Abstract
Adverse drug reactions play a substantial role in the etiology of gynecomastia. Gynecomastia as an adverse drug reaction, related to some cardiovascular drugs, has been reported in literature. Nebivolol is a third generation beta-blocker, and gynecomastia as an adverse effect on the consumption of this drug has not been reported in any article yet. We herein present the case of a 42-year-old male, who developed bilateral gynecomastia following nebivolol use and complete regression after discontinuation of nebivolol. Other reasons causing gynecomastia were excluded. Discontinuation of the responsible drug is quite sufficient with regard to the treatment of drug-induced gynecomastia, without any pharmacological or surgical treatment.
Keywords: Antihypertensive therapy, drug-induced gynecomastia, nebivolol
INTRODUCTION
Gynecomastia is the development of a fibroepithelial structure in the male breast due to several factors, with resemblance to female breast.[1] Gynecomastia may occur owing to physiological changes, systemic diseases, tumors, and medications. Angiotensin-converting enzyme inhibitors, spironolactone, digoxin, amiodarone, verapamil, diltiazem, and nifedipine are the medications commonly used in Cardiology practice and they are all reported to be the cause of gynecomastia.[2] Nebivolol is a third generation beta-1 selective blocker used for arterial hypertension and systolic heart failure. Nebivolol-induced gynecomastia has not been mentioned in the prescribing information nor has it ever been reported in literature as an adverse effect.
CASE REPORT
A 42-year-old male patient, who had never smoked or taken alcohol was prescribed 5 mg of nebivolol for essential hypertension. His blood pressure levels were all in the normal range during his follow-up. However, the patient presented to the Outpatient Clinic with a complaint of bilateral breast swelling and pain after two months of the medication [Figures 1 and 2]. Bilateral nodular gynecomastia and swollen glandular tissue in the retroareolar region were detected by mammography and breast ultrasonography [Figure 3]. The patient was referred to the Endocrinology and General Surgery Departments and diagnosed with grade II gynecomastia. The patient was interrogated for any drug or herbal preparate use, except nebivolol. Testicular and abdominal ultrasonography and computed tomography of adrenal glands were unremarkable. The hepatic, renal, and thyroid function tests and sex hormones levels were all in the normal range. Luteinizing hormone (6.3 mIU/mL), follicle stimulating hormone (5.8 mIU/mL), testosterone (6.3 ng/dL), thyroid stimulating hormone (2.4 μIU/mL), prolactin (4.1 ng/mL), and progesterone (0.43 ng/mL) were all in the normal range. Gynecomastia was thought to be induced by nebivolol and hence it was discontinued. Amlodipine (10 mg) once a day was administered. After three months of amlodipine, the gynecomastia had completely regressed and there was no swelling or pain anymore. A casualty assessment of definite was made according to the Naronjo's adverse drug reaction (ADR) probability scale, as in Table 1.[3]
Figure 1.

Bilateral gynecomastia
Figure 2.
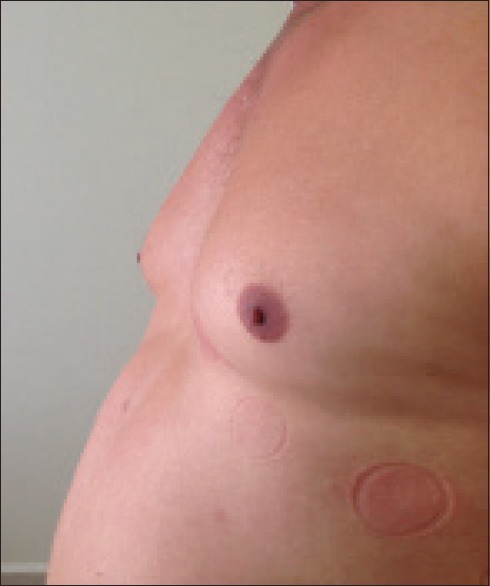
Bilateral gynecomastia
Figure 3.

