Abstract
We do not know the impact an ankle sprain has on physical activity levels across the lifespan. With the negative consequences of physical inactivity well established, understanding the effect of an ankle sprain on this outcome is critical. The objective of this study was to measure physical activity across the lifespan after a single ankle sprain in an animal model. Thirty male mice (CBA/J) were randomly placed into one of three groups: the transected calcaneofibular ligament (CFL) group, the transected anterior talofibular ligament (ATFL)/CFL group, and a SHAM group. Three days after surgery, all of the mice were individually housed in a cage containing a solid surface running wheel. Physical activity levels were recorded and averaged every week across the mouse’s lifespan. The SHAM mice ran significantly more distance each day compared to the remaining two running groups (post hoc p = 0.011). Daily duration was different between the three running groups (p = 0.048). The SHAM mice ran significantly more minutes each day compared to the remaining two running groups (post hoc p=0.046) while the ATFL/CFL mice ran significantly less minutes each day (post hoc p = 0.028) compared to both the SHAM and CFL only group. The SHAM mice ran at a faster daily speed versus the remaining two groups of mice (post hoc p = 0.019) and the ATFL/CFL mice ran significantly slower each day compared to the SHAM and CFL group (post hoc p = 0.005). The results of this study indicate that a single ankle sprain significantly decreases physical activity across the lifespan in mice. This decrease in physical activity can potentially lead to the development of numerous chronic diseases. An ankle sprain thus has the potential to lead to significant long term health risks if not treated appropriately.
Key points.
A single ankle significantly decreased physical activity levels in mice across the lifespan.
Decreased physical activity could significantly negatively impact overall health if not modified.
Initial treatment and rehabilitation of ankle sprains needs to be studied to determine ways to keep physical activity levels up after injury.
Key words: Ankle injury, physical activity, exercise, mice
Introduction
Although ankle sprains are the most common orthopedic musculoskeletal injury (Hootman et al., 2007) we do not currently understand how an ankle sprain can impact physical function as a person ages. This is despite the high percentage of patients that suffer from repetitive ankle sprains (up to 70%) and go on to develop chronic ankle instability (CAI) (up to 74%)(McKay, 2001). Long term patients that develop CAI often go on to develop post-traumatic ankle osteoarthritis (OA) (Hinterman et al., 2002; Hubbard et al., 2009). The high percentage of patients that develop these sequelae may be secondary to the approximately 50% of patients that suffer an ankle sprain and do not seek any medical treatment or evaluation (McKay, 2001). Without proper treatment and management of the acute ankle sprain proper healing and restoration of function will not occur, and an unstable ankle may result. This instability may impact physical activity as a person ages.
Physical inactivity is currently classified as one of the three highest risk behaviors in the development of cardiovascular disease, cancer, and other chronic diseases such as diabetes and obesity, and is the second highest alterable cause of cardiovascular arterial disease (Centers for Disease Control, 2012). With all the negative effects of physical inactivity known, there is surprisingly little research on the impact musculoskeletal injury has on physical activity levels. Acutely after a lateral ankle sprain research has demonstrated significant decreases in voluntary physical activity in a mouse model (Hubbard-Turner et al., 2012). The mice with both the severe and minor ankle sprain ran significantly less for the first week after injury compared to the SHAM group. From weeks two to four the severe sprain group ran significantly less than the minor sprain and SHAM group (Hubbard-Turner et al., 2012). This research demonstrates physical activity is impacted by an acute ankle sprain in the short term but there is a dearth of long term data available.
The study by Hubbard-Turner et al. (2012) was one of the first to quantify physical activity levels for four weeks after an ankle sprain. Although done in an animal model, the mouse model is an ideal model to prospectively measure lifelong physical activity levels. They offer a more cost effective means to look at long term changes after injury. Previous research examining the consequences of an ankle sprain have examined short term (1-4 weeks) changes, but have not focused on activity across the lifespan. If the short term decreases in physical activity continue across the lifespan, there is a risk for development of chronic disease secondary to physical inactivity. Therefore the purpose of this study was to examine the effects of surgically transecting the lateral ligaments of a mouse hindlimb and determine the effect of the injury on voluntary physical activity levels across the lifespan.
