Abstract
Patient: Male, 27
Final Diagnosis: Obstructive gastric pseudotumor
Symptoms: Dyspepsia • weight loss
Medication: Ganciclovir
Clinical Procedure: Infectious diseases treatments and surgical treatment
Specialty: Gastroenterology and Hepatology
Objective:
Rare co-existance of disease or pathology
Background:
Cytomegalovirus (CMV) is a common opportunistic pathogen in patients with HIV. It is also a major cause of gastrointestinal ulcers in patients with acquired immunodeficiency syndrome (AIDS). CMV pseudotumor in the stomach is a rare cause of digestive tract obstruction.
Case Report:
In this study we report a male patient infected with HIV in 2002. In 2014 he evolved C3 stage AIDS with prepyloric gastric ulcer which provoked deformity and pseudotumoral aspect of the gastric outlet. Endoscopic biopsy confirmed CMV infection. He underwent Roux-en-Y gastroenteroanastomosis with good recovery.
Conclusions:
CMV infection should be considered as an agent in gastric lesions in HIV-infected patients. Roux-en-Y gastroenteroanastomosis is a surgical option for this group of patients, allowing improvements in quality of life and decreasing risks of perioperative complications.
MeSH Keywords: AIDS-Related Opportunistic Infections, Cytomegalovirus Infections, HIV, Pyloric Stenosis, Stomach Ulcer
Background
Cytomegalovirus is a common opportunistic pathogen in patients infected with HIV, especially in those with low CD4+ lymphocyte count. It is a DNA virus and a member of the Herpesviridae family [1,2]. After primary infection, it remains hidden and can be reactivated [1]. Patients immunosuppressed by chemotherapy, immunosuppressive drugs, or acquired immunodeficiency syndrome (AIDS) may have severe disease affecting organs such as the retina, respiratory system, central nervous system, or gastrointestinal tract [1,3–5].
In the gastrointestinal tract, in particular, CMV virus most commonly affects the large bowel (47%), followed by the duodenum (21.7%), stomach (17.4%), esophagus (8.7%), and (rarely) small intestine (4.3%) [6]. In AIDS patients, CMV is a frequent cause of gastrointestinal ulcers [7,8]. CMV virus can cause vasculitis of the submucosal layer, resulting in thrombosis, ischemia, and mucosal ulceration. It can evolve to lumen narrowing and, eventually, gangrene and perforation of the affected organ [9].
The medical literature describes Kaposi’s sarcoma and non-Hodgkin’s lymphoma as causes of intestinal subocclusion in AIDS patients. However, there are few reports linking CMV virus as a cause of gastric or intestinal obstruction [10].
Roux-en-Y gastroenteroanastomosis can be a surgical option in dealing with gastric outlet obstruction. This technique offers shorter surgical time and fewer perioperative complications compared to surgical resection or other bypass procedures. Moreover, Roux-en-Y gastroenteroanastomosis is a surgical approach that can eliminate the alkaline gastritis and its long-term consequences that usually occur in classical gastroenteroanastomosis [11].
Case Report
Our Brazilian patient was a 27-year-old, white male when he sought our help in the first time in 2002. He had serological diagnosis of HIV infection in a screening test done due to a blood transfusion and was referred to the Infectious Disease Department of our Faculty. At this first visit, he was asymptomatic, and had never had opportunistic infections linked to HIV until that date. Also, he had a personal background of cigarette smoking and alcohol consumption. Physical examination did not show any specific sign or alteration. Furthermore, he cited a previous gastric ulcer treatment in another medical facility 1 year ago. This information was annotated as personal background and remained without further details afterwards. Laboratory exams were done (Tables 1, 2). Between 2002 and 2004, he had only 4 medical visits in an outpatient basis. In 1 of them, he complained of dyspepsia and therefore was treated with IBP (omeprazole). He was classified with A2 stage HIV infection at that time, and received no specific treatment for HIV.
Table 1.
