Abstract
Lactic acidosis occurs when lactate production exceeds its metabolism. There are many possible causes of lactic acidosis, and in any given patient, several causes may coexist. This Attending Rounds presents a case in point. Metformin’s role in the pathogenesis of lactic acidosis in patients with diabetes mellitus is complex, as the present case illustrates. The treatment of lactic acidosis is controversial, except for the imperative to remedy its underlying cause. The use of sodium bicarbonate to treat the often alarming metabolic derangements may be quite efficacious in that regard but is of questionable benefit to patients. Renal replacement therapies (RRTs) have particular appeal in this setting for a variety of reasons, but their effect on clinical outcomes is untested.
Keywords: acidosis, congestive heart failure, diabetes mellitus, dialysis, intoxication
Introduction
A 49-year-old man presented to the emergency department complaining of dyspnea for 2 days. He had a history of hypertension, type 2 diabetes mellitus, atrial fibrillation, and a severe dilated cardiomyopathy. He had been hospitalized several times in the previous year for decompensated congestive heart failure (most recently, 1 month earlier). The plasma creatinine concentration was 1.13 mg/dl on discharge.
Outpatient medications included insulin, digoxin, warfarin, spironolactone, metoprolol succinate, furosemide (80 mg two times per day; increased from 40 mg daily 1 month earlier), metolazone (2.5 mg daily; added 1 month earlier), and metformin (2500 mg in three divided doses; increased from 1000 mg 1 month earlier).
Physical examination revealed an obese man in moderate respiratory distress. The temperature was 36.8°C, BP was 119/83 mmHg, and heart rate was 96 per minute. Peripheral hemoglobin oxygen saturation was 97% on room air, with a respiratory rate of 26 per minute. The heart rhythm was irregularly irregular; there was no S3 or murmur. Jugular venous pressure was about 8 cm. There was 1+ edema at the ankles. A chest radiograph showed cardiomegaly and central venous prominence. The N-terminal pro-B-type natriuretic peptide level was 5137 pg/ml (reference range = 1–138 pg/ml). The peripheral hemoglobin concentration was 12.5 g/dl, the white blood cell count was 12,500/µl (76% granulocytes), and the platelet count was 332,000/µL. Initial plasma chemistries are shown in Table 1.
Table 1.
Initial plasma chemistry results
| Analyte | 1 mo Earlier | On Presentation | +2 h |
|---|---|---|---|
| Glucose (mg/dl) | 128 | 136 | 174 |
| BUN (mg/dl) | 22 | 29 | 34 |
| Creatinine (mg/dl) | 1.13 | 1.36 | 1.52 |
| Sodium (mmol/L) | 140 | 136 | 137 |
| Potassium (mmol/L) | 3.7 | 4.3 | 6.2 |
| Chloride (mmol/L) | 97 | 95 | 89 |
| Total CO2 (mmol/L) | 29 | 20 | 4 |
| Anion gap (mmol/L) | 14 | 21 | 44 |
| Lactate (mmol/L) | 18.7 | ||
| pH, arterial | 6.97 | ||
| pCO2, arterial (torr) | 26 | ||
| pO2, arterial (torr) | 134 | ||
| HCO3, arterial (mmol/L) | 8 |
The impression was decompensated congestive heart failure. After administration of furosemide (160 mg intravenously), the urine output increased to 320 ml over the next 1 hour. There was no improvement in the dyspnea. Within 2 hours, the patient’s BP fell to 100/64 mmHg; he became agitated and more tachypneic. An endotracheal tube was inserted, and he was placed on mechanical ventilation. The BP fell to 80/42 mmHg, and continuous intravenous infusions of dobutamine, epinephrine, and norepinephrine were begun along with intravenous infusions of vancomycin, levofloxacin, and metronidazole. The urine output fell to <5 ml/h. Repeat plasma chemistry results are shown in Table 1. The nephrology service was consulted.
Patient Summary
A middle-aged man with a history of type 2 diabetes mellitus and severe congestive heart failure presented with a 2-day history of dyspnea. Apart from tachypnea, he had normal vital signs and evidence of moderate fluid overload with vascular congestion. Initial plasma chemistries showed a plasma total CO2 and a small increase in the plasma creatinine concentration from his known baseline. The anion gap was high. Within 2 hours, the patient became agitated, his BP fell, and he required mechanical ventilation and medications to support his BP. Over the next several hours, he developed oliguria and a severe metabolic acidosis, with an anion gap of 44 mmol/L and a plasma lactate concentration of 18.7 mmol/L.
Discussion
Close examination of the initial plasma chemistries reveals that he had an anion gap of 21 mmol/L on presentation. The anion gap is well above the laboratory’s reference range of 8–14 mEq/L, and, with that as a basis, could theoretically represent an increase of 7–13 mEq/L over baseline. In reality, there is considerable variability in the usual anion gap of a given individual (1), and the best way to assess the change in the anion gap is to compare it not with the laboratory’s reference range but with the patient’s own usual anion gap. In this patient, there were ample old laboratory results to review, and his anion gap ranged between 10 and 12 mEq/L. Thus, the anion gap on presentation represented an increase of 9–11 mEq/L over his own baseline.
Initially, there was no mention of the high anion gap—clearly, an oversight. If one presumes that the high anion gap represents accumulation of the anion of an organic acid, which it does in most patients (2), then it is reasonable to expect the plasma total CO2 to fall concomitantly, because the accumulated protons titrate extracellular bicarbonate. Thus, the nearly normal total CO2 of 20 mmol/L may be seen as paradoxical and may have obscured the diagnosis of a high anion gap metabolic acidosis. There are several possible explanations for a discordance between the change in the anion gap and the change in the total CO2 (1). The most obvious in this patient is a superimposed metabolic alkalosis. The patient denied vomiting or alkali ingestion. Therefore, this is mostly likely caused by the use of combination loop and thiazide diuretics for the patient’s underlying congestive cardiomyopathy (3). The patient also had a respiratory acidosis, indicated by the pCO2 above expected value, and this amplified his acidemia.
