Abstract
Background
Chronic Obstructive Pulmonary Disease (COPD) moved from fourth to third commonest cause of death in the world between 1990 and 2010.
Methods
Using data from the Global Burden of Disease programme we quantified regional changes in number of COPD deaths and COPD mortality rates between 1990 and 2010. We estimated the proportion of the change attributable to gross national income (GNI)/capita and an index of cumulative smoking exposure, and quantified the difference in mortality rates attributable to demographic changes.
Findings
Despite a substantial decrease in COPD mortality rates COPD deaths fell only slightly, from 3 million in 1990 to 2.8 million in 2010 because the mean age of the population increased. The number of COPD deaths in 2010 would have risen to 5.2 million if the age-sex specific mortality rates had remained constant. Changes in smoking led to only a small increase in age-sex specific mortality rates, which were strongly associated with changes in GNI.
Interpretation
The increased burden of COPD mortality was mainly driven by changes in age distribution, but age-sex specific rates fell as incomes rose. The rapid response to increasing affluence suggests that changes in COPD mortality are not explained entirely by changes in early life.
Keywords: COPD, Poverty, Mortality, Smoking
INTRODUCTION
Global patterns of mortality are changing, mainly due to three related transformations: an epidemiological transition driven by change in global risks, with falling death rates in childhood associated with infectious disease, a demographic transition with more people surviving into later life, and a health care transition with improved effectiveness of both preventive and curative or palliative treatments. The reassessment of the Global Burden of Disease (GBD) in 2010 identified Chronic Obstructive Pulmonary Disease (COPD) as the third most common cause of death in the world,(1) up from fourth in 1990.(2) Understanding the reasons for this is important for identifying potential public health interventions. In particular, we need to know whether this change has been caused by an increase in the mortality rate or by an increase in the absolute number of deaths due to increased prevalence, for example associated with aging in the population. Alternatively, the change may be a relative one, with COPD mortality having declined less than mortality from other causes.
Cigarette smoking is regarded as the most important cause of COPD, with other inhaled hazards from occupational, domestic or outdoor sources thought to play an important role.(3) However, in a study of mortality in local authorities in England and Wales, Barker et al. noted that there was a strong association between childhood deaths from pneumonia and bronchitis in the 1920s and deaths from “COPD” in the 1970s, with high death rates from both conditions being strongly associated with poverty.(4) As this association was not noted for lung cancer, this relation was unlikely to be strongly confounded by cigarette smoking. Subsequently, by following up a cohort of men born in the 1920s, with very complete health records from the first year of life, they showed associations between early life factors and both death from COPD and adult lung function, associations that were, again, not explained by smoking habits.(5) More recently the BOLD study has shown a strong ecological association between national levels of poverty and both the prevalence of a low FVC and national COPD mortality rates.(6)
Many descriptions of national and regional mortality trends over time have been published over the years. (7-14) These have reported variable trends, mostly at a local level, and have rarely analysed the drivers of change. In this analysis we have examined trends in COPD mortality rates for all 21 GBD regions of the world. We have assessed the impact of population changes on the numbers of COPD deaths, and the associations between changes in age-sex specific mortality rates from COPD and changes in both national income and smoking. By analysing trends within regions across time, we have avoided much of the confounding between national income and other factors varying across regions that is inevitable in purely cross-sectional analyses.
METHODS
In the decade 1990-1999, data on mortality were collected by country and year in countries allocated to 21 GBD regions, from 566 vital registration sources, 27 surveillance reports and 1 verbal autopsy report. In the decade 2000-2010, mortality data were collected, by country and year in the same regions, from 894 vital registration sources, 24 surveillance reports and 8 verbal autopsy reports. Our definition for COPD in ICD-9 was 490-492, 494 and 496, and J40-J44, J47 in ICD-10. We used algorithms previously developed (15)to improve the quality of the information on mortality by redistributing vague and unsatisfactory causes of death (“garbage codes”) to more likely causes of death using similar methods in all countries and all years. The selection of the model used to ascribe deaths to specific causes was based on its out-of-sample predictive validity.(15) Our model of COPD mortality ascribes each death to a single cause.
For each region and for each age-sex specific group we analysed COPD mortality rates for 1990 and 2010, limiting the analyses to people aged 40 and over (18 groups per region, 9 for age and 2 for sex). For the same age-sex groups, we used information on regional mortality rates from lung cancer to create a proxy for smoking exposure, based on a modification of the method proposed by Peto.(16) This uses the strong association between smoking and lung cancer mortality to estimate the cumulative life time exposure to tobacco. Briefly, a common baseline mortality rate from lung cancer was assumed in the absence of smoking and estimated from the age-sex specific lung cancer mortality rates among non-smokers in the American Cancer Society’s Cancer Prevention Study II. Subtracting these figures from the age-sex specific lung cancer mortality rates recorded in the GBD data, we derived an index of cumulative smoking exposure for each group. This index, referred to as the smoking index, is expressed as a rate per 100 000. Gross National Income per capita (GNI) was estimated in United States dollars and adjusted for purchasing power parity using data from the World Bank.(17) The average GNI for each country in the region was weighted by the total population of each country to provide a regional value for 1990 and 2010. To compare COPD mortality rates across regions, we estimated age-standardised mortality rates by applying the regional age-specific rates to a standard world population covering the same age span (≥40 years). For each region, the number of deaths that would have been expected in 2010 if the population had been the same as in 1990 was calculated, by taking the age-sex specific COPD mortality rates for 2010 and applying them to the age and sex population distribution of 1990. The difference between this figure and the actual number of deaths in 2010 gave the change in deaths due to changes in the age-sex structure and size of the population.
