Abstract
Suprasellar hemangioblastoma (HBL) without von Hippel-Lindau (VHL) disease is extremely rare. A 51-year-old woman presented with headache and progressively deteriorating bilateral visual disturbance for 4 months. Magnetic resonance imaging (MRI) revealed a 2.5-cm solid mass in the suprasellar region with homogeneous contrast enhancement. Our preoperative presumptive diagnosis was meningioma. Resection of the tumor was achieved via a left pterional craniotomy. The tumor was reddish in appearance and relatively firm, and was extremely vascularized, which might provide extensive blood supply through small branches of the internal carotid artery. There was a clear border between the tumor and the pituitary stalk and optic nerves. Histopathologic examination showed that the tumor was well vascularized, consisting of a reticular mesh of numerous thin-walled capillaries and abundant stromal cells. Immunohistochemistry demonstrated the positive staining for CD34, vimentin (VIM), and neuron specific enolase (NSE) in the intratumoral capillaries, while negative staining of epithelial membrane antigen (EMA) and glial fibrillary acidic protein (GFAP) was observed. Based on these results, the patient was diagnosed as HBL. After the resection, the visual field defect in the left eye was markedly improved, and no tumor recurrence was noted in 1 year follow-up. When solid lesions are highly vascularized in the suprasellar region of patients, even though no VHL disease is present, the possibility of HBL should be taken into consideration. Moreover, craniotomy is a better treatment option for suprasellar HBL without VHL disease.
Keywords: Suprasellar region, hemangioblastoma, von Hippel-Lindau disease, craniotomy
Introduction
Hemangioblastomas (HBLs) are tumors of uncertain histogenesis which may be sporadic or associated with von Hippel-Lindau (VHL) disease [1]. The benign vascular tumors account for 1-2.5% of all intracranialneoplasms [2]. The cerebellum is the most frequent site for these tumors, however the tumors may also occur in the medulla oblongata and the spinal cord [3].
Suprasellar HBLs are rare, with only 18 cases reported to date [4]. Moreover, suprasellar HBL without VHL disease is extremely rare, and only 5 cases have been previously reported [4-8]. The most common presenting complaints of the disease include visual disturbance, headache, and endocrinological disturbance. Both the transsphenoidal approach and the craniotomy have been used to treat suprasellar HBL without VHL disease.
In this paper, another suprasellar HBL case without VHL disease is described. Additionally, all the HBL cases without VHL disease in the literature have been reviewed, and the diagnosis and the treatment of the disease have been discussed.
Case description
A 51-year-old woman was admitted to our hospital with a 4-month history of headache and progressively deteriorating bilateral visual disturbance. There was no family history of VHL disease. Endocrinological examination indicated as light hypocortisolism. Ophthalmological examination revealed a visual field defect in the left eye. General systemic examination failed to show any abnormality associated with VHL disease. Magnetic resonance imaging (MRI) demonstrated a 2.5-cm solid mass in the suprasellar region, which was isointense on T1-weighted images and hyperintense on T2-weighted images, with homogeneous contrast enhancement (Figure 1A-D).
Figure 1.
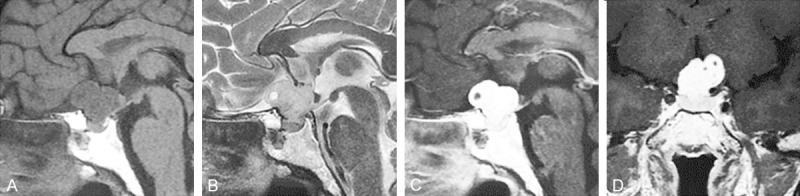
Magnetic resonance imaging (MRI) for the diagnosis of suprasellar HBL without VHL disease. A. T1-weightedsagittal image; B. T2-weightedsagittal image; C. Contrast enhanced T1-weightedsagittal image; D. Contrast enhanced T1-weightedcoronal image.
Total resection of the tumor was successfully accomplished via a left pterional craniotomy. During the operation, the tumor was reddish in appearance, and was relatively firm. It received an extensive blood supply from small branches of the internal carotid artery. There was a clear border between the tumor and the pituitary stalk as well as the optic nerves. Histopathologic analysis showed that the tumor was highly vascularized, consisting of a reticular mesh of numerous thin-walled capillaries and abundant stromal cells (Figure 2A). Immunohistochemistry demonstrated the expression of the endothelial-associated marker CD34 in the intratumoral capillaries (Figure 2B), and strong immunoreactivity for vimentin (VIM; Figure 2C) and neuron specific enolase (NSE; Figure 2) were also observed, which were the major intermediate filament proteins expressed by stromal cells in HBL. However, the tumor cells were negative for epithelial membrane antigen (EMA; Figure 2E) and glial fibrillary acidic protein (GFAP; Figure 2F).
