Abstract
Purpose
This formative research study describes the development and preliminary evaluation of a theory-guided, on-line multimedia psycho-educational program (PROGRESS) designed to facilitate adaptive coping among prostate cancer patients transitioning from treatment into long-term survivorship.
Methods
Guided by the Cognitive-Social Health Information Processing Model (C-SHIP) and using health communications best practices, we conducted a two phase, qualitative formative research study with early stage prostate cancer patients (n=29) to inform the web program development. Phase 1 included individual (n=5) and group (n=12) interviews to help determine intervention content and interface. Phase 2 employed iterative user/usability testing (n=12) to finalize the intervention. Interview data were independently coded and collectively analyzed to achieve consensus.
Results
Survivors expressed interest in action-oriented content on: (1) managing treatment side effects; (2) handling body image and co-morbidities related to overweight/obesity; (3) coping with emotional and communication issues; (4) tips to reduce disruptions of daily living activities, and (5) health skills training tools. Patients also desired the use of realistic and diverse survivor images.
Conclusions
Incorporation of an established theoretical framework, application of multimedia intervention development best practices, and an evidence-based approach to content and format, resulted in a psycho-educational tool that comprehensively addresses survivors' needs in a tailored fashion.
Implications for Cancer Survivors
The results suggest that an interactive web-based multimedia program is useful for survivors if it covers the key topics of symptom control, emotional well-being, and coping skills training; this tool has the potential to be disseminated and implemented as an adjunct to routine clinical care.
Keywords: Prostate Cancer, Survivorship, Web-based Health Intervention Development, Patient Activation, C-SHIP model, Health Adaptation and Surveillance
Introduction
Prostate cancer (Pca) is the most common solid tumor malignancy among American men, and it is estimated that 233,000 new cases of Pca will be diagnosed in 2014 [1]. Approximately 91% of new Pca cases are diagnosed in stages I or II, where the tumor is localized to the prostate [2]. Given the advances in technology for early diagnosis and treatment, the five-year survival rate for patients with early stage Pca is nearly 100% [3]. There were approximately 2.78 million men living with the disease in the U.S. in 2010, and this number has been steadily growing over the past 10 years [4]. Although men diagnosed with very early stage Pca can elect active surveillance as an option, the majority still choose treatment [5] with fewer than 10% of low-risk prostate cancer patients pursuing a delayed treatment approach [6]. Treatment of Pca has significant quality of life consequences for survivors and their families, warranting public health attention. In response, several organizations (including the American Society of Clinical Oncology and the National Comprehensive Cancer Network) have issued prostate cancer treatment guidelines. Most recently, the American Cancer Society (ACS) in July 2014 issued prostate cancer clinical care guidelines for primary care providers for long-term survivorship care emphasizing the need to address physical and psychosocial needs of prostate cancer survivors to enhance their survivorship experience [7]. These developments emphasize the need for scientifically sound survivorship resources to address Pca survivors' quality of life and transition of care concerns.
Regardless of the primary treatment chosen (i.e., prostatectomy or radiation therapy), Pca patients typically experience considerable physical, emotional, interpersonal, and practical challenges. These challenges include physical side effects, such as urinary, sexual and bowel dysfunctions [8]; psycho-social changes, such as anxiety, depression, and adjustment problems [9-13]; interpersonal issues, such as difficulties in patient-provider communication and disruption of intimate relationships with spouses and partners due to sexual dysfunctions and disease experiences [14, 15]; and practical issues, such as financial strain, medical insurance for future medical care, and obtaining legal assistance in cases of discriminatory workplace practices [16-18]. Although these complications can be difficult for patients to manage, there has been a paucity of healthcare resources to address patients' informational and psychosocial needs, beginning with the transition from active treatment to survivorship [19-21]. Indeed, only a minority of oncology care facilities offer survivorship clinic services, and only patients who have survived cancer for at least two years are generally referred to these services. Pca survivorship resources are therefore needed to facilitate re-entry into healthy survivorship, which involves having relevant knowledge, accurate expectations, adaptive emotions, and self-regulatory skills to manage life after Pca treatment [22, 23]. Internet and web-based educational platforms have enormous potential to address this void by serving as an adjunct to standard clinical care.
Access to the computer and the Internet is now ubiquitous and as of January 2014, 87% of American adults use the Internet [24]. For adults ages 50-64 years old, the usage remains high (88%), and even for those over 65 years old, over half (57%) use the Internet. Further, according to the latest data from the Pew Research Center's Internet & American Life Project, 35% of American adults report that they have gone online at least once, specifically to gain knowledge on what medical condition they or someone else might have. The advantages of website interventions include not only easy access, but also the ability to tailor information provided to the individual's post-treatment needs, to harness a variety of learning channels to enhance information processing (including multi-media approaches such as videos), and the option for patients to interact with the program in a dynamic and self-directed fashion that is optimal for patient-centered usage. Therefore, it is possible to easily reach diverse populations geographically and demographically and to provide appropriate, tailored and theory-guided informational content. While e-health interventions that focus on quality of life concerns of cancer survivors have been developed [25], a significant portion of this work has focused on breast cancer survivors [26-28] and/or has explored the use of the Internet as a forum for online-based support groups. The small number of eHealth programs that address prostate cancer survivorship are limited either in their focus (i.e., exclusively addressing treatment decision making [29] or sexuality/intimacy issues [30]), their functionality (i.e., not harnessing all of the currently available eHealth tools, e.g., interactivity, videos, audios, graphics, etc. [31, 32]), or their breadth of coverage (i.e., not addressing the full range of survivorship issues [33]).
Given the lack of e-health interventions currently available for Pca survivors at the transition from active treatment to survivorship, particularly those that include a comprehensive focus, functionality, and breadth of content, along with the high prevalence of Internet use among middle- to older-aged men, there is a missed opportunity to intervene by drawing on theory and evidence to address the informational, coping, and surveillance needs of early stage Pca survivors as they transition from treatment to long-term survivorship. To fill this gap in the care transition process, and to take advantage of the dissemination potential of use of the Internet, we report here the development of an innovative, theory-driven, web-based multimedia survivorship program designed to comprehensively address the Pca survivor-relevant domains of interest outlined above. This program, entitled the “Prostate Cancer Online Guide & Resource for Electronic Survivorship (PROGRESS)” is aimed at empowering Pca survivors to both effectively manage treatment-related side effects and adverse psychosocial sequelae, as well as to cope effectively with the interpersonal and practical challenges that are experienced as they transition from immediate to long-term survivorship. In addition, barriers identified by masculinity researchers [34-37] highlight a set of traditional masculinity scripts, including emotional stoicism, not asking questions, and following physician advice uncritically. By identifying and addressing these issues in a proactive manner, the goal is to improve patient knowledge, patient-physician communication, and ultimately health and wellness. The objective of this paper is to describe the rigorous development of PROGRESS, guided by evidence-based behavioral science theory [22, 23] and health communication (e.g., health literacy) best practices [38-40], and to present findings from the iterative focus groups/individual interviews and user/usability testing interviews.