To analyse the effects of GNI and smoking on COPD mortality rates in 1990 and 2010, and their impact on the change in COPD mortality between the two years, we used multilevel regression analysis accounting for the clustering of the data within regions. We first analysed the COPD mortality rate against GNI and smoking index, separately for 1990 and 2010, adjusting for age group and sex, with log transformation of the mortality rate, GNI and smoking index. In order to log transform the smoking index, we added a small constant (180 per 100 000) to ensure that all values were positive. We then analysed the change in COPD mortality rates using a multilevel linear regression model, with the log of the 2010/1990 mortality rate ratio as the outcome, with the corresponding 2010/1990 log ratios for GNI and smoking index as linear predictors, and adjusting for age group and sex as categorical predictors with additive effects. As a sensitivity analysis, we reran the analysis after dropping the regions where the change in GNI had been highest, namely East Asia, South-East Asia, Andean-Latin America, and Southern Latin-America.
We estimated the population attributable fraction of the change in COPD mortality for GNI and smoking, i.e. the percentage of the change in COPD mortality between 1990 and 2010 that was attributable to changes in GNI and smoking, using the program margins in Stata.(1) All analyses were undertaken in Stata 12.2. (StataCorp. 2011. Stata Statistical Software: Release 12. College Station, TX: StataCorp LP)
RESULTS
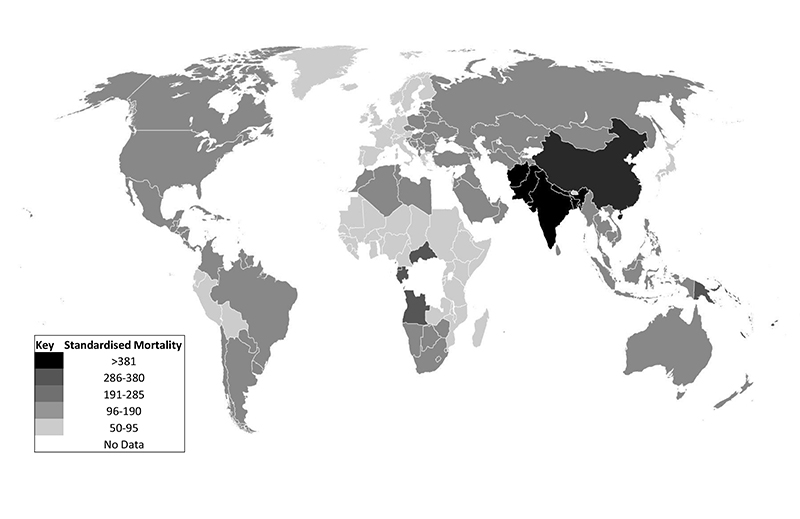
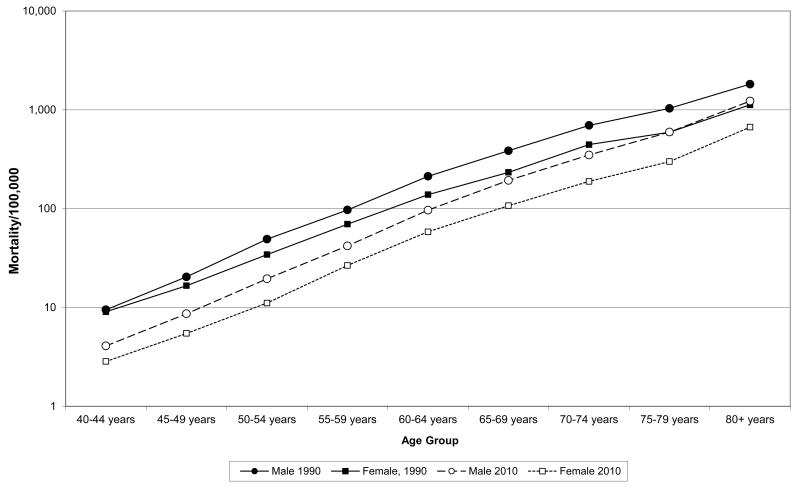
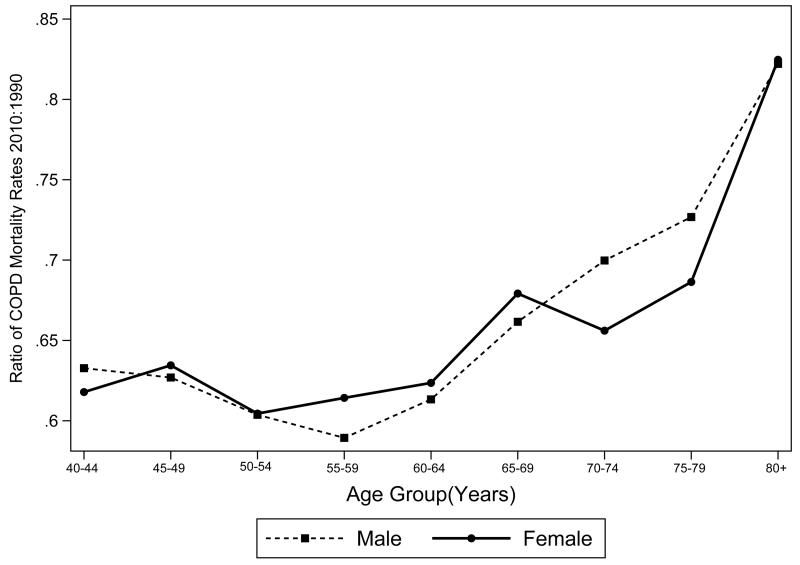
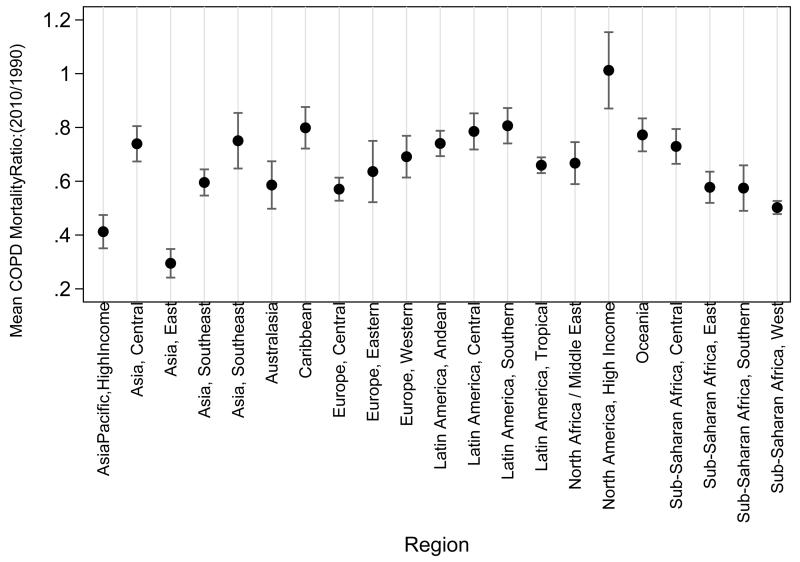
Most deaths from COPD occur in low income areas of the world including East, South and South-East Asia and Central Africa where age-standardised rates are also higher. (Figure 1) Globally the total number of COPD deaths fell from 3 million in 1990 to 2.8 million in 2010, a fall of 5·5%, whereas the expected number if age-sex specific rates had been maintained at 1990 levels was very much higher at 5.2 million (Table 1). There was a clear fall in age-sex specific rates for all groups between 1990 and 2010 (Figure 2a), and COPD mortality fell in all age groups, with the greatest relative falls in younger people (Figure 2b). There was substantial variation in the 2010/1990 ratio of mortality rates but clear reductions in all regions except the North America High Income region where the mortality rate remained stable (Figure 3).
