Abstract
Mammals are co-infected by multiple pathogens that interact through unknown mechanisms. We found that helminth infection, characterized by the induction of the cytokine interleukin-4 (IL-4) and the activation of the transcription factor Stat6, reactivated murine gammaherpesvirus infection in vivo. IL-4 promoted viral replication and blocked the antiviral effects of interferon-γ (IFNγ) by inducing Stat6 binding to the promoter for an important viral transcriptional transactivator. IL-4 also reactivated human Kaposi's sarcoma associated herpesvirus from latency in cultured cells. Exogenous IL-4 plus blockade of IFNγ reactivated latent murine gammaherpesvirus infection in vivo, suggesting a ‘two-signal’ model for viral reactivation. Thus chronic herpesvirus infection, a component of the mammalian virome, is regulated by the counterpoised actions of multiple cytokines on viral promoters that have evolved to sense host immune status.
Mammals are populated by many chronic viruses, termed the virome, which can regulate host physiology and disease susceptibility(1). For example, more than 90% of humans are latently infected with herpesviruses that, after clearance of acute infection, produce little infectious virus and often cause no overt disease. Like human gammaherpesviruses Epstein Barr Virus (EBV) and Kaposi’s sarcoma associated herpesvirus (KSHV), murine gammaherpesvirus-68 (MHV68) establishes lifelong latency. Studies in this model system showed that the cytokine IFNγ nhibits MHV68 replication and reactivation from macrophages, a major cellular site for latency(2–5), controls persistent replication in vivo(6), and is present at low amounts during latency(7).
Many people around the world are co-infected with herpesviruses and intestinal helminths. While herpesviruses can modulate immunity to harm or benefit the host(7–11), the effects of helminth co-infection on chronic herpesvirus infection are unexplored. Intestinal helminths generate strong T helper 2 (Th2)-driven cytokine responses, which counter the biological effects of IFNγ, and drive the activation of macrophages with an M2 (immunoregulatory) rather than an M1 (pro-inflammatory) phenotype(12). Parasitic worms may influence control of pathogens, including Mycobacterium tuberculosis, HIV, and Plasmodium species in humans, but there are few studies elucidating the mechanisms behind this immunomodulation(13). Thus, we considered the hypothesis that parasite infection would induce MHV68 reactivation in vivo.
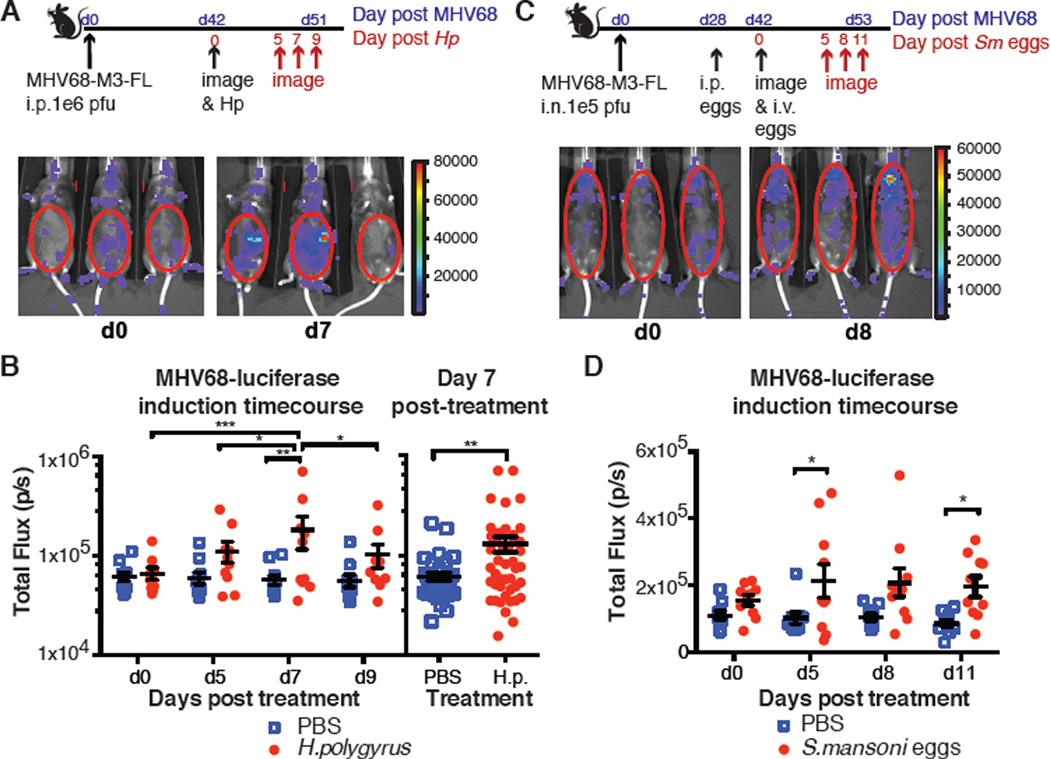
We examined the effects of acute infection with Heligmosomoides polygyrus or Schistosomiasis mansoni (Sm) egg administration on MHV68 reactivation from latency using a MHV68 virus expressing luciferase under the control of a lytic viral promoter upon reactivation from latency in vivo (MHV68-M3-FL)(14). Both acute H. polygyrus infection and Sm egg challenge reactivated MHV68 infection (Fig. 1A–D). Mice latently infected for over 100 days also showed increased luciferase expression following Sm egg challenge (fig. S1). By contrast, infection with the systemic bacteria, Listeria monocytogenes, did not stimulate viral reactivation (fig. S2). Thus, responses to either a nematode parasite or trematode eggs induced herpesvirus reactivation, suggesting a role for Th2 cytokines in viral reactivation.
