Abstract
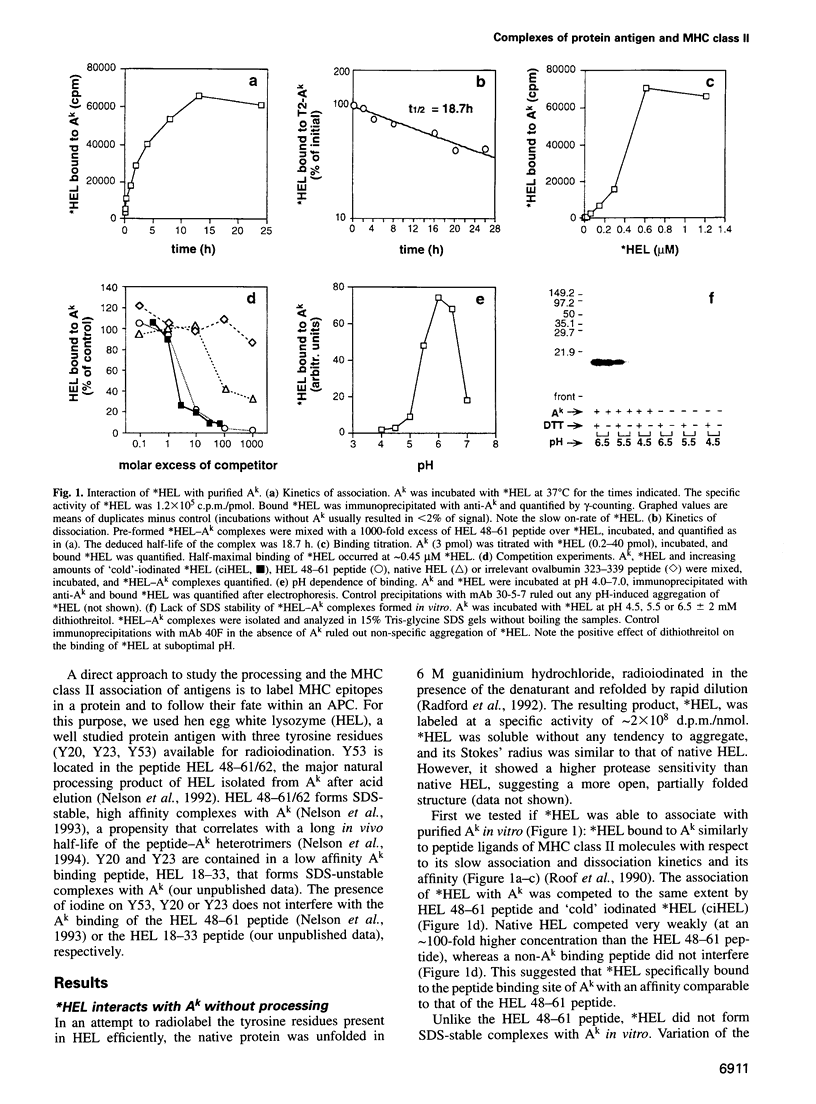
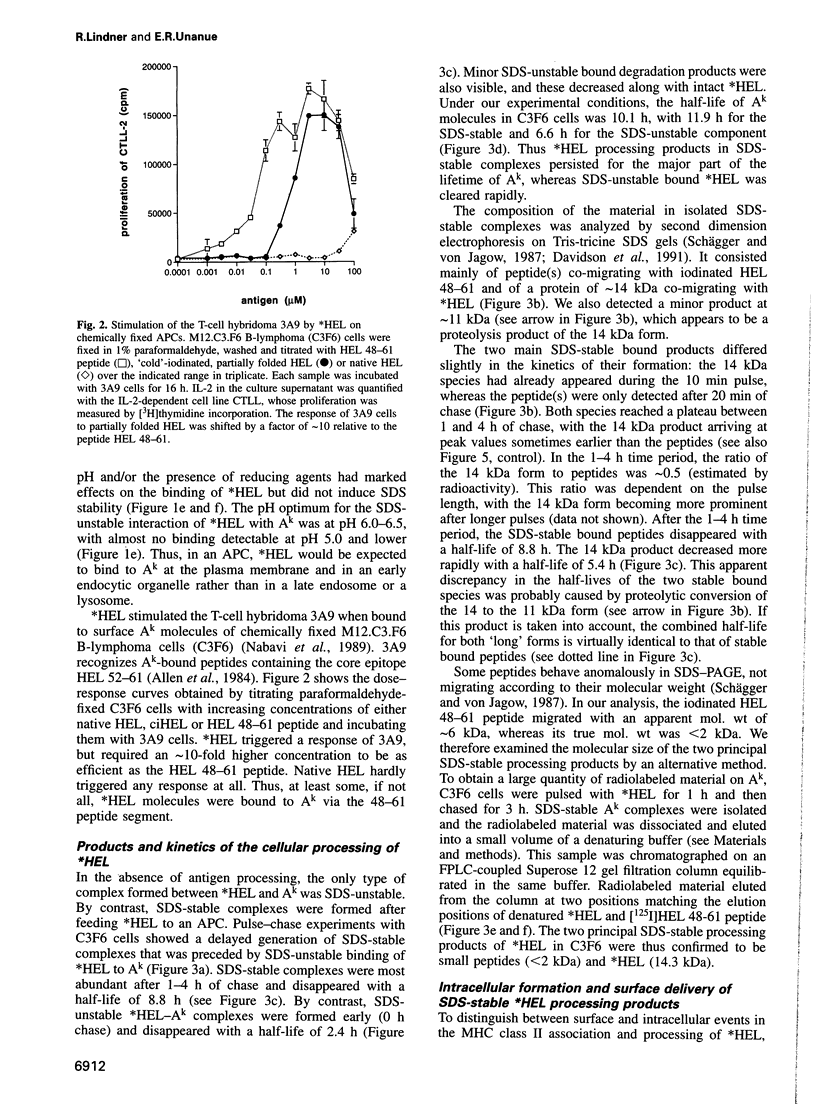
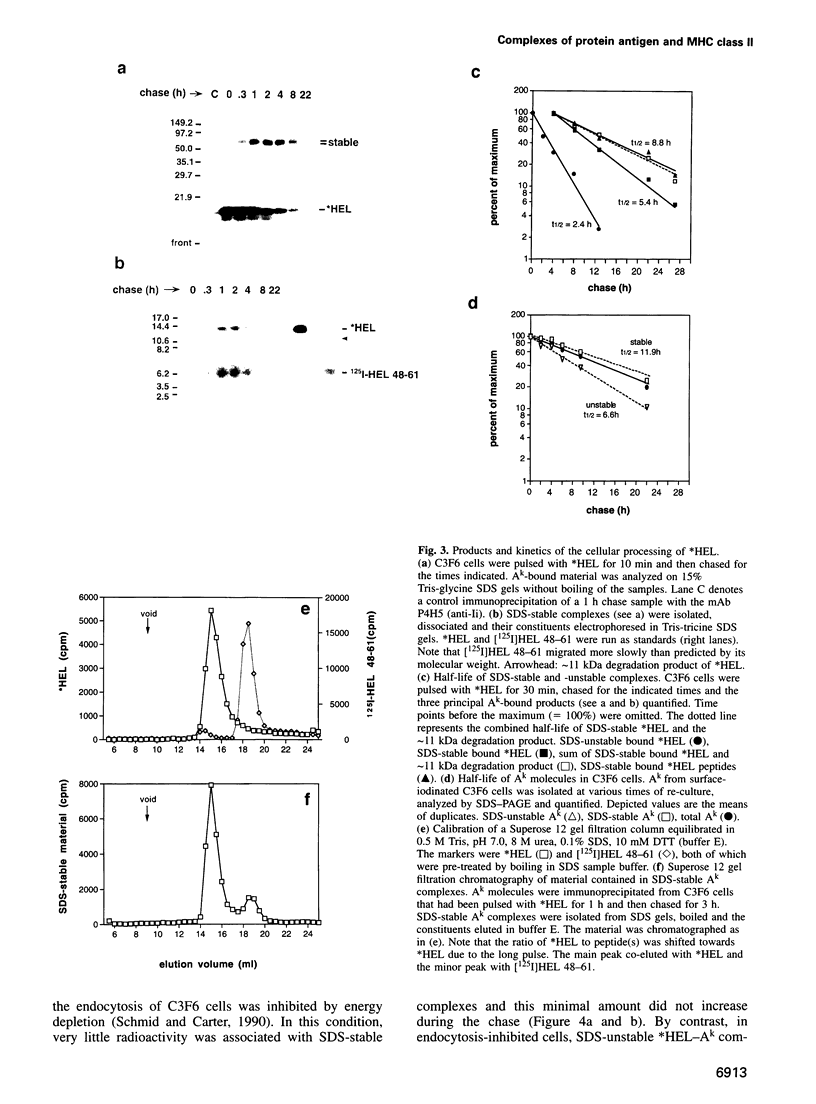
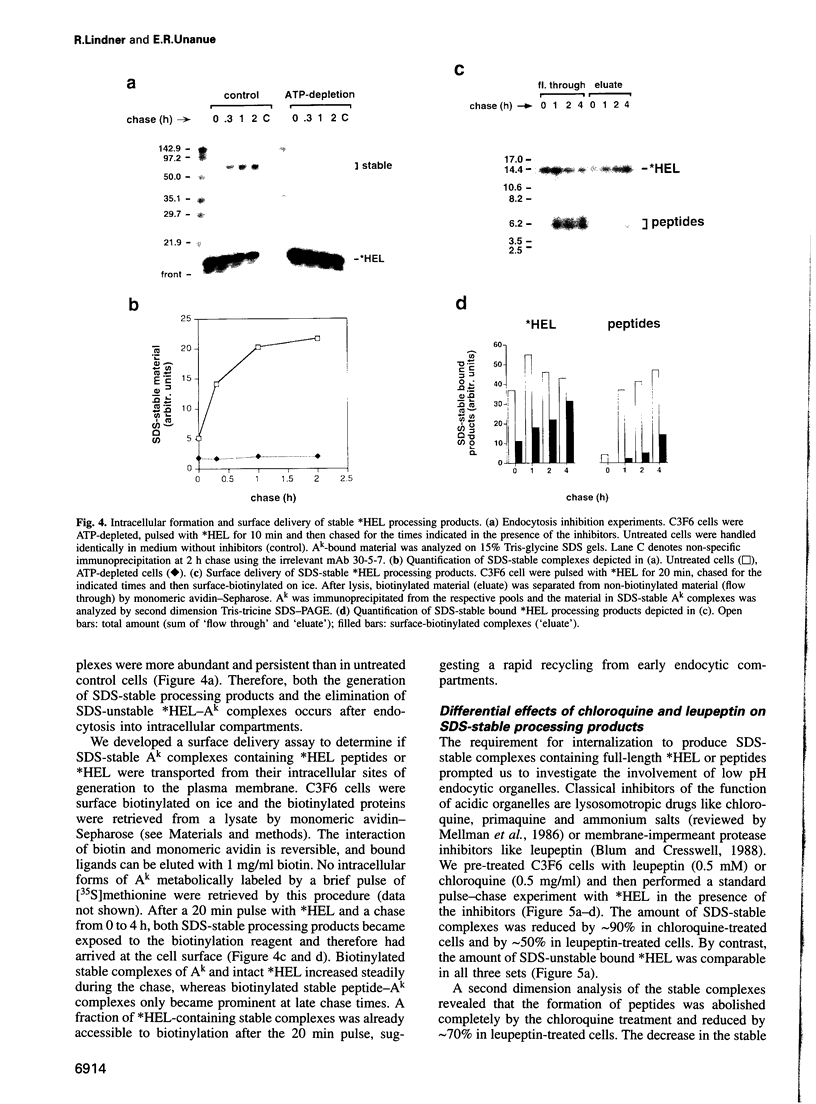
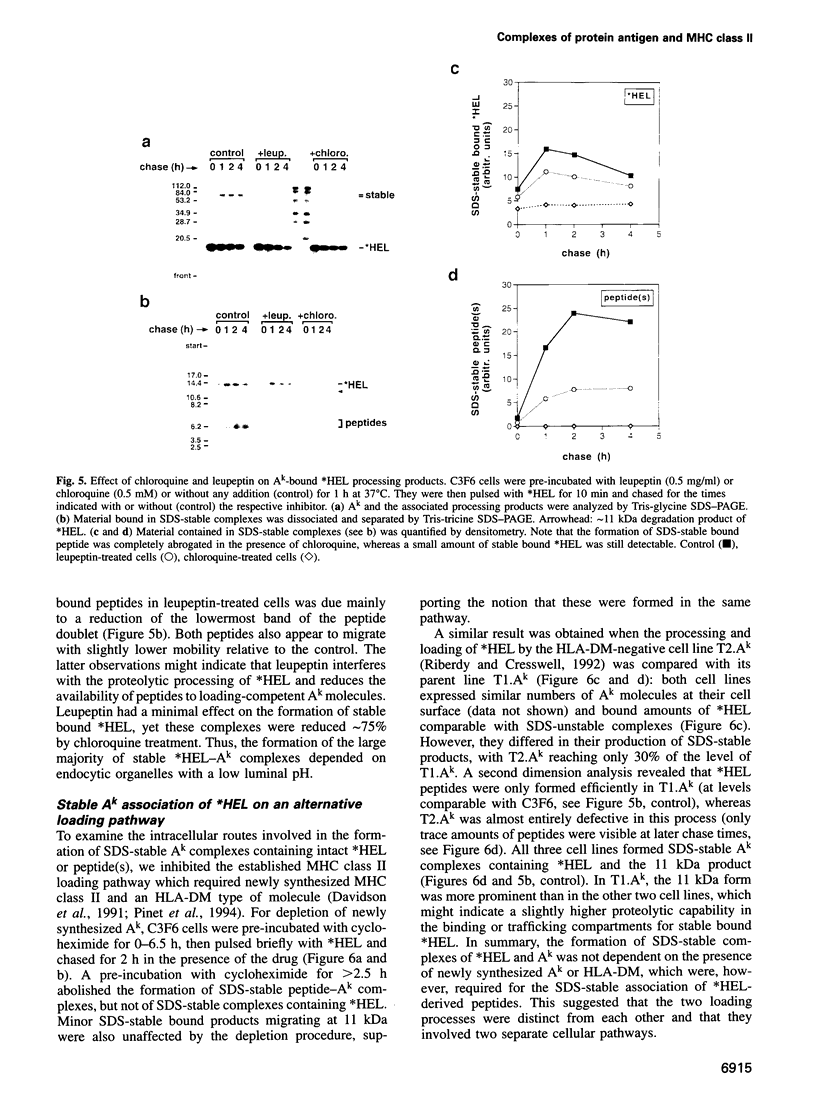
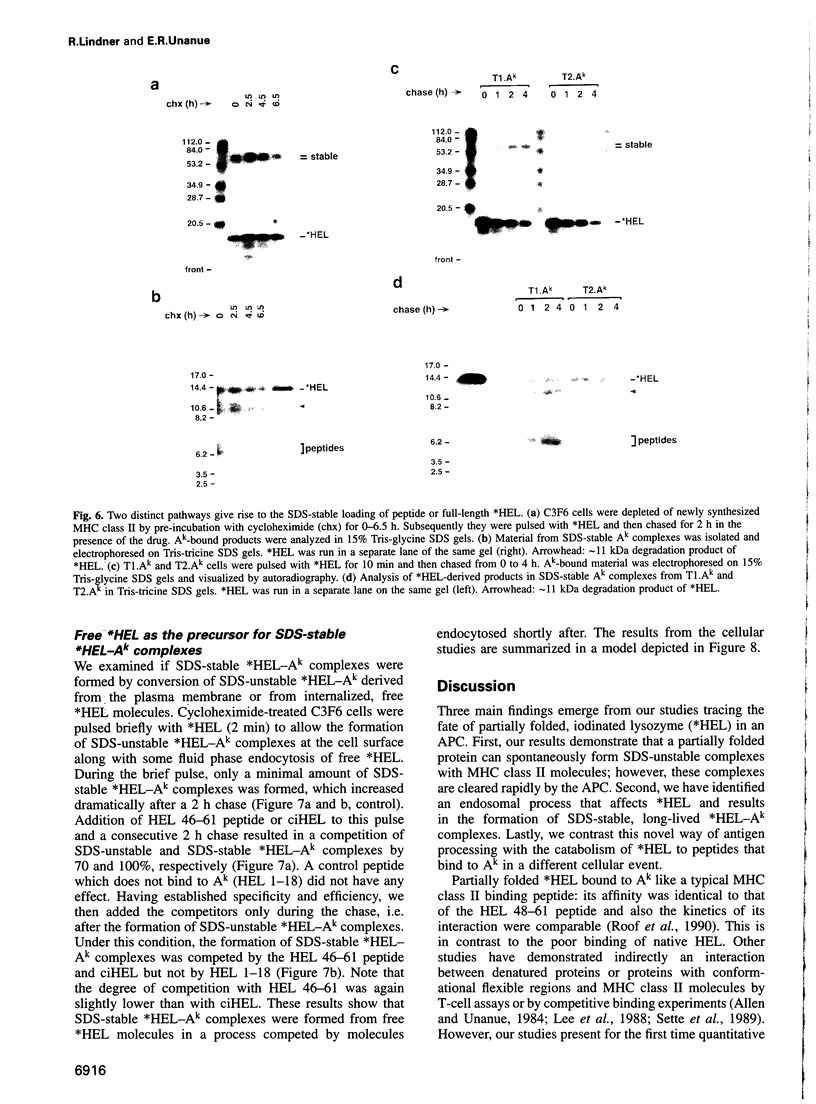
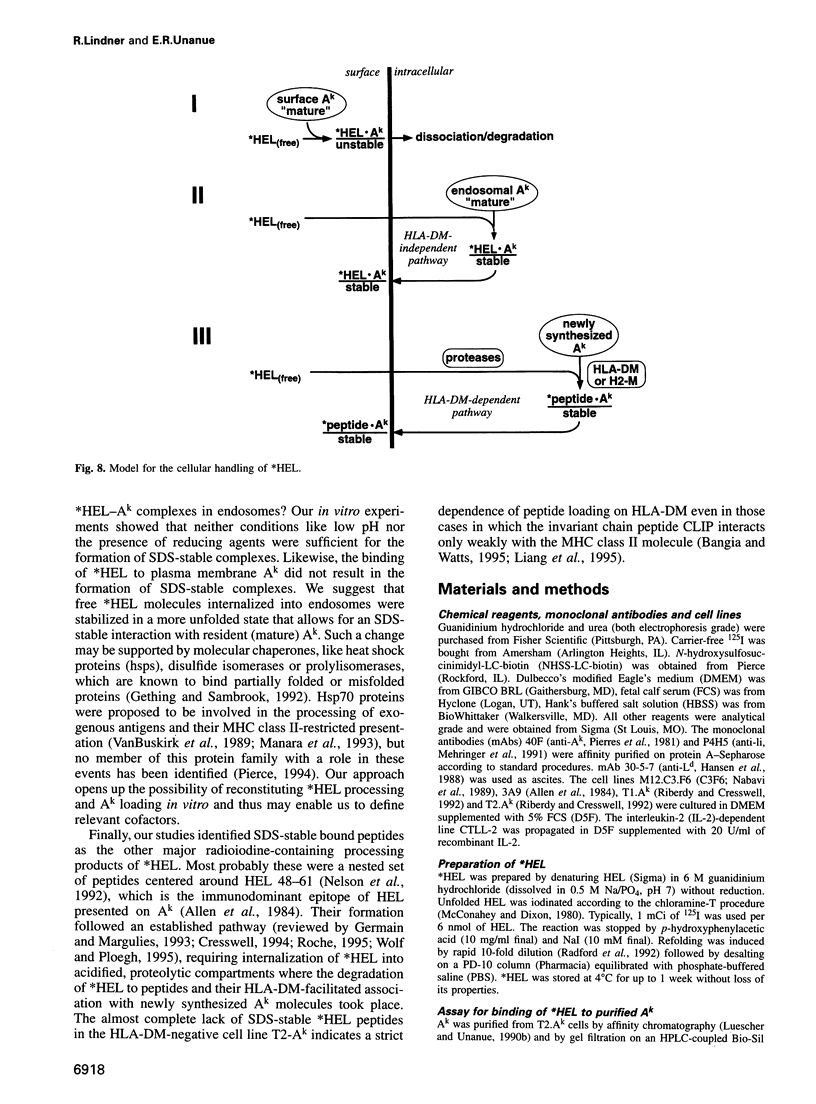
The peptide binding site of MHC class II molecules is open at both ends and, therefore, does not restrict the length of the bound ligand. Here we show that a partially folded protein antigen (*HEL) spontaneously formed SDS-unstable complexes with the purified MHC class II molecule I-Ak (Ak). These complexes were also detected on the surface of antigen-presenting cells (APCs) where they stimulated T cells. However, they rapidly disappeared after endocytosis. Intracellular processing of *HEL gave rise to SDS-stable, long-lived Ak complexes containing *HEL peptides and, unexpectedly, full-length *HEL. Both SDS-stable products were formed in low pH compartments and then transported to the plasma membrane. In contrast to *HEL peptides, the stable association of *HEL occurred in an alternative pathway that required mature class II molecules and did not involve HLA-DM or proteases. SDS-stable *HEL-Ak complexes were formed by a reaction of endosomal Ak with endocytosed *HEL, but not by direct conversion of SDS-unstable complexes derived from the plasma membrane. Our work establishes a fundamental difference between the two MHC class II loading pathways and for the first time demonstrates a full-length protein as a product of antigen processing.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Allen P. M., Strydom D. J., Unanue E. R. Processing of lysozyme by macrophages: identification of the determinant recognized by two T-cell hybridomas. Proc Natl Acad Sci U S A. 1984 Apr;81(8):2489–2493. doi: 10.1073/pnas.81.8.2489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Allen P. M., Unanue E. R. Differential requirements for antigen processing by macrophages for lysozyme-specific T cell hybridomas. J Immunol. 1984 Mar;132(3):1077–1079. [PubMed] [Google Scholar]