This patient quite clearly has a lactic acidosis. Common criteria for the diagnosis of lactic acidosis include a plasma lactate concentration in excess of 4 mmol/L (4), usually with plasma pH <7.35. By the time the nephrology service was consulted, the patient had a profound metabolic acidosis and extreme hyperlactatemia. The patient’s anion gap was high on presentation. Thus, he is likely to have had a lactic acidosis on arrival, which worsened rapidly. Interestingly, the prominent dyspnea, out of proportion to the physical manifestations of heart failure on presentation, may have been caused by accumulated lactic acid, which seems to stimulate ventilatory drive in patients with heart failure (5).
The second set of laboratory results shows that the change in the anion gap from the patient’s known baseline was about 30 mEq/L, whereas the plasma lactate concentration was about 19 mmol/L. Thus, the accumulated lactate accounted for slightly over one half of the excess anion gap. This raises the possibility of other causes of the high anion gap acidosis in this patient. Diabetic ketoacidosis, toxic alcohols, such as methanol and ethylene glycol, and salicylate intoxication could cause an anion gap metabolic acidosis of this severity (6). Plasma ketones and salicylates were not detected. A plasma osmolal gap (the difference between the measured and the estimated plasma osmolality) >20 mosmol/kg is a clue to the presence of an alcohol in the circulation (7). The estimated plasma osmolality was 296 mosmol/kg, and the measured plasma osmolality was 311 mosmol/kg. Pyroglutamic acidosis (8) should be considered in any patient presenting with a high anion gap acidosis. This is seen mostly in women (probably because of sex-associated enzymatic differences) who ingest acetaminophen regularly (8). Our patient gave no history of acetaminophen use.
It should be noted, however, that, among patients with an identified organic acidosis, much of the excess anion gap cannot be explained by the measured anions, even after assiduous investigation (2). Likewise, in patients with what seems to be pure lactic acidosis, the lactate concentration explains less than one half of the change in the anion gap (9). (The contribution of pyroglutamic and other organic acids was investigated in a population of critically ill patients with anion gap acidosis and found to be trivial [10].) Thus, although the large discrepancy between the change in the anion gap and the lactate concentration introduces some ambiguity, it is quite typical of patients with lactic acidosis.
There were two important questions for the nephrology consultant to answer. First, what is the likely cause of lactic acidosis in this patient? Second, what is the most effective treatment for the patient?
What Is the Likely Cause of Lactic Acidosis in This Patient?
The conventional clinical laboratory assay for lactic acid detects only l-lactic acid stereoisomer, and therefore, d-lactic acidosis presents as an unexplained high anion gap metabolic acidosis. That is not the case here, and therefore, we will concern ourselves only with l-lactic acidosis.
The causes of l-lactic acidosis (Table 2) can be divided into those associated with delivery of oxygen insufficient to meet demands of metabolizing tissues (type A lactic acidosis) and all others (type B lactic acidosis), including those associated with excessive lactate generation unrelated to oxygen delivery, impaired oxygen use, and impaired lactate metabolism.
Table 2.
Causes of lactic acidosis
| Type A lactic acidosis (oxygen supply: demand mismatch) |
| Shock |
| Sepsis |
| Seizure |
| Regional ischemia |
| Type B lactic acidosis |
| Excessive lactate production |
| Alcohols |
| Ethanol |
| Methanol |
| Ethylene glycol |
| Propylene glycol |
| Fructose metabolic defects |
| Diabetes mellitus |
| Impaired oxygen use |
| Disruption of mitochondrial oxidative phosphorylation |
| Cyanide intoxication |
| Carbon monoxide intoxication |
| Linezolid |
| Biguanides |
| Nucleoside analog reverse transcription inhibitors |
| Acetaminophen intoxication |
| Acquired defects of the TCA cycle |
| Thiamine deficiency |
| Nutritional |
| Cancer |
| Alcoholism |
| Gastrectomy |
| Congenital defects of |
| Pyruvate transport |
| TCA cycle enzymes |
| Pyruvate dehydrogenase complex |
| Impaired lactate use |
| Liver disease |
| Kidney disease |
TCA, tricarboxylic acid.
In the patient at hand, obvious causes of type A lactic acidosis seemed unlikely, at least initially. The patient was not hypotensive on arrival, and the peripheral oxygenation was normal. He did meet several criteria for systemic inflammatory response syndrome, including rapid heart and respiratory rates and leukocytosis, but there was no obvious source of infection, and cultures of blood and urine proved later to be sterile. This made sepsis as a cause of lactic acidosis less likely but did not exclude it. The patient was sensibly covered with broad spectrum antibiotics.
Although he had no history of alcohol use, ingestion of ethanol or another toxic alcohol should be considered in patients with lactic acidosis, and it was in this patient. Metabolism of alcohols by alcohol dehydrogenase lowers the redox potential and may, thus, predispose to lactic acidosis, and propylene glycol may be metabolized to some extent directly to lactate (11). Thus, lactate may contribute modestly to the high anion gap acidosis seen with toxic alcohols but would not cause lactic acidosis of this severity. Additional evidence against a role for toxic alcohols is the normal osmolal gap.
Both types 1 and 2 diabetes mellitus seem to predispose to the development of lactic acidosis (11,12). One large population-based study has shown an incidence of lactic acidosis of 3% among all patients with diabetes mellitus compared with 0.1% in the nondiabetic population (12). The causes of this predisposition are not known but have been postulated to include a propensity for tissue hypoxia because of large and small vessel disease and alterations in pyruvate metabolism (13). In the absence of another cause, however, diabetes will not result in lactic acidosis of the degree seen in this patient.
