Abstract
Ruptured aortic aneurysms uniformly require emergent attention. Historically, urgent surgical repair or medical management was the only treatment options. The development of covered stent grafts has introduced a third approach in the care of these critical patients. The clinical status of the patient and local physician expertise drive the treatment modalities in the majority of cases. The goal of therapy in these patients is to stabilize the patient as quickly as possible, establish maximum survival with minimum morbidity, and provide a long lasting result. The endovascular approach has become an acceptable treatment option in an increasing number of patients presenting with ruptured aneurysmal disease of both the descending thoracic and abdominal aorta. Major factors influencing treatment include patient clinical status, characteristics of the aorta, physician preference, institutional experience, and availability of appropriate equipment. Planning, experience, and the ability to improvise effective solutions are keys to the success of the procedure when endovascular techniques are utilized. Three separate cases, requiring intraprocedural improvisation, are presented followed by a review of the literature.
Keywords: ruptured aneurysm, emergent stent graft, EVAR, TEVAR, interventional radiology
CME Objective: On completion of this article, the reader will be able to identify the indications for placing emergent endovascular aortic stent grafts and know the current evidence for endovascular and open surgical outcomes following treatment of ruptured aortic aneurysms.
Accreditation: This activity has been planned and implemented in accordance with the Essential Areas and policies of the Accreditation Council for Continuing Medical Education through the joint providership of Tufts University School of Medicine (TUSM) and Thieme Medical Publishers, New York. TUSM is accredited by the ACCME to provide continuing medical education for physicians.
Credit: Tufts University School of Medicine designates this journal-based CME activity for a maximum of 1 AMA PRA Category 1 Credit™. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
The high mortality rate associated with ruptured aortic aneurysms instills a sense of urgency in physicians tasked to treat patients with this unfortunate condition. Elective management of both stable abdominal aortic aneurysms (AAAs) and thoracic aortic aneurysms (TAA) has incorporated open surgical techniques, endovascular procedures, and medical management. The treatment approach frequently depends on aneurysm characteristics, the underlying clinical status of the patient, and physician experience. In appropriate patients without aneurysm rupture, randomized controlled trials have demonstrated early survival benefits with elective endovascular AAA repair (EVAR) compared with the open surgical approach.1 2 Similarly, elective endovascular repair of TAA (TEVAR) is also associated with decreased morbidity and long-term aneurysm-related mortality.3 4 5 The luxury of a clinically stable patient without a ruptured aneurysm allows an organized, well-orchestrated treatment plan, frequently with second and third alternative treatment options. If questions or uncertainties arise, there is time to review the literature as well as to consult other experts in the field. Unfortunately, once an aortic aneurysm ruptures and clinical stability fades, urgent decisions are required. The choice between surgical and endovascular techniques involves careful, deliberate consideration of many factors, including surgical risks of a major invasive operation, the availability of an appropriate endovascular device to match the individual aneurysm anatomy, and physician and institutional experience in both open surgical and endovascular procedures.6 7 8 9 10 11 A ruptured aneurysm requires a more expeditious evaluation and implementation of a specific treatment plan.12 13 14
Case 1
An 88-year-old man presented to the emergency department (ED) with acute onset right flank pain. The pain had migrated from the right to the left flank, then to the mid-back in ∼24 hours. He had a past medical history of peripheral vascular disease, diabetes, hypertension, diverticulitis, hypothyroidism, and coronary artery disease. He had recently been seen by a vascular surgeon to discuss open versus endovascular options to repair an existing 6.3-cm AAA. Owing to a moderately elevated serum creatinine, no cross-sectional imaging was obtained at the time of the clinical evaluation. Findings from the physical exam at presentation to the emergency room included a heart rate of 75 beats per minute; blood pressure of 198/94 mm Hg; and 2+ palpable carotid, radial, dorsalis pedis, and posterior tibial pulses bilaterally. Limited abdominal ultrasound demonstrated a 7.8-cm AAA (enlarged from previous measurements) as well as a newly elevated troponin. Fig. 1 is a representative cross-sectional image from a noncontrast computed tomographic (CT) scan obtained at presentation that demonstrates high attenuation fluid in and around the AAA. This finding is highly concerning for acute hemorrhage and an evolving AAA rupture.
Fig. 1.
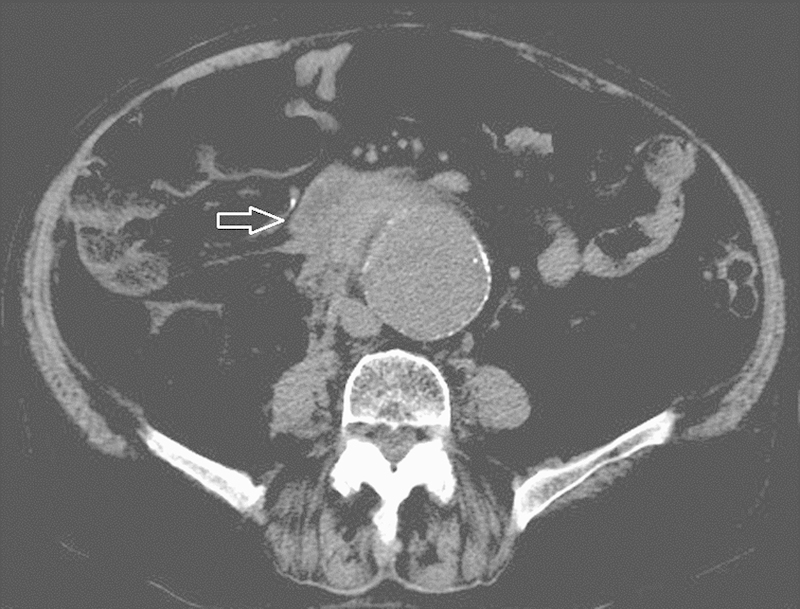
Computed tomographic image of the first patient's ruptured abdominal aortic aneurysm. Note the high attenuation fluid and stranding in the retroperitoneal fat (arrow).
Case 2
An 82-year-old man presented to the ED after a syncopal episode and loss of consciousness earlier the same day. He described diffuse abdominal pain, most pronounced with movement or palpation. He was initially hypotensive and volume resuscitation was started. Physical exam after 6 L of fluid resuscitation revealed a heart rate of 82 beats per minute; a blood pressure of 113/60 mm Hg; and “good” femoral, dorsalis pedis, and posterior tibial pulses bilaterally. Contrast-enhanced CT scan demonstrated a 7-cm AAA as well as a 5-cm left internal iliac artery aneurysm (Fig. 2). (Note that an elective common iliac artery repair is frequently performed at a diameter of 3.5 cm or larger.15) High-density fluid was present in the pelvis, around the left internal iliac arterial aneurysm, and extended up the paracolic gutters bilaterally. He has a past medical history of a 4-vessel cardiac bypass, congestive heart failure, and atrial fibrillation for which he was currently taking warfarin. He was unaware of his AAA or his left internal iliac arterial aneurysm.
