Abstract
The effect of probenecid on the transport of tetraethylammonium (TEA) was investigated in rabbit reanal cortical slices in an attempt to ascertain the interaction of organic anion with the organic cation transport system in proximal tubule. Probenecid reversibly inhibited TEA uptake by cortical slices in a dose-dependent manner over the concentration range of 1 and 5 mM. The efflux of TEA was not affected by the presence of 3 mM probenecid. Kinetic analysis indicated that probenecid decreased Vmax without a significant change in Km. Probenecid inhibited significantly tissue oxgen consumption at concentrations of 3 and 5 mM. However, probenecid did not significantly reduce TEA uptake in brush border and basolateral membrane vesicles prepared from renal cortex even at higher concentration of 10 mM.
These results indicate that probenecid reduces TEA uptake in cortical slices by inhibiting the tissue metabolism rather than by the interaction with the organic cation transporter.
Keywords: Probenecid, TEA (Tetraethylammonium)
INTRODUCTION
Many organic cations are actively secreted by renal proximal tubule of the mammalian kidney1,2). This secretion requires uptake from blood into the cell across the basolateral membrane of the proximal tubule epithelium and subsequent exit into the urine across the brush border mombrane. Studies with membrane vesicles isolated from the kidney cortex have consistently identified that organic cations are actively transported by a H+/organic cation exchange process across the brush border membrane3–6). Transport of organic cation across the basolateral membrane has also been demonstrated to be an active process in studies with intact proximal tubule7,8), although studies with basolateral membrane vesicles showed a facilitated diffusion process9).
Many studies have demonstrated that the organic cation transport system is specific to organic cations and is clearly different from the organic anion transport system1,10,11). However, several studies showed that probenecid, the classical competitve inhibitor of organic anion transport, also inhibits the renal transport of cimetidine, an organic cation, in in vivo and in vitro studies12,13). Recently, Hsyu et al14) demonstrated that probenecid inhibits N’-methylnicotinamide (NMN) uptake in a competitive fashion in renal brush border membrane vesicles, which suggests interaction of organic anion with the organic cation transport system.
This study, thus, was carried out to examine whether probenecid interacts with the organic cation transport system in basolateral membrane of proximal tubule. The effect of probenecid on TEA uptake was determined in slices and membrane vesicles prepared from rabbit kidney cortex.
MATERIALS AND METHODS
1. Slice Experiments
New Zealand white rabbits of both sexes, weighing 1.5–2.5 Kg, were used. Kidneys were removed just after the abdominal cavity was opened, and the renal artery was immediately perfused with an ice-cold isotonic saline solution containing 140 mM NaCl, 10 mM KCl and 1.5 mM CaCl2 to remove as much blood as possible. Thin (0.4–0.5 mm thick) slices of renal cortex were prepared using a Stadie-Riggs microtome and were stored in an ice-cold modified Cross-Taggart medium containing 130 mM NaCl, 10 mM KCl, 1.5 mM CaC12 and 20 mM Tris/HCl (pH 7.8). Approximately 50 mg (wet wt.) of slices were then transferred into a 20 ml beaker containing 4 ml of the modified Cross-Taggart medium and incubated with 10 μM 14C-TEA (Amersham, Alrington Heights, IL). The incubation was carried out in a Dubnoff metabolic shaker at 25°C under a 100% oxygen atmophere. Unless stated otherwise, the incubation period was 60 min.
Upon completion of incubation, slices were quickly removed from the beaker, blotted and weighed. After the tissus was solubilized in 1 N NaOH, the solution was neutralized with HCl. Aliquots of incubation media were treated similarly. The solution was then pipetted into a scintillation vial containing Aquasol (New England Nuclear) and the radioactivity was determined using a liquid scintillation counter (Packard Tricarb 300 C). The TEA uptakes by renal slices were expressed as the slice to medium (S/M) ratio: the concentration of the compound in the tissue (mole/g wet tissue) divided by that in the medium (mole/ml medium).
