Abstract
Background:
The current techniques for percutaneous coronary interventions (PCI) remain limited by restenosis. Recent studies have provided evidence of inflammation playing a role in the pathogenesis of cardiovascular disease.
Methods:
Whether inflammatory markers are predictors of subsequent restenosis were prospectively tested in 272 consecutive patients with angiographically proven coronary artery disease. Patients having undergone PCI at Chonnam National University Hospital, between Sept. 1999 and Mar. 2001, were divided into two groups according to the occurrence of restenosis on a follow-up coronary angiogram: Group I were patients with restenosis (n=99, 59.5±10.8 years, M:F=77:22) and Group II were those without restenosis (n=173, 58.8±10.2 years, M:F=131:42). The IgG seropositivity, cytomegalovirus (CMV) titers, C. pneumoniae, H. pylori and levels of C-reactive protein (CRP) were compared between the two groups.
Results:
There were no statistical differences in the seropositivity of the CMV IgG C. pneumoniae IgG and H. pylori IgG between the two groups (Groups I vs. II: 100 vs. 100%, 24.7 vs. 25.7% and 62.2 vs. 63.7%, respectively). Of the angiographic parameters, a low Thrombolysis In Myocardial Infarction (TIMI) flow (TIMI 0 or I) was more common in Group I than Group II (p=0.038). The patients with an elevated CRP (>0.5 mg/dL) were more common in Group I than Group II (57.6 vs. 36.4%, p=0.001), with the CRP values being higher in Group I than Group II (3.3±5.8 vs. 1.3±2.6 mg/dL, p=0.001). According to a multiple logistic regression analysis, the CRP was the only predictor of restenosis, with an odds ratio of 2.1169 (95% C.I. 1.2062–3.7154, p=0.009).
Conclusion:
The CRP value is the most important predictor of restenosis after PCI.
Keywords: Coronary Artery Disease, Restenosis, C-Reactive Protein, Percutaneous Coronary intervention
INTRODUCTION
It is well known that hypertension, diabetes, smoking, and hyperlipidemia are associated risk factors for atherosclerotic disease, including coronary artery disease, but in half the cases its etiology is hard to explain by only these conventional risk factors1). It has recently been reported that an inflammatory response is related to the development of atherosclerosis, which accelerates the rupture of atheromatous plaques and coronary artery thrombosis, thereby playing a major role in the pathogenesis of acute coronary syndrome2–4). It is now known that elevated levels of CRP in patients with coronary artery disease, increase the risk of future coronary events and that CRP itself is an independent risk factor for atherosclerotic disease such as myocardial infarction or stroke, occurring in healthy adults5–11). Factors stimulating the increase of CRP are not well known, but it has been suggested that chronic infections, especially with Chlamydia pneumoniae (C. pneumoniae), Cytomegalovirus, and Helicobacter pylori (H. pylori,) are associated4, 6, 8).
Percutaneous coronary intervention (PCI) has become established as a safe and effective method for the treatment of many coronary artery diseases, with a high primary success rate, but the problem of chronic restenosis following the procedure still remains to be resolved12–16). Cytomegalovirus (CMV) DNA has recently been reported to be found in the atheromatous plaques of coronary arteries, and previous infection with CMV has been suggested as an independent risk factor for restenosis17–19, 21), but further investigations into restenosis are required.
It has been reported from our laboratory, through the observation of associations between the markers of infection or inflammation, and coronary artery disease that the antibody titers of H. pylori were higher in patients with coronary artery disease, and the levels of CRP and ESR (erythrocyte sedimentation rate) were significantly elevated in patients with acute coronary syndrome, thereby explaining their role in the pathogenesis of acute coronary syndrome12).
The role of chronic infection or inflammation, in coronary artery disease was studied to ascertain the involvement of chronic infection (C. pneumoniae, CMV, H. pylori) in various coronary artery diseases, and to assess the relationship between chronic infection and the inflammatory markers, such as CRP to coronary restenosis.
MATERIALS AND METHODS
1. Subjects
Two hundred and seventy two patients (59.1±10.4 years of age, male : female = 208 : 64), with 345 lesions, who were first diagnosed as having coronary artery disease, and underwent PCI and follow-up coronary angiography, between March 1999 and March 2001, were divided into two groups according to the presence or absence of restenosis.
Patients were excluded from the study if they had undergone previous PCI or bypass surgery, even if they had undergone follow-up angiography, had other infectious or non-infectious inflammatory disease, underlying severe left ventricular dysfunction, had received or were receiving treatment for H. pylori eradication, or had no positive tests for infectious or inflammatory markers.
2. Methods
Immediately after admission, or early the following morning, fasting blood samples were collected prior to coronary angiography for titers of the IgG antibodies of C. pneumoniae, CMV, and H. pylori and the levels of CRP were also measured on the same day. From the findings of the follow-up coronary angiography, the subjects were divided into two groups; Group I, with restenosis, and Group II, without restenosis, and a comparatively analysis of the two groups was performed.