The patient had been hospitalized several times in the recent past, and there was no prior history of metabolic acidosis; therefore, a congenital error of metabolism can be confidently excluded. Liver enzymes were initially normal, and the renal function had declined only slightly from baseline at the time of presentation; therefore, liver and kidney failures are not tenable explanations for lactic acidosis. There was no exposure to medications other than those prescribed to him. Among the latter, metformin should be considered as a cause of his lactic acidosis.
Metformin as a Cause of Lactic Acidosis
Galega officinalis (goat’s rue or French lilac) was recognized in medieval times as a treatment for diabetes mellitus (14). On the basis of the discovery of its active ingredient, guanidine, several drugs of the biguanide class were formulated to treat diabetes mellitus beginning in the 1950s (15). The first drug of this class in wide use, phenformin, was withdrawn from the United States market in the 1970s because of an unacceptably high incidence of lactic acidosis. Phenformin is highly lipid soluble and caused lactic acidosis by crossing the mitochondrial membrane and inhibiting mitochondrial oxidative phosphorylation and also, by inhibiting gluconeogenesis (11,16). Its descendent, metformin, lacks phenformin’s lipid solubility. It was introduced to the United States market in 1995 after studies showed a 10- to 20-fold reduction in the predisposition to lactic acidosis (17).
Metformin is an insulin sensitizer (14,18); it is ineffective in the absence of insulin. It acts mainly by reducing hepatic gluconeogenesis (14,16,18), in large part by inhibiting mitochondrial oxidative phosphorylation (19) and mitochondrial glycerophosphate dehydrogenase (20). It also has some effect to increase peripheral glucose disposal (14,18). Unlike phenformin, metformin is not metabolized and is eliminated entirely by the kidney. Its plasma clearance is by glomerular filtration and to a greater extent, tubular secretion through a variety of transporters (18). Despite a tremendous volume of distribution, its half-time of elimination is estimated to be only 2.7 hours (18).
Because of its effect to reduce hepatic gluconeogenesis, for which lactate is a substrate, and because it acts partly by inhibiting mitochondrial oxidative phosphorylation and lowering the mitochondrial redox state (20), it is somewhat surprising that metformin administration has been found to cause only a slight increase in basal and postprandial plasma lactate concentrations (14,18). Among the many studies corroborating metformin’s low potential to cause lactic acidosis are a systematic review of 347 clinical trials, finding no patients with fatal or nonfatal lactic acidosis in 70,490 subject-years of metformin exposure (21), and a case-control study of over 50,000 patients with type 2 diabetes mellitus, yielding about three cases of lactic acidosis per 100,000 patient-years of metformin use—an incidence no different from that among patients using sulfonylurea drugs (22). Nonetheless, very shortly after the drug was marketed in the United States, a case series of metformin-associated lactic acidosis (MALA) was reported (23). This was followed by hundreds of case reports and a dozen case series over the past two decades (24). However, given the predisposition of patients with diabetes mellitus to hyperlactatemia and lactic acidosis (12), questions arose regarding metformin’s role in the reported lactic acidosis. Does metformin cause lactic acidosis, or is it merely associated with it?
The fact that metformin is capable of causing lactic acidosis is obvious from patients with pure metformin overdose (18). This serves to emphasize that, from both mechanistic and logical standpoints, metformin accumulation is the risk for lactic acidosis (15), with studies suggesting that a metformin plasma level of at least 40 mg/L (10 times the therapeutic level) may be necessary to cause lactic acidosis (25). On this basis, there are three possible relevant conditions: (1) metformin-independent lactic acidosis (in which metformin cannot possibly be implicated), (2) metformin-induced lactic acidosis (in which no other possible cause of lactic acidosis is present), and (3) MALA (in which metformin is among the factors that might have caused lactic acidosis) (18). Strictly speaking, these defined conditions require measurement of plasma metformin concentration; the latter two conditions may only be considered in the presence of a high metformin concentration. A plasma metformin concentration was not measured in this patient. At our institution (and I suspect, most other large, tertiary referral centers), plasma metformin measurement cannot be performed in-house. Specimens are sent to a reference laboratory, and results may take days to return. Thus, metformin levels usually cannot help to guide acute management. Furthermore, metformin levels correlate poorly with pH, plasma lactate levels, or clinical outcomes (26,27). A diagnosis of MALA in this patient is speculative and based on circumstantial evidence, but this working diagnosis may have important therapeutic implications.
Our patient had a number of risk factors for MALA (Table 3). His plasma creatinine on arrival to the emergency department was higher than his baseline, perhaps because of his increased diuretic regimen. He carried a diagnosis of severe congestive heart failure, with frequent episodes of decompensation, and was in decompensated congestive heart failure on admission. Whether a diagnosis of stable congestive heart failure should contraindicate the use of metformin is a matter of some debate (28). Mechanistically, however, decompensated congestive heart failure is likely to potentiate metformin’s contribution to lactic acidosis because of decreased tissue perfusion (5). Finally, the patient’s dose of metformin had been increased to the recommended maximum (even for patients with no risk factors) a few weeks before admission. This combination of factors placed the patient at extremely high risk of lactic acidosis. Therefore, MALA with heart failure and, possibly, sepsis was the presumed diagnosis.
Table 3.
Contraindications to metformin use (14)
| Impaired kidney function |
| Age >80 yr |
| Liver disease |
| Congestive heart failure |
| Respiratory disease with risk for hypoxemia |
| Heavy ethanol use |
| Intravascular radiocontrast administration |
What Is the Most Effective Treatment for the Patient?
