Abstract
The comparative study of neuroprotective effect of angiotensin converting enzyme inhibitors against scopolamine-induced neuroinflammation in albino Wistar rats was studied. Male albino rats were administered with scopolamine to induce memory impairment. The standard nootropic agent, piracetam (200 mg/kg b.w., [i.p.]), perindopril (0.1 mg/kg b.w., [i.p.]), enalapril (0.1 mg/kg b.w., [i.p.]), and ramipril (0.1 mg/kg b.w., [i.p.]) were administered in different group of animals for 5 days. On 5th day, scopolamine (1 mg/kg b.w., i.p.) was administered after 60 min of the last dose of test drug. Memory function was evaluated in Morris water maze (MWM) test and pole climbing test (PCT). Biochemical estimations like glutathione (GSH), malondialdehyde (MDA), and acetylcholinesterase activity in the brain were estimated after completion of behavior study. All three test groups shows improvement in learning and memory in comparison to control group. Perindopril treated group showed a more effective significant decrease in escape latency time and transfer latency time compared to enalapril and ramipril treated group on day 4 in MWM test and PCT, respectively. Perindopril shows a significant reduction in MDA level and acetylcholinesterase activity and a significant rise in GSH level compared to enalapril and ramipril. The finding of this study indicates that Perindopril is more effective in memory retention compared to enalapril and ramipril.
Keywords: Angiotensin converting enzyme inhibitors, comparative study, scopolamine-induced memory impairment
INTRODUCTION
Alzheimer's disease (AD) is a progressive neurodegenerative brain disorder that is slow in onset but leads to dementia, unusual behavior, personality changes and ultimately death.[1] Neuropathological examination of AD brain reveals extensive atrophy, accumulation of neurofibrilatory tangles and β-amyloid fibrillar deposits.[2] The National Institute of Health predicts, if the current trend continues, there will be more than 8.5 million AD patients by the year 2013 in USA alone.[3]
Formation of memory is the most complex process and involves multiple neuronal pathways and neurotransmitters.[4] The cholinergic neural system plays an important role in learning and memory in humans and animals.[5] Scopolamine, a nonselective muscarinic cholinergic antagonist, is a well-known centrally acting cholinergic probe, which causes impairment in learning and memory.[6] At present tacrine and donepezil are two reversible acetylcholinesterase (AChE) inhibitors approved by Food and Drug Administration for the treatment of mild to moderate dementia coupled with AD. Hepatotoxicity and high cost limit the use of tacrine. Donepezil has the advantage of long t1/2, about 70 h.[7]
Over the recent years, the brain renin-angiotensin system (RAS) has been shown to be involved in learning and memory consolidation. Continuous activation of the brain RAS impairs cognitive functions through stimulation of the AT1 receptor.[8] It has been found that the use of angiotensin-converting enzyme (ACE) inhibitors and angiotensin receptor blockers in AD patients is associated with slower rate of AD progression.[9]
Nootropics are agents that enhance the cognitive skills, and amnestics are the agent that disrupts the learning and memory process.[10] Nootropic agents such as piracetam, aniracetam, and cholinesterase inhibitors like donepezil are presently used for improving memory, mood, and behavior.[11]
MATERIALS AND METHODS
Drugs and Chemicals
Perindopril (Coversyl, Douglas Pharmaceuticals, Auckland, New Zealand), enalapril (Envas, Cadila Pharmaceuticals Ltd., Gujrat, India), ramipril (Ramistar, Lupin Pharmaceuticals Ltd., India) and piracetam (Sumocetam, Talent Healthcare, Haridwar, Uttarakhand, India) were dissolved in distilled water. Scopolamine (Sigma-Aldrich, MO, USA) was dissolved in normal saline.
Animals
The experiments were carried out with male Wistar rats weighing (150–200 g) obtained from the Laboratory Animal Services Division of Central Drug Research Institute, Lucknow, India. Research on experimental animals was conducted in accordance with the internationally accepted principles for laboratory animal use and care (1088/07/Committee for the Purpose of Control and Supervision of Experiments on Animals [CPCSEA]). They were kept in polyacrylic cages (22.5 cm × 37.5 cm) and were maintained under standard housing conditions (room temperature, 24–27°C, and humidity, 60–65%) with a 12-h light/12-h dark cycle. Food and water were available ad libitum. The experimental protocols were approved by the Institutional Animal Ethics Committee, which follow the guidelines of the CPCSEA and conform to the international norms of the Indian National Science Academy. Ethical norms were strictly followed during all experimental procedures.
