Abstract
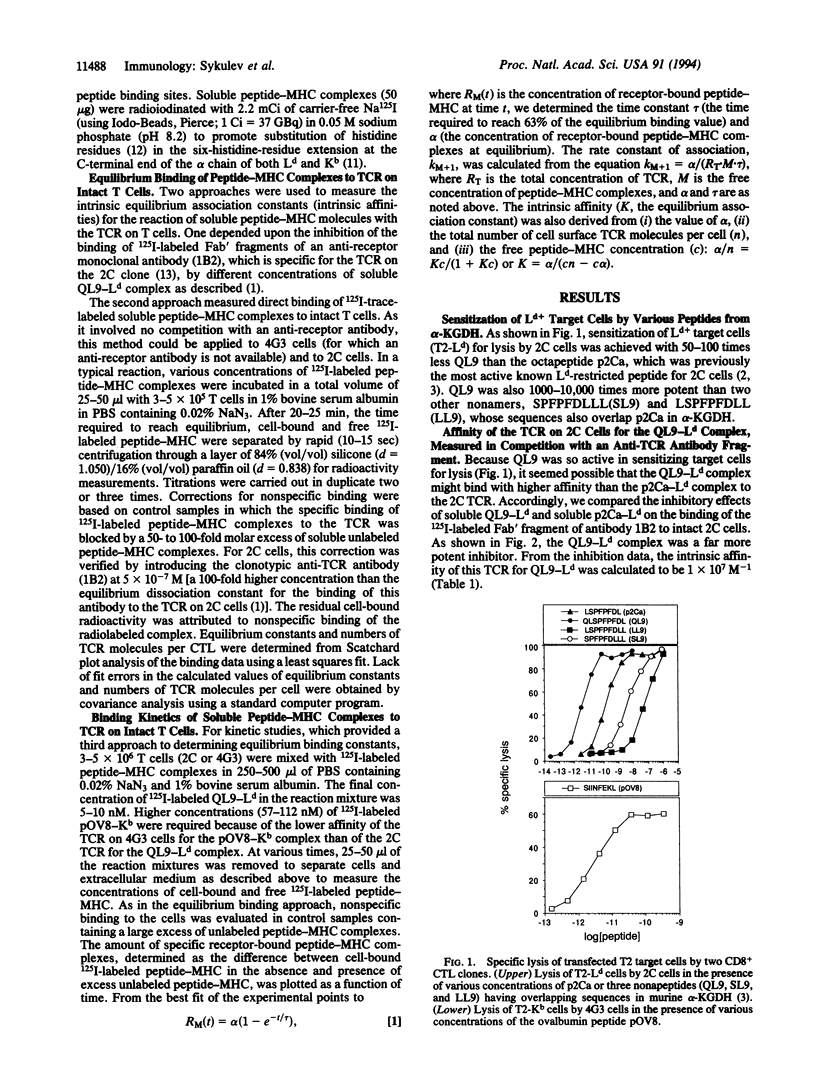
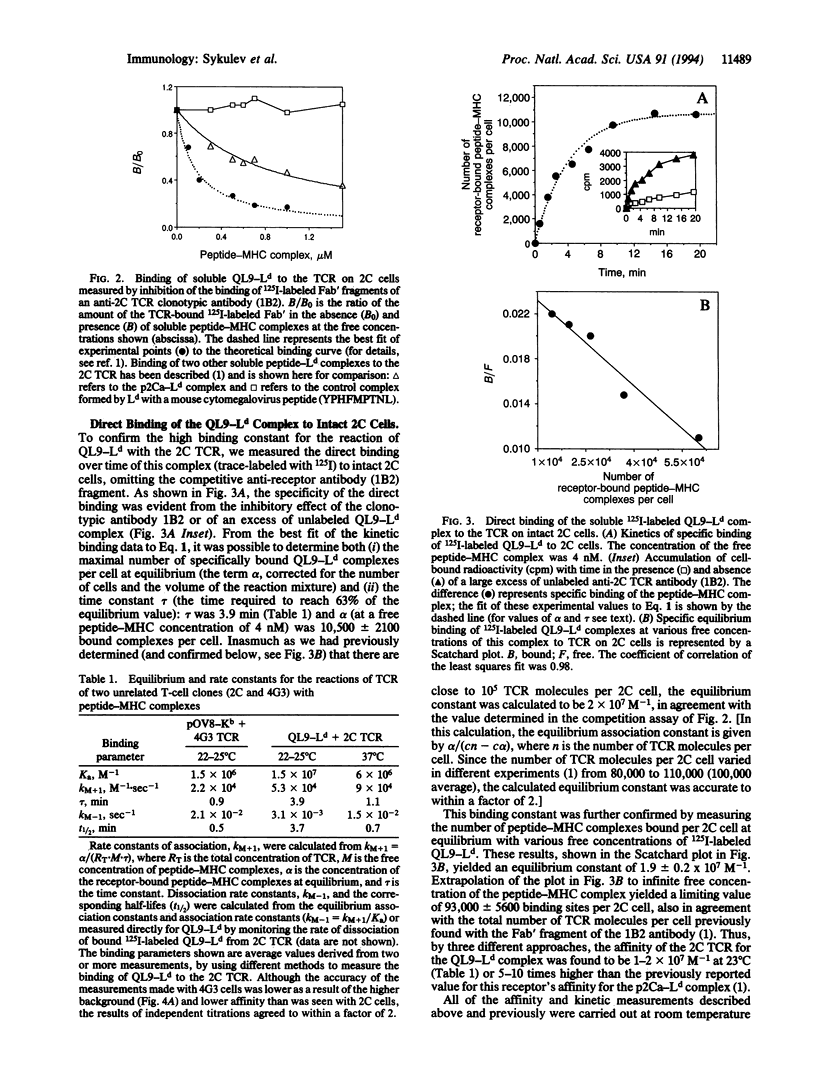
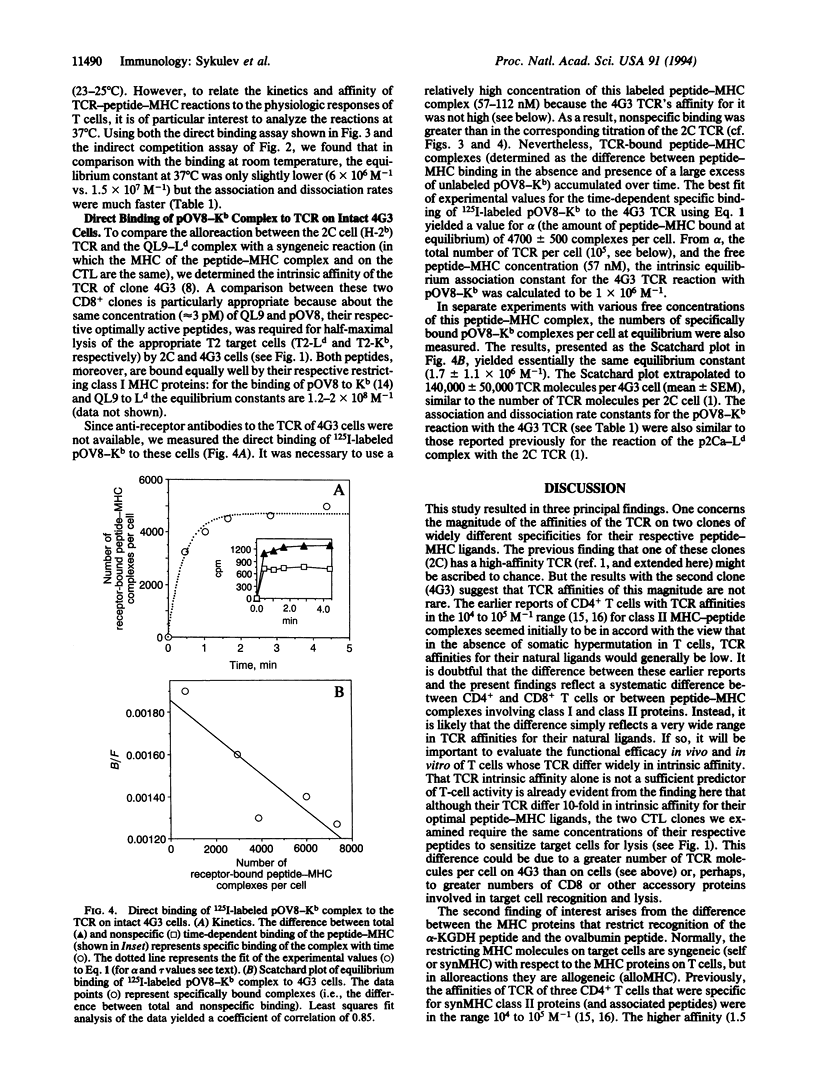
We report here that the intrinsic affinities of the antigen-specific T-cell receptors (TCR) of two unrelated CD8+ T-cell clones for their respective peptide-major histocompatibility complex (MHC) ligands are higher than the values generally thought to prevail for TCR. The TCR of one clone (2C) binds an allogeneic class I MHC protein (Ld) in association with an alpha-ketoglutarate dehydrogenase nonapeptide (QLSPFPFDL, termed QL9) with an intrinsic affinity (intrinsic equilibrium association constant) of 1-2 x 10(7) M-1. The TCR of the other clone (4G3) binds a syngeneic class I MHC protein (Kb) in association with an ovalbumin octapeptide (SIINFEKL, termed pOV8) with an intrinsic affinity of 1.5 x 10(6) M-1. A comparison of the two clones, combined with current views of T-cell repertoire selection in the thymus, leads us to propose that TCR affinities are generally likely to be higher for allogeneic MHC-peptide complexes than for syngeneic MHC-peptide complexes.
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alexander J., Payne J. A., Murray R., Frelinger J. A., Cresswell P. Differential transport requirements of HLA and H-2 class I glycoproteins. Immunogenetics. 1989;29(6):380–388. doi: 10.1007/BF00375866. [DOI] [PubMed] [Google Scholar]
- Ashton-Rickardt P. G., Bandeira A., Delaney J. R., Van Kaer L., Pircher H. P., Zinkernagel R. M., Tonegawa S. Evidence for a differential avidity model of T cell selection in the thymus. Cell. 1994 Feb 25;76(4):651–663. doi: 10.1016/0092-8674(94)90505-3. [DOI] [PubMed] [Google Scholar]