Apart from supportive care, there are several possible therapeutic approaches to patients with lactic acidosis in general: treatment of the underlying cause, raising the systemic pH, and removal of lactate from the blood. Only the first of these is not controversial. For patients with type A lactic acidosis, ameliorating the cause of lactic acidosis involves improving tissue perfusion. Patients with shock should be treated with fluids and vasoactive medications to optimize their hemodynamics. To complicate even this straightforward recommendation, however, it must be noted that catecholamines (epinephrine more than norepinephrine) cause an increase in plasma lactate concentration (29). Treatment of type B lactic acidosis may involve removal of the causative toxin as discussed below.
Alkali therapy often is used to treat the acidemia that typically accompanies lactic acidosis. This practice is based, at least partly, on the recognition that acidemia has important hemodynamic effects. It impairs myocardial contractility (30,31) and causes venoconstriction with central vascular congestion (32), arterial vasodilation and resistance to the vasoconstrictive effects of catecholamines, especially when the pH is <7.10–7.20 (33). Although alkalinization has theoretical appeal, no animal or human studies of lactic acidosis have shown an improvement in myocardial performance (33) or survival with sodium bicarbonate administration (34), even among patients with the most profound acidemia (35). Indeed, there is some evidence that sodium bicarbonate administration for lactic acidosis may be harmful (36,37). There are several possible reasons for this. First, bicarbonate may cause paradoxical intracellular acidification because of the increased generation of cell-permeant CO2 by mass action effect on the carbonic acid equilibrium (38). Second, the increase in extracellular pH reduces ionized calcium concentration because of chelation by albumin (39), impairing myocardial contractility (40). Third, alkalinization may reduce tissue oxygen delivery by the Bohr effect on hemoglobin oxygen affinity. Fourth, bicarbonate administration may increase lactate generation by upregulating the pH-dependent rate-limiting step in the glycolytic pathway. Fifth, administration of large volumes of sodium bicarbonate may cause intravascular fluid overload. All this notwithstanding, international critical care guidelines for sepsis recommend giving sodium bicarbonate if the blood pH is <7.15 (41).
Precise calculation of bicarbonate deficit would require an accurate estimate of the volume of distribution of bicarbonate. The apparent bicarbonate distribution space varies widely, however, and in inverse proportion to the bicarbonate concentration (42). Even if the deficit could be accurately calculated, the effect of administered bicarbonate on the final bicarbonate concentration may be far from the predicted value because of ongoing acid generation and the contraction of the apparent bicarbonate distribution space as the bicarbonate concentration rises. Therefore, it may be most practical to administer a short-term or bolus infusion of sodium bicarbonate (e.g., 1–2 mmol/kg body wt intravenously), rechecking the arterial blood gases and pH at the end of the infusion and frequently thereafter to guide additional therapy. Care must be taken to ensure the patient’s ventilation is appropriate at the start of the infusion (i.e., pCO2 no greater than the plasma bicarbonate concentration +15), because the bicarbonate infusion will lead to increased CO2 generation and may, thus, worsen the acidemia. Finally, continuous bicarbonate infusions carry a risk of overshoot alkalemia, especially if the patient’s underlying cause of lactic acidosis is improving and there is metabolism of the lactate to bicarbonate.
Tris-hydroxymethyaminomethane is an amino alcohol that buffers without generating CO2. It has the theoretical advantage, therefore, of avoiding both the superimposed respiratory acidosis that may accompany bicarbonate administration and its consequent intracellular acidification (32). There is no evidence to support the use of Tris-hydroxymethyaminomethane in this situation.
Renal replacement therapy (RRT) offers a number of theoretical and practical advantages over alkali infusion for the treatment of lactic acidosis. First, RRT can deliver large quantities of base without the risk of volume overload. Second, RRT may reduce plasma lactate concentration. (This may not necessarily be a benefit, because lactate serves as a preferred energy substrate for some tissues under conditions of metabolic stress and may, thus, confer an adaptive advantage [34].) Third, RRT has the potential to maintain the plasma ionized calcium concentration while raising pH, thus potentially improving myocardial contractility (43). There are few studies of RRT relevant to the treatment of lactic acidosis, and most are in the setting of sepsis with hemodynamic instability (44). Almost all involve continuous RRT (CRRT). Early uncontrolled studies showed the feasibility of CRRT modalities in the treatment of patients with renal failure and lactic acidosis (45–47) and emphasized the metabolic advantage of bicarbonate- over lactate-buffered dialysate (46). A recent single-center observational cohort study of 116 patients with AKI and lactic acidosis showed that CRRT using bicarbonate-buffered fluids can reduce blood lactate levels and raise pH (48). The effect of CRRT on survival in patients with lactic acidosis has never been assessed, and therefore, the use of RRT in this patient cannot be considered evidence based.
Our patient had lactic acidosis in association with suspected metformin intoxication. Some investigators (49) (but not all [18]) have shown a correlation between plasma metformin concentration and severity of acidosis, and most patients with MALA have impaired renal function. Thus, metformin removal by dialysis has figured prominently in the management of patients with MALA (50). Metformin is readily dialyzable, owing to its low molecular weight and lack of protein binding (44). Because of its very high volume of distribution and two-compartment elimination kinetics (50), metformin’s removal may be best accomplished with prolonged extracorporeal therapy, either intermittent (49) or continuous (51). In one series, it took an average of 15 hours of conventional hemodialysis to reduce metformin levels to <20% of predialysis levels (49). CRRT may be the preferred modality for patients with hemodynamic instability, such as in this patient. The ambiguities regarding the most efficacious modality of RRT in MALA and lactic acidosis in general point to the need for future controlled clinical trials in this area.