The tests for the anti-C. pneumoniae IgG antibodies were performed with C. pneumoniae IgG (Radim, Roma, Finland), and the results were interpreted as positive or negative. The tests for the anti-CMV IgG antibodies were performed using AxSYM (Abbott, Illinois, U.S.A.) and AxSYM CMV IgG reagent peak (Abbott, Illinois, U.S.A.), which were considered positive when the levels were greater than or equal to 15 AU/mL. The anti-H. pylori IgG antibodies were tested with Pyloriset EIA-G (Orion Diagnostica, Espoo, Finland), and were considered positive when the concentrations were greater than or equal to 300 U/mL. CRP was tested by Behring nephelometer analyzer II (Dade Behring Inc., Marburg, Germany) using N Latex CRP mono (Dade Behring Inc., Marburg, Germany) reagents and the normal reference range were regarded as less than 0.5 mg/dL.
3. Coronary angiography and intervention
Diagnostic coronary angiography was performed by puncturing the right (or left) femoral artery using the Seldinger method following local anesthesia of the inguinal area, or by insertion of a 6 French arterial sheath via the radial artery. On the coronary angiogram stenosis with an internal diameter greater than 50% was regarded as significant. From the coronary angiogram the location of the culprit arteries, Thrombolysis In Myocardial Infarction (TIMI) flow20) and the type of lesions were analyzed, according to the American College of Cardiology/American College of Cardiology (ACC/AHA) classification21). The reference vessel internal diameter, minimal luminal diameter, and luminal stenosis of the target arteries were measured using the on-line quantitative coronary angiogram program (Philips H5000, Netherlands), which was capable of edge detection. An elective or emergency PCI was performed according to the indications in patients showing stenosis greater than 50% in more than one of the three arteries from the coronary angiography.
4. Follow-up coronary angiography
Six month follow-up coronary angiography was taken in the subjects that had had undergone PCI, had a recurrence of symptoms, or had a positive test on exercise stress or other non-invasive tests during the follow-up period. Restenosis was defined as stenosis greater than 50% on the follow-up angiography in the arteries with luminal stenosis of less than 50% immediately after the PCI.
5. Statistical analysis
All data were described as the mean standard deviation. The nominal variables were analyzed by Chi-squared or Fisher's exact tests, and the various continuous variables compared by t-and Chi-squared tests. A p-value less than 0.05 was considered significant. Also, predictive factors were sought using a multiple, stepwise logistic regression analysis, after adjustment for age, sex, and risk factors of coronary artery disease.
RESULTS
1. Baseline clinical characteristics
Two hundred and seventy two patients who had undergone PCI, measurement of inflammatory markers, and follow-up coronary angiography, were divided into two groups; Group I, 99 patients with restenosis (59.56±10.82 years, 77 males and 22 females) and Group II, 173 patients without restenosis (58.89±10.22 years, 131 males and 42 females). Follow-up coronary angiography was performed after 5.5±1.5 and 6.2±5.5 months in groups I and II, respectively; no statistical differences were found between the two groups. The restenosis rates were 36.4 and 34.8% in relation to the number of patients (99 patients out of the total 272 patients) and lesions (101 arteries out of the total 290 arteries where PCI was performed), respectively. The initial clinical diagnoses, with significantly higher restenosis rates, were acute myocardial infarction (MI) and unstable angina pectoris rather than stable angina and old MI (p=0.023). Risk factors of atherosclerosis, such as smoking, diabetes, hypertension and hyperlipidemia, were not different between the two groups (Table 1).
Table 1.
Clinical characteristics of Groups I and II
| Group I | Group II | p value | |
|---|---|---|---|
| Patient number | 99 | 173 | |
| Age (years) | 59.6±10.8 | 58.9±10.2 | NS |
| Sex (%) | NS | ||
| Male | 77 (77.8) | 131 (75.5) | |
| Female | 22 (22.2) | 42 (24.3) | |
| Clinical diagnosis (%) | 0.023 | ||
| Acute myocardial infarction | 58 (58.6) | 80 (46.2) | |
| Unstable angina | 34 (34.3) | 57 (32.9) | |
| Stable angina | 6 (6.1) | 28 (16.2) | |
| Old myocardial infarction | 1 (1.0) | 8 (4.6) | |
| Risk factors (%) | |||
| Smoking | 57 (57.6) | 89 (54.3) | NS |
| Hypertension | 45 (45.5) | 56 (34.4) | NS |
| Diabetes meliitus | 23 (23.2) | 32 (19.4) | NS |
| Hyperlipidemia | 25 (25.8) | 41 (26.8) | NS |
| Ejection fraction (%) | 55.5±12.1 | 57.7±11.7 | NS |
| Number of diseased vessel(%) | NS | ||
| one vessel disease | 72 (72.1) | 140 (80.9) | |
| multivessel disease | 27 (27.3) | 33 (19.1) |
2. Associations between infection and post-PCI restenosis
The seropositivity for anti-C. pneumoniae IgG antibodies were 27.3 (27/99) and 26% (45/173) in groups I and II, respectively, with no differences found between the two groups. The seropositivity for anti-CMV IgG antibodies was 100% in both the groups and that for Anti-H. pylori IgG antibodies was 61.6% (61/99) and 63.6% (110/173) in groups I and II, respectively, with no statistical differences found. Titers for the anti-H. pylori antibodies were not different between the two groups (852.4±1332.4 vs. 809.4±931.7 U/mL, p=NS, Table 2).
