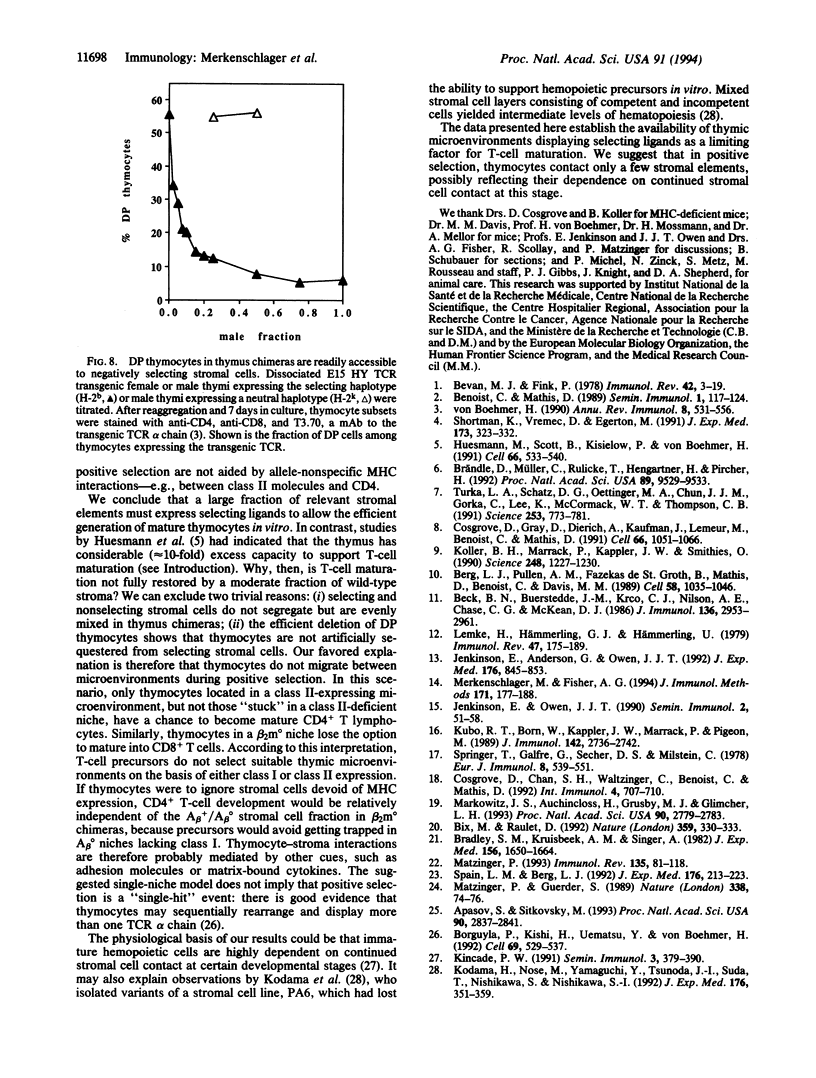
Abstract
Thymocyte maturation depends on interactions with thymic stromal elements expressing major histocompatibility complex (MHC) molecules. Mutant mouse strains lacking MHC class I (beta 2-microglobulin-null) or class II (A beta-null) expression fail to generate normal CD8 or CD4 T-cell populations and provide model systems for reconstitution experiments. We have constructed in vitro chimeras between normal and MHC-deficient thymi to evaluate the efficiency of positive selection. Unexpectedly, the generation of mature single-positive thymocytes was proportional to the fraction of wild-type (i.e., MHC-expressing) stroma over a wide range of chimerism. Similar results were obtained for the development of T-cell receptor-transgenic thymocytes in graded chimeras expressing selecting and nonselecting MHC alleles. These findings are best explained by hypothesizing that positive selection involves a rate-limiting step at which each thymocyte can interact with only one stromal cell niche.