Back to the Patient
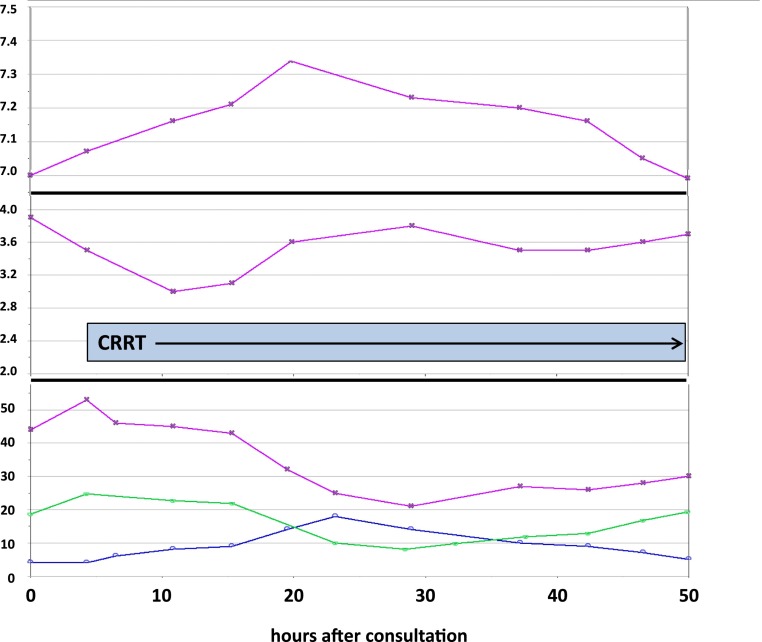
Figure 1 shows the patient’s biochemical course. He was receiving sodium bicarbonate intravenously at the time of the consultation, and the pH had risen from 6.97 to 7.08, with a concomitant fall in ionized calcium. During that time, there was a sharp decline in arterial pressure, prompting infusions of maximum doses of dopamine, dobutamine, epinephrine, phenylephrine, norepinephrine, and vasopressin. There was a sharp increase in transaminase concentrations (peak aspartate transaminase =14,650 IU/L), which were felt to be caused by passive hepatic congestion.
Figure 1.
Plasma chemistries over time. Top panel shows arterial pH, middle panel shows ionized calcium (reference range =4.4–5.0 mg/dl), and bottom panel shows total CO2 (blue line), anion gap (magenta line), and lactate (green line). CRRT, continuous RRT.
Because of the profound acidemia and hyperkalemia with oliguria in this hemodynamically unstable patient—and the possibility that metformin may have been implicated—we initiated continuous venovenous hemodialysis with a bicarbonate bath, as noted in Figure 1. With that, the anion gap and plasma lactate concentration fell, and the plasma total CO2, the pH, and with some delay, the ionized calcium rose. Despite the biochemical improvement, there was only a slight improvement in the BP, which remained in the range of 105/75 mmHg on maximum inotropic and pressor support. All biochemical parameters showed maximum improvement by about 24 hours of CRRT, after which they began to deteriorate along with the patient’s hemodynamics. The patient died on the third hospital day.
Questions
Ron Zanger, MD, Attending Nephrologist, Cooper University Health Care
Given this patient’s only modest renal impairment on presentation, significant metformin accumulation seems unlikely, even with his relatively high prescribed dose. Do you think metformin was likely to be the sole cause of his severe lactic acidosis?
Dr. Lawrence Weisberg
On arrival, the patient had impaired renal function, the duration of which was unclear. Therefore, it is unclear to what extent the metformin, prescribed at a very high dose, had accumulated. His lactic acidosis seemed out of proportion to the severity of his heart failure on presentation, suggesting an additional cause, the most likely of which is metformin. In the absence of a plasma metformin level, it is impossible to definitely assess the drug’s contribution. Having said that, however, our patient, like the majority of patients who present with lactic acidosis while taking metformin, had other risk factors for lactic acidosis (25,52), the most important of which may have been his decompensated congestive heart failure. The severe lactic acidosis, therefore, was probably the result of a nefarious synergy between the metformin and his decompensated congestive heart failure.
Jason A. Kline, MD, Attending Nephrologist, Cooper University Health Care
This patient had a dreadful clinical course. Is this typical of patients with MALA?
Dr. Lawrence Weisberg
The prognosis of patients with lactic acidosis, in general, is quite variable and depends on the underlying comorbidities, with mortality approaching 100% in patients with lactic acidosis after cardiac arrest (53). The mortality of patients with MALA seems to be lower than that of patients with lactic acidosis of other causes (54), but again, it depends on the circumstances. Patients whose lactic acidosis is caused by intentional metformin overdose have a very low mortality rate, whereas those for whom metformin is one of several causes of lactic acidosis—as is the case with our patient—have a mortality rate around 50% (49,54). Our patient’s course is perhaps best appreciated in two phases. In the first phase, he presented with rapidly progressive lactic acidosis out of proportion to his congestive heart failure, likely as a result of high metformin levels. The second phase was entered when the acidemia reached a critical level. At that point, the patient’s hemodynamics deteriorated drastically. This further reduced his organ perfusion and increased the generation of lactate, the clearance of which was impaired by acute kidney failure and hepatic congestion. The combination of severely impaired myocardial contractility and maximum doses of catecholamines, causing intense peripheral vasoconstriction, likely led to additional tissue hypoperfusion, establishing a vicious, downward spiral. This patient serves to emphasize that most patients with MALA have additional causes of lactic acidosis, the nature and severity of which determine their prognosis.
Christopher B. McFadden, Attending Nephrologist, Cooper University Health Care
Is metformin strictly contraindicated in patients with CKD, or can its dose be adjusted for renal function?