Basic laboratory tests are shown.
| Basic laboratory tests | |||
|---|---|---|---|
| 2002 | January, 2014 | Preoperative | |
| Serum Na+ (mEq/L) | 141 | 132 | 137 |
| Serum K+ (mEq/L) | 4.6 | 3 | 3.9 |
| Serum urea (mg/dL) | 32 | 28 | 22 |
| Serum creatinine (mg/dL) | 1.06 | 0.74 | 0.58 |
| Fasting serum glucose (mg/dL) | 85 | 80 | 85 |
| Red blood cell count (×106/mm3) | 5.37 | 4.08 | 4.45 |
| Hemoglobin (g/dL) | 16.2 | 10.5 | 13.2 |
| Hematocrit (%) | 46.4 | 31.4 | 39.3 |
| White blood cell count (×103/mm3) | 4.54 | 5.8 | 5.61 |
| Bands count (×103/mm3) | 0 | 0.46 (8%) | 0 |
| Neutrophils count (×103/mm3) | 2.54 (56%) | 4.29 (74%) | 3.92 (69.9%) |
| Lymphocytes count (×103/mm3) | 1.34 (29.7%) | 0.58 (10%) | 1.14 (20.3%) |
| Monocytes count (×103/mm3) | 0.55 (12.3%) | 0.46 (8%) | 0.49 (8.7%) |
| Eosinophils count (×103/mm3) | 0.08 (1.8%) | 0 | 0.04 (0.7%) |
| Basophils count (×103/mm3) | 0 (0.2%) | 0 | 0.02 (0.4%) |
| Platelets count (×103/mm3) | 199 | 181 | 284 |
| Aspartate transaminase (U/L) | 24 | 103 | 20 |
| Alanine transaminase (U/L) | 25 | 66 | 17 |
| Alkaline phosphatase (U/L) | 162 | 66 | 124 |
| Gamma-glutamyl transpeptidase (U/L) | 19 | 69 | 52 |
| Serum total bilirubin dosage (mg/dL) | 0.5 | 0.31 | 0.16 |
| Prothrombin time (seconds) | – | 13.2 | 14 |
| Prothrombin activity (%) | – | 105.09 | 105.95 |
| INR | – | 0.98 | 0.97 |
| Partial thromboplastin time (seconds) | – | 31.7 | 29.4 |
| R | – | 1.13 | 1.05 |
| Total serum cholesterol (mg/dL) | 153 | – | 131 |
| Serum HDL-cholesterol (mg/dL) | 45 | – | 44 |
| Serum LDL-cholesterol (mg/dL) | 97 | – | 73 |
| Serum VLDL-cholesterol (mg/dL) | 11 | – | 14 |
| Serum triglycerides (mg/dL) | 55 | – | 69 |
Trace mark (–) means that test was not done.
Table 2.
HIV infection laboratory tests are shown.
| HIV infection laboratory tests | ||||
|---|---|---|---|---|
| 2002 | January, 2014 | Preoperative | 6-months after surgery | |
| Viral load count | 21,737 | 221,762 | 395 | <40 |
| CD4 count | 455 | 65 | 143 | 537 |
| CD8 count | 717 | 689 | 383 | 1,115 |
| CD4/CD8 ratio | 0.63 | 0.09 | 0.37 | 0.48 |
Unfortunately, this patient abandoned his clinical follow-up between 2005 and 2013. He came back to our medical facility on January 2014. Between 2005 and 2013, he did not receive any specific treatment for HIV infection. He was 39 years old in 2014. He complained of dyspepsia, fever, and recent weight loss. At physical examination, there were augmented cervical lymph nodes, fever, and abdominal pain. There was also a remarkable weight loss of 19 kilograms (kg) compared to our 2002 data available. Consequently, it was decided to admit him to the Infectious Diseases ward. New laboratory tests were done, including counts of CD4, CD8, and viral copies (Tables 1, 2). According to this new clinical picture, he was diagnosed with C3 stage AIDS. Thus, antiretroviral therapy was initiated with tenofovir, lamivudine, and efavirenz.
Those cervical lymph nodes seen on physical examination were examined with fine-needle aspiration biopsy and were positive for Mycobacterium tuberculosis (M. tuberculosis) infection. Indeed, CT scan imaging demonstrated augmented lymph nodes in the cervical and abdominal regions. In addition, there were images compatible to pulmonary tuberculosis on CT scan, although sputum tests were negative for M. tuberculosis. Therefore, tuberculosis treatment was started with rifampicin, isoniazid, pyrazinamide, and ethambutol.