Fig. 1. Challenge with H. polygyrus and S. mansoni eggs reactivates MHV68.
(A) C57BL6/J mice were infected intraperitoneally (i.p.) with MHV68-M3-FL and challenged with H. polygyrus 42 days later. Mice were imaged prior to H. polygyrus infection (day 0) and 5, 7 and 9 days after. Three representative mice imaged on days 0 and 7 are shown. (B) Total flux (photons/second) was quantitated for mice in 2 independent experiments for the timecourse after infection with H. polygyrus. Data from 4 independent experiments at day 7 post H. polygyrus is also shown. (C) C57BL/6J mice were infected intranasally (i.n.) with MHV68-M3-FL. Diagram indicates timecourse of experiment and challenge with Sm eggs or PBS as a control. Mice were injected with D-Luciferin and imaged prior to intravenous (i.v.) challenge with Sm eggs (day 0). They were subsequently imaged 5, 8 and 11 days after challenge with Sm eggs. Three representative mice imaged on days 0 and 8 are shown. (D) Total flux (photons/second) was quantitated from mice in two independent experiments after Sm egg challenge. Symbols represent individual mice, and the mean and standard error are indicated * p<0.05, ** p<0.01, *** p<0.001 by 2-way repeated measures ANOVA with Tukey’s and Bonferroni’s post-test.
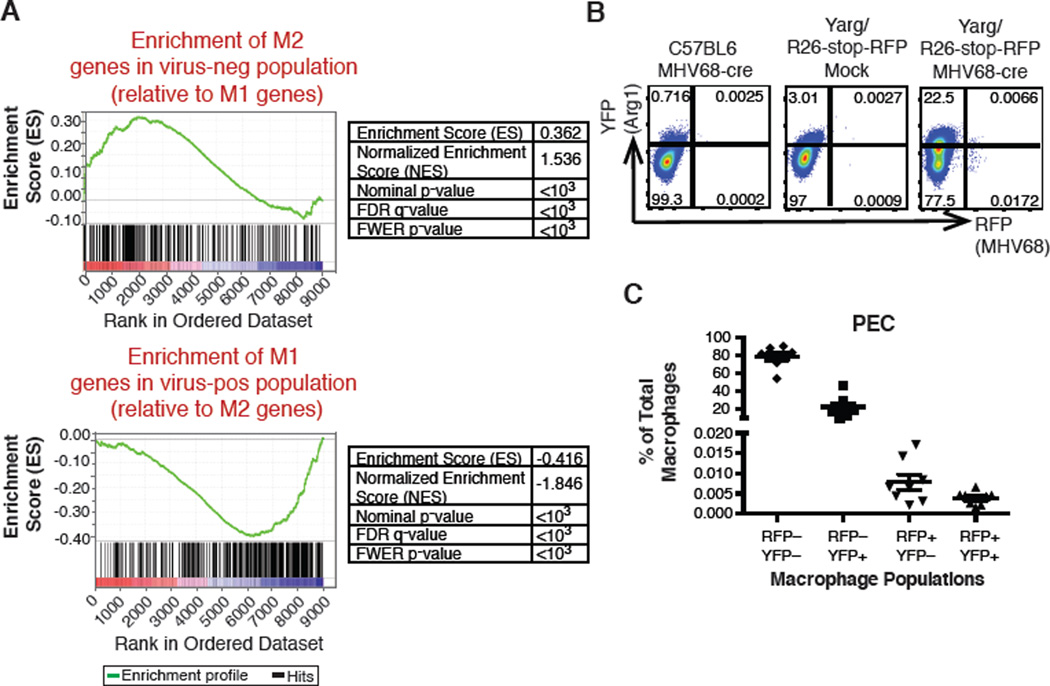
To determine whether Th2 cytokines affect latently infected macrophages we compared host gene expression patterns in virally infected and uninfected macrophages during chronic infection. We engineered MHV68 to express cre-recombinase (MHV68-cre) from a locus permitting heterologous gene expression without altering viral replication or reactivation(15) (fig. S3). Reporter mice in which fluorescent protein expression is induced by cre recombination (Rosa26-floxed stop-eYFP or tandem dimer (td)RFP(16)) were infected with MHV68-cre. Virus-positive and virus-negative cells sorted from latently infected mice (fig. S3G) were subjected to RNAseq analysis. Transcription in these cells was compared to that in bone marrow-derived macrophages (BMDMs) stimulated with IL-4 (M2) or IFNγ plus lipopolysaccharide (LPS) (M1). compared to untreated BMDMs (M0). Gene set enrichment analysis (GSEA) revealed that genes upregulated in M1 BMDMs were enriched in virus-positive macrophages whereas genes upregulated in M2 BMDMs were enriched in virus-negative cells (Fig. 2A, and table S1). This was consistent with the role of IFNγ, which drives M1 macrophage polarization, in inhibiting MHV68 replication and reactivation(3, 4). We therefore tested whether latent MHV68 infection was restricted to M1-type macrophages by infecting tandem dimer (td)RFP mice carrying the Arginase-1 (Arg1)-YFP reporter (YARG, a marker for macrophages stimulated with Th2 cytokines)(17) with MHV68-cre. Surprisingly, virus-positive macrophages were either Arg1-negative or Arg1-positive (Fig. 2B, C), suggesting that, despite the role for IFNγ in controlling chronic MHV68 infection, at least some virus-infected cells were exposed to cytokines that drive Arg-1 expression in vivo.
