Abstract
Mosquitoes are the most deadly vectors of parasites that cause diseases such as malaria, yellow fever, and filariasis. In view of the recent increased interest in developing plant origin insecticides as an alternative to chemical insecticides, the objective of this study was to determine the repellent activity of creams formulated with methanol crude extract (MCE), hexane fraction (HF), and ethyl acetate fractions (EAFs) of Ocimum gratissimum and Lantana camara leaves in single and combined actions against female Aedes aegypti. Evaluation was carried out in the net cages (30 by 30 by 30 cm) containing 60 blood-starved female mosquitoes each and were assayed in the laboratory condition following World Health Organization 2009 protocol. All formulations (single and mixture) were applied at 2, 4, 6, and 8 mg/cm2 in the exposed area of human hands. Only acetone + white soft paraffin served as negative control and odomos (12% DEET) as positive control. All the formulations presented good protection against mosquito bites without any allergic reaction by the human volunteers. The repellent activity was dependent on the strength of the extracts and fractions. Among the tested formulations, the maximum protection time was observed in MCE (120 min) and EAF (150 min) of O. gratissimum; MCE:MCE (150 min) and HF:HF (120 min) mixtures of both plants. In addition, MCE:MCE and HF:HF mixtures from both plants showed possible synergistic effect. From the results, the combination of O. gratissimum and L. camara to formulate natural mosquito repellent using small amount of extracts can be encouraging to be an alternative to conventional DEET.
Keywords: Ocimum gratissimum, Lantana camara, Aedes aegypti, mosquito repellent formulation, synergism
Mosquitoes are the most deadly vectors of parasites that cause diseases such as malaria, filariasis, Japanese encephalitis, dengue fever, dengue hemorrhagic fever, and yellow fever (Renugadevi et al. 2012). Aedes aegypti alone is a very important disease vector, transmitting the arbovirus that causes dengue fever and dengue hemorrhagic fever, chikungunya, and allergic skin reaction especially in children, which is endemic to Southeast Asia, the Pacific island area, the Americas, and Africa (Supratik et al. 2010).
Dengue fever is clinically difficult to diagnose especially in the developing countries with no established diagnostic facility and could easily be mistaken another disease (Baba et al. 2009). In Nigeria, misdiagnosis of Dengue (DEN) infection for malaria/typhoid has been detected (Baba et al. 2009). Still in Nigeria, the four forms of dengue (DEN-1, DEN-2, DEN-3, and DEN-4) have been detected in Ae. aegypti (Baba et al. 2009).
An outbreak of Yellow fever occurred on the Jos Plateau and adjacent areas in 1969, the first recognized epidemic of yellow fever in Nigeria since 1953. An estimated 252 patients with yellow fever were hospitalized. The case fatality ratio for hospitalized patients was approximately 40%. The diagnosis of yellow fever was confirmed by virus isolation, serology, or pathology in 55 patients. It was estimated that up to 100,000 cases of yellow fever may have occurred during the epidemic (Carey et al. 1972).
One commonly advocated approach for preventing mosquito bites is personal protection. This method allows an individual to select from (or combine) avoidance techniques, exclusion of mosquitoes with physical and chemical barriers, treatment of fabric with toxicants, and the use of topical (skin) repellents (Barnard and Xue 2004). Application of repellents to the skin is a common personal protection practice (Kazembe and Nkomo 2012). Numerous mosquito repellents under a variety of brand names are currently commercially available to consumers throughout the world. Most of them contain DEET (N,N-diethyl-m-toluamide), which has broad-spectrum activity and effectively repels most mosquitoes, biting flies, chiggers, fleas, and ticks. It is the most effective insect repellent available for human use (Fradin and Day 2002). Currently, DEET is formulated in aerosols, pump sprays, lotions, creams, liquids, sticks, roll-ons, and impregnated towelettes, with concentrations ranging from 5 to 100%. However, DEET has shown adverse effects when applied as directed. The most common problem is local skin irritation, including erythema and pruritis, at the site of application. One case of anaphylaxis after brief exposure to DEET has been reported (Osimitz and Murphy 1997). Many people who applied high concentrations of DEET on a daily basis have developed more severe adverse effects due to chronic exposure. These adverse effects included insomnia, muscle cramps, mood disturbances, and rashes (Osimitz and Murphy 1997). For this reason, extracts of several plants have been studied as possible mosquito repellents revealing the existence of natural repellents with good efficacy (Esimone et al. 2011).