- Amigorena S., Webster P., Drake J., Newcomb J., Cresswell P., Mellman I. Invariant chain cleavage and peptide loading in major histocompatibility complex class II vesicles. J Exp Med. 1995 May 1;181(5):1729–1741. doi: 10.1084/jem.181.5.1729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bangia N., Watts T. H. Evidence for invariant chain 85-101 (CLIP) binding in the antigen binding site of MHC class II molecules. Int Immunol. 1995 Oct;7(10):1585–1591. doi: 10.1093/intimm/7.10.1585. [DOI] [PubMed] [Google Scholar]
- Blum J. S., Cresswell P. Role for intracellular proteases in the processing and transport of class II HLA antigens. Proc Natl Acad Sci U S A. 1988 Jun;85(11):3975–3979. doi: 10.1073/pnas.85.11.3975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bonnerot C., Lankar D., Hanau D., Spehner D., Davoust J., Salamero J., Fridman W. H. Role of B cell receptor Ig alpha and Ig beta subunits in MHC class II-restricted antigen presentation. Immunity. 1995 Sep;3(3):335–347. doi: 10.1016/1074-7613(95)90118-3. [DOI] [PubMed] [Google Scholar]
- Brooks A. G., Campbell P. L., Reynolds P., Gautam A. M., McCluskey J. Antigen presentation and assembly by mouse I-Ak class II molecules in human APC containing deleted or mutated HLA DM genes. J Immunol. 1994 Dec 15;153(12):5382–5392. [PubMed] [Google Scholar]
- Brown J. H., Jardetzky T. S., Gorga J. C., Stern L. J., Urban R. G., Strominger J. L., Wiley D. C. Three-dimensional structure of the human class II histocompatibility antigen HLA-DR1. Nature. 1993 Jul 1;364(6432):33–39. doi: 10.1038/364033a0. [DOI] [PubMed] [Google Scholar]
- Busch R., Cloutier I., Sékaly R. P., Hämmerling G. J. Invariant chain protects class II histocompatibility antigens from binding intact polypeptides in the endoplasmic reticulum. EMBO J. 1996 Jan 15;15(2):418–428. [PMC free article] [PubMed] [Google Scholar]
- Buus S., Sette A., Colon S. M., Grey H. M. Autologous peptides constitutively occupy the antigen binding site on Ia. Science. 1988 Nov 18;242(4881):1045–1047. doi: 10.1126/science.3194755. [DOI] [PubMed] [Google Scholar]
