Abstract
Multiple sclerosis (MS) is an immune-mediated demyelinating disease of the central nervous system that has been linked with defects in regulatory T cell function. Therefore, strategies to selectively target pathogenic cells via enhanced regulatory T cell activity may provide therapeutic benefit. Kv1.3 is a voltage-gated potassium channel expressed on myelin-reactive T cells from MS patients. Kv1.3 KO mice are protected from experimental autoimmune encephalomyelitis (EAE), an animal model of MS, and Kv1.3 KO Th cells display suppressive capacity associated with increased IL-10. Herein, we demonstrate that MOG-specific Kv1.3 KO Th cells exhibit a unique regulatory phenotype characterized by high CD25, CTLA4, pSTAT5, FoxO1, and GATA1 expression without a corresponding increase in Foxp3. These phenotypic changes result from increased signaling through the IL-2 receptor. Moreover, MOG-specific Kv1.3 KO Th cells can ameliorate EAE following transfer to WT recipients in a manner that is partially dependent on IL-2 receptor and STAT5 signaling. The present study identifies a population of Foxp3-negative T cells with suppressive properties that arise in the absence of Kv1.3 and enhances the understanding of the molecular mechanism by which these cells are generated. This increased understanding could contribute to the development of novel therapies for MS patients that promote heightened immune regulation.
INTRODUCTION
Multiple sclerosis (MS) is an immune-mediated disease of the central nervous system (CNS) that results in demyelination and axonal loss(1, 2). Recent evidence indicates that regulatory T cells (Tregs) with decreased suppressive capacity may contribute to the ongoing inflammation that exists in MS (3-6). Studies in experimental autoimmune encephalomyelitis (EAE), an animal model commonly used to study MS, have also demonstrated an important role for Tregs in controlling susceptibility and severity of disease, and a recently identified population of FoxA1+ Tregs has been shown to arise in relapsing remitting MS patients who demonstrate a favorable clinical response to IFN-β therapy(7). A need exists for novel therapies that specifically target and eliminate pathogenic cells without compromising the protective immune response(8, 9). Treatment strategies focused on enhancing the functional Treg response are being actively explored for such purposes.
Kv1.3 is an outward rectifying voltage-gated potassium channel that has been shown to be important for maintaining the membrane potential by promoting a countercurrent efflux of potassium to allow influx of extracellular calcium through calcium release activated channels (1, 10-12). Kv1.3 has been shown to be highly expressed on antigen experienced T effector memory cells from MS patients and has been investigated as a therapeutic target for T cell-mediated autoimmune disease for over a decade (12-15). Studies performed in rodent and human T cells suggest that blockade of Kv1.3 may be beneficial for maintaining immune regulation and homeostasis(11, 16). Recently, we have demonstrated that gene deletion of Kv1.3 in mice results in significantly decreased incidence and severity of EAE. This decreased disease severity correlated with an increase in the frequency of IL-10-producing Kv1.3 KO Th cells that were able to suppress activation of effector T cells following immunization with myelin peptide(17). Importantly, this population of T cells does not express Foxp3 suggesting the identification of a potentially novel subset of T helper cells with suppressive properties. The molecular mechanisms underlying the development of this type of T cell are unclear.
Herein, we elucidated the molecular mechanisms contributing to the development of Foxp3 negative Th cells with suppressive properties that were identified in Kv1.3 KO mice and investigated the therapeutic potential of these cells in EAE. Our data demonstrate that antigenic stimulation of MOG-specific Kv1.3 KO Th cells in vitro results in significant upregulation of CD25 and CTLA4 in association with an increase in pSTAT5, nuclear FoxO1, and GATA1 expression. Importantly, this phenotypic change is not a result of impaired intracellular calcium flux as might be expected. Moreover, as these changes are not accompanied by increased expression of Foxp3, and differ in phenotype from TR1 cells based on high CD25 expression and increased IL-4 production, we believe that we have identified a novel subset of Th cells with regulatory capacity. Importantly, our data demonstrate that MOG-specific Kv1.3 KO Th cells are able to ameliorate EAE induction in vivo, at least in part, via an IL-2 receptor-dependent mechanism. Furthermore, unlike canonical Tregs, Kv1.3 KO Th cells secrete large amounts of IL-2 in vitro, which may render them a uniquely self-sustaining regulatory Th cell population. Further study of Kv1.3 KO Th cells could elucidate novel mechanisms or pathways to enhance regulation of immune responses in immune-mediated diseases such as MS.
MATERIALS AND METHODS
Mice
C57BL/6 (CD45.2+) and CD45.1+ congenic mice were purchased from the National Cancer Institute (Frederick, MD). Mice with a transgenic T cell receptor specific for MOG 35-55 (2D2 mice) were purchased from The Jackson Laboratories (Bar Harbor, ME). Kv1.3 KO mice on the C57BL/6 background were a kind gift from Dr. Leonard Kaczmarek (Yale University, New Haven, CT) and were bred and maintained in our animal facility. Kv1.3 KO mice expressing MOG-specific TCR (2D2-Kv1.3 KO) were generated and bred in our facility. Mice of 8 to 12 weeks of age were used in all of the experiments. Age-, sex-, and genotype-matched mice were used in all experiments as controls.
Induction of EAE
For EAE induction, mice were immunized subcutaneously over two sites on the lateral abdomen with 100 μg of myelin oligodendrocyte glycoprotein (MOG 35-55) peptide (The Johns Hopkins Peptide Synthesis Core Facility, Baltimore, MD) in Complete Freund's Adjuvant (CFA) containing 4 μg/ml Mycobacterium tuberculosis H37Ra (Difco Laboratories, Detroit, MI), followed by intraperitoneal injections of 250 ng of Bordetella pertussis toxin (List Biological Laboratories, Campbell, CA) on day 0 and 2 after immunization. Mice were scored daily for disease progression using the following scale: 0, no disease; 1, limp tail; 2, hind-limb weakness; 3, hind-limb paralysis; 4, quadriplegia or moribund; 5, death due to EAE.
In vivo suppression assay
For in vivo suppression assay, 2D2-WT and 2D2-Kv1.3 KO Th cells were cultured with irradiated WT splenocytes and 10 μg/ml MOG 35-55 for 72 hours. Ficoll gradient centrifugation was then used to remove dead cells, resulting in >90% pure CD4+ T cells, and 5 × 106 cells were transferred intraperitoneally into CD45.1+ congenic recipients a day prior to immunization. One day after transfer, the recipient mice were immunized to induce EAE as described above.
CFSE proliferation assay
Following isolation, 2D2-Kv1.3 KO CD4+ T cells were labeled with CFSE (Molecular Probes, Carlsbad, CA) according to manufacturer's instructions. Briefly, cells were resuspended at a concentration of 1 × 106 per ml in 0.1 % BSA in PBS and incubated with 0.25 μM CFSE at 37°C for 10 minutes. The staining was quenched with addition of 5 volumes of culture media containing 10% FBS. The cells were centrifuged, washed, and resuspended in complete RPMI 1640 media. The cells were then plated at 1 × 106 per ml and stimulated with 10 μg/ml MOG 35-55 and irradiated APCs in the presence or absence of 20 μg/ml rIL-2. The cells were cultured for 72 hours, stained with an anti-CD4-APC antibody (BD Biosciences, San Jose, CA), and run on the FACS Calibur flow cytometer to assess cell division. The data were analyzed using FlowJo software (Treestar, Ashland, OR).
In vitro Suppression Assay
CD4+ T cells were isolated from 2D2-Kv1.3 KO or 2D2-WT mice and stimulated with 10μg/ml MOG 35-55 +irradiated APCs in the presence of 10μg/ml anti-IL-2, 10μg/ml anti-IL-10, 20μg/ml anti-CTLA-4, or isotype control antibody. Immediately preceding re-stimulation, Kv1.3 KO or WT T cells were incubated with WT naïve CD4+ CFSE-labeled responder cells at a ratio of suppressors: responders = 5:1. The cells were stimulated with anti-CD3/CD28 beads for an additional 72 hours and proliferation of responder cells was assessed using a FACS Calibur flow cytometer. Data was analyzed using FlowJo software.