Methods
Animals
Thirty male mice (CBA/J), 5-6 weeks old, were purchased from Jackson Laboratory (JAX; Bar Harbor, ME). All mice were housed in the university vivarium (an AAALAC approved facility) with 12-h light/dark cycles with room temperatures and relative humidity standardized to 18-22°C and 20-40%, respectively. All mice were provided with water and standard chow (Harland Teklad 8604 Rodent Diet; Madison, WI) ad libitum. Each mouse was monitored every 24 hours for health.All study procedures were approved by the Institutional Animal Care and Use Committee. These facilities and animal care meet the standards required for appropriate treatment of animal subjects as outlined by the USDA, the Animal Welfare Act and the American College of Sports Medicine.
Surgery procedures
Each mouse was anesthetized with 4% isoflurane gas and supplemental oxygen. The right ankle of all mice were then shaved and cleaned with alcohol, followed by a Chlorhexidine scrub. After the site was prepped, the mouse remained under anesthesia and was moved to a sterile surgical field under a microscope and a warming lamp. For the calcaneal fibular ligament (CFL) only group, a small incision was made under the microscope using sterile equipment. The skin was retracted, the CFL was transected and the skin was closed using 2 drops of formulated cyanoacrylate surgical adhesive. For the anterior talofibular (ATFL)/CFL group, after the skin was retracted both the ATFL and CFL were transected and the skin was closed using 2 drops of formulated cyanoacrylate surgical adhesive. For the SHAM group, a small incision was made in the same place as the CFL only and ATFL/CFL groups; however, no ligaments were damaged and the skin was closed using 2 drops of formulated cyanoacrylate surgical adhesive. After the surgery was complete the mouse was removed from anesthesia and taken to a recovery area. Each mouse received a subcutaneous injection of 5.0mg/kg Carprofen (Rimadyl) diluted with saline, and were allowed to recover under a warming lamp until freely mobile. Mice were monitored every 24 hours after surgery and were given 12.5mg Carprofen (Rimadyl) tablets ad libitum for pain management throughout the first three days following surgery.
Physical activity measurement
Three days after surgery, all mice were individually housed in a cage containing a solid surface running wheel (127 mm, Ware Manufacturing, Phoenix, AZ), magnetic sensor, and digital odometer (Sigma Sport BC600, Olney, IL) that recorded the number of running wheel revolutions (Lightfoot et al., 2004; Turner et al., 2005). Daily running wheel measurements of duration (min) and distance (km) were recorded beginning four days after surgery. Recordings of physical activity were taken daily (6 days per week) throughout the lifespan of the mouse. Average daily speed (m/min) was calculated for each week.
Statistical analysis
Two-way ANOVAs (group x time) with Repeated Measures were performed to compare changes in physical activity (duration, distance, speed). Post hoc comparisons of between time means were performed using Tukey’s HSD tests. An alpha-level of p < 0.05 was used to determine significant effects for each analysis. All statistical analyses were performed using JMP Statistical Analysis software (SAS Institute, Cary, NC).
Results
The average lifespan for each group of mice was: SHAM group: 87.9 ± 16.1 weeks, CFL only group: 88.2 ± 9.4 weeks, and ATFL/CFL group: 92.4 ± 1.9 weeks. There were no significant differences (p = 0.07) in body weight or food consumption between the groups across the lifespan. There was no significant group by time interaction (p = 0.21, F = 0.604) for distance, duration or speed. There were main effects for group and time. For all three groups (ATFL/CFL, SHAM, CFL only) duration (p = 0.001), distance (p = 0.0001) and speed (p = 0.0001) significantly decreased across the lifespan.
Daily distance run was different between the groups (p = 0.013, F = 5.08; Figure 1A, Table 1). The SHAM mice ran significantly more distance each day compared to the remaining two running groups (post hoc p = 0.011, F = 7.44) while the ATFL/CFL mice ran significantly less distance each day than the remaining two running mice groups (post hoc p = 0.009, F = 7.80). Daily distance run decreased significantly throughout the lifespan (p < 0.0001, F = 16.02).
Figure 1.
