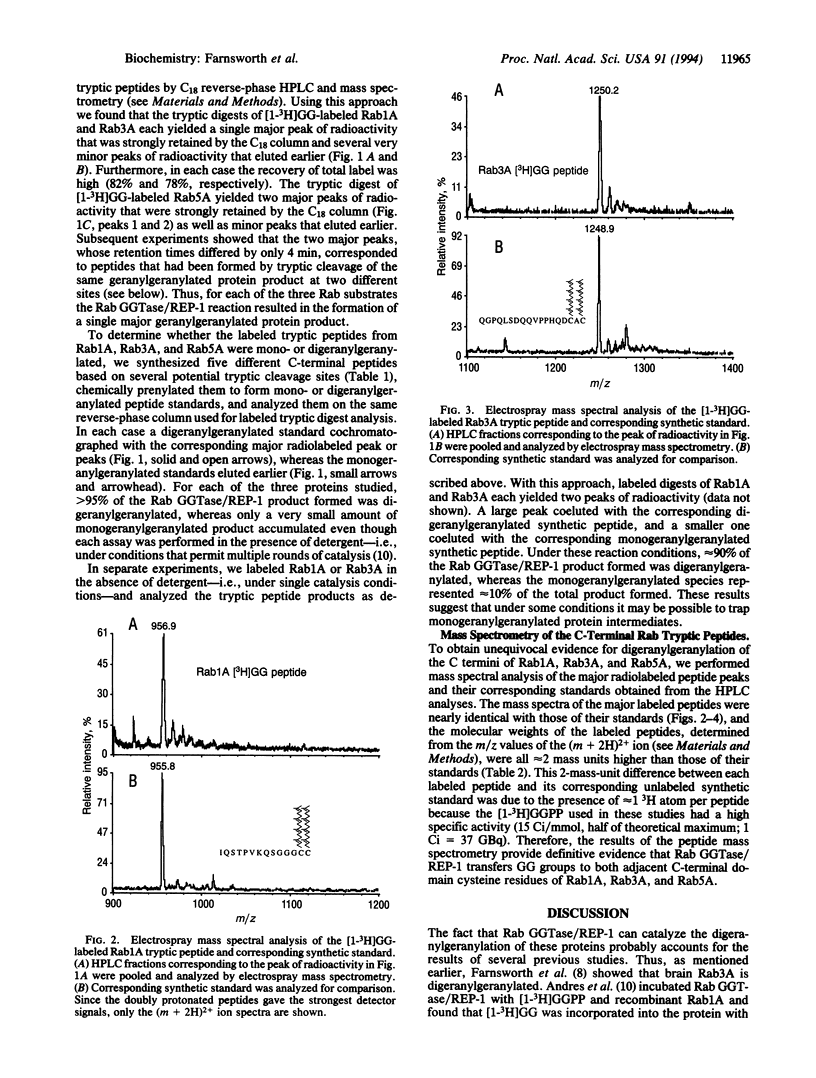
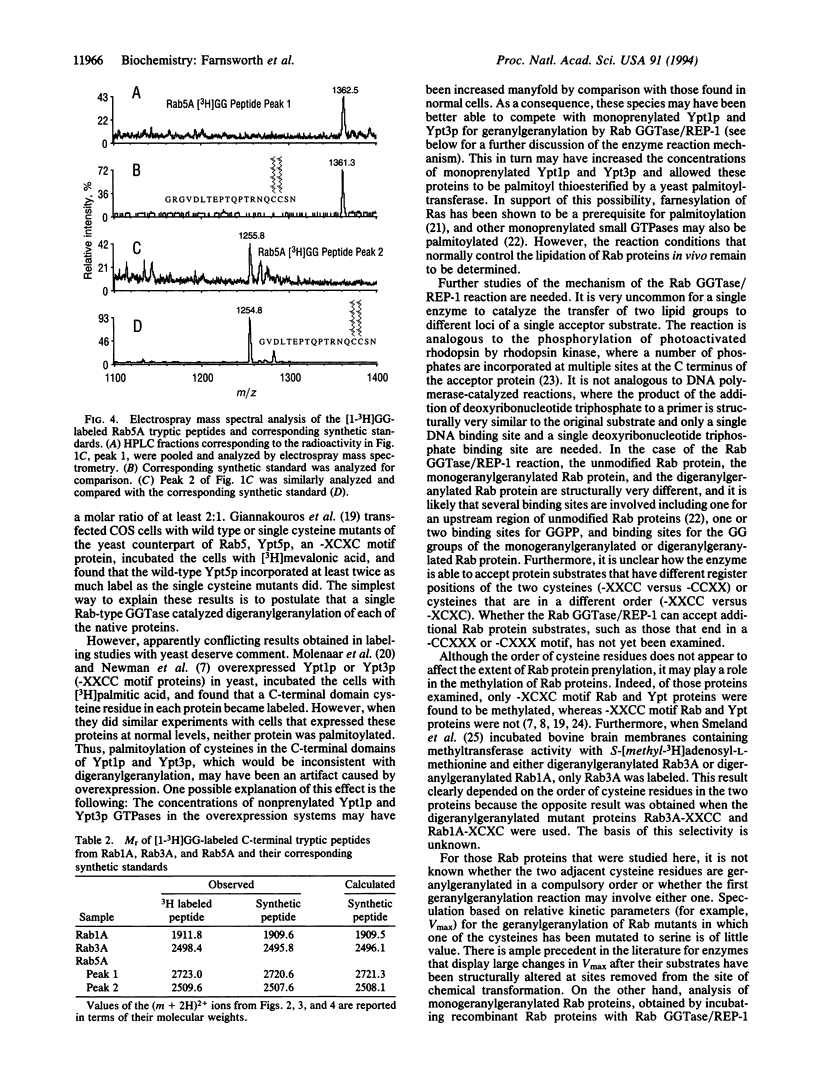
Abstract
Rab proteins are Ras-related small GTPases that are geranylgeranylated on cysteine residues located at or near their C termini. They differ from other geranylgeranylated small GTPases in several important respects. (i) Most Rab proteins contain two adjacent cysteine residues within one of the following C-terminal sequence motifs: -XXCC, -XCXC, or -CCXX; (ii) a Rab protein that ends in a -XCXC motif has been shown to be geranylgeranylated on both adjacent cysteine residues; and (iii) Rab proteins are substrates of a unique Rab-specific geranylgeranyltransferase. Whether this enzyme catalyzes the geranylgeranylation of both cysteines is unknown. We addressed this question by direct structural analysis of in vitro prenylated proteins. We incubated recombinant Rab geranylgeranyltransferase, Rab escort protein, and [1-3H]geranylgeranyl pyrophosphate with recombinant wild-type Rab1A (-XXCC), Rab3A (-XCXC), or Rab5A (-CCXX) and treated each labeled protein with trypsin. We then analyzed the resulting peptides by HPLC and electrospray mass spectrometry and found that for each protein both C-terminal adjacent cysteines were geranylgeranylated. These results indicate that Rab geranylgeranyltransferase/Rab escort protein catalyzes the geranylgeranylation of both cysteines in Rab proteins with three distinct C-terminal motifs and suggest that other Rab proteins with these motifs may be similarly modified.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Andres D. A., Seabra M. C., Brown M. S., Armstrong S. A., Smeland T. E., Cremers F. P., Goldstein J. L. cDNA cloning of component A of Rab geranylgeranyl transferase and demonstration of its role as a Rab escort protein. Cell. 1993 Jun 18;73(6):1091–1099. doi: 10.1016/0092-8674(93)90639-8. [DOI] [PubMed] [Google Scholar]
- Chavrier P., Parton R. G., Hauri H. P., Simons K., Zerial M. Localization of low molecular weight GTP binding proteins to exocytic and endocytic compartments. Cell. 1990 Jul 27;62(2):317–329. doi: 10.1016/0092-8674(90)90369-p. [DOI] [PubMed] [Google Scholar]
- Cremers F. P., Armstrong S. A., Seabra M. C., Brown M. S., Goldstein J. L. REP-2, a Rab escort protein encoded by the choroideremia-like gene. J Biol Chem. 1994 Jan 21;269(3):2111–2117. [PubMed] [Google Scholar]