In the kinetic analyses of active TEA uptake, the total and passive uptakes of PAH were measured separately, and the difference was taken as an active uptake15). For the determination of total uptake, slices were preincubated for 60 min under a 100% oxygen atmosphere and were then transferred for a 10-min incubation to the same medium containing various concentrations of TEA. The passive uptake was measured similarly but under a 100% nitrogen atmosphere in a medium containing 10 mM choline chloride.
The efflux of TEA from renal cortical slices was determined by modification of the technique of Farah et al.16). Slices were first loaded with TEA by incubating them in a medium containing 50 μM 14C-TEA for 60 min after which they were rinsed for 20 sec in a TEA-free medium in order to remove TEA adhering to the tissue surface. The slices were then transferred at 1 min intervals through a series of 30 beakers containing a TEA-free modified Cross-Taggart medium at 25°C. The experiment was performed in a Dubnoff metabolic shaker with a hinged plexiglas cover and 100% oxygen atmosphere. The quantity of TEA collected from each beaker as well as the amount of the compound remaining in the tissue was used to construct the efflux curve and to calculate rate constant.
2. Oxygen Consumption Measurement
The oxygen consumption of renal slices was measured with an oxygen monitor (Yellow Springs Instrument Co., Model 53). Approximately 50 mg of slices were incubated in a reaction vessel containing 4 ml of the modified Cross-Taggart medium saturated with oxygen at 25°C. Decrease in PO2 in the medium was measured using a Clark electrode for 15 min, and the rate of oxygen consumption was calculated. Oxgen consumption was measured in the absence (total consumption) and presence (ouabain-insensitive component) of 1 mM ouabain, and the difference was taken as ouabain-senstitive component.
3. Vesicle Experiments
Vesicle preparation
Brush border (BBMV) and basolateral (BLMV) membrane vesicles were simultaneously isolated from rabbit kidney cortex by Percoll-density gradient centrifugation and Mg2+-precipitation as described previously17).
The vesicles were suspended in the vesicle buffer, adjusted to yield a protein concentration of 6 mg/ml and stored at −70°C until use. The composition of vesicle buffer was 100 mM mannitol, 100 mM KCl and 20 mM Hepes/Tris (pH 6.0). Prior to transport studies, vesicles were preincubated at 37°C for 30 min to effectively load with appropriate buffer4).
The purity of vesicle preparations was evaluated by comparing marker enzyme activities in initial homogenates and final membrane vesicle preparations. NA-K-ATPase and alkaline phosphatase are well established marker enzymes for BLMV and BBMV, respectively. The enrichment of NA-K-ATPase in BLMV and that of alkaline phosphatase in BBMV was 13.85 and 10.33, respectively.
2) Transport Studies
TEA uptake by vesicles was measured by a rapid filtration technique. Briefly, the reaction was initiated by adding membrane vesicles to the incubation medium (a 1:10 dilution of membrane vesicle suspension) containing 14C-TEA at 25°C. The composition of the incubation medium was 100 mM mannitol, 100 KCl and 20 mM Hepes/Tris (pH 7.4). At 10 sec of the incubation, 100 μM aliquots were taken and quickly filtered under vacuum through Millipore fiters (HAWP, 0.45 μM pore size), which were soaked overnight in distilled water. The filters were then washed with 5 ml of ice-cold stop solution containing 100 mM mannitol, 100 mM KCl and 20 mM Hepes/Tris (pH 7.4) and dissolved in 1.0 ml of methoxyethanol. After addition of 10 ml of scintillation cocktail, the amount of radioactivity taken up by vesicles was determined by liquid scintillation spectrometry (Packard Tricarb 300C). Nonspecific binding of substrate to vesicle membrane was determined by the same filtration procedure after vesicles were incubated in distilled water containing 0.1% deoxycholate and radioactive substrate.
3) Enzyme Assays
Na-K-ATPase activity was measured by the method of Jørgensen and Skou18) and alkaline phosphatase activity by the method of Linhardt and Walter19). Protein was determined according to Bradford20), using γ-globulin as a standard.
4) Statistical analysis
All experiments were per-formed on more than three separate membrane preparations. Student’s t-test was used in statistical evaluation of the data and p values less than 0.05 were considered significant.