Fig. 2. IL-4 and IFNγ signatures identified in different macrophage populations during MHV68 infection.
(A) GSEA analysis of virus-positive and virus-negative cells sorted from the peritoneum of MHV68-infected mice compared to BMDMs stimulated with IL-4 or IFNγ/LPS. (B) C57BL/6J mice or YARG/R26-stop-RFP mice were either mock infected or infected with MHV68-cre. CD11b+F4/80+ cells were gated and examined for RFP and YFP expression. Representative plots from two independent experiments, with three to five mice per experiment. (C) Quantitation of FACS analysis in (B) with each symbol representing a single mouse.
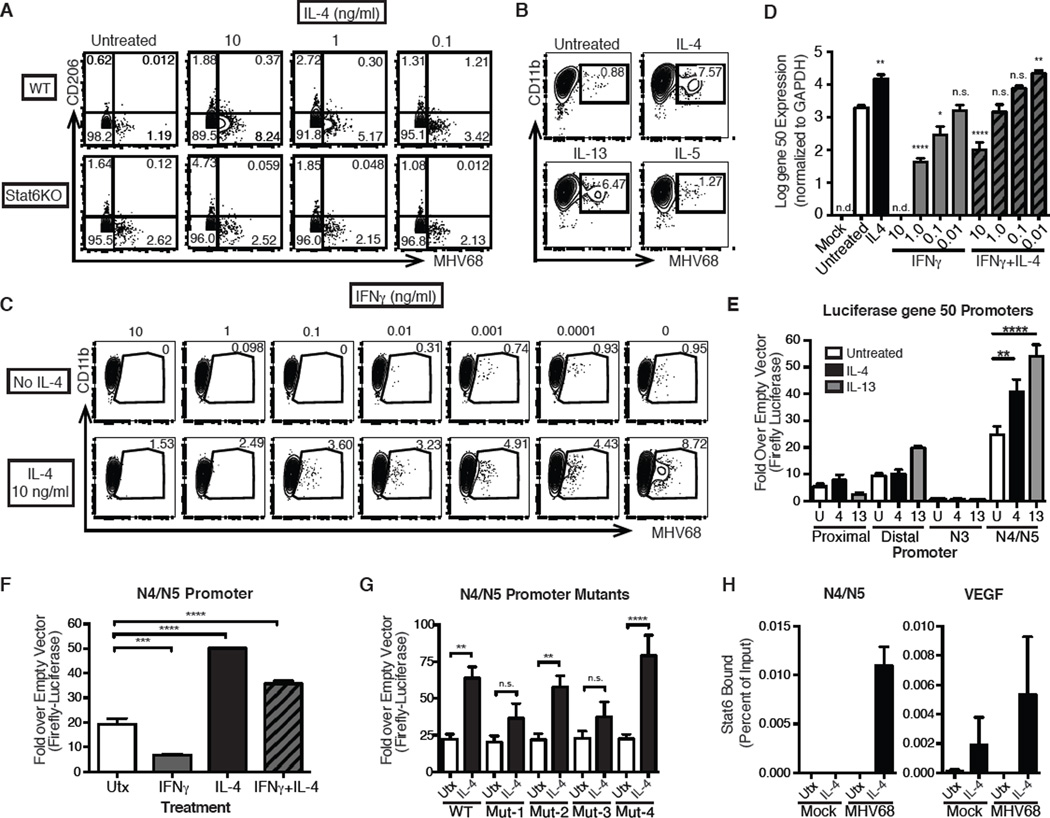
The presence of an IL-4 signature in some virus-infected macrophages along with the observation that Th2 cytokine-inducing parasites promoted reactivation from latency, suggested a role for IL-4 in viral infection. We tested this by determining the effect of IL-4 on MHV68 replication in BMDMs. Treatment with IL-4 increased Arg1 expression (fig. S4A) consistent with M2 polarization(18). As expected, few infected BMDMs expressed lytic viral antigens upon MHV68 infection(19). However, IL-4 pretreatment increased the number of BMDMs expressing viral proteins and enhanced viral replication (Fig. 3A, fig. S4B–D), and increased infection of transformed RAW264.7 macrophages (fig. S4E). Treatment with IL-4 after MHV68 infection increased viral replication (fig. S4F), indicating that IL-4 acts on replication rather than by increasing the number of infected cells. Enhancement of replication was dependent on the Th2-associated transcription factor Stat6 (Fig. 3A), and occurred with IL-13 stimulation, another Th2-associated cytokine that utilizes the IL-4 receptor α chain and signals via Stat6 (Fig. 3B, fig. S4D, G). The Th2 cytokine IL-5, which does not signal through Stat6, did not promote MHV68 replication (Fig. 3B).
Fig. 3. IL-4 promotes viral replication and antagonizes IFNγ suppression of viral replication through direct binding to a viral promoter.