Dr. Lawrence Weisberg
The metformin package insert contraindicates the use of the drug when the plasma creatinine concentration is ≥1.4 mg/dl in women and ≥1.5 mg/dl in men (55). Some investigators worry that such an absolute restriction deprives too many patients of a medication with a highly favorable risk to benefit profile (18,25). Several large studies suggest that an adjusted dose of metformin may be prescribed safely to patients whose eGFR is as low as 30 ml/min (15,56). Adam and O’Brien (25) have gone so far as to propose a dose-adjustment scheme for patients across the full range of kidney function, including dialysis dependence. However, such liberalization of metformin use is controversial (57). Furthermore, it must be borne in mind that CKD is the single biggest risk for AKI, which in turn, seems to pose the greatest risk for MALA (15).
William D. Sirover, MD, Attending Nephrologist, Cooper University Health Care
Why did the patient’s plasma potassium concentration rise acutely with development of the severe lactic acidosis?
Dr. Lawrence Weisberg
At first blush, this is somewhat unexpected, given the conventional wisdom that efflux of cellular potassium, with consequent hyperkalemia, accompanies mineral (hyperchloremic) acidosis but not organic acidosis, like lactic or ketoacidosis (58). Of note, however, is Fulop's original case series (59), which is widely cited in support of this contention, revealed that some patients with organic acidoses did develop hyperkalemia. Indeed, among patients with the highest potassium concentrations in that series were those with phenformin-associated lactic acidosis (59). It is, therefore, plausible, although speculative, that the biguanide-induced failure of ATP generation impairs Na,K-ATPase and allows net K efflux from cells. As with diabetic ketoacidosis, the acidemia per se does not cause the hyperkalemia; rather, there is a common cause of the acidosis and the hyperkalemia.
Disclosures
None.
Footnotes
Published online ahead of print. Publication date available at www.cjasn.org.
References
- 1.Kraut JA, Madias NE: Serum anion gap: Its uses and limitations in clinical medicine. Clin J Am Soc Nephrol 2: 162–174, 2007 [DOI] [PubMed] [Google Scholar]
- 2.Gabow PA, Kaehny WD, Fennessey PV, Goodman SI, Gross PA, Schrier RW: Diagnostic importance of an increased serum anion gap. N Engl J Med 303: 854–858, 1980 [DOI] [PubMed] [Google Scholar]
- 3.Peixoto AJ, Alpern RJ: Treatment of severe metabolic alkalosis in a patient with congestive heart failure. Am J Kidney Dis 61: 822–827, 2013 [DOI] [PubMed] [Google Scholar]
- 4.DuBose TD: Disorders of acid-base balance. In: Brenner & Rector's The Kidney, 9th Ed., edited by Maarten WT, Chertow GM, Marsden PA, Skorecki K, Yu ASL, Brenner BM, Philadelphia, Saunders, 2012, pp 595–639. [Google Scholar]
- 5.Tang WHW, Francis GS: Clinical evaluation of heart failure. In: Heart Failure: A Companion to Braunwald's Heart Disease, 2nd Ed., edited by Mann DL, Philadelphia, Saunders, 2010, p 516 [Google Scholar]
- 6.Moe OW, Fuster D: Clinical acid-base pathophysiology: Disorders of plasma anion gap. Best Pract Res Clin Endocrinol Metab 17: 559–574, 2003 [DOI] [PubMed] [Google Scholar]
- 7.Kraut JA, Kurtz I: Toxic alcohol ingestions: Clinical features, diagnosis, and management. Clin J Am Soc Nephrol 3: 208–225, 2008 [DOI] [PubMed] [Google Scholar]
- 8.Emmett M: Acetaminophen toxicity and 5-oxoproline (pyroglutamic acid): A tale of two cycles, one an ATP-depleting futile cycle and the other a useful cycle. Clin J Am Soc Nephrol 9: 191–200, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Levraut J, Bounatirou T, Ichai C, Ciais JF, Jambou P, Hechema R, Grimaud D: Reliability of anion gap as an indicator of blood lactate in critically ill patients. Intensive Care Med 23: 417–422, 1997 [DOI] [PubMed] [Google Scholar]
- 10.Moviat M, Terpstra AM, Ruitenbeek W, Kluijtmans LA, Pickkers P, van der Hoeven JG: Contribution of various metabolites to the “unmeasured” anions in critically ill patients with metabolic acidosis. Crit Care Med 36: 752–758, 2008 [DOI] [PubMed] [Google Scholar]
- 11.Adeva-Andany M, López-Ojén M, Funcasta-Calderón R, Ameneiros-Rodríguez E, Donapetry-García C, Vila-Altesor M, Rodríguez-Seijas J: Comprehensive review on lactate metabolism in human health. Mitochondrion 17: 76–100, 2014 [DOI] [PubMed] [Google Scholar]
- 12.Scale T, Harvey JN: Diabetes, metformin and lactic acidosis. Clin Endocrinol (Oxf) 74: 191–196, 2011 [DOI] [PubMed] [Google Scholar]
- 13.Gray LR, Tompkins SC, Taylor EB: Regulation of pyruvate metabolism and human disease. Cell Mol Life Sci 71: 2577–2604, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Bailey CJ, Turner RC: Metformin. N Engl J Med 334: 574–579, 1996 [DOI] [PubMed] [Google Scholar]
- 15.Lalau JD: Lactic acidosis induced by metformin: Incidence, management and prevention. Drug Saf 33: 727–740, 2010 [DOI] [PubMed] [Google Scholar]
