Abstract
Tracheobronchomalacia is a common congenital defect in which the walls of the trachea and bronchi lack of adequate cartilage required for support of the airways. Deletion of Wls, a cargo receptor mediating Wnt ligand secretion, in the embryonic endoderm using ShhCre mice inhibited formation of tracheal-bronchial cartilaginous rings. The normal dorsal-ventral patterning of tracheal mesenchyme was lost. Smooth muscle cells, identified by Acta2 staining, were aberrantly located in ventral mesenchyme of the trachea, normally the region of Sox9 expression in cartilage progenitors. Wnt/β-catenin activity, indicated by Axin2 LacZ reporter, was decreased in tracheal mesenchyme of Wlsf/f;ShhCre/+ embryos. Proliferation of chondroblasts was decreased and reciprocally, proliferation of smooth muscle cells was increased in Wlsf/f;ShhCre/+ tracheal tissue. Expression of Tbx4, Tbx5, Msx1 and Msx2, known to mediate cartilage and muscle patterning, were decreased in tracheal mesenchyme of Wlsf/f;ShhCre/+ embryos. Ex vivo studies demonstrated that Wnt7b and Wnt5a, expressed by the epithelium of developing trachea, and active Wnt/β-catenin signaling are required for tracheal chondrogenesis before formation of mesenchymal condensations. In conclusion, Wnt ligands produced by the tracheal epithelium pattern the tracheal mesenchyme via modulation of gene expression and cell proliferation required for proper tracheal cartilage and smooth muscle differentiation.
Keywords: Tracheobronchomalacia, cartilage, airways, Wls, Sox 9, αSMA
Introduction
The ventral region of the mammalian trachea is supported by incomplete circumferential cartilaginous rings and dorsally by the trachealis smooth muscle. Dorsal-ventral patterning of cartilage and muscle protects the upper airways from injury and provides flexibility during ventilation and cough, preventing airway collapse during the respiratory cycle. Tracheobronchomalacia is a congenital malformation in which the walls of the conducting airways lack adequate cartilage. While tracheobronchomalacia is a common disorder (1:3000/live births) (Boogaard et al., 2005; Kenny AP, 2013), its etiology is poorly understood. Tracheal and bronchial structures forming the airway are derived from the anterior foregut endoderm and its interactions with surrounding mesenchyme, as the airways separate from the esophagus during the embryonic period at approximately week 12 of gestation in humans (Fausett and Klingensmith, 2012). Given the prevalence of tracheobronchomalacia and the challenges associated with corrective surgery, understanding the mechanisms regulating the formation of the tracheal cartilage will provide the framework to prevent and treat this condition (Carden et al., 2005; Majid et al., 2010).
A recent study proposed that the precise apposition of chondroblasts and myoblasts establishes the boundaries between cartilage and muscle, thus generating a structure needed for rigidity and flexibility characteristic of the trachea (Hines et al 2013). It is presently unclear how dorsal-ventral patterning of developing tracheal mesenchyme is established. Cartilage development is initiated by the commitment of subsets of mesenchymal cells surrounding the developing trachea, towards a chondrogenic lineage (McAteer, 1984). Mesenchymal cells derived from the splanchnic mesoderm, positioned ventral to the developing tracheal tube express the transcription factor Sox9 as early as E9 (Elluru and Whitsett, 2004; Hines et al., 2013). Sox9 is a key regulator of cartilage and bone development (Akiyama, 2008; Akiyama et al., 2002) and its role in tracheal cartilage has been demonstrated (Arora et al., 2012; Elluru and Whitsett, 2004; Hines et al., 2013; Park et al., 2010; Turcatel et al., 2013). Signaling mechanisms controlling the initial specification of tracheal cartilage progenitors are poorly defined; however, a number of transcription factors, receptors and ligands that influence Sox9 expression in the mesenchyme of the trachea have been identified. The transcription factors Tbx4 and Tbx5, expressed by the tracheal mesenchyme promote Sox9 expression and mesenchymal condensations required for tracheal cartilage formation (Arora et al., 2012). Shh, a signaling molecule expressed by the tracheal epithelium, is required for expression of Sox9 in the ventral tracheal mesenchyme by a mechanism that involves modulation of Bmp4 signaling (Miller et al., 2004; Park et al., 2010). Epithelial transcription factor Sox2 is required for tracheal cartilage formation via induction of Shh signaling (Que et al., 2009; Tompkins et al., 2009). The calcium channel Cav3.2, present in both the epithelium and the mesenchyme of the trachea promotes Sox9 expression via the activation of the NFAT signaling pathway (Lin et al., 2014). The kinases Mek1 and Mek2, present in both epithelium and mesenchyme of developing trachea, induce Sox9 expression via activation of ERK/MAPK signaling (Boucherat et al., 2014). Mesenchymal Fgf10 and its epithelial receptor Fgfr2 mediate the periodic pattern of expression of Shh that influences the formation of cartilaginous rings (Sala et al., 2011). Fgf18, expressed by the tracheal mesenchyme, promotes proliferation of tracheal chondrocytes by inducing Sox9 expression (Elluru et al, 2009). Thus, epithelial-mesenchymal crosstalk is required for specification and differentiation of tracheal cartilage, resembling the mesenchymal-epithelial interactions that govern cell differentiation in developing lung (Shannon and Hyatt, 2004).
Wnt signaling plays a critical role in specification and early patterning of respiratory tract (Goss et al., 2009; Harris-Johnson et al., 2009; Miller et al., 2012). Germ line deletion of Wnt5a, which is expressed in the mesenchyme and epithelium of E11.5 developing trachea, disrupted cartilage rings and caused shortening of the trachea (Li et al., 2002). Likewise, ablation of the receptor ROR2, which mediates Wnt5a-induced Wnt signaling, caused miss patterning of cartilage and shortening of the trachea (Oishi et al., 2003). Deletion of β-catenin from the tracheal epithelium caused incomplete cartilaginous rings, while deletion of β-catenin from the tracheal mesenchyme produced a shorter trachea that underwent degeneration by E18.5 (Bell et al., 2008; De Langhe et al., 2008; Yin et al., 2008). Deletion of Wnt7b, expressed by the respiratory epithelium and known to mediate Wnt/β-catenin signaling, caused incomplete cartilaginous rings (Rajagopal et al., 2008). Wnt signaling is also critical for proliferation and differentiation of pulmonary muscle cells (Cohen et al., 2009; De Langhe et al., 2008; Goss et al., 2011; Shu et al., 2002; Yin et al., 2008). Wnt7b promotes vascular smooth muscle differentiation via a mechanism that involves PDGF signaling (Cohen et al., 2009), while Wnt2, produced by the mesenchyme, promotes differentiation of airway smooth muscle by inducing the expression of the ligand Fgf10 and Myocd (myocardin), a key regulator of smooth muscle differentiation (Goss et al., 2011). While the genetic data support a role for Wnt signaling in tracheal cartilage formation and suggest a putative role in tracheal muscle differentiation, precise mechanisms mediating these processes remain unclear.
To define the role of Wnt signaling in dorsal-ventral patterning of developing trachea, we generated a mouse model wherein Wntless (Wls), a cargo receptor that mediates Wnt ligand secretion (Banziger et al., 2006; Goodman et al., 2006), was conditionally deleted either in epithelial cells of the foregut endoderm or mesenchyme of the developing respiratory tract. Epithelial Wls was required for activation of canonical Wnt signaling in the tracheal mesenchyme. Wnt ligands produced by epithelial cells of the conducting airways were required for proper specification of the tracheal bronchial cartilage. Deletion of Wls from the tracheal epithelium increased proliferation of smooth muscle cells causing thickening and expansion of the muscle layer into the ventral region of the trachea at sites where cartilage is normally formed. Thus, epithelial Wnt signaling promotes dorsal-ventral patterning of the trachea, influencing differentiation and proliferation of tracheal cartilage and smooth muscle cells.
Materials and Methods
Mouse Breeding and Genotyping: Animals were housed in pathogen-free conditions and handled according to protocols approved by CCHMC Institutional Animal Care and Use Committee (Cincinnati, OH USA). Generation of the Wntless (Wls) conditional knockout (CKO) mouse has been described (Carpenter et al., 2010). Wlsf/f;ShhCre/+ embryos were obtain by breeding Wls f/f mice with ShhCre mice and breeding the resulting mice with Wlsf/f mice. Wlsf/f;Dermo1Cre/+ mutants were generated by breeding Wlsf/f mice with Dermo1Cre mice (Sosic et al., 2003) and breeding resultant mice with Wlsf/f. Wlsf/f;ShhCre/+;Axin2 LacZ mice were obtained by mating Wlsf/+;ShhCre/+ with Axin2LacZ (Joeng et al., 2011; Lustig et al., 2002). Wlsf/f;ShhCre/+;Rosa mT/mG mice were generated by breeding Wlsf/wt ShhCre+/−; with Rosa mT/mG mice (Muzumdar et al., 2007). Wlsf/f;Col2a1Cre/+ mutants were generated by breeding Wlsf/f mice with Col2a1Cre mice (Ovchinnikov et al., 2000) and breeding resultant mice with Wlsf/f mice. Sox9f/f;Col2a1Cre/+ and Sox9f/f;Dermo1Cre/+ embryos were obtained by crossing Sox9 f/f with Col2a1Cre or Dermo1Cre and mating resulting mice to Sox9 f/f mice. Genotypes of transgenic mice were determined by PCR using genomic DNA isolated from mouse tail or embryonic tissue. Primers utilized for genotyping have been provided as supplementary material (Table S1).
Histology, Immunohistochemistry and Immunofluorescence Staining: Embryonic tissue was fixed and embedded in paraffin or frozen using OCT. Sections (6um) were processed for H&E or DAB staining as described (Mucenski et al., 2003). For immunofluorescence, antigen retrieval was performed, when necessary, using 10 mM citrate buffer, pH 6, before slides were blocked for 2 hours in TBS containing 10% Donkey serum and 1% BSA. Following blocking, slides were incubated, overnight, at 4° in primary antibody diluted accordingly in blocking solution. After removal of unbound primary antibody, slides were incubated with secondary antibody at a dilution of 1:200 for one hour. Slides were then washed and coverslip using Vecta Shield mounting media with or without Dapi. Fluorescent staining was visualized and photographed using an automated fluorescence microscope (Zeiss). Source and dilution of primary antibodies utilized for this study has been provided as Supplementary data (Table S2).
