Abstract
The injured intestine is responsible for significant morbidity and mortality after severe trauma and burn; however, targeting the intestine with therapeutics aimed at decreasing injury has proven difficult. We hypothesized that we could use intravenous phage display technology to identify peptide sequences that target the injured intestinal mucosa in a murine model, and then confirm the cross-reactivity of this peptide sequence with ex vivo human gut. Four hours following 30% TBSA burn we performed an in vivo, intravenous systemic administration of phage library containing 1012 phage in balb/c mice to biopan for gut-targeting peptides. In vivo assessment of the candidate peptide sequences identified after 4 rounds of internalization was performed by injecting 1 × 1012 copies of each selected phage clone into sham or burned animals. Internalization into the gut was assessed using quantitative polymerase chain reaction. We then incubated this gut-targeting peptide sequence with human intestine and visualized fluorescence using confocal microscopy. We identified 3 gut-targeting peptide sequences which caused collapse of the phage library (4–1: SGHQLLLNKMP, 4–5: ILANDLTAPGPR, 4–11: SFKPSGLPAQSL). Sequence 4–5 was internalized into the intestinal mucosa of burned animals 9.3-fold higher than sham animals injected with the same sequence (2.9 × 105 vs. 3.1 × 104 particles per mg tissue). Sequences 4–1 and 4–11 were both internalized into the gut, but did not demonstrate specificity for the injured mucosa. Phage sequence 4–11 demonstrated cross-reactivity with human intestine. In the future, this gut-targeting peptide sequence could serve as a platform for the delivery of biotherapeutics.
Keywords: Burn, Intestinal injury, Gut targeting, Epithelial barrier, Drug delivery
1. Introduction
Diseases of the human intestinal tract are common world-wide, resulting in numerous acute and chronic medical conditions. Conditions such as inflammatory bowel disease, severe trauma and burn injury, colitis, and cancer cause alterations in the intestinal tract which contribute to both local and systemic illness [3,11,24,25]. Directly targeting various cell types within the gut for the purpose of delivering therapeutics has been a difficult problem in the treatment of these diseases. Therapeutics aimed at either preventing intestinal injury or quickly repairing intestinal integrity after injury could represent a significant advance for researchers and clinicians.
Severe burn injury causes a profound systemic inflammatory response which predisposes these patients to distant organ injury including lung injury and acute kidney injury [16,19]. Intestinal injury is thought to serve as the source of this SIRS reaction after injury, with the spread of pro-inflammatory mediators through the mesenteric lymph that drive the inflammatory response [13,14]. We have focused on the effects of severe burn injury on intestinal barrier integrity and intestinal inflammation in a murine model of 30% total body surface area burn, finding that severe burn increases intestinal permeability, causes histologic gut injury, alters the expression of intestinal tight junction proteins, and increases gut cytokine levels [4,6,7,9]. We have demonstrated that intestinal epithelial injury occurs at early time points following injury, which would require prompt intervention to alter these burn-induced changes [8]. Our focus is on identifying therapeutics which could be deployed in the clinic setting to limit gut inflammation and the SIRS response in patients sustaining severe injury.
Phage display is a molecular technique that has the ability to identify functional targeting ligands and their receptors. Diverse peptide libraries can be displayed on the surface of the M13 bacteriophage, and allows for the screening of a large number of candidate peptide sequences. Based on the screening strategy, peptide sequences which target a specific tissue, cell, or receptor type can be identified through multiple rounds of screening [23]. Biopanning approaches utilizing phage display have been used to identify cell targeting ligands and their corresponding receptors which can be tissue or organ specific [1,18]. Phage display allows for a relatively efficient, economical screening of a diverse library of peptide sequences, enriching for only those phage clones that are recovered from the tissue of interest.
We have previously performed biopanning using in vitro phage display techniques to identify peptide sequences which bind to the intestinal epithelium after severe burn injury [12]. In this study, we hypothesize that we can perform in vivo phage display, using a phage library of 12 amino acid peptide sequences delivered parenterally, to identify peptide sequences which bind and internalize into the injured intestinal epithelium. Our goal is to identify peptide sequences which can be used to deliver therapeutics directly to the cells of the injured intestinal epithelium.
2. Materials and methods
2.1. Animal model of severe burn
Male balb/c mice (Jackson laboratory, Sacramento, CA) were placed under general anesthesia using inhaled isoflurane. Animals underwent dorsal fur clipping prior to being placed in a template estimating 30% total body surface area (TBSA). Animals underwent steam burn for 7 s, and then were given a subcutaneous injection of 1.5 ml normal saline with buprenorphine (0.05 mg/kg) in a non-burned area. Animals were allowed to recover in their cage following injury and were provided free access to food and water. These experiments were approved by the University of California Animal Subjects Committee and are in accordance with guidelines established by the National Institutes for Health.