- 16.Stumvoll M, Nurjhan N, Perriello G, Dailey G, Gerich JE: Metabolic effects of metformin in non-insulin-dependent diabetes mellitus. N Engl J Med 333: 550–554, 1995 [DOI] [PubMed] [Google Scholar]
- 17.Lalau JD, Race JM: Lactic acidosis in metformin therapy: Searching for a link with metformin in reports of ‘metformin-associated lactic acidosis.’ Diabetes Obes Metab 3: 195–201, 2001 [DOI] [PubMed] [Google Scholar]
- 18.Lalau JD, Arnouts P, Sharif A, De Broe ME: Metformin and other antidiabetic agents in renal failure patients. Kidney Int 87: 308–322, 2015 [DOI] [PubMed] [Google Scholar]
- 19.Kirpichnikov D, McFarlane SI, Sowers JR: Metformin: An update. Ann Intern Med 137: 25–33, 2002 [DOI] [PubMed] [Google Scholar]
- 20.Madiraju AK, Erion DM, Rahimi Y, Zhang XM, Braddock DT, Albright RA, Prigaro BJ, Wood JL, Bhanot S, MacDonald MJ, Jurczak MJ, Camporez JP, Lee HY, Cline GW, Samuel VT, Kibbey RG, Shulman GI: Metformin suppresses gluconeogenesis by inhibiting mitochondrial glycerophosphate dehydrogenase. Nature 510: 542–546, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Salpeter SR, Greyber E, Pasternak GA, Salpeter EE: Risk of fatal and nonfatal lactic acidosis with metformin use in type 2 diabetes mellitus. Cochrane Database Syst Rev 4: CD002967, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bodmer M, Meier C, Krähenbühl S, Jick SS, Meier CR: Metformin, sulfonylureas, or other antidiabetes drugs and the risk of lactic acidosis or hypoglycemia: A nested case-control analysis. Diabetes Care 31: 2086–2091, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Misbin RI, Green L, Stadel BV, Gueriguian JL, Gubbi A, Fleming GA: Lactic acidosis in patients with diabetes treated with metformin. N Engl J Med 338: 265–266, 1998 [DOI] [PubMed] [Google Scholar]
- 24.Kajbaf F, Lalau JD: Mortality rate in so-called “metformin-associated lactic acidosis”: A review of the data since the 1960s. Pharmacoepidemiol Drug Saf 23: 1123–1127, 2014 [DOI] [PubMed] [Google Scholar]
- 25.Adam WR, O’Brien RC: A justification for less restrictive guidelines on the use of metformin in stable chronic renal failure. Diabet Med 31: 1032–1038, 2014 [DOI] [PubMed] [Google Scholar]
- 26.Lalau JD, Race JM: Lactic acidosis in metformin-treated patients. Prognostic value of arterial lactate levels and plasma metformin concentrations. Drug Saf 20: 377–384, 1999 [DOI] [PubMed] [Google Scholar]
- 27.Vecchio S, Giampreti A, Petrolini VM, Lonati D, Protti A, Papa P, Rognoni C, Valli A, Rocchi L, Rolandi L, Manzo L, Locatelli CA: Metformin accumulation: Lactic acidosis and high plasmatic metformin levels in a retrospective case series of 66 patients on chronic therapy. Clin Toxicol (Phila) 52: 129–135, 2014 [DOI] [PubMed] [Google Scholar]
- 28.Eurich DT, Weir DL, Majumdar SR, Tsuyuki RT, Johnson JA, Tjosvold L, Vanderloo SE, McAlister FA: Comparative safety and effectiveness of metformin in patients with diabetes mellitus and heart failure: Systematic review of observational studies involving 34,000 patients. Circ Heart Fail 6: 395–402, 2013 [DOI] [PubMed] [Google Scholar]
- 29.Levy B: Bench-to-bedside review: Is there a place for epinephrine in septic shock? Crit Care 9: 561–565, 2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Marsh JD, Margolis TI, Kim D: Mechanism of diminished contractile response to catecholamines during acidosis. Am J Physiol 254: H20–H27, 1988 [DOI] [PubMed] [Google Scholar]
- 31.Teplinsky K, O’Toole M, Olman M, Walley KR, Wood LD: Effect of lactic acidosis on canine hemodynamics and left ventricular function. Am J Physiol 258: H1193–H1199, 1990 [DOI] [PubMed] [Google Scholar]
- 32.Kraut JA, Kurtz I: Use of base in the treatment of severe acidemic states. Am J Kidney Dis 38: 703–727, 2001 [DOI] [PubMed] [Google Scholar]
- 33.Kraut JA, Madias NE: Treatment of acute metabolic acidosis: A pathophysiologic approach. Nat Rev Nephrol 8: 589–601, 2012 [DOI] [PubMed] [Google Scholar]
- 34.Rachoin JS, Weisberg LS, McFadden CB: Treatment of lactic acidosis: Appropriate confusion. J Hosp Med 5: E1–E7, 2010 [DOI] [PubMed] [Google Scholar]
- 35.Cooper DJ, Walley KR, Wiggs BR, Russell JA: Bicarbonate does not improve hemodynamics in critically ill patients who have lactic acidosis. A prospective, controlled clinical study. Ann Intern Med 112: 492–498, 1990 [DOI] [PubMed] [Google Scholar]
- 36.Arieff AI, Leach W, Park R, Lazarowitz VC: Systemic effects of NaHCO3 in experimental lactic acidosis in dogs. Am J Physiol 242: F586–F591, 1982 [DOI] [PubMed] [Google Scholar]
- 37.Zahler R, Barrett E, Majumdar S, Greene R, Gore JC: Lactic acidosis: Effect of treatment on intracellular pH and energetics in living rat heart. Am J Physiol 262: H1572–H1578, 1992 [DOI] [PubMed] [Google Scholar]
- 38.Levraut J, Garcia P, Giunti C, Ichai C, Bouregba M, Ciebiera JP, Payan P, Grimaud D: The increase in CO2 production induced by NaHCO3 depends on blood albumin and hemoglobin concentrations. Intensive Care Med 26: 558–564, 2000 [DOI] [PubMed] [Google Scholar]