Fig. 2.
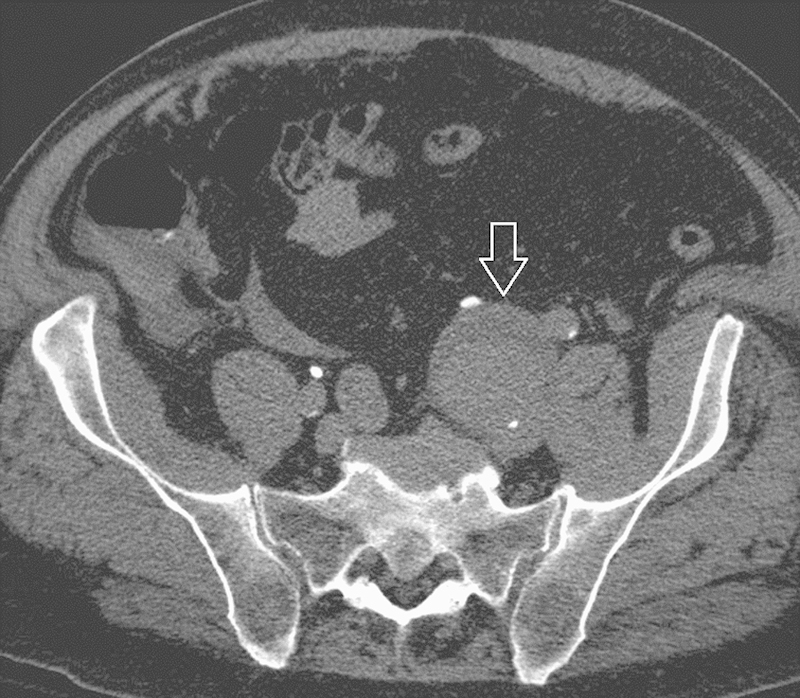
Computed tomographic image of the second patient's ruptured internal iliac artery aneurysm. There is high attenuation fluid adjacent to the dilated artery (arrow).
Case 3
A 73-year-old man presented to an outside hospital emergency department after two episodes of hemoptysis earlier in the day. In each episode, the patient reported one-half to two-thirds of a cup of blood loss. His history is significant for prior open surgical placement of a thoracic aortic graft in 1964 following a deceleration injury while flying a fighter aircraft in Vietnam. He also has a past medical history of coronary artery disease (status post–coronary artery bypass graft), chronic obstructive pulmonary disease, peripheral vascular disease, cerebrovascular disease, chronic renal insufficiency, hypertension, congestive heart failure, and hyperlipidemia. Initially, an aortoduodenal fistula was suspected; however, upper endoscopy was unremarkable. Subsequently, CT of the chest revealed a complex saccular contained rupture or pseudoaneurysm of the proximal descending aorta encroaching on the left main stem bronchus with what appeared to be free floating remnants of a disrupted surgical Dacron graft (Fig. 3). He was transferred to the authors' institution with admission to the intensive care unit (ICU). Physical exam revealed a heart rate of 65 beats per minute, blood pressure of 125/75 mm Hg, 2+ right femoral pulse, 1+ left femoral pulse, and nonpalpable dorsalis pedal pulses bilaterally.
Fig. 3.
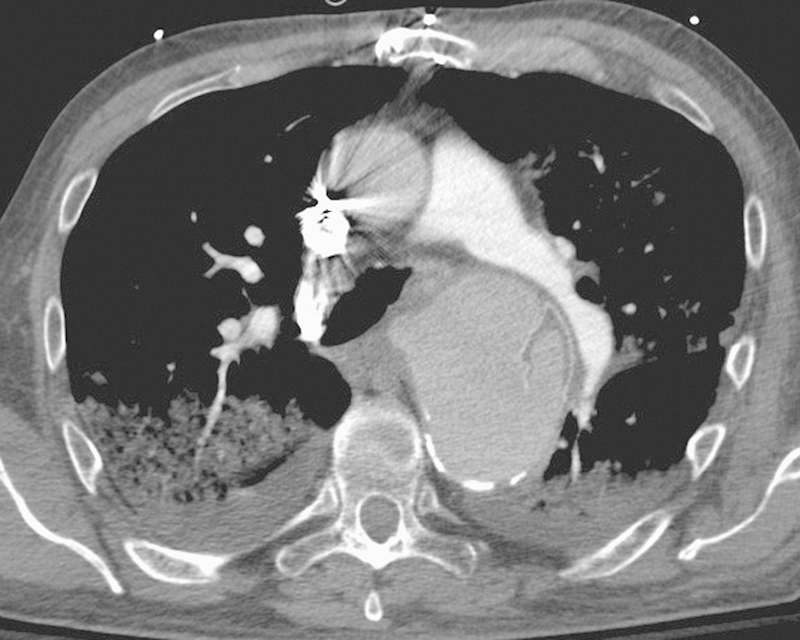
Contrast-enhanced computed tomographic image of the third patient's disrupted Dacron graft. Evidence of acute hemorrhage includes the mediastinal fat stranding, bilateral pleural infiltrates (due to communication with the bronchial tree), and high attenuation pleural fluid.
Patient Evaluation
Step 1: Identify the Rupture
Any patient entering an emergency department with pain and a history of aneurysmal disease will likely undergo cross-sectional imaging (although frequently not optimized to evaluate the arterial system). Identifying a rupture in patients with suboptimal cross-sectional imaging studies can be challenging. Ruptured vasculature frequently demonstrates an adjacent complex fluid collection with high attenuation. In the abdomen, this high attenuation fluid may represent a periaortic, retroperitoneal, or psoas hematoma. However, “stranding” of the retroperitoneal fat and loss of the para-aortic fat plane are also commonly seen in patients with aneurysm ruptures. Additional findings suggesting an acute rupture include a “crescent sign” (C-shape region of high attenuation within the mural thrombus/aortic wall) and “draping” of the posterior wall of the abdominal aorta along the vertebral bodies. The lateral aspects of the aorta fall onto the vertebral body rather than being pulled up and around the aortic circumference. The circumferential tension introduced by the aortic wall musculature introduces an angle of retroperitoneal fat between the vertebral body cortex and the curvature of the aorta.16 17 Similarly, with thoracic aneurysm, rupture concerning findings include associated intramural hematoma or “crescent sign,” “draping” of the thoracic aorta along adjacent vertebral bodies, and poor visualization of the posterior aortic wall. Thoracic aneurysm rupture is also associated with “stranding” of perianeurysmal fat, mediastinal hematoma, hemothorax (often on the left), and hemopericardium.18 19 To confirm the diagnosis of a ruptured aneurysm, a noncontrast CT scan is sufficient. However, a contrast-enhanced, arterial phase CT scan is recommended in all cases being considered for an endovascular repair. The contrast-enhanced study allows for the evaluation of the lumen diameter and distances between vessel origins. These measurements are vital in selecting the appropriate endograft. For thoracic endovascular repair, CT arteriography that includes the arch vessels through the femoral vessels is recommended for procedural planning.