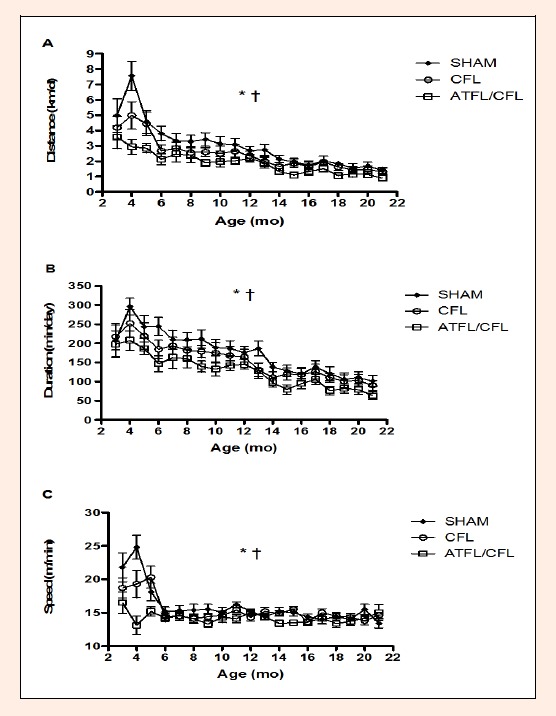
A. Mean (± SD) daily distance, B. daily duration, and C. average running speed for the three running wheel groups across the lifespan. * denotes significant difference between groups and † denotes significant difference with age (p<0.05).
Table 1.
Average (± SD) distance (km) of physical activity and effective sizes for each group across the lifespan.
| 3-5 mo | 6-8 mo | 9-11 mo | 12-14 mo | 15-17 mo | 18-21 mo | |
|---|---|---|---|---|---|---|
| Sham | 5.22 (2.43) | 3.41 (1.4) | 2.78 (.91) | 2.20 (.79) | 1.68 (.77) | 1.52 (.78) |
| CFL | 4.22 (1.93) | 2.69 (1.13) | 2.48 (.98) | 2.20 (.77) | 1.78 (.85) | 1.45 (.77) |
| Mean difference | 1.22 km | .79km | .3 km | 0 km | -.1km | .07km |
| Effect size | .413 | .512 | .305 | .006 | .113 | .090 |
| ATF/CFL | 3.02 (1.62 | 2.58 (1.23) | 2.01 (.86) | 1.78 (.76) | 1.41 (.66) | 1.15 (.57) |
| Mean difference | 2.2km | .83km | 1.6km | .42 km | .27 km | .37 km |
| Effect size | .906 | .597 | .846 | .528 | .354 | .475 |
Daily duration was different between the three running groups (p = 0.048, F = 3.42; Figure 1B, Table 2). The SHAM mice ran significantly more minutes each day compared to the remaining two running groups (post hoc p = 0.046, F = 4.36) while the ATFL/CFL mice ran significantly less minutes each day (post hoc p = 0.028, F = 5.38). Daily duration decreased significantly throughout the lifespan (p < 0.0001, F = 19.88).
Table 2.
Average (± SD) duration (min/day) of physical activity and effective sizes for each group across the lifespan.
| 3-5 mo | 6-8 mo | 9-11 mo | 12-14 mo | 15-17 mo | 18-21 mo | |
|---|---|---|---|---|---|---|
| Sham | 222.0 (71.0) | 212.4 (65.1) | 177.1 (47.5) | 149.8 (45.8) | 120.0 (44.1) | 103.6 (46.0) |
| CFL | 216.2 (89.3) | 181.3 (70.4) | 165.8 (53.0) | 145.5 (44.5) | 120.1 (50.4) | 98.8 (48.1) |
| Mean difference | 5.8 min | 31.1 min | 11.3 min | 3.5 min | 0 | 4.8 min |
| Effect size | .065 | 0.442 | .213 | .093 | .002 | .099 |
| ATF/CFL | 176.6 (71.8) | 170.1 (63.9) | 141.2 (49.4) | 121.3 (40.4) | 97.8 (39.3) | 79.3 (33.4) |
| Mean difference | 45.4 min | 42.3 min | 35.9 min | 28.5 min | 22.2 | 24.3 min |
| Effect size | .633 | .650 | .726 | .623 | .504 | .529 |
Average running speed was different between the groups (p = 0.012, F = 5.26; Figure 1C, Table 3). The SHAM mice ran at a faster daily speed versus the remaining two groups of mice (post hoc p = 0.019, F = 6.19) and the ATFL/CFL mice ran significantly slower each day compared to the CFL and SHAM mice (post hoc p = 0.005, F = 9.28). Average daily running speed slowed down with an increase in age (p < 0.0001, F = 14.82).