(A) BMDMs were untreated or treated with the indicated doses of IL-4 and infected with MHV68. 24 hours post infection cells were analyzed for expression of MHV68 lytic viral proteins and CD206 expression. Represents three independent experiments. (B) Pretreatment of BMDMs with IL-4, IL-13, or IL-5 prior to infection with MHV68 and FACS analysis. (C) BMDMs were pretreated with varying doses of IFNγ ± 10 ng/ml of IL-4. 24 hours post infection cells were analyzed for expression of lytic viral proteins. Represents three independent experiments. (D) Gene 50 expression was analyzed by RT-PCR in BMDMs pretreated with IL-4 and/or IFNγ. Expression was normalized to Gapdh. Represents four independent experiments. (E) RAW264.7 cells were transfected with vectors expressing luciferase under control of four different gene 50 promoters ((23, 24). Cells were then treated ± IL-4 or IL-13 for 24 hours, lysed and assayed for luciferase activity. (F) Cells were transfected with the N4/N5 promoter luciferase construct and treated with IL-4, IFNγ, or both. (G) N4/N5 luciferase mutants were transfected into RAW264.7 cells and assayed for sensitivity to IL-4. (H) RAW264.7 cells were infected with MHV68 at MOI=5 and treated with IL-4 for 8 hours. After chromatin immunoprecipitation with Stat6 antibody, quantitative PCR was performed for the N4/N5 promoter region or VEGF. Percent of input after normalizing to IgG control was calculated for both N4/N5 and VEGF. 1 experiment representative of 4 independent experiments is shown. (I) Schematic of N4/N5 luciferase construct with potential Stat-binding mutants. n.d. not detected. n.s. not significant. * p<0.05, ** p<0.01, *** p<0.001, **** p<0.0001 by T test or 1-way ANOVA with Sidak’s multiple comparisons test.
After treatment with IL-4, the majority of infected cells did not express the M2 markers CD206 or Arg1 (Fig. 3A, and fig. S4A), suggesting that not all IL-4-induced changes in macrophage differentiation are required for enhanced MHV68 replication(20). Etomoxir blocks IL-4-induced changes in fatty–acid oxidation(21) and upregulation of CD206 (fig. S5A) but did not block enhancement of MHV68 replication by IL-4 (fig. S5B). Moreover, IL-4 enhanced replication in the absence of PPARγ or ARG1, key proteins involved in M2 macrophage function, or iNOS, an essential protein in M1 macrophage function (fig. S5C–G)(20). Importantly, IL-4 antagonized IFNγ-mediated suppression of viral replication (Fig. 3C)(3). Because Stat6 antagonizes Stat1(22), we tested whether IL-4 promoted virus replication in the absence of Stat1. IL-4 increased virus replication in Stat1-deficient BMDMs (fig. S6).
Previously, we found that IFNγ-mediated suppression of viral replication was associated with inhibition of promoters driving expression of the essential viral latent-to-lytic switch gene (gene 50) (3, 23). Importantly, IL-4 antagonizes IFNγ-mediated suppression of gene 50 transcription (Fig. 3D). This effect was specific to the viral promoter because IL-4 did not block IFNγ-mediated induction of Nos2, and IFNγ did not inhibit IL-4-mediated induction of Arg1 and Relmα/Fizz1 (fig S7). Furthermore, IL-4 and IL-13 transactivated the gene 50 N4/N5 promoter (Fig. 3E)(24), and IL-4 antagonized IFNγ-mediated suppression of N4/N5 promoter (Fig. 3F). The effect of IL-4 on the N4/N5 promoter was diminished by mutation of two of four putative Stat-binding sites in the promoter (Fig. 3G, fig. S8). Further, chromatin immunoprecipitation experiments revealed that Stat6 bound to the N4/N5 promoter after IL-4 treatment of MHV68-infected cells (Fig. 3H). Taken together, these data suggest that activated Stat6 induced by IL-4/IL-13 promotes viral replication by binding to and acting on a viral promoter to induce expression of gene 50.