At about the same time, an upper digestive endoscopy (UDE) was performed. UDE showed an ulcer 2 cm in diameter, with deep fibrin in its base, erythematous and swollen edges comprising the pyloric channel, causing difficulty in endoscopic progression into the duodenum (Figure 1). Biopsy demonstrated cytological abnormalities consistent with CMV infection and concomitant presence of Helicobacter pylori (H. pylori) infection (Figure 2). Abnormalities were described such as endothelial cells with foamy cytoplasm and nuclear eosinophilic inclusion compatible with cytopathic viral effect by cytomegalovirus in ulcerated mucosa area. Consequently, the patient was treated with ganciclovir 5 mg/kg twice a day intravenously for 21 days. At the same time, H. pylori infection was treated with amoxicillin, clarithromycin, and omeprazole (triple therapy orally for 7 days).
Figure 1.

Endoscopic photo showing pre-pyloric gastric ulcer causing stenosis of pyloric channel (biopsy confirming ulcer etiology by CMV).
Figure 2.
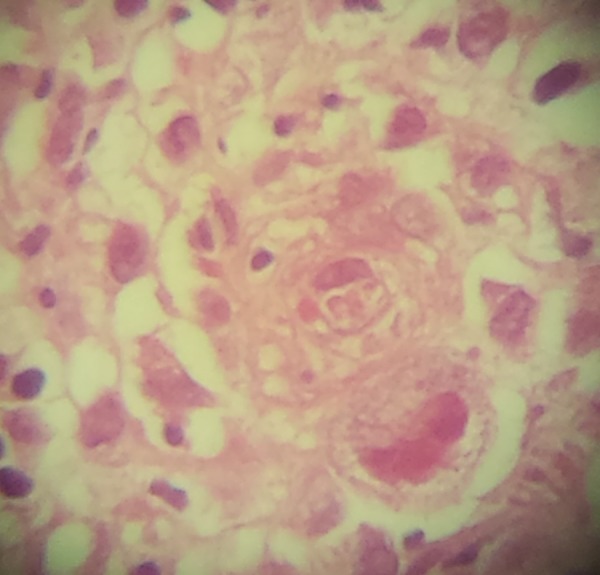
Photomicrography showing endothelial cells with foamy cytoplasm and nuclear eosinophilic inclusion compatible with cytopathic viral effect by cytomegalovirus in ulcerated mucosa area (hematoxylin-eosin, 400-fold increase).
After 22 days of in-hospital treatment, he was discharged without fever or abdominal pain, and continued clinical follow-up on an outpatient basis. He completed his treatment for CMV with ganciclovir when he was in hospital. Also, he received his treatment for H. pylori infection correctly. However, after 4 months of discharge, when he was still receiving treatment for M. tuberculosis infection, he presented to us with poor oral diet acceptance, vomiting, upper abdominal pain, and absence of weight gain. Therefore, UDE was repeated. It demonstrated gastric stasis and a pyloric ulcer that prevented progression into the duodenum. Nonetheless, this second biopsy did not show signs of CMV infection or H. pylori infection. In addition, the patient had some improvement in his HIV infection assessment (Table 2).
At that time, regarding the new clinical picture, the surgical team was asked to assess this patient. Considering his poor oral diet acceptance, persistent vomiting, absence of weight gain in spite of medical treatment, signs of pyloric obstruction on physical exam, and UDE, we decided to perform an elective laparotomy. One concern was uncertainty linked to oral medication absorption, including that of antiretroviral therapy. Another fact that helped our decision to surgery was the absence of CMV infection in the last biopsy. During abdominal examination, we observed a tumorous hard mass in the pyloric region, blocked by omentum, causing great deformity and distortion of the duodenum. An intraoperative biopsy was done and it was negative for malignancy. In consideration of this intense local blockage hindering the duodenal approach, we performed Roux-en-Y gastroenteroanastomosis in the posterior wall of the stomach with a 50-cm efferent loop.