Experimental design
From day 6th to 10th, memory function was evaluated by using Morris water maze (MWM) test and pole climbing test (PCT). All animals were sacrificed on the 11th day by cervical dislocation. Brains were isolated and biochemical estimation like glutathione (GSH), malondialdehyde (MDA), and acetylcholinesterase activity in the brain were carried out.
Behavior study
Morris water maze test
The MWM consists of a circular pool (1.8–2.0 m in diameter, 0.4–0.6 m in height), filled with water at 20°C–22°C to the depth of 0.3–0.4 m and made opaque white color. The pool was divided into four hypothetical quadrants. An escape platform was placed 1 cm below the water surface. Four different starting points for rats were placed around the perimeter of the pool. On each of the 5 training days, all four start points were used once in a pseudorandom sequence. The water maze was located on a large room with a number of extra maze visual cues. The trail began by placing the animal in the water facing the wall of the pool at one of the starting points. If the animals failed to escape on the platform within 120 s, it was gently placed there by the researcher and allowed to stay for 30 s. Each animal was subjected to a daily session of four trials for 5 consecutive days. Escape latency time (ELT) to locate the hidden platform in water maze was noted as an index of learning.[10]
Pole climbing test
Passive avoidance, based on negative reinforcement, was recorded to examine the long-term memory. The apparatus consisted of a box (27 cm × 27 cm × 27 cm) having three walls of wood and one wall of plexiglas, featuring a grid floor (made up of 3 mm stainless-steel rods set 8 mm apart), with a wooden pole (10 cm × 7 cm × 1.7 cm) in the center of the grid floor. Electric shock was delivered to the grid floor. When the rat was placed in the chamber and placed its paw on the grid floor, shock (foot shock: 50 Hz; 1.5 mA; 1 s) was delivered and transfer latency time (TLT) was recorded. TLT is defined as the time taken by the rat to step down and place all four paws on the grid floor.[4]
Locomotor activity
After a period of 15 min for acclimatization, animals were placed individually in the activity cage for 10 min, and their activity was monitored. The photocell counts were noted, and decrease or increase in locomotor activity was calculated.[3]
Biochemical estimation of markers of oxidative stress
After completion of the behavioral studies on day 11th, rats were sacrificed by cervical dislocation. Brain was removed from each rat, kept on an ice-cold plate.
Preparation of brain homogenate
The brains were washed and homogenized 10 times with ice-cold 0.1 M phosphate buffer (pH 7.4) and used to measure MDA, GSH, and AChE activity.[12]
Measurement of glutathione level
The GSH level was measured by its reaction with DTNB (Ellman reagent) to yield a yellow chromophore whose absorbance was measured spectrophotometrically. The brain homogenate was mixed with an equal amount of 10% trichloroacetic acid (TCA) and centrifuge at 200 × g for 10 min at 4°C. The supernatant was used for GSH estimation. To 0.1 ml of processed tissue sample, 2 ml of phosphate buffer (pH 8.4), 0.5 ml of DTNB, and 0.4 ml of double-distilled water were added, and the mixture was shaken vigorously on vortex. The absorbance was read at 412 nm within 15 min. The GSH level was expressed as nmol/mg protein.[13]
Measurement of malondialdehyde level
To a volume of 0.5 ml of tissue homogenate, 0.5 ml distilled water, and 1.0 ml 10% TCA were added, mixed well, and centrifuged at 3,000 g for 10 min. To 0.2 ml supernatant, 0.1 ml TBA was added. The total solution was placed in a water bath at 80°C for 40 min and then cooled at room temperature. The absorbance of the clear supernatant was measured at 532 nm in a spectrophotometer. The MDA level was expressed as nmol/mg protein.[13]
Measurement of acetylcholinesterase activity
A volume of 0.4 ml of aliquot of brain homogenate was incubated for 5 min with 2.7 ml of phosphate buffer and 0.1 ml of DTNB. Optical density (OD) was measured at 412 nm using ELISA microplate (Hemodiaz Lifesciences Pvt. Ltd., New Delhi, India). Then, 0.2 ml of freshly prepared acetylcholine iodide (pH 8.0) was added and change in OD was recorded for a period of 10 min at intervals of 2 min. Enzyme activity was calculated by using following formula.[6]
R = 5.74 × 10−4× ∆A/Co.
