Abstract
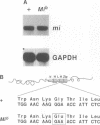
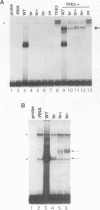
The mouse microphthalmia (mi) locus encodes a basic helix-loop-helix-leucine zipper (bHLH-Zip) transcription factor called MITF (microphthalmia transcription factor). Mutations at mi affect the development of several different cell types, including melanocytes, mast cells, osteoclasts and pigmented epithelial cells of the eye. Here we describe the phenotypic and molecular characterization of the semidominant Microphthalmia(brwnish) (Mi(b)) mutation. We show that this mutation primarily affects melanocytes and produces retinal degeneration. The mutation is a G to A transition leading to a Gly244Glu substitution in helix 2 of the HLH dimerization domain. This location is surprising since other semidominant mi mutations characterized to date have been shown to affect DNA binding or transcriptional activation domains of MITF and act as dominant negatives, while mutations that affect MITF dimerization are inherited recessively. Gel retardation assays showed that while the mutant MITF(Mi-b) protein retains its dimerization potential, it is defective in its ability to bind DNA. Computer modeling suggested that the Gly244Glu mutation might disrupt DNA binding by interfering with productive docking of the protein dimer onto DNA. The Mi(b) mutation therefore appears to dissociate a DNA recognition function of the HLH domain from its role in protein dimerization.
Full text
PDF









Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bentley N. J., Eisen T., Goding C. R. Melanocyte-specific expression of the human tyrosinase promoter: activation by the microphthalmia gene product and role of the initiator. Mol Cell Biol. 1994 Dec;14(12):7996–8006. doi: 10.1128/mcb.14.12.7996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boissy R. E., Boissy Y. L., Krakowsky J. M., Lamoreux M. L., Lingrel J. B., Nordlund J. J. Ocular pathology in mice with a transgenic insertion at the microphthalmia locus. J Submicrosc Cytol Pathol. 1993 Jul;25(3):319–332. [PubMed] [Google Scholar]
- Bok D., Hall M. O. The role of the pigment epithelium in the etiology of inherited retinal dystrophy in the rat. J Cell Biol. 1971 Jun;49(3):664–682. doi: 10.1083/jcb.49.3.664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Budd P. S., Jackson I. J. Structure of the mouse tyrosinase-related protein-2/dopachrome tautomerase (Tyrp2/Dct) gene and sequence of two novel slaty alleles. Genomics. 1995 Sep 1;29(1):35–43. doi: 10.1006/geno.1995.1212. [DOI] [PubMed] [Google Scholar]
- Casanova J. L., Pannetier C., Jaulin C., Kourilsky P. Optimal conditions for directly sequencing double-stranded PCR products with sequenase. Nucleic Acids Res. 1990 Jul 11;18(13):4028–4028. doi: 10.1093/nar/18.13.4028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Church G. M., Gilbert W. Genomic sequencing. Proc Natl Acad Sci U S A. 1984 Apr;81(7):1991–1995. doi: 10.1073/pnas.81.7.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dryja T. P., Finn J. T., Peng Y. W., McGee T. L., Berson E. L., Yau K. W. Mutations in the gene encoding the alpha subunit of the rod cGMP-gated channel in autosomal recessive retinitis pigmentosa. Proc Natl Acad Sci U S A. 1995 Oct 24;92(22):10177–10181. doi: 10.1073/pnas.92.22.10177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dryja T. P., McGee T. L., Reichel E., Hahn L. B., Cowley G. S., Yandell D. W., Sandberg M. A., Berson E. L. A point mutation of the rhodopsin gene in one form of retinitis pigmentosa. Nature. 1990 Jan 25;343(6256):364–366. doi: 10.1038/343364a0. [DOI] [PubMed] [Google Scholar]