Ocimum gratissimum L. (Lamiaceae) is an aromatic perennial herb wildly grown in Nigeria. It is commonly known as Scent leaf or “Nchuanwa” in Igbo, “Effirin” in Yoruba, and “Dai doyatagida” in Hausa (Orwa et al. 2009, Okoli et al. 2010). The plant is used as food spice (Okwu 2006) and for the treatment of many infections (Rabelo et al. 2003). In Nigeria, the repellent activity of ointments formulated with O. gratissimum oil has been reported against Ae. aegypti and Culex quinquefasciatus mosquitoes (Esimone et al. 2011).
Lantana camara L. is a flowering ornamental plant belonging to family Verbenaceae. In Nigeria, L. camara has as local names “Ewonadele” in Yoruba, “Kimbamahalba” in Hausa, and “Anya nnunu” in Igbo (Gabi et al. 2011). According to Sanjeeb et al. (2012), the plant is said to have antibacterial, antifungal, wound healing, antimotility, antiulcerogenic, hemolytic, antihyperglycemic, antifilarial, antiinflammatory, embryo toxicity, antiurolithiatic, anticancer and antiproliferative, antimutagenic, antioxidant, and mosquitoes-controlling activities. In this work, we investigated the single and combined repellent actions using the methanol crude extract (MCE), hexane fraction (HF), and ethyl acetate fraction (EAF) from O. gratissimum and L. camara against Ae. aegypti bites for those plants are almost everywhere within Nigeria.
Materials and Methods
Collection of Plant Materials, Extraction, and Fractionation
The fresh L. camara and O. gratissimum leaves were harvested from farms at Oko, Anambra State, Nigeria. The plants were identified by Dr. S. I. Okeke (PhD), a botanist at Federal Polytechnic, Oko, Anambra State, Nigeria. The plant materials were air dried at room (temperature of 28 ± 3°C, 79 ± 4% relative humidity [RH]) for 2 wk. The dried leaves were ground in powder using an electric grinder until the powder passed through a 0.4-mm mesh sieve. The extraction and fractionation procedures were carried out in accordance with the method adopted by Okoye and Osadede (2009). From the collection of plant material powder, 500 g for L. camara and 325 g for O. gratissimum were extracted for 3 d by cold maceration in methanol JQ American Corporation JQLAB, California (USA) shaking it thrice per day (morning, noon, and afternoon) in the laboratory of Pharmaceutical and Medicinal Chemistry. The maceration process was then repeated twice for maximal extraction. The suspension was filtered through Whatman No. 1 filter paper (size: 24 cm, England) Cole-Parmer Instrument Co. Ltd. (United Kingdom). The MCE (11.68% for L. camara and 6.23% for O. gratissimum) were concentrated to dryness in rotary vacuum evaporator RE300 (ROTAFLO, England) Fisher Scientific UK Ltd. (United Kingdom). The MCEs were adsorbed on silica gel (60–200 mesh) GeeJay Chemicals Ltd. (United Kingdom) and eluted in succession with hexane and ethyl acetate JQ American Corporation JQLAB, California (USA) to obtain HF (30.05% for L. camara and 26.11% for O. gratissimum) and EAF (24.59% for L. camara and 27.55% for O. gratissimum). All the extracts and fractions were stored in a refrigerator at −4°C American refrigeration Company, Inc., 149 River Street (USA) until used.
Formulation of Repellent Creams
The formulation procedure was set up as described (Adeniran and Fabiyi 2012). One gram of MCE, HF, and EAF of each plant material was weighted out in 5-ml tubes using Adventurer Scale, Ohaus Corp. Pine Brook, NJ; max cap: 210 g; readability: 0.0001 g Ohaus Corporation, New Jersy (USA); and diluted in 3 ml of acetone Sigma-Aldrich Co. LLC (USA) over night for proper dilution. For combination, 0.5 g of each MCE, HF, and EAF from each plant material were also weighted out in 5 ml tubes and diluted in 1.5 ml of acetone. The following day, 4 g of white soft paraffin Kerax Limited, (United Kingdom) (either for single extract or combined extract) was accurately weighted into a 100 ml beaker and melted in water bath at 60°C. At this stage, the aliquot was transferred into the molten white soft paraffin. The MCE of L. camara and O. gratissimum (0.5 g of extract in 1.5 ml of acetone each) were poured together into melted white soft paraffin making it at the same concentration as in single formulation (1 g of extract in 3 ml of acetone). The same procedure was performed for HF and EAF. The mixture was swirled properly to ensure that the extract or fraction was uniformly mixed with the white soft paraffin using a spatula before transferring in a screwed covered bottle. The combination was used to investigate the possibility of synergistic activity. The thick product (25 % w/w cream) was then taken for repellency test. The negative control was set up in poring 3 ml of acetone in 4 g of melted white soft paraffin without any trace of extract or fraction. Odomos (12% DEET), bought in a pharmacy at Awka, Anambra State, Nigeria, which is a conventional mosquito repellent was used as positive control to compare the test results with.