Figure 2.
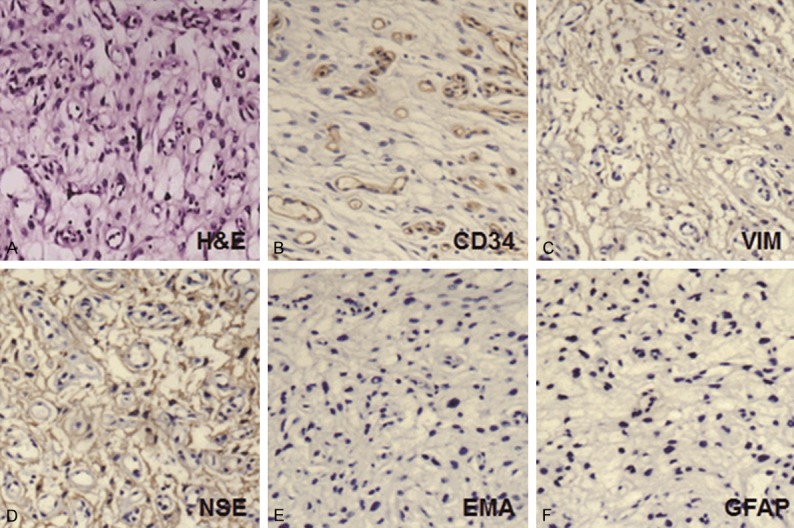
H&E staining and immunohistochemical analysis of suprasellar HBL without VHL disease. (A) H&E staining (×100); (B-F) Immunohistochemical analysis for CD34 (B), vimentin (VIM; C), neuron specific enolase (NSE; D), epithelial membrane antigen (EMA; E), and glial fibrillary acidic protein (GFAP; F) (×100).
After the resection, the visual field defect in the left eye of the patient was markedly improved Endocrinological examination showed hypopituitarism associated with transient diabetes insipidus. The patient received substitution therapy with hydrocortisone and transient therapy with desmopressin. A repeat MRI scan was performed at 1 year after surgery, and the results showed adequate chiasmatic decompression, with no tumor recurrence (Figure 3).
Figure 3.
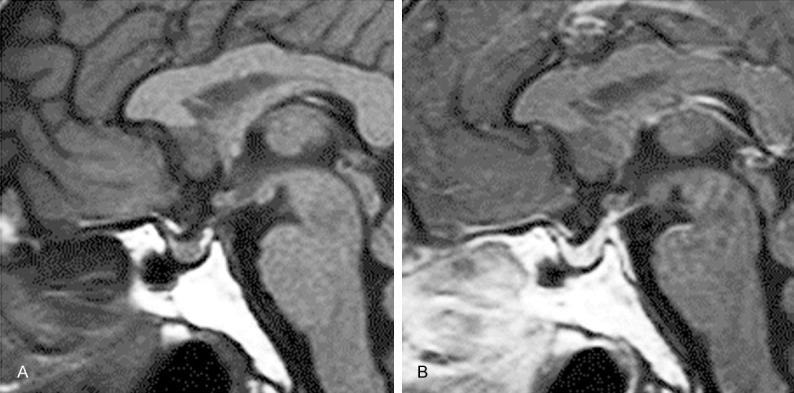
One year postoperative MRI of the patient. A. T1-weighted sagittal image; B. Contrast enhanced T1-weighted sagittal image.
Discussion
Up to now, only 18 cases of suprasellar HBL have been previously described [1,3,4,6,7-13]. In this paper, another case of HBL in the suprasellar region was reported. Out of these cases, 12 (63.2%) were associated with VHL disease, while 6 (31.6%) were not, and the remaining 1 (5.2%) was not specifically mentioned. These results suggest that the suprasellar HBL is prone to be linked with VHL disease. A recent study has reported that the average age of patients with sporadic HBL is 52.4 years, whereas the average age of patients with VHL-associated HBL is 35.8 years [14]. In accordance with this study, the average age of the 12 patients with VHL-associated HBL in the suprasellar region (31.7 years) has been found to be younger than the 6 suprasellar HBL patients without VHL disease (53.2 years). Moreover, it has been shown that 49% of the supratentorial HBL cases are cystic, while cysts are seldom (only 12.5%) seen in sellar and suprasellar HBL cases [8,15]. Furthermore, we have noticed that, all the tumors in the 6 suprasellar HBL cases without VHL disease are solid (Table 1).
Table 1.