- Cresswell P. Assembly, transport, and function of MHC class II molecules. Annu Rev Immunol. 1994;12:259–293. doi: 10.1146/annurev.iy.12.040194.001355. [DOI] [PubMed] [Google Scholar]
- Davidson H. W., Reid P. A., Lanzavecchia A., Watts C. Processed antigen binds to newly synthesized MHC class II molecules in antigen-specific B lymphocytes. Cell. 1991 Oct 4;67(1):105–116. doi: 10.1016/0092-8674(91)90575-j. [DOI] [PubMed] [Google Scholar]
- Denzin L. K., Cresswell P. HLA-DM induces CLIP dissociation from MHC class II alpha beta dimers and facilitates peptide loading. Cell. 1995 Jul 14;82(1):155–165. doi: 10.1016/0092-8674(95)90061-6. [DOI] [PubMed] [Google Scholar]
- Denzin L. K., Robbins N. F., Carboy-Newcomb C., Cresswell P. Assembly and intracellular transport of HLA-DM and correction of the class II antigen-processing defect in T2 cells. Immunity. 1994 Oct;1(7):595–606. doi: 10.1016/1074-7613(94)90049-3. [DOI] [PubMed] [Google Scholar]
- Engelhard V. H. Structure of peptides associated with class I and class II MHC molecules. Annu Rev Immunol. 1994;12:181–207. doi: 10.1146/annurev.iy.12.040194.001145. [DOI] [PubMed] [Google Scholar]
- Fling S. P., Arp B., Pious D. HLA-DMA and -DMB genes are both required for MHC class II/peptide complex formation in antigen-presenting cells. Nature. 1994 Apr 7;368(6471):554–558. doi: 10.1038/368554a0. [DOI] [PubMed] [Google Scholar]
- Germain R. N., Margulies D. H. The biochemistry and cell biology of antigen processing and presentation. Annu Rev Immunol. 1993;11:403–450. doi: 10.1146/annurev.iy.11.040193.002155. [DOI] [PubMed] [Google Scholar]
- Gething M. J., Sambrook J. Protein folding in the cell. Nature. 1992 Jan 2;355(6355):33–45. doi: 10.1038/355033a0. [DOI] [PubMed] [Google Scholar]
- Gruenberg J., Maxfield F. R. Membrane transport in the endocytic pathway. Curr Opin Cell Biol. 1995 Aug;7(4):552–563. doi: 10.1016/0955-0674(95)80013-1. [DOI] [PubMed] [Google Scholar]