Methods
The development process of PROGRESS was iterative and several of the steps described below occurred in tandem (see Figure 1): 1) comprehensive literature review and expert team members' input to determine major domains and content areas of Pca survivorship issues; 2) comprehensive review and identification of credible resources (on-line and printed) for the content areas; 3) development of text-based content based on credible resources and plain language writing style guide; 4) preliminary website interface design, such as layout, structure, graphics, coloring, etc.; 5) selection of communication channels (e.g. text, diagrams, video, audio, for different type of materials), based on expert team members' input and the extant literature; 6) collection of target users' feedback on the preliminary content areas and website design by focus groups and individual interviews; 7) development of the first version; 8) conduct of user/usability testing of the first version by individual interviews; 9) revision of the website based on user/usability testing results; and 10) further website revision and finalization.
Fig 1. Overall development and evaluation process of PROGRESS.
Guiding Theory
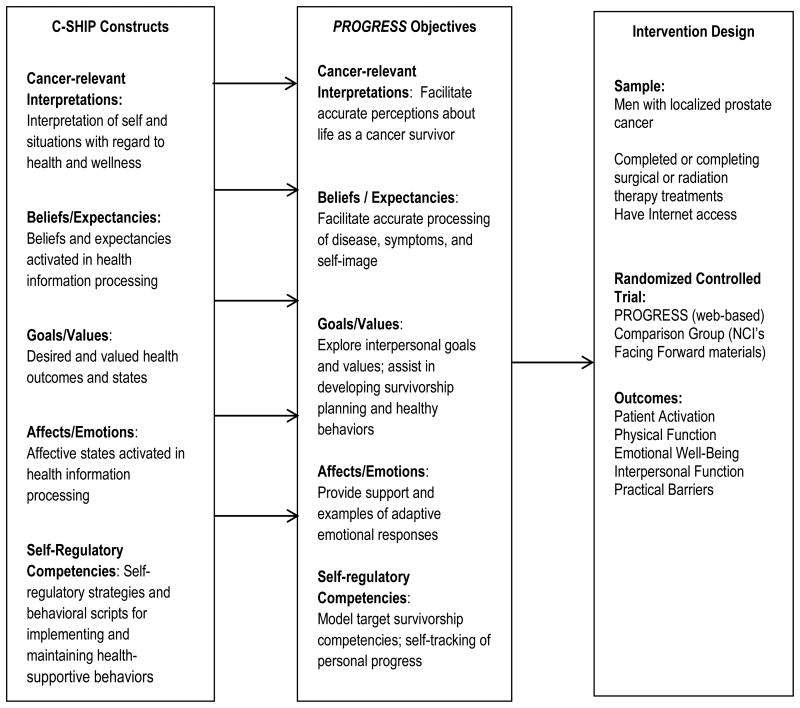
The content and design of PROGRESS was guided by the Cognitive-Social Health Information Processing Model (C-SHIP) (see Figure 2) developed by the research team, [41] which identifies the following key constructs of individual adaptation to health challenges: 1) cancer-relevant interpretations (i.e., interpretations of self and situations with regard to health; 2) belief/expectancies (i.e., beliefs/expectancies activated in health information processing; 3) goals/values (i.e.., desired and valued health outcomes and states); 4) affects/emotions (i.e., affective states activated in health information processing); and 5) self-regulatory competencies (e.g., self-regulatory strategies for implementing and maintaining health-supportive behaviors). We also based PROGRESS on prior C-SHIP guided work in cancer survivorship [42, 43] and coping. E-health intervention development strategies from the literature were also used to develop PROGRESS components (e.g., survivors' videos, stress management skills training), along with prior research that described application of theory and best practices in the design, development and preliminary testing of multimedia interventions, particularly two projects based on our prior work that developed eHealth platforms designed to support decision making prior to treatment of early stage prostate cancer patients, i.e., Healing Choices for Prostate Cancer [29] and the Prostate Interactive Educational System (PIES) [44], as well as two other projects of our team [28, 45].
Fig 2. Cognitive-Social Health Information Processing (C-SHIP) model as applied to the development of PROGRESS.
Preliminary content development and website design
Text Content Development
We first identified content domains to be addressed based on the extant literature and tailoring of the C-SHIP constructs to the issues for Pca survivorship, including : 1) Treatment type and their expected PSA changes, which indicate Pca status/progression; 2) Physical side effects (e.g. urinary and sexual dysfunction); 3) Emotional concerns (e.g. fear of recurrence); 3) Interpersonal concerns (e.g. communications with provider, family members); 4) Practical concerns (e.g. follow-up care, financial needs); and 5) Healthy lifestyle (e.g. nutrition, physical activities). A preliminary content map was developed and reviewed with collaborating physicians who treat Pca patients. See Supplemental Materials (On-line Resource 1) for the final full list of content areas under each domain.
The content map guided the writing process of all content of PROGRESS. An extensive process of identifying source material as well as original writing and revision was necessary since there are few vetted and validated resources for Pca survivors. In addition, extensive readability evaluation was conducted to ensure that the intervention met plain language standards. This evaluation included review of all content by members of the Office of Health Communications and Health Disparities (OHCHD) at FCCC, who have expertise in health literacy and communication. The software program, Health Literacy Advisor™, was employed to conduct this evaluation. This program calculates reading grade levels using the SMOG readability formula and also identifies, highlights and offers replacement text for complex terms and long sentences. All text was revised as needed to conform to an 8th-9th grade target level reading range and plain language best practices based on standards drawn from the literature [38-40] and medical and governmental agencies, (e.g., NCI, CDC, AHRQ, IOM, and AMA.)