Table 3.
Average (±SD) speed (m/min) of physical activity and effective sizes for each group across the lifespan.
| 3-5 mo | 6-8 mo | 9-11 mo | 12-14 mo | 15-17 mo | 18-21 mo | |
|---|---|---|---|---|---|---|
| Sham | 21.9 (5.4) | 15.5 (2.2) | 15.4 (1.8) | 14.4 (1.5) | 13.6 (1.6) | 14.3 (1.5) |
| CFL | 19.0 (4.2) | 14.7 (1.5) | 14.7 (1.4) | 15.0 (1.2) | 14.5 (1.2) | 14.4 (1.3) |
| Mean difference | 2.9 m | .8 m | .7 m | -.6 m | -.9 m | 0 m |
| Effect size | .528 | .386 | .418 | .422 | .505 | .106 |
| ATF/CFL | 16.3 (4.5) | 14.6 (2.0) | 13.9 (1.7) | 14.2 (1.6) | 13.8 (1.5) | 14.1 (1.9) |
| Mean difference | 5.6 m | .9 m | 1.5 m | .2 m | 0 m | .2 m |
| Effect size | 1.031 | .411 | .836 | .119 | .126 | .107 |
Effect sizes were calculated between the SHAM and CFL group and SHAM and ATFL/CFL group for wheel running distance (table 1), duration (Table 2) and speed (Table 3). The most clinical meaningful effect sizes were calculated between the SHAM and ATFL/CFL group. For distance effect sizes ranged from 0.354-0.906 across the lifespan. For duration, effect sizes ranged 0.504-0.726 across the lifespan, and for speed 0.107 – 1.031.
Discussion
This study represents the first use of inducing an ankle sprain in a mouse model and tracking physical activity levels across the lifespan. The results of this study indicate that a single severe ankle sprain significantly reduced physical activity levels across the lifespan compared to mice with a minor ankle sprain or the SHAM group. The greatest differences were between the ATFL/CFL groups (Tables 1 – 3). For distance ran on the running wheel, during the first half of the lifespan the mice without an ankle sprain (SHAM group) were running on average 2.23 km more per day compared to the ATF/CFL group. During the same time period the SHAM group was running on average 44.23 minutes more per day compared to the ATFL/CFL group. As the mice age and reach the last ¼ of the lifespan mean differences between the group decreases. But even at the end of life (last 3 months) the SHAM group was still running on average 24.3 minutes more per day compared to the ATFL/CFL group. This reduction in physical activity may have a significant impact on the development of chronic diseases and thus ways to improve physical activity across the lifespan after ankle sprains are needed.
Since this is the first study to examine physical activity after an ankle sprain across the lifespan, direct comparison to other studies is not possible. We are not aware of any other studies that have used other musculoskeletal injury models and measured physical activity. This lack of research on orthopedic injury and physical activity levels is quite concerning. Physical inactivity is one of three highest risk behaviors in the development of cardiovascular disease, cancer, and other chronic diseases such as diabetes and obesity, and is the second highest alterable cause of cardiovascular arterial disease (Centers for Disease Control, 2012). Understanding the impact of musculoskeletal injury on physical activity is crucial to monitoring recovery after injury. If patients recover appropriately after injury they should be able to return to near normal physical activity levels. The mice in this study (CBA/J) were used secondary to their relative high physical activity levels (Turner et al., 2005). If ligament healing occurred after injury then the mice should have been able to demonstrate normal wheel running activity.