- 39.Pedersen KO: Binding of calcium to serum albumin. II. Effect of pH via competitive hydrogen and calcium ion binding to the imidazole groups of albumin. Scand J Clin Lab Invest 29: 75–83, 1972 [DOI] [PubMed] [Google Scholar]
- 40.Lang RM, Fellner SK, Neumann A, Bushinsky DA, Borow KM: Left ventricular contractility varies directly with blood ionized calcium. Ann Intern Med 108: 524–529, 1988 [DOI] [PubMed] [Google Scholar]
- 41.Dellinger RP, Levy MM, Carlet JM, Bion J, Parker MM, Jaeschke R, Reinhart K, Angus DC, Brun-Buisson C, Beale R, Calandra T, Dhainaut JF, Gerlach H, Harvey M, Marini JJ, Marshall J, Ranieri M, Ramsay G, Sevransky J, Thompson BT, Townsend S, Vender JS, Zimmerman JL, Vincent JL; International Surviving Sepsis Campaign Guidelines Committee; American Association of Critical-Care Nurses; American College of Chest Physicians; American College of Emergency Physicians; Canadian Critical Care Society; European Society of Clinical Microbiology and Infectious Diseases; European Society of Intensive Care Medicine; European Respiratory Society; International Sepsis Forum; Japanese Association for Acute Medicine; Japanese Society of Intensive Care Medicine; Society of Critical Care Medicine; Society of Hospital Medicine; Surgical Infection Society; World Federation of Societies of Intensive and Critical Care Medicine: Surviving Sepsis Campaign: International guidelines for management of severe sepsis and septic shock: 2008. Crit Care Med 36: 296–327, 2008 [DOI] [PubMed] [Google Scholar]
- 42.Fernandez PC, Cohen RM, Feldman GM: The concept of bicarbonate distribution space: The crucial role of body buffers. Kidney Int 36: 747–752, 1989 [DOI] [PubMed] [Google Scholar]
- 43.Orchard CH, Kentish JC: Effects of changes of pH on the contractile function of cardiac muscle. Am J Physiol 258: C967–C981, 1990 [DOI] [PubMed] [Google Scholar]
- 44.Cerdá J, Tolwani A, Gibney N, Tiranathanagul K: Renal replacement therapy in special settings: Extracorporeal support devices in liver failure. Semin Dial 24: 197–202, 2011 [DOI] [PubMed] [Google Scholar]
- 45.Barton IK, Hilton PJ, Treacher DF, Bradley RD: Treatment of combined renal failure and lactic acidosis by haemofiltration. Clin Intensive Care 3: 196–198, 1992 [PubMed] [Google Scholar]
- 46.Hilton PJ, Taylor J, Forni LG, Treacher DF: Bicarbonate-based haemofiltration in the management of acute renal failure with lactic acidosis. QJM 91: 279–283, 1998 [DOI] [PubMed] [Google Scholar]
- 47.Kirschbaum B, Galishoff M, Reines HD: Lactic acidosis treated with continuous hemodiafiltration and regional citrate anticoagulation. Crit Care Med 20: 349–353, 1992 [DOI] [PubMed] [Google Scholar]
- 48.De Corte W, Vuylsteke S, De Waele JJ, Dhondt AW, Decruyenaere J, Vanholder R, Hoste EA: Severe lactic acidosis in critically ill patients with acute kidney injury treated with renal replacement therapy. J Crit Care 29: 650–655, 2014 [DOI] [PubMed] [Google Scholar]
- 49.Seidowsky A, Nseir S, Houdret N, Fourrier F: Metformin-associated lactic acidosis: A prognostic and therapeutic study. Crit Care Med 37: 2191–2196, 2009 [DOI] [PubMed] [Google Scholar]
- 50.Lalau JD, Andrejak M, Morinière P, Coevoet B, Debussche X, Westeel PF, Fournier A, Quichaud J: Hemodialysis in the treatment of lactic acidosis in diabetics treated by metformin: A study of metformin elimination. Int J Clin Pharmacol Ther Toxicol 27: 285–288, 1989 [PubMed] [Google Scholar]
- 51.Dichtwald S, Weinbroum AA, Sorkine P, Ekstein MP, Dahan E: Metformin-associated lactic acidosis following acute kidney injury. Efficacious treatment with continuous renal replacement therapy. Diabet Med 29: 245–250, 2012 [DOI] [PubMed] [Google Scholar]
- 52.van Berlo-van de Laar IR, Vermeij CG, Doorenbos CJ: Metformin associated lactic acidosis: Incidence and clinical correlation with metformin serum concentration measurements. J Clin Pharm Ther 36: 376–382, 2011 [DOI] [PubMed] [Google Scholar]
- 53.Andersen LW, Mackenhauer J, Roberts JC, Berg KM, Cocchi MN, Donnino MW: Etiology and therapeutic approach to elevated lactate levels. Mayo Clin Proc 88: 1127–1140, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Friesecke S, Abel P, Roser M, Felix SB, Runge S: Outcome of severe lactic acidosis associated with metformin accumulation. Crit Care 14: R226, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Glucophage: Glucophage Package Insert. Available at: http://packageinserts.bms.com/pi/pi_glucophage_xr.pdf. Accessed October 12, 2014
- 56.Kajbaf F, Arnouts P, de Broe M, Lalau JD: Metformin therapy and kidney disease: A review of guidelines and proposals for metformin withdrawal around the world. Pharmacoepidemiol Drug Saf 22: 1027–1035, 2013 [DOI] [PubMed] [Google Scholar]
- 57.Fantus IG: Metformin’s contraindications: Needed for now. CMAJ 173: 505–507, 2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Lee Hamm L, Hering-Smith KS, Nakhoul NL: Acid-base and potassium homeostasis. Semin Nephrol 33: 257–264, 2013 [DOI] [PubMed] [Google Scholar]
- 59.Fulop M: Serum potassium in lactic acidosis and ketoacidosis. N Engl J Med 300: 1087–1089, 1979 [DOI] [PubMed] [Google Scholar]