White soft paraffin is also known as white petroleum jelly. It is used in a variety of applications, which include pharmaceutical, cosmetic, and skin care uses. It is an odorless jelly that is impervious to water (Callahan 2014). The stratum corneum is an outer layer of the skin cells that retains moisture in the skin. This layer can be diminished or lost for a variety of reasons. These include aging and washing with soap and hot water (Callahan 2014). Applying white soft paraffin creates a substitute for the stratum corneum and the sebum that naturally keeps the skin moisturized. This improves the health of the skin, diminishes dryness, and prevents cracking (Callahan 2014).
Preparation of Mosquitoes for Repellence Test
Ae. aegypti larvae were a laboratory strain collected from World Health Organization (WHO)/National Arbovirus and Vector Research Centre Enugu, Enugu state, Nigeria, and reared in the insectary, Faculty of Pharmaceutical Sciences, Agulu; Nnamdi Azikiwe University, Awka; Anambra State, Nigeria. Female Ae. aegypti mosquitoes (60 in number), 7–10-d old, free from pathogens, were placed in 30 by 30 by 30 cm cages with a mosquito netting on top and two sides and a sleeve on one side used to introduce and retrieve mosquitoes using a mouth aspirator. The mosquitoes were fed on 10% sugar solution, starved for 24 h prior to the experiments and maintained at 32 ± 4°C, 83 ± 3% RH, with a photo period of 12:12 (L:D) h.
Preparation of Human Volunteers for Repellent Test
The laboratory repellence test was conducted using the standard human-bait technique of WHO (2009) and Kazembe and Nkomo (2012). Three human volunteers were used in the experiments. An area of 7 by 7 cm (49 cm2) was marked and cut open on plastic disposable hand gloves, manufactured by Ahmedabad, Gujarat (India). The edges of the cut area were lined with masking tape.
Determination of Landing Time
Landing time is the average time required for the first mosquito to land on the exposed area and attempt to take a blood meal. An untreated hand was exposed to the mosquitoes and the time to landing was recorded to determine the readiness of the mosquitoes to take blood. The procedure was repeated 10 times in each cage and the average landing time calculated:
Dose–Response Experiments
Because Ae. aegypti is a day biter, the tests were carried out between 8:00 and 16:00 Nigerian time, at 28–32°C and 71–82% RH. Before each test, the hand of a human subject was washed with unscented neutral soap, thoroughly rinsed, and allowed to dry 20 min before formulation application. The formulation was applied from the wrist to the fingertips. An attempt of the mosquito to insert its stylets was considered as a bite, recorded and then shaken off before imbibing any blood. A treated hand with acetone and white soft paraffin formulation served as a negative control. The left hand was used for treatment and the right one for negative control. The control hand was exposed to mosquitoes before the treated hand for 3 min and the number of mosquito bites recorded. To determine the dose–response experiments, a series of repellency experiments was performed starting with the application of 0.1 g of formulated cream on 49 cm2 cut off area (≈2 mg/cm2) dose of the formulation. The 2 mg/cm2 dose was applied on the open area of the hand and the percent repellency during the 3 min exposure time was determined. The dose was increased by 2 mg/cm2 for each succeeding experiment, calculating the percent repellence after each dose, until 0.4 g of formulated cream on 49 cm2 cut off area (≈8 mg/cm2). This was to check the minimum concentration of formulation, which gave complete protection from mosquito bites. To equalize chances to all formulations, 8 mg/cm2 was applied. The repellence percentage was then plotted against concentrations of formulations to check the minimal dose to be applied for total protection.
Protection Time Experiments
The treated hands with application of 8 mg/cm2 dose from previous section (dose–response experiments) were followed up for protection time procedure. The number of bites was counted over 3 min; every 30 min. Tests were discontinued after 180 min. The experiments were conducted in triplicate in separate cages. In each replicate, each volunteer was used to nullify any effect of skin differences on repellency. The protection percentage was calculated by using the following formula:
Where C is the number of mosquitoes that bit on the control and T is the number of mosquitoes that bit on the treated areas of the volunteer.