During the first four weeks after surgery the severe sprain group (ATFL/CFL) as well as the minor sprain (CFL only) had significant decreases in physical activity compared to the SHAM group (Hubbard-Turner et al., 2012). The SHAM group demonstrated wheel running activity levels similar to those reported in previous physical activity and mice studies within a week after the surgery (Turner et al., 2005). The CFL group had no significant differences in physical activity levels with the SHAM group after three weeks, whereas the ATFL/CFL group had significantly less physical activity across the lifespan. Although we did not measure ligament healing or joint stability, a lack of healing and thus an unstable joint may have negatively impacted physical activity levels. We know from human research that patients after an ankle sprain as well as those with chronic ankle instability (CAI) have significantly more laxity or joint instability compared to healthy controls (Brown et al., 2015; Hubbard et al., 2007; 2012; Croy et al., 2012). These changes in joint stability could also lead to the impairments in neuromuscular control seen in patients with CAI (Feger et al., 2015; Levin et al., 2015; Wikstrom et al., 2010). The combination of the joint instability and neuromuscular control impairments may decrease the ability to be physically active. Mice were given access to running wheels three days after the ankle sprain was induced. Introducing the wheel three days after injury was too soon. Since these mice are known for high activity levels (Turner et al., 2005), giving them a wheel could lead to lack of rest and thus an inability for the joint to heal after the ankle sprain was induced. It is unlikely that pain played much of role in the decreased physical activity levels. Although we did not objectively assess pain in the mice, the mice were walking normally within 72 hours post-surgery. Mice were also given 12.5mg Carprofen (Rimadyl) tablets ad libitum for pain management throughout the first three days following surgery. However it should be noted the tablets were untouched within 48 hours after surgery. The lack of continued ingestion of the tablets and return to walking without a limp subjectively speak to a lack of pain experienced by the mice.
In humans, physical activity levels were measured in a group of subjects with CAI (Hubbard-Turner and Turner, 2012). Subjects with CAI were given a pedometer to wear for 7 consecutive days to monitor physical activity levels. The study reported subjects with CAI took significantly less steps per week then the control group. Although this physical activity data was only taken over a period of a week, it indicates the overall impact an ankle sprain and the development of CAI may have on physical activity levels (Hubbard-Turner and Turner, 2012). If these physical activity levels continue to decline similar to the mice in the current study it may have a huge impact on the overall health and well-being of the patient.
Although there is a lack of physical activity data in those recovering from an ankle sprain or in subjects with CAI, there have been numerous studies using self-reported functional limitations in these patient populations. Research has demonstrated in patients after an ankle sprain and those that develop CAI score significantly less compared to their uninvolved limb and healthy controls on subjective scales like the Foot and Ankle Instability Index (FADI), the Foot and Ankle Instability Index Sport (FADIS), the Foot and Ankle Ability Measure (FAAM) as well as its Sports subscale (FAAMS) (Hubbard et al., 2006; 2007; Hubbard-Turner, 2012; Simon et al. 2014; Wikstrom et al., 2013) With the numerous subjective symptoms patients report with ankle instability (pain, giving way, weakness, instability) it is not surprising patients would score significantly less on subjective scales or that those subjective feelings would carry over to decreased physical activity levels. Research has also reported that subjects with lower scores on subjective indexes also had greater performance deficits on a figure-of 8 hop and side hop (Docherty et al., 2005). The Docherty et al. (2005) study also reported that 76% of subjects with ankle instability reported feelings of the ankle “being unstable” during at least one of the functional tasks they performed in the study. With these subjective feelings of ankle instability, and decreased functional performance it is not surprising that subjects would likely refrain from physical activity or at least physical activity that requires ballistic movements like landing from a jump and/or cutting. While speculative, if this link is present, it demonstrates that the bigger health concern may be the need to find methods to improve anindividual’s function and helping them restore physical activity levels, otherwise long term chronic disease development may occur.
The primary benefit of using a mouse model to induce an ankle sprain and follow physical activity across the lifespan is the highly reliability and repeatability of physical activity assessment with voluntary wheel running has (Knab et al., 2009). Mice are also a more cost effective and feasible way to monitor changes that occur long term after an ankle sprain. Previous research has utilized an ankle sprain model in rats (Hahm, 2007; Kim et al., 2008; Koo et al., 2002; 2008) and based on our results mice could also be used to successfully induce and follow after an ankle sprain. Additionally, since the mice are genetically identical and live in the same environment, changes in physical activity levels can be attributed to the ankle sprain and no other secondary variables. It is not possible to determine how the distance or duration ran on the running wheel equates to steps taken by human subjects. But is a way to assess overall activity levels between three different groups of mice, and try to understand how an ankle sprain impacts physical activity in these mice. Effect sizes were on the moderate to high range across the lifespan between the ATFL/CFL group compared to the SHAM group. Effect sizes between the SHAM and CFL only group were on the low to moderate side across the lifespan for distance, duration and speed of physical activity. The magnitude of the difference between the ATFL/CFL and the SHAM group was much greater then between the SHAM and CFL only group (Table 1 – 3). The more severe the damage the greater impact on physical activity levels.