Perfusion and Immunostaining
EAE mice receiving 2D2-Kv1.3 KO or 2D2-WT Th cells were anesthetized, perfused with HBSS, and blood, lymph nodes, spleens and brains were removed. CNS tissue was digested and enriched for mononuclear cells. Single cell suspensions were made from blood, lymph nodes, and spleens as described below. Collected cells were stimulated in media containing PMA (50 ng/ml; Sigma-Aldrich, St. Louis, MO), ionomycin (1 μg/ml; Sigma-Aldrich), and monensin (4 μl/6 ml; GolgiStop, BD Biosciences) at 37°C for 4-6 hrs. Cells were washed with FACS buffer (2% FBS in PBS) and surface stained with anti-CD4 (BD Biosciences, RM4-5) Percep or APC, anti-CD45.2 FITC, anti-CD11b APC, and anti-Ly6G PE(BD Bioscience, San Jose, CA and eBioscience, San Diego, CA). Flow cytometry was performed using a FACS Calibur flow cytometer and data were analyzed using FlowJo software (Treestar, Ashland, OR).
Cell culture and polarization
Spleens and lymph nodes were isolated from immunized or naïve mice, and single-cell suspensions were made by passing through a 70-μm nylon cell strainer (BD Biosciences, San Jose, CA). Th cells were isolated from splenocytes by negative selection using EasySep Mouse CD4+ T cell Enrichment Kit (StemCell Technologies, Vancouver, British Columbia, Canada), following the manufacturer's protocol. Cells were cultured in RPMI 1640 (Life Technologies, Grand Island, NY) supplemented with 10% v/v FBS (Life Technologies), 100 μg/ml penicillin and streptomycin (Quality Biological, Gaithersburg, MD), 0.5 μM 2-mercaptoethanol (Life Technologies), 10 mM HEPES buffer (Quality Biological), 1 mM sodium pyruvate (Sigma-Aldrich, St. Louis, MO), and MEM NEAA (Sigma-Aldrich). For polarization to Th1, cells were cultured in complete RPMI in the presence of 10 ng/ml IL-12. For polarization to Treg, cells were cultured in complete RPMI in the presence of 5 ng/ml TGF-β. For polarization to Th17, cells were cultured in IMDM-based medium in the presence of 20 ng/ml IL-6, 5 ng/ml TGF-β, and 20 ng/ml IL-23. For polarization to Th2, cells were cultured in complete RPMI in the presence of 20 ng/ml IL-4. Th0 cells were cultured in complete RPMI without polarizing cytokines. For blockade experiments, cells were cultured in the presence of 10μg/ml anti-IL-2, 10μg/ml anti-IL-10, 20μg/ml antic-CTLA-4, or isotype control antibody prior to stimulation with MOG 35-55 peptide. Exogenous rIL-2 was used at 20ng/ml.
Flow cytometry
For intracellular cytokine staining, cells were stimulated with Cell Stimulation Cocktail plus protein transport inhibitors (eBioscience, San Diego, CA) for 4 – 6 hours. Surface antigens were stained with the following antibodies: anti-CD4 (BD Biosciences, RM4-5), anti-CD44 (BD Biosciences, IM7), anti-CD62L (eBioscience, MEL-14), anti-CD45.1, and anti-CD25 (BD Biosciences, 7D4). For intracellular staining, cells were fixed and permeabilized with the Foxp3 staining buffer kit (eBioscience) and stained for intracellular cytokines and proteins with anti-IFN-γ (BD Biosciences, XMG1.2), anti-IL-17 (BioLegend, TC11-18H10.1), anti-CTLA-4, and anti-Foxp3 (eBioscience, FJK-16s) antibodies. Flow cytometric analyses were performed on a FACSCalibur instrument (BD Biosciences) and analyzed using FlowJo software (TreeStar, Ashland, OR).
Calcium flux
CD4+ T cells were cultured in complete RPMI 1640 and activated with anti-CD3/CD28-coated beads. The cells were either examined immediately ex vivo or after 72 hours of stimulation using a flow cytometric assay. To examine Ca2+ flux, the cells were loaded with the Ca2+ indicator dyes Fluo-4 and Fura Red, surface stained for CD4, and suspended in RPMI. Intracellular Ca2+ amounts were plotted as a median ratio of Fluo-4/Fura Red over time in order to minimize the variability in dye loading. To induce TCR signaling, CD4+ T cells were precoated with biotinylated anti-CD3ε antibody and treated with streptavidin.
ELISA
To quantify cytokine production, supernatants from cultured cells were collected after 72 hours, and the levels of IL-2, IFNγ, IL-17, IL-10, and IL-4 were quantified using ELISA kits according to manufacturer's instructions (BioLegend, San Diego, CA).
Western blotting
For whole cell protein extracts, cells were collected and lysed with RIPA buffer (Boston Bioproducts, Ashland, MA). Protein was quantitated using the BCA assay (Pierce, Rockford, IL). Lysates were electrophoretically separated using 12% Tris HCl gels (Biorad, Hercules, CA) and transferred to nitrocellulose membranes. Western blotting was performed using antibodies specific for the following proteins: pSTAT5, STAT5, pAkt, Akt, p56lck, phosphor-p56lck, pErk1/2, Erk1/2, pFoxO1/FoxO3a, FoxO1, GATA-1 (all from Cell Signaling Technologies, Danvers, MA), NFATc1, goat anti-rabbit IgG-HRP, and goat anti-mouse IgG-HRP. Average densitometric ratio was calculated for each blot by normalizing to an actin loading control using Image J software.
Statistical analysis
Statistical analysis was conducted using the GraphPad Prism software (GraphPad, San Diego, CA). Mann-Whitney test was used to compare data from noninterval scales, e.g. clinical EAE scores. Two-tailed Student t-test was used to analyze normally distributed data. Results were considered significant if the p value was < 0.05: * p < 0.05, ** p < 0.01, *** p < 0.001, NS = no significant difference. Error bars represent standard error of the mean (SEM) in all the figures.
Study Approval
All mice were maintained in a federally approved animal facility at The Johns Hopkins University (Baltimore, MD) in accordance with guidelines and approval from the Institutional Animal Care and Use Committee.
RESULTS
MOG-specific Kv1.3 KO Th cells express significantly more CD25 and CTLA4 but less Foxp3 than WT cells
Antigen-specific regulatory T cells have the potential to eliminate pathogenic autoreactive cells within the target organ more efficiently than polyclonal regulatory T cells(8). To determine whether Kv1.3 KO Th cells with specificity for myelin antigens retain the regulatory properties observed in Th cells from the Kv1.3 KO EAE animals (17), we crossed the Kv1.3 KO mouse to the 2D2 TCR transgenic mouse that expresses the Vα3.2/Vβ11 TCR specific for MOG 35-55 peptide (18). Directly ex-vivo, 2D2-WT and 2D2-Kv1.3 KO Th cells isolated from naïve animals exhibited equivalent expression of CD44, CD62L, CD25, CTLA4, and Foxp3 (Figure 1a,c). Following stimulation with MOG-35-55 in the presence of irradiated WT APCs (or KO APCs), 2D2-WT and 2D2-Kv1.3 KO Th cells expressed equivalent levels of the activation markers CD44 and CD62L, suggesting that the T cells from 2D2 Kv1.3 KO mice do not display a different threshold for activation compared to WT T cells. However, 2D2-Kv1.3 KO Th cells expressed significantly higher levels of CD25 than the 2D2-WT cells (Figure 1b,d). Interestingly, this CD25hi population of cells was not Foxp3+ but did exhibit increased expression of CTLA4, a molecule implicated in canonical regulatory T cell function. These data indicate that antigen-specific Kv1.3 KO Th cells have a phenotype consistent with the suppressive phenotype observed in the Kv1.3 KO EAE animals.
Figure 1. MOG-specific Kv1.3 KO Th cells express significantly more CD25 and CTLA4 but less Foxp3 than WT cells.
2D2-WT and 2D2-Kv1.3 KO Th cells were cultured with irradiated WT APCs and 10 μg/ml MOG 35-55 for 72 h. Expression of CD44, CD62L, CD25, CTLA4 and Foxp3 was analyzed by FACS analysis either directly ex vivo (a,c) or after 72 hours of stimulation (b,d). Plots are gated on CD4+ cells. Graphs below represent data from at least 3 independent experiments.