FIGURE 1. Age-Sex adjusted COPD Mortality/100,000 : 2010.
TABLE 1.
Number of COPD deaths, total population, age-standardised COPD mortality rate by region and year. “Expected” COPD deaths are calculated as the number of deaths we would have expected in 2010 if the population had been the same, in terms of age and sex, as in 1990
| Region | 1990 | 2010 | |||||
|---|---|---|---|---|---|---|---|
| COPD deaths | Population | Age-standardised COPD mortality/100,000 | COPD deaths | Population | Age-standardised COPD mortality/100,000 | “Expected ”COPD deaths in 2010 | |
| Asia Pacific, High Income | 30,075 | 69,100,000 | 91 | 45,041 | 95,700,000 | 55 | 72,297 |
| Asia, Central | 17,210 | 16,000,000 | 249 | 15,934 | 23,600,000 | 166 | 23,696 |
| Asia, East | 1,430,132 | 314,000,000 | 1044 | 967,505 | 563,000,000 | 419 | 2,500,856 |
| Asia, South | 874,239 | 233,000,000 | 980 | 1,009,473 | 418,000,000 | 639 | 1,591,997 |
| Asia, Southeast | 77,567 | 95,800,000 | 213 | 121,999 | 183,000,000 | 185 | 148,331 |
| Australasia | 7,357 | 7,660,514 | 175 | 8,773 | 12,000,000 | 104 | 13,669 |
| Caribbean | 4,178 | 7,459,902 | 110 | 6,525 | 12,500,000 | 105 | 6,873 |
| Europe, Central | 43,513 | 47,600,000 | 180 | 38,076 | 57,000,000 | 110 | 61,533 |
| Europe, Eastern | 97,358 | 88,300,000 | 246 | 60,063 | 98,700,000 | 125 | 121,998 |
| Europe, Western | 152,394 | 168,000,000 | 142 | 169,312 | 218,000,000 | 98 | 229,961 |
| Latin America, Andean | 2,974 | 7,878,512 | 99 | 5,294 | 14,400,000 | 82 | 6,358 |
| Latin America, Central | 24,604 | 32,500,000 | 180 | 47,789 | 65,500,000 | 165 | 47,131 |
| Latin America, Southern | 10,340 | 15,400,000 | 138 | 15,756 | 22,400,000 | 121 | 17,562 |
| Latin America, Tropical | 34,090 | 34,500,000 | 256 | 54,320 | 65,800,000 | 184 | 75,753 |
| North Africa / Middle East | 29,997 | 56,100,000 | 137 | 46,593 | 114,000,000 | 108 | 60,998 |
| North America, High Income | 105,704 | 106,000,000 | 163 | 167,299 | 158,000,000 | 159 | 157,969 |
| Oceania | 1,067 | 1,010,723 | 324 | 1,483 | 1,881,734 | 248 | 2,004 |
| Sub-Saharan Africa, Central | 7,592 | 9,093,840 | 237 | 9,992 | 15,300,000 | 192 | 12,900 |
| Sub-Saharan Africa, East | 18,259 | 34,800,000 | 143 | 21,014 | 60,400,000 | 94 | 33,310 |
| Sub-Saharan Africa, Southern | 8,170 | 9,741,673 | 251 | 9,625 | 16,500,000 | 167 | 15,178 |
| Sub-Saharan Africa, West | 18,238 | 36,000,000 | 144 | 16,011 | 59,000,000 | 75 | 31,543 |
| Total | 2,995,058 | 1,390,000,000 | 544 | 2,837,877 | 2,280,000,000 | 291 | 5,231,916 |
FIGURE 2.