Whole Mount X-galactosidase Staining
Embryonic lungs were dissected and fixed in 4% paraformaldehyde (PFA) in PBS for 30 minutes, then washed in PBS and stained two to three hours in X-gal staining solution. To stop the reaction, explants were washed in 3% dimethyl sulfoxide-PBS, rinsed in PBS, washed and stored in 70% ethanol. Explants were processed for paraffin embedding and sectioning. Slides were deparaffinized and counterstained with fast red.
In-Situ Hybridization
Procedure was performed according to a protocol developed by Advanced Cell Diagnostics (ACD) (Wang et al., 2012). In situ probes were designed by ACD. In brief, slides were baked and deparaffinized. In situ probes were added to the slides and hybridization was performed for 2 hours at 40°C followed by several rounds of amplification steps. Signal was detected by chromogenic reaction using DAB and slides were counter stained with Hematoxylin. All experiments were run simultaneously with a positive control probe recognizing transcripts for the housekeeping gene Ppib (which encodes for peptidylpropil isomerase B, an enzyme found in the endoplasmic reticulum) as well as a negative control probe that recognizes a bacterial transcript (Dapb). After mounting with permanent mounting media, slides were photographed using a wide field Nikon i90 microscope.
Lectin Staining
Samples were fixed overnight in PFA and rinsed multiple times in PBS. Afterwards, samples were incubated for 1 hour in blocking buffer comprised of 2% Goat Serum, 1% BSA, .3% Triton X and PBS. After removing blocking buffer, lectin PNA solution (in 10% normal goat serum and PBS) was added to the samples at a final concentration of 10 ug/ml. The samples were then covered in aluminum foil and incubated overnight at 4C. Finally, explants were rinsed with PBS multiple times and photographed using a Leica fluorescence dissecting microscope.
Whole Mount Immunofluorescence Staining and Confocal Microscopy. Samples were staining as previously described (Ahnfelt-Ronne et al., 2007). E11.5 and E13.5 tracheal lung explants were isolated and fixed overnight in 4% PFA and stored in methanol 100%. Samples were permeabilized using Dent’s Bleach and hydrated in a graded series of methanol. Explants were blocked using a commercial reagent (Invitrogen TSA kit blocking reagent). Primary antibody was diluted in blocking solution and samples were incubated overnight. After several washes, secondary antibody was applied for two hours at a 1:500 dilution. After washing, samples were dehydrated and stored in Methanol. Samples were cleared with Murray’s clear before imaging (Jahrling et al., 2009; Ott, 2008). Pictures were obtained using a confocal microscope (NikonA1Rsi). Image processing was performed using IMARIS software (BITPLANE Scientific Software).
Cell Proliferation and Cell Death: E11.5 pregnant mice were injected with BrdU at concentration 100ug/g of body weight. Embryos were isolated at day E12.5 or E13.5. Embryonic sections were labeled with BrdU, Nkx2.1, Sox9 and SMA antibodies to determine mitotic cells in tracheal epithelium and mesenchyme. Labeled cells and total cells were counted per each field photograph at 20X, and ratios of proliferating cells to total cells, defined as number of Sox9 and αSMA stained cells, were calculated. Average mitotic index was determined for five different samples. To determine cell death, TUNEL assay was performed on sections of E12.5, E13.5 and E14.5 embryos using a commercially available detection kit (Roche).
Embryonic Whole Tracheal Explant Culture: Embryonic tracheas were harvested at E11.5 and cultured at air-liquid interphase as described (Hyatt et al., 2004). Wnt5a, Wnt3a (200ng/ml), XAV-393 (1uM) or Bmp-2 (100ng/ml) were added to the media one hour after initiation of culture. Explants were harvested and processed for RNA isolation after 72 hrs. in culture.
Micromass Culture
Primary MEF cells were obtained from ATCC and cultured in Dulbecco’s modified Eagle’s medium (GIBCO) supplemented with 20% non-Heat inactivated fetal bovine serum (ATCC) along with 1% Penicillin/Streptomycin. Cells were plated into 12-well plates in micro-masses consisting of 125,000 cells/10ul. After three hours, cells were flooded with culture media. Wnt 3a, Wnt5a, Bmp-2 and LiCL were added to the media. Cells were harvested after 6 days of culture and process for RNA isolation.
RNA Extraction and RT-PCR. Gene expression was determined by quantitative RT-PCR. RNA was isolated from embryonic tracheal explants using a commercially available kit (RNAeasy mini kit or micro kit, Qiagen-Promega). Reverse transcription was performed according to manufacturer instructions (Verso Fisher Sci), and Taqman probes were utilized to detect differential expression using a StepOnePlus RT-PCR System.
Statistics: Quantitative data were presented as mean ± standard error. Experiments were repeated at least twice with a minimum of three biological replicates for each group. Statistically significant differences were determined by paired T-test or one-way ANOVA followed by post hoc pairwise multiple comparison procedures (Bonferroni method). Significance was set at P<0.05.
Results
Deletion of Wls from the respiratory tract epithelium caused tracheobronchomalacia
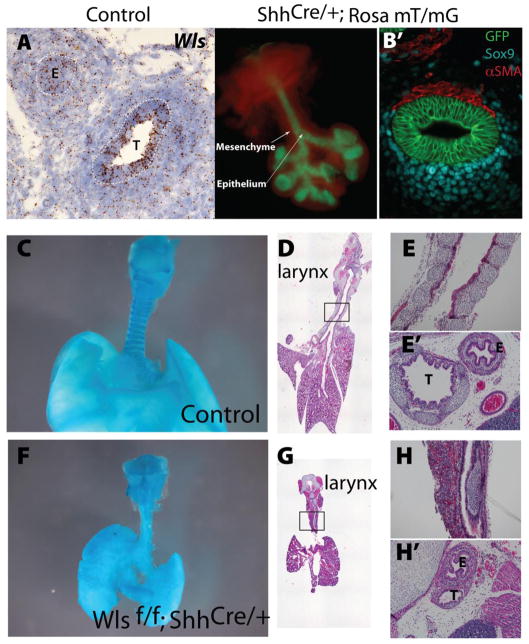
Wls is expressed in both epithelium and mesenchyme of developing trachea as determined by in situ hybridization (Fig.1 A). To define the role of Wnt signaling in upper airway differentiation Wls was deleted from the epithelial cells of the embryonic foregut endoderm by breeding Wls CKO mice (Carpenter et al, 2010) to the Shh Cre mice. Efficient and specific Cre mediated recombination in the trachea was demonstrated by detection of GFP in the epithelium of the respiratory tract of embryos at E11.5 after breeding the ShhCre mice to the Rosa mT/mG reporter mice (Fig.1 B). Deletion of Wls was confirmed by PCR using primers that recognize the mutant allele in tracheal tissue (Supp. Fig.1). Lungs of Wlsf/f;ShhCre/+ mice were hypoplastic (Fig.1 F,G compare to C,D) (Cornett et al., 2013). Tracheal and bronchial, but not laryngeal cartilage was absent as determined by Alcian blue (Fig.1 C,F) and H&E staining at E18.5 (Fig.1 D, E, G, H, E′ and H′). Cross sections of E18.5 Wlsf/f;ShhCre/+ embryos stained with H&E confirmed the lack of cartilage in the trachea and the presence of a distinct esophageal tube. Taken together, these data demonstrate that deletion of Wls from the epithelium of the developing trachea caused agenesis of the tracheal and bronchial cartilage, findings consistent with defects associated with tracheobronchomalacia.
Figure 1. Deletion of Wls from the embryonic foregut endoderm causes tracheal- bronchial cartilage agenesis.
In situ hybridization was performed on cross sections of E13.5 embryos. Representative image depicting expression of Wls in trachea and esophagus is shown. Dotted lines demark the tracheal and esophageal epithelium (A). Whole mount of tracheal lung explant depicting epithelial GFP expression driven by Cre mediated recombinase in Wlsf/f;ShhCre/+;Rosa mT/mG mice is shown. Cross section of the tracheal explant demonstrates the circumferential expression of GFP indicating sites of Cre mediated recombination (B). Alcian blue (C,F) and H & E (D,E,G,H) stainings of whole mounts and tracheal/lung sections demonstrate the near absence of cartilage in Wlsf/f;ShhCre/+ trachea and bronchi. Higher magnifications of regions in squares are shown in E and H. Cross sections of control (E′) and Wlsf/f;ShhCre/+ (H′) E18.5 embryos depict esophagus and trachea. Note the lack of cartilage in Wlsf/f;ShhCre/+ embryos and the position of trachea in close proximity to the esophagus.