Step 2: Endovascular Procedure or Open Surgery
The decision between an endovascular approach and open surgical repair should be made expeditiously owing to the high mortality associated with an aortic rupture. Several protocols have been put forth in determining the patient status and evaluation routine. Fig. 4 represents one institution's approach to the decision process with a primary deciding factor being patient clinical stability (i.e., is there time to evaluate the patient for a potential endovascular approach?). A contrary approach would focus on the patient comorbid factors (i.e., would the patient survive an open operation?). In the latter instance, the patient would go directly to the operating room (OR) unless contraindicated, in which case an endovascular approach would be considered prior to conservative/palliative care. A third approach involves placing an occlusion balloon within the vasculature to stabilize the patient and to allow time for a more complete evaluation. Unfortunately, all approaches introduce a selection bias into any comparative or cohort studies. Therefore, the institutional treatment algorithm becomes a key component in the evaluation of the published literature surrounding endovascular repair of ruptured aortic aneurysms. In many clinical settings, any combination of the aforementioned approaches may be utilized in determining if a patient even may be evaluated for placement of an endovascular device. Unless specific algorithms are designed and followed, the decision between open and endovascular approaches may boil down to interpersonal relationships or bias introduced from the last successful or unsuccessful case.
Fig. 4.
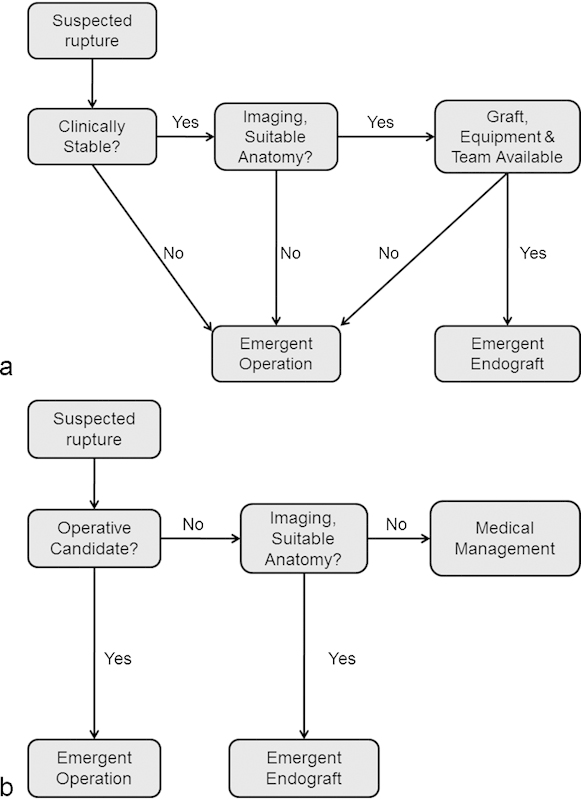
Algorithm (a) initiates the evaluation of the patient with a suspected rupture by determining if the patient is clinically stable enough to undergo evaluation for an endograft. Algorithm (b) determines if the patient is an operative candidate. These initial choices in the decision tree impart opposite biases into the survival curves for endograft and open surgical operation, defining one hospital's treatment protocol. Note the initial question in the decision tree can lead to selection bias.
Step 3: Key Endovascular Anatomy
Once the decision has been made to place an aortic endograft, the vascular anatomy must be critically and efficiently analyzed. Most studies and device manufacturers recommend an arterial phase contrast-enhanced CT arteriogram (CTA) to evaluate the key vasculature and choose an appropriate device. However, in some cases, magnetic resonance angiography (MRA) or catheter arteriography may provide enough information to select an appropriate endograft. Key components to evaluate in the vascular anatomy include the maximum aortic diameter (as well as the diameter and distance between the planned locations of the proximal and distal landing zones); aortic angulation; vessel wall calcification; diameter at the aortic bifurcation (for aortoiliac aneurysms); conformation of the aortic arch (for thoracic aneurysms); and the diameter and tortuosity of the vasculature required to deliver the anticipated device(s). The ability to rapidly perform three-dimensional reconstructions greatly assists in defining true diameters of vessels as well as determining accurate lengths required in selecting an appropriate endograft device. Previous experience frequently assists in making quick, efficacious evaluations and decisions in these instances. Once these measurements have been made, the operator must select the most compatible device from available equipment. Another key component to a successful endograft program requires the development of a primary as well as a quality “back-up” plan; having an alternative approach to the same case is extremely beneficial should unanticipated developments arise. Procedural events or clinical conditions that would necessitate abandoning the endovascular approach and conversion to an open surgical repair should be anticipated.
In general, vascular diameters do not change significantly from cross-sectional imaging measurements if appropriate three-dimensional reformats have been made. On the other hand, the length of devices required is occasionally modified due to tortuosity of the vessel and stiffness of guide wires. Rarely, one may be required to reverse the approach (main body of the endograft delivered from the right or the left) depending on access vessel diameter and tortuosity. When it comes to safe choices in device selection, one must remember that infolding from a too-large graft diameter can frequently be treated in vivo with repeat angioplasty and/or stenting. Too small of a graft diameter relative to the vessel can only be treated by additional stent grafts and modifying the intended “landing zone.” This change may result in covering renal or visceral vessels proximally and internal iliac arteries distally for abdominal repairs, or mediastinal great vessels proximally and mesenteric vessels distally for thoracic repairs. Similarly, endografts that are too short in length can always be extended with additional devices; however, there is almost no way to shorten a device percutaneously after deployment.
Procedure Details and Follow-up
Case 1
The patient's age, elevated troponin, and decreased renal function essentially excluded an emergent open surgical repair of his ruptured AAA. Risks and benefits of attempting endovascular repair versus conservative management (including renal failure and dialysis) were discussed with the patient and his family. The decision was made to proceed with the endovascular repair.
The noncontrast CT was used to identify and measure proximal and distal landing zones; the appropriate devices were available for use immediately. A contrast-enhanced CTA was not obtained due to the decreased renal function with a plan of identifying appropriate final measurements via intraoperative arteriograms. The diameter of the aorta at the level of the renal arteries as well as both common and external iliac arteries was determined from the noncontrast CT scan. Limited three-dimensional reconstructions were used to estimate appropriate lengths of devices required for the case. Lumen diameter and mural thrombus were unknown components at the time the patient was brought to the procedure room.