The patient had an uneventful postoperative course. Oral diet was initiated on the third postoperative day. Finally, he was discharged on the seventh day after surgery, with good acceptance of oral diet and improvement of the gastrointestinal symptoms. Six months after surgery, he is asymptomatic, with good oral diet acceptance, no vomiting, and recovered weight (Tables 3, 4).
Table 3.
Vital signs are shown.
| Vital signs evolution | ||||
|---|---|---|---|---|
| 2002 | January, 2014 | Preoperative | 6-months after surgery | |
| Pulse (beats/min) | 66 | 80 | 90 | 88 |
| Arterial pressure (mmHg) | 110×70 | 90×60 | 110×80 | 110×70 |
| Temperature (°C) | 36 | 38.8 | 36.5 | 36 |
| Respiratory frequency (breaths/min) | 16 | 20 | 18 | 16 |
Table 4.
Nutritional evolution is shown.
| Nutritional evolution | ||||
|---|---|---|---|---|
| 2002 | January, 2014 | Preoperative | 6 months after surgery | |
| Weight (kg) | 66 | 47 | 50.6 | 60.8 |
| Body mass index (kg/m2) | 22.6 | 16 | 17.3 | 20.8 |
| Serum albumin dosage (g/dl) | 3.3 | 2.7 | 3.7 | – |
Trace mark (–) means that test was not done.
Discussion
Gastroduodenal ulcers (GDU) are rare in HIV patients who present upper digestive tract symptoms [12]. In a study with 497 patients with these characteristics, only 23 (5%) presented GDU in the endoscopy. Moreover, CMV was the only organism significantly associated with GDU in HIV patients and H. pylori was an uncommon cause of ulcers in this group [13].
Other studies showed similar results [14]. A study conducted in Taiwan comparing prevalence of H. pylori in patients with and without HIV demonstrated infection in 70% to 100% in those HIV-negative patients with peptic ulcer. On the other hand, this same study measured H. pylori infection only in 0% to 49% of HIV-positive patients. The authors of this study estimated that approximately 70% of AIDS patients with GDU had CMV infection concomitantly. They concluded that in AIDS patients with upper digestive tract symptoms and low CD4+ count, CMV infection is more common than H. pylori infection [8].
Despite high prevalence of CMV infection in this group of patients, it rarely causes GDU with pseudotumoral effect leading to pyloric duodenal sub-stenosis [15]. We found in the medical literature 1 report of just 5 cases of gastric pseudotumor by CMV, of which, only 3 cases were reported causing stenosis by the lesion [10]. Kelesidis et al. described a case of duodenal pseudotumor solved by oral treatment with valganciclovir and reported another 14 cases of CMV pseudotumor in other parts of the gastrointestinal tract in their patients [20]. They cautiously concluded that this successful treatment with complete resolution of CMV’s pseudotumor in their HIV immunosuppressed patient with medication was a unique case in the literature. Finally, they highlighted the importance of remembering this clinical entity in the differential diagnosis in HIV patients [16].
Surgical treatment for duodenal stenosis, which can be caused by malignancy, peptic ulcer, or other specific agents, may involve procedures with surgical resection like partial gastrectomy or bypass techniques like pyloroplasty. Bypass surgery can be done by several approaches as described by Heineke-Mikulicz, Finney, Jaboulay, Finsterer-Bancroft, as well as gastroenteroanastomosis [17–20].
In difficult duodenal cases with pseudotumor and local blockage in the gastric outlet as we describe in this paper, we should consider a bypass like Roux-en-Y gastroenteroanastomosis. This surgical option avoids direct manipulation of the affected region, possibly averting some surgical complications like postoperative duodenal leaking. In addition, this bypass technique can offer improved acceptance of oral diet and, consequently, better nutritional status and quality of life.
Conventional gastroenteroanastomosis is performed with a side-to-side anastomosis between the stomach posterior wall and the jejunal loop [21]. However, this technique is linked with bile reflux directly into the stomach, causing dyspeptic symptoms and alkaline reflux gastritis [22]. Furthermore, chronic alkaline reflux gastritis can increase neoplasia risk in the gastric mucosa in the long term [23].