Team members from the Office of Health Communications and Health Disparities at FCCC also provided training in plain language writing techniques to all study staff who participated in writing content. In addition, they identified a list of credible resources and provided writing templates for each content area. These templates enabled team staff to write the text content in a consistent format. The team staff at three sites (Fox Chase Cancer Center (FCCC), Icahn School of Medicine at Mount Sinai (ISMMS), and Rutgers University (RU)) were assigned different content areas and a series of drafts were reviewed and edited by the OHCHD health literacy team. The majority of the content was developed by the research team; some skill building components (e.g. stress reduction, communication with partners, lifestyle changes) in text format were adapted from current evidence-based materials on other reliable professional websites.
Multi-media Content Development
The goal of audio and visual materials included on the website was to enhance comprehension of the educational materials as well as to increase self-efficacy of adaptive coping skills. For example, diagrams related to the male reproductive system and male Kegel exercise were placed next to the related text content to facilitate learning. Clinicians' and survivors' videos were produced to discuss common questions and challenges faced by the survivors, and coping/treatment options. The clinicians were from a variety of specialties (e.g., male reproductive medicine, clinical psychology) to provide converging perspectives on survivorship issues. Survivors were chosen for video segments to reflect racial diversity and experiences with different treatment types. The survivors' video production emphasized personal challenges in Pca survivorship and realistic adaptive coping strategies. Several skills building components were also presented in multi-media format, such as stress management and Kegel exercise audios. Tracking tools in graphic format for self-monitoring of symptoms and healthy lifestyle changes were also included to facilitate action-oriented strategies over time.
Website Interface Design
The website was designed and produced in collaboration with a software development company, NotSoldSeparately (NSS), which specializes in developing health-related software and has previously collaborated with the team on cancer e-health programs. The design staff from NSS began by communicating with the team to gather information on the conceptualization of the website intervention, the structure and tailoring of content, and expectations about the functionality of the intervention. Then, they produced an initial wireframe or mock-up of the website interface. The team, particularly specialists from FCCC's OHCHD, provided input regarding the visual design of the website, such as color, layout of the homepage and sub-web pages, as well as formatting of content, tabs/message boxes/cross-links throughout the website, using health literacy best practices for designing easy-to-use websites. After about five iterations, a preliminary wireframe and related screenshots of the website were developed to collect target users' input.
Survivor Information Needs
An iterative process was implemented to understand survivor needs and gather their feedback throughout the development process. Phase 1 focused on the gathering of input with regard to website content and design. Phase 2 focused on feedback about usability of the initial prototype developed.
Participants
Participants were recruited at study sites in Philadelphia (FCCC) and New York City (ISMMS). A total of 49 eligible men were approached for the study. There were 20 who chose not to participate in the study. Not all explained their reasons for decline; however, for those who did, reasons for refusal included: 1) distance to interviews (n=5); 2) no interest (n=4); 3) busy/no time (n=3); 4) privacy (n=1); and, 5) participation in another study (n=1). Eligibility criteria included men who: (1) had a localized diagnosis of Pca at disease stage <T3; (2) were currently being treated or had completed Pca treatment within the past 12 months; (3) were 55 to 85 years of age; (4) were able to communicate readily in English; (5) were able to give consent; and (6) were within traveling distance to one of the study sites. Patients who chose to engage in active surveillance were not included in the study.
Phase 1: Individual and group interviews to guide web-based intervention development
Instruments
A semi-structured focus group/individual interview guide was developed. The lead moderator or interviewer asked open-ended, grand tour questions designed to elicit survivors' suggestions for content, their prostate cancer survivorship needs in terms of transitioning from active treatment and then over the long-term, their preferences for the website program, as well as their reactions to different program designs.
Procedure
The group interviews were conducted by a male lead moderator assisted by another male note-taker to facilitate rapport building [34, 35] and were audio-recorded. The individual interviews were also conducted by a male interviewer and were also audio-recorded. Participants received a $20 gift card as compensation for their time and input. All audio recordings were subsequently transcribed, cross-checked for accuracy and summarized. Interviews focused on the content needs and preferences of the participants. Specifically, they were asked open-ended questions regarding the challenges they faced in the domains of physical side effects, emotional concerns, interpersonal concerns, and practical concerns related to Pca survivorship transition. They gathered target users' reactions and feedback to the preliminary content components and interface design of PROGRESS, by showing them draft screenshots of the website wireframes. Information collected from each group/individual was progressively incorporated into the wireframe revisions as the focus groups/interviews were conducted, to assist the development of the first version of the website in terms of content, format, and interface design. The group interviews lasted for 90 minutes each, one with surgery patients, one with radiation patients, and one with mixed surgery/radiation patients. Individual interviews lasted for 20-30 minutes each.
Phase 2: User/Usability Testing
Following the focus groups and interviews, we continued to further develop iterations of the website in terms of both content and features. The research team worked to incorporate patients' expressed needs and preferences for pertinent information, tools, and website features targeted to the population. After the first version of the intervention was completed, we conducted user/usability testing to ensure acceptability of formatting/design, relevance, and functionality of the website components. The goals of testing were to determine need for further content and format modification and fine-tuning of the website intervention.
Instruments
A user/usability testing guide was developed by FCCC OHCHD to detail the instructions and procedures for conducting the individual interviews. The guide design was based on the literature and NCI usability guidelines [46], as well as best practice models such as those described on NCI's website www.usability.gov. It included an introduction, consenting, the purpose of the testing and the evaluation process, as well as other open-ended questions for participants as they navigated the website for the first time. Participants were encouraged to visit any website areas that interested them, take as much time as they desired, give overall impressions of the website, and report which website aspects they liked best and least. A sample question was “Without clicking on anything, tell me what you expect to find in each section (point to the tabs at the top of the page)?”
Procedure
Each interview lasted for approximately 20-80 minutes, and it was conducted by a lead interviewer and audio-recorded. A note-taker was present to record the observations of the users' interactions (mainly non-verbal ones) with the website. Participants were asked to “Think Aloud” [47] as they used the website so that insights into their thought processes could be gathered. The lead interviewer observed participants' behaviors and comments during specific pre-determined questions on the interview guide, and asked user-specific questions based on difficulties/behaviors observed while the participant navigated the website. Morae software was available for user/usability interviews and used at the FCCC site only to record participants' mouse movements on the computer screen. Participants received a $20 gift card as compensation for their time and input. Audio recordings were subsequently transcribed, cross-checked for accuracy by three research staff members, and the testing findings summarized by the OHCHD specialist. User feedback on acceptability, usefulness and functionality of the intervention components was used to facilitate revision and refinement of the program prototype.