2.2. Screening of phage library
The Ph.D-12 Phage Display Peptide Library Kit (New England Biolabs, Cambridge, MA) was used to perform in vivo phage display in our animal model of burn injury. Animals underwent injection of the phage library in 200 µl of normal saline via tail vein at 4 h following 30% TBSA burn injury and were compared to sham. One hour following injection of the phage library, animals were placed under general anesthesia with isoflurane and a segment of the distal ileum was harvested and placed in an Eppendorf tube on ice for continued analysis.
2.3. Screening of candidate peptide sequences
Candidate peptide sequences identified through multiple rounds of biopanning were studied for internalization into the intestine after intravenous administration. Injection of 1 × 1010 to 1 × 1012 phage clones bearing the candidate peptide sequence was injected in 200 µl normal saline. The dose selection was made based on a combination of the cost and the practicality of preparing the doses of targeted particles to avoid the problems associated with aggregation and precipitation that can occurs at higher concentrations. Injections were performed via tail vein in sham and animals 4 h following 30% TBSA burn. The distal ileum was harvested 1 h following injection of phage.
2.4. Extraction of phage from intestinal specimens
Segments of distal ileum were placed in a 200 µl solution containing 50 mM glucose, 25 mM Tris–HCl (pH 8), and 10 mM EDTA. The tissue was then ground for 300 s. A solution containing 400 µl of 0.2 M NaOH and 1% (w/v) SDS was added and the Eppendorf tube was rotated for 10 min at 4 °C. A 300 µl solution containing 3 M potassium acetate, 2 M acetic acid (pH 4.8) was added prior to rotation for 15 min at 4 °C. The specimens were then spun in a centrifuge at 10,000 × g for 15 min and the supernatant removed. 750 µl of propan-2-ol was added and the specimen was again centrifuged for 15 min at 10,000 × g. The supernatant was removed and the pellet was allowed to dry for 10 min. The pellet was the suspended in RNAse/DNAse free water for a final concentration of 0.1 mg/µl tissue. Specimens were frozen at −80 °C until later analysis.
2.5. Quantitative polymerase chain reaction
Phage DNA from tissue (0.5 µl) was amplified by relative PCR (Invitrogen Platinum Blue PCR Supermix) and by quantitative PCR to visualize and quantitate the number of phage particles per milligram tissue. The acid precipitated DNA solution for each sample of intestinal tissue was diluted serially 10 fold over 4 magnitudes. Quantitative PCR was performed using the Bio-Rad iQ Sybr Green Supermix (170–8880). The number of particles per milligram protein was then calculated.
2.6. Screening of human intestinal tissue
Human intestinal segments obtained from autopsy specimens (n = 3) and frozen in liquid nitrogen. Intestinal segments were cut into sections 20 µm thick using a Reichert-Jung Cryocut 1800 (Reichert Microscopes, Depew, NY). The sections were fixed in 3.7% paraformaldehyde then rinsed in 1× PBS. The segments were blocked for 30 min in 3% Bovine Serum Albumin (BSA, Sigma, St. Louis, MO) then rinsed in PBS. Intestinal sections were incubated with phage bearing the candidate peptide sequence (1E + 10 ml−1 in 1% BSA) overnight at 4 °C. Sections were rinsed in PBS then incubated for 1 h with mouse anti-M13 antibody (Abcam, Cambridge, MA; ab6188) in 1% BSA at room temperature. After rinsing in PBS, sections were incubated with Alexa fluor 488 anti-mouse antibody (Invitrogen, Carlsbad, CA; A11029) in 1 BSA for 1 h at room temperature. Sections were rinsed and then incubated with 100 µl DAPI for 30 s. Sections were rinsed in PBS prior to the addition of Slow Fade. Images were viewed using an Olympus Fluoview FV1000 laser scanning confocal microscope.
2.7. Statistical analysis
Data is expressed as the mean ± the standard error of the mean (SEM). The statistical significance among groups f was determined using t-test or analysis of variance (ANOVA) with Bonferroni correction where appropriate. Statistical significance was defined as a p value <0.05.