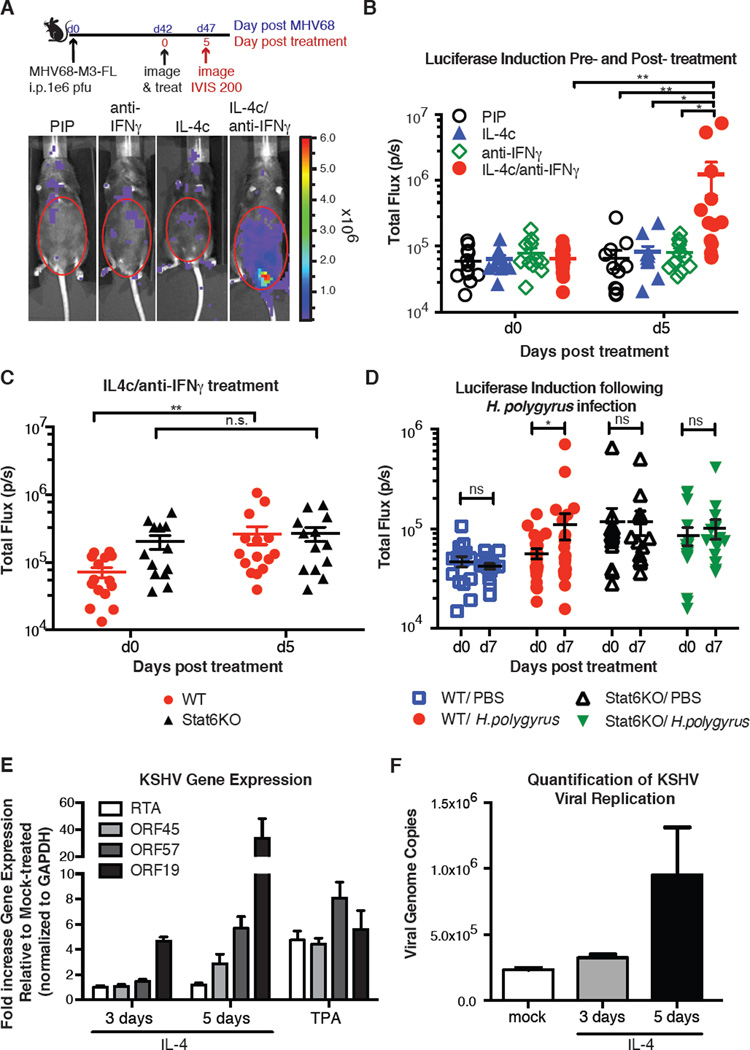
These counterbalancing effects of IFNγ and IL-4 on virus replication and viral promoter activity suggested a potential mechanism by which IL-4-inducing pathogens such as helminths promote reactivation. We therefore treated mice infected with MHV68-M3-FL virus with a blocking antibody to IFNγ (clone H22)(25), an isotype control antibody (clone PIP), long-lasting IL-4 complexes (IL4c)(26), or a combination of anti IFNγ and IL4c. No reactivation was observed after treatment with anti-IFNγ, IL4c, or PIP alone, indicating that a single signal was insufficient to reactivate virus in vivo. However, robust reactivation was observed in mice that received a combination of IL-4c and anti-IFNγ (Fig. 4A, B, fig. S9A). We next assayed reactivation using an independent assay(27, 28). Little or no preformed virus was detectable in tissues after treatment with PIP, IL4c, or anti-IFNγ alone(27), while treatment with IL-4c plus anti-IFNγ increased infectious virus (fig. S9B, C). Together these data supports a 'two-signal' mechanism by which co-infections could induce reactivation via induction of IL-4 and inhibition of Th1 responses(12).
Fig. 4. MHV68 and KSHV reactivation from latency is regulated by IL-4.
(A) C57BL6/J mice were infected with MHV68-M3-FL i.p. 42 days later mice were imaged for luciferase expression (d0) and then received isotype control (PIP), anti-IFNγ (H22), IL4c, or both anti-IFNγ and IL-4c. On day 44 mice received a second dose of IL4c or PBS. Mice were imaged five days after the first treatment and total flux from the abdominal region was quantitated. Four representative mice are shown. (B) Quantitation of total flux from three individual experiments described in (A) are shown. Each symbol represents an individual mouse. Bars are means, and error bars are standard errors. (C) WT or Stat6KO mice were treated as in (A) with anti-IFNγ/IL4c and imaged 5 days later. Bars are the means of individual mice (symbols), and error bars are standard error. (D) Experimental set-up was the same as Fig. 1A. Latently infected WT and Stat6KO mice infected with MHV68-M3-FL were infected with H. polygyrus or treated with PBS on day 42 and reactivation was quantitated by luciferase induction 7 days later. (E) BCBL-1 cells were treated with IL-4 for 3 or 5 days or TPA for 48 hours and viral gene expression was analyzed. Shown is the fold increase in gene expression over mock after normalization of GAPDH. Data from 3 independent experiments. (F) Supernatants from cells treated in (E) were collected and virus was isolated by centrifugation. Viral genome copy number was assayed by qPCR using serial diluted LANA expression plasmid as a standard curve. 1 experiment representative of 2 independent experiments is shown. For luciferase experiments: * p<0.05, ** p<0.01, n.s. not significant by 2-way repeated measures ANOVA with Tukey’s and Bonferroni’s post-test.
Increased reactivation after treatment with both IL4c/anti-IFNγ required Stat6 (Fig. 4C). We did not test the role of Stat1 or the IFNγ receptor because both are required to establish latency(6). To assess whether the effects of helminth infection on MHV68 reactivation also required Stat6, we challenged MHV68-infected Stat6KO mice with H. polygyrus. We found that helminth infection did not reactivate MHV68 from latency in Stat6KO mice, further supporting a two-signal model for control of gammaherpesvirus reactivation in vivo (Fig.4D).
Our results suggested a possible role for IL-4 in human gammaherpesvirus reactivation. We therefore tested whether IL-4 could reactivate the human gammaherpesvirus, KSHV in the BCBL-1 human B cell lymphoma cell line. We found that treatment with IL-4 increased immediate early (RTA, ORF45, and ORF57) and late viral transcripts (ORF19) (29)(Fig. 4E). RTA is the homolog in KSHV of MHV68 gene50, and ORF 45 and ORF57 are both transactivators, indicating a common role of IL-4 in regulating important viral transcriptional transactivators. Furthermore, IL-4 treatment of cells increased virus production (Fig. 4F), indicating that IL-4 is capable of inducing reactivation of KSHV.