Where,
R = Rate in moles of substrate hydrolyzed/min/g tissue,
∆A = Change in absorbance/min (mean change in absorbance from the 0 to 10 min was taken).
Co = Original concentration of the tissue (mg/ml).
Statistical analysis
Results were expressed as means with their standard error of the mean. The statistical significance of difference between the different groups was determined by one-way ANOVA followed by Bonferroni's post-hoc test using GraphPad Prism 5 software (GraphPad Inc., California, USA). The significance level for Bonferroni's multiple comparison test was set to 0.05 for three or more groups, and P < 0.05 was considered as statistically significant.
RESULTS
Effect on locomotor activity
Perindopril, enalapril, and ramipril do not show any significant changes in the locomotor activity of animals when tested on actophotometer before and after treatment as compared to control group.
Effect of drugs on scopolamine-induced memory impairment in Morris water maze test
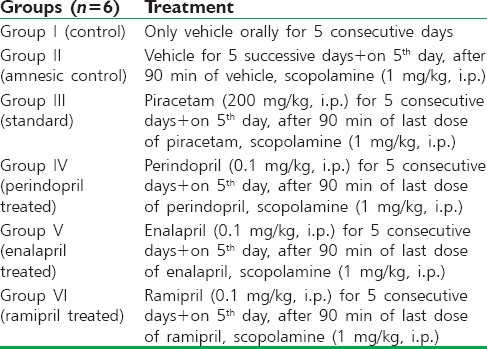
In control groups, a significant decrease in ELT on day 4 (P < 0.01) and day 5 (P < 0.001) was observed when compared to day 1 indicating spatial learning, however administration of scopolamine in amnesic control group at a dose of 1 mg/kg, i.p. caused memory impairment as there was no significant change (P < 0.01 and P < 0.001) in the ELT throughout all the days. Treatment with the standard nootropic drug piracetam (200 mg/kg, i.p.) prevented scopolamine-induced amnesia as indicated by a significant reduction in ELT on day 4 (P < 0.01) and day 5 (P < 0.001) when compared to day 1. Perindopril, enalapril, and ramipril treated group showed significant decrease in ELT on day 4 (P < 0.01) and day 5 (P < 0.001) compared to day 1 [Figure 1].
Figure 1.
Effect of drugs on scopolamine-induced memory impairment in rats using Morris water maze test. Values were expressed as mean escape latency time (s) ± standard error of the mean. *Significant difference (**P < 0.01 and ***P < 0.001) in comparison to day 1 of the respective groups
Effect of drugs on scopolamine-induced memory impairment in pole climbing test
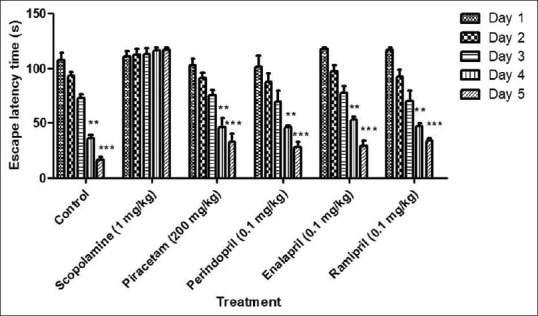
In control groups, there was a significant decrease (P < 0.01) in TLT on day 4 and 5 when compared to day 1. However, administration of scopolamine at a dose of 1 mg/kg, i.p. caused memory impairment as indicated by a no significant change (P < 0.01) in the TLT throughout all the days. The standard drug piracetam (200 mg/kg, i.p.) showed a significant reduction in the TLT on day 4 (P < 0.01) and day 5 (P < 0.01) when compared to day 1 indicating amelioration of scopolamine-induced memory impairment in rats. Perindopril, enalapril, and ramipril treated group showed a significant decrease in TLT on day 4 and day 5 (P < 0.01) compared to day 1 [Figure 2].