2D2-Kv1.3 KO Th cells secrete significantly more IL-10, IL-4, and IL-2 and significantly less IFN-γ and IL-17
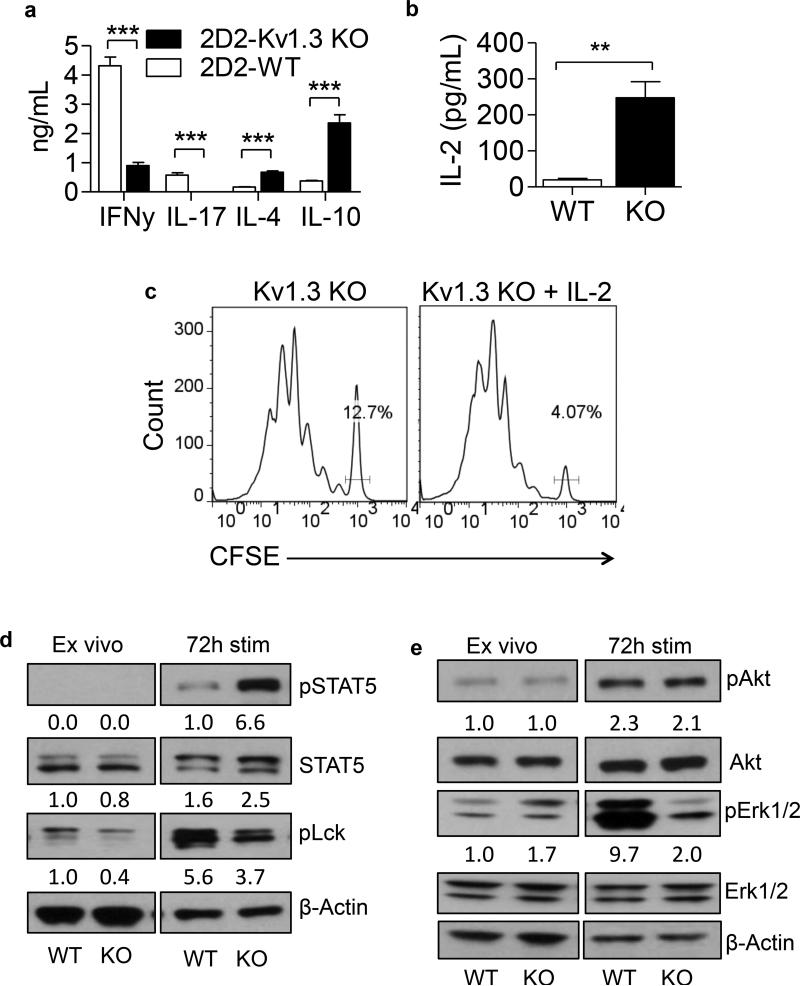
It has been well described in the literature that Th cells of the Th1 and Th17 lineages are detrimental in MS(19-24). Much effort in the field has been focused on deviating the phenotype of the pathogenic T cells away from Th1 and Th17 and toward Th2 and Treg (25-29). Moreover, antigen-specific regulatory T cells have the ability to suppress the production of IFN-γ and IL-17 from myelin-reactive T cells (8, 30). Further examination revealed that 2D2-Kv1.3 KO Th cells stimulated with MOG in vitro secrete increased amounts of IL-4 and IL-10 and significantly less of the pro-inflammatory cytokines IFN-γ and IL-17 than the 2D2-WT cells, consistent with deviation toward a regulatory or suppressive phenotype (Figure 2a). Importantly, 2D2-Kv1.3 KO Th cells also secreted significantly more IL-2 than the WT controls (Figure 2b) and displayed an enhanced proliferative response to IL-2 (Figure 2c), indicating that, unlike conventional regulatory T cells, the Kv1.3 KO Th cells may be able to sustain their own proliferation without exogenous IL-2.
Figure 2. 2D2-Kv1.3 KO Th cells display increased IL-10, IL-4, and IL-2 secretion and increased expression of pSTAT5 accompanied by decreased IFN-γ and IL-17 secretion and decreased phosphorylation of p56lck and Erk1/2.
2D2-WT and 2D2-Kv1.3 KO Th cells were stimulated with MOG 35-55 and irradiated APCs for 0 or 72 h. (a) Production of IFNγ, IL-17, IL-4, and IL-10 were analyzed by ELISA. (b) Production of IL-2 was analyzed by ELISA. (c) 2D2-Kv1.3 KO Th cells were CFSE labeled and stimulated with 10μg/ml MOG 35-55 + irradiated APCs for 72 h in the presence or absence of 20ng/ml rIL-2. CFSE dilution in CD4+ T cells was analyzed by FACS analysis. The percent of undivided cells is shown on the histograms. (d) STAT5, p56lck, β-Actin, and (e) Akt and Erk1/2 expression and phosphorylation were determined using western blotting. Numbers below bands indicate average densitometric ratio normalized to total protein (for phospho blots) or actin loading control.
Antigenic stimulation of 2D2-Kv1.3 KO Th cells results in increased phosphorylation of STAT5 and decreased phosphorylation of p56lck and Erk1/2
To further characterize the signaling pathway that culminates in increased CD25 and CTLA4 expression and IL-4, IL-10 and IL-2 production by Kv1.3 KO Th cells, we sought to examine TCR and IL-2 receptor (IL-2R) signaling following antigenic stimulation of 2D2-WT and 2D2-Kv1.3 KO Th cells. Expression and phosphorylation of STAT5, p56lck, Akt and Erk in 2D2-WT and 2D2-Kv1.3 KO Th cells were examined directly ex vivo or following 72 hours of stimulation with MOG 35-55 and irradiated WT APCs. Interestingly, we found significantly increased phosphorylation of STAT5 in 2D2-Kv1.3 KO Th cells compared to 2D2-WT controls (Figure 2d). This finding correlated with a slight decrease in phosphorylation of p56lck and a significant decrease in phosphorylation of Erk1/2 (Figure 2d and e). No significant changes in phosphorylation of AKT were observed. These data suggest that absence of Kv1.3 results in decreased signaling through the TCR and increased signaling through the IL-2R/STAT5 pathway. Importantly, these increases in pSTAT5 were observed in 2D2- Kv1.3 KO T cells regardless of whether cells were stimulated in the presence of WT or Kv1.3 KO APCs (Figure S1a and b), indicating that the Th cell suppressive phenotype is due to T cell-intrinsic signaling events.
The regulatory phenotype of Kv1.3 KO Th cells is not a result of depletion of intracellular calcium or changes in NFAT signaling
Kv1.3 plays a fundamental role in the activation of T cells through regulation of cytoplasmic free calcium levels (12). Thus, we sought to investigate whether the regulatory phenotype of Kv1.3 KO Th cells associated with decreased proximal TCR signaling could be attributed to defective calcium signaling, which could lead to the development of anergic T cells with suppressive capabilities. To this end, calcium flux induced by TCR cross-linking was evaluated in Kv1.3 KO and WT Th cells either directly ex vivo or following 72 hours of stimulation using a flow cytometric method. Surprisingly, no difference was observed in calcium flux in Kv1.3 KO T cells compared to WT controls (Figure 3a). To confirm that there is no defect in intracellular calcium signaling in Kv1.3 KO Th cells, Th cells were stimulated in vitro for 24 hours and expression and nuclear translocation of the calcium sensitive transcription factor NFAT were evaluated by western blotting. No difference in NFAT activation and translocation was observed, confirming that intracellular calcium signaling is unaffected in the absence of Kv1.3 (Figure 3b), likely due to compensation from other potassium or chloride channels (31). These data highlight an important role for Kv1.3 in TCR signaling that is independent of its role as an ion channel.
Figure 3. Kv1.3 is not required for TCR crosslinking-induced elevation in cytoplasmic free Ca2+ by naïve or effector T cells.
(a) WT (red line) and Kv1.3 KO (blue line) Th cells were loaded with the Ca2+ indicator dyes Fluo-4 and Fura Red, surface stained for CD4, and suspended in RPMI either directly ex vivo or 72 h after stimulation with anti-CD3/CD28-coated beads. Th cells were precoated with biotinylated anti-CD3ε antibody and treated with streptavidin (TCR) at the indicated time (arrow). Intracellular Ca2+ amounts were plotted as a median ratio of Fluo-4/Fura Red over time in order to minimize the variability in dye loading. (b) WT and Kv1.3 KO Th cells were stimulated with anti-CD3/CD28 beads for 24 hours. Nuclear and cytoplasmic extracts were prepared and NFATc1 expression and nuclear translocation were determined using western blotting. Numbers below bands indicate average densitometric ratio normalized to actin loading control.