Bilateral open surgical to cut down exposure to both common femoral arteries was obtained with the assistance of the vascular surgery team. Access from the right common femoral artery was uneventful. On the left, the guide wire would not advance into the left external iliac artery in spite of adequate arterial flow with an 18-gauge access needle. A pelvic arteriogram was performed to evaluate the left external iliac artery as well as to assess the landing zones of both iliac limbs of the endograft. Fig. 5 demonstrates the complete left external iliac arterial occlusion, the diameter of the bilateral common iliac arteries, and the location of the iliac bifurcations. Conversion to an aorto-uni-iliac procedure followed by a surgical right-to-left common femoral bypass procedure was considered. In that situation, the left common and internal iliac arterial supply and the corresponding pelvic perfusion would be sacrificed. Therefore, the decision was made to attempt recanalization of the external iliac artery.
Fig. 5.
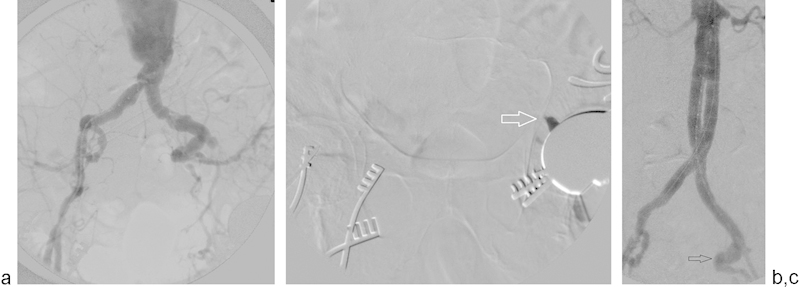
Pelvic arteriogram demonstrating a left external iliac arterial occlusion. The aortogram (a) demonstrates the patent left common and internal iliac arteries. Selective injection from a left common femoral approach (b) demonstrates the distal aspect of the external iliac artery occlusion (arrow) (as well as the left hip arthroplasty). An aorto-uni-iliac device in this patient would have likely resulted in significant buttock claudication. Extravascular and subintimal techniques were used in the occluded segment, allowing placement of a bifurcated abdominal aortic endograft device and preserving arterial flow into the left hemipelvis. Aortogram after repair (c) demonstrates continued perfusion through the left internal iliac artery (arrow) as well as contrast within the sheath in the subintimal space along the left external iliac artery.
Initial attempts at revascularization from the left common femoral artery were unsuccessful. A wire and catheter were positioned over the aortic bifurcation from the right common femoral arterial access into the left common iliac artery, and antegrade recanalization was attempted. In the blind stump of the proximal left external iliac artery, a hydrophilic wire was advanced into the subintimal space. The wire and catheter were gently advanced to the level of the recanalized left distal external iliac artery. Attempts to re-enter the lumen were made but eventually resulted in complete vessel perforation. The hydrophilic wire was identified within the open cut down site adjacent to the left common femoral artery. The options were reconsidered (abort the attempted endovascular repair completely, conversion to an aorto unifemoral device, or proceed as planned). Given that the subintimal tract was safely away from the origin of the left internal iliac artery in the external iliac arterial stump, the decision was made to expand the subintimal and extravascular tract enough to successfully allow placement of the contralateral limb of the device. After exchange for a stable working wire, a sheath was advanced through the tract and into the lumen of the left external and common iliac artery. The main body of the device was deployed uneventfully from the right common femoral arterial approach. The short limb of the main body was successfully cannulated from the complex left common femoral approach. The contralateral limb was deployed without incident, landing zones were secured with balloon angioplasty, and digital subtraction arteriography confirmed exclusion of the aneurysm with no residual leak into the left subintimal/extravascular space. Surprisingly, the “arteriotomy” on the left did not require suture-mediated closure. The patient was discharged in stable condition on postprocedure day 9; his delayed discharge was primarily due to contrast-induced nephropathy and cardiac evaluation for his non-ST segment elevation myocardial infarction.
Case 2
Compared with case 1, the second patient was clinically stable immediately after the initial resuscitation bolus, allowing for the contrast-enhanced CTA to be obtained. The CTA demonstrated the AAA as well as the very large internal iliac artery aneurysm. No active extravasation was identified; however, there was an interval increase in the amount of blood in the pelvis. He was clinically stable enough to allow adequate planning of an endograft procedure both with and without left internal iliac artery coil embolization. Given these results, plans were made to extend the AAA endograft in a manner that would exclude the internal iliac artery aneurysm. The decision to coil embolize the outflow branches of the internal iliac artery aneurysm would be based on the patient's intraoperative clinical stability.
Open surgical exposure of the common femoral arteries was obtained bilaterally with the assistance of the vascular surgery team, and stable vascular access was obtained with bilateral 7-Fr vascular sheaths. Given the patient's hemodynamic stability at this time, the decision was made to catheterize both anterior and posterior divisions of the left internal iliac artery to occlude potential aneurysm sack inflow in hopes of reducing risk of a type II endoleak. After multiple coils in both anterior and posterior divisions, no significant outflow from the aneurysm sac was identified and attention was turned to the AAA. The main device body was uneventfully deployed from a right common femoral access. The left limb was extended into the left external iliac artery, thereby excluding the left internal iliac artery aneurysm (Fig. 6). The patient was discharged on postprocedure day 4 in stable condition. Three-month follow-up imaging demonstrates interval resolution of the retroperitoneal blood and stable to slight interval decrease in both AAA and internal iliac arterial aneurysm sac sizes. Unfortunately, a small type II endoleak remained from a previously unseen pelvic branch in the left internal iliac arterial aneurysm; the endoleak and aneurysms are currently being followed for stability (Fig. 7).
Fig. 6.
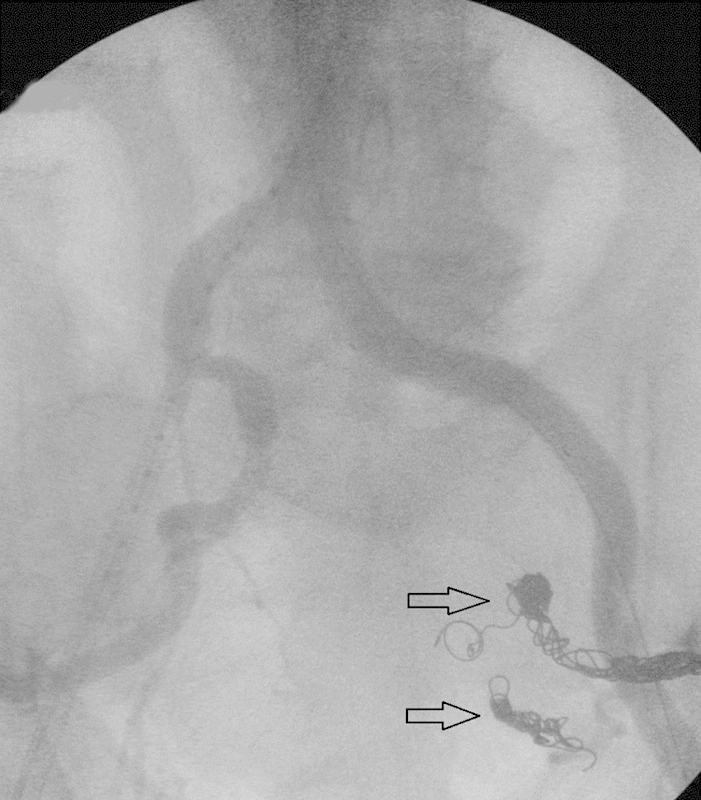
Arteriogram of the second patient's repaired aortoiliac aneurysm, demonstrating flow through the endograft, and occlusive coils in the anterior and posterior divisions of the left internal iliac artery (arrows).