Adverse Effect of the Formulations on the Skin of Human Volunteers
The effects on skin such as skin allergic reaction, irritation, skin sensitization, skin keratinization, and spots appearance were observed daily for 3 wk on each human volunteer.
Ethical Issues
This study was given an ethical approval from Anambra State University Teaching Hospital, Amaku, Awka; Anambra State, Nigeria Ethics Review Committee with the reference number of ANSUTH/AA/ECC/37. This acceptance was after the submitting of the detailed proposal of the present project to the Ethics Review Committee for proper study. All volunteers were given written consent forms signed in front of a witness who was not a study participant.
Statistical Analysis
The calculation of protection percent and the plotting graphs were obtained using Microsoft Office Excel 2007. The calculated protection percent results were submitted to SPSS 17.0 for Windows software (SPSS Inc., SPSS (Hong Kong) Ltd., Quarry Bay (Hong Kong)) to get the means of repellence.
Results
Dose–Response Experiments of Formulated Plant Extracts and Fractions as Mosquito Creams Against Ae. aegypti Bites
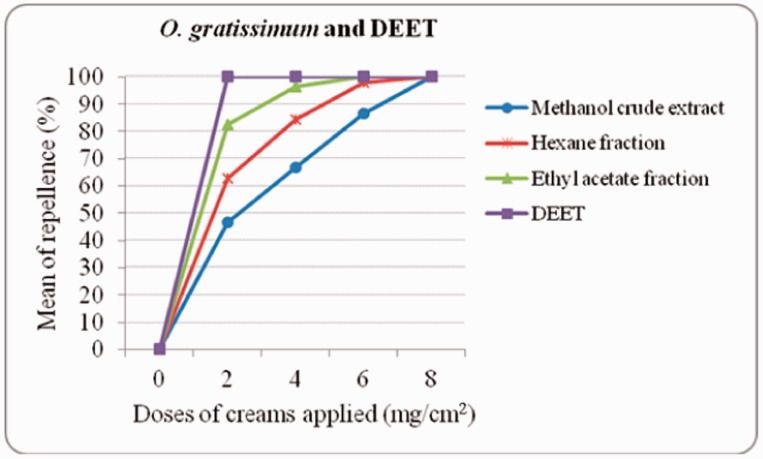
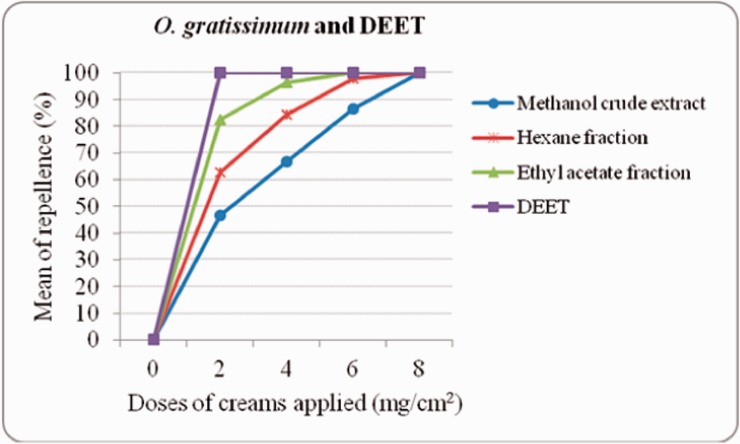
In the dose–response experiments of formulated O. gratissimum extract and fractions, EAF formulation showed good results with the minimal concentration used to achieve complete protection from mosquito bite (Fig. 1). At 2 mg/cm2 the lowest dose, the protection percent was 82%, whereas it was 63% and 47% for HF and MCE, respectively. The DEET control repelled female Ae. aegypti at 100% at the same dose of 2 mg/cm2. At 6 mg/cm2, no mosquito landed on exposed skin of volunteers, but it was at 8 mg/cm2 that HF and MCE recorded the same results.
Fig. 1.
Dose–response experiments of creams formulated with O. gratissimum crude extract and fractions against female Ae. aegypti bites.