A remarkable aspect of herpesvirus infection is its permanence despite ongoing immunity combined with the capacity to reactivate and spread to new hosts. This work illuminates one potential mechanism by which a gammaherpesvirus exhibits these two apparently disparate functions. Our data suggest that the virus evolved cytokine-responsive promoters to remain latent under some conditions (IFNγ-dominant) while reactivating under other conditions (IL-4-dominant). In this setting, co-infection may govern the outcome of reactivation by changing the balance in IL-4 and IFNγ, thus raising a potential issue with herpesvirus reactivation and proposed live helminth therapies(12). Additionally, our data illustrate one potential mechanism by which helminths and other Type 2 immune response-inducing parasites influence host control of another pathogen through M2 macrophage polarization(13). The fact that viral promoters for an essential gene are responsive to host cytokines implies that the viral genome evolved to sense the infection status of the host. We speculate that a similar mechanism for IL-4-induced reactivation of KSHV could also be true. Although not extensively studied, seroprevalence to KSHV is associated with hookworm and other parasitic infections in Uganda(30). Intriguingly, certain Burkitt’s lymphoma cell lines are reported to express EBV transcripts in response to IL-4(31).
Although mouse studies are done in specific pathogen-free animals, our data suggest that there is added complexity when multiple pathogens infect the same host, particularly in situations where one pathogen has the capacity to respond to specific immune signals generated to another pathogen to regulate chronic infection. Previously we showed that herpesvirus infection, a component of the mammalian virome(1) enhances resistance to some pathogens(7). Here we demonstrate the opposite effect, that co-infection regulates herpesvirus reactivation. These studies emphasize that immunity to chronic infection is a dynamic equilibrium regulated by co-infections, in part through highly evolved pathogen genomes with the capacity to sense host cytokines.
Supplementary Material
Acknowledgments
TAR was supported by Damon Runyon Postdoctoral Fellowship. This work was supported by grant U54 AI057160 and RO1 CA96511 to HWV, and grant AI032573 to EJP. The authors would like to thank R. Schreiber and K. Sheehan for supplying anti-IFNγ and PIP; G.Randolph and E. Gautier for PPARγ f/f×LyzMcre mice and helpful discussion; J. Urban for help setting up the H. polygyrus system; D. Kreamalmeyer for animal care and breeding; members of the Virgin lab for manuscript review and discussion; the Genome Technology Access Center at Washington University for sequencing; and the Flow Cytometry Core at Washington University for assistance with sorting. The data presented in this manuscript are tabulated in the main paper and in the supplementary materials. The accession number for RNAseq data on virus-infected cells is GSE58116, and the accession number for the BMDMs is GSE21895.
Footnotes
TAR designed the project, performed experiments, and wrote the paper. HWV assisted with project design and paper writing. BSW and SHS defined promoters and performed promoter assays. XZ created and characterized MHV68-cre virus, performed LD-PCR and western blots. HSC and RR performed KSHV experiments. MMH and MHK performed ChIP experiments. SCH and EJP helped design helminth experiments and supplied reagents. MDB did RT-PCR for gene 50 and host genes. AJ did flow cytometry with IL-4 in RAW264.7 cells. AK did western blots. CYL did plaque assays. GG and RX did initial bioinformatic analysis of gene expression by array, later replaced with RNASeq. PJM provided arginase mice. MNA analyzed sequencing data.
References and Notes
- 1.Virgin HW. The Virome in Mammalian Physiology and Disease. Cell. 2014;157:142–150. doi: 10.1016/j.cell.2014.02.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Flaño E, Husain SM, Sample JT, Woodland DL, Blackman MA. Latent Murine γ-Herpesvirus Infection Is Established in Activated B Cells, Dendritic Cells, and Macrophages. J. Immunol. 2000;165:1074–1081. doi: 10.4049/jimmunol.165.2.1074. [DOI] [PubMed] [Google Scholar]
- 3.Goodwin MM, Canny S, Steed A, Virgin HW. Murine gammaherpesvirus 68 has evolved gamma interferon and stat1-repressible promoters for the lytic switch gene 50. J. Virol. 2010;84:3711–3717. doi: 10.1128/JVI.02099-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Steed A, Buch T, Waisman A, Virgin HW. Gamma interferon blocks gammaherpesvirus reactivation from latency in a cell type-specific manner. J. Virol. 2007;81:6134–6140. doi: 10.1128/JVI.00108-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Weck K, Kim S, Virgin H., IV Macrophages are the major reservoir of latent murine gammaherpesvirus 68 in peritoneal cells. J. Virol. 1999 doi: 10.1128/jvi.73.4.3273-3283.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Tibbetts SA, van Dyk LF, Speck SH, Virgin HW. Immune control of the number and reactivation phenotype of cells latently infected with a gammaherpesvirus. J. Virol. 2002;76:7125–7132. doi: 10.1128/JVI.76.14.7125-7132.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Barton ES, et al. Herpesvirus latency confers symbiotic protection from bacterial infection. Nature. 2007;447:326–329. doi: 10.1038/nature05762. [DOI] [PubMed] [Google Scholar]