Table 2.
Comparisons of the seropositivity of the infectious and inflammatory markers between Groups I and Group II
| Group I (n=99) | Group II (n=173) | p value | |
|---|---|---|---|
| Chlamydia pneumoniae (%) | 27 (27.3) | 45 (26.0) | NS |
| Cytomegalovirus (%) | 99 (100) | 173 (100) | NS |
| Helicobacter pylori (%) | 61 (61.6) | 110 (63.6) | NS |
3. Associations between C-reactive protein and restenosis
Seropositivity of CRP was significantly higher in group I than II, at 57.6 (57/99) vs. 36.4% (63/173) (p=0.001) (Table 2). The serum levels were significantly higher in group I than II (3.38±5.80 vs. 1.36±2.62 mg/dL) (p=0.001) (Figure 1, Table 3). When only the patients with acute coronary syndrome were included, the CRP seropositivity was significantly higher in group I than II (60.9 (56/92) vs. 39.1% (36/92) (p=0.004)), and the serum levels were also significantly higher In group I than II, at 3.60±5.97 vs. 1.55±2.86 mg/dL respectively (p=0.003).
Figure 1.
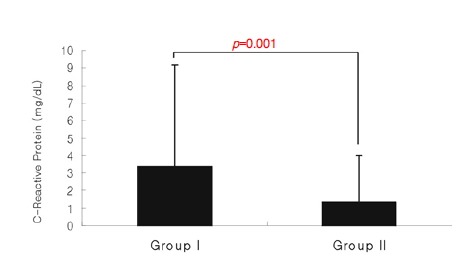
Values of C-reactive protein were significantly higher in the patients in Group I than in the patients in Group II.
Table 3.
Comparisons of the seropositivity and concentrations of C-reactive protein between Groups I and Group II
| Group I (n=99) | Group II (n=173) | p value | |
|---|---|---|---|
| C-reactive protein (mg/dL) | 3.385.80 | 1.362.62 | 0.001 |
| C-reactive protein positivity | 57 (57.6) | 63 (36.4) | 0.001 |
From the multiple logistic regression analysis the CRP was found to be an independent risk factor for the development of restenosis (p=0.009), even after adjustment for sex, age, smoking, hypertension, diabetes, hyperlipidemia, and seropositivity of H. pylori and C. pneumoniae, and a relative risk of 2.1169, with a 95% confidence interval of 1.2072–3.7154 (Table 7).
Table 7.
Analysis of predictive factors for the restenosis
| p value | OR (95% C.I.) | |
|---|---|---|
| C-reactive protein | 0.009 | 2.1169 (1.2062–3.7154) |
| sex | 0.5477 | 1.2566 (0.5968–2.6460) |
| smoking | 0.9602 | 0.9843 (0.5288–1.8323) |
| Diabetes mellitus | 0.2928 | 1.4409 (0.7296–2.8457) |
| Hypertension | 0.0602 | 1.8957 (1.0632–3.3802) |
| Dyslipidemia | 0.5003 | 0.8016 (0.4213–1.5250) |
| Chlamydia pneumoniae (+) | 0.9338 | 0.9733 (0.5144–1.8418) |
| Helicobacter pylori (+) | 0.9863 | 0.9951 (0.5660–1.7495) |
| age | 0.523 | 0.775 (0.490–1.226) |
| TIMI flow | 0.0691 | 0.6849 (0.4781–0.9812) |
| MLD (before PCI) | 0.3194 | 1.048 (0.609–1.802) |
| MLD (after PCI) | 0.7156 | 1.241 (0.794–1.940) |
C.I., confidence interval; OR, odds ratio; MLD, minimal luminal diameter; PCI, percutaneous coronary intervention
4. Comparison with coronary angiography
From the coronary angiography a total of 345 lesion arteries, in 272 patients, were found, of which PCI was performed in 290 arteries; 137 in the left anterior descending artery, 75 in the right coronary artery and 78 in the left circumflex artery. The TIMI flows were 0 in 44, I in 17, II in 103 and III in 104 cases, with the lesion types, according to the ACC/AHA classification, being A in 21, B1 in 145, and C in 43 cases. There were no significant differences in the distribution of the culprit arteries, number of arteries, or types of lesions between the two groups, but the TIMI flow was significantly lower in group I (p=0.038) (Table 4). However, this value was not statistically significant on the multiple regression analysis following adjustment for the variables, such as age, sex, smoking, diabetes, hypertension, hyperlipidemia, and clinical diagnosis.