- Hogquist K. A., Jameson S. C., Heath W. R., Howard J. L., Bevan M. J., Carbone F. R. T cell receptor antagonist peptides induce positive selection. Cell. 1994 Jan 14;76(1):17–27. doi: 10.1016/0092-8674(94)90169-4. [DOI] [PubMed] [Google Scholar]
- Jackson M. R., Song E. S., Yang Y., Peterson P. A. Empty and peptide-containing conformers of class I major histocompatibility complex molecules expressed in Drosophila melanogaster cells. Proc Natl Acad Sci U S A. 1992 Dec 15;89(24):12117–12121. doi: 10.1073/pnas.89.24.12117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kappler J. W., Roehm N., Marrack P. T cell tolerance by clonal elimination in the thymus. Cell. 1987 Apr 24;49(2):273–280. doi: 10.1016/0092-8674(87)90568-x. [DOI] [PubMed] [Google Scholar]
- Kisielow P., Blüthmann H., Staerz U. D., Steinmetz M., von Boehmer H. Tolerance in T-cell-receptor transgenic mice involves deletion of nonmature CD4+8+ thymocytes. Nature. 1988 Jun 23;333(6175):742–746. doi: 10.1038/333742a0. [DOI] [PubMed] [Google Scholar]
- Kranz D. M., Sherman D. H., Sitkovsky M. V., Pasternack M. S., Eisen H. N. Immunoprecipitation of cell surface structures of cloned cytotoxic T lymphocytes by clone-specific antisera. Proc Natl Acad Sci U S A. 1984 Jan;81(2):573–577. doi: 10.1073/pnas.81.2.573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kranz D. M., Tonegawa S., Eisen H. N. Attachment of an anti-receptor antibody to non-target cells renders them susceptible to lysis by a clone of cytotoxic T lymphocytes. Proc Natl Acad Sci U S A. 1984 Dec;81(24):7922–7926. doi: 10.1073/pnas.81.24.7922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lindahl