Usability testing was conducted with 12 participants. Based on the testing results, the research team made significant improvements to the web program content and incorporated key suggestions into the video clips of the website. Staff also provided feedback based on their observations of participants and the functionality of the website. Table 3 provides a list of revisions made to the website, organized by C-SHIP constructs and the relevant website components. When no further changes were suggested by the user-testing participants, the PROGRESS website was finalized. Supplemental Materials including the screenshots of the final PROGRESS homepage and other webpages for the major domains of the program are included in an on line appendix (see On-line Resource 1).
Table 3. Modification to PROGRESS prototype based on C-SHIP model and user/usability testing results.
| C-SHIP Constructs | Content Topic | Reactions | Functionality of Website/Incorporation of Feedback into Website |
|---|---|---|---|
| Cancer-relevant Interpretations | Treatment side effects | More information about radiation |
|
| Information on incontinence | |||
| Beliefs/Expectancies | Images | Make certain that photos and videos on the site reflect “regular” people and “people who look like me” |
|
| Survivorship Care Plan | Mixed reaction to providing an SCP. Some thought they would be useful. Others felt they duplicated systems they had already created to track their care. | ||
| Suggested calling the SCP a “tool for survivorship” | |||
| Goals/Values | Tailoring Content | Wanted content organized in a way that was relevant based on their treatment choice |
|
| Wanted content organized based on time from treatment | |||
| Affects/Emotions | Psychosocial Issues | Fear of recurrence |
|
| PSA anxiety | |||
| Self-Regulatory Competencies | Instructional Videos | Wanted an overview of the website at first log-in to help navigation |
|
| Wanted clearer instructions for the skills training components, like breathing exercises and Kegel exercise | |||
| Wanted credential information of physicians and health care providers on the videos | |||
| Tracking tools | Mixed reaction to tracking tools, some thought they would be useful, others felt they had already established methods to track PSA | ||
| Wanted clearer instructions on accessing and using the tracking tools | |||
| Practical Advice | Information on Kegel Exercises |
|
|
| Information on relaxation techniques (i.e., breathing exercises) | |||
| Information about how to plan for travel (i.e., carrying extra pads and wipes to cope with incontinence) |
Analyses
Both qualitative and descriptive quantitative methods were used to collect and analyze the development and preliminary evaluation data for PROGRESS. Phase 1 data were initially qualitatively analyzed using an inductive, crystallization-immersion process [48] followed by a content analytic process [49] once inductive themes had been identified. A group of 5 initial interviews (30% of the sample) were reviewed together by two researchers (SVH and AN) with qualitative expertise in sociology and health education and behavioral science research methods. They met weekly to discuss key issues that emerged from the transcripts, and through a consensus seeking process, identified a common list of codes and definitions, which were then applied to the analysis of the remaining Phase 1 interviews. The Phase 2 data were qualitatively analyzed using content analytic methods of data review, analysis and data reduction. Quantitative analysis was employed using descriptive statistics were generated using SPSS version 21 to describe and evaluate the socio-demographic characteristics of the study sample, as well as the usage patterns for the Phase 2 user/usability testing.
Results
Participant Characteristics
Table 1 presents separately the demographic characteristics of participants in Phases 1 and Phase 2 of the study, as well as the overall sample characteristics (N=29). It is noteworthy that African-Americans were oversampled (29%), and 50% of participants reported using the Internet daily, with 75% of them reporting searching the Internet sometimes or often for health information.
Table 1. Characteristics of study sample.
| Total (N=29) |
Phase 1 Website Development (Individual and Group Interviews) (n=17) |
Phase 2 User/Usability Testing (n=12) | ||||
|---|---|---|---|---|---|---|
| Frequency (n) |
Percentage (%) |
Frequency (n) |
Percentage (%) |
Frequency (n) |
Percentage (%) |
|
| Age | ||||||
| Median Range |
64.5 52-79 |
64.3 52-75 |
64.8 56-79 |
|||
| Race* | ||||||
| White | 18 | 64.3 | 12 | 75.0 | 6 | 50.0 |
| African-American | 8 | 28.6 | 2 | 12.5 | 6 | 50.0 |
| Other | 2 | 7.1 | 2 | 12.5 | ||
| Ethnicity* | ||||||
| Hispanic | 2 | 8.7 | 2 | 12.5 | ||
| Non-Hispanic | 21 | 91.3 | 14 | 87.5 | 7 | 100.0 |
| Treatment Type* | ||||||
| Radiation | 12 | 42.9 | 9 | 56.3 | 3 | 25.0 |
| Surgery | 16 | 57.1 | 7 | 43.7 | 9 | 75.0 |
| Years Since Tx* | ||||||
| 1 year | 11 | 50.0 | 4 | 26.7 | 7 | 100.0 |
| 2 years | 11 | 50.0 | 11 | 73.3 | ||
| Internet Use | ||||||
| Sometimes | --- | --- | --- | --- | 6 | 50.0 |
| Daily | --- | --- | --- | --- | 6 | 50.0 |
| Search Internet for Health Information | ||||||
| Sometimes | --- | --- | --- | --- | 8 | 66.7 |
| Often | --- | --- | --- | --- | 1 | 8.3 |
| Never | --- | --- | --- | --- | 3 | 25.0 |
There were individuals who preferred not to provide information concerning these variables; percentages reported concerning these variables are based only on participants who responded (therefore exclude consideration of missing data).
Results from each step in the developmental process
Phase 1 (Individual and Group Interviews)
Seventeen early stage Pca survivors were recruited from FCCC and ISMMS to gather information about patients' views on the post-treatment experience and their preferences to guide further program content development, interface design, and when to best implement the intervention. In this Phase of the study, we tested the preliminary development of PROGRESS iteratively through formative evaluation (i.e., individual and group interviews and continued expert input and stakeholder group feedback) to further develop the website content and layout. Examples of participant feedback and the corresponding survivorship life domains are shown in Table 2. Patients expressed physical functioning concerns that focused around managing treatment side effects and other health concerns and suggested they wanted information on health behaviors and practical advice to manage physical issues. They also discussed emotional well-being including body image and psychosocial concerns related to increased perceived susceptibility to cancer and fears of disease recurrence. Participants' also articulated interpersonal concerns (e.g., disruption of relationships with spouse/partner) and practical challenges (e.g., discriminatory workplace practices) and suggested a need for resources to address these issues. For example, they described needs for information to help deal with partners and changing self-perceptions. Surgical patients expressed a desire to understand the impact of pre-existing and newly developed co-morbidities on their recovery, while radiation patients expressed concerns about the radioactive nature of treatment. This feedback was crucial in developing new text and skills training content to address these concerns. We integrated the content developed with the initial content to produce the prototype web program, which was subsequently user-tested in Phase 2.