Table 4.
Angiographic and procedural characteristics in Groups I and II
| Group I (n=101) | Group II (n=189) | p value | |
|---|---|---|---|
| Target lesion | NS | ||
| Left anterior descending artery | 51 (50.5) | 86 (45.5) | |
| Right coronary artery | 22 (21.8) | 53 (28.0) | |
| Left circumflex artery | 28 (27.7) | 50 (26.5) | |
| TIMI flow grade | 0.038 | ||
| 0 | 24 (25.3) | 20 (11.6) | |
| I | 5 (5.3) | 12 (6.9) | |
| II | 33 (34.7) | 70 (40.5) | |
| III | 33 (34.7) | 71 (41.0) | |
| ACC/AHA types | NS | ||
| A | 6 (5.9) | 15 (7.9) | |
| B1 | 47 (46.5) | 98 (51.9) | |
| B2 | 31 (30.7) | 50 (26.5) | |
| C | 17 (16.8) | 26 (13.8) | |
| Reference diameter (mm) | 2.88±0.65 | 2.87±0.53 | NS |
| % percent diameter stenosis | 89.60±11.53 | 90.86±9.30 | NS |
| Minimal luminal diameter (mm) | |||
| Before PCI | 0.43±0.40 | 0.50±0.40 | NS |
| After PCI | 2.63±0.55 | 2.59±0.53 | NS |
| 6-month follow-up | 0.55±0.34 | 2.32±0.62 | 0.0001 |
| Stent implantation (%) | 66 (66.7%) | 118 (68.2%) | NS |
TIMI, Thrombolysis In Myocardial Infarction; ACC/AHA, American College of Cardiology/American Heart Association; PCI, percutaneous coronary intervention.
5. Correlation between infection and inflammation
The CRP seropositivities for the patients positive and negative for the anti-H. pylori IgG antibodies were 45.0 (77/171) and 42.6% (43/101), with serum levels of 1.96±3.42 and 2.22±4.63 mg/dL, respectively, with no differences found between the two groups. The CRP seropositivities for the patients positive and for the anti-C. Pneumoniae IgG antibodies were 43.1 (31/72) and 44.5% (89/200), with serum levels of 2.19±4.60 and 1.94±3.38 mg/dL, respectively, with no differences found between the two groups (Table 5). The Pearson correlation showed no significance between the anti-H. pylori IgG antibody titers and the CRP levels (r=0.272, p=0.637). The anti-CMV IgG antibody titers were divided, into the high-titer and the low-titer groups, according to the serum levels (>250 and <250 AU/mL, respectively), and were compared with the restenosis rate, CRP seropositivity, and CRP levels, but no significant differences were found between the two groups (Table 6). The median anti-H. pylori IgG antibody level was 411 U/mL, and when patients were subgrouped on the basis of this value, into the high- and low-titer groups (>411 and <411 U/mL, respectively) and compared with the restenosis rate, CRP seropositivity, and CRP levels, no statistical significance was found between the two groups (Table 6).
Table 5.
Association of C-reactive protein (CRP) and IgG seropositivity to Chlamydia pneumoniae and Helicobacter pylori
| C. pneumoniae (+) | C. pneumoniae (−) | p value | |
|---|---|---|---|
| Number (%) | 72 (26.5%) | 200 (73.5%) | NS |
| C-reactive protein>0.5 mg/dL | 43.1% (31/72) | 44.5% (89/200) | NS |
| C-reactive protein (mg/dL) | 2.02±3.43 | 2.12±4.43 | NS |
| Restenosis rate | 37.5% (27/72) | 36.0% (72/200) | NS |
| H. pylori (+) | H. pylori (−) | ||
| Number (%) | 171 (62.9%) | 101 (37.1%) | NS |
| C-reactive protein>0.5 mg/dL | 45.0% (77/171) | 42.6% (43/101) | NS |
| C-reactive protein (mg/dL) | 1.96±3.42 | 2.22±4.63 | NS |
| Restenosis rate | 35.7% (61/171) | 37.6% (38/101) | NS |
Table 6.