COPD mortality rates and 2010/1990 rate ratios, by sex and age group
(a) COPD mortality rates for 1990 and 2010 (b) 2010/1990 COPD mortality rate ratios
FIGURE 3.
2010/1990 ratio for COPD age-sex specific mortality rate, for each of the 21 regions
Changes in smoking index varied widely between regions, from a reduction of 15·3% in South Sub-Saharan Africa to an increase of 14·5% in East Asia (Table 2). GNI increased everywhere over this period of time, except in West Sub-Saharan Africa. In most places this increase was substantial and in East Asia it was over 700%. Plotting age-standardised COPD mortality rates by GNI showed a negative association. Because this appeared non-linear we transformed GNI to a log scale.
TABLE 2.
GNI per capita and smoking index by region and year, with % change in GNI and smoking index between 2010 and 1990. The % change was calculated as: (value2010- value1990/ value1990)*100
| Region | GNI Per Capita($) | Smoking Index | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Overall | Male | Female | ||||||||||
| 1990 | 2010 | %change | 1990 | 2010 | % Change | 1990 | 2010 | % Change | 1990 | 2010 | % Change | |
| Asia Pacific, High Income | 16,237 | 33,840 | 108·4 | 279·0 | 277·7 | −0·5 | 342·1 | 339·3 | −0·8 | 215·9 | 216·0 | 0·1 |
| Asia, Central | 2,444 | 3,329 | 36·2 | 251·6 | 223·2 | −11·3 | 311·0 | 261·6 | −15·9 | 192·1 | 184·9 | −3·8 |
| Asia, East | 884 | 7,733 | 774·7 | 279·1 | 319·7 | 14·5 | 316·7 | 382·1 | 20·7 | 241·6 | 257·2 | 6·5 |
| Asia, South | 864 | 3,084 | 256·9 | 187·2 | 195·1 | 4·2 | 197·7 | 208·1 | 5·3 | 176·7 | 182·2 | 3·1 |
| Asia, Southeast | 1,689 | 5,042 | 198·6 | 230·3 | 247·0 | 7·2 | 250·3 | 277·7 | 10·9 | 210·3 | 216·2 | 2·8 |
| Australasia | 16,259 | 35,200 | 116·5 | 305·2 | 266·2 | −12·8 | 379·2 | 300·4 | −20·8 | 231·3 | 232·0 | 0·3 |
| Caribbean | 3,595 | 6,692 | 86·1 | 265·5 | 259·5 | −2·2 | 307·0 | 293·9 | −4·3 | 223·9 | 225·1 | 0·5 |
| Europe, Central | 6,173 | 15,569 | 152·2 | 308·0 | 294·9 | −4·2 | 406·1 | 376·2 | −7·4 | 209·9 | 213·7 | 1·8 |
| Europe, Eastern | 7,466 | 15,907 | 113·1 | 316·9 | 275·2 | −13·2 | 436·3 | 360·3 | −17·4 | 197·5 | 190·0 | −3·8 |
| Europe, Western | 15,216 | 35,460 | 133·0 | 313·8 | 278·3 | −11·3 | 41·2 | 338·4 | −18·5 | 212·5 | 218·1 | 2·7 |
| Latin America, Andean | 3,033 | 7,754 | 155·7 | 202·7 | 193·0 | −4·8 | 210·3 | 192·1 | −8·6 | 195·1 | 193·9 | −0·6 |
| Latin America, Central | 6,330 | 11,458 | 81·0 | 208·7 | 203·2 | −2·6 | 223·4 | 213·1 | −4·6 | 194·0 | 193·4 | −0·3 |
| Latin America, Southern | 4,893 | 15,242 | 211·5 | 267·9 | 243·3 | −9·2 | 338·8 | 287·4 | −15·2 | 197·0 | 199·1 | 1·1 |
| Latin America, Tropical | 4,975 | 10,789 | 116·9 | 221·2 | 218·5 | −1·3 | 247·5 | 237·5 | −4·1 | 194·9 | 199·5 | 2·3 |
| North Africa / Middle East | 4,108 | 9,743 | 137·2 | 225·3 | 218·3 | −3·1 | 258·6 | 248·7 | −3·8 | 192·0 | 187·8 | −2·2 |
| North America, High Income | 22,450 | 46,337 | 106·4 | 353·5 | 315·2 | −10·8 | 427·9 | 348·1 | −18·7 | 279·1 | 282·3 | 1·1 |
| Oceania | 1,383 | 2,711 | 96·0 | 197·0 | 195·5 | −0·8 | 197·9 | 193·1 | −2·4 | 196·1 | 197·9 | 0·9 |
| Sub-Saharan Africa, Central | 927 | 949 | 2·5 | 181·3 | 182·1 | 0·4 | 183·2 | 185·0 | 1·0 | 179·5 | 179·3 | −0·1 |
| Sub-Saharan Africa, East | 561 | 1,294 | 130·8 | 166·9 | 167·1 | 0·1 | 159·6 | 161·1 | 1·0 | 174·2 | 173·1 | −0·7 |
| Sub-Saharan Africa, Southern | 5,193 | 9,854 | 89·7 | 256·3 | 217·2 | −15·3 | 300·9 | 238·4 | −20·8 | 211·7 | 195·9 | −7·4 |
| Sub-Saharan Africa, West | 854 | 769 | −9 ·9 | 168·1 | 166·4 | −1·0 | 166·7 | 164·4 | −1·4 | 169·6 | 168·5 | −0·6 |