- Farrar G. J., Kenna P., Jordan S. A., Kumar-Singh R., Humphries M. M., Sharp E. M., Sheils D. M., Humphries P. A three-base-pair deletion in the peripherin-RDS gene in one form of retinitis pigmentosa. Nature. 1991 Dec 12;354(6353):478–480. doi: 10.1038/354478a0. [DOI] [PubMed] [Google Scholar]
- Ferré-D'Amaré A. R., Pognonec P., Roeder R. G., Burley S. K. Structure and function of the b/HLH/Z domain of USF. EMBO J. 1994 Jan 1;13(1):180–189. doi: 10.1002/j.1460-2075.1994.tb06247.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferré-D'Amaré A. R., Prendergast G. C., Ziff E. B., Burley S. K. Recognition by Max of its cognate DNA through a dimeric b/HLH/Z domain. Nature. 1993 May 6;363(6424):38–45. doi: 10.1038/363038a0. [DOI] [PubMed] [Google Scholar]
- Hemesath T. J., Steingrímsson E., McGill G., Hansen M. J., Vaught J., Hodgkinson C. A., Arnheiter H., Copeland N. G., Jenkins N. A., Fisher D. E. microphthalmia, a critical factor in melanocyte development, defines a discrete transcription factor family. Genes Dev. 1994 Nov 15;8(22):2770–2780. doi: 10.1101/gad.8.22.2770. [DOI] [PubMed] [Google Scholar]
- Hodgkinson C. A., Moore K. J., Nakayama A., Steingrímsson E., Copeland N. G., Jenkins N. A., Arnheiter H. Mutations at the mouse microphthalmia locus are associated with defects in a gene encoding a novel basic-helix-loop-helix-zipper protein. Cell. 1993 Jul 30;74(2):395–404. doi: 10.1016/0092-8674(93)90429-t. [DOI] [PubMed] [Google Scholar]
- Huang S. H., Pittler S. J., Huang X., Oliveira L., Berson E. L., Dryja T. P. Autosomal recessive retinitis pigmentosa caused by mutations in the alpha subunit of rod cGMP phosphodiesterase. Nat Genet. 1995 Dec;11(4):468–471. doi: 10.1038/ng1295-468. [DOI] [PubMed] [Google Scholar]
- Hughes A. E., Newton V. E., Liu X. Z., Read A. P. A gene for Waardenburg syndrome type 2 maps close to the human homologue of the microphthalmia gene at chromosome 3p12-p14.1. Nat Genet. 1994 Aug;7(4):509–512. doi: 10.1038/ng0894-509. [DOI] [PubMed] [Google Scholar]
- Hughes M. J., Lingrel J. B., Krakowsky J. M., Anderson K. P. A helix-loop-helix transcription factor-like gene is located at the mi locus. J Biol Chem. 1993 Oct 5;268(28):20687–20690. [PubMed] [Google Scholar]
- Jackson I. J. A cDNA encoding tyrosinase-related protein maps to the brown locus in mouse. Proc Natl Acad Sci U S A. 1988 Jun;85(12):4392–4396. doi: 10.1073/pnas.85.12.4392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jenkins N. A., Copeland N. G., Taylor B. A., Lee B. K. Organization, distribution, and stability of endogenous ecotropic murine leukemia virus DNA sequences in chromosomes of Mus musculus. J Virol. 1982 Jul;43(1):26–36. doi: 10.1128/jvi.43.1.26-36.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kajiwara K., Berson E. L., Dryja T. P. Digenic retinitis pigmentosa due to mutations at the unlinked peripherin/RDS and ROM1 loci. Science. 1994 Jun 10;264(5165):1604–1608. doi: 10.1126/science.8202715. [DOI] [PubMed] [Google Scholar]
- Kajiwara K., Hahn L. B., Mukai S., Travis G. H., Berson E. L., Dryja T. P. Mutations in the human retinal degeneration slow gene in autosomal dominant retinitis pigmentosa. Nature. 1991 Dec 12;354(6353):480–483. doi: 10.1038/354480a0. [DOI] [PubMed] [Google Scholar]
- Li L. X., Turner J. E. Inherited retinal dystrophy in the RCS rat: prevention of photoreceptor degeneration by pigment epithelial cell transplantation. Exp Eye Res. 1988 Dec;47(6):911–917. doi: 10.1016/0014-4835(88)90073-5. [DOI] [PubMed] [Google Scholar]
- Liu X. Z., Newton V. E., Read A. P. Waardenburg syndrome type II: phenotypic findings and diagnostic criteria. Am J Med Genet. 1995 Jan 2;55(1):95–100. doi: 10.1002/ajmg.1320550123. [DOI] [PubMed] [Google Scholar]