Cases of suprasellar hemangioblastoma (HBL) without von Hippel-Lindau (VHL) disease
| No. | Age (yr)/sex | Symptoms/signs | Solid/cystic | Imaging examinations | Surgery | Follow-up | Reference |
|---|---|---|---|---|---|---|---|
| 1 | 28/F | Galactorrhoea, elevated PRL, Chiari-Frommel syndrome | Solid | CT: enhancing lesion; Angio: highly vascular lesion | Craniotomy, total resection | NS | Grisoli et al. [4] |
| 2 | 62/M | Visual disturbance | Solid | MRI: hyperintense on T2, homogeneous enhancement; Angio: tumor blush | Craniotomy, total resection | NS | Ikeda et al. [5] |
| 3 | 60/M | Visual loss, bitemporal hemianopsia, panhypopituitarism | Solid | MRI: hyperintense on T2, homogeneous enhancement | Transsphenoidal biopsy | NS | Rumboldt et al. [12] |
| 4 | 54/M | Headache, visual loss | Solid | MRI: isointense on T1, hyperintense on T2, homogeneous enhancement | Craniotomy, total resection | No tumor recurrence 5 yr | Peker et al. [11] |
| 5 | 64/F | Headache, visual disturbance | Solid | MRI: isointense on T1, hyperintense on T2, homogeneous enhancement; Angio: highly vascular lesion | Endoscopic transsphenoidal approach, subtotal resection | Secondary cerebrospinal fluid leak and hydrocephalus | Xie et al. [16] |
| 6 | 51/F | Headache, visual disturbance, hypocortisolism | Solid | MRI: isointense on T1, hyperintense on T2, homogeneous enhancement | Craniotomy, total resection | No tumor recurrence 1 yr | Present case |
Abbreviations: F, female; M, male; PRL: prolactin; Angio, angiography; CT, computed tomography; MRI, magnetic resonance imaging; NS, not stated.
The non-specific features of the suprasellar HBL make it really difficult to establish the correct preoperative diagnosis, especially in solitary tumor patients with no past or family history of VHL disease. MRI findings of the sellar region are available for 5 suprasellar HBL cases without VHL disease, which is mandatory for the diagnosis of the disease. The MRI results have demonstrated that, suprasellar HBL cases commonly appear isointense on T1-weighted images and hyperintense on T2-weighted images, with homogeneous enhancement on contrast enhanced T1-weighted images. However, these imaging characteristics of suprasellar HBL are similar to that of sellar meningioma or pituitary tumor, which may lead to misdiagnosis. In the case of suprasellar HBL reported herein, we noted that contrast-enhanced T1-weighted images could reveal the diaphragm of sella turcica between the tumor and the pituitary gland, which might contribute to the differential diagnosis. Moreover, signal void were observed within the tumor, which was consistent with the features of intratumoral vascular structure in suprasellar HBL cases previously described [4,7,8]. In addition, angiography can be helpful in diagnosing suprasellar HBL. In the reported 6 cases suprasellar HBL without VHL disease, 3 cases have been subjected to angiography, both of which show highly vascular lesion and dense tumor blush. Definitive diagnosis of suprasellar HBL could be made based on the pathologic examination. Our results from immunohistochemistry showed that, the intratumoral capillaries were strongly positive for the CD34 staining, and the HBL stromal cells were positive for the staining of VIM and NSE. However, HBL was negative for the staining of EMA or GFAP, which might contribute to the differential diagnosis.
Although Lonser et al. [11] have reported that the surgical intervention could be reserved for pituitary stalk HBL until associated signs or symptoms occur, the microsurgical resection remains one of the major treatment options for symptomatic and sporadic HBL cases in the suprasellar region. Both the transsphenoidal approach and craniotomy have been reported in the treatment of suprasellar HBL [2,5,6,8-10,12,13,16]. There are 4 cases of suprasellar HBL without VHL disease that have undergone craniotomy in previous studies and our own, and all of them have achieved total tumor removal.
However, the other 2 suprasellar HBL patients without VHL disease, who have underwent surgery via the transsphenoidal approach, only achieved biopsy or subtotal tumor resection. Moreover, the transsphenoidal approach could be associated with more complications, including secondary cerebrospinal fluid leak, significant arterial hemorrhage, and postoperative communicating hydrocephalus [4]. These findings suggest that the transsphenoidal approach might not be the most appropriate treatment choice for suprasellar HBL without VHL disease.
Conclusion
When solid lesions are highly vascularized in the suprasellar region of patients, even though no VHL disease is present, the possibility of HBL should be taken into consideration. Furthermore, craniotomy might be a better treatment option for suprasellar HBL without VHL disease.
Disclosure of conflict of interest
None.