We perform a modified gastroenteroanastomosis since 1998. It consists of a precolic side-to-side gastrojejunostomy and a Roux-en-Y end-to-side jejuno-jejunal anastomosis, with an efferent loop measuring about 50 cm. This procedure is usually performed in patients with unresectable malignancy [11].
Accordingly, HIV-positive patients presenting GDU must be surveyed with endoscopic biopsies to assess presence of opportunistic infections, notably CMV infection. In addition, H. pylori infection is an uncommon cause of ulcer disease in these patients [7,8]. Thus, patients who are not responsive to traditional ulcer disease treatment should be promptly reviewed by endoscopy with several biopsies to assess the presence of specific agents like CMV [8].
In conclusion, Roux-en-Y gastroenteroanastomosis can be a good option in preventing bilious vomiting compared to classic gastroenteroanastomosis. Additionally, dyspeptic symptoms linked to alkaline gastritis and malignancy risk decrease in the long term. It is a safe technique that promotes weight gain and improves patient quality of life.
Conclusions
In summary, we described a gastric pseudotumor case causing pyloric stenosis in an advanced stage AIDS patient. Other opportunistic diseases affected him as well. Biopsy demonstrated that CMV infection was linked to gastric pseudotumor, a very rare manifestation according to medical literature. Besides, this gastric outlet obstruction impaired his treatment with oral medications. Lastly, we advocate Roux-en-Y gastroenteroanastomosis as a good alternative to surgical treatment. This approach is linked to lower risk of operative complications compared to classic gastroenteroanastomosis and provides good quality of life.
Acknowledgments
We acknowledge the Pathology Department, Diagnostic Center for Digestive Diseases (Gastrocentro), State University of Campinas (Unicamp), especially Dr. Rita Barbosa de Carvalho, M.D., who kindly provided us the photomicrograph that illustrates this paper.
Footnotes
Disclosure
The authors declare no conflict of interest.
References:
- 1.Galiatsatos P, Shrier I, Lamoureux E, Szilagyi A. Meta-analysis of outcome of cytomegalovirus colitis in immunocompetent hosts. Dig Dis Sci. 2005;50(4):609–16. doi: 10.1007/s10620-005-2544-6. [DOI] [PubMed] [Google Scholar]
- 2.Bang S, Park YB, Kang BS, et al. CMV enteritis causing ileal perforation in underlying lupus enteritis. Clin Rheumatol. 2004;23(1):69–72. doi: 10.1007/s10067-003-0825-z. [DOI] [PubMed] [Google Scholar]
- 3.Michalopoulos N, Triantafillopoulou K, Beretouli E, et al. Small bowel perforation due to CMV enteritis infection in an HIV-positive patient. BMC Res Notes. 2013;6:45. doi: 10.1186/1756-0500-6-45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Tamura J, Arakaki S, Shibata D, Maeshiro T. Cytomegalovirus-associated gastric ulcer: a diagnostic challenge in a patient of fulminant hepatitis with steroid pulse therapy. BMJ Case Rep. 2013;2013:bcr2013010501. doi: 10.1136/bcr-2013-010501. pii: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Mori H, Fujihara S, Nishiyama N, et al. Cytomegalovirus-associated gastric ulcer: a side effect of steroid injections for pyloric stenosis. World J Gastroenterol. 2013;19(7):1143–46. doi: 10.3748/wjg.v19.i7.1143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kawate S, Ohwada S, Sano T, et al. Ileal perforation caused by cytomegalo-virus infection in a patient with recurrent gastric cancer: report of a case. Surg Today. 2002;32(12):1088–90. doi: 10.1007/s005950200220. [DOI] [PubMed] [Google Scholar]