FoxO1 nuclear localization and GATA1 expression are increased following stimulation of Kv1.3 KO Th cells in vitro
Forkhead box O (FoxO) transcription factors have been demonstrated to regulate cell cycle progression, survival and homeostasis (32, 33). Under conditions of strong TCR stimulation, FoxO proteins are phosphorylated by a variety of molecules including Akt, Erk, and Sgk1, and shuttled from the nucleus to the cytoplasm, where they may be targeted for degradation (34). FoxO proteins have been demonstrated to control immune homeostasis and function of regulatory T cells via unique transcriptional programs that are not dependent on Foxp3 (35). Moreover, FoxO1 has been shown to regulate transcription of CTLA4 directly and to repress IFN-γ expression in Tregs (33, 35). To investigate whether FoxO1 expression was increased in Kv1.3 KO Th cells and could thus be inducing or repressing transcription of regulatory or inflammatory genes, respectively, we examined the phosphorylation and nuclear translocation of FoxO1 in Kv1.3 KO, WT, 2D2-Kv1.3 KO, and 2D2-WT Th cells. We observed a significant increase in nuclear FoxO1 expression, which correlated directly with decreased phosphorylation of the protein in 2D2-Kv1.3 KO T cells as well as Kv1.3 KO non-TCR transgenic T cells (Figure 4a-b). Moreover, it has recently been described that the transcriptional signature of regulatory T cells is comprised of multiple transcription factors that act in a redundant fashion (36). One of the transcription factors in addition to Foxp3 that comprises this signature is GATA1. Because Kv1.3 KO T cells do not express increased Foxp3, we investigated the possibility that perhaps one or more of the other Treg signature molecules might play a role in promoting the regulatory phenotype observed. To this end, CD4+ T cells from Kv1.3 KO or WT mice were stimulated in vitro, and GATA1 expression was examined by western blotting. Interestingly, Kv1.3 KO T cells consistently express significantly more GATA1 than WT T cells (Figure 4c), supporting a novel regulatory role for GATA1 not only in lymphocytes, but specifically in Kv1.3-deficient T cells.
Figure 4. FoxO1 nuclear localization and GATA1 expression are increased following stimulation of Kv1.3 KO Th cells in vitro.
(a) WT and Kv1.3 KO Th cells were stimulated for 24 hours. Expression of FoxO1 and β-Actin was examined in cytoplasmic and nuclear extracts. (b) 2D2-WT and 2D2-Kv1.3 KO Th cells were stimulated with MOG and irradiated APCs for 0 or 72 hours. Expression of pFoxO1/pFoxO3a, FoxO1, and β-Actin was examined in total cell lysates by western blotting. (c) WT and Kv1.3 KO Th cells were stimulated for 24, 48, or 72 hours. Expression of GATA1 and β-Actin were evaluated by western blotting. Numbers below bands indicate average densitometric ratio normalized to actin loading control.
Kv1.3 KO Th cells maintain a stable phenotype in a highly inflammatory environment and consistently express increased FoxO1 and GATA1 transcription factors
Next, we sought to assess whether the regulatory phenotype of Kv1.3 KO Th cells is maintained in the face of large amounts of pro-inflammatory cytokines as would occur during immune-mediated diseases such as MS. To this end, we polarized WT and Kv1.3 KO Th cells to Th0, Th1, Th2, Th17, and Treg subtypes and then examined expression of signature cytokines as well as pSTAT5, FoxO1, and GATA1 under these conditions. When polarized to Th1 or Th17, Kv1.3 KO Th cells produced significantly less IFN-γ and IL-17, respectively, than the WT Th cells (Figure 5a and b). Importantly, under all polarization conditions, Kv1.3 KO Th cells expressed significantly more pSTAT5, less pFoxO1, and more GATA1 than the WT controls (Figure 5c). Additionally, Kv1.3 KO or WT CD4+ T cells were stimulated for 72 hours, rested, and restimulated in the presence of polarizing cytokines. Kv1.3 KO T cells consistently expressed decreased IFN-γ and IL-17, and more IL-10 under Th1-polarizing conditions (Figure 5d). These data indicate that Kv1.3 KO Th cells maintain their regulatory phenotype even in the presence of strong polarizing cytokines, demonstrating phenotypic stability.
Figure 5. Kv1.3 KO Th cells maintain a stable phenotype in a highly inflammatory environment and consistently express increased FoxO1 and GATA1 transcription factors.
(a) WT and Kv1.3 KO Th cells were stimulated with anti-CD3/CD28-coated beads for 72h in the presence of 10 ng/ml IL-12 (Th1). Production of IFN-γ and IL-17 was quantified by FACS analysis. Plots are gated on CD4+ cells and IFNγ MFI, normalized to WT from three independent experiments, is shown. (b) WT and Kv1.3 KO Th cells were stimulated for 72 hours in the presence of anti-IL-4, anti-IFN-γ, TGF-β, IL-6, and IL-23 (Th17). Proportions of IFN-γ and IL-17 positive cells were quantified by FACS analysis. Plots are gated on CD4+ and IL-17 MFI, normalized to WT from three independent experiments, is shown. (c) WT and Kv1.3 KO Th cells were stimulated and polarized to Th0, Th1, Th2, Th17, and Treg for 72 hours. Expression of GATA1, pFoxO1/pFoxO3a, FoxO1, pSTAT5, and β-Actin were examined in total cell lysates by western blotting. Numbers below bands indicate average densitometric ratio normalized to actin loading control. (d) WT (white bars) and Kv1.3 KO (black bars) Th cells were stimulated for 72 hours in vitro, rested and restimulated in the presence of 10 ng/ml IL-12 (Th1) for 72 hours. Secretion of IFN-γ, IL-17, and IL-10 were evaluated by ELISA. Graphs represent data from 2 independent experiments and error bars depict SEM. *** p < 0.001.
Kv1.3 KO Th cells do not have an intrinsic defect in the ability to express Foxp3
Because the Kv1.3 KO Th cells have many properties common to Foxp3+ regulatory T cells, with the exception that they do not express Foxp3, it was important to determine whether Th cells can express Foxp3 in the absence of Kv1.3. To this end, we polarized Kv1.3 KO and WT Th cells to a Treg phenotype in vitro using TGF-β. Under polarizing conditions, Kv1.3 KO T cells can express equivalent amounts of Foxp3 as WT T cells, suggesting that Kv1.3 deficiency does not inhibit Foxp3 expression (Figure S2a and b). Instead, following TCR stimulation, Kv1.3 KO Th cells appear to develop regulatory capacity independently of Foxp3, supporting the hypothesis that this population of T cells constitutes a novel subset of T helper cells with suppressive properties.