Association of C-reactive protein, titer of Cytomegalovirus, Helicobacter pylori and restenosis
| Cytomegalovirus High Titer | Cytomegalovirus Low Titer | p value | |
|---|---|---|---|
| Number (%) | 134 (49.3%) | 138 (50.7%) | NS |
| C-reactive protein>0.5 mg/dL | 38.8% (52/134) | 49.3% (68/138) | NS |
| C-reactive protein (mg/dL) | 1.83±4.03 | 2.35±4.32 | NS |
| Restenosis rate | 32.8% (44/134) | 39.9% (55/138) | NS |
| Helicobacter pylori High Titer | Helicobacter pylori Low Titer | ||
| Number (%) | 136 (50%) | 136 (50) | NS |
| C-reactive protein>0.5 mg/dL | 41.9% (57/136) | 46.3% (63/136) | NS |
| C-reactive protein (mg/dL) | 2.19±4.77 | 1.99±3.52 | NS |
| Restenosis rate | 35.3% (48/136) | 37.5% (51/136) | NS |
CMV high titer, >250 AU/mL; low titer, <250 AU/mL, H. pylori high titer, >411 U/mL; low titer, <411 U/mL
DISCUSSION
The current study has shown that the serum levels of CRP following PCI were significantly higher in those patients with restenosis and that it was an independent risk factor of restenosis following adjustment for the other variables. Conversely, there was also no significant correlation between restenosis and infections with CMV, H. pylori, and C. pneumoniae, or between restenosis and the anti-CMV and -H. pylori IgG antibodies titers. Furthermore, increased levels of CRP had no correlation with the seropositivity of the IgG antibodies for H. pylori, C. pneumoniae, CMV or with the anti-CMV and -H. pylori antibody titers.
PCI has become established as a safe and effective treatment for coronary artery atherosclerosis, but chronic restenosis following PCI remains to be resolved. The experimental or clinical mechanisms suggested for restenosis are the proliferation of smooth muscle cells, neointimal hyperplasia, vascular remodeling, and thrombosis19–22).
There have been many studies on the relationship of inflammation to atherosclerosis and coronary artery disease, but its accurate role remains to be clarified. However, inflammation is an important factor in determining the stability of atherosclerotic atheromatous plaques22), and the results of this study confirms the reports from many large-scale prospective epidemiologic studies that the CRP level is associated with the increased incidences of future cardiac events occurring in patients with a history of ischemic heart disease5, 7, 9, 24), stable angina pectoris25), or myocardial infarction26).
A report that aspirin27) and statin26) reduced the risks of future cardiac events in patients with increased CRP levels implies that inflammation has an effect on the disease course, independently of conventional risk factors of coronary artery disease, supporting the currently proposed hypotheses. CMV DNA sequences have been discovered in large parts of the tissues of restenotic coronary lesions28), and besides the reports of CMV seropositivity being a significant independent risk factor for restenosis17), increased levels of CRP have also been reported as an independent risk factor for mortality from coronary artery disease28). In the current study, all the subjects were positive for the anti-CMV IgG antibody, and divided into the high- and low-titer groups according to their titer values. When this factor was compared with the restenosis rate there were no significant statistical differences were found, which was consistent with the results of Manegold, et al30).
Since the report of Saikku et al31), in 1988, showing the incidence of seropositivity of anti-C. pneumoniae antibodies were high in patients with coronary artery disease; there have been other seroepidemiologic studies on the associations between anti-C. pneumoniae antibodies and coronary artery disease. Gupta et al32, 33), in 1997, reported that increased anti-C. pneumoniae antibody titers were related to cardiovascular complications, with the eradication of C. pneumoniae, using azithromycin, not only decreased the antibody titers, but also the cardiovascular complications. Conversely, Gurfinkel et al reported that roxithromycin failed to decrease the complications of coronary artery disease in patients with acute non-Q-wave coronary syndrome34). However, a recent prospective epidemiologic study revealed that 300 mg of roxithoromycin administered two hours after coronary stenting, for four weeks, significantly decreased the rate of restenosis in patients with high anti-C. pneumoniae antibody titers35). Neointimal formation, due to injury following PCI, is induced by inflammation, and in animal experiments has been suppressed by anti-inflammatory cytokines, such as interleukin 1036), tissue factor pathway inhibitor-136), antibodies of beta2-leukocyte integrin Mac-1 and by suppression of ICAM-1-supported adhesion through genetic disruption of Mac-138). Cellular infection by C. pnueumoniae plays an important role in the neointimal formation through the upregulation of these surface receptors, and the use of antibiotics can inhibit neointimal formation by inhibiting the vascular infection. In this study, owing to the lack of data on the anti-C. pneumoniae antibody titers, their effects on restenosis could not be confirmed. Therefore, further investigation would seem necessary. In relation to the seropositivity of the antibodies, there were no significant differences between the two groups, and no correlation with the inflammatory markers was shown. It would therefore appear that other factors causing inflammation, or the effects of inflammation itself on restenosis, should be considered.
In a study conducted at our laboratory, the anti-H. pylori antibody titers were increased in patients with coronary artery disease, with 64% seropositivity of its antibodies12). In this study, the rates were 61 and 63.3% in groups I and II, respectively, with no significant difference between the groups. Furthermore, when the patients were divided into the high-and low-titer groups, on the basis of the median level of the titers, there was also no significant difference between the groups. It has been proposed that H. pylori infection was a strong risk factor of restenosis39), but studies on its relation to restenosis have not been sufficient, which has resulted in various conflicting claims relating to this factor40). From the results of this study other conclusions could be drawn if new methods should be used to assess the extent of infection, even though the rate of restenosis and the seropositivity of IgG antibodies or the titers of inflammatory markers were not related.