3. Results
3.1. Intravenous phage displays identifies peptide sequences which internalize into the gut epithelium
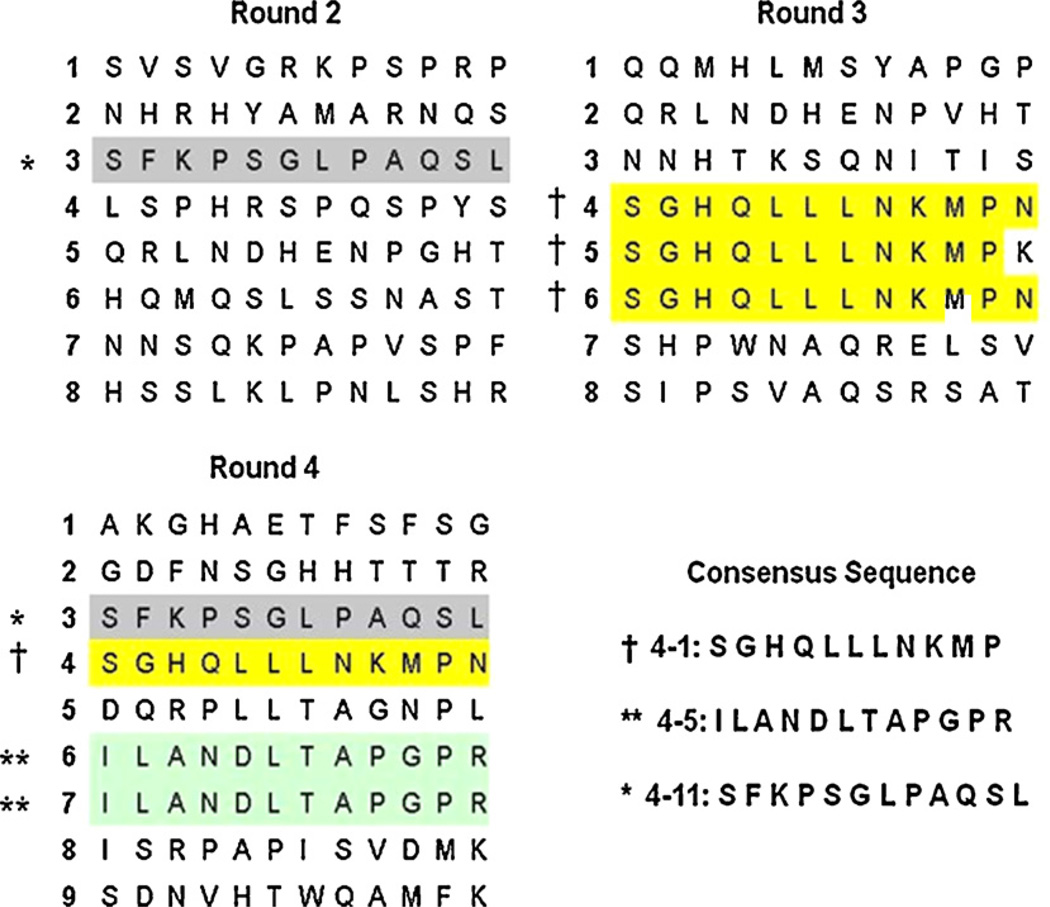
Four hours following 30% TBSA burn we performed in vivo, intravenous systemic administration of a phage library to biopan for gut barrier targeting peptides. A total of 4 rounds of panning were performed in order to enrich for our pool of candidate intestinal mucosa targeting clones. We successfully identified 3 gut targeting peptide sequences (4–1, 4–5, and 4–11) which caused collapse of the library, meaning the same sequence was identified in multiple rounds of screening. DNA sequencing of the recovered phage clones was performed on a small number of clones in order to identify the peptide sequences which were internalized into the intestinal epithelium following burn (Fig. 1). Sequences 4–1, 4–5, and 4–11, which caused collapse of the phage library, were chosen for further in vivo testing.
Fig. 1.
Candidate gut barrier targeting peptide sequences identified during in vivo, intravenous phage screening. DNA sequencing of the recovered phage clones was performed on a small number of clones in order to identify the peptide sequences that were internalized into the intestinal epithelium following burn. Highlighted sequences are found in multiple phage clones recovered from the gut epithelium. (For interpretation of the references to color in this figure legend, the reader is referred to the web version of the article.)
3.2. Gut targeting peptides identified through IV phage screening are internalized into the injured intestine
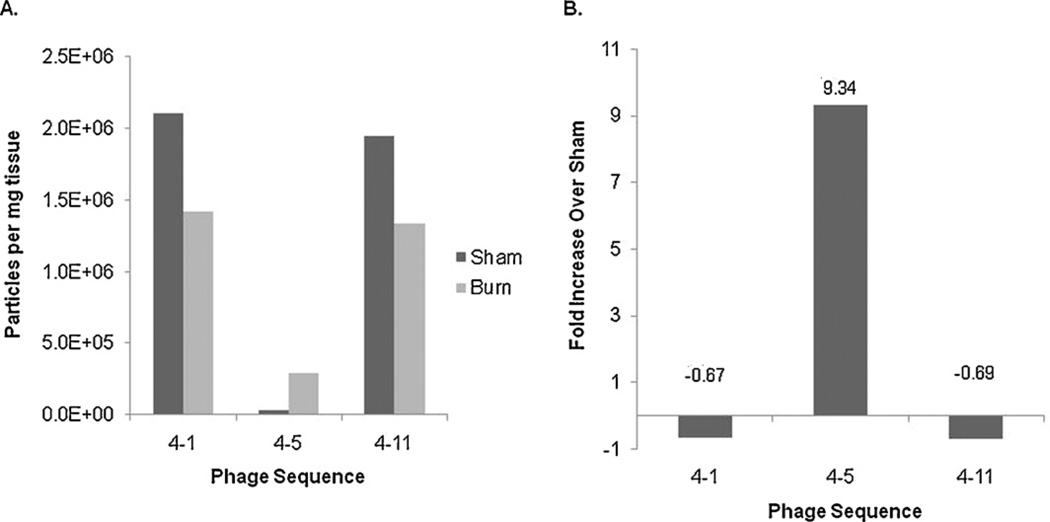
Candidate peptide sequences identified during our intravenous phage screening were tested in vivo following severe burn. Each candidate peptide sequence was administered intravenously 4 h following severe burn to determine its ability to internalize into the injured gut epithelium. We have previously demonstrated that severe burn injury causes increased histologic gut injury and intestinal barrier injury at this time-point [6,7,10]. Here, we were interested in assessing not only whether each candidate peptide sequence had the ability to internalize into the gut, but also whether each sequence preferentially internalized into the injured versus normal intestine. Phage expressing each of the candidate peptide sequences of interest was recovered for the intestine in both sham and burn animals to screen for sequences which would demonstrate tropism for the burn-injured gut (Fig. 2A). The degree of phage recovery varied for each clone, with sequence 4–1 and 4–11 internalizing into the gut to a greater degree than sequence 4–5. Sequence 4–5 did demonstrate evidence of internalizing preferentially into the injured intestine, with a 9.3-fold increase in recovery from the gut of burn injured animals compared to sham (Fig. 2B).