Further research needs to examine the mechanisms responsible for the decreased physical activity levels (lack of healing, neuromuscular impairment) as well as other markers of health and chronic disease development (cardiovascular, pulmonary function). Potential interventions could include delaying introduction of wheel so mice can have some controlled rest, we could also look at immobilizing the joint to allow healing, and potentially introduce balance exercises to help improve neuromuscular control.
Conclusion
A single severe ankle sprain significantly decreased physical activity levels across the lifespan in mice. This significant decrease in wheel running activity may lead to the development of numerous chronic diseases. Although the initial ankle sprain may be thought of as a relatively minor or insignificant injury, if not treated appropriately, or with lack of guidance with rehabilitation and exercise prescription, physical activity levels across the lifespan may decrease. Decreased physical activity is a significant public health concern given the cost associated with treating chronic disease development as well as the further loss of function the patient may experience.
Acknowledgements
This study was funded by the UNC Charlotte Faculty Grant Research Fund. There are no financial conflicts of interest.
Biographies
Tricia HUBBARD-TURNER
Employment
The University of North Carolina @ Charlotte
Degree
PhD
Research interests
Ankle instability, ankle osteoarthritis, physical activity after injury
E-mail: thubbar1@uncc.edu
Erik A. WIKSTROM
Employment
The University of North Carolina @ Charlotte
Degree
PhD
Research interests
Neuromuscular adaptations after an ankle sprain
Sophie GUDERIAN
Employment
University of Delaware
Degree
MS
Research interests
Exercise
Michael J. TURNER
Employment
The University of North Carolina @ Charlotte
Degree
PhD
Research interests
Cardiovascular changes in aging and physical activity
References
- Brown C.N., Rosen A.M., Ko J. (2015) Ankle ligament laxity and stiffness in chronic ankle instability. Foot and Ankle International 36, 565-572. [DOI] [PubMed] [Google Scholar]
- Centers for Disease Control and Prevention. (2012) Available from URL: http://www.cdc.gov/nccdphp/press/ [Google Scholar]
- Croy T., Saliba S.A., Saliba E., Anderson M.W., Hertel J. (2012) Differences in lateral ankle laxity measured via stress ultrasonography in individuals with chronic ankle instability, ankle sprain copers, and healthy individuals. Journal of Orthopedic and Sport Physical Therapy 42, 593-600. [DOI] [PubMed] [Google Scholar]
- Docherty C.L., Arnold B.L., Gansneder B.M., Hurwitz S., Gieck J. (2005) Functional-performance deficits in volunteers with functional ankle instability. Journal of Athletic Training 40, 30-34. [PMC free article] [PubMed] [Google Scholar]
- Feger M.A., Donovan L., Hart J.M., Hertel J. (2015) Lower extremity muscle activiation in patients with or without chronic ankle instability during walking. Journal of Athletic Training 50, 350-357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hahm T.S. (2007) The effect of 2 Hz and 100 Hz electrical stimulation of acupoint on ankle sprain in rats. Journal of Korean Medical Science 22, 347-351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hinterman B., Boss A., Schafer D. (2002) Arthroscopic findings in patients with chronic ankle instability. American Journal of Sports Medicine 30, 402-409. [DOI] [PubMed] [Google Scholar]
- Hootman J.M., Dick R., Agel J. (2007) Epidemiology of collegiate injuries for 15 sports: summary andrecommendations for injury prevention initiatives. Journal of Athletic Training 42(2), 311-319. [PMC free article] [PubMed] [Google Scholar]
- Hubbard T.J., Hertel J., Sherbondy P. (2006) Anterior fibular displacement with chronic ankle instability. Journal of Orthopaedic and Sport Physical Therapy 36, 3-9. [DOI] [PubMed] [Google Scholar]
- Hubbard T.J., Hicks-Little C.A., Cordova M.L. (2009) Mechanical and sensorimotor implications with ankle osteoarthritis. Archives Physical Medicine and Rehabilitation 90, 1136-1141. [DOI] [PubMed] [Google Scholar]