- Cremers F. P., van de Pol D. J., van Kerkhoff L. P., Wieringa B., Ropers H. H. Cloning of a gene that is rearranged in patients with choroideraemia. Nature. 1990 Oct 18;347(6294):674–677. doi: 10.1038/347674a0. [DOI] [PubMed] [Google Scholar]
- Farnsworth C. C., Kawata M., Yoshida Y., Takai Y., Gelb M. H., Glomset J. A. C terminus of the small GTP-binding protein smg p25A contains two geranylgeranylated cysteine residues and a methyl ester. Proc Natl Acad Sci U S A. 1991 Jul 15;88(14):6196–6200. doi: 10.1073/pnas.88.14.6196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giannakouros T., Newman C. M., Craighead M. W., Armstrong J., Magee A. I. Post-translational processing of Schizosaccharomyces pombe YPT5 protein. In vitro and in vivo analysis of processing mutants. J Biol Chem. 1993 Nov 15;268(32):24467–24474. [PubMed] [Google Scholar]
- Glomset J. A., Farnsworth C. C. Role of protein modification reactions in programming interactions between ras-related GTPases and cell membranes. Annu Rev Cell Biol. 1994;10:181–205. doi: 10.1146/annurev.cb.10.110194.001145. [DOI] [PubMed] [Google Scholar]
- Hancock J. F., Magee A. I., Childs J. E., Marshall C. J. All ras proteins are polyisoprenylated but only some are palmitoylated. Cell. 1989 Jun 30;57(7):1167–1177. doi: 10.1016/0092-8674(89)90054-8. [DOI] [PubMed] [Google Scholar]
- Khosravi-Far R., Lutz R. J., Cox A. D., Conroy L., Bourne J. R., Sinensky M., Balch W. E., Buss J. E., Der C. J. Isoprenoid modification of rab proteins terminating in CC or CXC motifs. Proc Natl Acad Sci U S A. 1991 Jul 15;88(14):6264–6268. doi: 10.1073/pnas.88.14.6264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kinsella B. T., Maltese W. A. rab GTP-binding proteins implicated in vesicular transport are isoprenylated in vitro at cysteines within a novel carboxyl-terminal motif. J Biol Chem. 1991 May 5;266(13):8540–8544. [PubMed] [Google Scholar]
- Kinsella B. T., Maltese W. A. rab GTP-binding proteins with three different carboxyl-terminal cysteine motifs are modified in vivo by 20-carbon isoprenoids. J Biol Chem. 1992 Feb 25;267(6):3940–3945. [PubMed] [Google Scholar]
- Molenaar C. M., Prange R., Gallwitz D. A carboxyl-terminal cysteine residue is required for palmitic acid binding and biological activity of the ras-related yeast YPT1 protein. EMBO J. 1988 Apr;7(4):971–976. doi: 10.1002/j.1460-2075.1988.tb02903.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newman C. M., Giannakouros T., Hancock J. F., Fawell E. H., Armstrong J., Magee A. I. Post-translational processing of Schizosaccharomyces pombe YPT proteins. J Biol Chem. 1992 Jun 5;267(16):11329–11336. [PubMed] [Google Scholar]