Figure 2.
Effect of drugs on scopolamine-induced memory impairment in rats using pole climbing test. Values were expressed as mean transfer latency time (s) ± standard error of the mean. *Significant difference **P < 0.01 in comparison to day 1 of the respective groups
Effect of drugs on glutathione level
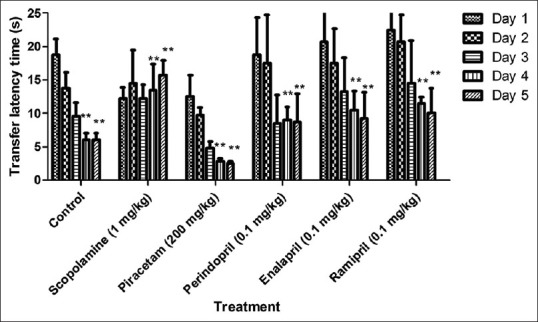
Glutathione was estimated in brain after the completion of behavioral studies. A significant fall (P < 0.001) in the level of GSH was observed in the amnesic control group as compared to the control group. There was a significant rise (P < 0.001), (P < 0.01) and (P < 0.01) in the level of GSH in brains of group treated with perindopril, enalapril and ramipril in comparison to scopolamine group. Standard group showed significant rise (P < 0.05) in GSH level in scopolamine-treated animals [Figure 3].
Figure 3.
Effect of drugs on glutathione (GSH) level. Values were expressed as mean GSH level (μg/mg protein) ± standard error of the mean. ***P < 0.001 versus control group, #P < 0.05, ##P < 0.01 and ###P < 0.001 versus amnesic control
Effect of drugs on malondialdehyde level
The MDA level was estimated in rat brain after the completion of behavioral studies. MDA level increased significantly (P < 0.001) in the brain of scopolamine-treated rats as compared to the control group. On the other hand, piracetam significantly decreased (P < 0.001) the MDA level in comparison to scopolamine group. Preventive treatment with 0.1 mg/kg b.w., i.p. of perindopril, enalapril and ramipril significantly decreased (P < 0.001) the MDA level in brains of scopolamine injected rats [Figure 4].
Figure 4.
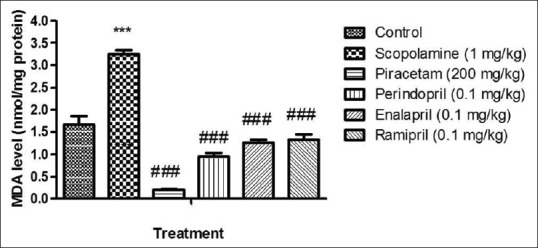
Effect of drugs on malondialdehyde (MDA) level. Values were expressed as mean MDA level (nmol/mg protein) ± standard error of the mean. ***P < 0.001 versus control group, ###P < 0.001 versus amnesic control
Effect of drugs on acetylcholinesterase activity
There was a significant increase (P < 0.001) in AChE activity in brain of scopolamine-treated group as compared to control. Piracetam significantly decreased (P < 0.001) AChE activity in brain of scopolamine injected rat. Preventive treatment of perindopril, enalapril and ramipril significantly decreased (P < 0.001) the AChE activity in brains of scopolamine-treated rats [Figure 5].
Figure 5.