Canonical Wnt signaling is active in tracheal mesenchyme and is severely impaired after epithelial deletion of Wls
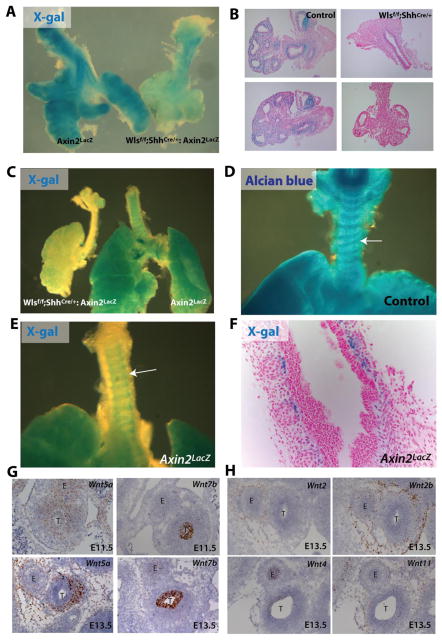
Wls is a cargo receptor mediating secretion of Wnt ligands from the producing cells (Banziger et al., 2006). However, it remains unclear whether Wls differentially promotes secretion of Wnt ligands eliciting β-catenin dependent or independent signaling. To assess whether Wnt ligands produced by the tracheal epithelium induce canonical Wnt response, Wlsf/f;ShhCre/+ mice were crossed to Axin2Lac-Z mice. Axin promoter activity was decreased after deletion of Wls from the epithelium. X-gal staining was present in the subepithelial mesenchymal cells of conducting airways and lung periphery of E12.5 control embryos. In contrast, no staining was detected in Wlsf/f;ShhCre/+ embryos (Fig.2 A,B). At E14.5, X-gal staining was detected in the mesenchyme of the trachea resembling the pattern of cartilaginous rings (Fig.2C). Alcian blue staining and Axin2 promoter activity were present in the tracheal mesenchyme at sites of chondrogenesis (Fig.2D); however, Axin2 LacZ staining was excluded from mesenchymal condensations and was confined to the sub-epithelial mesenchyme of developing trachea. Similarly to findings at E12.5, Axin2 promoter activity was not detected in tracheas of E14.5 Wlsf/f;ShhCre/+ embryos (Fig. C).
Figure 2. Decreased mesenchymal canonical Wnt signaling after deletion of epithelial Wls.
X-gal staining was performed on trachea-lung explants of E12.5 Axin2LacZ and Wlsf/f;ShhCre/+;Axin2LacZ mice. X-gal staining was primarily observed in mesenchyme of Axin2LacZ mice, while staining was nearly absent in tissue of Wlsf/f;ShhCre/+;Axin2LacZ mice (A,B). At E14.5, X-gal staining was detected in the tracheal mesenchyme of control embryos in a pattern similar to that of the cartilaginous rings. In contrast, no staining was detected in the trachea of Wlsf/f;ShhCre/+ embryos (C). E14.5 control tracheal explants were stained with Alcian blue to determine sites of chondrogenesis. The staining pattern is similar to the X-gal staining of Axin2LacZ mice (D). X-gal staining was restricted to the periphery of tracheal mesenchymal condensations (E, F). In situ hybridization was performed to determine expression pattern of several Wnt ligand mRNA. At E11.5 Wnt5a was detected in both epithelium and mesenchyme of developing trachea. At E13.5 Wnt5a RNA was enriched in the ventral mesenchyme of developing trachea, while Wnt7b was restricted to the epithelium of E11.5 and E13.5 trachea (G). Wnt2 and Wnt2b mRNA were detected in dorsal and lateral mesenchyme surrounding the trachea. Wnt4 mRNA was detected at low levels in the tracheal epithelium, while Wnt11 was present in tracheal mesenchyme of E13.5 embryos (H).
While the expression pattern of Wnt ligands in developing lung is well established, less is known about the temporal-spatial expression of Wnt ligands in the trachea. At E11.5 Wnt5a was expressed in both epithelium and mesenchyme of the trachea. Wnt7b was expressed in a circumferential pattern in tracheal epithelium of E11.5 and E13.5 embryos. In contrast, by E13.5 Wnt5a was expressed predominantly in ventral mesenchyme of the trachea, but it was also detected to lesser degree in the epithelium of the trachea (Fig.2G). At E13.5 Wnt2 and Wnt2b were present in tracheal mesenchyme. Wnt2b was enriched in the dorsal and lateral regions of the tracheal mesenchyme. Wnt2 and Wnt2b were also detected in the mesenchyme of the developing distal lung (Supp. Fig.2). Wnt4 and Wnt11 were detected at low levels in the epithelium and mesenchyme of the trachea respectively (Fig.2H). Wnt11 was also detected in ventral body wall confirming our previous studies (Supp. Fig.2) (Snowball et al., 2015). In situ probes recognizing a housekeeper gene Ppib and a bacterial gene Dapb, were run in parallel and served as a positive and negative experimental control (Suppl. Fig2). Data presented demonstrate that epithelial Wls is required for Wnt/β-catenin signaling in developing tracheal mesenchyme and that Wnt7b and Wnt5a are abundantly expressed in the E11.5 tracheal epithelium where they are likely to participate in mesenchymal tracheal patterning downstream of Wls.
Epithelial Wls regulates dorsal ventral patterning of the tracheal mesenchyme
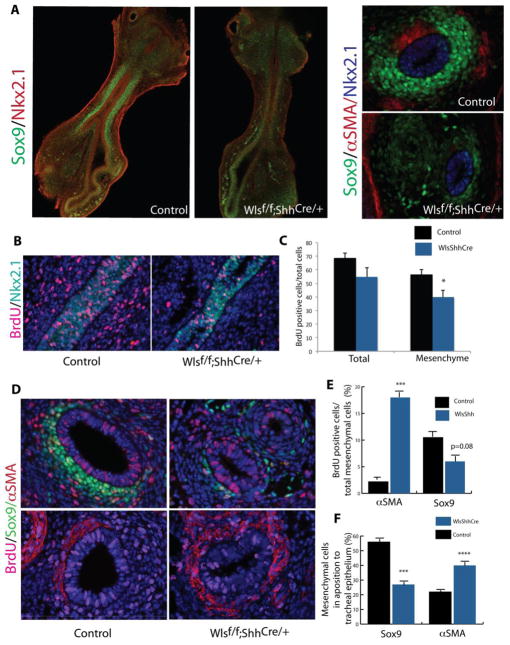
Since epithelial deletion of Wls drastically impaired cartilage at E18.5, we sought to identify the timing of cartilage and smooth muscle patterning at earliest stages. In the mouse, tracheal cartilage is well formed by E15 (Park et al., 2010; Tiozzo et al., 2009). At E13.5 in Wlsf/f;ShhCre/+ embryos smooth muscle layer, detected by αSMA staining, was expanded into the ventral aspect of the trachea and cartilage was absent (Fig.3 A and B). At E14.5, the precise patterning of the normal trachea in control mice was evidenced by a dorsal stripe of muscle. Cartilage was present as partial circumferential rings on the ventral side of the trachea. In contrast, in tracheas of Wlsf/f;ShhCre/+ mice, αSMA staining was expanded to the ventral side of the trachea (Fig.3 C, D, E, F). Epithelial cells lining the trachea stained for TTF-1 in Wlsf/f;ShhCre/+ embryos, demonstrating maintenance of respiratory epithelial cell identity. In Wlsf/f;ShhCre/+ embryos, the orientation of muscle fibers was disrupted and organized parallel to the longitudinal axis of the trachea. In contrast, in control embryos, muscle fibers of the trachealis muscle were oriented transversally to the longitudinal axis of the trachea (Fig.3 E′ versus F′). Few Sox9 stained cells were observed randomly scattered throughout the dorsal mesenchyme of the Wlsf/f;ShhCre/+ tracheas (Fig.3F′). These cells were also positive for Sox10 (data not shown). At E18.5, virtually no Sox9 positive cells were present in the tracheal mesenchyme (Supp. Fig.3). Sox9 transcripts were diminished while Acta2 transcripts were increased in tracheal tissue isolated from Wlsf/f;ShhCre/+ embryos (Fig.3 G). These findings are in agreement with the previously described expression profile of Sox9 and αSMA in Wlsf/f;ShhCre/+ tracheas. These data support the concept that Wnt ligands, dependent upon Wls secretion from endodermally derived cells, promote cartilage formation and may repress smooth muscle on the ventral side of the trachea, as well as influence the arrangement of tracheal smooth muscle.
Figure 3. Loss of epithelial Wls-mediated signaling disrupts dorsal-ventral patterning of the trachea.
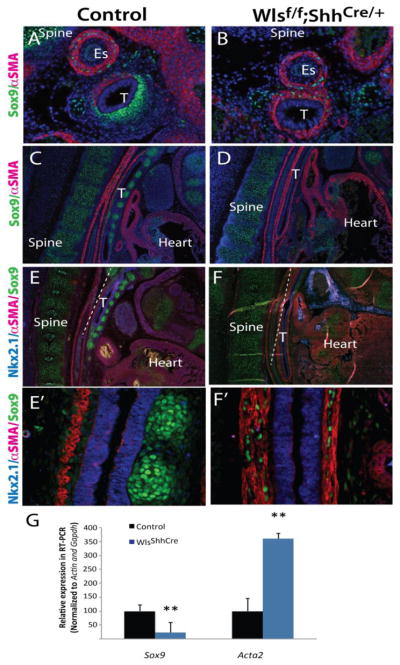
Longitudinal and cross sections of E13.5 (A,B) and E14.5 (C,D,E,F) embryos were stained with Sox9, aSMA and Nkx2.1 antibodies. DAPI was utilized to visualized cell nuclei. Sox9 was absent in the tracheal mesenchyme but expressed in vertebre cartilage of Wlsf/f;ShhCre/+ embryos. αSMA staining was expanded into the ventral region of the trachea of Wlsf/f;ShhCre/+ embryos (B,D,F). In developing trachea of Wlsf/f;ShhCre/+ embryos, muscle fibers run parallel to the longitudinal axis of the trachea in contrast to the transversal arrangements of the fibers observed in control trachea (E′, F′). Sox9 was decreased and Acta2 mRNA increased in tracheal explants of Wlsf/f;ShhCre/+ embryos at E14.5 (G). N=4, **p<0.01, T-trachea Es= esophagus.