- Hubbard T.J., Kramer L.C., Hertel J., Denegar .C.R. (2007). Contributing Factors to Chronic Ankle Instability. Foot and Ankle International, 28,343-355 [DOI] [PubMed] [Google Scholar]
- Hubbard-Turner T. (2012) The relationship between Mechanical Ankle Joint Laxity and Subjective Function. Foot and Ankle International 33, 852-856. [DOI] [PubMed] [Google Scholar]
- Hubbard-Turner T., Turner M.J. (2012) Physical activity levels in those with chronic ankle instability. Journal of Orthopedic and Sport Physical Therapy 43, A23. [Google Scholar]
- Hubbard-Turner T., Wikstrom E.A., Turner M.J. (2012) Acute Ankle Sprain in a Mouse Model. Medicine and Science in Sport and Exercise 4, 1623-1628. [DOI] [PubMed] [Google Scholar]
- Kim H.Y., Wang J., Chung K., Chung J.M. (2008) A surgical ankle sprain pain model in the rat: Effects of morphine and indomethacin. Neuroscience Letters 444, 161-164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knab A.M., Bowen R.S., Moore-Harrison T., Hamilton A.T., Turner M.J., Lightfoot J.T. (2009) Repeatability of exercise behaviors in mice. Physiological Behavior 98(4), 433-440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Konradsen L., Olesen S., Hansen H. (1998) Ankle sensorimotor control and eversion strength after acute ankle inversion injuries. American Journal of Sports Medicine 26, 72-77. [DOI] [PubMed] [Google Scholar]
- Koo S.T., Lim K.S., Chung K., Ju K., Chung J.M. (2008) Electroacupuncture-induced analgesia in a rat model of ankle sprain pain is mediated by spinal α-adrenceptors. Pain 135, 11-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koo S.T., Park Y.I., Lim K.S., Chung K., Chung J.M. (2002) Acupuncture analgesia in a new rat model of ankle sprain pain. Pain 99, 423-431. [DOI] [PubMed] [Google Scholar]
- Levin O., Vanwanseele B., Thijsen J.R., Helsen W.F., Staes F.F., Duysens J. (2015) Proactive and reactive neuromuscular control in subjects with chronic ankle instability: evidence from a pilot study on landing. Gait and Posture 41, 106-111. [DOI] [PubMed] [Google Scholar]
- Lightfoot J.T., Turner M.J., Daves M, Vordermark A., Kleeberger S.R. (2004) Genetic influence on daily wheel running activity level. Physiological Genomics 19(3), 270-276. [DOI] [PubMed] [Google Scholar]
- McKay G.D. (2001) Ankle injuries in basketball: injury rate and risk factors. British Journal of Sports Medicine 35, 103-108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schaap G.R., de Keizer G., Marti K. (1989) Inversion trauma of the ankle. Archives Orthopedic Trauma Surgery 108(5), 272-275. [DOI] [PubMed] [Google Scholar]
- Simon J., Donahue M., Docherty C.L. (2014) Critical review of self-reported functional ankle instability measures: a follow-up. Physical Therapy in Sport 15, 97-100. [DOI] [PubMed] [Google Scholar]
- Turner M.J., Kleeberger S.R., Lightfoot J.T. (2005) Influence of genetic background on daily running-wheel activity differs with aging. Physiological Genomics 22, 76-78. [DOI] [PubMed] [Google Scholar]
- Verhagen R.A., de Keizer G., van Dijk C.N. (1995) Long-term follow-up of inversion trauma of the ankle. Archives Orthopedic Trauma Surgery 114, 92-96. [DOI] [PubMed] [Google Scholar]
- Wikstrom E.A., Fournier K.A., McKeon P.O. (2010) Postural control differs between those with and without chronic ankle instability. Gait Posture 32, 82–86. [DOI] [PubMed] [Google Scholar]
- Wikstrom E.A., Hubbard-Turner T., McKeon P.O. (2013) Understanding and treating lateral ankle sprains and their consequences: a constraints-based approach. Sports Medicine 46, 385-393. [DOI] [PubMed] [Google Scholar]
- Wikstrom E.A., Tillman M.D., Chmielewski T.L., Cauraugh J.H., Naugle K.E., Borsa P.A. (2009) Self-assessed disability and functional performance in individuals with and without ankle instability: a case control study. Journal of Orthopedicand Sports Physical Therapy 39, 458-467. [DOI] [PubMed] [Google Scholar]