Fig. 2.
Candidate peptide sequences were screened in vivo following severe burn to identify sequences which demonstrate burn specificity. An injection containing 1 × 1012 copies of each selected clone were injected via tail vein into sham or animals 4 h following burn. A total of 3 different peptide sequences (4–1, 4–5, and 4–11) were tested based on results from in vivo biopanning. (A) The amount of phage bearing each candidate peptide sequence that was recovered from the intestine was quantified. Data is expressed as phage particles per mg of intestinal tissue. (B) The relative quantity of phage displaying each candidate peptide sequence that was recovered from burn-injured animals compared to sham was measured.
3.3. The ability to deliver phage to the injured gut is dose dependent
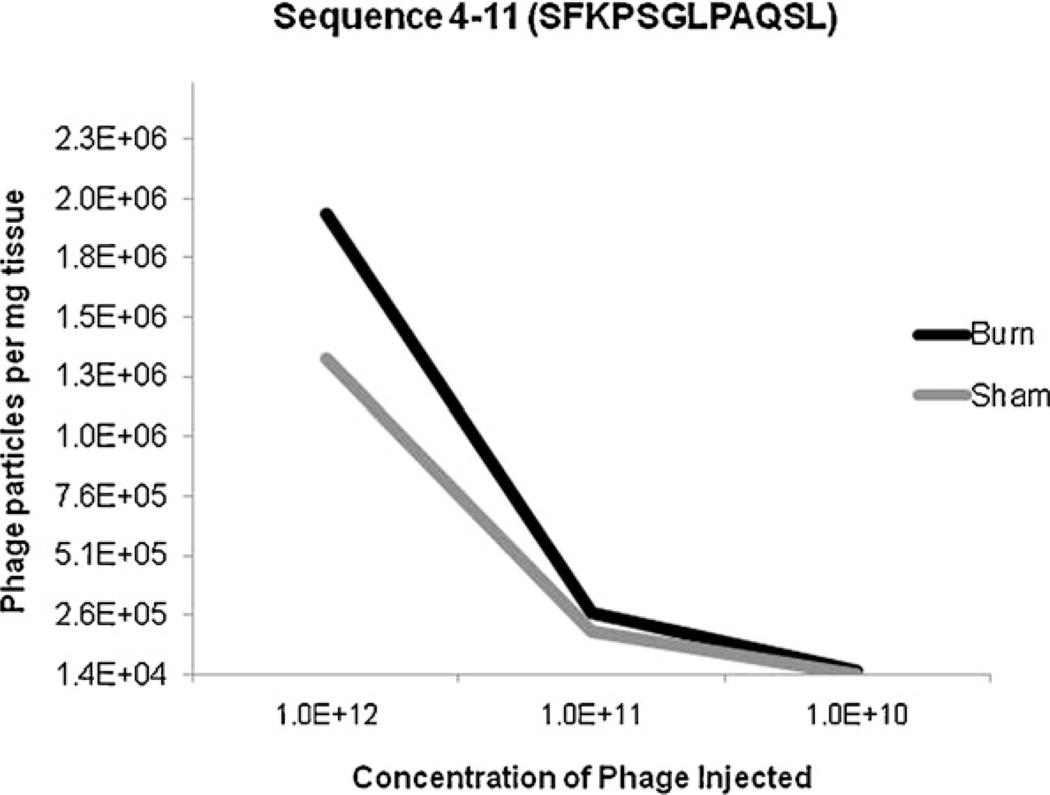
Clinically, it would be ideal to identify peptide sequences which preferentially bind to epitopes displayed by the burn injured gut. The pharmacokinetics and the concentration of phage injected can have a significant effect on phage pharmacokinetics. We injected increasing concentrations of phage expressing sequence 4–11 to determine if we could maximize phage targeting to the burn-injured gut. There is a linear relationship between concentration of phage injected and the amount of phage internalization into the gut. There is a concentration dependent difference in phage internalization into the gut of both sham and burn injured animals (Fig. 3). There is increased internalization of sequence 4–11 at the highest concentration of phage injected in the burn injured animal compared to sham, which may reflect increased tropism for this sequence in the injured gut.