- Lowings P., Yavuzer U., Goding C. R. Positive and negative elements regulate a melanocyte-specific promoter. Mol Cell Biol. 1992 Aug;12(8):3653–3662. doi: 10.1128/mcb.12.8.3653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McLaughlin M. E., Sandberg M. A., Berson E. L., Dryja T. P. Recessive mutations in the gene encoding the beta-subunit of rod phosphodiesterase in patients with retinitis pigmentosa. Nat Genet. 1993 Jun;4(2):130–134. doi: 10.1038/ng0693-130. [DOI] [PubMed] [Google Scholar]
- Moore K. J. Insight into the microphthalmia gene. Trends Genet. 1995 Nov;11(11):442–448. doi: 10.1016/s0168-9525(00)89143-x. [DOI] [PubMed] [Google Scholar]
- Packer S. O. The eye and skeletal effects of two mutant alleles at the microphthalmia locus of Mus musculus. J Exp Zool. 1967 Jun;165(1):21–45. doi: 10.1002/jez.1401650103. [DOI] [PubMed] [Google Scholar]
- Shibahara S., Taguchi H., Muller R. M., Shibata K., Cohen T., Tomita Y., Tagami H. Structural organization of the pigment cell-specific gene located at the brown locus in mouse. Its promoter activity and alternatively spliced transcript. J Biol Chem. 1991 Aug 25;266(24):15895–15901. [PubMed] [Google Scholar]
- Smith S. B., Cope B. K., McCoy J. R., McCool D. J., Defoe D. M. Reduction of phagosomes in the vitiligo (C57BL/6-mivit/mivit) mouse model of retinal degeneration. Invest Ophthalmol Vis Sci. 1994 Sep;35(10):3625–3632. [PubMed] [Google Scholar]
- Steingrímsson E., Moore K. J., Lamoreux M. L., Ferré-D'Amaré A. R., Burley S. K., Zimring D. C., Skow L. C., Hodgkinson C. A., Arnheiter H., Copeland N. G. Molecular basis of mouse microphthalmia (mi) mutations helps explain their developmental and phenotypic consequences. Nat Genet. 1994 Nov;8(3):256–263. doi: 10.1038/ng1194-256. [DOI] [PubMed] [Google Scholar]
- Tachibana M., Perez-Jurado L. A., Nakayama A., Hodgkinson C. A., Li X., Schneider M., Miki T., Fex J., Francke U., Arnheiter H. Cloning of MITF, the human homolog of the mouse microphthalmia gene and assignment to chromosome 3p14.1-p12.3. Hum Mol Genet. 1994 Apr;3(4):553–557. doi: 10.1093/hmg/3.4.553. [DOI] [PubMed] [Google Scholar]
- Takebayashi K., Chida K., Tsukamoto I., Morii E., Munakata H., Arnheiter H., Kuroki T., Kitamura Y., Nomura S. The recessive phenotype displayed by a dominant negative microphthalmia-associated transcription factor mutant is a result of impaired nucleation potential. Mol Cell Biol. 1996 Mar;16(3):1203–1211. doi: 10.1128/mcb.16.3.1203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tassabehji M., Newton V. E., Read A. P. Waardenburg syndrome type 2 caused by mutations in the human microphthalmia (MITF) gene. Nat Genet. 1994 Nov;8(3):251–255. doi: 10.1038/ng1194-251. [DOI] [PubMed] [Google Scholar]
- Tso M. O., Zhang C., Abler A. S., Chang C. J., Wong F., Chang G. Q., Lam T. T. Apoptosis leads to photoreceptor degeneration in inherited retinal dystrophy of RCS rats. Invest Ophthalmol Vis Sci. 1994 May;35(6):2693–2699. [PubMed] [Google Scholar]
- Voronova A., Baltimore D. Mutations that disrupt DNA binding and dimer formation in the E47 helix-loop-helix protein map to distinct domains. Proc Natl Acad Sci U S A. 1990 Jun;87(12):4722–4726. doi: 10.1073/pnas.87.12.4722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yasumoto K., Yokoyama K., Shibata K., Tomita Y., Shibahara S. Microphthalmia-associated transcription factor as a regulator for melanocyte-specific transcription of the human tyrosinase gene. Mol Cell Biol. 1994 Dec;14(12):8058–8070. doi: 10.1128/mcb.14.12.8058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yokoyama K., Yasumoto K., Suzuki H., Shibahara S. Cloning of the human DOPAchrome tautomerase/tyrosinase-related protein 2 gene and identification of two regulatory regions required for its pigment cell-specific expression. J Biol Chem. 1994 Oct 28;269(43):27080–27087. [PubMed] [Google Scholar]