References
- 1.Bohling TPK, Haltia MJ, Alitalo K, Neumann HPH. Von Hippel-Lindaudisease and capillary hemangioblastoma. In: Kleihues P, Cavanee W, editors. Pathology and Genetics. Tumors of the Nervous System: World Health Organization Classification of Tumors. Lyon: IARC press; 2000. pp. 223–6. [Google Scholar]
- 2.Wasenko JJ, Rodziewicz GS. Suprasellar hemangioblastoma in Von Hippel-Lindau disease: a case report. Clin Imaging. 2003;27:18–22. doi: 10.1016/s0899-7071(02)00537-5. [DOI] [PubMed] [Google Scholar]
- 3.Bilge T, Özveren F, Senol S, Bilge S, Barut S, Karakaslar O, Aydin Y. Von Hippel-Lindau disease: analysis of two families. Neurochirurgia. 1992;35:204–6. doi: 10.1055/s-2008-1052279. [DOI] [PubMed] [Google Scholar]
- 4.Xie T, Zhang XB, Hu F, Wang XJ, Wang J, Yu Yong, Chen LL. Suprasellar hemangioblastoma mimicking acraniopharyngioma: result of extended endoscopictranssphenoidal approach-case report. Neurol Med Chir (Tokyo) 2013;53:735–9. doi: 10.2176/nmc.cr2011-0016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Grisoli F, Gambarelli D, Raybaud C, Guibout M, Leclercq T. Suprasellar hemangioblastoma. Surg Neurol. 1984;22:257–62. doi: 10.1016/0090-3019(84)90010-7. [DOI] [PubMed] [Google Scholar]
- 6.Ikeda M, Asada M, Yamashita H, Ishikawa A, Tamaki N. A case of suprasellar hemangioblastoma with thoracic meningioma. No Shinkei Geka. 2001;29:679–83. [PubMed] [Google Scholar]
- 7.Peker S, Kurtaya-Yapicier O, Sun I, Sav A, Pamir MN. Suprasellar haemangioblastoma. Report of two cases and review of the literature. J Clin Neurosci. 2005;12:85–9. doi: 10.1016/j.jocn.2004.02.025. [DOI] [PubMed] [Google Scholar]
- 8.Rumboldt Z, Gnjidić Z, Talan-Hranilović J, Vrkljan M. Intrasellar hemangioblastoma: characteristic prominent vessels on MR imaging. AJR Am J Roentgenol. 2003;180:1480–1. doi: 10.2214/ajr.180.5.1801480. [DOI] [PubMed] [Google Scholar]
- 9.Goto T, Nishi T, Kunitoku N, Yamamoto K, Kitamura I, Takeshima H, Kochi M, Nakazato Y, Kuratsu J, Ushio Y. Suprasellar hemangioblastoma in a patient with von Hippel-Lindau disease confirmed by germline mutation study: case report and review of the literature. Surg Neurol. 2001;56:22–6. doi: 10.1016/s0090-3019(01)00482-7. [DOI] [PubMed] [Google Scholar]
- 10.Kouri JG, Chen MY, Watson JC, Oldfield EH. Resection of suprasellar tumors using a modified transphenoidal approach. Report of four cases. J Neurosurg. 2000;92:1028–35. doi: 10.3171/jns.2000.92.6.1028. [DOI] [PubMed] [Google Scholar]
- 11.Lonser RR, Butman JA, Kiringoda R, Song D, Oldfield EH. Pituitary stalk hemangioblastomas in von Hippel-Lindau disease. J Neurosurg. 2009;110:350–3. doi: 10.3171/2008.4.17532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Miyata S, Mikami T, Minamida Y, Akiyama Y, Houkin K. Suprasellar hemangioblastoma. J Neuroophthalmol. 2008;28:325–6. doi: 10.1097/WNO.0b013e318183c633. [DOI] [PubMed] [Google Scholar]
- 13.O’Reilly GV, Rumbaugh CL, Bowens M, Kido DK, Naheedy MH. Supratentorial haemangioblastoma: the diagnostic roles of computed tomography and angiography. Clin Radiol. 1981;32:389–92. doi: 10.1016/s0009-9260(81)80276-0. [DOI] [PubMed] [Google Scholar]
- 14.Schär R, Vajtai I, Sahli R, Seiler RW. Manifestation of a sellar hemangioblatoma due to pituitary apoplexy: a case report. J Med Case Reports. 2011;5:496. doi: 10.1186/1752-1947-5-496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Iplikcioglu AC, Yaradanakul V, Trakya U. Supratentorial haemangioblastoma: appearances on MR imaging. Br J Neurosurg. 1997;11:576–8. doi: 10.1080/02688699745772. [DOI] [PubMed] [Google Scholar]
- 16.Sawin PD, Follett KA, Wen BC, Laws ER Jr. Symptomatic intrasellar hemangioblastoma in a child treated with subtotal resection and adjuvant radiosurgery. Case report. J Neurosurg. 1996;84:1046–50. doi: 10.3171/jns.1996.84.6.1046. [DOI] [PubMed] [Google Scholar]