- 7.Varsky CG, Correa MC, Sarmiento N, et al. Prevalence and etiology of gastroduodenal ulcer in HIV-positive patients: a comparative study of 497 symptomatic subjects evaluated by endoscopy. Am J Gastroenterol. 1998;93(6):935–40. doi: 10.1111/j.1572-0241.1998.00282.x. [DOI] [PubMed] [Google Scholar]
- 8.Chiu HM, Wu MS, Hung CC, et al. Low prevalence of Helicobacter pylori but high prevalence of cytomegalovirus-associated peptic ulcer disease in AIDS patients: Comparative study of symptomatic subjects evaluated by endoscopy and CD4 counts. J Gastroenterol Hepatol. 2004;19(4):423–28. doi: 10.1111/j.1440-1746.2003.03278.x. [DOI] [PubMed] [Google Scholar]
- 9.Spencer SP, Power N. The acute abdomen in the immune compromised host. Cancer Imaging. 2008;8:93–101. doi: 10.1102/1470-7330.2008.0013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Mohan H, Bal A, Garg S, Dalal U. Cytomegalovirus-associated pseudotumor simulating gastric malignancy in acquired immunodeficiency syndrome: a case report with review of literature. Jpn J Infect Dis. 2007;60(2–3):134–36. [PubMed] [Google Scholar]
- 11.Andreollo NA, Lopes LR, Coelho Neto JdS. Silveira HJV: Roux-en-Y gastroenteroanastomosis in the treatment of stenosing and advanced gastric adenocarcinoma. Arq Bras Cir Dig. 2010;23:90–93. [Google Scholar]
- 12.Murray RN, Parker A, Kadakia SC, et al. Cytomegalovirus in upper gastrointestinal ulcers. J Clin Gastroenterol. 1994;19(3):198–201. doi: 10.1097/00004836-199410000-00005. [DOI] [PubMed] [Google Scholar]
- 13.Vachon GC, Brown BS, Kim C, Chessin LN. CMV gastric ulcer as the presenting manifestation of AIDS. Am J Gastroenterol. 1995;90(2):319–21. [PubMed] [Google Scholar]
- 14.Lv FJ, Luo XL, Meng X, et al. A low prevalence of H. pylori and endoscopic findings in HIV-positive Chinese patients with gastrointestinal symptoms. World J Gastroenterol. 2007;13(41):5492–96. doi: 10.3748/wjg.v13.i41.5492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Cullen MH, Stansfeld AG. Cytomegalovirus-associated gastric ulcer simulating malignancy. Postgrad Med J. 1979;55(641):210–11. doi: 10.1136/pgmj.55.641.210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kelesidis T, Tozzi S, Mitty R, et al. Cytomegalovirus pseudotumor of the duodenum in a patient with AIDS: an unrecognized and potentially treatable clinical entity. Int J Infect Dis. 2010;14(4):e274–82. doi: 10.1016/j.ijid.2009.04.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hallenbeck GA. What is the best elective operation for duodenal ulcer? Can Med Assoc J. 1970;103(12):1255–62. [PMC free article] [PubMed] [Google Scholar]
- 18.Soreide K, Sarr MG, Soreide JA. Pyloroplasty for benign gastric outlet obstruction – indications and techniques. Scand J Surg. 2006;95(1):11–16. doi: 10.1177/145749690609500103. [DOI] [PubMed] [Google Scholar]
- 19.Kauffman GL., Jr Duodenal ulcer disease: treatment by surgery, antibiotics, or both. Adv Surg. 2000;34:121–35. [PubMed] [Google Scholar]
- 20.Feldman SD, Wise L, Ballinger WF. Review of elective surgical treatment of chronic duodenal ulcer. World J Surg. 1977;1(1):9–15. doi: 10.1007/BF01654719. [DOI] [PubMed] [Google Scholar]
- 21.Cohen MM. Choice of gastric drainage procedures. Can J Surg. 1975;18(5):414–19. [PubMed] [Google Scholar]
- 22.Burden WR, Hodges RP, Hsu M, O’Leary JP. Alkaline reflux gastritis. Surg Clin North Am. 1991;71(1):33–44. doi: 10.1016/s0039-6109(16)45331-4. [DOI] [PubMed] [Google Scholar]
- 23.Sitarz R, Maciejewski R, Polkowski WP, Offerhaus GJ. Gastroenterostoma after Billroth antrectomy as a premalignant condition. World J Gastroenterol. 2012;18(25):3201–6. doi: 10.3748/wjg.v18.i25.3201. [DOI] [PMC free article] [PubMed] [Google Scholar]