Epithelial Wnt signaling regulates Sox9, mesenchymal cell proliferation and mesenchymal cells condensations
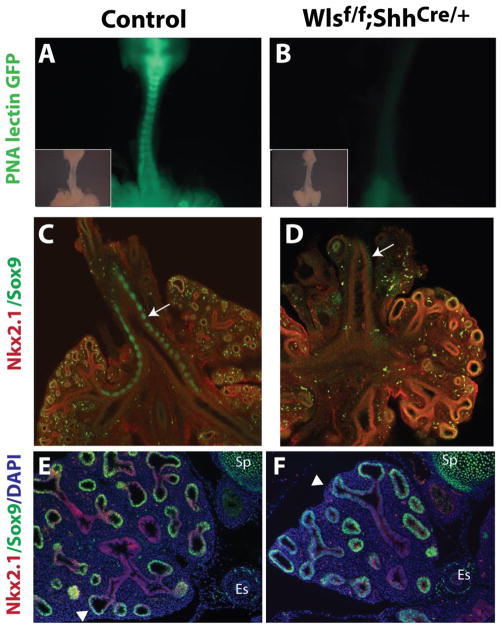
A critical event during cartilage formation is the condensation of mesenchymal cells into chondrogenic nodules (Bi et al., 1999). In developing trachea, condensations are established by E13.5. Whole mount tracheal lung explants were stained with PNA (Peanut Agglutinin Lectin) to label condensing cells (Gillotte et al., 2003). At E13.5, a clear pattern of condensed mesenchymal cells resembling cartilaginous rings was readily distinguished (Fig.4 A, arrow), while in Wlsf/f;ShhCre/+ tracheas condensations were seldom detected (Fig.4 B, arrow). These data were supported by whole mount staining of control tracheal lung explants wherein Sox9 was strongly expressed in mesenchymal condensations (Fig.4 C, arrow). In contrast, in tracheas of Wlsf/f;ShhCre/+ embryos Sox9 staining was weakly detected in subepithelial mesenchyme (Fig.4 D arrow). Sox9 was strongly expressed in the respiratory epithelium of the lung (arrow head in Fig.4 E and F). These data indicate that epithelial Wls is required for signaling to tracheal mesenchymal cells to influence Sox9 expression and to form mesenchymal condensations on the ventral side of the trachea.
Figure 4. Epithelial Wls is required for prechondrogenic mesenchymal condensations.
Tracheal lung explants of E13.5 embryos were stained with PNA lectin. Lack of staining demonstrates absence of mesenchymal condensations in tracheas of Wlsf/f;ShhCre/+ embryos (arrow B vs. arrow A). Insets in A and B correspond to bright field images of whole mounts stained with PNA lectin. Confocal images of E13.5 whole mounts show decreased Sox9 staining in tracheal mesenchyme (arrow in C, and D). Sox9 staining in cells of the peripheral lung overlapped with Nkx2.1 in both control and Wlsf/f;ShhCre/+ pulmonary tissue (arrowhead in G and H). Es= esophagus, Br=Bronchus, Sp=spine.
At E11.5, Sox9 staining was present in the tracheal mesenchyme of control and reduced in Wls deficient embryos (Fig.5 A). Since Wnt signaling modulates cell proliferation, we reasoned that the lack of Sox9 positive cells could be related to decreased mesenchymal cell proliferation or increased cell death. At E12.5, cell proliferation was decreased in the mesenchyme of the Wlsf/f;ShhCre/+ tracheas (Fig.5 B and C). To test whether loss of epithelial Wls mediated signaling differentially affected proliferation of chondroblasts or myoblasts, we quantified cells expressing Sox9 or αSMA in cross sections of tracheas from E13.5 embryos. BrdU labeling demonstrated increased numbers of αSMA but decrease numbers of Sox9 proliferating cells. Likewise, numbers of Sox9 stained cells were decreased and those staining for αSMA stained cells were increased (Fig.5 D and E). Moreover, the number of Sox9 cells in close apposition with the tracheal epithelium was decreased and αSMA stained mesenchymal cells in close proximity to the tracheal epithelium were increased in Wlsf/f;ShhCre/+ embryos, demonstrating the malpositioning of muscle cells on the ventral side of the trachea (Fig.5 F). TUNEL studies did not detect differences in cell death between controls and Wlsf/f;ShhCre/+ tracheal tissue (data not shown). Thus, these data demonstrate that epithelial Wls controls mesenchymal cell proliferation, modulating proliferation and numbers of chondrogenic and myogenic cells in the embryonic tracheal mesenchyme.
Figure 5. Epithelial Wnt ligands modulate mesenchymal cell proliferation.
Confocal images of E11.5 tracheal lung explants stained for Sox9 and Nkx2.1 are shown. Sox9 staining was nearly absent in the tracheal mesenchyme of Wlsf/f;ShhCre/+ mice. Transverse sections of tracheal tissue depict the extent of Sox9 expression and the lack of aSMA staining in Wlsf/f;ShhCre/+ at E11.5. Dams were injected at E11.5 with BrdU to determine cell proliferation. Longitudinal sections of E12.5 tracheas were stained for BrdU and Nkx2.1; proliferation was decreased in mesenchyme of Wlsf/f;ShhCre/+ embryos (B,C). Smooth muscle cell proliferation was increased at E13.5, while proliferation of Sox9 stained cells was reduced in the tracheal mesenchyme of Wlsf/f;ShhCre/+ embryos (D, E). Increased numbers of αSMA stained cells were found in direct apposition to the tracheal epithelium. Numbers of Sox9 stained cells in direct contact with the tracheal epithelium were significantly reduced (F).
Mesenchymal deletion of Sox9 is not sufficient to promote expansion of tracheal smooth muscle
To assess whether Wls effects on tracheal mesenchyme patterning depend on Sox9, we deleted Sox9 from the developing tracheal mesenchyme using Dermo1Cre and Col2a1Cre mice. Deletion of Sox9 from the mesenchyme of the trachea using Dermo1Cre mice, reduced the number and altered the shape of cartilaginous rings, resulting in a shorter trachea, findings resembling the Wnt5a loss of function (Li et al., 2002) (Supp. Fig. 4 H). Col2a1 is a direct target of Sox9 and it is expressed in developing trachea at E10.5 in a similar distribution to that of Sox9 (Elluru et al., 2009; Elluru and Whitsett, 2004). We utilized Col2a1Cre mice to delete Sox9 in chondrocyte progenitors. While cartilage formation was severely impaired, deletion of Sox9 did not cause robust expansion of the muscle layer into the ventral side of the trachea (Fig. 6 B,D, F, H and Supp. Fig.4 F), findings contrasting the phenotype observed in Wlsf/f;ShhCre/+ embryos (Fig.3B). We also determined that Acta2 levels were not altered in tracheas of E14.5 Sox9f/f;Col2a1Cre/+ embryos (Fig.6 I). These data demonstrate that while Sox9 is required for tracheal cartilage development, deletion of Sox9 did not cause expansion of the muscle layer into the ventral side of the trachea as seen in the Wlsf/f;ShhCre/+ embryos.
Figure 6. Mesenchymal deletion of Sox9 from developing trachea does not promote expansion of tracheal muscle layer.
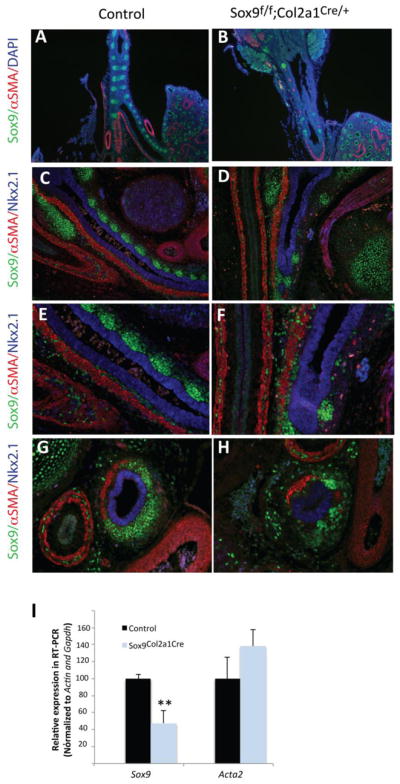
Sox9 was deleted using Col2a1Cre mice, resulting in tracheal cartilage agenesis (B,D,F). No expansion of muscle into the ventral mesenchyme of the trachea was observed (D,F). RT-PCR analysis demonstrates that Sox9 mRNA is decreased while Acta2 mRNA was not significantly changed in E14.5 Sox9f/f;Col2a1Cre/+ trachea (G). N=4 **p<0.01.
To test whether mesenchymal Wls influenced tracheal patterning, Wls was conditionally deleted using Dermo1Cre mice. Dermo1Cre efficiently targets the mesenchyme surrounding the E10.5 foregut as well as mesenchyme of the E12.5 developing trachea, as verified by GFP staining in Dermo1-Cre;Rosa mT/mG mice (Supp. Fig. 5A). Neither Sox9 mRNA nor staining were altered by mesenchymal deletion of Wls (Supp Fig.5). Expression levels and pattern of αSMA staining were unaltered. Deletion of Wls from chondrocyte precursors, using Col2a1Cre mice did not affect cartilage formation or tracheal patterning (Supp. Fig5). These data suggest that secretion of mesenchymal Wnt ligands may play a redundant role in regards to tracheal patterning, supporting the concept that the epithelial Wls mediated signaling directs mesenchymal patterning of the trachea.