Fig. 3.
Therapeutic window for targeting the burn-injured intestine. Increasing concentrations of phage expressing sequence 4–11 injected intravenously into sham (gray line) and burned animals (black line). For sequence 4–11, the greatest therapeutic window for recovery in the gut between sham and burned animals is seen when 1 × 1012 phage are injected.
3.4. Phage can be targeted preferentially internalize into the burn-injured intestinal epithelium
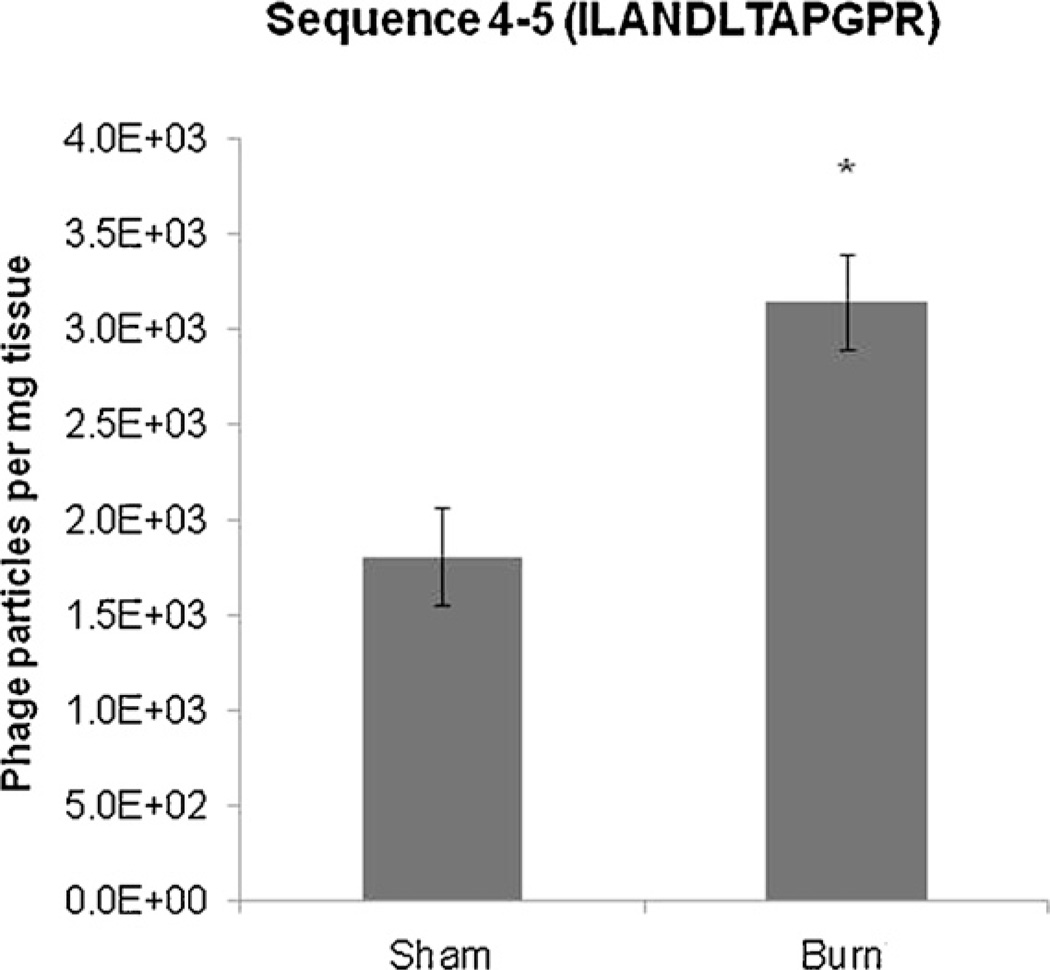
Sequence 4–5 was identified during in vivo screening as a candidate sequence which demonstrated specificity for the burn-injured gut. Phage bearing candidate peptide sequence 4–5 was tested in both sham and animals 4 h following burn injury to confirm our preliminary screening results. Candidate sequence 4–5 (ILANDLTAPGPR) was internalized into the intestinal mucosa of burned animals higher than sham animals injected with the same sequence (Fig. 4).
Fig. 4.
Phage sequences can be designed to internalize into burn injured intestine tissue. Quantitative PCR of intestinal tissue harvested from animals 4 h after burn injury following intravenous injection of phage. Candidate peptide sequence 4–5 (ILANDLTAPGPR) targets the burn-injured gut to a greater degree compared to sham. *p < 0.05, t-test.