K. F., Wilson D. B. Histocompatibility antigen-activated cytotoxic T lymphocytes. II. Estimates of the frequency and specificity of precursors. J Exp Med. 1977 Mar 1;145(3):508–522. doi: 10.1084/jem.145.3.508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsui K., Boniface J. J., Reay P. A., Schild H., Fazekas de St Groth B., Davis M. M. Low affinity interaction of peptide-MHC complexes with T cell receptors. Science. 1991 Dec 20;254(5039):1788–1791. doi: 10.1126/science.1763329. [DOI] [PubMed] [Google Scholar]
- Matsumura M., Saito Y., Jackson M. R., Song E. S., Peterson P. A. In vitro peptide binding to soluble empty class I major histocompatibility complex molecules isolated from transfected Drosophila melanogaster cells. J Biol Chem. 1992 Nov 25;267(33):23589–23595. [PubMed] [Google Scholar]
- Matzinger P., Bevan M. J. Hypothesis: why do so many lymphocytes respond to major histocompatibility antigens? Cell Immunol. 1977 Mar 1;29(1):1–5. doi: 10.1016/0008-8749(77)90269-6. [DOI] [PubMed] [Google Scholar]
- Moore M. W., Carbone F. R., Bevan M. J. Introduction of soluble protein into the class I pathway of antigen processing and presentation. Cell. 1988 Sep 9;54(6):777–785. doi: 10.1016/s0092-8674(88)91043-4. [DOI] [PubMed] [Google Scholar]
- Rötzschke O., Falk K., Stevanović S., Jung G., Walden P., Rammensee H. G. Exact prediction of a natural T cell epitope. Eur J Immunol. 1991 Nov;21(11):2891–2894. doi: 10.1002/eji.1830211136. [DOI] [PubMed] [Google Scholar]
- Sha W. C., Nelson C. A., Newberry R. D., Kranz D. M., Russell J. H., Loh D. Y. Positive and negative selection of an antigen receptor on T cells in transgenic mice. Nature. 1988 Nov 3;336(6194):73–76. doi: 10.1038/336073a0. [DOI] [PubMed] [Google Scholar]