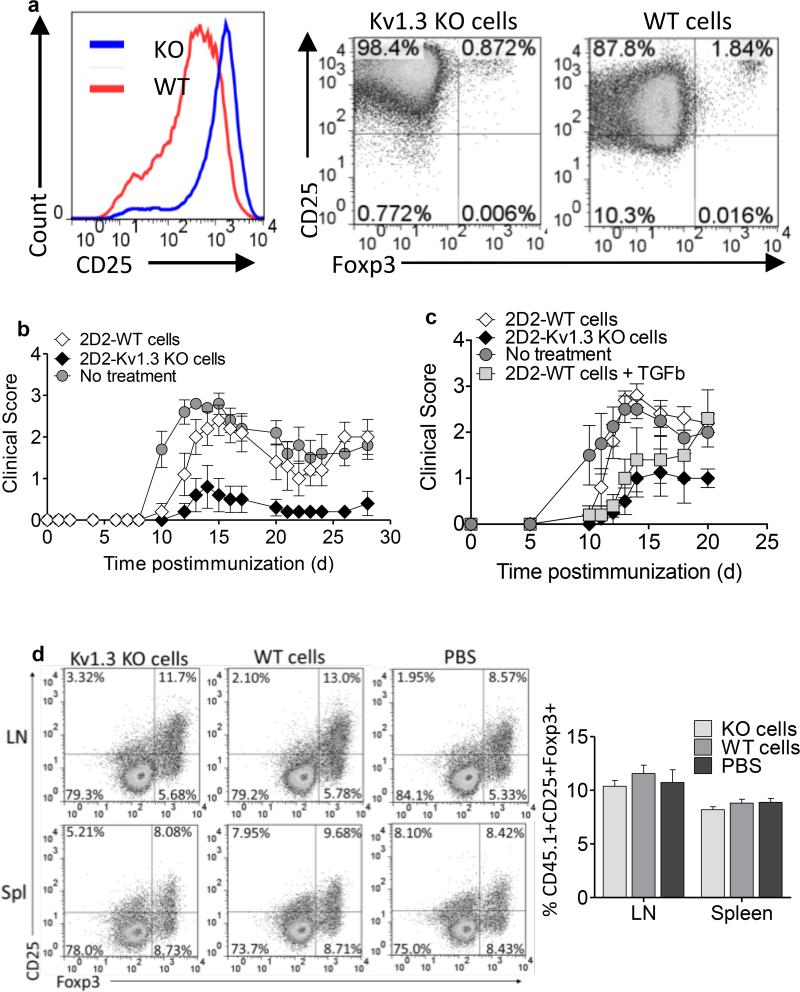
Adoptive transfer of activated 2D2-Kv1.3 KO Th cells ameliorates EAE but does not induce expansion of endogenous Foxp3+ Tregs
Foxp3+ peripherally-induced Tregs have been demonstrated to reduce the severity of EAE when transferred at the time of immunization (37). To determine if our antigen-specific Kv1.3 KO Th cells could also reduce the severity of EAE in a similar manner, we transferred 2D2-Kv1.3 KO and 2D2-WT Th cells, which were stimulated with MOG 35-55 peptide and irradiated WT APCs for 72 hours in vitro, into WT animals and then immunized the recipients to induce EAE. Prior to transfer, cells were subjected to ficoll density centrifugation to remove dead APCs resulting in greater than 90% pure CD4+ T cells. Examination of phenotype prior to transfer revealed that 2D2-Kv1.3 KO Th cells expressed significantly higher amounts of CD25 than 2D2-WT cells, but did not express Foxp3. 2D2-WT cells were also Foxp3 negative (Figure 6a). Remarkably, animals that received activated 2D2-Kv1.3 KO T cells at the time of immunization exhibited a significantly decreased incidence and severity of EAE compared to immunized animals receiving 2D2-WT T cells or no cells (Figure 6b). This decreased disease severity could not be attributed to impaired migration of transferred Kv1.3 KO Th cells or host CD4+ T cells to the CNS, as equal number of CD4+ T cells were detected in the blood, lymph nodes, and spleens of mice at both 7 and 14 days post-immunization (Figure S3a and b). In a parallel experiment 2D2-Kv1.3KO Th cells were compared with 2D2-WT Th cells polarized to a Foxp3+ Treg phenotype prior to transfer for their ability to suppress EAE. Kv1.3 KO Th cells suppressed disease equally to WT Tregs and, importantly, maintained their suppressive phenotype longer than the Foxp3+ Tregs in vivo (Figure 6c). To explore the mechanism by which transfer of 2D2-Kv1.3 KO T cells confers protection from EAE, we investigated whether transfer of these T cells induced expansion of endogenous Tregs, which would, in turn, protect the mice from EAE. For this purpose, 2D2-Kv1.3 KO and 2D2-WT CD4+ T cells were transferred into CD45.1 congenic recipients, which were immunized with MOG peptide to induce EAE. The percentage of CD45.1+, CD4+, CD25+, Foxp3+ cells was examined via flow cytometry at peak of disease. No differences were detected in the percentage of host Tregs from either the spleen or draining lymph nodes of mice receiving either WT or Kv1.3 KO T cells despite significantly decreased incidence of disease in animals receiving Kv1.3 KO cells (Figure 6d). These data have powerful implications for the ability of Foxp3-negative antigen-specific Kv1.3 KO Th cells to be protective in autoimmune disease via a mechanism that is not dependent on host regulatory T cells.
Figure 6. Adoptive transfer of activated 2D2-Kv1.3 KO Th cells ameliorates EAE but does not induce expansion of endogenous Foxp3+ Tregs.
2D2-WT and 2D2-Kv1.3 KO Th cells were stimulated with MOG 35-55 and irradiated WT APCs for 72 hours. Th cells were isolated by ficoll gradient. (a) CD25 and Foxp3 expression were evaluated by FACS analysis. (b) Ficolled 2D2-WT or 2D2-Kv1.3 KO Th cells were injected into WT recipients at the time of immunization with MOG 35-55. (c) Kv1.3 KO and 2D2-WT Th cells were stimulated with MOG 35-55 and irradiated WT APCs for 72hours with or without 5ng/ml TGF-β. Following isolation by ficoll gradient Th cells were transferred to WT C57BL/6 recipients at the time of immunization. Animals were monitored for EAE clinical scores. (d) 2D2-WT or 2D2-Kv1.3 KO Th cells were stimulated as described above and transferred into CD45.1 congenic recipients at time of immunization. CD45.1+ CD4+ cells from recipient lymph nodes and spleen were analyzed by FACS analysis at peak of disease for the expansion of endogenous CD25+Foxp3+ Tregs. Graph is representative of 5 animals per group and 2 independent experiments.
We also investigated the possibility that the transfer of Kv1.3 KO Th cells resulted in the induction of myleiod derived suppressor cells (MDSCs) in the receipent animals, thereby contributing to the decreased disease severity. However, while the percentage of CD11b+ cells was increased in the mice receiving WT T cells, likely due to an increased number of infiltrating monocytes in EAE animals, we saw no differences in the percentages of CD11b+Ly6G+ cells in the brains of mice receiving Kv1.3 KO Th cells at the peak of disease compared to mice receiving WT Th cells (Figure S3c). These data suggest that induction of MDSCs is not the likely mechanism of Kv1.3 KO Th cell mediated suppression.
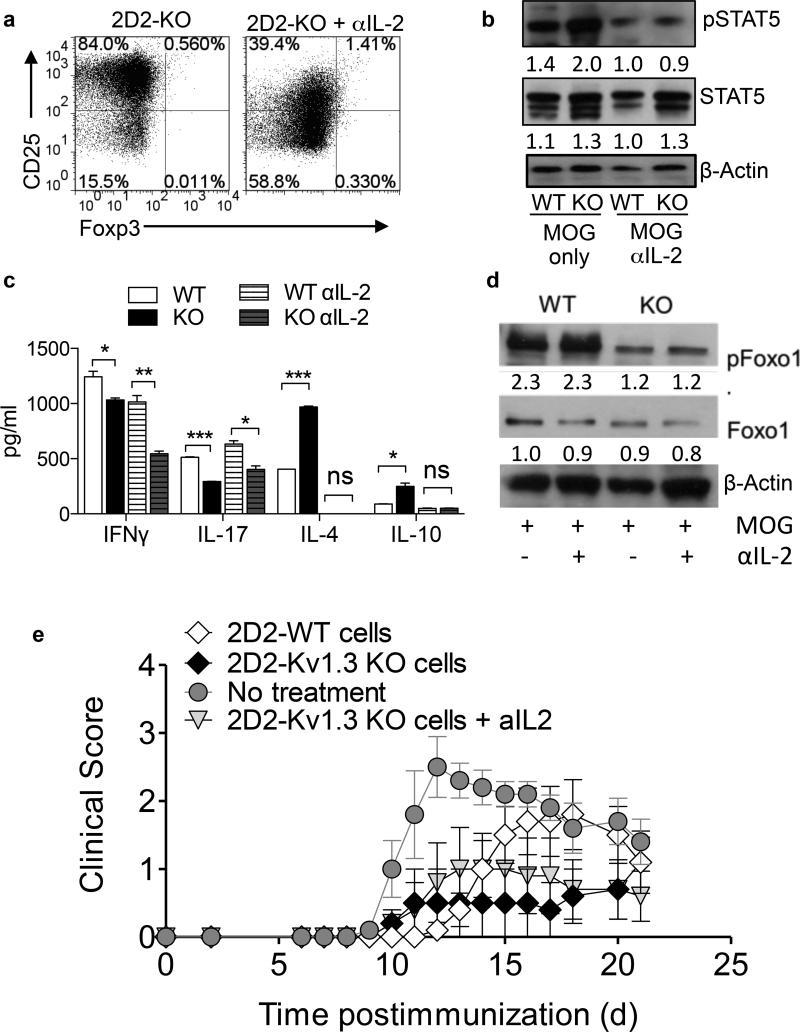
Neutralization of IL-2 results in a partial loss of the regulatory phenotype in Kv1.3 KO Th cells
To determine whether 2D2-Kv1.3 KO Th cells are dependent upon IL-2 for the development of their regulatory phenotype, we neutralized IL-2 using a blocking antibody in 2D2-WT and 2D2-Kv1.3 KO Th cell cultures during a 72-hour stimulation with MOG 35-55 and irradiated APCs and examined the effects on CD25, pSTAT5, and pFoxO1 expression as well as cytokine production and suppressive function. IL-2 neutralization was confirmed by ELISA (Figure S4a). We observed no increase in CD25 or pSTAT5 in Kv1.3 KO cells when IL-2 was blocked (Figure 7a and b) and no increase in IL-10 or IL-4 secretion (Figure 7c), suggesting that IL-2 signaling plays an important role in the development of the regulatory phenotype observed in the absence of Kv1.3. Accordingly, when IL-2 was neutralized, the ability of Kv1.3 KO Th cells to suppress the proliferation of WT effector T cells was impaired in an in vitro suppression assay. This suppressive quality was not affected when IL-10 or CTLA-4 were inhibited, supporting an important role for IL-2 in Kv1.3 KO Th cell mediated suppression (Figure S4b,c,d). Interestingly, 2D2-Kv1.3 KO T cells continued to secrete less IFN-γ and IL-17 than WT controls, which continued to secrete significant amounts of IFN-γ and IL-17 even when IL-2 was neutralized (Figure 7c), indicating that neutralization of IL-2 did not globally impair the function of the Th cells, but rather inhibited a specific signaling pathway. Importantly, neutralization of IL-2 also did not affect FoxO1 expression, suggesting that the effect of IL-2 neutralization is not broad but rather pathway-specific (Figure 7d).