Many studies are being performed on the restenosis-reducing effects of statin or aspirin, based on the result that elevated CRP levels are an independent risk factor for restenosis. It is also speculated that a better understanding of the degrees of inflammation could serve to prevent future restenosis.
REFERENCES
- 1.Grundy SM, Pasternak R, Greenland P, Smith S, Fuster V. Assessment of cardiovascular risk by use of multiple risk factor assessment equations: A statement for healthcare professionals from the American Heart Association and the American College of Cardiology. Circulation. 1999;100:1481–1492. doi: 10.1161/01.cir.100.13.1481. [DOI] [PubMed] [Google Scholar]
- 2.Alexander RW. Inflammation and coronary artery disease. N Engl J Med. 1994;331:468–469. doi: 10.1056/NEJM199408183310709. [DOI] [PubMed] [Google Scholar]
- 3.Van der Wal AC, Becker AE, Van der Loos CM, Das PK. Site of intimal rupture or erosion of thrombosed coronary atherosclerotic plaques is characterized by an inflammatory process irrespective of the dominant plaque morphology. Circulation. 1994;89:36–44. doi: 10.1161/01.cir.89.1.36. [DOI] [PubMed] [Google Scholar]
- 4.Libby P, Ridker PM. Novel inflammatory markers of coronary risk. Circulation. 1999;100:1148–1150. doi: 10.1161/01.cir.100.11.1148. [DOI] [PubMed] [Google Scholar]
- 5.Liuzzo G, Biasucci LM, Gallimore JR, Grillo RL, Rebuzzi AG, Pepys MB, Maseri A. The prognostic value of C-reactive protein and serum amyloid A protein in severe unstable angina. N Engl J Med. 1994;331:417–424. doi: 10.1056/NEJM199408183310701. [DOI] [PubMed] [Google Scholar]
- 6.Ridker PM. High-sensitivity C-reactive protein. Circulation. 2001;103:1813–1818. doi: 10.1161/01.cir.103.13.1813. [DOI] [PubMed] [Google Scholar]
- 7.Toss H, Lindahl B, Siegbahn A, Wallentin L. Prognostic influence of increased fibrinogen and C-reactive protein levels in unstable coronary artery disease. Circulation. 1997;96:4204–4210. doi: 10.1161/01.cir.96.12.4204. [DOI] [PubMed] [Google Scholar]
- 8.Anderson JL, Carlquist JF, Muhlestein JB, Horne BD, Elmer SP. Evaluation of C-reactive protein, an inflammatory marker, and infectious serology as risk factors for coronary artery disease and myocardial infarction. J Am Coll Cardiol. 1998;32:35–41. doi: 10.1016/s0735-1097(98)00203-4. [DOI] [PubMed] [Google Scholar]
- 9.Morrow DA, Rifai N, Antman EM, Weiner DL, Mccabe CH, Cannon CP, Braunwald E. C-reactive protein is a potent predictor of mortality independently of and in combination with troponin T in acute coronary syndromes: A TIMI11A sub-study. J Am Coll Cardiol. 1998;31:1460–1465. doi: 10.1016/s0735-1097(98)00136-3. [DOI] [PubMed] [Google Scholar]
- 10.Abdelmouttaleb I, Danchin N, Illardo C, Aimone I, Angioü M, Lozniewski A, Lonbinoux J, Le Faon A, Greant JL. C-reactive protein and coronary artery disease: Additional evidence of the implication of an inflammatory process in acute coronary syndromes. Am Heart J. 1999;137:346–351. doi: 10.1053/hj.1999.v137.92052. [DOI] [PubMed] [Google Scholar]
- 11.Koenig W, Sund M, Frohlich M, Fischer HG, Lowel H, Doring A, Hutchinson WL, Pepys MB. C-reactive protein, a sensitive marker of inflammation, predicts future risk of coronary heart disease in initially healthy middle-aged man: Results from the MONICA (Monitoring trends and determinants in cardiovascular disease) Ausburg cohort study, 1984 to 1992. Circulation. 1999;19:237–242. doi: 10.1161/01.cir.99.2.237. [DOI] [PubMed] [Google Scholar]
- 12.Kim KH, Jeong MH, Shin JH, Joo SB, Kim W, Lee SW, Kim KH, Kim NH, Cho JH, Park JC, Ahn YK, Na KJ, Cho JG, Ahn BH, Park JC, Kang JC. The Role of Chronic infection and Inflammation in Korean Patients with Coronary Artery Disease. Korean Circulation J. 2000;30:1007–1016. [Google Scholar]