3.5. Candidate peptide sequences identified through IV phage display using a murine model exhibit cross-reactivity with human intestinal tissue
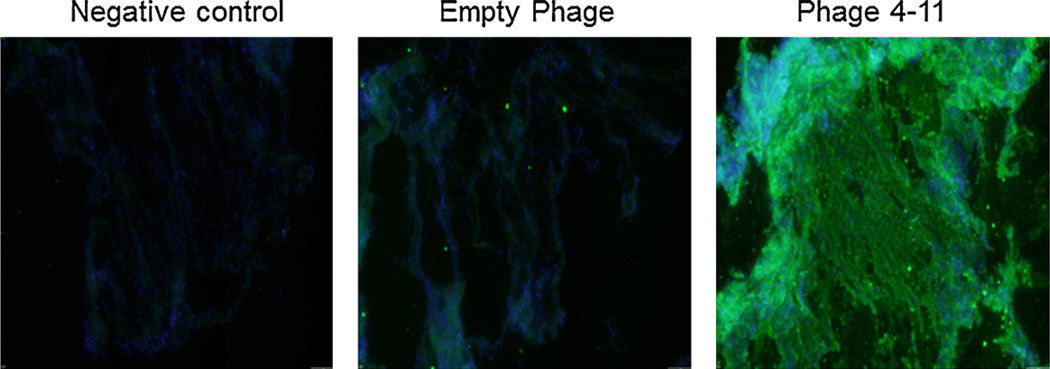
To demonstrate that peptides sequences identified through IV phage screening in a murine model will have translational potential in human intestine, we assessed the ability of our candidate peptide sequence to bind sections of human gut post-mortem. Sections of resected human small intestine were fixed and incubated with phage displaying candidate sequence 4–11 and imaged using immunofluorescence microscopy (Fig. 5). A section of gut was stained with the secondary antibody (anti-M13) and the fluorescent antibody (Alexa Fluor 488) to ensure that there was not any non-specific binding of those antibodies to the intestinal tissue (Negative Control). Sections of human gut incubated with phage containing no peptide sequence (Empty Phage) were also used as a control. Phage containing sequence 4–11 binds to human intestine to a greater degree than control. These results suggest that the candidate gut targeting peptide sequence identified in our murine model of burn could have similar effects in the human gut.
Fig. 5.
Peptide sequences identified using phage display in our murine model also bind to human gut tissue. Human small intestine was incubated with phage sequence 4–11 (SFKPSGLPAQSL) then stained with fluorescent antibodies (Alexa 488, green) for visualization using confocal microscopy. Sections of human gut incubated with the secondary and fluorescent antibodies alone (Negative Control) and with the phage containing no peptide sequence (Empty Phage) were used as controls. The binding of our candidate peptide sequence suggests that there is cross-reactivity between human tissue and peptide sequence identified through phage screening in a murine model. (For interpretation of the references to color in this figure legend, the reader is referred to the web version of the article.)
4. Discussion
The intestinal epithelium plays an important role in regulating host defense by maintaining an intact barrier which protects the gut from bacteria and potentially deleterious luminal contents. Loss of intestinal barrier function and subsequent intestinal inflammation has been implicated in disease processes including inflammatory bowel disease and severe trauma/burn. It is recognized that the injured gut may serve as a driving force behind the systemic inflammatory response that is seen following shock [21]. Therefore, effectively targeting the injured intestinal epithelium with therapeutics designed to limit or prevent gut inflammation could have clinical utility in a number of pathologic conditions.
In this series of experiment, we utilized a phage library displaying 12 amino acid peptide sequences to biopan for sequences, which when administered intravenously following burn, would internalize into the intestinal epithelium. Using an in vivo model, our phage screening strategy identified 3 unique candidate peptide sequences which were internalized into the gut after intravenous administration. One of those sequences (4–5, ILANDLTAPGPR) was recovered from the injured gut to a significantly greater degree than from the intestine of sham animals. Identifying sequences which specifically target the injured intestine could have important therapeutic implications. This could allow for better targeting of therapeutics to only those areas of the gut that have been damaged, and possibly limit the potential side effects and toxicities that could occur from delivery of a therapeutic to normal tissue. This could be especially relevant in the gut where injury tends to occur in a patchy distribution, with intervening segments of normal tissue.
Phage display of peptide sequences has wide-ranging applications for identifying therapeutic targets in humans. Phage display has previously been used to prevent angiogenesis in cancer cells by targeting tumor vasculature with TNF-α [26]. Phage display has also been used to identify antibodies which target specific receptors in mice, with cross-reactivity documented between the human and mouse sequence [27]. To assess the translational potential of gut-targeting peptide sequences identified in a murine model for cross-reactivity with human tissues, we incubated sections of human intestine with fluorescent-labeled Phage bearing a candidate peptide sequence identified through screening of the peptide library. Immunofluorescence microscopy was used to image the human gut, with greater binding of the peptide sequence compared to controls. This finding suggests that peptide targets identified from murine phage screening may have cross-reactivity in human tissue. This highlights the translational potential of further studies in vivo studies using this animal model.