- Hansen T. H., Myers N. B., Lee D. R. Studies of two antigenic forms of Ld with disparate beta 2-microglobulin (beta 2m) associations suggest that beta 2m facilitate the folding of the alpha 1 and alpha 2 domains during de novo synthesis. J Immunol. 1988 May 15;140(10):3522–3527. [PubMed] [Google Scholar]
- Harding C. V., Roof R. W., Allen P. M., Unanue E. R. Effects of pH and polysaccharides on peptide binding to class II major histocompatibility complex molecules. Proc Natl Acad Sci U S A. 1991 Apr 1;88(7):2740–2744. doi: 10.1073/pnas.88.7.2740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karlsson L., Péléraux A., Lindstedt R., Liljedahl M., Peterson P. A. Reconstitution of an operational MHC class II compartment in nonantigen-presenting cells. Science. 1994 Dec 2;266(5190):1569–1573. doi: 10.1126/science.7985028. [DOI] [PubMed] [Google Scholar]
- Lee P., Matsueda G. R., Allen P. M. T cell recognition of fibrinogen. A determinant on the A alpha-chain does not require processing. J Immunol. 1988 Feb 15;140(4):1063–1068. [PubMed] [Google Scholar]
- Liang M. N., Beeson C., Mason K., McConnell H. M. Kinetics of the reactions between the invariant chain (85-99) peptide and proteins of the murine class II MHC. Int Immunol. 1995 Sep;7(9):1397–1404. doi: 10.1093/intimm/7.9.1397. [DOI] [PubMed] [Google Scholar]
- Luescher I. F., Crimmins D. L., Schwartz B. D., Unanue E. R. The sites in the I-Ak histocompatibility molecule photoaffinity labeled by an immunogenic lysozyme peptide. J Biol Chem. 1990 Jul 5;265(19):11177–11184. [PubMed] [Google Scholar]
- Luescher I. F., Unanue E. R. Purification and photoaffinity labeling of the I-Ak histocompatibility molecule. J Immunol Methods. 1990 Dec 31;135(1-2):233–245. doi: 10.1016/0022-1759(90)90277-3. [DOI] [PubMed] [Google Scholar]
- Manara G. C., Sansoni P., Badiali-De Giorgi L., Gallinella G., Ferrari C., Brianti V., Fagnoni F. F., Ruegg C. L., De Panfilis G., Pasquinelli G. New insights suggesting a possible role of a heat shock protein 70-kD family-related protein in antigen processing/presentation phenomenon in humans. Blood. 1993 Nov 1;82(9):2865–2871. [PubMed] [Google Scholar]