Table 2. Cancer-relevant content issues cited among Phase 1 participants.
| Survivorship Life Domain | Topic | Proposed Content | Quote |
|---|---|---|---|
| Physical Function | Practical Advice | Information on Kegel Exercises | “..how to do a Kegel exercise right. I think it's important” |
| Information on relaxation techniques | Desired content on, “those different exercises and breathing exercises” | ||
| Health Concerns | Impact of Pca on other co-morbid conditions | “I asked my family doctor to take care of my diabetes for me…he said that he would, but I'd have to go to the (endocrinologist) at least once a year” | |
| Treatment Side Effect Concerns | Hot Flashes | “I had a little bit of difficulty because it's…the hormonal treatment…if I get to the point where I'm having excessive hot flashes and I'm doing all this, I'd quit” | |
| Incontinence and urinary function | “surgery always causes impotence, incontinence ….because everybody is a different” | ||
| Sexual dysfunction | “I'm 64 so I'd still like to go and have sex…there's pain… I spoke with my doctor…and…told him that I have blood that came with the semen and this is normal because you went through the procedure” | ||
| Fatigue | “I believe that fatigue is part of the treatment, what they did” | ||
| Health Behaviors | Physical Activity | “…the best thing that they told me to do was walk. And the day after the operation, I started to walk. And it kept the weight off” | |
| Emotional Well-Being | Body Image | Weight issues | “I'm overweight…I have some anxiety about how my performance is gonna be when I'm totally feeling better” |
| Concerns about incontinence and how to manage daily challenges due to treatment side effects | “..information…given to me in terms of …incontinence…right on the money” | ||
| Psychosocial Issues | Anxiety about rising PSA | “You've got PSA anxiety… I imagined other people probably have the same issue” | |
| Communication issues with family and partners | “…about communication with your spouse…I think the doctor would be able to help if you said that you're having a problem” | ||
| Increased perceived susceptibility | “I felt bad because I always felt invulnerable” | ||
| Interpersonal Concerns | Disruption of relationships with spouse/partner | Resources to offer emotional support to spouse | “Your wife, spouse, sometimes they take it a little harder than the men do…I think that would be very helpful to have some type of support information available to know what you expect” |
| Impact of treatment side effects on relationships | “We're in this relationship for about five years …before I had this operation or even knew that I had the cancer, we were very active. And it's fallen off…some things aren't working the way they used to” | ||
| Practical Barriers | Discriminatory workplace practices | Employment discrimination after diagnosis | “I had a very practical concern in that I got fired right after my employer found out that I had cancer and required an operation” |
Phase 2 (User/Usability Testing)
Twelve early stage prostate cancer survivors were recruited from FCCC and ISMMS to pre-test and give feedback on the revised web program content, visuals, approaches, and functionality. Table 3 shows illustrative user feedback from user testing and modifications our research team employed to address them, guided by the relevant C-SHIP theory constructs. For example, self-efficacy and self-regulatory competencies were addressed using a multi-faceted approach that included selection of images, revised instructions on skills training videos/audios, and tracking tools. First, given the desire for a simple but comprehensive interface that reflected the diversity of patients (e.g., age, weight status, race/ethnicity), we added more “everyday looking” survivors and their friends/families in the graphics on the web program. Second, to address patients' immediate and long-term survivorship beliefs and expectations in a way that survivors could relate to, “My Survivorship Care Plan” was re-named “My Care Plan” and the content under that tab was re-organized (e.g., healthy lifestyle change information and tools were separated from treatment plan summary or other health care needs). Third, to ensure that participants' goals and values were taken into account, we simplified the list of website topics that the user could select as “My topics” by re-categorizing/shortening the list and removing topics that were more relevant to the pre-treatment stage. Affects and emotions were addressed by providing normalization and support in the text content, and providing coping tools such as graphs for tracking weight and Prostate-Specific Antigen (PSA) levels for monitoring recurrence risk. Finally, cancer-relevant interpretations were facilitated by editing titles of video clips to better reflect the content of the videos, presenting videos categorized by radiation or surgery treatment types, categorizing text or video content into their corresponding survivorship time periods (1-2 months, 3-4 months, 5-6 months and beyond since treatment completion), and providing detailed information and management tips for treatment-related side effects.
Preliminary Usage
The user/usability testing interviews provided preliminary usage data on PROGRESS. Participants (n=12) were encouraged to freely navigate the website according to their interests and take as much time as they wished to view or use the content. In testing sessions, the median time spent per participant testing the entire web program was 46 minutes (range of 20-79 minutes). Certain webpages received more attention than others. For example, participants spent the most time, from 3-23 minutes browsing content in the Topics section. In all user/usability testing sessions, 67% of participants visited all tabs/sections of the website, with 25% of participants visiting the Topics tab first and 42% of participants visiting the Track My Progress tab first. In addition, one-third of the participants in the user/usability testing sessions watched the “What to expect” videos first. Participants said they found the information to be “comprehensive” and “helpful.”
Discussion
The PROGRESS program was developed to facilitate the transition from treatment to survivorship phase and to enhance social and emotional coping of a diverse group of early-stage prostate cancer survivors. We followed an iterative and theory-guided developmental process, along with a multidisciplinary expert team approach to develop the evidenced-informed multimedia prostate cancer survivorship web program. The C-SHIP theoretical framework [22] guided the selection of content, and a rigorous, user-centered process, based on health communications best practices, was employed to design the computer interface. This formative research adds to the emerging and existing literature [29, 44, 45] on best practices in the development of user-centered, e-health tools, as well as provides a unique contribution to the cancer survivorship literature focused on the comprehensive psychosocial management of prostate cancer survivors. The program thus serves as a prototype for assessing and addressing prostate cancer survivors' needs and goals as they transition from active treatment and experience the evolving challenges of the survivorship experience.