Fig. 7.
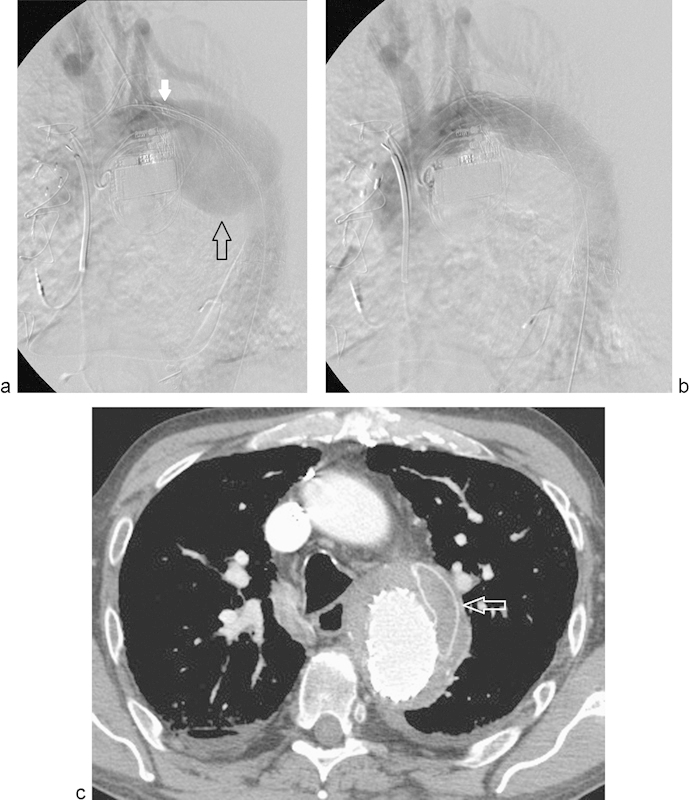
Arteriogram (a) demonstrates the large contained rupture in the descending thoracic aorta (open black arrow) as well as the initial stent graft deployment forming the seal at the proximal landing zone (solid white arrow). Arteriogram (b) demonstrates the exclusion of the aortic defect after placement of the second stent bridging the proximal landing zone with sufficient overlap into the distal stent graft. Poststenting computed tomography (c) demonstrates the excluded aortic injury as well as the initial open surgical graft material (arrow).
Case 3
The patient was hemodynamically stable upon presentation to the ICU with blood pressure controlled by β-blocker infusion. Given his complex comorbid conditions, he was considered to be a poor candidate for open surgical intervention. Technical challenges of endovascular repair included a limited proximal landing zone and the presence of the calcified Dacron graft within the pseudoaneurysm. CT imaging suggested the Dacron graft had partial attachment at both the proximal and distal anastomoses. There was the additional risk of stent graft infection as the pseudoaneurysm was in communication with the airway. Risks and benefits of attempting endovascular repair were discussed with the patient, and the decision was made to proceed.
Percutaneous access of the right common femoral artery was obtained uneventfully. Right brachial artery access was obtained for placement of the diagnostic flush catheter. Digital subtraction angiography (DSA) evaluation of the descending thoracic aorta was performed, although the images were somewhat compromised by the patient's previously placed automatic implantable cardioverter defibrillator. Once the anatomy was well delineated and in consultation with the cardiothoracic surgical team, the decision was made to place 31 × 100 mm and 34 × 100 mm thoracic stent grafts to adequately exclude the 7-cm aortic injury and surgical graft. A 1.5-cm neck was present between the origin of the left subclavian artery and the thoracic aortic injury. The initial 31 × 100 mm device was deployed, covering the distal two-thirds of the lesion with ∼3 cm of overlap in the normal distal thoracic aorta. Exchange was then made for the 34 × 100 mm device that was deployed at the left subclavian artery origin. Postdeployment DSA evaluation demonstrated normal flow through the great vessels, stent graft, and the distal descending thoracic aorta. No endoleak was identified (Fig. 7). The patient did well but had a somewhat prolonged ICU stay, largely due to his pre-stent overall medical condition. He was discharged in stable condition on postprocedure day 10.
Discussion
The first two cases demonstrate situations that require a flexible approach in treating ruptured aortoiliac aneurysms. The first case may have just been converted to a unilateral repair with exclusion of the left pelvic vasculature followed by a right to left femoral to femoral bypass graft. The risk of the subintimal/extravascular delivery of the contralateral limb seemed low compared with the high probability of left buttock claudication with the alternative procedure. Therefore, the attempted recanalization was performed. Similarly, the second case may not have absolutely required coil embolization of the internal iliac branches if the patient became hemodynamically unstable during the case. In the third case, the patient presented with a unique problem of rupture of an existing Dacron graft. Late fiber deterioration of Dacron grafts has been reported and may have led to the situation encountered in this case.20 In placing the endograft, the origin of the left subclavian artery could have been covered if an adequate seal had not been obtained, potentially requiring a carotid-subclavian arterial bypass. Important to note in all cases is the cooperation among vascular surgery, cardiothoracic surgery, and interventional radiology teams; working together in the best interest of the patient should always outweigh the political differences outside the procedure room.
Literature Review
Endovascular treatment of ruptured AAA was first reported in 1994.21 Since that time, several studies have been published evaluating the safety and efficacy of an endovascular approach in the treatment of ruptured AAA. While most reports are observational studies or meta-analyses, a few randomized controlled trials have been conducted.
The IMPROVE trial is the largest randomized controlled trial to evaluate endovascular versus open repair of ruptured aortic aneurysms.22 This trial demonstrated similar 30-day mortality rates in patients with clinically diagnosed ruptured aortic aneurysms (35.4% for endovascular strategy and 37.4% for open repair), as well as those patients with imaging-confirmed rupture (36.4 and 40.6%, respectively). Interestingly, subgroup analysis indicated that an endovascular strategy might be more beneficial in women compared with men. The 30-day mortality for women was 37% for endovascular strategy and 57% for open repair, compared with rates of 35 and 32% for men. This study did show that patients in the endovascular strategy group were discharged earlier and more likely discharged to home (94% in the endovascular strategy group and 77% in the open repair group) rather than to an extended care facility. The endovascular strategy group and open repair group were shown to have similar overall costs at 30 days.