Canonical Wnt signaling modulates Sox9 in prechondrogenic tracheal explants
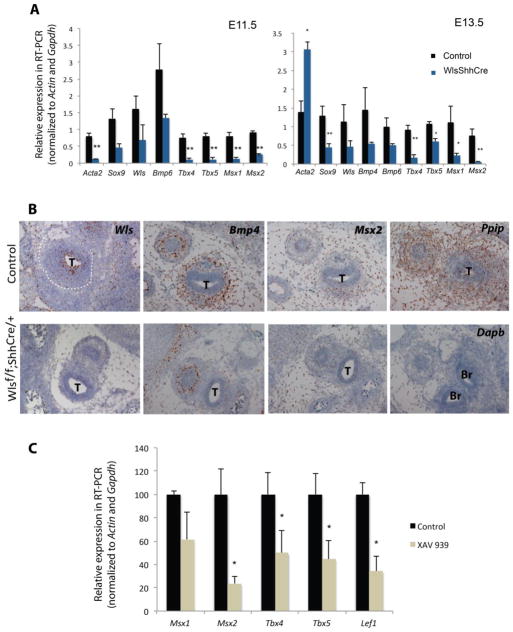
It is established that canonical Wnt signaling modulates Sox9 activity during endochondral bone formation (Akiyama et al., 2004; Day and Yang, 2008; Hill et al., 2005). To test the hypothesis that epithelial Wnt signaling regulates Sox9 expression to induce tracheal chondrogenesis, we utilized ex vivo tracheal explants cultures. We tested whether Wlsf/f;ShhCre/+ tracheal explants isolated at E11.5 and incubated for 72hrs. were responsive to Bmp-2, a chondrogenic signal (Lengner et al., 2004; Pan et al., 2008). Bmp-2 induced Sox9 and Col2a1 expression. Neither addition of Wnt3a (a Wnt ligand that similarly to Wnt7b typically induces Wnt/β-catenin signaling in respiratory tract) nor Wnt5a alone induced Sox9 in tracheal explants (Fig.7A); however, addition of both ligands to the culture media robustly induced Sox9 and to lesser degree Col2a1 (Fig.7B). Acta2 mRNA was not significantly changed by addition of Wnt3a or Wnt5a (Fig.7A and B). When E11.5 control tracheal explants were incubated in the presence of Wnt canonical signaling inhibitor XAV 939 (Huang et al., 2009), Sox9 expression was modestly decreased and Col2a1 was sharply decreased (Fig.7C). To test if Wnt signaling modulates Sox9 expression after formation of mesenchymal condensations, MEF (mouse embryonic fibroblasts) cells were seeded in micromasses and treated with Bmp-2 to induced Sox9 (Lengner et al., 2004). In this system, incubation in presence of LiCl, an activator of Wnt/β-catenin signaling (Klein and Melton, 1996), repressed Sox9 and Col2a1 and did not alter Acta2 (Fig. 7D). Similar results were obtained when tracheal explants isolated at E13.5 (stage when mesenchymal condensations are formed), were treated with LiCl resulting in decreased Sox9 and Col2a1 mRNAs (data not shown). These data suggest that Wnt/β-catenin signaling plays differential roles during tracheal chondrogenesis, and that Wnt signaling is necessary to promote Sox9 and Col2a1 in prechondrogenic tracheal mesenchyme ex vivo.
Figure 7. Canonical Wnt signaling modulates expression of Sox9 ex vivo and in vitro.
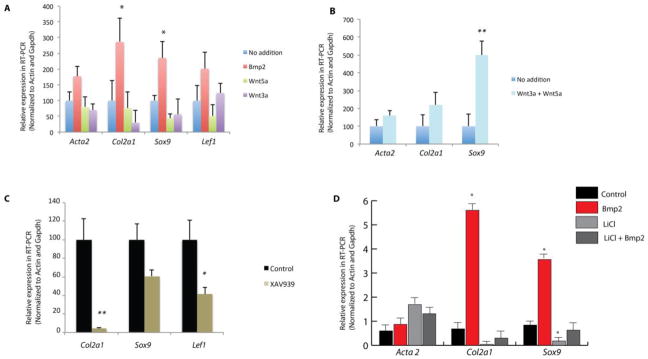
Bmp-2 (100ng/ml) induced Sox9 expression in Wlsf/f;ShhCre/+ tracheal explants, while Wnt3a or Wnt5a (200 ng/ml) did not induce Sox9 in tracheal explants. (A) Addition of Wnt3a and Wnt5a to culture media of Wlsf/f;ShhCre/+ explants increased Sox9 and Col2a1 mRNA (B). Control tracheal explants were cultured in the presence of the canonical Wnt antagonist XAV-939. Addition of XAV-939 (1uM) reduced expression of Col2a1, Lef1 (a direct canonical Wnt target) and to lesser degree Sox9 (C). In MEF micromass cultures, addition of Bmp-2 robustly induced Sox9 and Col2a1. Addition of Bmp-2 and LiCl repressed Bmp-2 induced Sox9 in MEF micromasses (D). N=5 * p<0.05 ** p<0.02 vs. C.
Epithelial Wls mediated signaling is necessary for expression of factors required for tracheal patterning of cartilage and muscle
Since addition of Bmp-2 induced Sox9 and Col2a1 in explants and in micromass cultures, we asked whether BMP signaling was altered in tracheas of Wlsf/f;ShhCre/+ embryos. Bmp4 and Bmp6 mRNA levels were modestly decreased in the mesenchyme of developing tracheas of E11.5 and E13.5 Wlsf/f;ShhCre/+ as determined by quantitative RT-PCR and in situ hybridization (Fig.8A and B). Furthermore, treatment of tracheal explants from Wlsf/f;ShhCre/+ embryos with Bmp-2, strongly induced Sox9 expression (Fig.7B). Msx1, Msx2, Tbx4 and Tbx5, transcription factors known to influence cartilage and smooth muscle development, were reduced in Wlsf/f;ShhCre/+ tracheal tissue (Fig.8A and B). At E13.5 Wls mRNA was decreased in the tracheal mesenchyme of Wlsf/f;ShhCre/+ as determined by in situ hybridization, suggesting that Wls may influence its own expression (Fig.8B). To test whether Wnt/β-catenin signaling modulated the expression of genes regulating chondrogenesis ex vivo, control tracheal explants isolated at E11.5 were incubated with XAV 939 for 72 hrs. resulting in decreased expression of Msx2, Tbx4 and Tbx5 (Fig.8 C). Taken together, these data support the concept that epithelial Wls influences expression of genes required for cartilage differentiation at least in part, via canonical Wnt signaling.
Figure 8. Canonical Wnt signaling influences expression of genes controlling cartilage differentiation.
Quantitative RT-PCR performed on E11.5 and E13.5 tracheas of control and Wlsf/f;ShhCre/+ embryos demonstrated decreased expression of genes required for cartilage patterning, as well as increased levels of Acta2 at E13.5, N= 3 *p<0.05, ** p<0.01 (A). Analysis of Wls, Bmp4 and Msx2 mRNA by in situ hybridization is shown. Bmp4 and Msx2 localized to the ventral region of the trachea where cartilage will be formed. Epithelial deletion of Wls decreased Bmp4 and Msx2 mRNA in ventral tracheal mesenchyme. Wls RNA was decreased in tracheal epithelial cells of Wlsf/f;ShhCre/+ mice. Ppib transcripts, positive control, were detected throughout the section while Dapb transcripts, negative control, were not detected (B). Incubation of tracheal explants with XAV-939 decreased Msx2, Tbx4 and Tbx5 and Lef1 mRNAs. Representative experiments of explant cultures are shown. N=3 *p<0.05 (C). T= Trachea, Br= Bronchus.
Discussion
In the present study, we demonstrated that endodermal Wls mediated signaling is required for tracheal cartilage development. Epithelial Wls influenced the expression of pro-chondrogenic genes in part by activation of Wnt/β-catenin signaling in tracheal mesenchyme. Epithelial Wls mediated signaling limited the proliferation of muscle layer into the ventral and lateral regions of the trachea. Therefore, precise regulation of Wnt ligand secretion from the tracheal bronchial epithelium is required for normal patterning of tracheal- bronchial cartilage.
Initiation of the chondrogenic program in developing trachea requires Wnt signaling. While commitment of tracheal mesenchymal cells towards the chondrogenic lineage is defined by the expression of Sox9, the events upstream of Sox9 expression are less well understood. Present data demonstrated that epithelial Wls is required for Sox9 expression and formation of tracheal cartilage (Fig. 1 and 3). We determined that Wnt/β-catenin signaling was operative in the sub-epithelial mesenchyme of the trachea of control embryos, and that epithelial deletion of Wls severely impaired Wnt/β-catenin activity in the tracheal mesenchyme of Wlsf/f;ShhCre/+ embryos (Fig.2).
Our data support the concept that epithelial Wls influences gene expression required for chondrogenesis. Expression of Bmp4 and Bmp6 was reduced, to some extent, in Wlsf/f;ShhCre/+ embryos. It has been shown that BMP signaling plays a critical role in cartilage formation during limb development (Barna and Niswander, 2007; Duprez et al., 1996; Karamboulas et al., 2010). At early stages of respiratory tract development, Bmp4 is expressed in the anterior foregut and it is required for outgrowth of the trachea (Li et al., 2008). Moreover, Bmp4 is expressed in the ventral region of the prechondrogenic tracheal mesenchyme (this study and (Park et al., 2010), where cartilage will be formed. Our studies showed that Bmp-2 (a ligand with 80% homology to Bmp-4) induced Sox9 expression in tracheal explants of control and Wlsf/f;ShhCre/+ embryos (Fig.7 A). Present data also suggest that in tracheal development, both Bmp and Wnt signaling influence the differentiation of tracheal mesenchyme into cartilage (Fig. 7 and Fig. 8). These data are supported by recent studies demonstrating that the interplay between Bmp and Wnt signaling determines the sites of expression of Sox9 during patterning of digits (Raspopovic et al., 2014). Our data also demonstrated that epithelial Wls is necessary for Tbx4 and Tbx5 expression. Tbx4 and Tbx5, are known to promote tracheal cartilage development by inducing Sox9 expression as well as promoting mesenchymal condensations (Arora et al., 2012). It is unclear whether Tbx4 and Tbx5 are direct targets of Wnt signaling. Our in vitro studies showed that pharmacological inhibition of Wnt/β-catenin signaling in prechondrogenic control tracheal explants decreased the mRNA levels of Sox9, Col2a1, Tbx4 and Tbx5 thus confirming the requirement of canonical Wnt signaling in the tracheal mesenchyme for establishment of the chondrogenic program (Fig.8). These data are in agreement with the concept that Wnt signaling plays a dynamic role in cartilage development and both anomalously increased or diminished levels of Wnt signaling will disrupt chondrogenesis (Ling et al., 2009; Liu et al., 2008). Previous in vivo and in vitro studies suggest that β-catenin activity fluctuates during chondrogenesis, being higher in prechondrogenic mesenchyme, and at lower levels during chondrocyte maturation (Gaur et al., 2006; Ling et al., 2009).