Figure 7. Neutralization of IL-2 results in a partial loss of the regulatory phenotype in Kv1.3 KO Th cells.
2D2-WT and 2D2-Kv1.3 KO Th cells were stimulated with MOG 35-55 and irradiated APCs for 72 hours in the presence of 10 μg/ml anti-IL-2 antibody or isotype control. CD25, Foxp3 (a), pSTAT5 and STAT5 expression (b), cytokine production (c), and FoxO1 expression (d) were examined. Numbers below bands indicate average densitometric ratio normalized to actin loading control. (e) Cells described above were transferred to C57BL/6 WT recipients at the time of immunization for EAE and clinical score was evaluated.
Next, we sought to determine whether IL-2 signaling and resulting induction of pSTAT5 and CD25 are required for 2D2-Kv1.3 KO cells to ameliorate EAE. To this end, we neutralized IL-2 during the in vitro pre-activation of 2D2-Kv1.3 KO and 2D2-WT T cells and then transferred these cells into WT recipients a day prior to immunization. Re-activation in the absence of IL-2 only partially impaired 2D2-Kv1.3 KO T cells from being able to ameliorate EAE, indicating that IL-2R signaling during antigenic stimulation contributes to the induction of a regulatory phenotype in 2D2-Kv1.3 KO Th cells. (Figure 7e).
DISCUSSION
Impaired suppressive function of Tregs has been implicated in the immunopathogenesis of MS (3-5). In the present study, we delineate the mechanism by which a unique population of Foxp3 negative Th cells develops in the absence of Kv1.3 and characterize the regulatory effects of this cell population on T cell function and disease using the EAE model. Our data suggest that CD25hi, CTLA4hi, IL-10 producing Foxp3 negative T cells with suppressive properties are generated in the absence of Kv1.3 as a consequence of disrupted TCR/integrin signaling during formation of the immunological synapse rather than impaired voltage-gated potassium flux and corresponding changes in intracellular calcium signaling. This lack of change in intracellular calcium signaling is not completely unsurprising as Kv1.3 is deleted during development of the mouse, thereby resulting in compensation from chloride channels as published previously(31). Moreover, decreased activation and phosphorylation of proximal TCR signaling molecules, including Erk1/2 and p56lck, was observed, suggesting a role for impaired TCR signaling in the development of this cell population. Not surprisingly, because phosphorylation of these proximal TCR signaling molecules is responsible for nuclear exclusion of FoxO transcription factors, the observed alteration in TCR signaling correlated with increased nuclear localization of FoxO proteins in the absence of Kv1.3. In addition to increased nuclear FoxO1 expression, increased expression of GATA1, a transcription factor thought to contribute to the Treg signature but not extensively studied in lymphocytes, was observed. The phenotype of this novel cell population remained stable in the presence of strong inflammatory conditions. Importantly, we also demonstrate that these MOG-specific non-canonical regulatory T cells from Kv1.3 KO animals can ameliorate EAE, and that this suppressive capacity is partially dependent on endogenous IL-2 signaling. Overall, our data demonstrate the development of a novel population of T cells with regulatory capacity that arise in the absence of Kv1.3 and point to FoxO1 and GATA1 as novel targets for induction of antigen-specific regulatory T cells during immune-mediated disease.
To determine which transcription factors, in lieu of Foxp3, may be driving transcription of regulatory factors such as IL-10, CTLA-4 and CD25 in the absence of Kv1.3, we examined FoxO family members. Our previously published data indicate that phosphorylation of SMAD3 is increased in Kv1.3 KO Th cells stimulated in vitro (11), and our new data demonstrate that FoxO1 expression is also increased in response to in vitro antigenic stimulation. Synergy between SMAD and FoxO transcription factors has been shown to be important for the regulatory effects of TGF-β in immune cells. FoxO1 plays an important role in maintaining T cell homeostasis, induction of tolerance, and can dictate regulatory T cell lineage commitment (32). FoxO1 has also been implicated in both the repression of IFN-γ and IL-17/RORγt and in the transactivation of genes essential for regulatory T cell function, such as CTLA4 and Foxp3 itself (38). Our data demonstrate increased nuclear localization of FoxO1, which correlates directly with decreased phosphorylation of the protein in Th cells from Kv1.3 KO and 2D2-Kv1.3 KO mice following stimulation in vitro. We hypothesize that FoxO1 could directly, or via interaction with other transcription factors, such as GATA1, induce transcription of CD25 and/or IL-10. GATA1 was one of several transcription factors identified recently to contribute to the transcriptional signature of Foxp3+ Tregs (36). Because GATA1 expression was found to be significantly increased in Kv1.3 KO T cells, it is possible that it can compensate for the lack of Foxp3 expression in these cells. In future studies, it will be important to determine whether induction of FoxO1, GATA1, or a combination of these two transcription factors is sufficient to convey a protective phenotype to 2D2-WT T cells. These data highlight the potential for the development of novel drugs that specifically induce the expression of these transcription factors in immune-mediated disease.
Increased IL-2 signaling through the high affinity IL-2R contributes to the differentiation of regulatory T cells. Engagement of TCR and peptide/MHC complexes leads to the activation of a multitude of downstream signaling molecules including protein tyrosine kinases and adaptor molecules such as p56lck, Zap70, and SLP76. These downstream signaling events culminate in the production of cytokines such as IL-2 and IL-4. Binding of IL-2 to IL-2R, and to a lesser extent, interaction of IL-4 with its receptor, plays a critical role in sustaining T cell activation and, importantly, immune homeostasis. It has been shown that one of the major roles of IL-2 is to induce activation of STAT5 (39-42). STAT5 proteins have been demonstrated to be crucial for the differentiation of Tregs and Th2 cells and, indeed, deletion of STAT5 results in the development of T cells with a hyperactivated phenotype very similar to that of the autoimmune phenotype in IL-2Rβ deficient mice (40-42). Our data demonstrate increased expression of CD25 and increased phosphorylation of STAT5, which correlates with decreased phosphorylation of p56lck and Erk1/2. Kv1.3 has been demonstrated to play an important role in formation of the immunological synapse and p56lck is known to interact directly with the hDlg adaptor protein of Kv1.3, promoting signaling through the TCR during T cell activation (43-47). Disruption of this interaction, as would occur in the absence of Kv1.3, could result in the relocalization of p56lck to the IL-2R complex, thereby preferentially promoting signaling through the IL-2R as opposed to the TCR and contributing to the differentiation of T cells with regulatory properties. As Erk has been demonstrated to phosphorylate FoxO1 and exclude it from the nucleus where it may be targeted for degradation (34), it is possible that the reduction in Erk1/2 phosphorylation that is observed in Kv1.3 KO Th cells is directly responsible for regulation of FoxO1 in the absence of Kv1.3.