- McConahey P. J., Dixon F. J. Radioiodination of proteins by the use of the chloramine-T method. Methods Enzymol. 1980;70(A):210–213. doi: 10.1016/s0076-6879(80)70050-2. [DOI] [PubMed] [Google Scholar]
- Mehringer J. H., Harris M. R., Kindle C. S., McCourt D. W., Cullen S. E. Characterization of fragments of the murine Ia-associated invariant chain. J Immunol. 1991 Feb 1;146(3):920–927. [PubMed] [Google Scholar]
- Mellman I., Fuchs R., Helenius A. Acidification of the endocytic and exocytic pathways. Annu Rev Biochem. 1986;55:663–700. doi: 10.1146/annurev.bi.55.070186.003311. [DOI] [PubMed] [Google Scholar]
- Morris P., Shaman J., Attaya M., Amaya M., Goodman S., Bergman C., Monaco J. J., Mellins E. An essential role for HLA-DM in antigen presentation by class II major histocompatibility molecules. Nature. 1994 Apr 7;368(6471):551–554. doi: 10.1038/368551a0. [DOI] [PubMed] [Google Scholar]
- Nabavi N., Ghogawala Z., Myer A., Griffith I. J., Wade W. F., Chen Z. Z., McKean D. J., Glimcher L. H. Antigen presentation abrogated in cells expressing truncated Ia molecules. J Immunol. 1989 Mar 1;142(5):1444–1447. [PubMed] [Google Scholar]
- Nadimi F., Moreno J., Momburg F., Heuser A., Fuchs S., Adorini L., Hämmerling G. J. Antigen presentation of hen egg-white lysozyme but not of ribonuclease A is augmented by the major histocompatibility complex class II-associated invariant chain. Eur J Immunol. 1991 May;21(5):1255–1263. doi: 10.1002/eji.1830210524. [DOI] [PubMed] [Google Scholar]
- Nelson C. A., Petzold S. J., Unanue E. R. Identification of two distinct properties of class II major histocompatibility complex-associated peptides. Proc Natl Acad Sci U S A. 1993 Feb 15;90(4):1227–1231. doi: 10.1073/pnas.90.4.1227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nelson C. A., Petzold S. J., Unanue E. R. Peptides determine the lifespan of MHC class II molecules in the antigen-presenting cell. Nature. 1994 Sep 15;371(6494):250–252. doi: 10.1038/371250a0. [DOI] [PubMed] [Google Scholar]