RESULTS
1. Effect Probenecid on TEA Uptake in Slices
Fig. 1 shows the time course of TEA uptake by renal cortical slices in the presence and absence of 3 mM probenecid. Probenecid significantly inhibited TEA uptake. To examine the concentration dependence for the inhibitory effect of probenecid, TEA uptake was measured in the presence of various concentrations of probenecid. The results are shown in Fig. 2. Relative S/M ratios of TEA at the end of 60 min incubation were plotted against probenecid concentration in the medium. Probenecid significantly inhibited TEA uptake at higher concentrations than 1 mM.
Fig. 1.
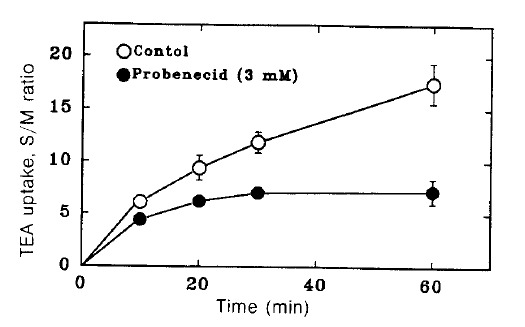
Time course of TEA uptake by renal cortical slices in the hresence and absence of 3 mM probenecid. Each point represents the mean ± SE of three experiments.
Fig. 2.
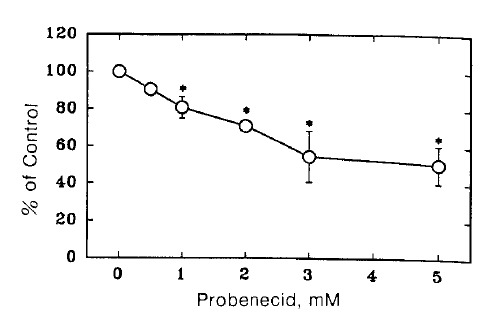
Effect of probenecid on TEA uptake by renal cortical slices were incubated for 60 min at 25°C in a medium containing 10 μM 14C-TEA under 100% oxygen atmosphere. Each point represents the mean ± SE of four experiments. *pä0.05 compared with the control.
2. Reversibility of Probenecid Inhibition
To determine wheter the effect of probenecid on TEA uptake was reversible, the slices were treated with probenecid of 1 and 2 mM concentrations for 30 min and incubated in the presence and absence of probenecid. As shown in Fig. 3, when probenecid treated slices were inculbated in the absence of probenecid, TEA uptake was not significantly altered. These results indicate that probenecid induces a reversible inhibition of TEA uptake.
Fig. 3.
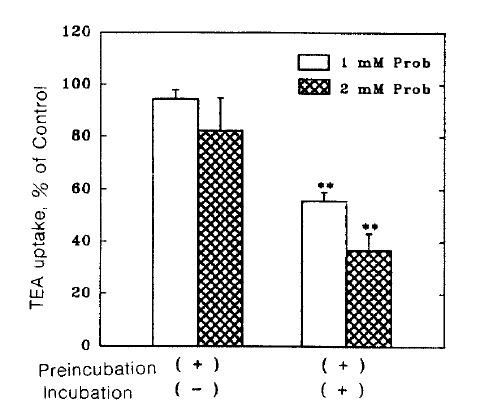
Reversibility of the inhibitory effect of probenecid on TEA uptake. Slices were preincubated for 30 min with probenecid and then incubated for 60 min in the presence (+) and absence(−) of probenecid. Data are expressed as percent of control uptake measured in the absence of probenecid in both preincu bated and incubated media. Each point represents the mean ± SE of four experiments. **pä0.01 compared with the control.
2. Effect Probenecid on TEA Efflux
The efflux rate constant was determined in the presence and absence of probenecid of 3 mM probenecid in the medium. In all paired experiments, the efflux rate constants did not alter in the presence of probenecid (0.028±0.03 vs. 0.027±0.003 per min). This indicates that the inhibitory effect of probenecid on the steadystate accumulation of TEA (Fig. 1) is due primarily to an inhibition of the influx of TEA from the medium into the cell across the basolateral membrane.