- 8.Nguyen Y, McGuffie BA, Anderson VE, Weinberg JB. Gammaherpesvirus modulation of mouse adenovirus type 1 pathogenesis. Virology. 2008;380:182–190. doi: 10.1016/j.virol.2008.07.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Saito F, et al. MHV68 Latency Modulates the Host Immune Response to Influenza A Virus. Inflammation. 2013;36:1295–1303. doi: 10.1007/s10753-013-9668-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Peacock JW, Elsawa SF, Petty CC, Hickey WF, Bost KL. Exacerbation of experimental autoimmune encephalomyelitis in rodents infected with murine gammaherpesvirus-68. Eur. J. Immunol. 2003;33:1849–1858. doi: 10.1002/eji.200323148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Virgin HW, Wherry EJ, Ahmed R. Redefining chronic viral infection. Cell. 2009;138:30–50. doi: 10.1016/j.cell.2009.06.036. [DOI] [PubMed] [Google Scholar]
- 12.McSorley HJ, Hewitson JP, Maizels RM. Immunomodulation by helminth parasites: defining mechanisms and mediators. Int. J. Parasitol. 2013;43:301–310. doi: 10.1016/j.ijpara.2012.11.011. [DOI] [PubMed] [Google Scholar]
- 13.Salgame P, Yap GS, Gause WC. Effect of helminth-induced immunity on infections with microbial pathogens. Nat. Immunol. 2013;14:1118–1126. doi: 10.1038/ni.2736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hwang S, et al. Persistent gammaherpesvirus replication and dynamic interaction with the host in vivo. J. Virol. 2008;82:12498–12509. doi: 10.1128/JVI.01152-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Moser JM, Upton JW, Allen RD, Wilson CB, Speck SH. Role of B-cell proliferation in the establishment of gammaherpesvirus latency. J. Virol. 2005;79:9480–9491. doi: 10.1128/JVI.79.15.9480-9491.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Luche H, Weber O, Nageswara Rao T, Blum C, Fehling HJ. Faithful activation of an extra-bright red fluorescent protein in “knock-in” Cre-reporter mice ideally suited for lineage tracing studies. Eur. J. Immunol. 2007;37:43–53. doi: 10.1002/eji.200636745. [DOI] [PubMed] [Google Scholar]
- 17.Reese TA, et al. Chitin induces accumulation in tissue of innate immune cells associated with allergy. Nature. 2007;447:92–96. doi: 10.1038/nature05746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Loke P, et al. IL-4 dependent alternatively-activated macrophages have a distinctive in vivo gene expression phenotype. BMC Immunol. 2002;3:7. doi: 10.1186/1471-2172-3-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Goodwin MM, et al. Histone deacetylases and the nuclear receptor corepressor regulate lytic-latent switch gene 50 in murine gammaherpesvirus 68-infected macrophages. J. Virol. 2010;84:12039–12047. doi: 10.1128/JVI.00396-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Gordon S, Martinez FO. Alternative Activation of Macrophages: Mechanism and Functions. Immunity. 2010;32:593–604. doi: 10.1016/j.immuni.2010.05.007. [DOI] [PubMed] [Google Scholar]
- 21.Vats D, et al. Oxidative metabolism and PGC-1beta attenuate macrophage-mediated inflammation. Cell Metab. 2006;4:13–24. doi: 10.1016/j.cmet.2006.05.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ohmori Y. Interleukin-4/STAT6 Represses STAT1 and NF-kappa B-dependent Transcription through Distinct Mechanisms. Journal of Biological Chemistry. 2000;275:38095–38103. doi: 10.1074/jbc.M006227200. [DOI] [PubMed] [Google Scholar]
- 23.Gray KS, Allen RD, Farrell ML, Forrest JC, Speck SH. Alternatively initiated gene 50/RTA transcripts expressed during murine and human gammaherpesvirus reactivation from latency. J. Virol. 2009;83:314–328. doi: 10.1128/JVI.01444-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wakeman BS, et al. Identification of alternative transcripts encoding the essential murine gammaherpesvirus lytic transactivator RTA. J. Virol. 2014 doi: 10.1128/JVI.03110-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Harty JT, Schreiber RD, Bevan MJ. CD8 T cells can protect against an intracellular bacterium in an interferon gamma-independent fashion. Proc Natl Acad Sci U S A. 1992;89:11612–11616. doi: 10.1073/pnas.89.23.11612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Jenkins SJ, et al. Local macrophage proliferation, rather than recruitment from the blood, is a signature of TH2 inflammation. Science. 2011;332:1284–1288. doi: 10.1126/science.1204351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Weck KE, Barkon ML, Yoo LI, Speck SH, HW IVV. Mature B cells are required for acute splenic infection, but not for establishment of latency, by murine gammaherpesvirus 68. J. Virol. 1996;70:6775–6780. doi: 10.1128/jvi.70.10.6775-6780.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.See supporting materials and methods in Science online.