Importantly, we demonstrate that activated myelin specific Kv1.3 KO Th cells, which maintain suppressive properties in the presence of strong inflammatory conditions, can suppress the development of EAE when transferred to WT animals at the time of immunization. This suppression is not dependent upon the expansion of endogenous Foxp3+ Tregs in the host, is maintained even when Foxp3+ Treg suppression wanes, and is at least partially dependent upon IL-2R signaling. The exact mechanisms by which these cells suppress EAE in vivo, including the requirement for IL-10, need to be addressed in future experiments. However, these data have strong implications for this unique population of T cells as suppressors of the aberrant immune response in MS. Recently, a population of regulatory cells named FoxA1+ T cells was identified in MS patients that demonstrated responsiveness to IFN-β therapy (7). This population of cells is also Foxp3 negative, but, unlike the population identified in our studies, does not secrete increased amounts of IL-10. The identification of such a novel population of Tregs in the context of CNS autoimmunity supports the possiblilty that yet to be discovered populations of T cells with regulatory properties, such as those described herein, exist, and could arise naturally in response to disease specific triggers. Determining whether a natural correlate of Kv1.3 KO Tregs exists in humans will be the focus of future studies.
Overall, our data indicate that deletion of Kv1.3 results in the development of a unique population of Th cells via increased STAT5 phosphorylation, IL-2R signaling, and expression of FoxO1 and GATA1 transcription factors. Importantly, this population of antigen-specific T cells with suppressive properties arises without increased expression of Foxp3, highlighting the contribution of the aforementioned transcription factors to the signature of these cells. In addition, these results establish an in vivo model system in which to study the role of specific molecules and transcription factors, such as FoxO1, GATA1, STAT5, and IL-2, in the differentiation of this previously undescribed population of T cells. This study increases our understanding of how a novel subset of T cells with suppressive properties, which is not dependent on Foxp3, arises in the absence of Kv1.3. This mechanistic insight sheds light on how effector T cells may be manipulated in vivo to suppress unwanted inflammation in immune-mediated diseases in an antigen-specific manner and may pave the way for developing novel effective therapies targeted at disease-specific cells.
Supplementary Material
Acknowledgments
Supported by the US National Institutes of Health (R01 NS041435 to P.A.C.) and the Silverman Foundation.
REFERENCES
- 1.Wulff H, Calabresi PA, Allie R, Yun S, Pennington M, Beeton C, Chandy KG. The voltage-gated Kv1.3 K(+) channel in effector memory T cells as new target for MS. The Journal of clinical investigation. 2003;111:1703–1713. doi: 10.1172/JCI16921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lovett-Racke AE, Trotter JL, Lauber J, Perrin PJ, June CH, Racke MK. Decreased dependence of myelin basic protein-reactive T cells on CD28-mediated costimulation in multiple sclerosis patients. A marker of activated/memory T cells. The Journal of clinical investigation. 1998;101:725–730. doi: 10.1172/JCI1528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Korn T, Reddy J, Gao W, Bettelli E, Awasthi A, Petersen TR, Backstrom BT, Sobel RA, Wucherpfennig KW, Strom TB, Oukka M, Kuchroo VK. Myelin-specific regulatory T cells accumulate in the CNS but fail to control autoimmune inflammation. Nature medicine. 2007;13:423–431. doi: 10.1038/nm1564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Lowther DE, Hafler DA. Regulatory T cells in the central nervous system. Immunological reviews. 2012;248:156–169. doi: 10.1111/j.1600-065X.2012.01130.x. [DOI] [PubMed] [Google Scholar]
- 5.Viglietta V, Baecher-Allan C, Weiner HL, Hafler DA. Loss of functional suppression by CD4+CD25+ regulatory T cells in patients with multiple sclerosis. The Journal of experimental medicine. 2004;199:971–979. doi: 10.1084/jem.20031579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Josefowicz SZ, Lu LF, Rudensky AY. Regulatory T cells: mechanisms of differentiation and function. Annual review of immunology. 2012;30:531–564. doi: 10.1146/annurev.immunol.25.022106.141623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Liu Y, Carlsson R, Comabella M, Wang J, Kosicki M, Carrion B, Hasan M, Wu X, Montalban X, Dziegiel MH, Sellebjerg F, Sorensen PS, Helin K, Issazadeh-Navikas S. FoxA1 directs the lineage and immunosuppressive properties of a novel regulatory T cell population in EAE and MS. Nature medicine. 2014;20:272–282. doi: 10.1038/nm.3485. [DOI] [PubMed] [Google Scholar]
- 8.Miller SD, Turley DM, Podojil JR. Antigen-specific tolerance strategies for the prevention and treatment of autoimmune disease. Nature reviews. Immunology. 2007;7:665–677. doi: 10.1038/nri2153. [DOI] [PubMed] [Google Scholar]
- 9.Yong VW, Marks S. The interplay between the immune and central nervous systems in neuronal injury. Neurology. 2010;74(Suppl 1):S9–S16. doi: 10.1212/WNL.0b013e3181c97d04. [DOI] [PubMed] [Google Scholar]
- 10.Hu L, Pennington M, Jiang Q, Whartenby KA, Calabresi PA. Characterization of the functional properties of the voltage-gated potassium channel Kv1.3 in human CD4+ T lymphocytes. Journal of immunology. 2007;179:4563–4570. doi: 10.4049/jimmunol.179.7.4563. [DOI] [PubMed] [Google Scholar]
- 11.Hu L, Gocke AR, Knapp E, Rosenzweig JM, Grishkan IV, Baxi EG, Zhang H, Margolick JB, Whartenby KA, Calabresi PA. Functional blockade of the voltage-gated potassium channel Kv1.3 mediates reversion of T effector to central memory lymphocytes through SMAD3/p21cip1 signaling. The Journal of biological chemistry. 2012;287:1261–1268. doi: 10.1074/jbc.M111.296798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Cahalan MD, Chandy KG. The functional network of ion channels in T lymphocytes. Immunological reviews. 2009;231:59–87. doi: 10.1111/j.1600-065X.2009.00816.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Beeton C, Pennington MW, Wulff H, Singh S, Nugent D, Crossley G, Khaytin I, Calabresi PA, Chen CY, Gutman GA, Chandy KG. Targeting effector memory T cells with a selective peptide inhibitor of Kv1.3 channels for therapy of autoimmune diseases. Molecular pharmacology. 2005;67:1369–1381. doi: 10.1124/mol.104.008193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Beeton C, Wulff H, Standifer NE, Azam P, Mullen KM, Pennington MW, Kolski-Andreaco A, Wei E, Grino A, Counts DR, Wang PH, LeeHealey CJ, Sankaranarayanan SAB,A, Homerick D, Roeck WW, Tehranzadeh J, Stanhope KL, Zimin P, Havel PJ, Griffey S, Knaus HG, Nepom GT, Gutman GA, Calabresi PA, Chandy KG. Kv1.3 channels are a therapeutic target for T cell-mediated autoimmune diseases. Proceedings of the National Academy of Sciences of the United States of America. 2006;103:17414–17419. doi: 10.1073/pnas.0605136103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wulff H, Castle NA, Pardo LA. Voltage-gated potassium channels as therapeutic targets. Nature reviews. Drug discovery. 2009;8:982–1001. doi: 10.1038/nrd2983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Nicolaou SA, Neumeier L, Steckly A, Kucher V, Takimoto K, Conforti L. Localization of Kv1.3 channels in the immunological synapse modulates the calcium response to antigen stimulation in T lymphocytes. Journal of immunology. 2009;183:6296–6302. doi: 10.4049/jimmunol.0900613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gocke AR, Lebson LA, Grishkan IV, Hu L, Nguyen HM, Whartenby KA, Chandy KG, Calabresi PA. Kv1.3 deletion biases T cells toward an immunoregulatory phenotype and renders mice resistant to autoimmune encephalomyelitis. Journal of immunology. 2012;188:5877–5886. doi: 10.4049/jimmunol.1103095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bettelli E, Pagany M, Weiner HL, Linington C, Sobel RA, Kuchroo VK. Myelin oligodendrocyte glycoprotein-specific T cell receptor transgenic mice develop spontaneous autoimmune optic neuritis. The Journal of experimental medicine. 