4. Effect of Probenecid on Kinetics of TEA Uptake
Fig. 4. depicts Lineweaver-Burk plot of TEA uptake in the presence of 2 mM probenecid. Apparently probencid decreased the Vmax 1.87±0.29 to 1.0±0.14 μmole/g/10 min (Pä0.05) without a significant change in Km (0.33±0.07 vs. 0.29±0.08 mM).
Fig. 4.
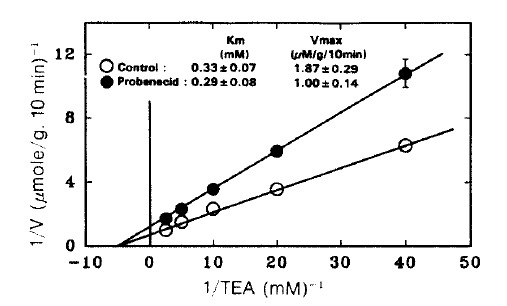
Lineweaver-Burk plot of the initial rate of active TEA accumulation into renal cortical slices in the presence and absence of 3 mM probenecid. TEA accumulation was determined during a 10-min incubation in a medium containing various concentrations of PAH and was corrected for passive accumulation which was determined in the presence of 10 mM choline chloride under 100% N2 atmosphere. Each point represents the mean ± SE of four experiments.
5. Effect of TEA on Probenecid Inhition of p-aminohippurate (PAH) Uptake
If the probenecid inhibiton of TEA uptake resulted from the interaction of organic anion with the organic cation transporter, the inhibitory effect of probenecid on PAH uptake could be reduced by TEA. The possibility was determined in Fig. 5. TEA did not affect PAH uptake as well as the inhibitory effect of probenecid on PAH uptake.
Fig. 5.
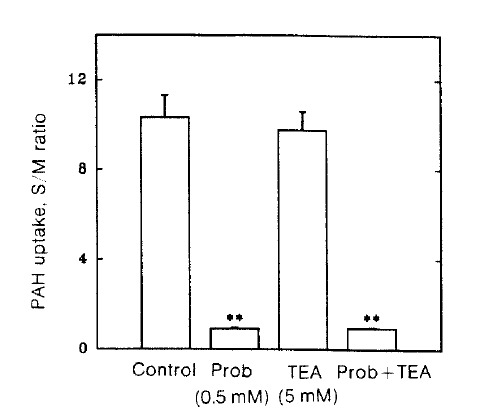
Effect of TEA on the probenecid inhibition of p-aminohippurate (PAH) uptake in renal cortical slices. PAH uptake of 75 μM 14C-PAH was determined for 60 min at 25°C in a medium containing 130 mM NaCl, 10 mM KCl, 1.5 mM CaCl2, 5 mM Na acetate and 20 mM Tris/HCl (pH 7.8) in the presence and absence of organic three experiments. **pä0.01 compared with control.
6. Effect of Probenecid on Na-K-ATPase Activity and Oxygen Consumption
The effect of probencid on Na-K-ATPase activity in the basolateral membrane is shown in Table 1. Probenecid did not alter enzyme activity at the concentration for inhibition of TEA uptake.
Table 1.
Effect of Probenecid on Microsomal Na+-K+-ATPase Activity
| Probenecid (mM) | Na-K-ATPase Activity (μmole Pi/mg protein, hr) |
|---|---|
| 0 (Control) | 43.04±3.62 |
| 2 | 46.84±4.27 |
| 5 | 44.30±5.18 |
All values are the mean ± SE of three determinations.
Fig. 6 shows changes in tissue oxygen consumption of renal cortical slices in the presence of various concentrations of probenecid. Probenecid inhibited significantly both ouabain sensitive and insensitive oxygen consumption at 3 and 5 mM.
Fig. 6.
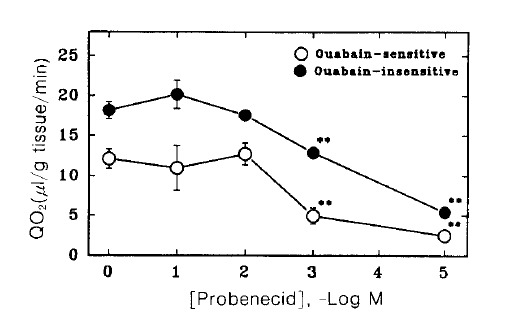
Effect of probenecid on oxygen consumption in renal cortical slices. Oxygen consumption was measured in the presence (ouabain-insensitive) and absence (total) of 1 mM ouabain, and the difference was taken as ouabain-sensitive component. Each point represents the mean ± SE of five experiments. **pä0.01 compared with the control.