Full text
PDF
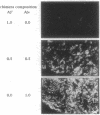
Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Apasov S., Sitkovsky M. Highly lytic CD8+, alpha beta T-cell receptor cytotoxic T cells with major histocompatibility complex (MHC) class I antigen-directed cytotoxicity in beta 2-microglobulin, MHC class I-deficient mice. Proc Natl Acad Sci U S A. 1993 Apr 1;90(7):2837–2841. doi: 10.1073/pnas.90.7.2837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beck B. N., Buerstedde J. M., Krco C. J., Nilson A. E., Chase C. G., McKean D. J. Characterization of cell lines expressing mutant I-Ab and I-Ak molecules allows the definition of distinct serologic epitopes on A alpha and A beta polypeptides. J Immunol. 1986 Apr 15;136(8):2953–2961. [PubMed] [Google Scholar]
- Benoist C., Mathis D. Positive and negative selection of the T cell repertoire in MHC class II transgenic mice. Semin Immunol. 1989 Nov;1(2):117–124. [PubMed] [Google Scholar]
- Berg L. J., Pullen A. M., Fazekas de St Groth B., Mathis D., Benoist C., Davis M. M. Antigen/MHC-specific T cells are preferentially exported from the thymus in the presence of their MHC ligand. Cell. 1989 Sep 22;58(6):1035–1046. doi: 10.1016/0092-8674(89)90502-3. [DOI] [PubMed] [Google Scholar]
- Bevan M. J., Fink P. J. The influence of thymus H-2 antigens on the specificity of maturing killer and helper cells. Immunol Rev. 1978;42:3–19. doi: 10.1111/j.1600-065x.1978.tb00256.x. [DOI] [PubMed] [Google Scholar]
- Bix M., Raulet D. Inefficient positive selection of T cells directed by haematopoietic cells. Nature. 1992 Sep 24;359(6393):330–333. doi: 10.1038/359330a0. [DOI] [PubMed] [Google Scholar]
- Borgulya P., Kishi H., Uematsu Y., von Boehmer H. Exclusion and inclusion of alpha and beta T cell receptor alleles. Cell. 1992 May 1;69(3):529–537. doi: 10.1016/0092-8674(92)90453-j. [DOI] [PubMed] [Google Scholar]
- Bradley S. M., Kruisbeek A. M., Singer A. Cytotoxic T lymphocyte responses in allogeneic radiation bone marrow chimeras. The chimeric host strictly dictates the self-repertoire of Ia-restricted T cells but not H-2K/D-restricted T cells. J Exp Med. 1982 Dec 1;156(6):1650–1664. doi: 10.1084/jem.156.6.1650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brändle D., Müller C., Rülicke T., Hengartner H., Pircher H. Engagement of the T-cell receptor during positive selection in the thymus down-regulates RAG-1 expression. Proc Natl Acad Sci U S A. 1992 Oct 15;89(20):9529–9533. doi: 10.1073/pnas.89.20.9529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cosgrove D., Chan S. H., Waltzinger C., Benoist C., Mathis D. The thymic compartment responsible for positive selection of CD4+ T cells. Int Immunol. 1992 Jun;4(6):707–710. doi: 10.1093/intimm/4.6.707. [DOI] [PubMed] [Google Scholar]
- Cosgrove D., Gray D., Dierich A., Kaufman J., Lemeur M., Benoist C., Mathis D. Mice lacking MHC class II molecules. Cell. 1991 Sep 6;66(5):1051–1066. doi: 10.1016/0092-8674(91)90448-8. [DOI] [PubMed] [Google Scholar]
- Huesmann M., Scott B., Kisielow P., von Boehmer H. Kinetics and efficacy of positive selection in the thymus of normal and T cell receptor transgenic mice. Cell. 1991 Aug 9;66(3):533–540. doi: 10.1016/0092-8674(81)90016-7. [DOI] [PubMed] [Google Scholar]
- Jenkinson E. J., Anderson G., Owen J. J. Studies on T cell maturation on defined thymic stromal cell populations in vitro. J Exp Med. 1992 Sep 1;176(3):845–853. doi: 10.1084/jem.176.3.845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jenkinson E. J., Owen J. J. T-cell differentiation in thymus organ cultures. Semin Immunol. 1990 Jan;2(1):51–58. [PubMed] [Google Scholar]