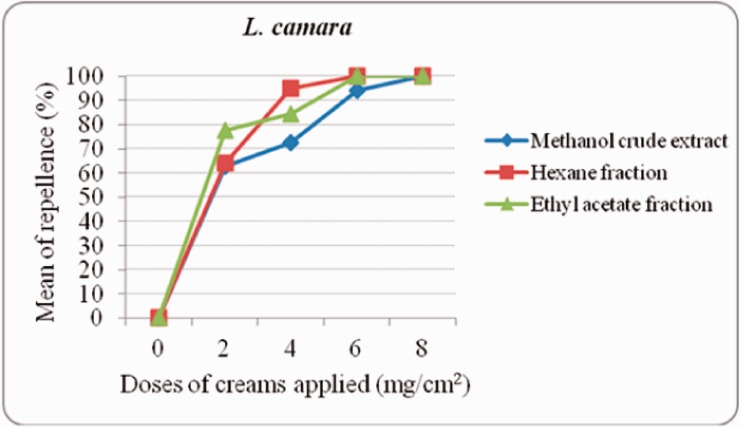
Concerning L. camara at 2 mg/cm2, EAF exhibited 78% protection, whereas 64% and 63% were registered by HF and MCE, respectively (Fig. 2). At 6 mg/cm2, HF and EAF provided 100% protection each, but it was at 8 mg/cm2 that the same record of protection was figured out by MCE.
Fig. 2.
Dose–response experiments of creams formulated with L. camara crude extract and fractions against female Ae. aegypti bites.
In combination of both plants formulated extracts and fractions, MCE recorded better protection (90%) at the lowest dose of 2 mg/cm2 followed by HF and EAF with 79% and 56% repellency, respectively (Fig. 3). Total protection was observed at 4, 6, and 8 mg/cm2 by MCE, HF, and EAF, respectively.
Fig. 3.
Dose–response experiments of creams formulated with O. gratissimum + L. camara crude extracts and fractions against female Ae. aegypti bites.
Protection Time of Creams Formulated with Plant Extracts and Fractions as Mosquito Repellents Against Ae. aegypti Bites
At the end of the proper repellence experiments, the EAF formulation of O. gratissimum showed total protection up to 150 min, but 120 min and 60 min for MCE and HF, respectively (Table 1). The same total protection for L. camara extract and fractions was 60 min. In the combination of two plant extracts and fractions, MCE and HF produced 100% protection up to 150 min each and only 30 min for EAF. From the beginning to the end of the experiments, no mosquito landed on the hands treated with odomos (DEET) (Table 1).
Table 1.
Protection times of creams formulated with O. gratissimum and L. camara crude extracts and fractions as mosquito repellents against female Ae. aegypti bites
| Mean % of bite-repellence of plant extracts formulations against Ae. aegypti |
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Time (min) |
O. gratissimum (Og) |
L. camara (Lc) |
Og + Lc |
DEET | ||||||
| MCE | HF | EAF | MCE | HF | EAF | MCE | HF | EAF | ||
| 0 | 100.00 | 100.00 | 100.00 | 100.00 | 100.00 | 100.00 | 100.00 | 100.00 | 100.00 | 100.0 |
| 30 | 100.00 | 100.00 | 100.00 | 100.00 | 100.00 | 100.00 | 100.00 | 100.00 | 100.00 | 100.0 |
| 60 | 100.00 | 100.00 | 100.00 | 100.00 | 100.00 | 100.00 | 100.00 | 100.00 | 88.89 | 100.0 |
| 90 | 100.00 | 92.16 | 100.00 | 66.67 | 87.18 | 86.67 | 100.00 | 100.00 | 77.78 | 100.0 |
| 120 | 100.00 | 78.43 | 100.00 | 39.21 | 51.28 | 75.56 | 100.00 | 100.00 | 63.89 | 100.0 |
| 150 | 82.22 | 62.74 | 100.00 | 39.22 | 43.59 | 60.00 | 100.00 | 83.33 | 50.00 | 100.0 |
| 180 | 75.55 | 23.53 | 91.23 | 29.41 | 25.64 | 42.22 | 89.58 | 61.90 | 44.44 | 100.0 |
MCE: methanol crude extract; HF: hexane fraction; EAF: ethyl acetate fraction; Number of replicates: 3.
Investigation for Possibility of Synergism in the Repellence Activity
The possibility of synergistic action between both plants showed that the total protection of L. camara MCE was 60 min after which it dropped down to 29% (Table 1). The O. gratissimum MCE lasted up to 120 min of total protection and started falling down to 76% at the end of the experiment. In combination of both plants, the total protection from Ae. aegypti bites was up to 150 min after which it slightly decreased at 90% at the end of the experiment. There may be a possible synergistic effect of MCE formulation between L. camara and O. gratissimum (Table 1).