The PROGRESS website was designed to facilitate engagement from men holding a number of different assumptions about what it means to be men in terms of their socially constructed masculinity scripts [34, 35, 50-52]. Key masculinity scripts voiced by participants in this study included reticence regarding seeking external assistance with the sequelae associated with prostate cancer as well as some active avoidance of emotional support. Such masculinity scripts pose significant challenges to the use of quality of life and self-management interventions. As a result, they need to be addressed. We therefore chose to address these scripts using a variety of strategies throughout the PROGRESS website.
Notably, prostate cancer survivors in this study reported a number of unmet needs that they felt a program like PR0GRESS had the potential to mitigate. In particular, patients wanted a variety of concrete strategies for managing treatment side effects such as fatigue and sexual dysfunction, addressing body image-related concerns and co-morbidities related to overweight/obesity, resolving psychosocial issues such as communication with family and partners and feelings of increased susceptibility, and dealing with disruptions to daily living activities. These observations provided key insights into how to best tailor the program to meet patient goals, by fostering and enhancing the active self-management component of the PROGRESS intervention to engage and sustain interest in the program. Even the choice of the program title was carefully weighed by the team in terms of what it denoted. The ultimate selection of the term PROGRESS is in line with, and highlights, the recognition of the desire for a “practical,” action-oriented approach to survivorship among prostate patients. This type of approach – which provides actionable content and engages users in ways that supports and encourages goal setting and self-management resources for a range of non-medical follow-up and behavioral and lifestyle challenges – is consistent with our findings on the use of print materials such as Facing Forward [53]. We found that the most preferred aspect of the material was the component that stresses self-management and coping strategies [54, 55], which differs somewhat from the main thrust of existing programs for early stage breast cancer patients.
In addition to self-management issues, participants in this study articulated the importance of targeting and tailoring content to be personally relevant to their survivorship journey in several different ways. Their suggestion that entry into the website be categorized by treatment type, as well as time from treatment completion, provided insight on how to present and organize intervention content and the importance of modifying the content as their survivorship experience, needs, and challenges evolved over time. This input supports the emerging literature on the importance of creating web-based cancer survivorship programs that address users' needs [32, 33, 49] specific to their diagnosis and treatment plan. Additionally, patient feedback on their desire to see patient-relevant images and visual representations of the material incorporated in to the website reinforces findings from the breast cancer survivorship literature [56] that it is important for patients to feel like they can relate to the content presented. For example, some of the original stock images of men and situations used in our website drafts elicited responses such as “they do not look like me” or “they look too happy.” Based on this feedback, the finished product was systematically modified to more accurately represent the diversity of prostate cancer survivors in terms of such factors as race/ethnicity, weight status, expression of mood, attractiveness, etc. By making the web interface more personally relatable our goal was to provide an intervention that is better positioned to promote deeper social cognitive processing to promote coping. These results are consistent with the masculinity literature that it is important to recognize that there are multiple masculinities which are culturally bounded by factors such as race/ethnicity and socioeconomic status [50]. They are also consistent with the findings of other studies which suggest that eHealth and mHealth interventions need to be guided by a patient-centered perspective where the needs and desires of the users are front and center in the development, as well as in the implementation, process [32-34].
In addition to contributing to the research literature on best practices in the development of user-centered, e-health tools on best practices [45], this study fills an important gap in the prostate cancer survivorship literature, given the limited on-line resources available for men dealing with prostate cancer post-treatment that incorporate clinical recommendations for best practice in the care of long-term prostate cancer survivors [7]. In comparison with breast cancer, there are few eHealth resources for men with prostate cancer to access. To our knowledge, there are only a handful of eHealth programs for men with prostate cancer [29-31, 33, 44]. All of them were developed prior to the development of ACS' long-term prostate cancer survivorship care guidelines. The PROGRESS website is the only web-based intervention that addresses all of the domains specified in the new ACS guidelines, from a patient-centered perspective, and therefore adds significantly to the resource base for prostate cancer survivors who are transitioning from active treatment into long-term survivorship.
While our study provides important, new information on the design of a theory informed and patient-guided intervention for an underserved survivorship group, there are a few limitations to note. First, our sample size is small and socioeconomic status (i.e., income, education, occupation) of participants was not systematically collected; therefore, it is difficult for us to address the representativeness of the patient population. Second, the web-based program is in English and therefore all of the participants in the user testing were English-speaking. Third, information was not systematically collected concerning receipt of hormone therapy. Finally, while we are currently limited in terms of our ability to discuss dissemination and reach, we are presently testing this intervention at several clinical sites with a racially and ethnically diverse population in a randomized controlled trial. If proven to have acceptability, utility and efficacy, the results would warrant additional study in terms of implementation and dissemination in a broad range of clinical and community settings. In future research it will also be important to integrate the PROGRESS support system with the clinical health record and to evaluate its function.
This paper highlights the utilization of a theory-based and best practice approach, as well as a user-centered iterative process, in the development of an eHealth tool. The findings suggest the importance of using converging data derived from survivor and expert input to enhance the development, personal relevance, and tailoring of the multimedia content and approach. Perhaps most importantly, the self-management focus of PROGRESS offers a scalable, psychosocial intervention that goes beyond the approaches taken in existing research and clinical settings and has high potential as an adjunct to routine clinical survivorship care. This type of eHealth tool potentially supports the promise of eHealth programs to provide complementary support to patients within the clinical context and outreach to survivors beyond the health care system.
Supplementary Material
Acknowledgments
This research was supported by National Cancer Institute grants, R01 CA158019 to Dr. Miller and the Fox Chase Cancer Center Behavioral Research Core Facility P30-CA06927. Dr. Hudson was supported by the following awards from the National Cancer Institute: K01 CA 131500, R03 CA154063, and R01 CA176838. Dr. Hui was supported by National Cancer Institute grant R03 CA159903. Dr. Diefenbach was supported by the following awards from the National Cancer Institute: 1R01 CA158019-01, 1R21 CA155963, 1R21 CA164807; and W81XWH-11-1-0604, from the Department of Defense. We would like to acknowledge the contribution of James Williams and the patients who participated in this study or appeared in the patient videos for the web program development. We thank the clinicians who appeared in the videos, Drs. Natan Bar-Chama, Neil Grafstein, and Christian Nelson. Other research team members, including Margaret Atchison, Javier Muniz, Craig Walt, Megan Grau, Eric Shaw, Sean O'Sullivan, Martin Cohen, Jennifer Burns, and Matt Hall, also made contributions to this study. We also thank Mary Anne Ryan for her technical and administrative assistance. Last but not least, we thank the team members from NotSoldSeparately.com, Kevin Durr, Joe Ifi, Mayr Budny, Dan Alvare, Anthony Wojtkowiak, for their help in the technical process of the website development and implementation.