- 13.Jeong MH, Kim SH, Ahn YK, Cho JG, Park JC, Na KJ, Kang JC†. Predictive factors for the second restenosis after coronary interventions. Cathet Cardiovasc intervent. 2000;50:34–39. doi: 10.1002/(sici)1522-726x(200005)50:1<34::aid-ccd7>3.0.co;2-l. [DOI] [PubMed] [Google Scholar]
- 14.Jeong MH, Ahn YK, Cho JG, Park JC, Na KJ, Kang JC. Successful coronary stent implantation using local nitric oxide donor delivery. J Interven Cardiol. 2000;13:191–196. [Google Scholar]
- 15.King SB., III Role of new technology in balloon angioplasty. Circulation. 1991;84:2574–2579. doi: 10.1161/01.cir.84.6.2574. [DOI] [PubMed] [Google Scholar]
- 16.Blum A, Giladi M, Weinberg M, Kaplan G, Pasternack H, Laniado S, Miller H. High anth-cytomegalovirus IgG antibody titer is associated with coronary artery disease and may predict post-coronary balloon angioplasty restenosis. Am J Cardiol. 1998;81:866–868. doi: 10.1016/s0002-9149(98)00019-8. [DOI] [PubMed] [Google Scholar]
- 17.Zhou YF, Leon MB, Waclawiw MA, Popma JJ, Yu ZX, Finkel T, Epstein SE. Association between prior cytomegalovirus infection and the risk of restenosis after coronary atherectomy. N Engl J Med. 1996;335:624–630. doi: 10.1056/NEJM199608293350903. [DOI] [PubMed] [Google Scholar]
- 18.Carlsson J, Miketic S, Mueller KH, Brom J, Von Essen R, Tebbe U. Previous cytomegalovirus or Chlamydia pneumoniae infection and risk of restenosis after percutaneous transluminal coronary angioplasty. Lancet. 1997;350:1225–1232. doi: 10.1016/S0140-6736(05)63456-3. [DOI] [PubMed] [Google Scholar]
- 19.Schwartz RS, Holmes DR, Topol EJ. The restenosis paradigm revisited : and alternative proposal for cellular mechanism. J Am Coll Cardiol. 1992;20:1284–1293. doi: 10.1016/0735-1097(92)90389-5. [DOI] [PubMed] [Google Scholar]
- 20.TIMI study group The Thrombolysis In Myocardial Infarction (TIMI) Trial. N Engl J Med. 1985;312:932–936. doi: 10.1056/NEJM198504043121437. [DOI] [PubMed] [Google Scholar]
- 21.Scott NA, Cipolla GD, Dunn B, Martin FH, Simonet L, Wilcox JN. Identification of a potential role for the adventitia in vascular lesion formation after balloon overstretch injury of porcine coronary proliferative restenosis model. J Am Coll Cardiol. 1996;24:2178–2187. doi: 10.1161/01.cir.93.12.2178. [DOI] [PubMed] [Google Scholar]
- 22.Bauters C, Meurice T, Hamon M, Lablanche JM, Bertrand ME. Mechanisms and prevention of restenosis: from experimental models to clinical practice. Cardiovasc Res. 1996;31:835–846. [PubMed] [Google Scholar]
- 23.Ross R. Atherosclerosis: an inflammatory disease. N Engl J Med. 1996;340:115–126. doi: 10.1056/NEJM199901143400207. [DOI] [PubMed] [Google Scholar]
- 24.Lindahl B, Toss H, Siegbahn A, Venge P, Wallentin L. Markers of myocardial damage and inflammation in relation to long-term mortality in unstable coronary artery disease. FRISC study Group. FRagmin during Instability in Coronary Artery Disease. N Engl J Med. 2000;19343:1139–1147. doi: 10.1056/NEJM200010193431602. [DOI] [PubMed] [Google Scholar]
- 25.Haverkate F, Thompson SG, Pyke SD, Gallimore JR, Pepys MB. Production of C-reactive protein and risk of coronary events in stable and unstable angina. European Concerted Action on Thrombosis and Disabilities Angina Pectoris Study Group. Lancet. 1997;15349:462–446. doi: 10.1016/s0140-6736(96)07591-5. [DOI] [PubMed] [Google Scholar]
- 26.Ridker PM, Rifai N, Pfeffer MA, Sacks FM, Moye LA, Goldman S, Flaker GC, Braunwald E. Inflammation, pravastatin, and the risk of coronary events after myocardial infarction in patients with average cholesterol levels. Cholesterol and Recurrent Events (CARE) Investigators. Circulation. 1998;198:839–844. doi: 10.1161/01.cir.98.9.839. [DOI] [PubMed] [Google Scholar]
- 27.Ridker PM, Cushman M, Stampfer MJ, Tracy RP, Hennekens CH. Inflammation, aspirin, and the risk of cardiovascular disease in apparently healthy men. N Engl J Med. 1997;3336:973–979. doi: 10.1056/NEJM199704033361401. [DOI] [PubMed] [Google Scholar]