Multilevel regression analysis, adjusting for age and sex, showed a highly statistically significant association of smoking index and GNI with COPD mortality rate for both years (Table 3). Since the model is a log-log regression (exposure and outcome both log transformed), the coefficients for GNI and smoking represent the % change in COPD mortality rate given a certain % change in GNI or smoking, holding all other variables constant. For example in the analysis of 1990, if we consider the effect of a 50% increase in exposure (1.5 times), the GNI coefficient of −0·47 means a decrease in mortality rate of 17% (1·5−0·47=0·83, i.e. 17% reduction); similarly, the smoking coefficient of 0.43 means a 19% increase in mortality rate. Corresponding figures for 2010 were 12% (3 to 19%) decrease for a 50% increase in GNI and 17% (12 to 21%) increase for a 50% increase in smoking index. As expected, for both 1990 and 2010 the model showed COPD mortality rates were higher in older age groups and in men, independently from smoking.
TABLE 3.
Results of the multilevel regression models for COPD mortality rate separately for 1990 and 2010, and for the 2010/1990 COPD mortality rate ratio
| Variable | (log) COPD Mortality Rate | Variable | (log) 2010/1990 COPD Mortality Rate Ratio | ||||
|---|---|---|---|---|---|---|---|
| 1990 | 2010 | ||||||
| Coefficient (95%CI) | p-value | Coefficient (95%CI) | p-value | Coefficient (95%CI) | p-value | ||
| (log) Smoking Index | 0·43 (0·33 to 0·52) | <0·001 | 0·38 (0·28 to 0·48) | <0·001 | (log) 2010/1990 Smoking Index Ratio | 0·004 (0·002 to 0·004) | <0·001 |
| (log) GNI | −0·47 (−0·68 to −0·26) | <0·001 | −0·31 (−0·53 to −0·08) | 0·01 | (log) 2010/1990 GNI ratio | −0·34 (−0·60 to −0·06) | 0·02 |
| Age | Age | ||||||
| 40-44 | Reference | Reference | 40-44 | Reference | |||
| 45-49 | 0·69 (0·56 to 0·81) | <0·001 | 0·72 (0·58 to 0·86) | <0·001 | 45-49 | 0·04 (−0·04 to 0·11) | 0·38 |
| 50-54 | 1·47 (1·35 to 1·59) | <0·001 | 1·47 (1·34 to 1·61) | <0.001 | 50-54 | 0·01 (−0·07 to 0·09) | 0·76 |
| 55-59 | 2·16 (2·03 to 2·28) | <0·001 | 2·19 (2·05 to 2·32) | <0.001 | 55-59 | 0·05 (−0·03 to 0·13) | 0·24 |
| 60-64 | 2·81 (2·68 to 2·94) | <0·001 | 2·88 (2·74 to 3·01) | <0·001 | 60-64 | 0·10 (0·02 to 0·18) | 0·02 |
| 65-69 | 3·60 (3·47 to 3·72) | <0·001 | 3·76 (3·62 to 3·89) | <0·001 | 65-69 | 0·19 (0·11 to 0·27) | <0·001 |
| 70-74 | 3.91 (3·78 to 4·04) | <0·001 | 4·08 (3·94 to 4·22) | <0·001 | 70-74 | 0·20 (0·12 to 0·28) | <0·001 |
| 75-79 | 4·42 (4·29 to 4·55) | <0·001 | 4·62 (4·48 to 4·77) | <0·001 | 75-79 | 0·21 (0·13 to 0·29) | <0·001 |
| 80+ | 5·11 (4·98 to 5·24) | <0.001 | 5·43 (5·28 to 5·57) | <0·001 | 80+ | 0·26 (0·18 to 0·34) | <0·001 |
| Sex | Sex | ||||||
| Male | Reference | Reference | Male | Reference | |||
| Female | −0·56 (−0·62 to −0·50) | <0·001 | −0·64 (−0·70 to −0·57) | <0·001 | Female | −0·12 (−0·16 to −0·07) | <0·001 |
The multilevel regression analysis of the ratio of COPD mortality rates between 2010 and 1990, also analysed as a log-log model, showed a 13% decrease in mortality rate for a 50% increase in GNI from 1990 to 2010 (p=0.02). The effect of change in smoking index (2010/1990 ratio) on change in COPD mortality was also statistically significant, although its magnitude was much smaller, 0·16% (0·08 to 0·17%) increase in COPD mortality rate for a 50% increase in smoking index (Table 3). In agreement with Figure 2, the multilevel regression model showed large between-region variation in COPD mortality, with an intraclass correlation coefficient of 0·69. When we dropped from the analysis the four regions with the highest change in GNI we obtained the same coefficient for the smoking index and only a small change in the coefficient for GNI, with a 16% (4% to 25%) decrease in COPD mortality rate for a 50% increase in GNI from 1990 to 2010. This remained statistically significant.