Footnotes
Conflict Of Interest The authors declare that they have no conflict of interest.
References
- 1.American Cancer Society. Cancer Facts & Figures 2014. Atlanta: American Cancer Society; 2014. p. 2014. [Google Scholar]
- 2.Jemal A, et al. Cancer statistics, 2009. CA Cancer J Clin. 2009;59(4):225–49. doi: 10.3322/caac.20006. [DOI] [PubMed] [Google Scholar]
- 3.Siegel R, et al. Cancer treatment and survivorship statistics, 2012. CA Cancer J Clin. 2012;62(4):220–41. doi: 10.3322/caac.21149. [DOI] [PubMed] [Google Scholar]
- 4.A.A.C. Society, editor. Society., A.C. American Cancer Society. Cancer Treatment and Survivorship Facts & Figures 2014-2015. Atlanta: American Cancer Society; 2014. Atlanta, Georgia: 2012. [Google Scholar]
- 5.Anandadas CN, et al. Early prostate cancer--which treatment do men prefer and why? BJU Int. 2011;107(11):1762–8. doi: 10.1111/j.1464-410X.2010.09833.x. [DOI] [PubMed] [Google Scholar]
- 6.Singer EA, et al. Active surveillance for prostate cancer: past, present and future. Curr Opin Oncol. 2012;24(3):243–50. doi: 10.1097/CCO.0b013e3283527f99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Skolarus TA, et al. American Cancer Society prostate cancer survivorship care guidelines. CA Cancer J Clin. 2014;64(4):225–49. doi: 10.3322/caac.21234. [DOI] [PubMed] [Google Scholar]
- 8.Bacon CG, et al. The association of treatment-related symptoms with quality-of-life outcomes for localized prostate carcinoma patients. Cancer. 2002;94(3):862–71. doi: 10.1002/cncr.10248. [DOI] [PubMed] [Google Scholar]
- 9.Lintz K, et al. Prostate cancer patients' support and psychological care needs: Survey from a non-surgical oncology clinic. Psychooncology. 2003;12(8):769–83. doi: 10.1002/pon.702. [DOI] [PubMed] [Google Scholar]
- 10.Wootten AC, et al. Psychological adjustment of survivors of localised prostate cancer: investigating the role of dyadic adjustment, cognitive appraisal and coping style. Psychooncology. 2007;16(11):994–1002. doi: 10.1002/pon.1159. [DOI] [PubMed] [Google Scholar]
- 11.Bennett G, Badger TA. Depression in men with prostate cancer. Oncol Nurs Forum. 2005;32(3):545–56. doi: 10.1188/05.ONF.545-556. [DOI] [PubMed] [Google Scholar]
- 12.Chipperfield K, et al. Factors associated with adherence to physical activity guidelines in patients with prostate cancer. Psychooncology. 2013;22(11):2478–86. doi: 10.1002/pon.3310. [DOI] [PubMed] [Google Scholar]
- 13.Badger TA, et al. Who benefits from a psychosocial counselling versus educational intervention to improve psychological quality of life in prostate cancer survivors? Psychol Health. 2013;28(3):336–54. doi: 10.1080/08870446.2012.731058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Cegala DJ, Post DM. The impact of patients' participation on physicians' patient-centered communication. Patient Educ Couns. 2009;77(2):202–8. doi: 10.1016/j.pec.2009.03.025. [DOI] [PubMed] [Google Scholar]
- 15.Grunfeld E, Earle CC. The interface between primary and oncology specialty care: treatment through survivorship. J Natl Cancer Inst Monogr. 2010;2010(40):25–30. doi: 10.1093/jncimonographs/lgq002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Grant M, et al. Educating health care professionals to provide institutional changes in cancer survivorship care. J Cancer Educ. 2012;27(2):226–32. doi: 10.1007/s13187-012-0314-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bradley CJ, et al. Employment and cancer: findings from a longitudinal study of breast and prostate cancer survivors. Cancer Invest. 2007;25(1):47–54. doi: 10.1080/07357900601130664. [DOI] [PubMed] [Google Scholar]
- 18.Grunfeld EA, et al. “The only way I know how to live is to work”: a qualitative study of work following treatment for prostate cancer. Health Psychol. 2013;32(1):75–82. doi: 10.1037/a0030387. [DOI] [PubMed] [Google Scholar]
- 19.Boberg EW, et al. Assessing the unmet information, support and care delivery needs of men with prostate cancer. Patient Educ Couns. 2003;49(3):233–42. doi: 10.1016/s0738-3991(02)00183-0. [DOI] [PubMed] [Google Scholar]
- 20.Smith DP, et al. Age, health, and education determine supportive care needs of men younger than 70 years with prostate cancer. J Clin Oncol. 2007;25(18):2560–6. doi: 10.1200/JCO.2006.09.8046. [DOI] [PubMed] [Google Scholar]
- 21.Stanton AL. What happens now? Psychosocial care for cancer survivors after medical treatment completion. J Clin Oncol. 2012;30(11):1215–20. doi: 10.1200/JCO.2011.39.7406. [DOI] [PubMed] [Google Scholar]
- 22.Miller SM, D MA. The Cognitive-Social Health Information-Processing (C-SHIP_ Model: A theoretical Framework for Research in Behavioral Oncology. In: Krantz David S, B A., editors. Technology and Methods in Behaviroal Medicine. Mahwah, NJ: Lawrence Erlbaum Associates, Inc; 1998. pp. 219–44. 1998. [Google Scholar]
- 23.Miller SM, et al. Tailoring psychosocial interventions to the individual's health information-processing style: The influence of monitoring versus blunting in cancer risk and disease. In: Andersen ABBL, editor. Psychosocial interventions for cancer. American Psychological Association; Washington, DC, US: 2001. pp. 343–362. [Google Scholar]
- 24.Project, P.R.I. Internet User Demographics, As of January 2014, 87% of American adults use the internet. Internet and Tech. 2014 [cited 2014 April]; Available from: http://www.pewinternet.org/data-trend/internet-use/latest-stats.