Wnt ligands act synergistically to induce tracheal cartilage
Wls is required for secretion and activity of all lipid modified Wnt ligands (Najdi et al., 2012). Our studies shown that Wnt7b, Wnt5a and to lesser degree Wnt4 transcripts were found in a circumferential expression pattern in the tracheal epithelium at E11.5 (Fig.2). Deletion of Wnt7b was associated with abnormal cartilaginous rings (Rajagopal et al., 2008). Mice carrying a null allele of Wnt5a developed a short trachea with anomalous, discontinuous cartilaginous rings (Li et al., 2002), resembling the tracheal phenotype observed in Sox9f/f;Dermo1Cre/+ mice. It is presently unknown whether Wnt4 participates in tracheal patterning, however mutations in WNT4 are associated with lung hypoplasia (Mandel et al., 2008). Thus, the lack of cartilage observed in tracheas of Wlsf/f;ShhCre/+ embryos may result from the combined loss of Wnt7b and Wnt5a signaling, a model supported by our in vitro data, wherein addition of both Wnt3a and Wnt5a induced expression of Sox9 and Col2a1 in Wlsf/f;ShhCre/+ tracheal explants. Thus Wnt/β-catenin activity induced by epithelial Wnt ligands is required, at least in part, for commitment of mesenchymal cells towards the chondrogenic lineage (Fig. 7 and Fig. 8).
Wnt signaling is required for differentiation and patterning of the trachealis muscle It is unknown how the muscle lineage is initially specified in the tracheal mesenchyme. In peripheral lung, Wnt7b and Wnt2 play essential roles in establishment of vascular and airway smooth muscle (Cohen et al., 2009; Goss et al., 2011). In our model, deletion of Wls from the epithelium of the trachea resulted in expansion of muscle layer into the ventral aspect of the trachea findings not seen in other experimental models of tracheomalacia (Hines et al., 2013; Turcatel et al., 2013). Our studies showed that deletion of Sox9 from tracheal mesenchyme using the Col2a1Cre mice caused severe loss of cartilage; however, expansion of αSMA staining into the ventral side of the trachea was not observed (Fig.6). Epithelial deletion of Wls caused a sharp change in proliferation profile, resulting in increased proliferation of αSMA stained cells (myoblasts) accompanied by decreased proliferation of Sox9 stained cells (chondroblasts). This proliferation profile contrasts with that observed in control developing tracheas, wherein Sox9 stained cells (chondroblasts) proliferation is higher when compared to αSMA stained cells (myoblasts) proliferation (Fig.5). Thus, epithelial Wls influences mesenchymal cell proliferation to balance myoblast and chondroblast growth in developing trachea.
Wnt signaling may pattern tracheal muscle by inducing genes that antagonize muscle development in the ventral mesenchyme of the trachea. The transcription factors Msx1 and Msx2, targets of BMP signaling and known to play many roles in embryonic development, were expressed in the ventral aspect of the trachea (Alappat et al., 2003; Ishii et al., 2005). Msx1 and Msx2 were decreased in Wlsf/f;ShhCre/+ tracheal tissue. Msx2 mRNA was decreased in control prechondrogenic tracheal explants incubated with the canonical Wnt inhibitor XAV 939 (Fig.8). Studies on vascular development and disease have shown that Msx1 and Msx2 actively repressed smooth muscle differentiation (Cheng et al., 2014; Hayashi et al., 2006; Lallemand et al., 2005). Thus, these data suggest that Msx1 and Msx2 may antagonize the formation of smooth muscle in the ventral mesenchyme of the trachea.
Besides effects in tracheal muscle patterning, deletion of epithelial Wls changed the orientation of the muscle fibers of the trachealis muscle, being oriented parallel to the longitudinal tracheal axis, rather than the transverse orientation observed in trachealis muscle of control embryos (Fig.3). This abnormal arrangement of the muscle layer resembles the phenotype previously described by deletion of the transmembrane protein Tmem 16; however, in the latter model of tracheomalacia, expansion of muscle into the ventral aspect of the trachea was not described (Rock et al., 2008). Thus, Wnt signaling from the epithelium plays a dual role in trachealis muscle development: preventing muscle differentiation in the ventral tracheal mesenchyme and being required for normal morphology of the trachealis muscle fibers.
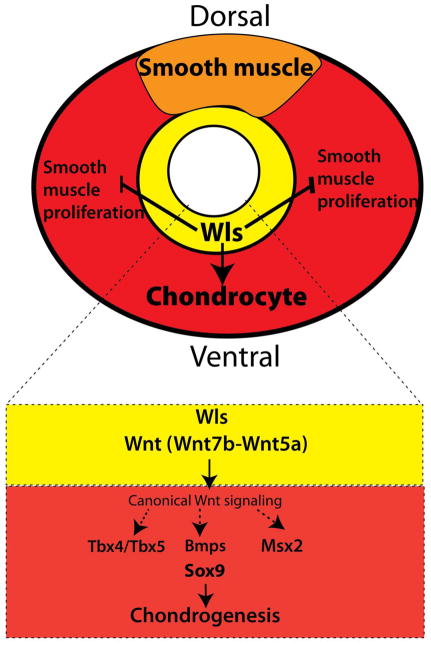
In conclusion, we propose a model wherein epithelial Wnt signaling from endodermal progenitors of the respiratory tract mediates dorsal ventral patterning of the trachea. Epithelial Wnt ligands including Wnt7b and Wnt5a activate Wnt/β-catenin in the mesenchyme of the developing trachea to influence expression of chondrogenic factors including Tbx4, Tbx5 Msx1 Msx2 Sox9 and Col2a1, genes required for initiation of chondrogenesis in the ventral region of the trachea. Epithelial Wls also differentially promotes cell proliferation of mesenchymal cells and prevents the anomalous proliferation of muscle cells into the ventral region of the trachea. By influencing differentiation of pre-chondrogenic mesenchyme and cell proliferation, epithelial Wnt ligands direct dorsal ventral patterning of developing trachea. Taken together, abnormalities in secretion of Wnt ligands produced by the epithelial cells of the upper airways, provides a mechanism underlying congenital tracheobronchomalacia.
Supplementary Material
Figure 9. Model for dorsal ventral patterning of developing trachea.
Epithelial Wls promotes cartilage development and prevents the expansion of muscle cells into the ventral side of the trachea. Wnt ligands produced by the epithelium promote canonical Wnt signaling in the tracheal mesenchyme, influencing the expression of transcription factors required for differentiation of mesenchymal cells into cartilage. Epithelial Wls regulates mesenchymal proliferation and modulates proliferation of smooth muscle cells. Thus, by promoting cartilage differentiation and modulating smooth muscle cell proliferation, epithelial Wnt signaling directs the patterning of the tracheal mesenchyme.
Highlights.
Wls is required for dorsal-ventral patterning of the developing trachea.
Wnt ligands from tracheal epithelium promote proliferation of tracheal mesenchyme.
Wnt/β catenin signaling is necessary for tracheal chondrogenesis.
Epithelial Wnt ligands are necessary for trachealis muscle formation and patterning
Acknowledgments
We would like to thank Dr. Aaron Zorn for useful comments on this project, and Jacqueline Akech (ACD) for advice on in situ hybridization protocol. We also acknowledge the assistance of Mr. Chuck Crimmel with graphic design and Mike Muntifering and Matt Kofron with confocal imaging. This work was partially supported by National Institutes of Health (K01HL115447 to D.S., U01 HL122642 and R01 HL095580 to J.A.W.)