Finally, we estimated the percentage of the change in COPD mortality that was attributable to the change in GNI and smoking exposure. Changes in GNI and smoking exposure accounted for 26·2% (95% CI: 4.7 to 47.7%) and 3·9% (3.2 to 4.6%) of the change in COPD mortality, respectively.
DISCUSSION
The relative increased burden from COPD mortality compared with mortality from other causes is largely due to changes in population structure. The apparent paradox that COPD has risen to be the third most common cause of death at a time when age standardised mortality rates from COPD have been falling is explained by the relatively faster decline in all-cause mortality. Between 1990 and 2010 the total number of COPD deaths fell from 3 to 2.8 million a fall of 5.5% while during the same period deaths due to all causes fell by 17%. The increase in COPD mortality due to population aging explains why the absolute number of COPD deaths has decreased only slightly although age-sex specific mortality rates have almost everywhere been falling. Much of this fall is associated with changes in gross national income (GNI), countries with the greatest improvement in GNI having the greatest relative improvement in COPD mortality rates. Changes over time in the cumulative exposure to cigarette smoking have had relatively little impact.
The ecological design of this analysis limits conclusions to inferences about populations rather than individuals, but unlike most ecological studies the longitudinal design and analysis of within-region changes over time largely overcomes the usual problem of confounding with other large between-region differences which is typical of many ecological studies.
Since the middle of the 19th century, the analysis of cause of death data has provided important insights into disease burden and the causes of ill health.(19) The data are nevertheless limited by incomplete records and inaccuracies in disease coding. Our definition of COPD deaths is deliberately broad, including all chronic lower respiratory tract causes whether bronchitic or obstructive, with the sole exception of asthma. Narrower definitions are more prone to misclassification and to local variation in the practice of both medicine and cause of death coding. With chronic diseases in particular, the assumption of a single underlying cause of death is often unrealistic, and those certifying the cause of death generally have limited information on which to determine the underlying cause of death and limited options on what cause to code it to. Poor practice often exacerbates these limitations. The protocols of the Global Burden of Disease programme have been developed to minimise these disadvantages, and the algorithms used to improve the reliability of these data, taking account of changes in the quality of cause of death coding and changes in the ICD classification over time, (15) have been extensively tested by out-of-sample tests of their predictive power.
The smoking index used provides a summary of smoking experience over the life time for each age group and has been widely used, the original paper being cited over 700 times. However, the estimates of cigarette smoking are indirect and depend on a high and consistent proportion of lung cancer deaths being attributable to smoking. This is an approximation. The population attributable fraction of lung cancer in men associated with smoking has been estimated as 92% in industrialised regions but as only 67% in developing regions.(20)
Although the method would be improved by using local estimates of “non-smoking” lung cancer, these are not universally available and we have kept to a single estimate. Any bias introduced by this simplification would have only small effects on within-region comparisons between 1990 and 2010. Use of this index is better than attempting to reconstruct smoking exposure over a lifetime from alternative sources such as surveys and sales, providing a relatively consistent source of information on the cumulative lifetime exposure to tobacco.
The downward trend for age-sex specific mortality rates from COPD has been demonstrated for the United Kingdom. Marks and Burney (7) reviewed mortality rates for respiratory causes of death from the beginning of the 20th century and showed that mortality rates began to decline starting around the time of the First World War. The effect of the smoking epidemic was to slow, or halt, this decline more than to lead to an absolute increase in rates. Both Adair et al.(11) and Erbas et al.(12) showed similar results in Australia, with much lower and declining rates in the first half of the 20th century, followed by rising rates up until 1970-1990 among men and 1990-2000 among women. Pride and Soriano also noted falling rates for bronchitis between 1931 and 1965 among 45-64 years olds in the UK, at a time when death rates were rising from lung cancer.(8) The strong association between COPD mortality and poverty is not new. In the United Kingdom this association was demonstrated at an ecological level by Barker et al.(5) and was shown to be far stronger than the association between poverty and lung cancer. At an individual level, the social class gradient of the mortality rate for COPD has been steeper than those for lung cancer, or even for tuberculosis. (21)
The strong association we observed between changes in COPD mortality and changes in GNI and the relatively weak association with changes in smoking index depend on the rate of change in GNI and smoking index as well as on the strength of their association with COPD mortality. The positive association of COPD mortality with the smoking index in both years and the positive association between changes in the smoking index and changes in COPD mortality confirm the relevance of cigarette smoking to COPD mortality rates. The relatively small impact of smoking changes is largely due to the small change in global smoking rates, with improvements in some regions being offset by major increases in smoking in the populous regions of South, East and South East Asia. Nevertheless the far greater impact of GNI requires some further explanation.