Bilateral glandular tissue growth in the retroareolar region of the breast demonstrated in mammography
Table 1.
Naranjo's ADR probability scale and its score in the present case

DISCUSSION
Gynecomastia is characterized by enlargement of the male breast, caused by glandular proliferation and fat deposition. The key point of gynecomastia development is the imbalance between the estrogen and androgen. Physiological gynecomastia may be seen in the neonatal period or during puberty or senility.[4] Our patient is a 42-year-old man, who does not belong to any of these periods. Cirrhosis, chronic renal failure necessitating hemodialysis, hyperthyroidism, and testicular or adrenal tumors are among the causes of gynecomastia.[2,4] None of these illnesses were confirmed in our patient. Medication-induced gynecomastia comprises of 10–20% of all gynecomastias.[2] Medication-induced gynecomastia generally regresses after medication discontinuation, as has happened in our case. Finasteride, isoniazide, ethionamide, ketoconazole, metronidazole, captopril, enalapril, reserpine, diazepam, tricyclic antidepressants, spironolactone, digoxin, amiodarone, verapamil, diltiazem, and nifedipine have all been reported to induce gynecomastia.[5,6] Spironolactone antagonizes the androgen receptors and leads to gynecomastia.[7] On the other hand, the mechanisms of gynecomastia caused by calcium channel blockers and amiodarone are not well-described.[6] We do not have an idea of how nebivolol causes gynecomastia.
Nebivolol is a frequently used beta blocker that is used to treat hypertension and heart failure. These patients may also use spironolactone, digoxin, nifedipine, verapamil, diltiazem, captopril or enalapril for the same, all of which may also cause gynecomastia as a rare side effect. Adverse drug reaction must be initially considered as the cause of gynecomastia, if any of these drugs have been taken. However, the above-mentioned etiologies leading to gynecomastia must also be sought. It is not easy to specify which drug causes gynecomatia, when many of the above-stated drugs are used, although digoxin and spironolactone are the potential supposed drugs that cause gynecomastia. Any disease that may result in gynecomastia has not been ascertained and our patient was not using any medication except nebivolol, therefore, we suspected that nebivolol was the agent responsible for the gynecomastia. Drug-induced gynecomastias regress after discontinuation of the drug responsible for them and any medical or surgical treatment is not principally necessary.
CONCLUSION
Nebivolol, a third generation beta-1 selective blocker rarely induces gynecomastia and discontinuation of the drug can improve the condition.
Footnotes
Source of Support: Nil
Conflict of Interest: None declared.
REFERENCES
- 1.Donegan WL. Common benign contitions of the breast. Gynecomastia. In: Donegan WL, Spratt JS Jr, editors. Cancer of the Breast. London: W.B. Saunders; 1995. pp. 100–3. [Google Scholar]
- 2.Braunstein GD. Gynecomastia. N Engl J Med. 1993;328:490–5. doi: 10.1056/NEJM199302183280708. [DOI] [PubMed] [Google Scholar]
- 3.Naranjo CA, Busto U, Sellers EM, Sandor P, Ruiz I, Roberts EA, et al. A method of estimating the probability of adverse drug reactions. Clin Pharmacol Ther. 1981;30:239–45. doi: 10.1038/clpt.1981.154. [DOI] [PubMed] [Google Scholar]
- 4.Carlson HE. Gynecomastia. N Engl J Med. 1980;303:795–9. doi: 10.1056/NEJM198010023031405. [DOI] [PubMed] [Google Scholar]
- 5.Thomson DF, Carter JR. Drug-induced gynecomastia. Pharmacotherapy. 1993;13:37–45. [PubMed] [Google Scholar]
- 6.Eckman A, Dobs A. Drug-induced gynecomastia. Expert Opin Drug Saf. 2008;7:691–702. doi: 10.1517/14740330802442382. [DOI] [PubMed] [Google Scholar]
- 7.Goldman RD. Drug-induced gynecomastia in children and adolescents. Can Fam Physician. 2010;56:344–5. [PMC free article] [PubMed] [Google Scholar]