7. Effect of Probenecid on TEA Uptake in Plasma Membrane Vesicles
Table 2 summarizes the effect of probenecid on TEA uptake in brush border (BBMV) and basolateral (BLMV) membrane vesicles. Since it has been demonstrated that TEA uptake is driven by a H+/TEA exchange process in BBMV and by a potential-dependent process in BLMV4), TEA uptake was measured in the presence of an outwardly directed H+ gradient in BBMV and BLMV. Probenecid did not significantly reduce the initial (10 sec) uptake of TEA even at concentration as high as 10 mM in both membranes. The equilibrium value measured for 60 min incubation was also unaltered by probenecid.
Table 2.
Effect of Probenecid of TEA Uptake in BBMV and BLMV
| Initial | Equilibrium | |||
|---|---|---|---|---|
| Probenecid conc. (mM) |
|
|||
| Uptake (pmole/mg. 10s) | Inhibition (%) | Uptake (pmole/mg. 60 min) | Inhibition (%) | |
| BBMV | ||||
| 0 (Control) | 1272.30±61.48 | 0 | 739.65±36.42 | 0 |
| 1 | 140.45±117.62 | 11.12±9.16 | 660.63±43.91 | 10.24±5.26 |
| 5 | 056.93±69.18 | 17.10±5.49 | 715.96±28.45 | 3.55±3.87 |
| 10 | 086.33±77.48 | 14.02±6.20 | 683.04±25.55 | 7.83±3.39 |
| BLMV | ||||
| 0 (Control) | 804.97±48.34 | 0 | 895.59±73.22 | 0 |
| 5 | 829.56±42.03 | −3.43±4.96 | 953.65±79.59 | −6.48±8.26 |
| 10 | 950.15±57.95 | −11.37±7.34 | 868.53±47.64 | 3.02±5.36 |
Membrane vesicles were preloaded with 100 mM mannitol, 100 mM KCl and 20 mM Mes/Tris (PH 6.0). Uptake of 50 μM 14)C-TEA was measured for 10 sec and 60 min in a buffer containing in 100 mM mannitol, 100 mM KCl and 20 mM Hepes/Tris (PH 7.5) in the presence and absence of various concentrations of probenecid (Prob)
DISCUSSION
Although probenecid has used as the prototypic inhibitor of organic anion tranport, this compound also inhibits the transport of organic cations such as cimetidine and MNM12–14) However, it is unclear whether the observed effect of probenecid is due to a direct action on the organic cation transport system.
The present study demonstrated that probenecid significantly inhibited TEA uptake by renal cortical slices at concentration higher than 1 mM. The kinetic analysis showed that probenecid inhibited TEA influx in a noncompetitive manner, in which it significantly decreased Vmax while it had no effect on Km for TEA. These results suggest that the inhibitory effect of probenecid is not due to mutual interaction between probencid and TEA on the organic cation carrier.
Pakarinen & Runeberg21) demonstrated that probenecid inhibits the accumulation of dicarboxylates and oxygen consumption in kidney cortical slices. In this study, probenecid also inhibited significantly tissue oxygen consumption in cortical slices. Since the transport of TEA uptake across the basolateral membrane of the renal proiximal tubule is affected by the transmembrane potential generated at the expense of metabolic energy2,22), it would be reduced by an agent which impairs the cellular metabolism of the membrane potential. Thus, the results of the present study suggest that the inhibition of TEA uptake by probenecid was not due to a direct effect of this compound on the transport system but to changes in cellular metabolism. From slice experiments, however, it is difficult to distinguish whether the inhibitory effect of probencid is due to an effect on cellular metabolism of to a direct interaction of probenecid with transport system. Thus, studies with membrane vesicles were chosen since the influence of cellular metabolism on transport process is excluded in the isolated vesicle system. Probenecid did not significantly inhibit the uptake of TEA in BBMV and BLMV even to concentrations as high as 10 mM (Table 2).