- Nelson C. A., Roof R. W., McCourt D. W., Unanue E. R. Identification of the naturally processed form of hen egg white lysozyme bound to the murine major histocompatibility complex class II molecule I-Ak. Proc Natl Acad Sci U S A. 1992 Aug 15;89(16):7380–7383. doi: 10.1073/pnas.89.16.7380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pierce S. K. Molecular chaperones in the processing and presentation of antigen to helper T cells. Experientia. 1994 Nov 30;50(11-12):1026–1030. doi: 10.1007/BF01923457. [DOI] [PubMed] [Google Scholar]
- Pierre P., Denzin L. K., Hammond C., Drake J. R., Amigorena S., Cresswell P., Mellman I. HLA-DM is localized to conventional and unconventional MHC class II-containing endocytic compartments. Immunity. 1996 Mar;4(3):229–239. doi: 10.1016/s1074-7613(00)80431-8. [DOI] [PubMed] [Google Scholar]
- Pierres M., Devaux C., Dosseto M., Marchetto S. Clonal analysis of B- and T-cell responses to Ia antigens. I. Topology of epitope regions on I-Ak and I-Ek molecules analyzed with 35 monoclonal alloantibodies. Immunogenetics. 1981 Dec;14(6):481–495. doi: 10.1007/BF00350120. [DOI] [PubMed] [Google Scholar]
- Pinet V., Malnati M. S., Long E. O. Two processing pathways for the MHC class II-restricted presentation of exogenous influenza virus antigen. J Immunol. 1994 May 15;152(10):4852–4860. [PubMed] [Google Scholar]
- Pinet V., Vergelli M., Martin R., Bakke O., Long E. O. Antigen presentation mediated by recycling of surface HLA-DR molecules. Nature. 1995 Jun 15;375(6532):603–606. doi: 10.1038/375603a0. [DOI] [PubMed] [Google Scholar]
- Radford S. E., Dobson C. M., Evans P. A. The folding of hen lysozyme involves partially structured intermediates and multiple pathways. Nature. 1992 Jul 23;358(6384):302–307. doi: 10.1038/358302a0. [DOI] [PubMed] [Google Scholar]