- 29.Nakamura H, et al. Global changes in Kaposi's sarcoma-associated virus gene expression patterns following expression of a tetracycline-inducible Rta transactivator. J. Virol. 2003;77:4205–4220. doi: 10.1128/JVI.77.7.4205-4220.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wakeham K, et al. Parasite infection is associated with Kaposi's sarcoma associated herpesvirus (KSHV) in Ugandan women. Infect. Agents Cancer. 2011;6:15. doi: 10.1186/1750-9378-6-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kis LL, et al. STAT6 signaling pathway activated by the cytokines IL-4 and IL-13 induces expression of the Epstein-Barr virus-encoded protein LMP-1 in absence of EBNA-2: implications for the type II EBV latent gene expression in Hodgkin lymphoma. Blood. 2011;117:165–174. doi: 10.1182/blood-2010-01-265272. [DOI] [PubMed] [Google Scholar]
- 32.Kaplan MH, Schindler U, Smiley ST, Grusby MJ. Stat6 is required for mediating responses to IL-4 and for development of Th2 cells. Immunity. 1996;4:313–319. doi: 10.1016/s1074-7613(00)80439-2. [DOI] [PubMed] [Google Scholar]
- 33.Odegaard JI, et al. Macrophage-specific PPARgamma controls alternative activation and improves insulin resistance. Nature. 2007;447:1116–1120. doi: 10.1038/nature05894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Pesce JT, et al. Arginase-1-expressing macrophages suppress Th2 cytokine-driven inflammation and fibrosis. PLoS Pathog. 2009;5:e1000371. doi: 10.1371/journal.ppat.1000371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Durbin JE, Hackenmiller R, Simon MC, Levy DE. Targeted disruption of the mouse Stat1 gene results in compromised innate immunity to viral disease. Cell. 1996;84:443–450. doi: 10.1016/s0092-8674(00)81289-1. [DOI] [PubMed] [Google Scholar]
- 36.Nair MG, et al. Alternatively activated macrophage-derived RELM-{alpha} is a negative regulator of type 2 inflammation in the lung. J. Exp. Med. 2009;206:937–952. doi: 10.1084/jem.20082048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Warming S, Costantino N, Court DL, Jenkins NA, Copeland NG. Simple and highly efficient BAC recombineering using galK selection. Nucleic Acids Res.e. 2005;33:e36. doi: 10.1093/nar/gni035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Qian Z, Xuan B, Hong TT, Yu D. The full-length protein encoded by human cytomegalovirus gene UL117 is required for the proper maturation of viral replication compartments. J. Virol. 2008;82:3452–3465. doi: 10.1128/JVI.01964-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Adler H, Messerle M, Wagner M, Koszinowski UH. Cloning and mutagenesis of the murine gammaherpesvirus 68 genome as an infectious bacterial artificial chromosome. J. Virol. 2000;74:6964–6974. doi: 10.1128/jvi.74.15.6964-6974.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Adler H, Messerle M, Koszinowski UH. Virus reconstituted from infectious bacterial artificial chromosome (BAC)-cloned murine gammaherpesvirus 68 acquires wild-type properties in vivo only after excision of BAC vector sequences. J. Virol. 2001;75:5692–5696. doi: 10.1128/JVI.75.12.5692-5696.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Trapnell C, Pachter L, Salzberg SL. TopHat: discovering splice junctions with RNA-Seq. Bioinformatics. 2009;25:1105–1111. doi: 10.1093/bioinformatics/btp120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Trapnell C, et al. Transcript assembly and quantification by RNA-Seq reveals unannotated transcripts and isoform switching during cell differentiation. Nature Biotechnology. 2010;28:511–515. doi: 10.1038/nbt.1621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Subramanian A, et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci U S A. 2005;102:15545–15550. doi: 10.1073/pnas.0506580102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Takeshita S, Kaji K, Kudo A. Identification and characterization of the new osteoclast progenitor with macrophage phenotypes being able to differentiate into mature osteoclasts. J. Bone Miner. Res. 2000;15:1477–1488. doi: 10.1359/jbmr.2000.15.8.1477. [DOI] [PubMed] [Google Scholar]
- 45.Weck KE, et al. Murine γ-herpesvirus 68 causes severe large-vessel arteritis in mice lacking interferon-γ responsiveness: A new model for virus-induced vascular disease. Nat Med. 1997;3:1346–1353. doi: 10.1038/nm1297-1346. [DOI] [PubMed] [Google Scholar]
- 46.Tarakanova VL, Molleston JM, Goodwin M, Virgin HW, IV, et al. MHV68 complement regulatory protein facilitates MHV68 replication in primary macrophages in a complement independent manner. Virology. 2010;396:323–328. doi: 10.1016/j.virol.2009.10.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Goswami R, Kaplan MH. Gcn5 is required for PU.1-dependent IL-9 induction in Th9 cells. J. Immunol. 2012;189:3026–3033. doi: 10.4049/jimmunol.1201496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Renne R, et al. Lytic growth of Kaposi's sarcoma-associated herpesvirus (human herpesvirus 8) in culture. Nat Med. 1996;2:342–346. doi: 10.1038/nm0396-342. [DOI] [PubMed] [Google Scholar]
- 49.Yoo SM, Zhou F-C, Ye F-C, Pan H-Y, Gao S-J. Early and sustained expression of latent and host modulating genes in coordinated transcriptional program of KSHV productive primary infection of human primary endothelial cells. Virology. 2005;343:47–64. doi: 10.1016/j.virol.2005.08.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Lei X, et al. Regulation of NF-kappaB inhibitor IkappaBalpha and viral replication by a KSHV microRNA. Nat. Cell Biol. 2010;12:193–199. doi: 10.1038/ncb2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Majerciak V, et al. Kaposi's sarcoma-associated herpesvirus ORF57 interacts with cellular RNA export cofactors RBM15 and OTT3 to promote expression of viral ORF59. J. Virol. 2011;85:1528–1540. doi: 10.1128/JVI.01709-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Persson LM, Wilson AC. Wide-scale use of Notch signaling factor CSL/RBP-Jkappa in RTA-mediated activation of Kaposi's sarcoma-associated herpesvirus lytic genes. J. Virol. 2010;84:1334–1347. doi: 10.1128/JVI.01301-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Ye F-C, et al. Kaposi's sarcoma-associated herpesvirus latent gene vFLIP inhibits viral lytic replication through NF-kappaB-mediated suppression of the AP-1 pathway: a novel mechanism of virus control of latency. J. Virol. 2008;82:4235–4249. doi: 10.1128/JVI.02370-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.