A smaller randomized controlled trial from the Netherlands also demonstrated similar 30-day outcomes for endovascular repair and open repair of ruptured AAAs.23 In this study, 116 patients with confirmed ruptured AAA and anatomy suitable for EVAR were randomized to either endovascular or open repair. The primary outcome of combined death and severe complications at 30 days was 42% for endovascular repair and 47% for open repair; mortality rates at 30 days were 21% for endovascular repair and 25% for open repair. While overall severe complications were not significantly different between the two groups, renal insufficiency requiring temporary or permanent dialysis occurred less frequently with the endovascular repair group (11 vs. 31%, p = 0.01). Survival analysis with median long-term follow-up of 1,533 days did not show any difference in mortality rates between the groups.
Hinchliffe et al describe the first randomized controlled trial of patients with ruptured AAAs who were also considered “fit for open repair.”24 This collection of patients was randomized into endovascular or open surgical groups. Of the 103 patients evaluated, 32 were recruited and randomized into the study. They reported no significant difference in 30-day mortality rates (53% for both groups) or hospital stay (10 vs. 12 days, respectively). The study did report the median time between diagnosis and the operation was 75 minutes and 100 minutes for the endovascular and open surgical groups, respectively, demonstrating that obtaining a preoperative CT does not delay treatment.
A Cochrane review of the three existing randomized controlled trials supported the finding of similar 30-day mortality rates for endovascular and open repair of patients with the clinical or radiological diagnosis of ruptured AAA.25 The Cochrane authors concluded, however, that there is a paucity of data and that more high-quality, randomized controlled trials are needed. Conclusions regarding differences in complication rates or long-term outcomes could not be made due to the lack of data.
In contrast to the few randomized controlled trials, a recent large meta-analysis evaluating 41 studies demonstrated a significantly decreased incidence of in-hospital mortality for patients undergoing endovascular repair of ruptured AAA compared with open repair.26 The meta-analysis population was composed of 59,941 patients, of whom 8,201 patients underwent EVAR while 51,740 patients underwent open repair; in-hospital mortality rates were 30% for EVAR and 42% for open repair. Perioperative respiratory complications were reported in 19 of the studies, which found a significantly decreased incidence in the EVAR population compared with the open repair patients (6 vs. 11%, respectively). The risk of acute renal failure was also significantly less in EVAR patients compared with open repair patients (16 vs. 26%, respectively) in the eight studies reporting this outcome. Rates of secondary interventions were similar in the two groups.
A large retrospective cohort study examined national level data over a 10-year period comparing endovascular and open repair of ruptured AAA.27 This study included 42,126 patients, of whom 8,140 underwent EVAR and 33,986 underwent open repair. Demographics of the two groups did differ in that the endovascular cohort was older than the open repair cohort and had more patients with multiple comorbidities. Despite these unfavorable characteristics, EVAR was associated with significantly lower in-hospital mortality rates (25.9% compared with 39.1% for open repair) and shorter length of hospital stay (10.4 vs. 13.7 days for open repair). Additionally, more patients in the EVAR group were discharged directly to home (36.8% compared with 21.5% for open repair) and fewer patients required institutional rehabilitation (26.3% compared with 29.1% for open repair). Trend analysis demonstrated a significant increase in the use of EVAR for ruptured AAA (6.6% in 2001 to 42.1% in 2010). Additionally, there was a decrease in hospital mortality for both EVAR and open repair over the study period (41.3–25.8% for EVAR and 40.5–30.8% for open repair).
Another national level study examined perioperative and long-term mortality as well as short- and long-term complications in patients undergoing endovascular versus open repair of ruptured AAA in the United States Medicare population.28 This study included 10,998 patients, of whom 1,126 underwent EVAR and 9,872 underwent open repair. Propensity score matching yielded 1,099 patient pairs. Using the matched cohorts, perioperative mortality rates were significantly lower for EVAR patients compared with open repair (33.8 vs. 47.7%), respectively. Many postoperative complications were also significantly less common in the EVAR group, including pneumonia, acute renal failure, respiratory failure requiring tracheostomy, and gastrointestinal complications of colon resection and mesenteric ischemia. Patients in the EVAR group also had improved long-term survival with a survival benefit persisting for >4 years. Patients treated with EVAR did have a significantly higher reintervention rate for AAA than those treated with open repair. This study also demonstrated a significantly increased use of EVAR to treat ruptured AAA over time (6% of repairs in 2001 vs. 31% of repairs in 2008). Perioperative mortality also decreased in both groups over time.
Gupta et al recently published a report analyzing differential outcomes for stable and unstable patients presenting with ruptured AAA.29 Patients were identified from the National Surgical Quality Improvement Program database from 2005 to 2010, with 54.8% (793/1,447 patients) classified as stable and 45.2% (654/1,447 patients) classified as unstable. They found that both cohorts had significantly improved 30-day mortality rates for patients undergoing EVAR compared with open surgical repair (16.4 vs. 26.3% for stable patients and 35.6 vs. 52.8% for unstable patients, respectively). EVAR was also associated with lower overall morbidity rates than open repair in both stable and unstable cohorts. Not surprisingly, the unstable patient cohort had less favorable outcomes compared with the stable cohort.
Another recent study also supports a survival benefit of EVAR over open surgical repair of ruptured AAA.30 This study examined ruptured infrarenal AAAs in 283 patients, of whom 120 underwent EVAR and 163 underwent open repair. The EVAR group had significantly lower 30-day mortality rates (24.2% compared with 44.2% for open repair), and this survival benefit persisted at 5 years of follow-up. Interestingly, when analyzed by gender, EVAR was not associated with significantly improved 30-day mortality for women. In the subset of patients who survived at least 30 days, long-term survival was not significantly different between the two treatment groups. EVAR patients had less mean estimated blood loss; however, they required a significantly increased rate of reintervention.
Although many investigators have examined the safety and efficacy of an endovascular approach for emergent repair of the descending thoracic aorta, no randomized controlled trials have yet been published. A multicenter retrospective analysis was performed to compare endovascular repair versus open surgery of ruptured descending TAA.31 This study included 161 patients over a 15-year period, of whom 92 were treated with TEVAR and 69 were treated with open repair. The primary composite outcome of death, stroke, or permanent paraplegia was significantly lower in the TEVAR group compared with open surgical repair (21.7 vs. 36.2%, respectively); however, 30-day mortality rates of 17.4% for TEVAR and 24.6% for open repair did not reach significance. TEVAR was associated with lower rates of pulmonary complications and acute renal failure as well as shorter median hospital stays.