- Novick P., Brennwald P. Friends and family: the role of the Rab GTPases in vesicular traffic. Cell. 1993 Nov 19;75(4):597–601. doi: 10.1016/0092-8674(93)90478-9. [DOI] [PubMed] [Google Scholar]
- Palczewski K., Benovic J. L. G-protein-coupled receptor kinases. Trends Biochem Sci. 1991 Oct;16(10):387–391. doi: 10.1016/0968-0004(91)90157-q. [DOI] [PubMed] [Google Scholar]
- Rossi G., Yu J. A., Newman A. P., Ferro-Novick S. Dependence of Ypt1 and Sec4 membrane attachment on Bet2. Nature. 1991 May 9;351(6322):158–161. doi: 10.1038/351158a0. [DOI] [PubMed] [Google Scholar]
- Seabra M. C., Brown M. S., Goldstein J. L. Retinal degeneration in choroideremia: deficiency of rab geranylgeranyl transferase. Science. 1993 Jan 15;259(5093):377–381. doi: 10.1126/science.8380507. [DOI] [PubMed] [Google Scholar]
- Seabra M. C., Brown M. S., Slaughter C. A., Südhof T. C., Goldstein J. L. Purification of component A of Rab geranylgeranyl transferase: possible identity with the choroideremia gene product. Cell. 1992 Sep 18;70(6):1049–1057. doi: 10.1016/0092-8674(92)90253-9. [DOI] [PubMed] [Google Scholar]
- Seabra M. C., Goldstein J. L., Südhof T. C., Brown M. S. Rab geranylgeranyl transferase. A multisubunit enzyme that prenylates GTP-binding proteins terminating in Cys-X-Cys or Cys-Cys. J Biol Chem. 1992 Jul 15;267(20):14497–14503. [PubMed] [Google Scholar]
- Simons K., Zerial M. Rab proteins and the road maps for intracellular transport. Neuron. 1993 Nov;11(5):789–799. doi: 10.1016/0896-6273(93)90109-5. [DOI] [PubMed] [Google Scholar]
- Smeland T. E., Seabra M. C., Goldstein J. L., Brown M. S. Geranylgeranylated Rab proteins terminating in Cys-Ala-Cys, but not Cys-Cys, are carboxyl-methylated by bovine brain membranes in vitro. Proc Natl Acad Sci U S A. 1994 Oct 25;91(22):10712–10716. doi: 10.1073/pnas.91.22.10712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wei C., Lutz R., Sinensky M., Macara I. G. p23rab2, a ras-like GTPase with a -GGGCC C-terminus, is isoprenylated but not detectably carboxymethylated in NIH3T3 cells. Oncogene. 1992 Mar;7(3):467–473. [PubMed] [Google Scholar]
- Zahraoui A., Touchot N., Chardin P., Tavitian A. The human Rab genes encode a family of GTP-binding proteins related to yeast YPT1 and SEC4 products involved in secretion. J Biol Chem. 1989 Jul 25;264(21):12394–12401. [PubMed] [Google Scholar]