2003;197:1073–1081. doi: 10.1084/jem.20021603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Yang Y, Weiner J, Liu Y, Smith AJ, Huss DJ, Winger R, Peng H, Cravens PD, Racke MK, Lovett-Racke AE. T-bet is essential for encephalitogenicity of both Th1 and Th17 cells. The Journal of experimental medicine. 2009;206:1549–1564. doi: 10.1084/jem.20082584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Bettelli E, Korn T, Oukka M, Kuchroo VK. Induction and effector functions of T(H)17 cells. Nature. 2008;453:1051–1057. doi: 10.1038/nature07036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Bettelli E, Kuchroo VK. IL-12- and IL-23-induced T helper cell subsets: birds of the same feather flock together. The Journal of experimental medicine. 2005;201:169–171. doi: 10.1084/jem.20042279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Dardalhon V, Korn T, Kuchroo VK, Anderson AC. Role of Th1 and Th17 cells in organ-specific autoimmunity. Journal of autoimmunity. 2008;31:252–256. doi: 10.1016/j.jaut.2008.04.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Gocke AR, Cravens PD, Ben LH, Hussain RZ, Northrop SC, Racke MK, Lovett-Racke AE. T-bet regulates the fate of Th1 and Th17 lymphocytes in autoimmunity. Journal of immunology. 2007;178:1341–1348. doi: 10.4049/jimmunol.178.3.1341. [DOI] [PubMed] [Google Scholar]
- 24.Lovett-Racke AE, Rocchini AE, Choy J, Northrop SC, Hussain RZ, Ratts RB, Sikder D, Racke MK. Silencing T-bet defines a critical role in the differentiation of autoreactive T lymphocytes. Immunity. 2004;21:719–731. doi: 10.1016/j.immuni.2004.09.010. [DOI] [PubMed] [Google Scholar]
- 25.Racke MK, Bonomo A, Scott DE, Cannella B, Levine A, Raine CS, Shevach EM, Rocken M. Cytokine-induced immune deviation as a therapy for inflammatory autoimmune disease. The Journal of experimental medicine. 1994;180:1961–1966. doi: 10.1084/jem.180.5.1961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Young DA, Lowe LD, Booth SS, Whitters MJ, Nicholson L, Kuchroo VK, Collins M. IL-4, IL-10, IL-13, and TGF-beta from an altered peptide ligand-specific Th2 cell clone down-regulate adoptive transfer of experimental autoimmune encephalomyelitis. Journal of immunology. 2000;164:3563–3572. doi: 10.4049/jimmunol.164.7.3563. [DOI] [PubMed] [Google Scholar]
- 27.Anderson AC, Reddy J, Nazareno R, Sobel RA, Nicholson LB, Kuchroo VK. IL-10 plays an important role in the homeostatic regulation of the autoreactive repertoire in naive mice. Journal of immunology. 2004;173:828–834. doi: 10.4049/jimmunol.173.2.828. [DOI] [PubMed] [Google Scholar]
- 28.Bettelli E, Das MP, Howard ED, Weiner HL, Sobel RA, Kuchroo VK. IL-10 is critical in the regulation of autoimmune encephalomyelitis as demonstrated by studies of IL-10- and IL-4-deficient and transgenic mice. Journal of immunology. 1998;161:3299–3306. [PubMed] [Google Scholar]
- 29.Chen Y, Kuchroo VK, Inobe J, Hafler DA, Weiner HL. Regulatory T cell clones induced by oral tolerance: suppression of autoimmune encephalomyelitis. Science. 1994;265:1237–1240. doi: 10.1126/science.7520605. [DOI] [PubMed] [Google Scholar]
- 30.Mills KH, McGuirk P. Antigen-specific regulatory T cells--their induction and role in infection. Seminars in immunology. 2004;16:107–117. doi: 10.1016/j.smim.2003.12.006. [DOI] [PubMed] [Google Scholar]
- 31.Koni PA, Khanna R, Chang MC, Tang MD, Kaczmarek LK, Schlichter LC, Flavella RA. Compensatory anion currents in Kv1.3 channel-deficient thymocytes. The Journal of biological chemistry. 2003;278:39443–39451. doi: 10.1074/jbc.M304879200. [DOI] [PubMed] [Google Scholar]
- 32.Ouyang W, Beckett O, Flavell RA, Li MO. An essential role of the Forkhead-box transcription factor Foxo1 in control of T cell homeostasis and tolerance. Immunity. 2009;30:358–371. doi: 10.1016/j.immuni.2009.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Hedrick SM, Hess Michelini R, Doedens AL, Goldrath AW, Stone EL. FOXO transcription factors throughout T cell biology. Nature reviews. Immunology. 2012;12:649–661. doi: 10.1038/nri3278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Zhao Y, Wang Y, Zhu WG. Applications of post-translational modifications of FoxO family proteins in biological functions. Journal of molecular cell biology. 2011;3:276–282. doi: 10.1093/jmcb/mjr013. [DOI] [PubMed] [Google Scholar]
- 35.Ouyang W, Liao W, Luo CT, Yin N, Huse M, Kim MV, Peng M, Chan P, Ma Q, Mo Y, Meijer D, Zhao K, Rudensky AY, Atwal G, Zhang MQ, Li MO. Novel Foxo1-dependent transcriptional programs control T(reg) cell function. Nature. 2012;491:554–559. doi: 10.1038/nature11581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Fu W, Ergun A, Lu T, Hill JA, Haxhinasto S, Fassett MS, Gazit R, Adoro S, Glimcher L, Chan S, Kastner P, Rossi D, Collins JJ, Mathis D, Benoist C. A multiply redundant genetic switch ‘locks in’ the transcriptional signature of regulatory T cells. Nature immunology. 2012;13:972–980. doi: 10.1038/ni.2420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Stephens LA, Malpass KH, Anderton SM. Curing CNS autoimmune disease with myelin-reactive Foxp3+ Treg. European journal of immunology. 2009;39:1108–1117. doi: 10.1002/eji.200839073. [DOI] [PubMed] [Google Scholar]
- 38.Hedrick SM. Positive selection in the thymus: an enigma wrapped in a mystery. Journal of immunology. 2012;188:2043–2045. doi: 10.4049/jimmunol.1200077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Paul WE, Zhu J. How are T(H)2-type immune responses initiated and amplified? Nature reviews. Immunology. 2010;10:225–235. doi: 10.1038/nri2735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Moriggl R, Sexl V, Piekorz R, Topham D, Ihle JN. Stat5 activation is uniquely associated with cytokine signaling in peripheral T cells. Immunity. 1999;11:225–230. doi: 10.1016/s1074-7613(00)80097-7. [DOI] [PubMed] [Google Scholar]
- 41.Moriggl R, Topham DJ, Teglund S, Sexl V, McKay C, Wang D, Hoffmeyer A, van Deursen J, Sangster MY, Bunting KD, Grosveld GC, Ihle JN. Stat5 is required for IL-2-induced cell cycle progression of peripheral T cells. Immunity. 1999;10:249–259. doi: 10.1016/s1074-7613(00)80025-4. [DOI] [PubMed] [Google Scholar]
- 42.Suzuki H, Kundig TM, Furlonger C, Wakeham A, Timms E, Matsuyama T, Schmits R, Simard JJ, Ohashi PS, Griesser H, et al. Deregulated T cell activation and autoimmunity in mice lacking interleukin-2 receptor beta. Science. 1995;268:1472–1476. doi: 10.1126/science.7770771. [DOI] [PubMed] [Google Scholar]
- 43.Toth A, Szilagyi O, Krasznai Z, Panyi G, Hajdu P. Functional consequences of Kv1.3 ion channel rearrangement into the immunological synapse. Immunology letters. 2009;125:15–21. doi: 10.1016/j.imlet.2009.05.004. [DOI] [PubMed] [Google Scholar]
- 44.Panyi G, Bagdany M, Bodnar A, Vamosi G, Szentesi G, Jenei A, Matyus L, Varga S, Waldmann TA, Gaspar R, Damjanovich S. Colocalization and nonrandom distribution of Kv1.3 potassium channels and CD3 molecules in the plasma membrane of human T lymphocytes. Proceedings of the National Academy of Sciences of the United States of America. 2003;100:2592–2597. doi: 10.1073/pnas.0438057100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Panyi G, Vamosi G, Bacso Z, Bagdany M, Bodnar A, Varga Z, Gaspar R, Matyus L, Damjanovich S. Kv1.3 potassium channels are localized in the immunological synapse formed between cytotoxic and target cells. Proceedings of the National Academy of Sciences of the United States of America. 2004;101:1285–1290. doi: 10.1073/pnas.0307421100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Nicolaou SA, Szigligeti P, Neumeier L, Lee SM, Duncan HJ, Kant SK, Mongey AB, Filipovich AH, Conforti L. Altered dynamics of Kv1.3 channel compartmentalization in the immunological synapse in systemic lupus erythematosus. Journal of immunology. 2007;179:346–356. doi: 10.4049/jimmunol.179.1.346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Reneer MC, Estes DJ, Velez-Ortega AC, Norris A, Mayer M, Marti F. Peripherally induced human regulatory T cells uncouple Kv1.3 activation from TCR-associated signaling. European journal of immunology. 2011;41:3170–3175. doi: 10.1002/eji.201141492. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.