Oral administration of therapeutics targeting the GI tract is often ideal. The enteral route of administration must take into account the area of the GI tract targeted, including the effect of pH and the presence of digestive enzymes which may affect bioavailability [22]. Enteral administration can be problematic in the presence of intestinal pathology. Intestinal pathophysiology such as impaired blood flow, decreased absorption, and increased barrier permeability can negatively affect the delivery of therapeutics to the intestine. For this reason, we designed our phage screening protocol to identify peptide sequences which target the intestinal epithelium after intravenous administration of the phage library. Parenteral administration of gut-targeting biotherapeutics could be ideal in the setting of severe trauma or burn injury, where intestinal injury is known to occur at early time-points following injury. Targeted therapy, delivered directly to cells of the GI tract, could be given parenterally during the initial resuscitation phase of treatment. Delivery of therapeutics to the gut in the early period post-injury could prevent or limit intestinal inflammation which drives the later systemic inflammatory response. The peptide sequences identified here could serve as a platform to deliver any number of possible therapies to the gut; including medications, growth factors, targeted antibodies, microspheres, or nanoparticles [2,5,20,27].
The peptide sequences that were identified using our phage screening strategy were only a sample of the clones that were recovered during biopanning. In further studies we plan to characterize more of the peptides sequences which cause collapse of the phage library. It is quite possible that we may identify sequences in our ongoing characterization of these phage clones which demonstrate improved specificity to the injured intestine. Prior studies have demonstrated the ability to identify peptide sequences which target the normal intestinal barrier in an animal model [15,17].
5. Conclusion
The results demonstrated here show that characterizing gut-targeting peptide sequences through intravenous phage display is feasible, identifying sequences which are internalized into both the normal and injured intestine. Importantly, we have demonstrated that gut-targeting peptide sequence identified using phage screening in a murine model, have cross reactivity with human intestinal tissue. In the future, peptide sequences identified through phage display may serve as a platform to deliver targeted therapies which limit intestinal inflammation and injury.
References
- 1.Baird A, Eliceiri BP, Gonzalez AM, Johanson CE, Leadbeater W, Stopa EG. Targeting the choroid plexus-CSF-brain nexus using peptides identified by phage display. Methods Mol Biol. 2011;686:483–498. doi: 10.1007/978-1-60761-938-3_25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Barrett KE. Building better bugs to deliver biologics in intestinal inflammation. Gut. 2010;59:427–428. doi: 10.1136/gut.2009.195016. [DOI] [PubMed] [Google Scholar]
- 3.Boman BM, Huang E. Human colon cancer stem cells: a new paradigm in gastrointestinal oncology. J Clin Oncol. 2008;26:2828–2838. doi: 10.1200/JCO.2008.17.6941. [DOI] [PubMed] [Google Scholar]
- 4.Chen C, Wang P, Su Q, Wang S, Wang F. Myosin light chain kinase mediates intestinal barrier disruption following burn injury. PLoS One. 2012;7:e34946. doi: 10.1371/journal.pone.0034946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Clark JA, Doelle SM, Halpern MD, Saunders TA, Holubec H, Dvorak K, et al. Intestinal barrier failure during experimental necrotizing enterocolitis: protective effect of EGF treatment. Am J Physiol Gastrointest Liver Physiol. 2006;291:G938–G949. doi: 10.1152/ajpgi.00090.2006. [DOI] [PubMed] [Google Scholar]
- 6.Costantini TW, Bansal V, Krzyzaniak M, Putnam JG, Peterson CY, Loomis WH, et al. Vagal nerve stimulation protects against burn-induced intestinal injury through activation of enteric glia cells. Am J Physiol Gastrointest Liver Physiol. 2010;299:G1308–G1318. doi: 10.1152/ajpgi.00156.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Costantini TW, Bansal V, Peterson CY, Loomis WH, Putnam JG, Rankin F, et al. Efferent vagal nerve stimulation attenuates gut barrier injury after burn: modulation of intestinal occludin expression. J Trauma. 2010;68:1349–1354. doi: 10.1097/TA.0b013e3181dccea0. discussion 54–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Costantini TW, Eliceiri BP, Peterson CY, Loomis WH, Putnam JG, Baird A, et al. Quantitative assessment of intestinal injury using a novel in vivo, near-infrared imaging technique. Mol Imaging. 2010;9:30–39. [PMC free article] [PubMed] [Google Scholar]