- Sykulev Y., Brunmark A., Jackson M., Cohen R. J., Peterson P. A., Eisen H. N. Kinetics and affinity of reactions between an antigen-specific T cell receptor and peptide-MHC complexes. Immunity. 1994 Apr;1(1):15–22. doi: 10.1016/1074-7613(94)90005-1. [DOI] [PubMed] [Google Scholar]
- Tsomides T. J., Eisen H. N. Stoichiometric labeling of peptides by iodination on tyrosyl or histidyl residues. Anal Biochem. 1993 Apr;210(1):129–135. doi: 10.1006/abio.1993.1162. [DOI] [PubMed] [Google Scholar]
- Udaka K., Tsomides T. J., Eisen H. N. A naturally occurring peptide recognized by alloreactive CD8+ cytotoxic T lymphocytes in association with a class I MHC protein. Cell. 1992 Jun 12;69(6):989–998. doi: 10.1016/0092-8674(92)90617-l. [DOI] [PubMed] [Google Scholar]
- Udaka K., Tsomides T. J., Walden P., Fukusen N., Eisen H. N. A ubiquitous protein is the source of naturally occurring peptides that are recognized by a CD8+ T-cell clone. Proc Natl Acad Sci U S A. 1993 Dec 1;90(23):11272–11276. doi: 10.1073/pnas.90.23.11272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walden P. R., Eisen H. N. Cognate peptides induce self-destruction of CD8+ cytolytic T lymphocytes. Proc Natl Acad Sci U S A. 1990 Nov;87(22):9015–9019. doi: 10.1073/pnas.87.22.9015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weber S., Traunecker A., Oliveri F., Gerhard W., Karjalainen K. Specific low-affinity recognition of major histocompatibility complex plus peptide by soluble T-cell receptor. Nature. 1992 Apr 30;356(6372):793–796. doi: 10.1038/356793a0. [DOI] [PubMed] [Google Scholar]