In the same vein but this time with HF, the total protection of both plants was 60 min. Thereafter, it decreased to 24% and 27% for O. gratissimum and L. camara, respectively, at the very end of the experiments. In their combination, the repellent activity persisted up to 120 min of total protection after which it slowly came down to 62% at the end of the experiment. That equally may be a possible synergistic activity of HF formulation between L. camara and O. gratissimum (Table 1).
However, the EAF of O. gratissimum repelled Ae. aegypti at 100% till 150 min and decreased to 91% after 180 min. Meanwhile, the total protection of L. camara EAF and the mixed EAF of the two plants was 60 and 30 min, respectively, which may be a possible antagonistic action of EAF between O. gratissimum and L. camara to repel female Ae. aegypti (Table 1).
No adverse effects on the exposed skin of human volunteers were observed throughout the 3-wk period after the application of the formulations.
Discussion
Many mosquito-borne diseases, such as malaria, dengue fever, and yellow, are serious public health problems in tropical regions, especially in Africa. These diseases are transmitted to human beings through mosquito bite only. Prevention of mosquito bites is one of the main strategies to control or minimize incidence of these diseases. The use of insect repellents can provide practical and economical means of preventing mosquito-borne diseases and dead. It is important not only for local people in disease risk areas as Nigeria but also for travelers who are vulnerable to diseases spread by mosquito vectors when they visit and seek leisure away from their home countries. Many mosquito formulations have been made containing essential oil of plant materials (Egunyomi et al. 2010, Esimone et al. 2011, Adeniran and Fabiyi 2012, Dickens and Bohbot 2013). The inconvenient of such product is that essential oil has short time of effectiveness because of its rapid volatility. Vanillin is usually added to increase the protection time of the oils (Apiwat et al. 2001). In this work, creams formulated as mosquito repellents from MCE, HF, and EAFs of O. gratissimum and L. camara against Ae. aegypti bites were made.
In previous findings, ethyl acetate and methanol extracts of O. gratissimum leaf showed good mosquito larvicidal activity against Culex gelidus and Culex quinquefasciatus. When adults of the same mosquito species were exposed to the concentration of 500 µg/ml ethyl acetate and methanol extracts for 1 h, above 90% mortality was recorded after 24 h of recovery period (Chinnaperumal and Rahuman 2010). In Nigeria, formulated ointments from O. gratissimum leaf essential oil were tested against Ae. aegypti on rat. The formulation results of 1 g of petroleum jelly in 1 ml and 1.5 ml of essential oil showed 100% protection and that in 0.5 ml essential oil, 93%. The toxicity study on the shaved rat skin showed no obvious effect for 3 wk (Esimone et al. 2011). In this study, 1 g of extract or fraction in 4 g of white soft paraffin provided total protection up to 120 min for MCE and 60 min for HF. No adverse effects were observed on the human volunteers throughout 3 wk after the application of the formulations. Confidence in the safety repellent efficiency of this study was thus established.
In India, the petroleum ether extract of L. camara showed larvicidal activity against Ae. aegypti recording 100% mortality after 48 h of incubation with LC50 of 251 μg/ml (Anitha and Geethapriya 2012). Still in India, the repellent effect of L. camara flowers was evaluated against Aedes mosquitoes. The flower extract in coconut oil provided 94.5% protection from Aedes albopictus and Ae. aegypti bites. The mean protection time was 1.9 h. No adverse effects were observed on the human volunteers 3 mo after the application of the formulations (Dua et al. 1996). In Nigeria, hexane and methanol extracts of L. camara leaf recorded complete protection after 30 min of 2 mg/ml application on guinea pigs (Egunyomi et al. 2010). In this study, MCE and HF of L. camara showed total protection of 60 min. No adverse effects were also observed on the skin of human volunteers 3 wk after the application of the formulations. This confirmed the promising safety and repellent activity of L. camara claimed by some people in the rural areas of Nigeria when they planted it near the entrance of their house.
In this study, MCE and HF showed possible synergistic effect while EAF exhibited possible antagonistic action within O. gratissimum–L. camara mixture. The same results were also observed by Kazembe and Nkomo (2012), when they combined essential oils from Blumea alata, Bidens pilosa, and Chenopodium ambrosioides fresh leaves for mosquito repellents. A clear case of synergism was established in the mixture of B. alata with C. ambrosioides.
