spear LP. (2000). The adolescent brain and age-related behavioral manifestations. Neurosci Biobehav Rev 24:417–463. [DOI] [PubMed] [Google Scholar]
- Thiel KJ, Pentkowski NS, Peartree NA, Painter MR, Neisewander JL. (2010). Environmental living conditions introduced during forced abstinence alter cocaine-seeking behavior and Fos protein expression. Neuroscience 171:1187–1196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trezza V, Baarendse PJ, Vanderschuren LJ. (2010). The pleasures of play: pharmacological insights into social reward mechanisms. Trends Pharmacol Sci 31:463–469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trezza V, Campolongo P, Vanderschuren LJ. (2011). Evaluating the rewarding nature of social interactions in laboratory animals. Dev Cogn Neurosci 1:444–458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tzschentke TM. (2007). Measuring reward with the conditioned place preference (CPP) paradigm: update of the last decade. Addict Biol 12:227–462. [DOI] [PubMed] [Google Scholar]
- Vanderschuren LJ, Niesink RJ, Van Ree JM. (1997). The neurobiology of social play behavior in rats. Neurosci Biobehav Rev 21:309–326. [DOI] [PubMed] [Google Scholar]
- van den Brink W. (2012). Evidence-based pharmacological treatment of substance use disorders and pathological gambling. Curr Drug Abuse Rev 5:3–31. [DOI] [PubMed] [Google Scholar]
- van Kerkhof LWM, Damsteegt R, Trezza V, Gao P, Voorn P, Vanderschuren LJMJ, Saria A. (2011). Corticostriatal systems activated during social play behaviour in adolescent rats. Soc Neurosci Abs 943:20. [Google Scholar]
- van Kerkhof LW, Damsteegt R, Trezza V, Voorn P, Vanderschuren LJ. (2013). Social play behavior in adolescent rats is mediated by functional activity in medial prefrontal cortex and striatum. Neuropsychopharmacology 38:1899–1909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Voorn P, Vanderschuren LJ, Groenewegen HJ, Robbins TW, Pennartz CM. (2004). Putting a spin on the dorsal-ventral divide of the striatum. Trends Neurosci 27:468–474. [DOI] [PubMed] [Google Scholar]
- World Health Organization (1992). Tenth revision of the international classification of diseases (ICD-10). Geneva: World Health Organisation. [Google Scholar]
- Yates JR, Beckmann JS, Meyer AC, Bardo MT. (2013). Concurrent choice for social interaction and amphetamine using conditioned place preference in rats: effects of age and housing condition. Drug Alcohol Depend 129:240–246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zahm DS, Heimer L. (1993). Specificity in the efferent projections of the nucleus accumbens in the rat: comparison of the rostral pole projection patterns with those of the core and shell. J Comp Neurol 327:220–232. [DOI] [PubMed] [Google Scholar]
- Zahm DS, Parsley KP, Schwartz ZM, Cheng AY. (2013). On lateral septum-like characteristics of outputs from the accumbal hedonic ‘hotspot’ of Pecina and Berridge with commentary on the transitional nature of basal forebrain ‘boundaries’. J Comp Neurol 521:50–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zavala AR, Osredkar T, Joyce JN, Neisewander JL. (2008). Upregulation of Arc mRNA expression in the prefrontal cortex following cue-induced reinstatement of extinguished cocaine-seeking behavior. Synapse 62:421–431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zernig G, Saria A, Kurz M, O’Malley SS. (2000). Handbook of alcoholism. Boca Raton: CRC Press. [Google Scholar]
- Zernig G, Giacomuzzi S, Riemer Y, Wakonigg G, Sturm K, Saria A. (2003). Intravenous drug injection habits: drug users’ self-reports versus researchers’ perception. Pharmacology 68:49–56. [DOI] [PubMed] [Google Scholar]
- Zernig G, Ahmed SH, Cardinal RN, Morgan D, Acquas E, Foltin RW, et al. (2007). Explaining the escalation of drug use in substance dependence: models and appropriate animal laboratory tests. Pharmacology 80:65–119. [DOI] [PubMed] [Google Scholar]
- Zernig G, Fritz M, Klement S, El Rawas R, Saria A. (2011). Social interaction- vs cocaine conditioned place preference is associated with a differential activation of nucleus accumbens core cholinergic interneurons. Drug Alcohol Depend 16:273. [Google Scholar]
- Zernig G, Kummer KK, Prast JM. (2013). Dyadic social interaction as an alternative reward to cocaine. Front Psychiatry 4:100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zlebnik NE, Carroll ME. (2015). Effects of the combination of wheel running and atomoxetine on cue- and cocaine-primed reinstatement in rats selected for high or low impulsivity. Psychopharmacology (Berl) 232:1049–1059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zlebnik NE, Anker JJ, Carroll ME. (2012). Exercise to reduce the escalation of cocaine self-administration in adolescent and adult rats. Psychopharmacology (Berl) 224:387–400. [DOI] [PMC free article] [PubMed] [Google Scholar]