A study evaluating national level data evaluated in-hospital outcomes of TEVAR compared with open surgical repair of ruptured TAA.32 A total of 923 patients were included: 364 underwent TEVAR and 559 underwent open repair. The TEVAR group was noted to be significantly older and a higher proportion was women, white, receiving Medicare, and had higher Deyo comorbidity scores. Unadjusted in-hospital mortality rates, overall complication rates, and failure to rescue (death after a complication) rates were similar in the two groups. TEVAR was associated with a significantly shorter hospital stay and higher likelihood of discharge directly to home. Unfortunately, factors such as the severity of rupture and hemodynamic stability of the patients were not included in the database used for analysis, preventing further risk-adjusted analysis.
A single-center study analyzed outcomes of open versus endovascular repair of ruptured nontraumatic descending thoracic aortas in 69 patients.33 The pathology included rupture from fusiform aneurysm, saccular aneurysm/ulcer, and dissection. The TEVAR group consisted of 35 patients deemed high risk for open repair or with extremely favorable anatomy for endovascular repair; the remaining 34 patients underwent open surgical repair. The TEVAR group was older, had smaller aortic dimensions, and had an increased incidence of chronic obstructive pulmonary disease. The open repair group more frequently required intervention on the aortic arch. There was a trend toward decreased early mortality rates in the TEVAR group, but this did not reach statistical significance (11.4 vs. 26.5%). Two independent predictors of a composite end-point (early death, stroke, permanent paraplegia, need for dialysis, and/or tracheostomy) were hemodynamic instability at presentation and open surgical repair. TEVAR was associated with a significant shorter hospital stay; however, it also had a higher need for aortic reintervention. Long-term survival analysis was not significantly different between the two groups.
One recent study compared endovascular treatment to open repair in100 patients with acute thoracic aortic catastrophes.34 The patients had a heterogeneous group of pathologies with 41% presenting with ruptured degenerative aneurysms. Of the 100 patients, 76 underwent TEVAR while 24 underwent open surgical repair. Overall 30-day mortality rates were significantly reduced in the TEVAR group compared with open repair (8 vs. 29%, respectively) and the survival benefit persisted at 5-year follow-up. Respiratory complications were also significantly lower in the TEVAR group (16 vs. 48% for open repair), while the incidence of postoperative stroke, spinal ischemia, myocardial infarction, and acute renal failure were similar in the two groups. Reintervention rates were similar in both treatment groups.
Conclusion
All three cases presented here demonstrate the need to be “flexible” and willing to improvise when it comes to emergent aortic endograft procedures. A diverse experience with multiple devices and procedures provides enough confidence to improvise in clinically unstable situations. In reviewing the literature, there are significant data supporting the safety and efficacy of aortic endografts in the acute/ruptured aneurysm setting. For ruptured AAAs, clinical trials have demonstrated equivalent short-term mortality rates for EVAR and open surgical repair, although numerous observational studies suggest improved outcomes for patients undergoing EVAR. In the thoracic aorta, clinical trials for treatment of ruptured TAAs are lacking, but the existing evidence supports similar improved outcomes for patients undergoing TEVAR. In endovascular repair, emergent aortic stent grafts may become standard of care in patients with appropriate vascular anatomy.
References
- 1.Greenhalgh R M Brown L C Kwong G P Powell J T Thompson S G; EVAR trial participants. Comparison of endovascular aneurysm repair with open repair in patients with abdominal aortic aneurysm (EVAR trial 1), 30-day operative mortality results: randomised controlled trial Lancet 20043649437843–848. [DOI] [PubMed] [Google Scholar]
- 2.Prinssen M, Verhoeven E L, Buth J. et al. A randomized trial comparing conventional and endovascular repair of abdominal aortic aneurysms. N Engl J Med. 2004;351(16):1607–1618. doi: 10.1056/NEJMoa042002. [DOI] [PubMed] [Google Scholar]
- 3.Fairman R M, Criado F, Farber M. et al. Pivotal results of the Medtronic Vascular Talent Thoracic Stent Graft System: the VALOR trial. J Vasc Surg. 2008;48(3):546–554. doi: 10.1016/j.jvs.2008.03.061. [DOI] [PubMed] [Google Scholar]
- 4.Makaroun M S Dillavou E D Wheatley G H Cambria R P; Gore TAG Investigators. Five-year results of endovascular treatment with the Gore TAG device compared with open repair of thoracic aortic aneurysms J Vasc Surg 2008475912–918. [DOI] [PubMed] [Google Scholar]
- 5.Matsumura J S Cambria R P Dake M D Moore R D Svensson L G Snyder S; TX2 Clinical Trial Investigators. International controlled clinical trial of thoracic endovascular aneurysm repair with the Zenith TX2 endovascular graft: 1-year results J Vasc Surg 2008472247–257., discussion 257 [DOI] [PubMed] [Google Scholar]
- 6.Ballotta E, Da Giau G, Bridda A, Gruppo M, Pauletto A, Martella B. Open abdominal aortic aneurysm repair in octogenarians before and after the adoption of endovascular grafting procedures. J Vasc Surg. 2008;47(1):23–30. doi: 10.1016/j.jvs.2007.08.054. [DOI] [PubMed] [Google Scholar]
- 7.Bown M J, Sutton A J, Bell P RF, Sayers R D. A meta-analysis of 50 years of ruptured abdominal aortic aneurysm repair. Br J Surg. 2002;89(6):714–730. doi: 10.1046/j.1365-2168.2002.02122.x. [DOI] [PubMed] [Google Scholar]
- 8.Chahwan S Comerota A J Pigott J P Scheuermann B W Burrow J Wojnarowski D Elective treatment of abdominal aortic aneurysm with endovascular or open repair: the first decade J Vasc Surg 2007452258–262., discussion 262 [DOI] [PubMed] [Google Scholar]
- 9.Lederle F A, Johnson G R, Wilson S E. et al. Rupture rate of large abdominal aortic aneurysms in patients refusing or unfit for elective repair. JAMA. 2002;287(22):2968–2972. doi: 10.1001/jama.287.22.2968. [DOI] [PubMed] [Google Scholar]