Though the mechanism of action of the formulated repellents in this study was not yet established, several lines of evidence suggest that insect repellent molecules reduce mosquito-host contacts by interacting with odorants and odorant receptors, thereby ultimately affecting olfactory-driven behaviors (Bohbot et al. 2011). The first detailed mode of action of repellents was summarized by Davis (1985). Techniques were available for single cell recordings from olfactory receptor neurons (ORNs) on the antennae of mosquitoes, and a number of repellent compounds were tested for their activity on these cells. Based mostly on these electrophysiological studies, Davis hypothesized that repellents had their effect by modifying or blocking responses of ORNs normally sensitive to attractants. The same point was made by Dickens and Bohbot (2013). In this study, one or more compounds should be responsible of modifying or blocking responses of ORNs normally sensitive to attractants.
In conclusion, the use of repellents by travelers (civilian and military) may reduce the occurrence of mosquito borne diseases in temperate areas. In country like Nigeria, where foreign trading business and tourism are major sources of national income, people may use creams formulated with O. gratissimum and L. camara MCE, HF, and EAFs, lonely or in combination as mosquito repellents at the place of DEET to increase their pleasure and comfort. In military operations, they may reduce the diseases and annoyance to personnel, who can then complete their operations more efficiently. They can therefore be viewed as a tool with a specific role in protecting people from mosquito-borne diseases. However, a lot of works need to be done: removing the greenness of the extracts and fractions, extend the work to other mosquito species though it is basically known that Aedes genus is the most resistant mosquito to repel, do further fractionation to get the molecule(s) mostly responsible of repellence, and field trial.
Aknowledgments
We gratefully acknowledge the guidance of Dr. SI Okeke (PhD), a botanist at Federal Polytechnic, Oko, Anambra State, Nigeria, during the harvesting of the leaves. We thank the volunteers for their assistance in repellent tests and WHO/National Arbovirus and Vector Research Centre Enugu, Enugu state, Nigeria, for provision of mosquito colony.
References Cited
- Adeniran O. I., Fabiyi E. 2012. A cream formulation of an effective mosquito repellent: a topical product from lemongrass oil (Cymbopogon citratus) Stapf. J. Nat. Prod. Plant Resour., 2, 322–327. [Google Scholar]
- Anitha R., Geethapriya D. 2012. Larvicidal activity of plant extracts on Aedes Aegypti L. Asian Pac. J. Trop. Biomed., 2(Suppl 3), S1578–S1582. [Google Scholar]
- Apiwat T., Steve D., Wratten R., Roderic S., Usavadee T., Yenchit T. 2001. Repellency of volatile oils from plants against three mosquito vectors. J. Vector Ecol., 26, 76–82. [PubMed] [Google Scholar]
- Baba M. M., Saron M.-F., Vorndam A. V., Adeniji J. A., Diop O., Olaleye D. 2009. Dengue virus infections in patients suspected of malaria/typhoid in Nigeria. J. Am. Sci., 5, 129–134. [Google Scholar]
- Barnard D. R., Xue R. D. 2004. Laboratory evaluation of mosquito repellents against Aedes albopictus, Culex nigripalpus, and Ochlerotatus triseriatus (Diptera: Culicidae). J. Med. Entomol., 41, 726–730. [DOI] [PubMed] [Google Scholar]
- Bohbot J. D., Fu L., Le T. C., Chauhan K. R., Cantrell C. L., Dickens J. C. 2011. Multiple activities of insect repellents on odorant receptors in mosquitoes. Med. Vet. Entomol., 25, 436–444. [DOI] [PubMed] [Google Scholar]
- Callahan, R. 2014. Benefits of white soft paraffin. ( http://www.livestrong.com/article/190070-benefits-of-white-soft-paraffin/).