- 9.Costantini TW, Krzyzaniak M, Cheadle GA, Putnam JG, Hageny AM, Lopez N, et al. Targeting alpha-7 nicotinic acetylcholine receptor in the enteric nervous system: a cholinergic agonist prevents gut barrier failure after severe burn injury. Am J Pathol. 2012;181:478–486. doi: 10.1016/j.ajpath.2012.04.005. [DOI] [PubMed] [Google Scholar]
- 10.Costantini TW, Loomis WH, Putnam JG, Drusinsky D, Deree J, Choi S, et al. Burn-induced gut barrier injury is attenuated by phosphodiesterase inhibition: effects on tight junction structural proteins. Shock. 2009;31:416–422. doi: 10.1097/SHK.0b013e3181863080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Costantini TW, Peterson CY, Kroll L, Loomis WH, Putnam JG, Wolf P, et al. Burns, inflammation, and intestinal injury: protective effects of an anti-inflammatory resuscitation strategy. J Trauma. 2009;67:1162–1168. doi: 10.1097/TA.0b013e3181ba3577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Costantini TW, Putnam JG, Sawada R, Baird A, Loomis WH, Eliceiri BP, et al. Targeting the gut barrier: identification of a homing peptide sequence for delivery into the injured intestinal epithelial cell. Surgery. 2009;146:206–212. doi: 10.1016/j.surg.2009.05.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Deitch EA, Shi HP, Lu Q, Feketeova E, Skurnick J, Xu DZ. Mesenteric lymph from burned rats induces endothelial cell injury and activates neutrophils. Crit Care Med. 2004;32:533–538. doi: 10.1097/01.CCM.0000109773.00644.F4. [DOI] [PubMed] [Google Scholar]
- 14.Deitch EA, Xu D, Kaise VL. Role of the gut in the development of injury- and shock induced SIRS and MODS: the gut-lymph hypothesis, a review. Front Biosci. 2006;11:520–528. doi: 10.2741/1816. [DOI] [PubMed] [Google Scholar]
- 15.Duerr DM, White SJ, Schluesener HJ. Identification of peptide sequences that induce the transport of phage across the gastrointestinal mucosal barrier. J Virol Methods. 2004;116:177–180. doi: 10.1016/j.jviromet.2003.11.012. [DOI] [PubMed] [Google Scholar]
- 16.Jeschke MG, Gauglitz GG, Kulp GA, Finnerty CC, Williams FN, Kraft R, et al. Long-term persistance of the pathophysiologic response to severe burn injury. PLoS One. 2011;6:e21245. doi: 10.1371/journal.pone.0021245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kang SK, Woo JH, Kim MK, Woo SS, Choi JH, Lee HG, et al. Identification of a peptide sequence that improves transport of macromolecules across the intestinal mucosal barrier targeting goblet cells. J Biotechnol. 2008;135:210–216. doi: 10.1016/j.jbiotec.2008.01.021. [DOI] [PubMed] [Google Scholar]
- 18.Kolonin MG, Sun J, Do KA, Vidal CI, Ji Y, Baggerly KA, et al. Synchronous selection of homing peptides for multiple tissues by in vivo phage display. FASEB J. 2006;20:979–981. doi: 10.1096/fj.05-5186fje. [DOI] [PubMed] [Google Scholar]
- 19.Kraft R, Herndon DN, Al-Mousawi AM, Williams FN, Finnerty CC, Jeschke MG. Burn size and survival probability in paediatric patients in modern burn care: a prospective observational cohort study. Lancet. 2012;379:1013–1021. doi: 10.1016/S0140-6736(11)61345-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Makhlof A, Tozuka Y, Takeuchi H. pH-Sensitive nanospheres for colon-specific drug delivery in experimentally-induced colitis rat model. Eur J Pharm Biopharm. 2009 doi: 10.1016/j.ejpb.2008.12.013. [DOI] [PubMed] [Google Scholar]
- 21.Masuno T, Moore EE, Cheng AM, Sarin EL, Banerjee A. Bioactivity of postshock mesenteric lymph depends on the depth and duration of hemorrhagic shock. Shock. 2006;26:285–289. doi: 10.1097/01.shk.0000223132.72135.52. [DOI] [PubMed] [Google Scholar]
- 22.Pinto JF. Site-specific drug delivery systems within the gastro-intestinal tract: from the mouth to the colon. Int J Pharm. 2010;395:44–52. doi: 10.1016/j.ijpharm.2010.05.003. [DOI] [PubMed] [Google Scholar]
- 23.Sergeeva A, Kolonin MG, Molldrem JJ, Pasqualini R, Arap W. Display technologies: application for the discovery of drug and gene delivery agents. Adv Drug Deliv Rev. 2006;58:1622–1654. doi: 10.1016/j.addr.2006.09.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Shen L, Su L, Turner JR. Mechanisms and functional implications of intestinal barrier defects. Dig Dis. 2009;27:443–449. doi: 10.1159/000233282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Su L, Shen L, Clayburgh DR, Nalle SC, Sullivan EA, Meddings JB, et al. Targeted epithelial tight junction dysfunction causes immune activation and contributes to development of experimental colitis. Gastroenterology. 2009;136:551–563. doi: 10.1053/j.gastro.2008.10.081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Tandle A, Hanna E, Lorang D, Hajitou A, Moya CA, Pasqualini R, et al. Tumor vasculature-targeted delivery of tumor necrosis factor-alpha. Cancer. 2009;115:128–139. doi: 10.1002/cncr.24001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Wu Y, Cain-Hom C, Choy L, Hagenbeek TJ, de Leon GP, Chen Y, et al. Therapeutic antibody targeting of individual Notch receptors. Nature. 2010;464:1052–1057. doi: 10.1038/nature08878. [DOI] [PubMed] [Google Scholar]