- 28.Speir E, Modali R, Huang ES, Leon MB, Shawl F, Finkel T, Epstein SE. Potential role of human cytomegalovirus and p53 interaction in coronary restenosis. Science. 1994;265:391–394. doi: 10.1126/science.8023160. [DOI] [PubMed] [Google Scholar]
- 29.Muhlestein JB, Home BD, Cariquist JF, Madsen TE, Bair TL, Pearson RR, Anderson JL. Cytomegalovirus seropositivity and C-reactive protein have independent and combined predictive value for mortality in patients with angiographically demonstrated coronary artery disease. Circulation. 2000;102:1917–1923. doi: 10.1161/01.cir.102.16.1917. [DOI] [PubMed] [Google Scholar]
- 30.Manegold C, Alwazzeh M, Jablonowski H, Adams O, Medve M, Seidlitz B, Hoiland U, Haussinger D, Strauer BE, Heinzen MP. Prior cytomegalovirus infection and the risk of restenosis after percutaneous transluminal coronary balloon angioplasty. Circulation. 1999;99:1290–1294. doi: 10.1161/01.cir.99.10.1290. [DOI] [PubMed] [Google Scholar]
- 31.Saikku P, Leinonen M, Mattila K, Ekman MR, Nieminen MS, Makera PH, Huttunen JK, Valtonen V. Serological evidence of an association of a novel Chlamydia, TWAR, with chronic coronary heart disease. Lancet. 1988;29:983–986. doi: 10.1016/s0140-6736(88)90741-6. [DOI] [PubMed] [Google Scholar]
- 32.Gupta S. Chlamydia pneumoniae, monocyte activation and azithromycin in coronary heart disease. Am Heart J. 1999;138:539–541. doi: 10.1016/s0002-8703(99)70296-6. [DOI] [PubMed] [Google Scholar]
- 33.Gupta S, Leatham EW, Carrington M, Mendall MA, Kaski JC, Camm AJ. Elevated Chalymydia pneumonia antibodies, cardiovascular events, and azithromycin in male survivors of myocardial infarction. Circulation. 1997;96:404–407. doi: 10.1161/01.cir.96.2.404. [DOI] [PubMed] [Google Scholar]
- 34.Gurfinkel E, Bozovich G, Beck E, Testa E, Livellara B, Mautner B. Treatment with the antibiotic roxithromycin in patients with acute non-Q-wave coronary syndromes: the final report of the ROXIS study. Eur Heart J. 1999;20:121–127. doi: 10.1053/euhj.1998.1283. [DOI] [PubMed] [Google Scholar]
- 35.Neumann FJ, Kastrati A, Miethke T, Gisela PM, Mehilli J, Valina C, jogethaei N, da Conta Clarissa P, Wagner H, Sohomig A. Treatment of Chlamydia pneumoniae infection with roxithromycin and effect on neointima proliferation after coronary stent placement (ISAR-3): a randomized, double-blind, placebo- controlled trial. Lancet. 2001;357:2085–2089. doi: 10.1016/s0140-6736(00)05181-3. [DOI] [PubMed] [Google Scholar]
- 36.Feldman LJ, Aguirre L, Ziol M, Bridon JP, Novo N, Michael JB, Steg PG. Interleukin-10 inhibits intimal hyperplasia after angioplasty or stent implantation in hypercholesterolemic rabbits. Circulation. 2000;101:908–916. doi: 10.1161/01.cir.101.8.908. [DOI] [PubMed] [Google Scholar]
- 37.Oltrona L, Speidel CM, Recchia D, Wickline SA, Eisenberg PR, Abendschein DR. Inhibition of tissue factor-mediated coagulation markedly attenuates stenosis after balloon-induced arterial injury in minipings. Circulation. 1997;96:646–652. doi: 10.1161/01.cir.96.2.646. [DOI] [PubMed] [Google Scholar]
- 38.Simon Dl, Dhen Z, Seifert P, Edelman ER, Ballantyne CM, Rogers C. Decreased neointimal formation in Mac-a (−/−) mice reveals a role for inflammation in vascular repair after angioplasty. J Clin Invest. 2000;105:293–300. doi: 10.1172/JCI7811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Buffon A, Liuzzo GM, Caligiuri G, Grillo RL, Angiolillo DJ, Summaria F. Association between prior Helicobacter pylori infection and the risk of restenosis after coronary angioplasty [abstract] Circulation. 1997;96:650. [Google Scholar]
- 40.Carlsson J, Miketic S, Brom J, Ross R, Bachmann H, Tebbe U. Prior cytomegalovirus, Chlamydia pneumoniae or Helicobacter pylori infection and the risk of restenosis after percutaneous transluminal coronary angioplasty. Int J Cardiol. 2000;73:165–171. doi: 10.1016/s0167-5273(00)00216-3. [DOI] [PubMed] [Google Scholar]