- Carey D. E., Kemp G. E., Troup J. M., White H. A., Smith E. A., Addy R., Fom A.L.M.D., Pifer J., Jones E. M., Brls P., Shope R. E. 1972. Epidemiological aspects of the 1969 yellow fever epidemic in Nigeria. Bull. World Health Organ., 46, 645–651. [PMC free article] [PubMed] [Google Scholar]
- Chinnaperumal K., Rahuman A. A. 2010. Larvicidal and adulticidal potential of medicinal plant extracts from south India against vectors. Asian Pac. J. Trop. Med., 3, 948–953. [Google Scholar]
- Davis E. E. 1985. Insect repellents: concepts of their mode of action relative to potential sensory mechanisms in mosquitoes (Diptera: Culicidae). J. Med. Entomol., 22, 237–243. [DOI] [PubMed] [Google Scholar]
- Dickens J. C., Bohbot J. D. 2013. Mini review: mode of action of mosquito repellents. Pestic. Biochem. Physiol., 106, 149–155. [Google Scholar]
- Dua V. K., Gupta N. C., Pandey A. C., Sharma V. P. 1996. Repellency of Lantana camara (Verbenaceae) flowers against Aedes mosquitoes. J. Am. Mosq. Control Assoc., 12, 406–408. [PubMed] [Google Scholar]
- Egunyomi A., Gbadamosi I. T., Osiname K. O. 2010. Comparative effectiveness of ethnobotanical mosquito repellents used in Ibadan, Nigeria. J. Appl. Biosci., 36, 2383–2388. [Google Scholar]
- Esimone C. O., Attama A. A., Ngwu G., Iloabanafo C. A., Momoh M. A., Onaku L. O. 2011. Mosquito repellent activity of herbal ointments formulated with Occimum gratissimum oil. J. Pharm. Res., 4, 3442–3444. [Google Scholar]
- Fradin M. S., Day J. F. 2002. Comparative efficacy of insect repellents against mosquito bites. N. Engl. J. Med., 347, 13–18. [DOI] [PubMed] [Google Scholar]
- Gabi B., Adewumi A.A.J., Aina V. O. 2011. Phytochemical characterization and in-vivo anti-malaria activity of Lantana camara leaf extract. Br. J. Pharmacol. Toxicol., 2, 277–282. [Google Scholar]
- Kazembe T. C., Nkomo S. 2012. Use of Blumea alata, Bidens pilosa and Chenopodium ambrosioides as mosquito repellents and mosquitocides. Bull. Environ. Pharmacol. Life Sci., 1, 59–66. [Google Scholar]
- Okoli C. O., Ezike A. C., Agwagah O. C., Akah P. A. 2010. Anticonvulsant and anxiolytic evaluation of leaf extracts of Ocimum gratissimum, a culinary herb. Pharmacognosy Res., 2, 36–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okoye F.B.C., Osadede O. P. 2009. Study on the mechanism of anti-inflammatory activity of the extracts and fractions of Alchornea floribunda leaves. Asian Pac. J. Trop. Med., 2, 7–14. [Google Scholar]
- Okwu D. E. 2006. The potentials of Ocimum gratissimum, Pengluria extensa and Tetrapleurea tetraptera as spices and flavouring agents. J. Chem. Soc. Niger., 31, 38–42. [Google Scholar]
- Orwa, C., A. Mutua, R. Kindt, R. Jamnadass, and S. Anthony. 2009. Agroforestree Database: a tree reference and selection guide version 4.0. World Agro-forestry Centre, Kenya. ( http://www.worldagroforestry.org/resources/databases/agroforestree).
- Osimitz T. G., Murphy J. V. 1997. Neurological effects associated with use of the insect repellent N, N-diethyl-m-toluamide (DEET). J. Toxicol. Clin. Toxicol., 35, 435–441. [DOI] [PubMed] [Google Scholar]
- Rabelo M., Souza E. P., Soares P.M.G., Miranda A. V., Matos F. A., Criddle D. N. 2003. Antinociceptive properties of the essential oil of Ocimum gratissimum L. (Lamiacae) in mice. Braz. J. Med. Biol. Res., 36, 521–524. [DOI] [PubMed] [Google Scholar]
- Renugadevi G., Ramanathan T., Shanmuga P. R., Thirunavukkarasu P. 2012. Studies on combined effect of mangrove plants against three dangerous mosquitoes. Int. J. Pharm. Biol. Arch., 3, 357–362. [Google Scholar]
- Sanjeeb K., Gaurav K., Loganathan K., Kokati V.B.R. 2012. A Review on medicinal properties of Lantana camara Linn. Res. J. Pharm. Technol., 5, 711–715. [Google Scholar]
- Supratik C., Someshwar S., Goutam C. 2010. Mosquito larvacidal effect of orthophosporic acid and lactic acid individually or their combined form on Aedes aegypti. Asian Pac. J. Trop. Med., 3, 954–956. [Google Scholar]
- World Health Organization (WHO) 2009. Guidelines for efficacy testing of mosquito repellents for human skin. WHO_HTM_NTD_WHOPES_2009.4_eng. Geneva, Switzerland: World Health Organization, 30 p.