The Burden of Obstructive Lung Disease (BOLD) study has shown a strong ecological association between the prevalence of airway obstruction and the prevalence of smoking, but has shown no such association with poverty. However it did show a strong association between the prevalence of poverty and the prevalence of a low Forced Vital Capacity (FVC), and the prevalence of a low FVC increases sharply in countries with a per capita GNI below around $15 000 per annum.(6) Data from the United States has also shown that survival is much more strongly linked to the FVC than to the FEV1/FVC ratio, with the latter showing very little association with mortality once smoking has been adjusted for.(22) Although some discount the low FVC in poor countries as confounded by race, evidence, again from the United States, shows that the outcome from a low FVC is no different in African Americans and white Americans after adjusting for age, sex and height.(23) These studies suggest that the high mortality from COPD in low income countries is associated with a low FVC and not with obstruction, as has generally been assumed, and raises an important question over the origins of this low FVC. There is good evidence that a low ventilatory function in later life is associated with low birth weight (5,24-27) and possibly with early infections,(4,5,28) and these conditions are associated with poverty.(4,29,30) However, COPD mortality appears to respond immediately to improvements in GNI, and it is therefore unlikely that the changes noted here are solely related to effects from poverty in early life. The mechanism of this more immediate effect, and whether it is mediated through general changes in lifestyle including environmental changes, or through access to health care, is unclear. It is unlikely that it is due to specific changes in care for COPD, but general access to support could be part of an explanation.
These findings have important implications for both policy and research. The association between COPD mortality and smoking emphasises the need to drive down smoking rates in all regions, particularly those with rising smoking rates, but our results show that this will not be enough. The strong association with poverty, particularly in the poorest regions of the world, demonstrate that poverty reduction will be critical in reversing the growing importance of COPD mortality. Meantime further research is required to understand what mediates the effects of poverty on COPD mortality.
In conclusion, the increase in the global burden of COPD mortality between 1990 and 2010 has been due to changes in the age distribution, while improved economic conditions, particularly in the poorest countries, have been associated with a fall in age-sex specific COPD mortality rates. The change in global smoking rates has been relatively small over the last 20 years, with falls in some areas offset by increases in others. This accounts for the relatively small influence of changes in smoking habits on changes in global COPD mortality.
Acknowledgements
The authors would like to acknowledge Christopher Murray, Rafael Lozano, Nicholas J. Kassebaum, and Spencer L. James for their contributions to this work.
Funding:
This work was supported by funding from the Bill and Melinda Gates Foundation. Jaymini Patel is supported by the Wellcome Trust (085790/Z/08/Z).
Footnotes
Conflicts of interest:
There are no conflicts of interest with the contents of this paper.
REFERENCES
- (1).Lozano R, Naghavi M, Foreman K, Lira S, Shibuya K, Aboyans V, et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet. 2012;380(9859):2095–2128. doi: 10.1016/S0140-6736(12)61728-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (2).Murray C. Alternative projections of mortality and disability by cause 1990-2020: Global Burden of Disease Study. Lancet. 1997;349:1498–1504. doi: 10.1016/S0140-6736(96)07492-2. [DOI] [PubMed] [Google Scholar]
- (3).Eisner M, Anthonisen N, Coultas D, Kuenzli N, Perez-Padilla R, Postma D, et al. An official American Thoracic Society public policy statement: Novel risk factors and the global burden of chronic obstructive pulmonary disease. Amer J Respir Crit Care Med. 2010;182(5):693–718. doi: 10.1164/rccm.200811-1757ST. [DOI] [PubMed] [Google Scholar]
- (4).Barker DJP, Osmond C. Childhood respiratory infection and adult chronic bronchitis in England and Wales. B M J. 1986;293:1271–1275. doi: 10.1136/bmj.293.6557.1271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (5).Barker DJP, Godfrey KM, Fall C, Osmond C, Winter PD, Shaheen SO. Relation of birth weight and childhood respiratory infection to adult lung function and death from chronic obstructive airways disease. B M J. 1991;303:671–675. doi: 10.1136/bmj.303.6804.671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (6).Burney P, Jithoo A, Kato B, Janson C, Mannino D, Niżankowska-Mogilnicka E, et al. Chronic Obstructive Pulmonary Disease Mortality and Prevalence: the associations with smoking and poverty - a BOLD analysis. Thorax. 2014;69(5):465–473. doi: 10.1136/thoraxjnl-2013-204460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (7).Marks G, Burney P. Diseases of the Respiratory System. In: Charlton J, editor. The Health of Adult Britain: 1841-1994. The Stationery Office; London: 1997. [Google Scholar]
- (8).Pride NB, Soriano JB. Chronic obstructive pulmonary disease in the United Kingdom: trends in mortality, morbidity and smoking. Curr Opin Pulm Med. 2002;8:95–101. doi: 10.1097/00063198-200203000-00003. [DOI] [PubMed] [Google Scholar]