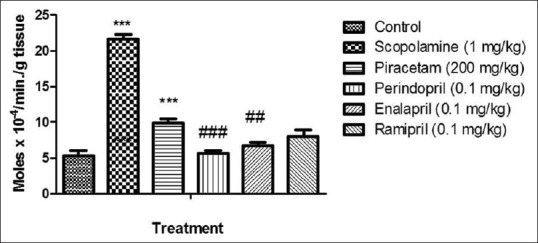
Effect of drugs on acetylcholinesterase (AChE) activity. Values were expressed as mean AChE activity (moles × 10−4/min/g tissue) ± standard error of the mean. ***P < 0.001 versus control group, ##P < 0.01 and ###P < 0.001 versus amnesic control
DISCUSSION
Alzheimer's disease is a neurodegenerative disorder associated with a decline in cognitive abilities. It is well-known that cholinergic neuronal system plays an important role in cognitive deficits associated with AD, ageing, and neurodegenerative disease.[4] Many anticholinergic drugs, such as, scopolamine, induce a transient disruption of memory in humans and experimental animals by blocking postsynaptic muscarinic receptors. This effect can be antagonized by cholinomimetics, such as physostigmine which increase brain ACh content.[14]
The present work was undertaken to study the effect of ACE inhibitors on memory function of animals for their establishment in AD. ACE plays a crucial role in memory deficits induced by i.p. Scopolamine because treatment with perindopril, enalapril, ramipril, which are an ACE inhibitors, prevented memory impairment, oxidative stress, and cholinergic dysfunction in rats.
The administration of the antimuscarinic agent Scopolamine produces transient memory deficits.[6] Scopolamine interferes with memory and cognitive function in human and experimental animals by blocking muscarinic receptors.[5] Scopolamine-induced impairment of the memory is due to cholinergic deficits in certain brain areas and appears to be unrelated to oxygen free radical.[15]
Clinical studies have shown an elevated ACE activity in various brain regions of AD patients.[1] Elevated brain Angiotensin II may interfere with Ach release that in turn interferes with cognitive function. It also interferes with potassium mediated release of Ach from rat entorhinal cortex slices.[16] ACE inhibitor prevents the conversion of Ang I to Ang II and prevents the vasoconstrictor action of Ang II.
From the behavior study, MWM test and PCT, it is clearly seen that there was a decrease in ELT and TLT in the scopolamine-treated group. Administration of perindopril, enalapril, and ramipril improved the memory loss effect of scopolamine.
Oxidative stress in brain generates oxygen radical like superoxide anion, hydroxyl radical, and hydrogen peroxide, which act on polyunsaturated fatty acids in brain, thereby propagating the lipid peroxidation. The major antioxidant and oxidative free radical scavenging enzyme like GSH, SOD and catalase plays an important role to reduce oxidative stress in brain.[6] In the present study rats after scopolamine treatment showed a significantly raise the brain levels of MDA, which is the indication of lipid peroxidation and free radical generation. At the same time, there was a significant decrease in levels of GSH, a tripeptides found in all cells, which reacts with free radicals and protect cells from superoxide radical, hydroxyl radical, and singlet oxygen. Pretreatment of perindopril, enalapril, and ramipril reduced the MDA level and increased GSH level in brain after scopolamine treatment.
The neurotransmitter acetylcholine is degraded by the enzyme AChE. Therefore, the use of AChE inhibitors is the most effective pharmacological approach for the symptomatic treatment of AD.[13] This activity tends to allow the more maintenance of acetylcholine in the brain, which is important for the cognitive function, learning, and memory. Scopolamine-induced amnesic animal shown a rise in AChE enzyme activity while ACE inhibitors inhibit the AChE enzyme activity.
Angiotensin converting enzyme inhibitors perindopril, enalapril, and ramipril has acetylcholinesterase enzyme inhibiting activity.
CONCLUSION
Angiotensin converting enzyme plays a crucial role in memory deficits induced by i.p. Scopolamine because treatment with perindopril, enalapril, ramipril, which are an ACE inhibitors, prevented memory impairment, oxidative stress and cholinergic dysfunction in rats. Perindopril was found to be more effective in memory retention compared to enalapril and ramipril.
ACKNOWLEDGMENTS
The authors are thankful to Hygia Institute of Pharmaceutical Education and Research, Lucknow, India for providing facilities to carry out this research. The Authors are also thankful to CDRI, Lucknow for providing animals.
Footnotes
Source of Support: Nil
Conflict of Interest: Nil.