Contrary to what was observed in this study, Hsyu et al14) found that probenecid inhibits competitively MNM transport in rabbit renal BBMV and concluded that the inhibition is due to a mechanism involved in the interaction of organic anions with the organic cation transporter. The reason for this discrepancy is not clear at present. However, this may be due to differences in the transport system for NMN and TEA. Renal slice accumulation of NMN is different from that of TEA in rabbits, the former being transported by a passive mechanism while the latter by an active process23). It has been demonstrated that renal brush border membranes also possess multiple carrier systems for organic cations24).
In this study, probencid reduced slightly TEA uptake in BBMV, although not significant. TEA uptake by slices was also inhibited by probenecid concentrations (1 and 2 mM) which did not affect the oxygen consumption. These results may be due to non-specific membrane effect of probenecid. Nonspecific effect of probenecid on membrane transpory systems has been demonstrated by several workers. Kippen et al25) reported that probenecid inhibits significantly glucose uptake by renal BBMV. Ullrich et al26) also demonstrated that this compound inhibits markedly succinate transport in intact proximal tubule.
In conclusion, the present study demonstrates that probenecid inhibits significantly TEA uptake in cortical slices but not in plasma membrane vesicles. The inhibitory effect results from an influence on other cell processes such as cellular metabolism rather than a direct interction with the organic cation transporter.
REFERENCES
- 1.Rennick BR. Renal tubule transport of organic cations. Am J Physiol. 1991;240:F83. doi: 10.1152/ajprenal.1981.240.2.F83. [DOI] [PubMed] [Google Scholar]
- 2.Rennick BR, Farah A. Studies on the renal tubule transport of tetraethylammonium ion in the dog. J Pharmacol Exp Ther. 1956;116:287. [PubMed] [Google Scholar]
- 3.Holohan PD, Ross CR. Mechanisms of organic cation transport in kidney plasma membrane vesicles. II. △pH studies. J Pharmacol Exp Ther. 1981;216:294. [PubMed] [Google Scholar]
- 4.Jung JS, Kim YK, Lee SH. Characteristics of tetraethylammonium transport in rabbit renal plasma-membrane vesicles. Biochem J. 1989;259:377. doi: 10.1042/bj2590377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Takano M, Inui K-I, Okano T, Saito H, Hori R. Carrier-mediated transport systems of tetraethylammonium in rat renal brush border and basolateral membrane vesicles. Biochim Biophys Acta. 1984;773:113. doi: 10.1016/0005-2736(84)90556-x. [DOI] [PubMed] [Google Scholar]
- 6.Wright SH. Transport of N’-methylnicotinamide across brush border membrane vesicles from rabbit kidney. Am J Physiol. 1985;249:F-903. doi: 10.1152/ajprenal.1985.249.6.F903. [DOI] [PubMed] [Google Scholar]
- 7.Schäli C, Schild L, Overney J, Roch-Ramel F. Secretion of tetraethylammonium by proximal tubules of rabbit kidneys. Am J Physiol. 1984;254:F238. doi: 10.1152/ajprenal.1983.245.2.F238. [DOI] [PubMed] [Google Scholar]
- 8.Tarloff JB, Brand PH. Active tetraethylammonium uptake across the basolateral membrane of rabbit proximal tubule. Am J Physiol. 1986;251:F141. doi: 10.1152/ajprenal.1986.251.1.F141. [DOI] [PubMed] [Google Scholar]
- 9.Ross CR, Holohan PD. Transport of organic anions and cations in isolated renal plasma membranes. Ann Rev Pharmacol Toxicol. 1983;23:65. doi: 10.1146/annurev.pa.23.040183.000433. [DOI] [PubMed] [Google Scholar]