- (9).Lopez AD, Shibuya K, Rao C, C.D. Mathers CD, A.L. Hansell AL, L.S. Held LS, et al. Chronic obstructive pulmonary disease: current burden and future projections. Eur Respir J. 2006;27:397–412. doi: 10.1183/09031936.06.00025805. [DOI] [PubMed] [Google Scholar]
- (10).Tan WC, Seale P, Ip M, Shim Y, Chiang C-, Ng T-, et al. Trends in COPD mortality and hospitalizations in countries and regions of Asia-Pacific. Respirology. 2009;14(1):90–97. doi: 10.1111/j.1440-1843.2008.01415.x. [DOI] [PubMed] [Google Scholar]
- (11).Adair T, Hoy D, Dettrick Z, Lopez AD. 100 years of mortality due to chronic obstructive pulmonary disease in Australia: the role of tobacco consumption. Int J Tuber Lung Dis. 2012;16(12):1699–1705. doi: 10.5588/ijtld.12.0251. [DOI] [PubMed] [Google Scholar]
- (12).Erbas B, Ullah S, Hyndman RJ, Scollo M, Abramson M. Forecasts of COPD mortalty in Australia:2006-2025. BMC Medical Research Methodology. 2012;12(17) doi: 10.1186/1471-2288-12-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (13).Pham T, Ozasa K, Kubo T, Fujino Y, Sakata R, Grant EJ, et al. Age-Period-Cohort Analysis of Chronic Obstructive Pulmonary Disease Mortality in Japan, 1950-2004. J Epidemiol. 2012;22(4):302–307. doi: 10.2188/jea.JE20110092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (14).López-Campos JL, Ruiz-Ramos M, Soriano JB. Mortality trends in chronic obstructive pulmonary disease in Europe, 1994-2010: a joinpoint regression analysis. Lancet Respir Med. 2014;2(1):54–62. doi: 10.1016/S2213-2600(13)70232-7. [DOI] [PubMed] [Google Scholar]
- (15).Naghavi M, Makela S, Foreman F, O’Brien J, Pourmalek F, Lozano R. Algorithms for enhancing public health utility of national causes-of-death data. Population Health Metrics. 2010;8:9. doi: 10.1186/1478-7954-8-9. 2010;8:9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (16).Peto R, Lopez AD, Boreham J, Thun M, Heath C. Mortality from tobacco in developed countries: indirect estimation from national vital statistics. Lancet. 1992;339(8804):1268–1278. doi: 10.1016/0140-6736(92)91600-d. [DOI] [PubMed] [Google Scholar]
- (17).World Bank [Accessed 3/6, 2012];Open Data: Indicators: Gross National Income per capita, PPP (current international $) Available at: http://data.worldbank.org/indicator/NY.GNP.PCAP.PP.CD.
- (18).Williams R. Using the margins command to estimate and interpret adjusted predictions and marginal effects. Stata Journal. 2012;12(2):308–31. [Google Scholar]
- (19).Greenhow EH. Papers relating to the sanitary state of the people of England: being an enquiry into the different proportions of death produced by certain diseases in different districts of England. HMSO; London: 1858. [1858]. [Google Scholar]
- (20).Ezzati M, Lopez AD. Estimates of global mortality attributable to smoking in 2000. Lancet. 2003;362:847–852. doi: 10.1016/S0140-6736(03)14338-3. [DOI] [PubMed] [Google Scholar]
- (21).Great Britain. Office of Population Censuses and Surveys . Occupational Mortality: The Registrar General’s Decennial Supplement for Great Britain, 1979-80, 1982-83, Great Britain. H.M. Stationery Office; London: 1986. Office of Population Censuses and Surveys. [Google Scholar]
- (22).Burney PGJ, Hooper R. Forced vital capacity, airway obstruction and survival in a general population sample from the USA. Thorax. 2011;66(1):49–54. doi: 10.1136/thx.2010.147041. [DOI] [PubMed] [Google Scholar]
- (23).Burney PGJ, Hooper RJ. The use of ethnically specific norms for ventilatory function in African-American and white populations. Int J Epidemiol. 2012;41(3):782–790. doi: 10.1093/ije/dys011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (24).Lawlor DA, Ebrahim S, Davey SG. Association of birth weight with adult lung function: findings from the British Women’s Heart and Health Study and a meta-analysis. [Review] [40 refs] Thorax. 2005 Oct;60(10):851–858. doi: 10.1136/thx.2005.042408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (25).Qi-Qiang H, Tze-Wai W, Lin D, Zhuo-Qin J, Yang G, Guo-Zhen L, et al. Birth weight and lung function in a cohort of Chinese school children. Pediatr Pulmonol. 2009;44(7):662–8. doi: 10.1002/ppul.21035. [DOI] [PubMed] [Google Scholar]
- (26).Canoy D, Pekkanen J, Elliott P, Pouta A, Laitinen J, Hartikainen A, et al. Early growth and adult respiratory function in men and women followed from the fetal period to adulthood. Thorax. 2007;62(5):396–402. doi: 10.1136/thx.2006.066241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (27).Pei L, Chen G, Mi J, Zhang T, Song X, Chen J, et al. Low birth weight and lung function in adulthood: retrospective cohort study in china, 1948-1996. Pediatrics. 2010;125(4):e899–905. doi: 10.1542/peds.2008-3086. [DOI] [PubMed] [Google Scholar]
- (28).Shaheen SO, Barker DJ, Shiell AW, Crocker FJ, Wield GA, Holgate ST. The relationship between pneumonia in early childhood and impaired lung function in late adult life. Amer J Respir Crit Care Med. 1994 Mar;149(3 Part 1):616–619. doi: 10.1164/ajrccm.149.3.8118627. [DOI] [PubMed] [Google Scholar]
- (29).Kramer MS, Seguin L, Lydon J, Goulet L. Socioeconomic disparities in pregnancy outcome: why do the poor fare so poorly? Paediatr Perinat Epidemiol. 2000;14(3):194–210. doi: 10.1046/j.1365-3016.2000.00266.x. [DOI] [PubMed] [Google Scholar]
- (30).Thorn LKAM, Minamisava R, Nouer SS, Ribeiro LH, Andrade A. Pneumonia and poverty: a prospective population-based study among children in Brazil. BMC Infectious Diseases. 2011;11:180. doi: 10.1186/1471-2334-11-180. [DOI] [PMC free article] [PubMed] [Google Scholar]