- Riberdy J. M., Cresswell P. The antigen-processing mutant T2 suggests a role for MHC-linked genes in class II antigen presentation. J Immunol. 1992 Apr 15;148(8):2586–2590. [PubMed] [Google Scholar]
- Roche P. A. HLA-DM: an in vivo facilitator of MHC class II peptide loading. Immunity. 1995 Sep;3(3):259–262. doi: 10.1016/1074-7613(95)90111-6. [DOI] [PubMed] [Google Scholar]
- Roof R. W., Luescher I. F., Unanue E. R. Phospholipids enhance the binding of peptides to class II major histocompatibility molecules. Proc Natl Acad Sci U S A. 1990 Mar;87(5):1735–1739. doi: 10.1073/pnas.87.5.1735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sadegh-Nasseri S., Stern L. J., Wiley D. C., Germain R. N. MHC class II function preserved by low-affinity peptide interactions preceding stable binding. Nature. 1994 Aug 25;370(6491):647–650. doi: 10.1038/370647a0. [DOI] [PubMed] [Google Scholar]
- Sanderson F., Kleijmeer M. J., Kelly A., Verwoerd D., Tulp A., Neefjes J. J., Geuze H. J., Trowsdale J. Accumulation of HLA-DM, a regulator of antigen presentation, in MHC class II compartments. Science. 1994 Dec 2;266(5190):1566–1569. doi: 10.1126/science.7985027. [DOI] [PubMed] [Google Scholar]
- Schmid S. L., Carter L. L. ATP is required for receptor-mediated endocytosis in intact cells. J Cell Biol. 1990 Dec;111(6 Pt 1):2307–2318. doi: 10.1083/jcb.111.6.2307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmid S. L., Jackson M. R. Immunology. Making class II presentable. Nature. 1994 May 12;369(6476):103–104. doi: 10.1038/369103a0. [DOI] [PubMed] [Google Scholar]
- Schägger H., von Jagow G. Tricine-sodium dodecyl sulfate-polyacrylamide gel electrophoresis for the separation of proteins in the range from 1 to 100 kDa. Anal Biochem. 1987 Nov 1;166(2):368–379. doi: 10.1016/0003-2697(87)90587-2. [DOI] [PubMed] [Google Scholar]
- Sette A., Adorini L., Colon S. M., Buus S., Grey H. M. Capacity of intact proteins to bind to MHC class II molecules. J Immunol. 1989 Aug 15;143(4):1265–1267. [PubMed] [Google Scholar]