REFERENCES
- 1.Kulkarni PD, Ghaisa MM, Chivate ND, Sankpal PS. Memory enhancing activity of C. pariera in mice. Int J Pharm Pharm Sci. 2011;3:206–11. [Google Scholar]
- 2.Gavimath CC, Havannavar V, Hulekal P, Pattar P, Joshi H. Anti-amnesic potentials of Calotropis procera in mice. Pharmacologyonline. 2009;3:457–62. [Google Scholar]
- 3.Joshi H, Parle M. Antiamnesic effects of Desmodium gangeticum in mice. Yakugaku Zasshi. 2006;126:795–804. doi: 10.1248/yakushi.126.795. [DOI] [PubMed] [Google Scholar]
- 4.Kulkarni KS, Kasture SB, Mengi SA. Efficacy study of Prunus amygdalus (almond) nuts in scopolamine-induced amnesia in rats. Indian J Pharmacol. 2010;42:168–73. doi: 10.4103/0253-7613.66841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kang SY, Lee KY, Park MJ, Kim YC, Markelonis GJ, Oh TH, et al. Decursin from Angelica gigas mitigates amnesia induced by scopolamine in mice. Neurobiol Learn Mem. 2003;79:11–8. doi: 10.1016/s1074-7427(02)00007-2. [DOI] [PubMed] [Google Scholar]
- 6.Kanwal A, Mehla J, Kuncha M, Naidu VG. Anti-amnesic activity of Vitex negundo in scopolamine induced amnesia in rats. Pharmacol Pharm. 2010;1:1–8. [Google Scholar]
- 7.Shintani EY, Uchida KM. Donepezil: An anticholinesterase inhibitor for Alzheimer's disease. Am J Health Syst Pharm. 1997;54:2805–10. doi: 10.1093/ajhp/54.24.2805. [DOI] [PubMed] [Google Scholar]
- 8.Deb D, Nayak V, Bairy KL, Rao M, Shetty J, Hedge MV, et al. Antiamnesic and neuroprotective effects of low dose of ramipril and losartan in scopolamine induced amnesia model of dementia. Res J Pharm Biol Chem Sci. 2013;4:1174–82. [Google Scholar]
- 9.Singh B, Sharma B, Jaggi AS, Singh N. Attenuating effect of lisinopril and telmisartan in intracerebroventricular streptozotocin induced experimental dementia of Alzheimer's disease type: Possible involvement of PPAR-γ agonistic property. J Renin Angiotensin Aldosterone Syst. 2013;14:124–36. doi: 10.1177/1470320312459977. [DOI] [PubMed] [Google Scholar]
- 10.Kshirsagar SN. Nootropic activity of dried seeds kernels of Caesalpinia crista Linn against scopolamine induced amnesia in mice. Int J PharmTech Res. 2011;3:104–9. [Google Scholar]
- 11.Joshi H, Parle M. Nootropic activity of calyces of Hibiscus sabdariffa Linn. Iran J Pharmacol Ther. 2006;5:15–20. [Google Scholar]
- 12.Tota S, Nath C, Najmi AK, Shukla R, Hanif K. Inhibition of central angiotensin converting enzyme ameliorates scopolamine induced memory impairment in mice: Role of cholinergic neurotransmission, cerebral blood flow and brain energy metabolism. Behav Brain Res. 2012;232:66–76. doi: 10.1016/j.bbr.2012.03.015. [DOI] [PubMed] [Google Scholar]
- 13.Jawaid T, Shakya AK, Siddiqui HH, Kamal M. Evaluation of Cucurbita maxima extract against scopolamine-induced amnesia in rats: Implication of tumour necrosis factor alpha. Z Naturforsch C. 2014;69:407–17. doi: 10.5560/znc.2014-0003. [DOI] [PubMed] [Google Scholar]
- 14.Hirokawa S, Nose M, Ishige A, Amagaya S, Oyama T, Ogihara Y. Effect of Hachimi-jio-gan on scopolamine-induced memory impairment and on acetylcholine content in rat brain. J Ethnopharmacol. 1996;50:77–84. doi: 10.1016/0378-8741(95)01332-6. [DOI] [PubMed] [Google Scholar]
- 15.Morgan SZ, Squire LR. Neuroanatomy of memory. Ann Rev Neurosci. 1993;16:547–63. doi: 10.1146/annurev.ne.16.030193.002555. [DOI] [PubMed] [Google Scholar]
- 16.Indumathy S, Kavimani S, Raman KV. Role of angiotensin antagonists in memory enhancement. Int J Pharm Biosci. 2010;1:1–6. [Google Scholar]