- Kincade P. W. Molecular interactions between stromal cells and B lymphocyte precursors. Semin Immunol. 1991 Nov;3(6):379–390. [PubMed] [Google Scholar]
- Kodama H., Nose M., Yamaguchi Y., Tsunoda J., Suda T., Nishikawa S., Nishikawa S. In vitro proliferation of primitive hemopoietic stem cells supported by stromal cells: evidence for the presence of a mechanism(s) other than that involving c-kit receptor and its ligand. J Exp Med. 1992 Aug 1;176(2):351–361. doi: 10.1084/jem.176.2.351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koller B. H., Marrack P., Kappler J. W., Smithies O. Normal development of mice deficient in beta 2M, MHC class I proteins, and CD8+ T cells. Science. 1990 Jun 8;248(4960):1227–1230. doi: 10.1126/science.2112266. [DOI] [PubMed] [Google Scholar]
- Kubo R. T., Born W., Kappler J. W., Marrack P., Pigeon M. Characterization of a monoclonal antibody which detects all murine alpha beta T cell receptors. J Immunol. 1989 Apr 15;142(8):2736–2742. [PubMed] [Google Scholar]
- Lemke H., Hämmerling G. J., Hämmerling U. Fine specificity analysis with monoclonal antibodies of antigens controlled by the major histocompatibility complex and by the Qa/TL region in mice. Immunol Rev. 1979;47:175–206. doi: 10.1111/j.1600-065x.1979.tb00293.x. [DOI] [PubMed] [Google Scholar]
- Markowitz J. S., Auchincloss H., Jr, Grusby M. J., Glimcher L. H. Class II-positive hematopoietic cells cannot mediate positive selection of CD4+ T lymphocytes in class II-deficient mice. Proc Natl Acad Sci U S A. 1993 Apr 1;90(7):2779–2783. doi: 10.1073/pnas.90.7.2779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matzinger P., Guerder S. Does T-cell tolerance require a dedicated antigen-presenting cell? Nature. 1989 Mar 2;338(6210):74–76. doi: 10.1038/338074a0. [DOI] [PubMed] [Google Scholar]
- Matzinger P. Why positive selection? Immunol Rev. 1993 Oct;135:81–117. doi: 10.1111/j.1600-065x.1993.tb00645.x. [DOI] [PubMed] [Google Scholar]
- Merkenschlager M., Fisher A. G. In vitro construction of graded thymus chimeras. J Immunol Methods. 1994 May 16;171(2):177–188. doi: 10.1016/0022-1759(94)90038-8. [DOI] [PubMed] [Google Scholar]
- Shortman K., Vremec D., Egerton M. The kinetics of T cell antigen receptor expression by subgroups of CD4+8+ thymocytes: delineation of CD4+8+3(2+) thymocytes as post-selection intermediates leading to mature T cells. J Exp Med. 1991 Feb 1;173(2):323–332. doi: 10.1084/jem.173.2.323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spain L. M., Berg L. J. Developmental regulation of thymocyte susceptibility to deletion by "self"-peptide. J Exp Med. 1992 Jul 1;176(1):213–223. doi: 10.1084/jem.176.1.213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Springer T., Galfrè G., Secher D. S., Milstein C. Monoclonal xenogeneic antibodies to murine cell surface antigens: identification of novel leukocyte differentiation antigens. Eur J Immunol. 1978 Aug;8(8):539–551. doi: 10.1002/eji.1830080802. [DOI] [PubMed] [Google Scholar]
- Turka L. A., Schatz D. G., Oettinger M. A., Chun J. J., Gorka C., Lee K., McCormack W. T., Thompson C. B. Thymocyte expression of RAG-1 and RAG-2: termination by T cell receptor cross-linking. Science. 1991 Aug 16;253(5021):778–781. doi: 10.1126/science.1831564. [DOI] [PubMed] [Google Scholar]
- von Boehmer H. Developmental biology of T cells in T cell-receptor transgenic mice. Annu Rev Immunol. 1990;8:531–556. doi: 10.1146/annurev.iy.08.040190.002531. [DOI] [PubMed] [Google Scholar]