- Sherman M. A., Weber D. A., Jensen P. E. DM enhances peptide binding to class II MHC by release of invariant chain-derived peptide. Immunity. 1995 Aug;3(2):197–205. doi: 10.1016/1074-7613(95)90089-6. [DOI] [PubMed] [Google Scholar]
- Sloan V. S., Cameron P., Porter G., Gammon M., Amaya M., Mellins E., Zaller D. M. Mediation by HLA-DM of dissociation of peptides from HLA-DR. Nature. 1995 Jun 29;375(6534):802–806. doi: 10.1038/375802a0. [DOI] [PubMed] [Google Scholar]
- Srinivasan M., Marsh E. W., Pierce S. K. Characterization of naturally processed antigen bound to major histocompatibility complex class II molecules. Proc Natl Acad Sci U S A. 1991 Sep 15;88(18):7928–7932. doi: 10.1073/pnas.88.18.7928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stern L. J., Brown J. H., Jardetzky T. S., Gorga J. C., Urban R. G., Strominger J. L., Wiley D. C. Crystal structure of the human class II MHC protein HLA-DR1 complexed with an influenza virus peptide. Nature. 1994 Mar 17;368(6468):215–221. doi: 10.1038/368215a0. [DOI] [PubMed] [Google Scholar]
- Tulp A., Verwoerd D., Dobberstein B., Ploegh H. L., Pieters J. Isolation and characterization of the intracellular MHC class II compartment. Nature. 1994 May 12;369(6476):120–126. doi: 10.1038/369120a0. [DOI] [PubMed] [Google Scholar]
- Vanbuskirk A., Crump B. L., Margoliash E., Pierce S. K. A peptide binding protein having a role in antigen presentation is a member of the HSP70 heat shock family. J Exp Med. 1989 Dec 1;170(6):1799–1809. doi: 10.1084/jem.170.6.1799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wolf P. R., Ploegh H. L. How MHC class II molecules acquire peptide cargo: biosynthesis and trafficking through the endocytic pathway. Annu Rev Cell Dev Biol. 1995;11:267–306. doi: 10.1146/annurev.cb.11.110195.001411. [DOI] [PubMed] [Google Scholar]
- Ziegler H. K., Unanue E. R. Decrease in macrophage antigen catabolism caused by ammonia and chloroquine is associated with inhibition of antigen presentation to T cells. Proc Natl Acad Sci U S A. 1982 Jan;79(1):175–178. doi: 10.1073/pnas.79.1.175. [DOI] [PMC free article] [PubMed] [Google Scholar]