- 10.Dua A, Furlough C L, Ray H, Sharma S, Upchurch G R, Desai S S. The effect of hospital factors on mortality rates after abdominal aortic aneurysm repair. J Vasc Surg. 2014;60(6):1446–1451. doi: 10.1016/j.jvs.2014.08.111. [DOI] [PubMed] [Google Scholar]
- 11.Findeiss L K, Cody M E. Endovascular repair of thoracic aortic aneurysms. Semin Intervent Radiol. 2011;28(1):107–117. doi: 10.1055/s-0031-1273945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Mehta M Taggert J Darling R C III et al. Establishing a protocol for endovascular treatment of ruptured abdominal aortic aneurysms: outcomes of a prospective analysis J Vasc Surg 20064411–8., discussion 8 [DOI] [PubMed] [Google Scholar]
- 13.Roche-Nagle G, Lindsay T F. Endovascular ruptured abdominal aortic aneurysm repair—setting up your hospital for an endovascular approach. Surgeon. 2010;8(1):39–43. doi: 10.1016/j.surge.2009.10.020. [DOI] [PubMed] [Google Scholar]
- 14.Coselli J S, Gopaldas R R. Ruptured thoracic aneurysms: to stent or not to stent? Circulation. 2010;121(25):2705–2707. doi: 10.1161/CIRCULATIONAHA.110.961631. [DOI] [PubMed] [Google Scholar]
- 15.Huang Y Gloviczki P Duncan A A et al. Common iliac artery aneurysm: expansion rate and results of open surgical and endovascular repair J Vasc Surg 20084761203–1210., discussion 1210–1211 [DOI] [PubMed] [Google Scholar]
- 16.Rakita D, Newatia A, Hines J J, Siegel D N, Friedman B. Spectrum of CT findings in rupture and impending rupture of abdominal aortic aneurysms. Radiographics. 2007;27(2):497–507. doi: 10.1148/rg.272065026. [DOI] [PubMed] [Google Scholar]
- 17.Roy J, Labruto F, Beckman M O, Danielson J, Johansson G, Swedenborg J. Bleeding into the intraluminal thrombus in abdominal aortic aneurysms is associated with rupture. J Vasc Surg. 2008;48(5):1108–1113. doi: 10.1016/j.jvs.2008.06.063. [DOI] [PubMed] [Google Scholar]
- 18.Hallinan J T, Anil G. Multi-detector computed tomography in the diagnosis and management of acute aortic syndromes. World J Radiol. 2014;6(6):355–365. doi: 10.4329/wjr.v6.i6.355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Chiu K W, Lakshminarayan R, Ettles D F. Acute aortic syndrome: CT findings. Clin Radiol. 2013;68(7):741–748. doi: 10.1016/j.crad.2013.03.001. [DOI] [PubMed] [Google Scholar]
- 20.Berger K, Sauvage L R. Late fiber deterioration in Dacron arterial grafts. Ann Surg. 1981;193(4):477–491. [PMC free article] [PubMed] [Google Scholar]
- 21.Yusuf S W, Whitaker S C, Chuter T A, Wenham P W, Hopkinson B R. Emergency endovascular repair of leaking aortic aneurysm. Lancet. 1994;344(8937):1645. doi: 10.1016/s0140-6736(94)90443-x. [DOI] [PubMed] [Google Scholar]
- 22.Powell J T, Sweeting M J, Thompson M M. et al. Endovascular or open repair strategy for ruptured abdominal aortic aneurysm: 30 day outcomes from IMPROVE randomised trial. BMJ. 2014;348:f7661. doi: 10.1136/bmj.f7661. [DOI] [PubMed] [Google Scholar]
- 23.Reimerink J J, Hoornweg L L, Vahl A C. et al. Endovascular repair versus open repair of ruptured abdominal aortic aneurysms: a multicenter randomized controlled trial. Ann Surg. 2013;258(2):248–256. doi: 10.1097/SLA.0b013e31828d4b76. [DOI] [PubMed] [Google Scholar]
- 24.Hinchliffe R J Bruijstens L MacSweeney S TR Braithwaite B D A randomised trial of endovascular and open surgery for ruptured abdominal aortic aneurysm - results of a pilot study and lessons learned for future studies Eur J Vasc Endovasc Surg 2006325506–513., discussion 514–515 [DOI] [PubMed] [Google Scholar]
- 25.Badger S, Bedenis R, Blair P H, Ellis P, Kee F, Harkin D W. Endovascular treatment for ruptured abdominal aortic aneurysm. Cochrane Database Syst Rev. 2014;7:CD005261. doi: 10.1002/14651858.CD005261.pub3. [DOI] [PubMed] [Google Scholar]
- 26.Antoniou G A, Georgiadis G S, Antoniou S A. et al. Endovascular repair for ruptured abdominal aortic aneurysm confers an early survival benefit over open repair. J Vasc Surg. 2013;58(4):1091–1105. doi: 10.1016/j.jvs.2013.07.109. [DOI] [PubMed] [Google Scholar]
- 27.Mohan P P, Hamblin M H. Comparison of endovascular and open repair of ruptured abdominal aortic aneurysm in the United States in the past decade. Cardiovasc Intervent Radiol. 2014;37(2):337–342. doi: 10.1007/s00270-013-0665-4. [DOI] [PubMed] [Google Scholar]
- 28.Edwards S T, Schermerhorn M L, O'Malley A J. et al. Comparative effectiveness of endovascular versus open repair of ruptured abdominal aortic aneurysm in the Medicare population. J Vasc Surg. 2014;59(3):575–582. doi: 10.1016/j.jvs.2013.08.093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Gupta P K, Ramanan B, Engelbert T L, Tefera G, Hoch J R, Kent K C. A comparison of open surgery versus endovascular repair of unstable ruptured abdominal aortic aneurysms. J Vasc Surg. 2014;60(6):1439–1445. doi: 10.1016/j.jvs.2014.06.122. [DOI] [PubMed] [Google Scholar]
- 30.Mehta M, Byrne J, Darling R C III. et al. Endovascular repair of ruptured infrarenal abdominal aortic aneurysm is associated with lower 30-day mortality and better 5-year survival rates than open surgical repair. J Vasc Surg. 2013;57(2):368–375. doi: 10.1016/j.jvs.2012.09.003. [DOI] [PubMed] [Google Scholar]
- 31.Jonker F H, Verhagen H J, Lin P H. et al. Open surgery versus endovascular repair of ruptured thoracic aortic aneurysms. J Vasc Surg. 2011;53(5):1210–1216. doi: 10.1016/j.jvs.2010.10.135. [DOI] [PubMed] [Google Scholar]
- 32.Gopaldas R R, Dao T K, LeMaire S A, Huh J, Coselli J S. Endovascular versus open repair of ruptured descending thoracic aortic aneurysms: a nationwide risk-adjusted study of 923 patients. J Thorac Cardiovasc Surg. 2011;142(5):1010–1018. doi: 10.1016/j.jtcvs.2011.08.014. [DOI] [PubMed] [Google Scholar]
- 33.Patel H J, Williams D M, Upchurch G R Jr, Dasika N L, Deeb G M. A comparative analysis of open and endovascular repair for the ruptured descending thoracic aorta. J Vasc Surg. 2009;50(6):1265–1270. doi: 10.1016/j.jvs.2009.07.091. [DOI] [PubMed] [Google Scholar]
- 34.Naughton P A, Park M S, Morasch M D. et al. Emergent repair of acute thoracic aortic catastrophes: a comparative analysis. Arch Surg. 2012;147(3):243–249. doi: 10.1001/archsurg.2011.1476. [DOI] [PMC free article] [PubMed] [Google Scholar]