- 25.Sanchez MA, et al. A systematic review of eHealth cancer prevention and control interventions: new technology, same methods and designs? Transl Behav Med. 2013;3(4):392–401. doi: 10.1007/s13142-013-0224-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Carpenter KM, et al. An online stress management workbook for breast cancer. J Behav Med. 2012 doi: 10.1007/s10865-012-9481-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Pauwels E, et al. Design and process evaluation of an informative website tailored to breast cancer survivors' and intimate partners' post-treatment care needs. BMC Res Notes. 2012;5:548. doi: 10.1186/1756-0500-5-548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Wen KY, et al. The development and preliminary testing of a multimedia patient-provider survivorship communication module for breast cancer survivors. Patient Educ Couns. 2012;88(2):344–9. doi: 10.1016/j.pec.2012.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Marcus AC, et al. Cancer patient and survivor research from the cancer information service research consortium: a preview of three large randomized trials and initial lessons learned. J Health Commun. 2013;18(5):543–62. doi: 10.1080/10810730.2012.743629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Schover LR, et al. A randomized trial of internet-based versus traditional sexual counseling for couples after localized prostate cancer treatment. Cancer. 2012;118(2):500–9. doi: 10.1002/cncr.26308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Van Bogaert D, et al. The Development of an eHealth tool suite for prostate cancer patients and their partners. J Support Oncol. 2012;10(5):202–8. doi: 10.1016/j.suponc.2012.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.McPherson DA, Herxheimer DA. healthtalk.org. [cited 2014; Available from: http://www.healthtalk.org.
- 33.Wootten A, et al. Development, feasibility and usability of an online psychological intervention for men with prostate cancwer: My Road Ahead. Internet Interventions. 2014;1 [Google Scholar]
- 34.Burns SM, Mahalik JR. Understanding how masculine gender scripts may contribute to men's adjustment following treatment for prostate cancer. Am J Mens Health. 2007;1(4):250–61. doi: 10.1177/1557988306293380. [DOI] [PubMed] [Google Scholar]
- 35.Wall D, Kristjanson L. Men, culture and hegemonic masculinity: understanding the experience of prostate cancer. Nurs Inq. 2005;12(2):87–97. doi: 10.1111/j.1440-1800.2005.00258.x. [DOI] [PubMed] [Google Scholar]
- 36.Mankowski ES, Maton KI. A community psychology of men and masculinity: historical and conceptual review. Am J Community Psychol. 2010;45(1-2):73–86. doi: 10.1007/s10464-009-9288-y. [DOI] [PubMed] [Google Scholar]
- 37.Mroz LW, Oliffe JL, Davison BJ. Masculinities and patient perspectives of communication about active surveillance for prostate cancer. Health Psychol. 2013;32(1):83–90. doi: 10.1037/a0029934. [DOI] [PubMed] [Google Scholar]
- 38.Rudd RE. Guidelines for Creating Materials-Resources for Developing and Assessing Materials. 2014 Available from: www.hsph.harvard.edu/healthliteracy.edu.
- 39.Osborne H. Health Literacy From A to Z, Second Edition: Practical Ways to Communicate your Health Message. Burlington, MA: Jones&Bartlett Learning; 2011. [Google Scholar]
- 40.Doak C, Doak L, Root J. Teaching patients with low literacy skills 1985. Philadelphia: JB Lippincott; [Google Scholar]
- 41.Venderbos LD, et al. A longitudinal study on the impact of active surveillance for prostate cancer on anxiety and distress levels. Psychooncology. 2014 doi: 10.1002/pon.3657. [DOI] [PubMed] [Google Scholar]
- 42.Hudson SV, et al. Cancer Survivors and the Patient-Centered Medical Home. Transl Behav Med. 2012;2(3):322–331. doi: 10.1007/s13142-012-0138-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Hudson SV, et al. Adult cancer survivors discuss follow-up in primary care: ‘not what i want, but maybe what i need’. Ann Fam Med. 2012;10(5):418–27. doi: 10.1370/afm.1379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Diefenbach MA, et al. Acceptability and preliminary feasibility of an internet/CD-ROM-based education and decision program for early-stage prostate cancer patients: randomized pilot study. J Med Internet Res. 2012;14(1):e6. doi: 10.2196/jmir.1891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Fleisher L, et al. Application of best practice approaches for designing decision support tools: the preparatory education about clinical trials (PRE-ACT) study. Patient Educ Couns. 2014;96(1):63–71. doi: 10.1016/j.pec.2014.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Neilsen J. Usability Engineering. Cambridge, MA: Elsevier; 1994. [Google Scholar]
- 47.Jaspers MW, et al. The think aloud method: a guide to user interface design. Int J Med Inform. 2004;73(11-12):781–95. doi: 10.1016/j.ijmedinf.2004.08.003. [DOI] [PubMed] [Google Scholar]
- 48.Crabtree BF, Miller WL. Doing qualitative research. 2nd. Thousand Oaks, Calif.: Sage Publications; 1999. p. xvii.p. 406. [Google Scholar]
- 49.Miles MB, Huberman AM, Saldaña J. Qualitative data analysis : a methods sourcebook. Third. Thousand Oaks, Califorinia: SAGE Publications, Inc.; 2014. p. xxiii.p. 381. [Google Scholar]
- 50.Connell R. Gender, health and theory: Conceptualizing the issue, in local and world perspective. Social Science & Medicine. 2012;74(11):1675–1683. doi: 10.1016/j.socscimed.2011.06.006. [DOI] [PubMed] [Google Scholar]
- 51.Connell RW. Masculinities. Berkeley, CA: University of California Press; 1995. [Google Scholar]
- 52.Connell RW, Messerschmidt JW. Hegemonic masculinity: rethinking the concept. Gender & Society. 2005;19(6):829–859. [Google Scholar]
- 53.Buzaglo JS, et al. Evaluation of the efficacy and usability of NCI's Facing Forward booklet in the cancer community setting. J Cancer Surviv. 2013;7(1):63–73. doi: 10.1007/s11764-012-0245-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Coleman MT, Newton KS. Supporting self-management in patients with chronic illness. Am Fam Physician. 2005;72(8):1503–10. [PubMed] [Google Scholar]
- 55.Weiss DS, Marmar CR. The Impact of Event Scale-Revised. In: Wilson JP, Keane TM, editors. Assessing psychological trauma and PTSD. Guilford; New York: 1996. [Google Scholar]
- 56.Perez M, et al. A novel intervention using interactive technology and personal narratives to reduce cancer disparities: African American breast cancer survivor stories. J Cancer Surviv. 2014;8(1):21–30. doi: 10.1007/s11764-013-0308-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.