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Ahnfelt-Ronne J, Jorgensen MC, Hald J, Madsen OD, Serup P, Hecksher-Sorensen J. An improved method for three-dimensional reconstruction of protein expression patterns in intact mouse and chicken embryos and organs. J Histochem Cytochem. 2007;55:925–930. doi: 10.1369/jhc.7A7226.2007. [DOI] [PubMed] [Google Scholar]
- Akiyama H. Control of chondrogenesis by the transcription factor Sox9. Mod Rheumatol. 2008;18:213–219. doi: 10.1007/s10165-008-0048-x. [DOI] [PubMed] [Google Scholar]
- Akiyama H, Chaboissier MC, Martin JF, Schedl A, de Crombrugghe B. The transcription factor Sox9 has essential roles in successive steps of the chondrocyte differentiation pathway and is required for expression of Sox5 and Sox6. Genes Dev. 2002;16:2813–2828. doi: 10.1101/gad.1017802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Akiyama H, Lyons JP, Mori-Akiyama Y, Yang X, Zhang R, Zhang Z, Deng JM, Taketo MM, Nakamura T, Behringer RR, McCrea PD, de Crombrugghe B. Interactions between Sox9 and beta-catenin control chondrocyte differentiation. Genes Dev. 2004;18:1072–1087. doi: 10.1101/gad.1171104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alappat S, Zhang ZY, Chen YP. Msx homeobox gene family and craniofacial development. Cell Res. 2003;13:429–442. doi: 10.1038/sj.cr.7290185. [DOI] [PubMed] [Google Scholar]
- Arora R, Metzger RJ, Papaioannou VE. Multiple roles and interactions of Tbx4 and Tbx5 in development of the respiratory system. PLoS Genet. 2012;8:e1002866. doi: 10.1371/journal.pgen.1002866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Banziger C, Soldini D, Schutt C, Zipperlen P, Hausmann G, Basler K. Wntless, a conserved membrane protein dedicated to the secretion of Wnt proteins from signaling cells. Cell. 2006;125:509–522. doi: 10.1016/j.cell.2006.02.049. [DOI] [PubMed] [Google Scholar]
- Barna M, Niswander L. Visualization of cartilage formation: insight into cellular properties of skeletal progenitors and chondrodysplasia syndromes. Dev Cell. 2007;12:931–941. doi: 10.1016/j.devcel.2007.04.016. [DOI] [PubMed] [Google Scholar]
- Bell SM, Schreiner CM, Wert SE, Mucenski ML, Scott WJ, Whitsett JA. R-spondin 2 is required for normal laryngeal-tracheal, lung and limb morphogenesis. Development. 2008;135:1049–1058. doi: 10.1242/dev.013359. [DOI] [PubMed] [Google Scholar]
- Bi W, Deng JM, Zhang Z, Behringer RR, de Crombrugghe B. Sox9 is required for cartilage formation. Nat Genet. 1999;22:85–89. doi: 10.1038/8792. [DOI] [PubMed] [Google Scholar]
- Boogaard R, Huijsmans SH, Pijnenburg MW, Tiddens HA, de Jongste JC, Merkus PJ. Tracheomalacia and bronchomalacia in children: incidence and patient characteristics. Chest. 2005;128:3391–3397. doi: 10.1378/chest.128.5.3391. [DOI] [PubMed] [Google Scholar]
- Boucherat O, Nadeau V, Berube-Simard FA, Charron J, Jeannotte L. Crucial requirement of ERK/MAPK signaling in respiratory tract development. Development. 2014;141:3197–3211. doi: 10.1242/dev.110254. [DOI] [PubMed] [Google Scholar]
- Carden KA, Boiselle PM, Waltz DA, Ernst A. Tracheomalacia and tracheobronchomalacia in children and adults: an in-depth review. Chest. 2005;127:984–1005. doi: 10.1378/chest.127.3.984. [DOI] [PubMed] [Google Scholar]
- Carpenter AC, Rao S, Wells JM, Campbell K, Lang RA. Generation of mice with a conditional null allele for Wntless. Genesis. 2010:1–5. doi: 10.1002/dvg.20651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng SL, Behrmann A, Shao JS, Ramachandran B, Krchma K, Arredondo YB, Kovacs A, Mead M, Maxson R, Towler DA. Targeted Reduction of Vascular Msx1 And Msx2 Mitigates Arteriosclerotic Calcification And Aortic Stiffness In LDLR-Deficient Mice Fed Diabetogenic Diets. Diabetes. 2014 doi: 10.2337/db14-0326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cohen ED, Ihida-Stansbury K, Lu MM, Panettieri RA, Jones PL, Morrisey EE. Wnt signaling regulates smooth muscle precursor development in the mouse lung via a tenascin C/PDGFR pathway. J Clin Invest. 2009;119:2538–2549. doi: 10.1172/JCI38079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cornett B, Snowball J, Varisco BM, Lang R, Whitsett J, Sinner D. Wntless is required for peripheral lung differentiation and pulmonary vascular development. Dev Biol. 2013;379:38–52. doi: 10.1016/j.ydbio.2013.03.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Day TF, Yang Y. Wnt and hedgehog signaling pathways in bone development. J Bone Joint Surg Am. 2008;90(Suppl 1):19–24. doi: 10.2106/JBJS.G.01174. [DOI] [PubMed] [Google Scholar]
- De Langhe SP, Carraro G, Tefft D, Li C, Xu X, Chai Y, Minoo P, Hajihosseini MK, Drouin J, Kaartinen V, Bellusci S. Formation and differentiation of multiple mesenchymal lineages during lung development is regulated by beta-catenin signaling. PLoS One. 2008;3:e1516. doi: 10.1371/journal.pone.0001516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duprez DM, Coltey M, Amthor H, Brickell PM, Tickle C. Bone morphogenetic protein-2 (BMP-2) inhibits muscle development and promotes cartilage formation in chick limb bud cultures. Dev Biol. 1996;174:448–452. doi: 10.1006/dbio.1996.0087. [DOI] [PubMed] [Google Scholar]
- Elluru RG, Thompson F, Reece A. Fibroblast growth factor 18 gives growth and directional cues to airway cartilage. Laryngoscope. 2009;119:1153–1165. doi: 10.1002/lary.20157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elluru RG, Whitsett JA. Potential role of Sox9 in patterning tracheal cartilage ring formation in an embryonic mouse model. Arch Otolaryngol Head Neck Surg. 2004;130:732–736. doi: 10.1001/archotol.130.6.732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fausett SR, Klingensmith J. Compartmentalization of the foregut tube: developmental origins of the trachea and esophagus. Wiley Interdiscip Rev Dev Biol. 2012;1:184–202. doi: 10.1002/wdev.12. [DOI] [PubMed] [Google Scholar]
- Gaur T, Rich L, Lengner CJ, Hussain S, Trevant B, Ayers D, Stein JL, Bodine PV, Komm BS, Stein GS, Lian JB. Secreted frizzled related protein 1 regulates Wnt signaling for BMP2 induced chondrocyte differentiation. J Cell Physiol. 2006;208:87–96. doi: 10.1002/jcp.20637. [DOI] [PubMed] [Google Scholar]
- Gillotte DM, Fox PL, Mjaatvedt CH, Hoffman S, Capehart AA. An in vitro method for analysis of chondrogenesis in limb mesenchyme from individual transgenic (hdf) embryos. Methods Cell Sci. 2003;25:97–104. doi: 10.1007/s11022-004-9803-3. [DOI] [PubMed] [Google Scholar]
- Goodman RM, Thombre S, Firtina Z, Gray D, Betts D, Roebuck J, Spana EP, Selva EM. Sprinter: a novel transmembrane protein required for Wg secretion and signaling. Development. 2006;133:4901–4911. doi: 10.1242/dev.02674. [DOI] [PubMed] [Google Scholar]
- Goss AM, Tian Y, Cheng L, Yang J, Zhou D, Cohen ED, Morrisey EE. Wnt2 signaling is necessary and sufficient to activate the airway smooth muscle program in the lung by regulating myocardin/Mrtf-B and Fgf10 expression. Dev Biol. 2011;356:541–552. doi: 10.1016/j.ydbio.2011.06.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goss AM, Tian Y, Tsukiyama T, Cohen ED, Zhou D, Lu MM, Yamaguchi TP, Morrisey EE. Wnt2/2b and beta-catenin signaling are necessary and sufficient to specify lung progenitors in the foregut. Dev Cell. 2009;17:290–298. doi: 10.1016/j.devcel.2009.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harris-Johnson KS, Domyan ET, Vezina CM, Sun X. beta-Catenin promotes respiratory progenitor identity in mouse foregut. Proc Natl Acad Sci U S A. 2009;106:16287–16292. doi: 10.1073/pnas.0902274106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hayashi K, Nakamura S, Nishida W, Sobue K. Bone morphogenetic protein-induced MSX1 and MSX2 inhibit myocardin-dependent smooth muscle gene transcription. Mol Cell Biol. 2006;26:9456–9470. doi: 10.1128/MCB.00759-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hill TP, Spater D, Taketo MM, Birchmeier W, Hartmann C. Canonical Wnt/beta-catenin signaling prevents osteoblasts from differentiating into chondrocytes. Dev Cell. 2005;8:727–738. doi: 10.1016/j.devcel.2005.02.013. [DOI] [PubMed] [Google Scholar]
- Hines EA, Jones MK, Verheyden JM, Harvey JF, Sun X. Establishment of smooth muscle and cartilage juxtaposition in the developing mouse upper airways. Proc Natl Acad Sci U S A. 2013;110:19444–19449. doi: 10.1073/pnas.1313223110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang SM, Mishina YM, Liu S, Cheung A, Stegmeier F, Michaud GA, Charlat O, Wiellette E, Zhang Y, Wiessner S, Hild M, Shi X, Wilson CJ, Mickanin C, Myer V, Fazal A, Tomlinson R, Serluca F, Shao W, Cheng H, Shultz M, Rau C, Schirle M, Schlegl J, Ghidelli S, Fawell S, Lu C, Curtis D, Kirschner MW, Lengauer C, Finan PM, Tallarico JA, Bouwmeester T, Porter JA, Bauer A, Cong F. Tankyrase inhibition stabilizes axin and antagonizes Wnt signalling. Nature. 2009;461:614–620. doi: 10.1038/nature08356. [DOI] [PubMed] [Google Scholar]
- Hyatt BA, Shangguan X, Shannon JM. FGF-10 induces SP-C and Bmp4 and regulates proximal-distal patterning in embryonic tracheal epithelium. Am J Physiol Lung Cell Mol Physiol. 2004;287:L1116–1126. doi: 10.1152/ajplung.00033.2004. [DOI] [PubMed] [Google Scholar]
- Ishii M, Han J, Yen HY, Sucov HM, Chai Y, Maxson RE., Jr Combined deficiencies of Msx1 and Msx2 cause impaired patterning and survival of the cranial neural crest. Development. 2005;132:4937–4950. doi: 10.1242/dev.02072. [DOI] [PubMed] [Google Scholar]