- 10.Besseghir K, Mosig D, Roch-Ramel Transport of the organic cation N’-methylnicotinamide by the rabbit proximal tubule. I. Accumulation on the isolated nonperfused tubule. J Pharmacol Exp Ther. 1989;253:444. [PubMed] [Google Scholar]
- 11.Kinsella JL, Hologan PD, Pessah NI, Ross CR. Transport of organic ions in renal cortical luminal and antiluminal membrne vesicles. J Pharmacol Exp Ther. 1979;209:443. [PubMed] [Google Scholar]
- 12.Cacini W, Keller MB, Grund VR. Accmulation of cimetidine by kidney cortical slices. J Pharmacol Exp Ther. 1982;221:342. [PubMed] [Google Scholar]
- 13.McKinney TD, Myers P, Speeg KV. Cimetidine secretion by rabbit renal tubules in vitro. Am J Physiol. 1981;241:F69. doi: 10.1152/ajprenal.1981.241.1.F69. [DOI] [PubMed] [Google Scholar]
- 14.Hsyu P-H, Gisclon LG, Hui AC, Giacomini KM. Interactions of organic anions with the organic cation transpoter in renal BBMV. Am J Physiol. 1988;254:F56. doi: 10.1152/ajprenal.1988.254.1.F56. [DOI] [PubMed] [Google Scholar]
- 15.Kim YK, Park YS, Lee SH. Effect of harmaline on organic ion transport in rabbit renal cortical slices. Arch Int Pharmacodyn Ther. 1988;294:259. [PubMed] [Google Scholar]
- 16.Farah A, Frazer M, Porter E. Studies on the uptake of N’-methylnicotinamide by renal slices of dog. J Pharmacol Exp Ther. 1959;126:202. [PubMed] [Google Scholar]
- 17.Lee HY, Kim KR, Woo JS, Kim YK, Rark YS. Transport of organic compounds in renal plasma membrane vesicles of cadmium intoxicated rats. Kid Int. 1990;37:727. doi: 10.1038/ki.1990.39. [DOI] [PubMed] [Google Scholar]
- 18.Jørhensen PL, Skou JC. Purification and characterization of (Na+-K+)-ATPase in preparations from the outer medulla of rabbit kidney. Biochem Biophys Acta. 1971;233:366. doi: 10.1016/0005-2736(71)90334-8. [DOI] [PubMed] [Google Scholar]
- 19.Linhardt K, Walter K. Phosphatases. In: Bergeyer HU, editor. Methods in Enzymatic Analysis. New York: Academic; 1963. pp. 783–785. [Google Scholar]
- 20.Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976;72:248. doi: 10.1006/abio.1976.9999. [DOI] [PubMed] [Google Scholar]
- 21.Pakarinen A, Runeberg L. Effects of phenolsulfonphthalein and probenecid on the uptake and utilization of citrate and α-ketoglutarate by kidney, in vitro. Biochem Pharmacol. 1969;18:2439. doi: 10.1016/0006-2952(69)90360-8. [DOI] [PubMed] [Google Scholar]
- 22.Nechay BR, Pardee LM. Inhibition of N’-methylnicotinamide secretion by ouabain in the chicken kidney. J Pharmacol Exp Ther. 1965;147:270. [PubMed] [Google Scholar]
- 23.Berndt WO. Organic base transport: a comparative study. Pharmacolgy. 1981;22:251. doi: 10.1159/000137497. [DOI] [PubMed] [Google Scholar]
- 24.Miyamoto Y, Tiruppathi C, Ganapathy V, Leibach FH. Multiple transport systems for organic cations in renal brush border membrane vesicles. Am J Physiol. 1989;256:F540. doi: 10.1152/ajprenal.1989.256.4.F540. [DOI] [PubMed] [Google Scholar]
- 25.Kippen I, Hirayama B, Klinenberg JR, Wright EM. Transport of p-aminohippuric acid, uric acid and glucose in highly purified rabbit renal brush border membranes. Biochim Biophys Acta. 1979;556:161. doi: 10.1016/0005-2736(79)90428-0. [DOI] [PubMed] [Google Scholar]
- 26.Ullrich KJ, Fasold H, Rumrich G, Kläss S. Secretion and contraluminal uptake of dicarboxylic acids in the proximal convolution of rat kidney. Pflugers Arch. 1984;400:241. doi: 10.1007/BF00581554. [DOI] [PubMed] [Google Scholar]