- Jahrling N, Becker K, Dodt HU. 3D-reconstruction of blood vessels by ultramicroscopy. Organogenesis. 2009;5:145–148. doi: 10.4161/org.5.4.10403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joeng KS, Schumacher CA, Zylstra-Diegel CR, Long F, Williams BO. Lrp5 and Lrp6 redundantly control skeletal development in the mouse embryo. Dev Biol. 2011;359:222–229. doi: 10.1016/j.ydbio.2011.08.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karamboulas K, Dranse HJ, Underhill TM. Regulation of BMP-dependent chondrogenesis in early limb mesenchyme by TGFbeta signals. J Cell Sci. 2010;123:2068–2076. doi: 10.1242/jcs.062901. [DOI] [PubMed] [Google Scholar]
- Kenny APTM, Hall E, Woodrooffe K, Lai W, Meinzen-Den J, Hopkin RJ, Greenberg JM. Cincinnati Regional Incidence, Morbidity, and Mortality of Neonatal Foregut Defects and High Coincidence with Cardiovascular Malformations. J Neonatal Biol. 2013;2:7. [Google Scholar]
- Klein PS, Melton DA. A molecular mechanism for the effect of lithium on development. Proc Natl Acad Sci U S A. 1996;93:8455–8459. doi: 10.1073/pnas.93.16.8455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lallemand Y, Nicola MA, Ramos C, Bach A, Cloment CS, Robert B. Analysis of Msx1; Msx2 double mutants reveals multiple roles for Msx genes in limb development. Development. 2005;132:3003–3014. doi: 10.1242/dev.01877. [DOI] [PubMed] [Google Scholar]
- Lengner CJ, Lepper C, van Wijnen AJ, Stein JL, Stein GS, Lian JB. Primary mouse embryonic fibroblasts: a model of mesenchymal cartilage formation. J Cell Physiol. 2004;200:327–333. doi: 10.1002/jcp.20118. [DOI] [PubMed] [Google Scholar]
- Li C, Xiao J, Hormi K, Borok Z, Minoo P. Wnt5a participates in distal lung morphogenesis. Dev Biol. 2002;248:68–81. doi: 10.1006/dbio.2002.0729. [DOI] [PubMed] [Google Scholar]
- Li Y, Gordon J, Manley NR, Litingtung Y, Chiang C. Bmp4 is required for tracheal formation: a novel mouse model for tracheal agenesis. Dev Biol. 2008;322:145–155. doi: 10.1016/j.ydbio.2008.07.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin SS, Tzeng BH, Lee KR, Smith RJ, Campbell KP, Chen CC. Cav3.2 T-type calcium channel is required for the NFAT-dependent Sox9 expression in tracheal cartilage. Proc Natl Acad Sci U S A. 2014;111:E1990–1998. doi: 10.1073/pnas.1323112111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ling L, Nurcombe V, Cool SM. Wnt signaling controls the fate of mesenchymal stem cells. Gene. 2009;433:1–7. doi: 10.1016/j.gene.2008.12.008. [DOI] [PubMed] [Google Scholar]
- Liu F, Kohlmeier S, Wang CY. Wnt signaling and skeletal development. Cell Signal. 2008;20:999–1009. doi: 10.1016/j.cellsig.2007.11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lustig B, Jerchow B, Sachs M, Weiler S, Pietsch T, Karsten U, van de Wetering M, Clevers H, Schlag PM, Birchmeier W, Behrens J. Negative feedback loop of Wnt signaling through upregulation of conductin/axin2 in colorectal and liver tumors. Mol Cell Biol. 2002;22:1184–1193. doi: 10.1128/MCB.22.4.1184-1193.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Majid A, Fernandez L, Fernandez-Bussy S, Herth F, Ernst A. Tracheobronchomalacia. Arch Bronconeumol. 2010;46:196–202. doi: 10.1016/j.arbres.2009.10.011. [DOI] [PubMed] [Google Scholar]
- Mandel H, Shemer R, Borochowitz ZU, Okopnik M, Knopf C, Indelman M, Drugan A, Tiosano D, Gershoni-Baruch R, Choder M, Sprecher E. SERKAL syndrome: an autosomal-recessive disorder caused by a loss-of-function mutation in WNT4. Am J Hum Genet. 2008;82:39–47. doi: 10.1016/j.ajhg.2007.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McAteer JA. Tracheal morphogenesis and fetal development of the mucociliary epithelium of the rat. Scan Electron Microsc, 1995–2008. 1984 [PubMed] [Google Scholar]
- Miller LA, Wert SE, Clark JC, Xu Y, Perl AK, Whitsett JA. Role of Sonic hedgehog in patterning of tracheal-bronchial cartilage and the peripheral lung. Dev Dyn. 2004;231:57–71. doi: 10.1002/dvdy.20105. [DOI] [PubMed] [Google Scholar]
- Miller MF, Cohen ED, Baggs JE, Lu MM, Hogenesch JB, Morrisey EE. Wnt ligands signal in a cooperative manner to promote foregut organogenesis. Proc Natl Acad Sci U S A. 2012;109:15348–15353. doi: 10.1073/pnas.1201583109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mucenski ML, Wert SE, Nation JM, Loudy DE, Huelsken J, Birchmeier W, Morrisey EE, Whitsett JA. beta-Catenin is required for specification of proximal/distal cell fate during lung morphogenesis. J Biol Chem. 2003;278:40231–40238. doi: 10.1074/jbc.M305892200. [DOI] [PubMed] [Google Scholar]
- Muzumdar MD, Tasic B, Miyamichi K, Li L, Luo L. A global double-fluorescent Cre reporter mouse. Genesis. 2007;45:593–605. doi: 10.1002/dvg.20335. [DOI] [PubMed] [Google Scholar]
- Najdi R, Proffitt K, Sprowl S, Kaur S, Yu J, Covey TM, Virshup DM, Waterman ML. A uniform human Wnt expression library reveals a shared secretory pathway and unique signaling activities. Differentiation. 2012;84:203–213. doi: 10.1016/j.diff.2012.06.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oishi I, Suzuki H, Onishi N, Takada R, Kani S, Ohkawara B, Koshida I, Suzuki K, Yamada G, Schwabe GC, Mundlos S, Shibuya H, Takada S, Minami Y. The receptor tyrosine kinase Ror2 is involved in non-canonical Wnt5a/JNK signalling pathway. Genes Cells. 2003;8:645–654. doi: 10.1046/j.1365-2443.2003.00662.x. [DOI] [PubMed] [Google Scholar]
- Ott SR. Confocal microscopy in large insect brains: zinc-formaldehyde fixation improves synapsin immunostaining and preservation of morphology in whole-mounts. J Neurosci Methods. 2008;172:220–230. doi: 10.1016/j.jneumeth.2008.04.031. [DOI] [PubMed] [Google Scholar]
- Ovchinnikov DA, Deng JM, Ogunrinu G, Behringer RR. Col2a1-directed expression of Cre recombinase in differentiating chondrocytes in transgenic mice. Genesis. 2000;26:145–146. [PubMed] [Google Scholar]
- Pan Q, Yu Y, Chen Q, Li C, Wu H, Wan Y, Ma J, Sun F. Sox9, a key transcription factor of bone morphogenetic protein-2-induced chondrogenesis, is activated through BMP pathway and a CCAAT box in the proximal promoter. J Cell Physiol. 2008;217:228–241. doi: 10.1002/jcp.21496. [DOI] [PubMed] [Google Scholar]
- Park J, Zhang JJ, Moro A, Kushida M, Wegner M, Kim PC. Regulation of Sox9 by Sonic Hedgehog (Shh) is essential for patterning and formation of tracheal cartilage. Dev Dyn. 2010;239:514–526. doi: 10.1002/dvdy.22192. [DOI] [PubMed] [Google Scholar]
- Que J, Luo X, Schwartz RJ, Hogan BL. Multiple roles for Sox2 in the developing and adult mouse trachea. Development. 2009;136:1899–1907. doi: 10.1242/dev.034629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rajagopal J, Carroll TJ, Guseh JS, Bores SA, Blank LJ, Anderson WJ, Yu J, Zhou Q, McMahon AP, Melton DA. Wnt7b stimulates embryonic lung growth by coordinately increasing the replication of epithelium and mesenchyme. Development. 2008;135:1625–1634. doi: 10.1242/dev.015495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raspopovic J, Marcon L, Russo L, Sharpe J. Modeling digits. Digit patterning is controlled by a Bmp-Sox9-Wnt Turing network modulated by morphogen gradients. Science. 2014;345:566–570. doi: 10.1126/science.1252960. [DOI] [PubMed] [Google Scholar]
- Rock JR, Futtner CR, Harfe BD. The transmembrane protein TMEM16A is required for normal development of the murine trachea. Dev Biol. 2008;321:141–149. doi: 10.1016/j.ydbio.2008.06.009. [DOI] [PubMed] [Google Scholar]
- Sala FG, Del Moral PM, Tiozzo C, Alam DA, Warburton D, Grikscheit T, Veltmaat JM, Bellusci S. FGF10 controls the patterning of the tracheal cartilage rings via Shh. Development. 2011;138:273–282. doi: 10.1242/dev.051680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shannon JM, Hyatt BA. Epithelial-mesenchymal interactions in the developing lung. Annu Rev Physiol. 2004;66:625–645. doi: 10.1146/annurev.physiol.66.032102.135749. [DOI] [PubMed] [Google Scholar]
- Shu W, Jiang YQ, Lu MM, Morrisey EE. Wnt7b regulates mesenchymal proliferation and vascular development in the lung. Development. 2002;129:4831–4842. doi: 10.1242/dev.129.20.4831. [DOI] [PubMed] [Google Scholar]
- Snowball J, Ambalavanan M, Cornett B, Lang R, Whitsett J, Sinner D. Mesenchymal Wnt signaling promotes formation of sternum and thoracic body wall. Dev Biol. 2015;401:264–275. doi: 10.1016/j.ydbio.2015.02.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sosic D, Richardson JA, Yu K, Ornitz DM, Olson EN. Twist regulates cytokine gene expression through a negative feedback loop that represses NF-kappaB activity. Cell. 2003;112:169–180. doi: 10.1016/s0092-8674(03)00002-3. [DOI] [PubMed] [Google Scholar]
- Tiozzo C, De Langhe S, Carraro G, Alam DA, Nagy A, Wigfall C, Hajihosseini MK, Warburton D, Minoo P, Bellusci S. Fibroblast growth factor 10 plays a causative role in the tracheal cartilage defects in a mouse model of Apert syndrome. Pediatr Res. 2009;66:386–390. doi: 10.1203/PDR.0b013e3181b45580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tompkins DH, Besnard V, Lange AW, Wert SE, Keiser AR, Smith AN, Lang R, Whitsett JA. Sox2 is required for maintenance and differentiation of bronchiolar Clara, ciliated, and goblet cells. PLoS One. 2009;4:e8248. doi: 10.1371/journal.pone.0008248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turcatel G, Rubin N, Menke DB, Martin G, Shi W, Warburton D. Lung mesenchymal expression of Sox9 plays a critical role in tracheal development. BMC Biol. 2013;11:117. doi: 10.1186/1741-7007-11-117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang F, Flanagan J, Su N, Wang LC, Bui S, Nielson A, Wu X, Vo HT, Ma XJ, Luo Y. RNAscope: a novel in situ RNA analysis platform for formalin-fixed, paraffin-embedded tissues. J Mol Diagn. 2012;14:22–29. doi: 10.1016/j.jmoldx.2011.08.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yin Y, White AC, Huh SH, Hilton MJ, Kanazawa H, Long F, Ornitz DM. An FGF-WNT gene regulatory network controls lung mesenchyme development. Dev Biol. 2008;319:426–436. doi: 10.1016/j.ydbio.2008.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.